Abstract
Background. Transcranial direct current stimulation (tDCS) has been suggested to improve poststroke recovery. However, its effects on bimanual motor learning after stroke have not previously been explored. Objective. We investigated whether dual-tDCS of the primary motor cortex (M1), with cathodal and anodal tDCS applied over undamaged and damaged hemispheres, respectively, improves learning and retention of a new bimanual cooperative motor skill in stroke patients. Method. Twenty-one chronic hemiparetic patients were recruited for a randomized, double-blinded, cross-over, sham-controlled trial. While receiving real or sham dual-tDCS, they trained on a bimanual cooperative task called CIRCUIT. Changes in performance were quantified via bimanual speed/accuracy trade-off (Bi-SAT) and bimanual coordination factor (Bi-Co) before, during, and 0, 30, and 60 minutes after dual-tDCS, as well as one week later to measure retention. A generalization test then followed, where patients were asked to complete a new CIRCUIT layout. Results. The patients were able to learn and retain the bimanual cooperative skill. However, a general linear mixed model did not detect a significant difference in retention between the real and sham dual-tDCS conditions for either Bi-SAT or Bi-Co. Similarly, no difference in generalization was detected for Bi-SAT or Bi-Co. Conclusion. The chronic hemiparetic stroke patients learned and retained the complex bimanual cooperative task and generalized the newly acquired skills to other tasks, indicating that bimanual CIRCUIT training is promising as a neurorehabilitation approach. However, bimanual motor skill learning was not enhanced by dual-tDCS in these patients.
Keywords
Introduction
A majority of survivors of unilateral stroke are left with lifelong impairments, including hemiparesis affecting motor control over the contralesional side. In particular, impairments in the motor and sensory functions of the contralesional upper limb (UL) severely impinge on crucial daily activities 1 requiring bimanual coordination such as driving or buttoning a shirt, which stroke sufferers learn to compensate for through increased use of their nonparetic hand. 2 Restoring bimanual coordination after stroke is essential for recovering in daily activities. While most stroke survivors retain the ability to perform certain bilateral movements, 3 including continuous bimanual tasks, 4 the nonparetic hand frequently slows down in order to maintain temporal and spatial coupling with the affected hand.5,6
Most bimanual studies have focused on symmetrical hand movements and neurorehabilitation with bilateral training involving symmetrical coupling can aid in the functional recovery of the paretic hand, 7 and this approach now serves as the basis for many rehabilitation therapies.8,9 However, many activities in daily life require both hands to cooperate asymmetrically to accomplish a common goal, 4 and it is these that are particularly impaired following a stroke. 10 It is for this reason that some have suggested that the emphasis should also be placed on asymmetrical bimanual coordination in poststroke UL neurorehabilitation. 11
In healthy individuals, high-level bimanual proficiency is acquired through motor skill learning, which involves the development of new (bimanual) skills through reinforcement processes involving trials and error, and the ability to retain and retrieve these skills when needed. Once a skill has been learned and mastered, it can then be performed through a smooth sequence of movements regardless of the tempo, with some degree of automation, and a shift in the speed/accuracy trade-off (SAT) function. 12 The individual will also possess the capacity to generalize the skill to unfamiliar contexts or to perform it with different effectors. For bimanual cooperative skills, subjects have to learn how to coordinate both ULs, establishing bimanual control policies dedicated to each specific task/skill. Bimanual cooperative skills rely on both the structural and functional integrity of the central nervous system. 13 However, despite its importance, the question of how stroke survivors learn new bimanual cooperative skills has received relatively little attention. 11
The degree of deficit in bimanual coordination in stroke survivors depends on the nature of the task, location and extent of the lesion, and severity of sensorimotor impairments.14,15 A unilateral stroke may disturb the balance of interhemispheric interactions, resulting in an abnormal inhibition of the ipsilesional hemisphere by the contralesional side. 16 Unlike the interhemispheric imbalance model, the vicariation model suggests a compensatory role for the contralesional hemisphere. 17 Finally the bimodal balance-recovery model 18 suggests that in less impaired stroke survivors, the interhemispheric imbalance model predicts well the increased recruitment of the ipsilesional hemisphere whereas, in more impaired stroke survivors, vicariation leads to increased recruitment of the contralesional hemisphere.
Noninvasive brain stimulation (NIBS) has been used with the objective of modulating brain excitability/activity19,20 in order to restore balanced interactions. 21 One promising NIBS method is transcranial direct current stimulation (tDCS). Although its effect size is modest, 22 tDCS is effective in enhancing motor function, motor learning,23,24 and generalization 25 in healthy individuals. Combined with neurorehabilitation in stroke survivors, tDCS significantly improves motor recovery compared with sham tDCS.26,27 Crucially, tDCS enhances motor skill learning involving a paretic UL28-30 and the long-term retention of that skill.30,31
Several studies have shown that tDCS enhances unimanual motor learning and recovery after stroke.29,30 A recent review article has suggested the potential of tDCS in improving bimanual motor performance and/or motor learning in healthy individuals. 32 In stroke survivors, a pilot study of 5 patients with stroke or traumatic brain injury has suggested that the combination of dual-tDCS and robotics improves bimanual motor control. 33 Another study has demonstrated that anodal tDCS over the ipsilesional hemisphere prevented the temporary decline in bimanual performance caused by contralesional inhibitory repetitive transcranial magnetic stimulation (rTMS). 34 However, the effect of tDCS on bimanual motor skill learning in stroke has not been systematically explored. Dual-tDCS improved unilateral motor function and learning with the paretic UL30,35 and resulted in a trend toward normalization of the functional magnetic resonance imaging (fMRI) patterns,36,37 in coherence with the interhemispheric imbalance model. In the present study, as a first step to explore bimanual learning, based on the interhemispheric imbalance model, we chose to investigate whether dual-tDCS enhanced bimanual cooperative skill learning, retention and generalization in chronic hemiparetic stroke survivors.
Methods
Population
A total of 21 chronic stroke survivors were included after providing written informed consent (Supplementary Figure 1, CONSORT flowchart, available online). To be included, the patients had to (1) be a chronic stroke (>6 months) sufferer, (2) be aged between 18 and 80 years, (3) present with a motor deficit in the UL, and (4) demonstrate a lesion on brain imaging. Patients with (1) craniotomy/a skull defects, (2) epilepsy, (3) intracranial metallic devices, (4) difficulty in understanding or executing commands, (5) drug/alcohol addiction, or (6) who were pregnant, were excluded. Of the 21 patients, 19 had an ischemic stroke and 2 had a hemorrhagic stroke (Table 1, Supplementary Figure 2). Residual hand dexterity was measured with the Box & Blocks test (B&B) and the residual bimanual ability with the ABILHAND questionnaire. 38 The level of neurological impairment was quantified using the National Institutes of Health Stroke Scale (NIHSS), and that of activity limitation with the modified Rankin Scale (mRS). The study was performed in accordance with the Declaration of Helsinki and all procedures described were approved by the Comité d’éthique médicale, CHU UCL Namur (Mont-Godinne), approval number: B039201317382.
Chronic stroke patient’s characteristics.
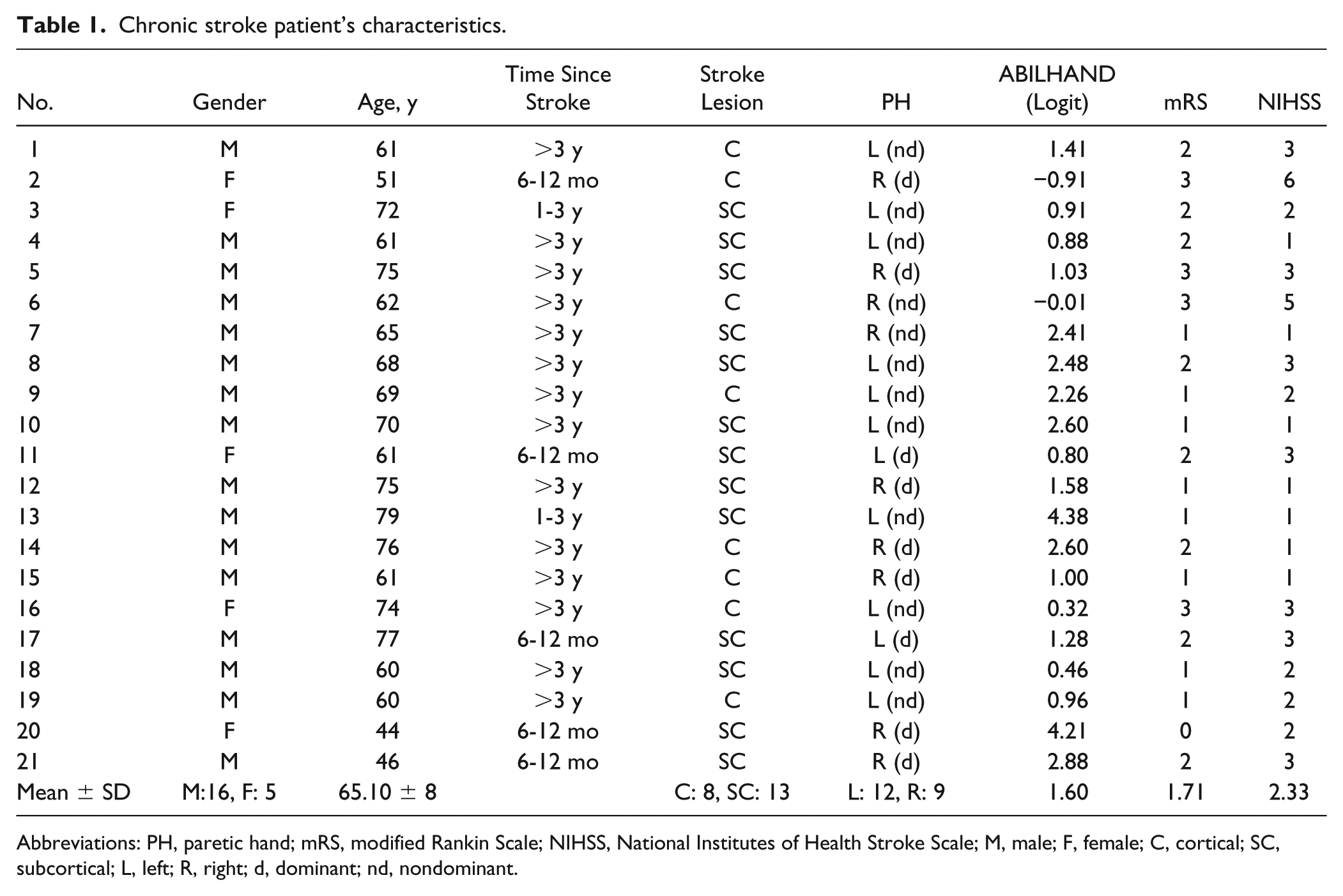
Abbreviations: PH, paretic hand; mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale; M, male; F, female; C, cortical; SC, subcortical; L, left; R, right; d, dominant; nd, nondominant.
Study Protocol
This study had a randomized and balanced, double-blinded, placebo-controlled, and cross-over design (RCT) and consisted of 2 blocks: (1) real and (2) sham dual-tDCS separated by at least 1 week (Supplementary Figure 1). The order of stimulation (real/sham or sham/real) and the orientation controlled by the patients’ paretic UL (lateral or anteroposterior) were randomized. The 2 blocks were identical except for the type of stimulation during the training session that was randomized (real/sham or sham/real). Each block consisted of an intervention session and a retention session 1 week later (Figure 1). Therefore, between the training sessions (ie, between the real and sham stimulation), there was a 2-week interval.
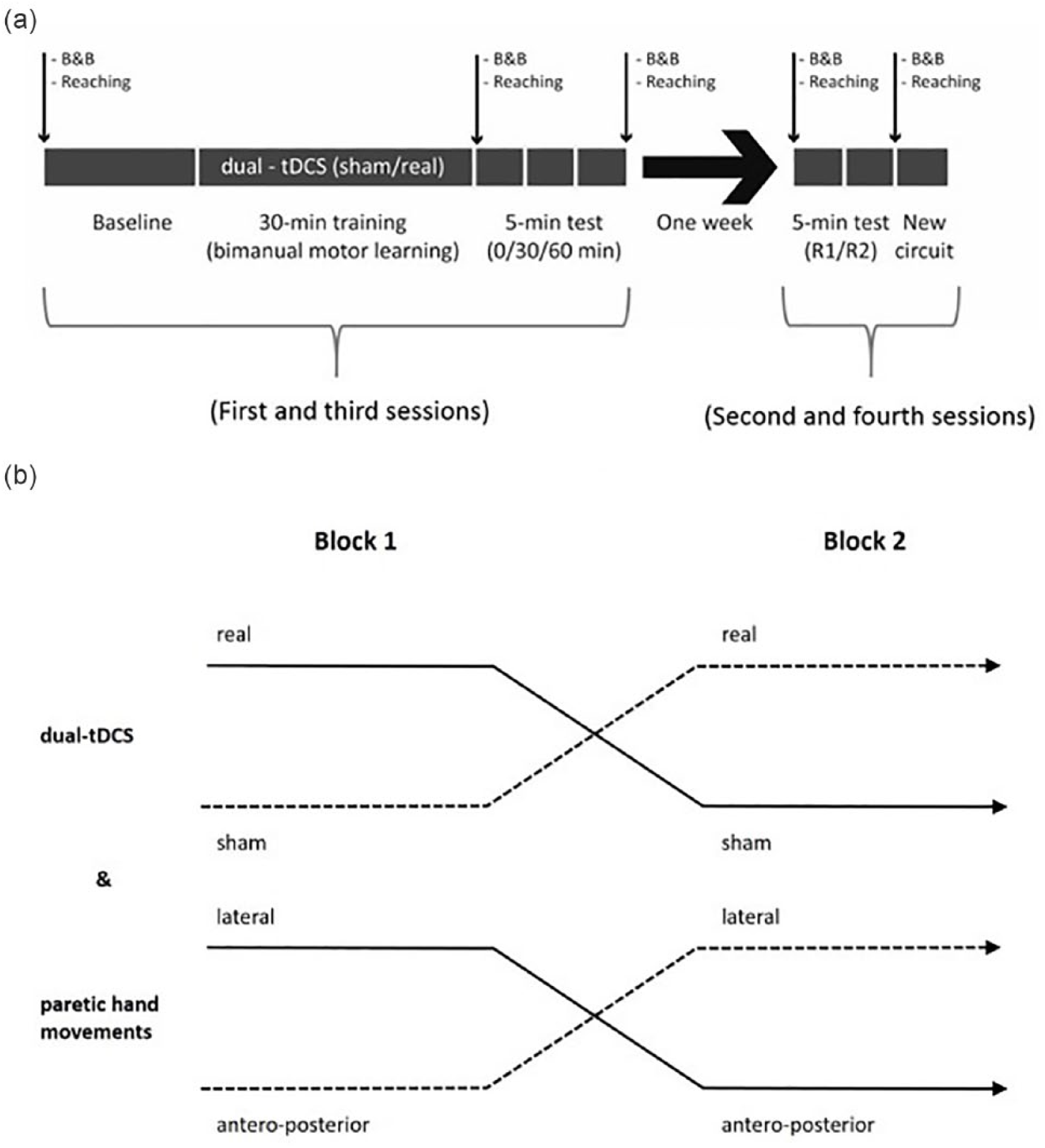
Study design. (a) This panel represents one block. Each experimental block (real and sham) consisted of an intervention session (combining training and stimulation) and a retention session performed after 1 week. CIRCUIT performance was tested at baseline, after, and 30 minutes, 60 minutes, and 1 week after training (retention blocks R1 and R2). Generalization was evaluated via the B&B (Box & Blocks) test, a bimanual REACHING task (small arrows), and with a new bimanual CIRCUIT task. (b) The order of the blocks (crossover design) was randomized and balanced for both dual-tDCS (transcranial direct current stimulation) and paretic hand movements.
At all sessions, patients were allowed 1 minute to familiarize or refamiliarize with the game using a simple square layout (Figure 2). During the intervention sessions, the patients performed a baseline CIRCUIT game (Figure 1), consisting of three 1-minute blocks, separated by 30 seconds of rest. Afterward, they trained on CIRCUIT for 30 minutes under real or sham dual-tDCS. Training consisted of 1-minute training blocks alternating with 30-second rest blocks; these blocks were grouped by 5 as T1, T2, . . .,T5. Tests were performed immediately after, and 30 and 60 minutes later, and were conducted in a manner identical to the baseline test. The retention sessions, performed a week later, consisted of 2 identical retention tests, R1 and R2, each consisting of three 1-minute blocks on the CIRCUIT layout on which the patients had been trained. Generalization was evaluated through (1) a bimanual cooperative REACHING task; (2) a unimanual B&B test (performed during baseline evaluation, immediately after training, and 30 and 60 minutes after training, and at the beginning and end of the retention sessions, Figure 1); and (3) three 1-minute blocks on a new CIRCUIT layout, completed at the end of the retention sessions.
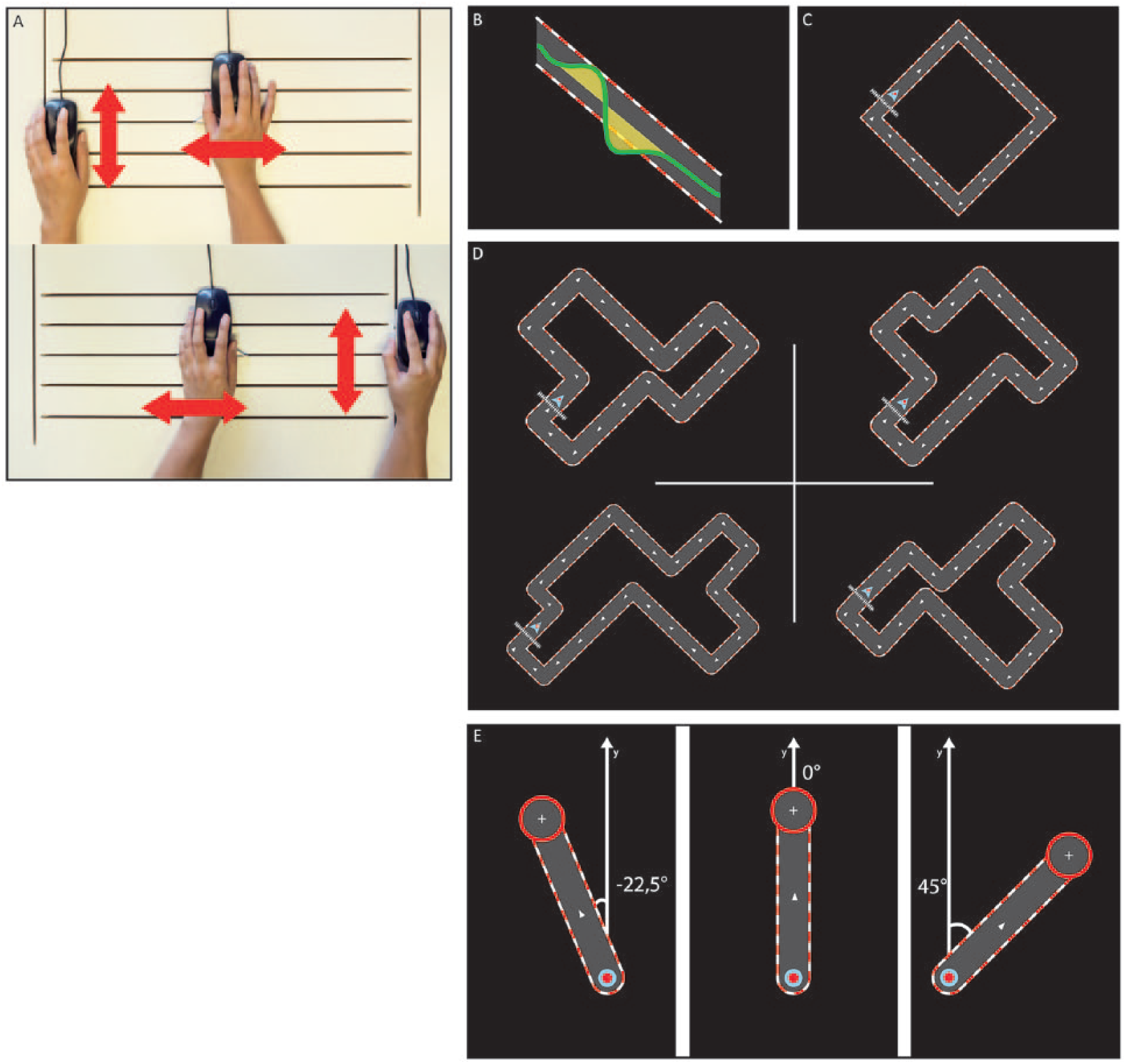
Bimanual tasks. (A) For the bimanual CIRCUIT task, one hand controls horizontal displacement (L/R) while the other hand controls vertical displacement (up/down). The computer mice were customized with small metallic ridges that glided along horizontal/vertical grooves carved into a custom-made board, physically constraining each hand’s axis. Coordinated movements of both upper limbs (ULs) were necessary to control the cursor. (B) Error was calculated as the surface area (cm2) enclosed between the real cursor trajectory and the midline of the track. (C) A simple square was used for familiarization with the task environment. (D) Four different CIRCUIT layouts of identical length and complexity were randomly chosen for use in the training and retention sessions, and for generalization assessments for which a new CIRCUIT layout was presented. (E) For the REACHING test, patients were asked to bimanually reach toward each of the 5 targets (set at ±45°, ±22.5°, and 0° to the home position), which appeared in a pseudo-randomized order, a total of 8 times each. Note that hitting the ±22.5° targets required a different bimanual control policy compared to the ±45° targets (which was identical as that of the CIRCUIT), and that hitting the 0° target was actually a unimanual task.
Dual-tDCS
One-milliampere tDCS was delivered using an Eldith DC-stimulator (NeuroConn, Ilmenau, Germany) via 2 electrodes (35 cm2) embedded in saline-soaked sponges located over the primary motor cortex (M1) of both hemispheres (dual-tDCS). The anode (cathode) was located over the ipsilesional (contralesional) M1, based on the C3-C4 positions of the international 10/20 electroencephalography system. Real stimulation started with an 8-second ramp-up to 1 mA for 30 minutes and finished with an 8-second ramp-down. For sham tDCS, the same ramp-up was followed by just 30 seconds of stimulation, followed by an 8-second ramp-down. Eldith codes (real/sham) were used to deliver dual-tDCS in a double-blind fashion.
Bimanual Cooperative Skill (CIRCUIT) Learning
The goal of the CIRCUIT task is to navigate a cursor across a complex “racetrack” displayed on a computer screen as fast and accurately as possible. Participants were instructed that both speed and accuracy were important to improve the score and that the ideal accuracy was staying inside of the circuit. In the bimanual version of the CIRCUIT game30,36,39,40 used in this study, subjects controlled the position of the cursor through the coordinated displacement of 2 computer mice, one of which was physically limited to lateral movements across a horizontal plane, and the other one to antero-posterior movements only (Figure 2).
To minimize carryover effects, one CIRCUIT was used for intervention and retention in sessions 1-2 and another randomly chosen CIRCUIT of identical length and difficulty was used for sessions 3-4, and the bimanual control policy (i.e., whether the paretic UL controlled the lateral or anteroposterior displacement of the cursor) was randomized between sessions 1-2 and 3-4. (Fig 2).
Bimanual REACHING
Generalization was further evaluated through a bimanual REACHING task. Using the same control rules as that used for the CIRCUIT exercises (ie, whether the paretic UL controlled lateral or anteroposterior cursor displacement), the patients were asked to move a cursor from a home position to 5 circular targets arrayed at the ±22.5°, ±45°, and 0° positions as quickly as possible, with each target appearing 8 times in a pseudo-randomized order.
In CIRCUIT, the ideal bimanual coordination required an equal contribution of both UL because the segments of the circuits were angled ±45°. REACHING with the ±45° targets evaluated generalization with an identical bimanual control policy and REACHING with the ±22.5° targets evaluated generalization with different bimanual control policies because the ±22.5° targets required another relative proportion of ULs’ movements. The 0° target was actually a “unimanual” task (Figure 2 and Supplementary Materials).
Randomization
Randomization was performed by a third person using minimization software (http://rct.mui.ac.ir/q), who then provided the other researcher in sessions 1-2 and 3-4 with instructions regarding the (1) Eldith codes to be used for dual-tDCS (real-sham/sham-real) and (2) the orientation controlled by the patients’ paretic UL (lateral/anteroposterior). The randomization criteria were: gender (male/female), age (18-50/50-70/>70 years), time since stroke (6-12 months/1-3 years/> 3 years), stroke laterality (dominant/nondominant hemisphere), UL deficit severity (near-normal/useful hand/afunctional hand/minimal arm movements) and stroke’s localization (cortical/subcortical). Table 1 includes the detailed characteristics of patients at inclusion.
Data Processing
The data were processed with Matlab (2015b, The MathWorks, Inc). Although the “relative phase” has been established for cyclic/rhythmic bimanual tasks,41,42 our task was nonrhythmic and noncyclic, necessitating the definition of 2 new bimanual outcomes:
1. Bimanual speed-accuracy trade-off (Bi-SAT):
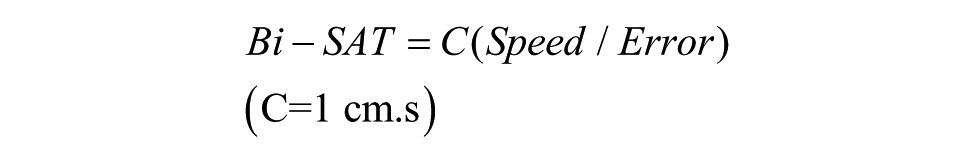
in which speed (cm/s) was calculated as the first derivative of cursor position on the screen, the cursor position was calculated from the combination of the positions of the 2 computer mice, and error was calculated as the surface area (cm2) enclosed between the recorded trajectory and the ideal trajectory defined by the mid-line of the track (Figure 2), as well as
2. Bimanual coordination factor (Bi-Co):
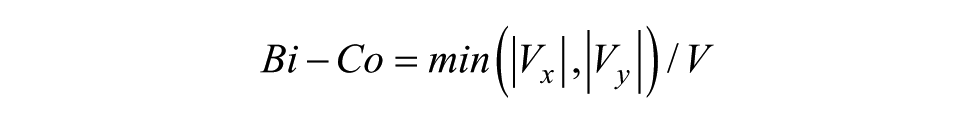
in which
For analyzing REACHING, the Bi-Co formula was adapted for targets set at ±22.5° angles (see Supplementary Materials). Finally, Bi-Co was not calculated for targets placed at 0° to the home position (unimanual REACHING).
After analyzing the data for each sample (20 ms) for both CIRCUIT and REACHING exercises, Bi-SAT and Bi-Co values were averaged for each 1-minute block per patient, following which the mean was calculated for each test (ie, baseline, T1, T2, T3, T4, T5, immediately after, after30, after60, R1, and R2).
To quantify the learning during the intervention sessions, the retention after one week and the generalization to a new CIRCUIT layout, the following formula was used:
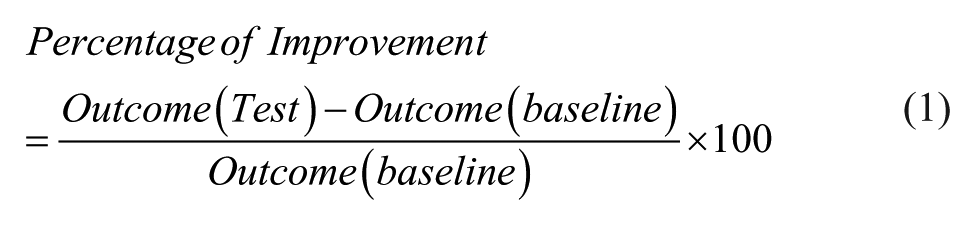
where “test” can either refer to the test performed 60 minutes after tDCS (after60), or R1, or generalization at the end of the retention sessions.
Statistical Analysis
Since Bi-SAT did not have a normal distribution, the data were presented on a logarithmic scale, and statistical tests were performed using a logarithmic transformation. Statistical tests for Bi-Co were performed on raw data, as it was symmetrically distributed.
Using the software package R, a generalized linear mixed model (GLMM) was carried out on the baseline data for Bi-SAT and Bi-Co to probe for potential carryover effects resulting from (1) order in which the stimulation was performed or (2) the control rules assigned to the paretic UL (direction of movements). Individuals were modeled as random effect, while [time], [time × stimulation order] interaction, [time × direction order] interaction, and [time × stimulation order × direction order] interaction were modeled as fixed effects.
The primary outcome was the retention at 1-week (R1) of the bimanual motor skill compared to baseline, using 2 other GLMMs for Bi-SAT and Bi-Co. Learning and retention of the CIRCUIT was investigated with the main effect of time in these 2 GLMMs.
Generalization was assessed through performance on new CIRCUIT layouts, REACHING and B&B. The effects of dual-tDCS on Bi-SAT and Bi-Co were investigated through a GLMM comparing the results from R2 and from the test conducted with new CIRCUIT layouts. A second GLMM compared bimanual REACHING results for the baseline and the first retention tests. A third GLMM compared the number of blocks transferred in the B&B test by both the paretic and nonparetic ULs during the baseline test and the first retention test.
Results
Bimanual CIRCUIT
The bimanual cooperative skill was learned and retained at the group level, as evidenced by the improvements in Bi-SAT (t61 df =8.25; P < .001) and Bi-Co (t61 df =4.99; P < .001; the main effect of time). Patients in both real and sham dual-tDCS conditions were able to learn and retain the bimanual cooperative skill (Figure 3, Supplementary Table 2), interpatient variability was similar for both real and sham tDCS as well. While both groups showed an overall improvement, there was slight deterioration in a few patients (see Supplementary Results). Dual-tDCS did not enhance the amount of learning when comparing baseline to R1 (Figure 4). A statistically significant difference between real and sham dual-tDCS for Bi-SAT (t61 df = 0.80; P = .42) or Bi-Co (t61 df =0.47; P = .63) was not detected by GLMM (Table 2).
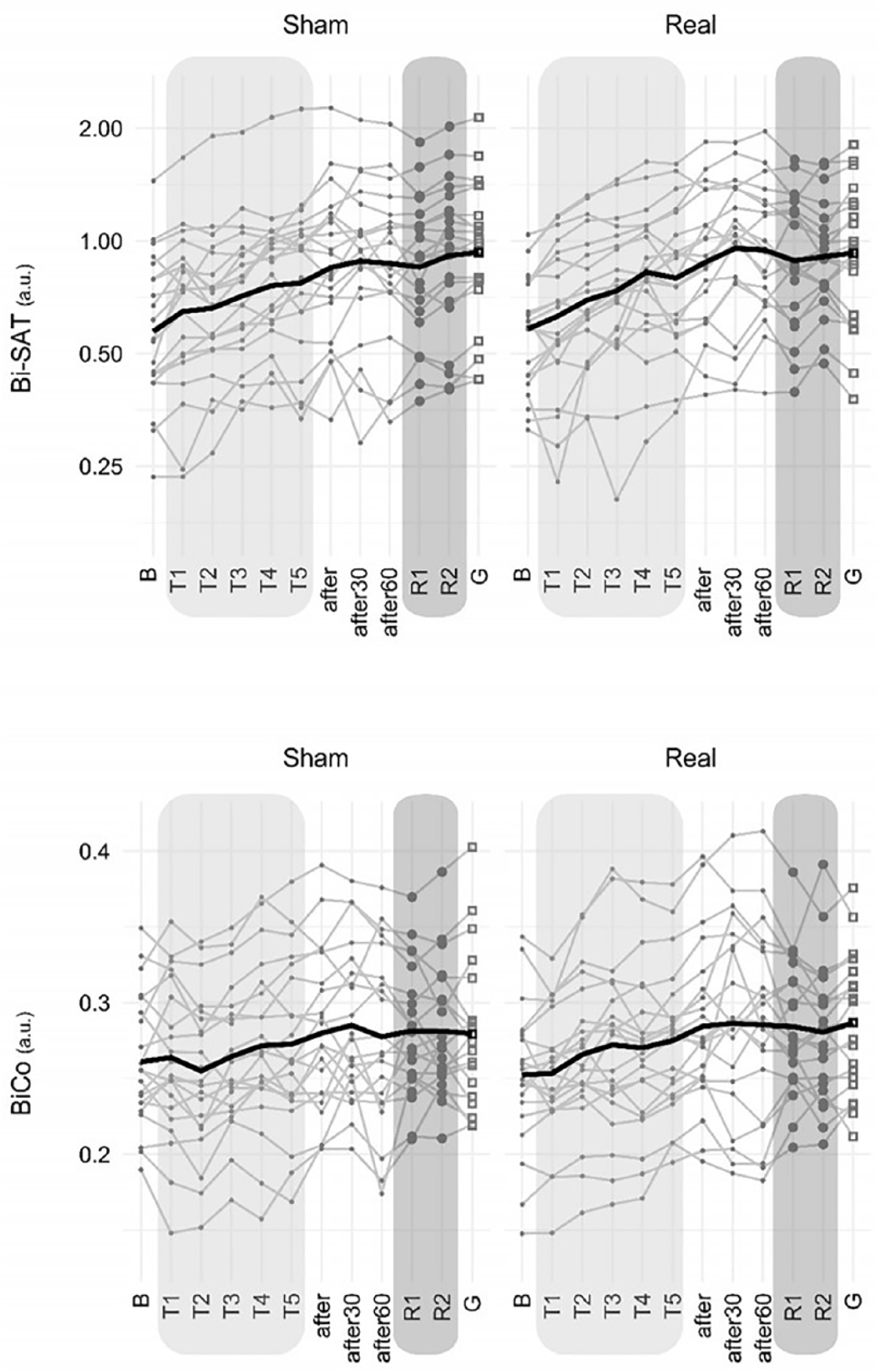
Improvement of Bi-SAT and Bi-Co on the CIRCUIT exercise for sham and real dual-tDCS (transcranial direct current stimulation). Bi-SAT and Bi-Co (expressed in arbitrary units, a.u.), respectively, quantify bimanual speed/accuracy trade-off (SAT) and coordination between the movements of the 2 hands. Thin gray lines correspond to individual patients, and the thick black line to the group mean. B, baseline; T1, training 1 (four 1-minute training blocks), T2-3-4-5, training 2-3-4-5; after, after stimulation; after30, 30 minutes after stimulation; after60, 60 minutes after stimulation; R1, retention test 1 (three 1-minute blocks), R2, retention test 2 (three 1-minute blocks); G, generalization (using a new CIRCUIT layout). Bi-SAT data are presented on a logarithmic scale and analyzed via logarithmic transformation.
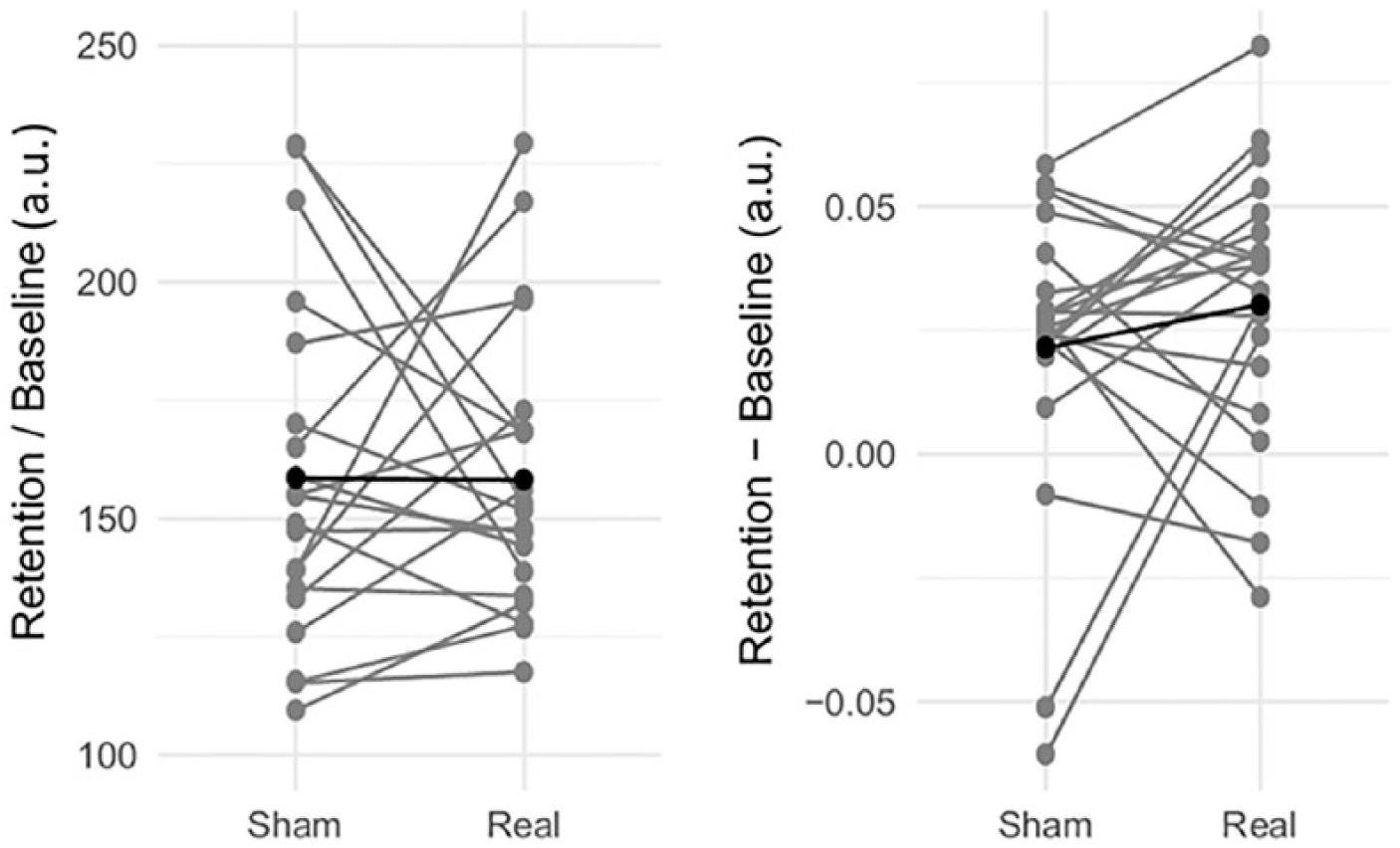
Retention test (R1) results for real and sham dual-tDCS (transcranial direct current stimulation) conditions. Comparison of the improvement in Bi-SAT (bimanual speed/accuracy trade-off) and Bi-Co (bimanual coordination) at the first retention test (R1; vs baseline values) in real and sham dual-tDCS conditions in stroke patients. As Bi-SAT data were subjected to a logarithmic transformation, this was calculated as R1/B, while Bi-Co improvement was calculated as R1-B. Gray and black lines represent individual patients and the group mean, respectively.
Summary of Results.
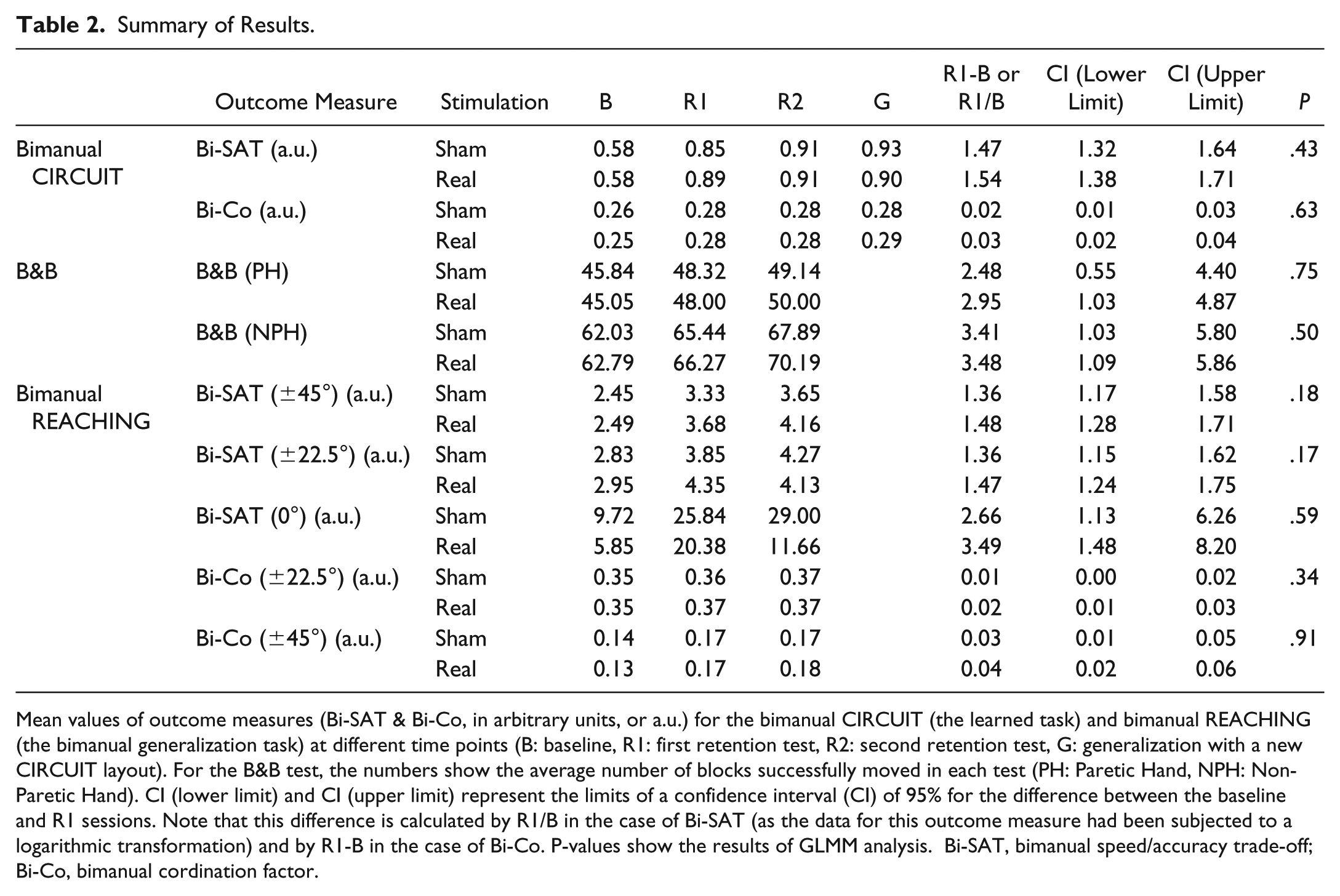
Mean values of outcome measures (Bi-SAT & Bi-Co, in arbitrary units, or a.u.) for the bimanual CIRCUIT (the learned task) and bimanual REACHING (the bimanual generalization task) at different time points (B: baseline, R1: first retention test, R2: second retention test, G: generalization with a new CIRCUIT layout). For the B&B test, the numbers show the average number of blocks successfully moved in each test (PH: Paretic Hand, NPH: Non-Paretic Hand). CI (lower limit) and CI (upper limit) represent the limits of a confidence interval (CI) of 95% for the difference between the baseline and R1 sessions. Note that this difference is calculated by R1/B in the case of Bi-SAT (as the data for this outcome measure had been subjected to a logarithmic transformation) and by R1-B in the case of Bi-Co. P-values show the results of GLMM analysis. Bi-SAT, bimanual speed/accuracy trade-off; Bi-Co, bimanual cordination factor.
The first GLMM did not detect a carryover effect. There was no difference between the baselines of the first and third sessions regardless of the dual-tDCS stimulation order (real/sham) for Bi-SAT or Bi-Co (see Supplementary Table 1). Similarly, no difference was detected based on the order of paretic UL’s direction of movements in Bi-SAT or Bi-Co. Finally, there was no [time × stimulation order × direction order] interaction for Bi-SAT or Bi-Co (Supplementary Table 1).
Generalization
First, generalization of the learned bimanual control policies was evaluated with a CIRCUIT layout unfamiliar to the patients, at the end of the retention sessions. While the patients maintained their performance on the new layout (Equation 1, Supplementary Table 2) demonstrating a large generalization, GLMM analysis revealed that real dual-tDCS did not appear to have a significant effect on Bi-SAT (t226 df = −0.94; P = .34) or Bi-Co (t228 df = 1.17; P = .24) compared with the sham tDCS.
Second, generalization was evaluated via a bimanual REACHING task. Similarly, although the patients improved in this task, the second GLMM analysis did not detect a significant effect of stimulation on the patients’ ability to hit any of the targets (±45°, ± 22.5°, and 0°; Supplementary Figure 3 and Supplementary Table 3, see details in Supplementary Results).
Third, generalization to unimanual activity was assessed with the B&B. The paretic and nonparetic UL performance improved by 7% ± 6% (mean ± SD) and 6% ± 5%, respectively, in the real dual-tDCS condition, as compared with 7% ± 8% and 5% ± 5% in the sham tDCS condition. The third GLMM analysis failed to indicate a statistically significant difference in the improvements between the 2 tDCS conditions for either the paretic (t60 df = 0.51; P = .61) or the nonparetic UL (t60 df = 0.05; P = .95; Supplementary Figure 4).
Discussion
In this double-blind, sham-controlled, crossover RCT involving 21 chronic hemiparetic stroke survivors, we found that dual-tDCS did not enhance learning of a new bimanual cooperative skill compared to sham stimulation. Nevertheless, the patients achieved learning and retention of new and complex bimanual control policies, including lasting generalization, as evidenced by their improved performance in untrained bimanual and unimanual tasks.
Dual-tDCS and Bimanual Skill Learning
In previous studies with hemiparetic stroke survivors, dual-tDCS enhanced online unimanual CIRCUIT motor skill learning with the paretic UL, as well as its retention 1 week later30,36,37 during the R1 session, with an effect size of 4.12 [2.96, 5.27] (Hedges’ g ± sg calculated as standardized mean differences). Here, dual-tDCS enhanced neither learning nor 1-week retention of bimanual CIRCUIT skill compared with a sham treatment (effect size: 0.1 [−0.5, 0.7]). What could be the reason for this discrepancy? Subtle differences between patients’ residual aptitudes could be a cause, but this seems unlikely as both the sample size and patient characteristics were similar to those of previous studies.
Alternatively, dual-tDCS delivered to the both M1 might not be well-suited to enhance bimanual skill learning in chronic hemiparetic stroke survivors. While several tDCS studies have demonstrated enhancements of UL motor recovery or learning after stroke,28,30,35-37,43 an increasing number of teams have reported contrasting results,44-46 and meta-analyses suggest that the effect of tDCS on poststroke motor function may either be inconsistent 47 or dose dependent. 48 As such, it is possible that the intensity we used (1 mA) was not sufficient. In healthy individuals, tDCS delivered to the left M1 or dorsolateral prefrontal cortex (DLPFC) 49 and high-definition anodal tDCS delivered over M1 or parietal cortex was ineffective in improving bimanual learning of a coordination task. 50 However, in our opinion, the absence of tDCS effect on bimanual motor skill control/learning in healthy individuals cannot predict the absence of tDCS effect in stroke survivors performing the same task. Indeed, because of functional impairments, there is more room for improvements in (bimanual) motor learning in stroke survivors, that is, much less ceiling effect as in healthy individuals. Finally, in the bimanual CIRCUIT task, both proximal and distal UL muscles were involved, as typically observed in most bimanual cooperative tasks. Despite the neurophysiological differences between distal and proximal UL muscles,51,52 using the same dual-tDCS montage in chronic stroke survivors, we observed improvement in the unimanual version of the CIRCUIT task,30,36,37 suggesting that this issue was not overcritical for the CIRCUIT task.
The model of poststroke interhemispheric imbalance has long driven the field of NIBS. However, it should be refined to account for the efficiency of the residual corticospinal tract, the role of noncorticospinal projections, and the recruitment of premotor areas from the (un)damaged hemisphere,18,53-56 factors that may require an additional level of precision in patient selection and stratification in trials combining NIBS and neurorehabilitation. The current model of interhemispheric imbalance may not be transposed wholesale to bimanual skills. Bimanual cooperative tasks are operated by neural coupling that involves the cortical areas and descending pathways from both hemispheres.13,57 After a unilateral stroke, the M1 and premotor areas of the undamaged hemisphere may become the new central hub of a reconfigured network coordinating bimanual actions. M1, the supplementary motor area (SMA), cerebellum, and premotor cortex (PMC) are involved in simple bilateral movements, and the bilateral secondary somatosensory (S2) plays a key role in bimanual cooperative tasks. 57 After a stroke, the neural coupling from the paretic to nonparetic side is impaired but the coupling from the nonparetic to paretic side is preserved. 13 Given that our bimanual task had a cooperative nature, it is thus possible that applying cathodal tDCS on the undamaged hemisphere may actually cause a deterioration in bimanual motor control, especially when considering the more bilaterally organized motor control of proximal UL muscles and the potentially negative impact that cathodal tDCS can exert on proximal UL control in severely impaired stroke survivors, 58 or at least cancel out the improvements resulting from anodal tDCS on the damaged hemisphere.18,54,59
In healthy individuals, dual-tDCS montages—regardless of whether the anode was placed over the contralateral or ipsilateral hemisphere—improved unimanual motor sequence learning nearly twice as effectively as “classical” unilateral tDCS montages. 60 This suggests the hemispheres function cooperatively, rather than competitively. Following a stroke, the undamaged hemisphere may become even more important for bimanual cooperative tasks. Therefore, the interhemispheric/competitive model may no longer apply once the reconfiguration of the network regulating bimanual actions has occurred after a stroke.
Bimanual Cooperative Learning After Stroke
Despite the lack of effect of dual-tDCS, the patients were able to learn (during the training session) and retain (1 week later) the new bimanual control policies under both tDCS conditions, and showed an almost complete ability to generalize these policies to a new, untrained, bimanual CIRCUIT layout and to other new bimanual and unimanual tasks. Thus, two hallmarks of motor skill learning—retention and generalization 61 —were observed, as in healthy individuals training on the bimanual version of the CIRCUIT (without tDCS). 40
The bimanual REACHING task quantified skill generalization toward the completion of a new task involving either identical (±45°) or different (±22.5°) bimanual control policies. Whereas the improvement of Bi-SAT and Bi-Co were similar for the ±45° angles, performances on Bi-Co were better for ±22.5° angles. These latter targets required a proportionally higher amount of anteroposterior displacement, which might have been “easier” to perform with the paretic UL, compared with lateral movements.
Therapeutic Implications
Our study demonstrated that bimanual CIRCUIT training, along with other bimanual neurorehabilitation methods such as Active-Passive Bilateral Priming (APBP), 62 Bilateral Arm Training with Rhythmic Auditory Cueing (BATRAC), 8 or, in children with cerebral palsy, Hand and Arm Bimanual Intensive Therapy Including Lower Extremity (HABIT-ILE), 63 allowed stroke survivors to learn, retain and generalize complex bimanual cooperative skills. Thus, the implementation of complex bimanual movements in neurorehabilitation should be encouraged, especially as skill generalization/transfer was so apparent. This could also apply to the improvement of unilateral motor function (i.e., B&B) through the learning of bimanual cooperative skills as here, or by training involving rhythmic movement. 64
Limitations
Although our sample size was modest, it was on par with most studies in the field, especially when considering the crossover design. Nevertheless, the comparison of the effect sizes of dual-tDCS on bimanual motor skill learning with those of our previous experiments on unimanual paretic UL motor skill learning led to a firm conclusion that as applied here, dual-tDCS does not enhance bimanual cooperative motor skill learning in stroke survivors. In addition, the patients had variable residual impairments and strokes of different etiologies, sizes, and locations. While this heterogeneity may explain the lack of effect, it could be argued that these results more accurately reflect what would be expected for a “real-life” population of stroke survivors. Finally, it might have been more desirable to provide training over several sessions, as well as testing tDCS with different intensities and dosages. However, before launching long trials, shorter studies are needed for optimization.
Conclusion
Dual-tDCS did not enhance the learning of a bimanual cooperative skill in chronic hemiparetic stroke survivors. The model of interhemispheric imbalance, the dominant framework for testing the potential of NIBS to improve paretic UL recovery, cannot be translated from unimanual to bimanual motor control, raising the need for testing alternative bimanual cooperation recovery models.11,65
Nevertheless, the stroke survivors were able to learn and retain a new complex bimanual cooperative skill under both real and sham dual-tDCS, and they could generalize their performance improvements to a similar bimanual CIRCUIT task, a bimanual REACHING task, and a unilateral B&B task, all tasks for which they were not trained. The inclusion of complex bimanual tasks should therefore be considered for future neurorehabilitation strategies.
Supplemental Material
Supplementary_Figure_1 – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Figure_1 for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Figure_2 – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Figure_2 for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Figure_3 – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Figure_3 for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Figure_4 – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Figure_4 for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Method – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Method for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Results – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Results for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Table_1 – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Table_1 for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_Table_2 – Supplemental material for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial
Supplemental material, Supplementary_Table_2 for Learning a Bimanual Cooperative Skill in Chronic Stroke Under Noninvasive Brain Stimulation: A Randomized Controlled Trial by Maral Yeganeh Doost, Jean-Jacques Orban de Xivry, Benoît Herman, Léna Vanthournhout, Audrey Riga, Benoît Bihin, Jacques Jamart, Patrice Laloux, Jean-Marc Raymackers and Yves Vandermeeren in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We are grateful to the stroke survivors who participated in the project, and to the nursing team of the Stroke Unit/Neurology Department for flexibility and assistance. We thank Stéphanie Lefebvre for her precious input in the design of the CIRCUIT task and Wojciech Gradkowski for the development of the software.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work of YV was supported by the following grants: Fonds National de la Recherche Scientifique–FNRS F 5/4/150/5 MCF/BAF – 84-89 1.R.506.16 & 1.R.506.18, Fonds de la Recherche Scientifique Médicale (FRSM) 3.4.525.08.F, Fonds Spécial de Recherche (FSR) from the Université catholique de Louvain (UCLouvain), Fondation Van Goethem-Brichant, and Fondation Mont-Godinne (2015, 2016, 2018). The work of MYD was supported by the FRNS-FRIA n° F 3/5/5- MCF/ROI/BC-19727 and F 3/5/5- MCF/XH/FC-17514. The work of JJOdX is supported by an internal grant of the KU Leuven (STG/14/054) and by the FWO (1519916N). The work of AR was supported by Fonds Spécial de Recherche (FSR) grant from the Université catholique de Louvain (UCLouvain) 2016-2018.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
