Abstract
Background. Recent evidence from both monkey and human studies suggests that the reticulospinal tract may contribute to recovery of arm and hand function after stroke. In this study, we evaluated a marker of reticulospinal output in stroke survivors with varying degrees of motor recovery. Methods. We recruited 95 consecutive stroke patients presenting 6 months to 12 years after their index stroke, and 19 heathy control subjects. Subjects were asked to respond to a light flash with a rapid wrist flexion; at random, the flash was paired with either a quiet or loud (startling) sound. The mean difference in electromyogram response time after flash with quiet sound compared with flash with loud sound measured the StartReact effect. Upper limb function was assessed by the Action Research Arm Test (ARAT), spasticity was graded using the Modified Ashworth Scale (MAS) and active wrist angular movement using an electrogoniometer. Results. StartReact was significantly larger in stroke patients than healthy participants (78.4 vs 45.0 ms, P < .005). StartReact showed a significant negative correlation with the ARAT score and degree of active wrist movement. The StartReact effect was significantly larger in patients with higher spasticity scores. Conclusion. We speculate that in some patients with severe damage to their corticospinal tract, recovery led to strengthening of reticulospinal connections and an enhanced StartReact effect, but this did not occur for patients with milder impairment who could use surviving corticospinal connections to mediate recovery.
Keywords
Introduction
After suffering a stroke, individuals pass through defined stages of recovery of upper limb function,1,2 with early spasticity and synergistic co-activation yielding to more fractionated forms of muscle activation. A single patient may fail to progress beyond a given recovery phase, leaving them with residual disability. The pattern of recovery suggests contributions from two underlying systems, 3 one contributing mainly strength, and the other most important for dexterity. Given recent evidence for a role of the reticulospinal tract in recovery of hand function,4,5 these systems may map onto reticulospinal and corticospinal tracts, respectively. 6 In stroke patients with little residual corticospinal tract, who remain heavily reliant on reticulospinal pathways even late in their recovery, the increased contribution from this pathway may lead to movement synergies,7,8 which limit the range of functional movements such as reaching.9,10
Noninvasive approaches to assess reticulospinal function in humans are limited. One potentially useful method is the StartReact paradigm, which measures the shortening of a voluntary reaction time when a visual cue is paired with a loud sound. 11 Given the proven role of the reticulospinal tract in overt acoustic startle responses, StartReact has been hypothesized to reflect reticulospinal activity. Increased StartReact effects are seen in patients with hereditary spastic paraplegia, 12 in patients after spinal cord injury for gross but not fine grasp responses, 13 and in stroke patients 14 in agreement with an enhancement of reticulospinal pathways in these conditions. In this study, we applied the StartReact method to the wrist flexor muscles of stroke patients and correlated the degree of reaction time shortening with measures of upper limb function.
Methods
We studied 95 consecutively recruited subjects (mean age 53 years; 67 male) who had recovered for 6 months to 12 years after a hemorrhagic or ischemic stroke, and 19 healthy controls. All subjects provided written informed consent, and procedures were approved by the Institutional Ethics Committee of the Institute of Neurosciences, Kolkata, India (reference number I-NK/EthicsComm/46/2016 dated April 2, 2016). Stroke patients with brainstem involvement assessed at stroke onset by magnetic resonance imaging, or those with visual or auditory impairment or individuals who had received botulinum toxin therapy for spasticity in the preceding 3 months were excluded. To avoid difficulties in understanding the task requirements, patients were screened for cognitive impairment using the mini mental state examination and excluded if they scored below 18. Stroke was determined from magnetic resonance imaging as cortical in 24% of patients, subcortical in 40%, and mixed in the remainder.
For the more affected side, upper limb function was assessed by the Action Research Arm Test (ARAT) 15 and spasticity at the wrist by the Modified Ashworth Scale (MAS). 16 The angular range of active wrist movement was measured using an electrogoniometer (SG 75, Biometrics Ltd, Newport, UK). All assessments were performed by the same individual (author SC), without knowledge of the StartReact results.
StartReact was assessed using surface electromyogram (EMG) recordings from electrodes placed over the forearm flexor compartment on the more affected side. Signals were amplified (30-2000 Hz bandpass, gain 2K) and sampled to hard disc at 5 kHz for offline analysis (1401 interface with Spike2 software, Cambridge Electronic Design, Cambridge, UK). Subjects sat with the affected arm flexed ~90° at the elbow with the forearm in a cushioned plastic cast fixed to a table. The hand was held semiprone with fingers extended and restrained between 2 vertical plastic blocks, whose spacing could be adjusted to clamp the hand firmly but comfortably in position. This arrangement was clamped to a strain gauge suitable for isometric wrist flexion. Subjects were instructed to attend to a red light-emitting diode (LED) placed ~1 m in front of them, and to make a rapid wrist isometric flexion movement after it illuminated (LED on time 50 ms). At random, the LED flash was delivered alone, or paired with a quiet (80 dB; 500 Hz, 50 ms) or loud acoustic stimulus (110 dB). These intensities are comparable to those that have been used in previous studies.12,17 The loud sound evoked a clear startle in the subjects on initial presentation. Before the start of recordings, subjects were familiarized with the task by practicing 3 to 4 responses to the LED flash. Five consecutive loud sounds were then delivered without task performance, to habituate the overt startle reaction. Both practice and habituation trials were discarded without analysis. Twenty responses in each condition, in randomized order, were then recorded with a 5-second intertrial interval (see Figure 1A and B for example single trial data).
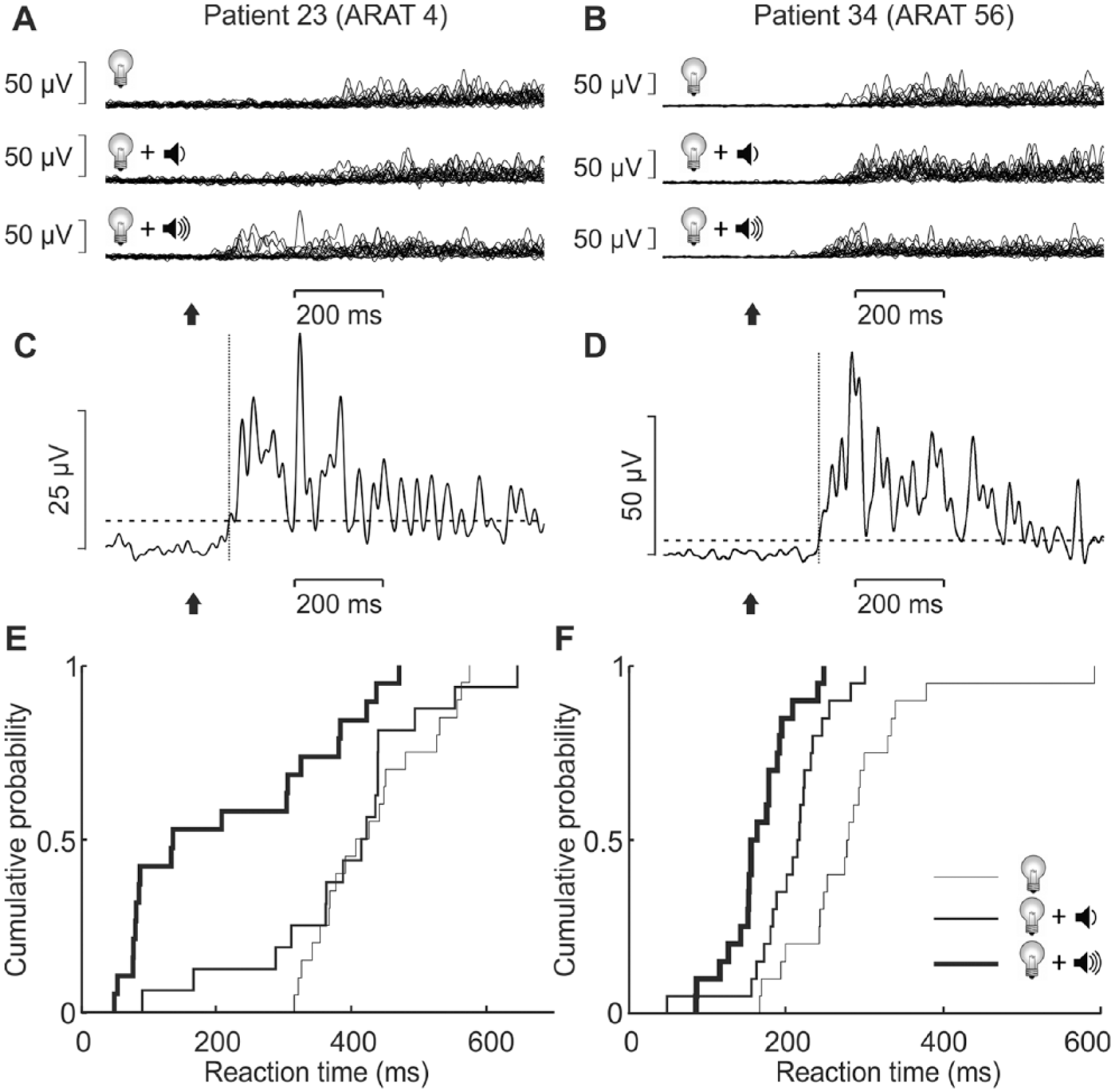
Representative data from 2 patients. Left column shows data from patient 23 who had severely impaired upper limb function, with an Action Research Arm Test (ARAT) score of 4/57; right column from patient 34 who had minimal impairment (ARAT 56/57). (A, B) Electromyography (EMG) recordings from the forearm flexors. Recordings have been rectified and low-pass filtered; trials are overlain for the 3 tested conditions of visual reaction time (VRT, light flash cue only), visual-auditory reaction time (VART, light flash plus quiet sound cue), and visual-startle reaction time (VSRT, light flash plus loud sound cue). (C, D) Single trial EMG responses, indicating the threshold set to the mean plus 3 standard deviations (SDs) of the baseline (horizontal dashed line), and the detected reaction time (vertical dotted line). Arrows indicate cue onset time. (E, F) cumulative distribution of detected reaction times in the 3 conditions. Note the greater separation between VART (medium line) and VSRT (thick line) in (E) than in (F).
The reaction time from stimulus to the first increase in EMG activity above baseline (defined as crossing pre-stimulus baseline plus 3 standard deviations [SDs], see Figure 1C and D) was measured on each trial automatically using a custom Matlab script, which also displayed sweeps and allowed erroneously assigned times (caused, eg, by electrical noise artifacts) to be manually corrected or excluded. We designated measures after LED flash alone the visual reaction time (VRT); when paired with quiet sound the visual-auditory reaction time (VART), and with the loud sound the visual-startle reaction time (VSRT).
In a few stroke patients, we repeated these measurements with a wrist extension movement in response to the cue, measuring reaction time from EMG collected from the forearm extensor. Such measurements were not possible in the majority of patients, due to extensor weakness, and are reported only briefly at the end of the Results section for completeness.
Results
The demographic and disease profile of 95 stroke patients are presented in Table 1. Out of 95 stroke patients, useable reaction time data was obtained from 46. In the remainder, reliable measures could not be made because either they complained of fatigue after a few trials and could not complete the task or their wrist flexor EMG signal was too weak to be usable. The mean ARAT score for the patients who could perform the task successfully was, as expected, higher (22.5, SD 21.4) compared to those who failed to produce usable StartReact data (8.6, SD 9.8).
Summary of Patient Demographics and Disease Profile. a
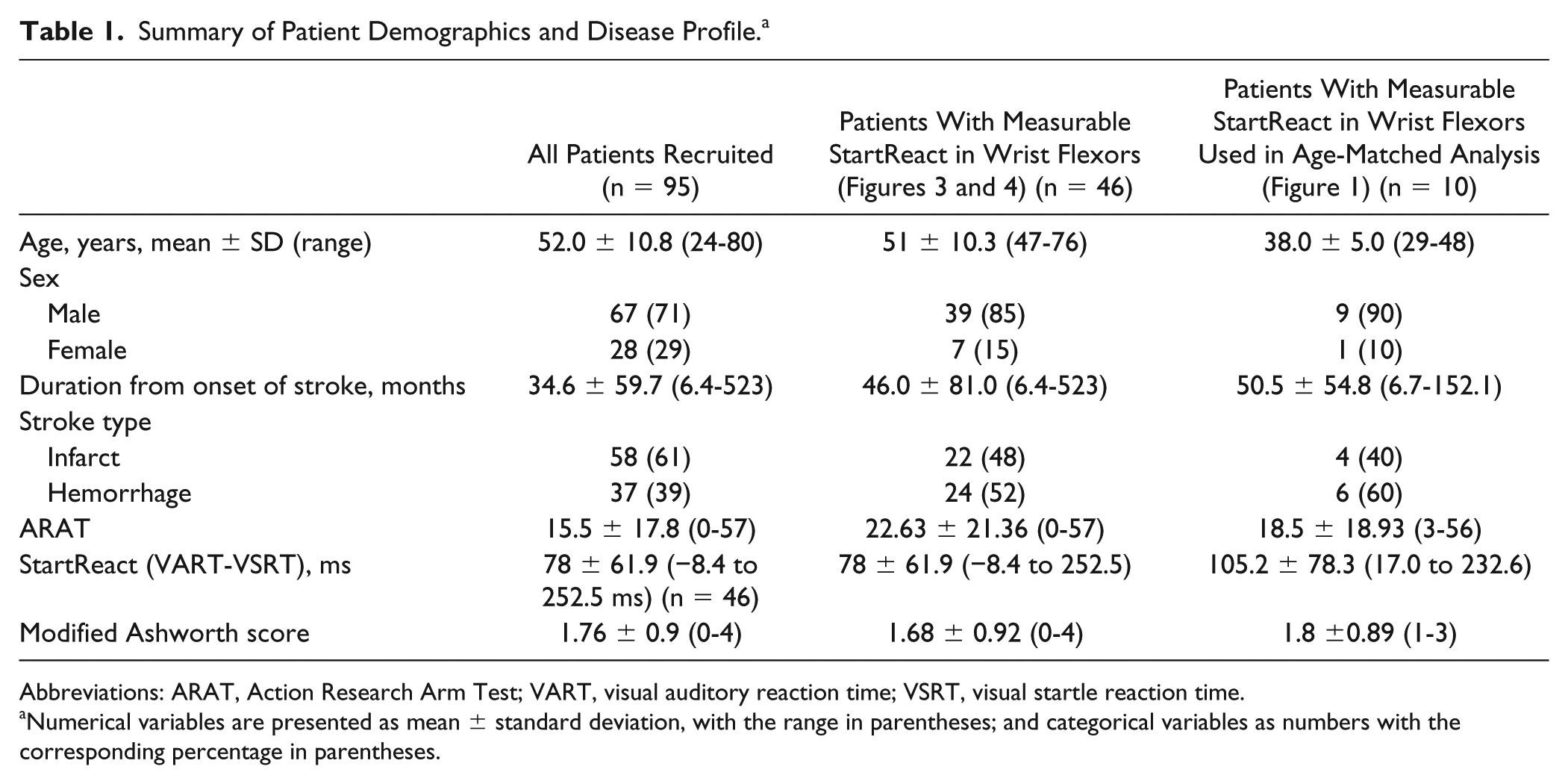
Abbreviations: ARAT, Action Research Arm Test; VART, visual auditory reaction time; VSRT, visual startle reaction time.
Numerical variables are presented as mean ± standard deviation, with the range in parentheses; and categorical variables as numbers with the corresponding percentage in parentheses.
Figure 1E and F illustrates results from 2 patients with very different residual impairment, as cumulative distribution plots of single trial measures of VRT, VART, and VSRT. Patient 23 (Figure 1E) had severely impaired upper limb function (ARAT 4/57), and there was a wide separation between the curves for VART and VSRT (mean difference 170 ms). By contrast, patient 34 (ARAT 56/57; Figure 1F) had smaller separation between these curves, and the mean VART-VSRT difference was only 43 ms, within the range seen in healthy subjects.
Figure 2A presents the comparison of reaction time measures between the healthy participants and stroke survivors. Unsurprisingly, all reaction times were significantly prolonged in the stroke subjects. We used the difference between VART and VSRT as an estimate of the StartReact effect. This was also greater on average in the stroke subjects (all comparisons P < .005; t test with Benjamini-Hochberg correction for multiple comparisons). This result was unaltered when we limited the comparison to a subset of stroke survivors matched to the age of the healthy participants (average age 35 years in healthy and 38 years in stroke, ages not significantly different, P = .399; Figure 2B). Through analysis of covariance, we found that stroke was a significant predictor of reaction times and the StartReact effect even after adjusting for age as covariate (P < .05).
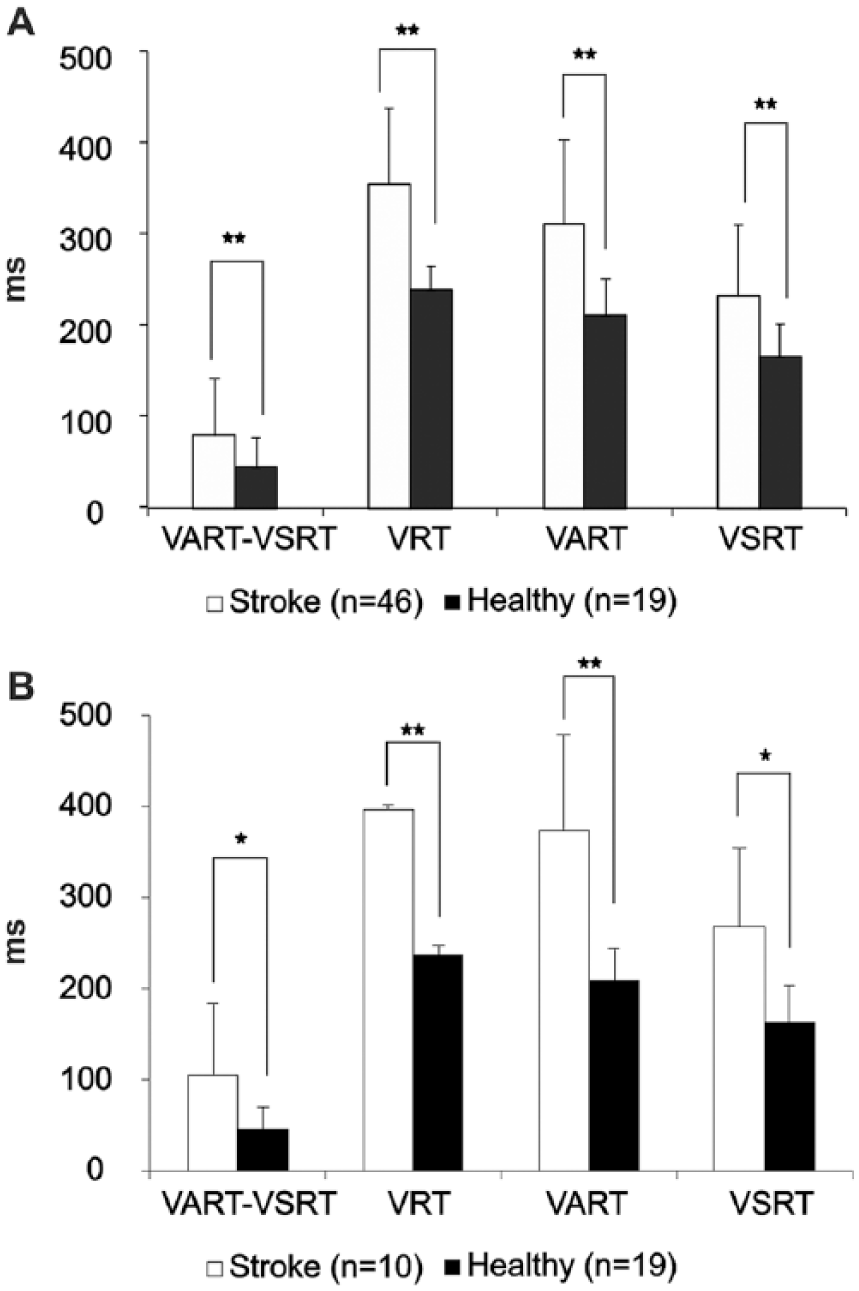
Comparison of StartReact responses between healthy subjects and stroke survivors. (A) Comparison between all 46 stroke survivors in which VSRT-VART could be measured, and 19 healthy participants. (B) Comparison between 19 healthy participants and 10 age-matched stroke survivors. In both cases, stroke survivors demonstrated a significantly larger StartReact effect (VART-VSRT) compared with healthy participants (unpaired t-test). They also showed early visual reaction times (VRT), visual auditory reaction times (VART), and visual startle reaction times (VSRT). Error bars plot SD. The normality of the data was tested by 1-sample Kolmogorov-Smirnov test (P > .05). The homogeneity of variance was assessed by Levene’s test for equality of variance and accordingly the type of unpaired t test was selected. P value of unpaired t test is indicated by **P < .005; *P < .05, corrected for multiple comparisons by the Benjamini-Hochberg procedure.
Although on average stroke subjects had a significantly greater StartReact effect, there was considerable heterogeneity between individuals, as illustrated already by the 2 subjects shown in Figure 1. To confirm the impression that patients with greater functional impairment had a larger StartReact effect, we measured the correlation between StartReact (ie, VART-VSRT difference) and measures of upper limb function. Scatterplots of StartReact effect versus the overall ARAT score, and active angular range of motion are shown in Figure 3A and B, respectively; in each case, the correlation was significant. The subdomains of the ARAT score, and the degree of wrist flexion, were also significantly correlated with StartReact (see Table 2). Although these correlations indicate an association between the functional measures and StartReact, this is clearly not well represented by a linear relationship. For the plots in Figure 3, it appears that patients fell into 2 categories: those with StartReact in the normal range (indicated by the bar underneath the abscissa) had highly variable functional scores (ARAT, Figure 3A, mean 27.1, SD 21.5; active range of motion, Figure 3B, mean 61.7°, SD 40.1°; n = 37). Those with a StartReact effect greater than the normal range had reduced, and less variable functional scores (ARAT, mean 4.3, SD 3.6; active range of motion, mean 22.9°, SD 12.8°; n = 9). These differences were significant (difference in mean, unpaired t test, P = .0001 for both ARAT and range of motion; difference in variance, Levene’s test for equality of variance, both P = .0001).
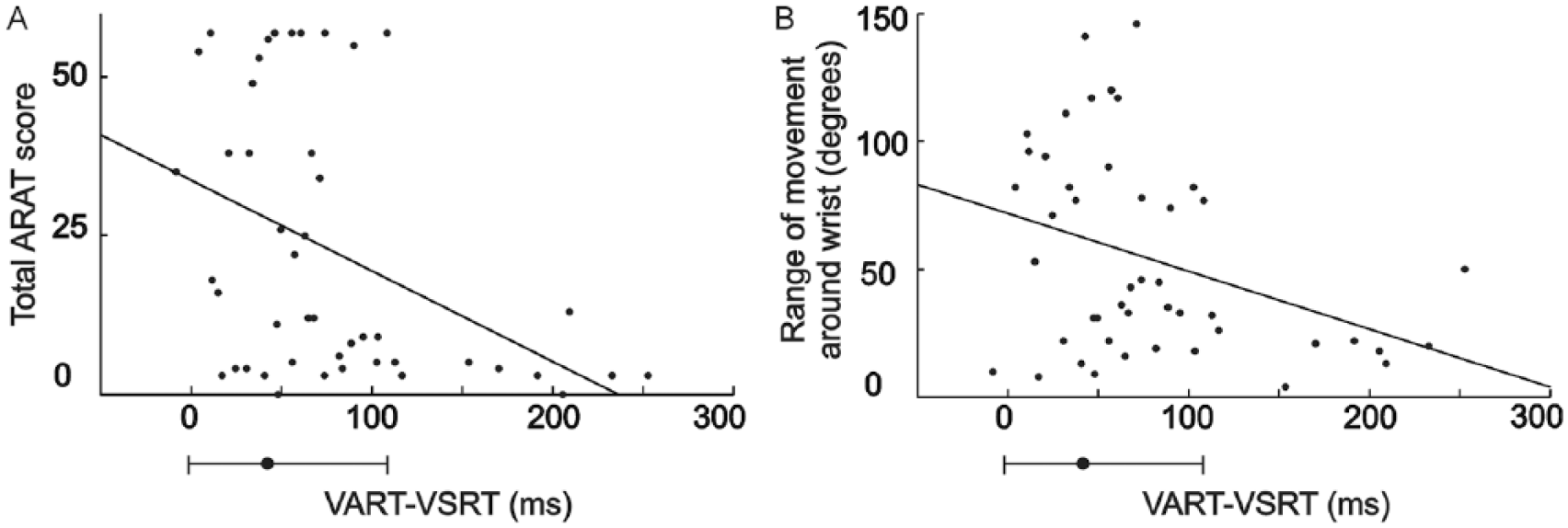
Scatterplots showing correlation between startle response and measures of upper limb function in stroke survivors. Scatterplots summarizing the correlation between StartReact (VART-VSRT) and (A) ARAT score (maximum 57), (B) active angular range of movement around the wrist. Note the significant negative correlation between VART-VSRT and total ARAT score (ρ = −0.393; P = .007) and VART-VSRT and active angular wrist movement (ρ = −0.303; P = .043). The horizontal error bar below the abscissa indicates the median and 95% range of VART-VSRT in healthy individuals. ARAT, Action Research Arm Test; VART, visual auditory response time; VSRT, visual startle reaction time. Results are presented for 46 patients in whom VART-VSRT could be measured.
Summary of Cross-Correlation Analysis: Measures of Upper Limb Function and StartReact. a
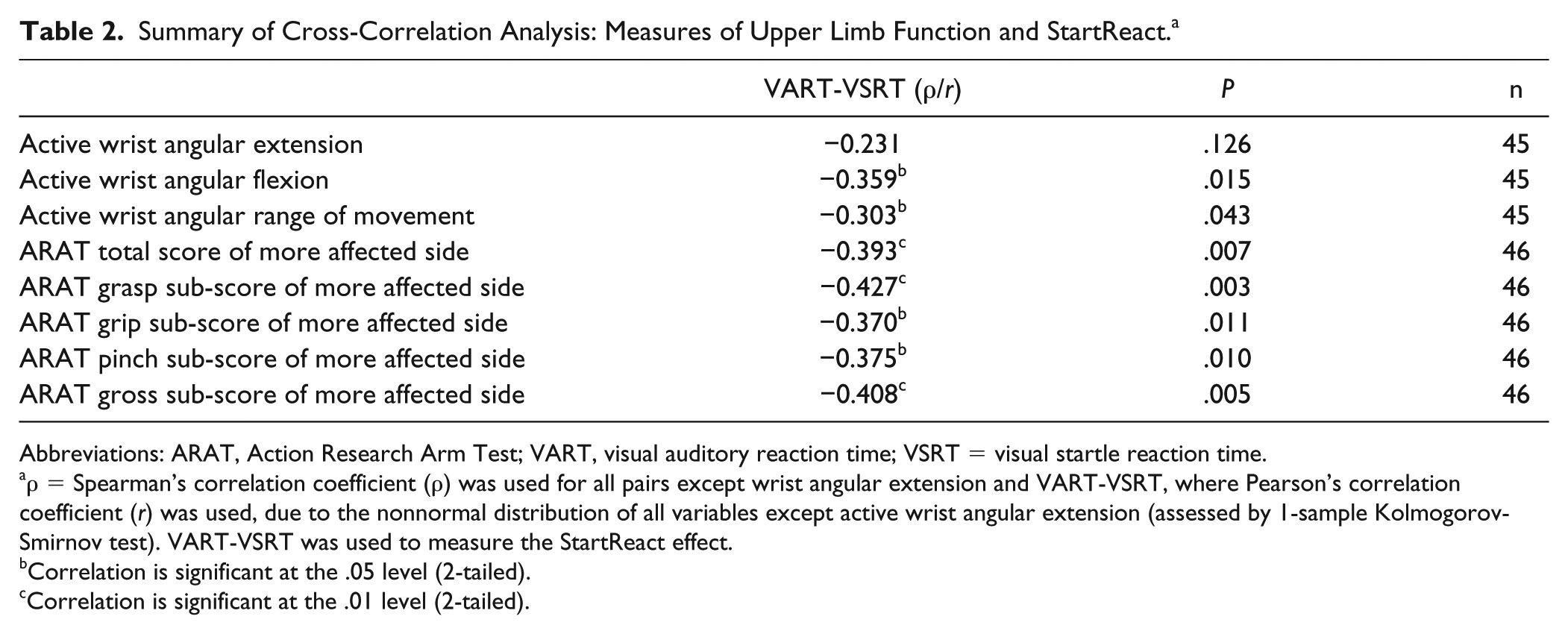
Abbreviations: ARAT, Action Research Arm Test; VART, visual auditory reaction time; VSRT = visual startle reaction time.
ρ = Spearman’s correlation coefficient (ρ) was used for all pairs except wrist angular extension and VART-VSRT, where Pearson’s correlation coefficient (r) was used, due to the nonnormal distribution of all variables except active wrist angular extension (assessed by 1-sample Kolmogorov-Smirnov test). VART-VSRT was used to measure the StartReact effect.
Correlation is significant at the .05 level (2-tailed).
Correlation is significant at the .01 level (2-tailed).
One possible explanation for the increased StartReact (probably reflecting an increase in reticulospinal outputs) in patients with more limited hand/wrist function is that movement was more impaired by spasticity in these individuals. The pathogenesis of spasticity is thought to involve the reticulospinal tract. 18 To explore this, patients were separated into mild (MAS 0-1; n = 15) and moderate/severe (MAS 1.5-4; n = 31) levels of spasticity. There was a significantly larger StartReact effect in the patients with greater spasticity (P < .05, t test; Figure 4).
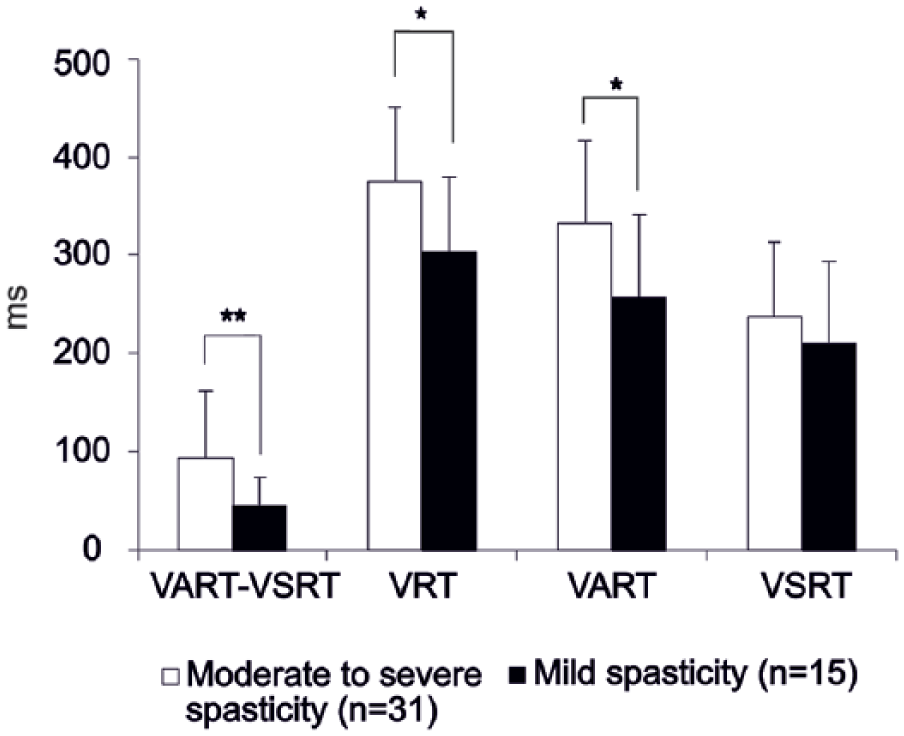
Comparison of StartReact responses in stroke patients grouped according to severity of spasticity. Bar chart comparing the effect of spasticity on various reaction time parameters. Patients with higher spasticity (Modified Ashworth score >1) showed significantly larger StartReact measure (VSRT-VART) compared with patients with mild upper limb spasticity (Modified Ashworth score 0-1) according to the unpaired t test (P values are included in the bar chart). Error bars plot SD. VRT, visual response time; VART, visual auditory reaction time; VSRT, visual startle reaction time. Data were assessed as normally distributed (Kolmogorov-Smirnov test, P > .05). The homogeneity of variance was assessed by Levene’s test for equality of variance and the type of unpaired t test was selected accordingly. P value of unpaired t test indicated by **P < .005; *P < .05, corrected for multiple comparisons by the Benjamini-Hochberg procedure. Results are presented for 46 patients in whom VART-VSRT could be measured.
In addition to measuring StartReact with a wrist flexion response, we also attempted to repeat the measurement using a wrist extension response and recording EMG from the forearm extensor muscles. This proved impossible in almost all stroke survivors, due to the profound extensor weakness commonly seen after stroke. However, in 13 patients, usable data were obtained. These patients were among the least impaired in our sample (ARAT mean 43.4, SD 17.0, range 9-57). There was no significant difference between Startreact measures made from the flexor and extensor muscles in these subjects (VSRT-VART for extensors mean 34.0 ms, SD 29.3 ms, for flexors mean 52.3 ms, SD 34.0 ms; P > .05, paired t test).
Discussion
Several previous reports have investigated reticulospinal contributions to stroke recovery using overt startle responses in more proximal muscles. Startle is enhanced in stroke patients compared with controls, 19 and correlates positively with spasticity19,20 and negatively with strength in the biceps muscle. 19 Within a single patient, the startle response in biceps is elicited more frequently on the more affected side. 20 Stroke patients show slowed voluntary reaction times in the biceps and triceps muscles compared with healthy subjects, but reaction times following startling cues are similar to healthy people; this suggests that the StartReact effect for elbow muscles is greater in stroke survivors. Additionally, stroke survivors attempting an elbow extension movement generate excess and inappropriate elbow flexor activity when the movement is triggered by a startling cue; this suggests that an overt startle reflex may overlie and interfere with startle-triggered movements in these individuals. 21
Recent work in monkey has demonstrated a role for the reticulospinal tract in hand movements as well as grosser movements involving upper arm muscles,22-24 and that these connections can strengthen during functional recovery. 4 Consistent with this, the StartReact effect can be seen in hand muscles.25,26 Our finding of enhanced StartReact in stroke patients for a wrist flexion movement, and a correlation with spasticity, is consistent with the previous studies.
How should we interpret the negative correlation between measures of upper limb function and StartReact? The obvious conclusion might be that enhancement of reticulospinal function is detrimental and produces deficits in upper limb function. Such a conclusion may be supported by the positive correlation with spasticity (Figure 4). Previous work has suggested that movement synergies, which often impair movements in stroke survivors, 9 have an origin in the strengthening of reticulospinal outflow after corticospinal damage.27-29 It is known that the reticulospinal tract is less able to generate fractionated patterns of independent muscle activation than the corticospinal tract.6,30,31 If reticulospinal connections have only detrimental effects, this would suggest that therapy to improve upper limb function should concentrate on reducing reticulospinal drive.
It is important to remember, however, that the demonstration of an association between reticulospinal enhancement and poor upper limb function does not tell us anything about cause and effect. An alternative explanation is possible, based on the idea of separate systems to produce recovery of strength and dexterity, putatively linked to the reticulospinal and corticospinal tracts, respectively. 3 On this view, where cortical damage is slight, recovery can proceed using spared corticospinal fibers from the primary motor cortex adjacent to the infarct, 32 or (less effectively) from nonprimary motor areas.33,34 Such patients would be expected to recover considerable dexterity, and score well on the ARAT. There would be no need for enhanced reticulospinal connections. By contrast, where cortical damage is extensive, few spared corticospinal resources will remain. Recovery must then use the “second best” alternative of reticulospinal connections, which can generate only an approximation of the desired motor output 6 leading to synergistic patterns of muscle activation,27-29,35 which are nonetheless preferable to paralysis.28,36 Such patients would have poor dexterity, and may have spasticity generated by aberrant reticulospinal outflow. Both factors would produce poor ARAT scores. This view is in accord with other work on ipsilateral motor evoked potentials following transcranial magnetic brain stimulation, which are most likely carried by a cortico-reticulospinal route.4,37-39 Patients with such ipsilateral responses typically show poorer recovery. 40 On this view, an intervention that strengthens reticulospinal outputs would aid recovery, but only in severely affected individuals. More moderately affected stroke survivors should instead focus on strengthening residual corticospinal output, which may allow recovery without interfering synergies. This idea is supported by a previous report, which attempted to suppress the activity of the ipsilateral cortex using transcranial direct current stimulation. 41 Such stimulation improved upper limb control for patients with mild impairment and limited damaged to the corticospinal tract assessed by magnetic resonance imaging but led to some loss of limb control in those with modest to severe impairment and greater corticospinal damage. A recent clinical trial suggested that optimizing the use of the residual corticospinal tract (over the contralesional cortico-reticulospinal tract) plays an important role in the improvement of reaching function in individuals with moderate to severe stroke. 42
The correlation coefficients that we observed between measures of hand function and StartReact were modest (ρ = −0.3 to −0.39, Figure 3); this would be consistent with the correlation being mediated indirectly via a third variable (ie, extent of corticospinal tract damage). In contrast, a previous study 28 measured the correlation between the Fugl-Meyer score (a measure of impairment) and a laterality index of brain activation (presumably measuring contralateral cortico-reticulospinal recruitment) and found stronger correlations (r2 = 0.57 to 0.93). This would be consistent with a more direct mediation.
It is of interest that, if we consider only the most severely impaired subjects with active range of motion below 60°, there appears to be a bimodal distribution of StartReact effect (Figure 3B). It has been previously reported that severely impaired stroke survivors fall into 2 groups: those who do, and do not, follow the “proportional recovery rule” and recover ~70% of lost function. 43 Given that we made our measurements at least 6 months after stroke, it is likely that recovery had reached a plateau. The subjects who remained severely impaired in our cohort are thus likely to be the “nonfitters” to the proportional recovery rule; it is known that these individuals are likely to have the most impaired corticospinal tract. 44 Our results may therefore suggest that these individuals can be further divided into those that have, and have not, substantially strengthened their reticulospinal outflow. This could be affected by many factors, most obviously by the detailed location of the lesion. In agreement with this, a previous report showed that more severely affected patients had, on average, an increased output from the ipsilateral hemisphere to the paretic limb compared with less impaired patients; however, there was considerable variability within the severely affected group. 45
In this study, we concentrated on wrist flexion movements. This is in contrast to a previous report by Honeycutt et al, 46 which examined StartReact during wrist extension and found no difference between stroke survivors and healthy controls. However, wrist extension is frequently impaired after stroke, with many stroke survivors unable to make an extension effort. Measurement of StartReact in wrist extension necessarily therefore involves recruitment of less impaired individuals; in agreement with this, in the work by Honeycutt et al, 46 7 of 8 subjects had Chedoke hand scores ≥4 (out of a maximum of 7); the single subject with a Chedoke score of 3 exhibited no StartReact effect. Our own limited data set from a few patients capable of wrist extension also found no difference in StartReact between flexion and extension responses. Reticulospinal connections to forearm extensors do not seem to strengthen after corticospinal lesions, 4 which may explain the lack of extensor function in individuals unable to recruit sufficient spared corticospinal fibers to the recovery process.
Footnotes
Authors’ Note
Statistical analysis was done by Prof Stuart Baker and Dr Supriyo Choudhury.
Author Contributions
Supriyo Choudhury: Study concept and design, acquisition of data, analysis and interpretation, critical revision of the manuscript for important intellectual content
A. Shobhana: Patient diagnosis, critical revision of the manuscript for important intellectual content
Ravi Singh: acquisition of data, analysis and interpretation, critical revision of the manuscript for important intellectual content
Dwaipayan Sen: Patient diagnosis, critical revision of the manuscript for important intellectual content
Sidharth Shankar Anand: Patient diagnosis, critical revision of the manuscript for important intellectual content
Shantanu Shubham: Patient diagnosis, critical revision of the manuscript for important intellectual content
Mark R. Baker: Study concept and design, acquisition of data, analysis and interpretation, critical revision of the manuscript for important intellectual content
Hrishikesh Kumar: Study concept and design, acquisition of data, analysis and interpretation, critical revision of the manuscript for important intellectual content, study supervision
Stuart Baker: Study concept and design, acquisition of data, analysis and interpretation, critical revision of the manuscript for important intellectual content, study supervision
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for study was provided by Wellcome Trust grant 101002 and MRC grant MR/P023967/1 to SNB.
