Abstract
Keywords
Introduction
Upper-limb impairment, a frequent disabling consequence of stroke 1 affects participation and quality of life. 2 Improvements in upper-limb impairment can be attributed to sensorimotor learning and adaptive plasticity in the nervous system 3 that extend well past the acute stages.4,5 A promising new rehabilitation approach uses virtual reality technology, a multisensorial experience that permits feedback-based motor learning in a computer-generated virtual environment (VE). 6 Virtual reality technology permits individualization of training environments, 7 combining intensity, variability, specificity, motivation, and inter-activity of practice. These factors may enhance experience-dependent neuroplasticity, influence motor learning, and improve rehabilitation outcomes.8,9
VEs can be manipulated to provide meaningful visual, auditory, and haptic/tactile feedback to the learner. Feedback can be delivered as enhanced information about knowledge of results (KR) and of performance (KP). 10 Task practice in VEs with feedback may increase activation in the ipsilesional hemisphere.11,12 VEs can thus incorporate important attributes linked to maximizing poststroke motor recovery and learning. 13 Authors concur that using virtual reality for stroke rehabilitation is feasible, 14 but the effectiveness 15 and added value of using this over other interventions need to be documented using rigorous research designs such as randomized control trials (RCTs).
Studies have compared virtual reality intervention effectiveness to no 12 or conventional treatment. 5 However, because exercise intensity is a critical factor in motor recovery,16,17 it needs to be controlled for when comparing treatment effectiveness between different training environments. We addressed the question of whether and to what extent intensity-matched training in a VE results in similar or better outcomes than training in a physical environment (PE). This was addressed by creating a VE that was equivalent to a PE in terms of type of practice, practice intensity, and feedback. Training environments only differed in that the VE had an additional visual effect (target size increased temporarily to indicate a successful trial), encouraged movement in a functional context (shopping task), and included a game score for monitoring success. We investigated the effectiveness of these additional VE attributes on upper-limb recovery in chronic stroke. We hypothesized that upper-limb impairment, activity, and use would improve more after VE compared with PE training. Preliminary results have appeared in abstract form. 18
Methods
Participants were included in this double-blind RCT if they (1) were between 40 and 80 years, (2) sustained a single ischemic or hemorrhagic stroke 6 to 60 months previously, (3) scored 3 to 6/7 on the Chedoke-McMaster Stroke Assessment 19 arm subscale, and (4) had no other neurologic or neuromuscular/orthopedic problems affecting the upper limb and trunk. Individuals were excluded if they had (1) brainstem/cerebellar lesions, (2) comprehension difficulties, and (3) marked apraxia, attention, or visual field deficits. Participants signed informed consent forms approved by the institutional review board of the Center for Interdisciplinary Research in Rehabilitation of Greater Montréal.
Participants were stratified at baseline by severity (Fugl-Meyer Stroke Assessment Upper Limb [FMA] score ±5 points) 20 and age (±5 years) and block randomized to VE or PE training groups. Randomization, clinical evaluations, training, and data analysis were done by different individuals uninvolved with and blinded to other study aspects.
Intervention
Participants in both groups (PE, VE) repetitively pointed toward 6 targets placed just beyond arm’s length, without physically touching the target in either environment. Earlier work by Cirstea et al 21 showed that reaching training-based improvement in movement variables (eg, error) led to an asymptotic level only after 30 to 35 repetitions, depending on arm motor impairment level. This number was doubled to ensure high practice intensity. The total number—72 trials—was divided into 3 blocks of 24, with 5-minute interblock intervals. A prerecorded verbal randomized target sequence was played back on the computer, instructing participants to point to the next target. Each training session averaged 45 minutes in both environments. The acquisition phase was 12 days spaced over 4 weeks (3 times/wk).
For both environments, participants sat with the back supported and hips and knees flexed to 90°, shoulder abducted (20°) and internally rotated, elbow slightly flexed, forearm pronated, and wrist in neutral. Prior to movement, the index finger was placed ipsilaterally on a 41.5-cm high platform, 10 cm lateral to the hip (starting position, Figure 1A). This position was chosen to encourage full-range shoulder and elbow movement during pointing. A research assistant seated behind the participant ensured that the arm starting position was similar before each trial during interventions and kinematic assessments. Participants were instructed to point as fast and as accurately as possible.
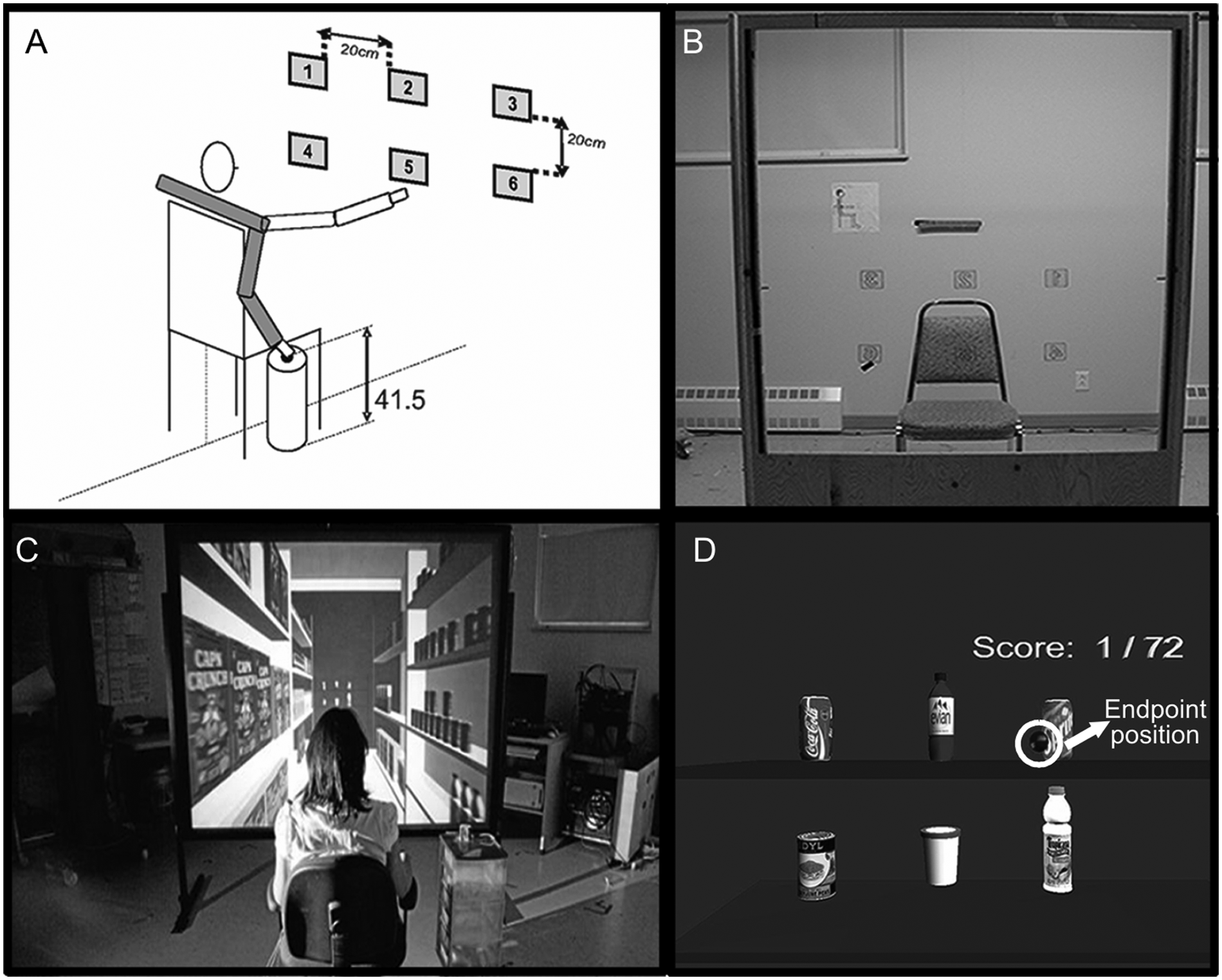
Experimental set-up: A. Participant’s starting position in relation to the targets; target 5 was the lower middle, and target 3 was the upper ipsilateral target for right arm movements. B. Physical training environment with 6 numbered targets. Initial (C) and final (D) virtual training environment—3D scene of 6 consumer products on a supermarket shelf projected on a large screen.
Physical Environment
In the PE, 6 square (6 × 6 cm2) numbered targets were mounted on a wooden frame and arranged in a 2-row by 3-column grid numbered 1 to 6 (Figure 1B), which was placed just beyond arm’s length (measured from the medial axilla to index fingertip) for each participant. This ensured that there was no contact between the finger and the target (no haptic feedback). Target collision was detected by the Computer Assisted Rehabilitation Environment (CAREN) software (Motek BV, Amsterdam) when the index fingertip approached a critical distance from the target center, defined as ±4 cm3 for the initial condition (but see progression below). Grid midpoint, the center of the CAREN-Optotrak global axis system, was aligned with the sternum at a height of 90° shoulder flexion. Targets were located in middle (T2-upper, T5-lower), ipsilateral (T3-upper,T6-lower), and contralateral (T1-upper,T4-lower) arm work spaces separated by a 20-cm edge-to-edge distance (Figure 1A). During practice, arm and trunk positions were tracked using electromagnetic sensors (Fastrak, Colchester, Vermont) on the fingertip, ipsilateral acromion, and midsternum. Positional data were used to generate feedback about movement precision, speed, and trunk displacement by the CAREN software.
Feedback was provided to facilitate motor learning. It consisted of terminal (end-of-movement) auditory plus visual KR about movement precision and speed, and concurrent auditory KP about compensatory trunk use. Positive KR (“ping” sound) was delivered if movement met accuracy and speed criteria without excessive trunk displacement (≤5 cm). 22 A buzzer sounded (negative KR) if participants were unable to point to the correct target within the allotted time. If trunk movement exceeded 5 cm, a “whoosh” sound was heard (KP) regardless of whether movement was within accuracy and speed constraints. During practice, individual accuracy and speed criteria were progressed from low to high (accuracy, 4 to 1 cm3 from target center; speed, 6-1 s) when participants were successful ≥75% of the time.
Virtual Environment
A 3D VE (CAREN) simulated a supermarket scene. To provide a context for the shopping task, the VE scene initially depicted a supermarket aisle filled with products viewed from one end (Figure 1C). An animation sequence then simulated the participant moving through the aisle. At the end of the aisle, the scene stopped moving and participants saw 6 consumer products—2 soda cans, water, juice, ice cream, and peas—arranged on 2 shelves (Figure 1D). The objects on the shelves were placed at the same height in the visual field as the PE targets to avoid differences in distance perception. 23 To further improve depth perception, the products had the same shape and size dimensions with respect to the participant’s actual hand as in the real world. 24
The environment was calibrated such that the VE objects appeared to be at the same distance as the PE targets—that is, just beyond arm’s length. The virtual target grid and the hand, arm, and trunk positions of the participants were calibrated in the same CAREN-Optotrak global axis system. As in the PE, the center of the virtual grid was placed just beyond arm’s length, and target collision detection was extended in the
Assessments
Clinical and kinematic assessments were done before (PRE), immediately after (POST), and at retention testing (RET), 3 months after the intervention, to evaluate changes at 2 levels of the International Classification of Functioning: Structure/Function and Activity. 25 The primary outcome was change in arm motor impairment measured clinically and kinematically, and secondary outcomes were changes in activity levels. Motivation was assessed at POST only. All assessments were reliable and valid.20,22,26-33
Clinical Assessments
Motor impairment was measured with the FMA and Reaching Performance Scale for Stroke (RPSS). The arm section of FMA20,26 (0-66 points) assesses movement made in and out of synergy patterns, coordination, speed, and reflexes. Participants were classified into 2 motor severity groups: mild (FMA ≥ 50 points; PE, n = 5; VE, n = 7) and moderate-to-severe (FMA ≤ 49 points; PE, n = 11; VE, n = 9).22,27 The RPSS 28 evaluates reach-to-grasp performance for objects placed within (RPSSclose) and beyond (RPSSfar) arm length. A total of 6 movement components—trunk, shoulder and elbow movement, end point smoothness, prehension quality—are scored on 4-point scales (18 points/task). Arm motor activity was evaluated with the Wolf Motor Function Test—Functional Assessment Scale (WMFT-FAS)29,30 as the mean value of 15 tasks scored on 6-point scales. This scale was chosen over the timed portion of the WMFT because it takes movement quality into account, such that tasks performed with abnormal movement patterns are scored lower. Arm use was assessed through a structured interview with the Motor Activity Log Amount Scale (MAL-AS)31,32 mean scores.
The MAL-AS measures how much the more-impaired arm was used in 30 tasks over the previous week. Each item was scored on a 6-point scale where 0 =
Kinematic Data Acquisition and Analysis
Kinematic testing was done in separate sessions in a physical set-up. Arm and trunk kinematics were recorded from 25 pointing trials each toward the lower-middle (LM) target (T5) and upper-ipsilateral (UI) target (T1 or T3 depending on the more affected side; Figure 1A). Target positions required different combinations of joint excursions to reach into different parts of the workspace. Elbow extension was combined with a greater range of shoulder horizontal adduction (ShHor) for LM and with a greater range of shoulder flexion for UI, although reaches to both targets required all 3 movements. Outcomes were measured at 2 levels: motor performance (end point velocity, precision) and movement pattern (joint angular excursions, trunk displacement). 4 Movements were recorded with Optotrak 3020 (Northern Digital Corp, 100 Hz, 6 s, Waterloo, Canada), and 6 infrared-emitting diodes (IREDs) were placed on the fingertip (end point), wrist styloid, lateral epicondyle, ipsilateral and contralateral acromions, and midsternum.
For motor performance outcomes, end point tangential velocity was computed from the velocity vector amplitude obtained from the finger marker. Movement beginning and end were defined as times at which the velocity exceeded and remained above or fell and remained below 10% of the peak velocity. Precision was computed as the root mean square error of the absolute distance between the final end point position and target center.
For movement pattern outcomes, 3 joint angles were computed at the end of the movement—elbow extension, ShHor, and shoulder flexion based on vectors formed by IRED pairs. For elbow movement, vectors were defined between wrist and elbow and elbow and ipsilateral shoulder IREDs (full extension = 180°). ShHor was measured as the angle between vectors formed by ipsilateral shoulder-elbow IREDs and a line traced by the contralateral-ipsilateral shoulder IREDs projected horizontally (full adduction = 0°). For shoulder flexion, vectors were defined between elbow and ipsilateral shoulder IREDs and the vertical through the ipsilateral shoulder IRED (arm alongside body = 0°). Trunk movement was measured as forward sagittal displacement (mm) of the sternal IRED.
Statistical Analysis
Descriptive statistics highlighted main demographic characteristics and motivation levels. Distributions were inspected to verify normality assumptions and to identify potential outliers and influential observations. Homogeneity of variance assumptions were assessed with Levene tests. Demographic and initial characteristics were compared using independent
Results
A total of 32 participants completed all PRE and POST assessments and 25 (PE, 13; VE, 12) completed RET (see CONSORT statement available online as Supplemental Material; nnr.sagepub.com). Participants in both environments were similar in age, sex, side affected, duration poststroke, type of stroke, clinical scores (Table 1), and initial kinematic outcomes. All participants reported being comfortable throughout the training and did not experience any side effects. There were overall effects of time (Figure 2), training environment (Figure 3), and severity group (Figure 4).
Demographic and Clinical Characteristics of Study Participants at Baseline a
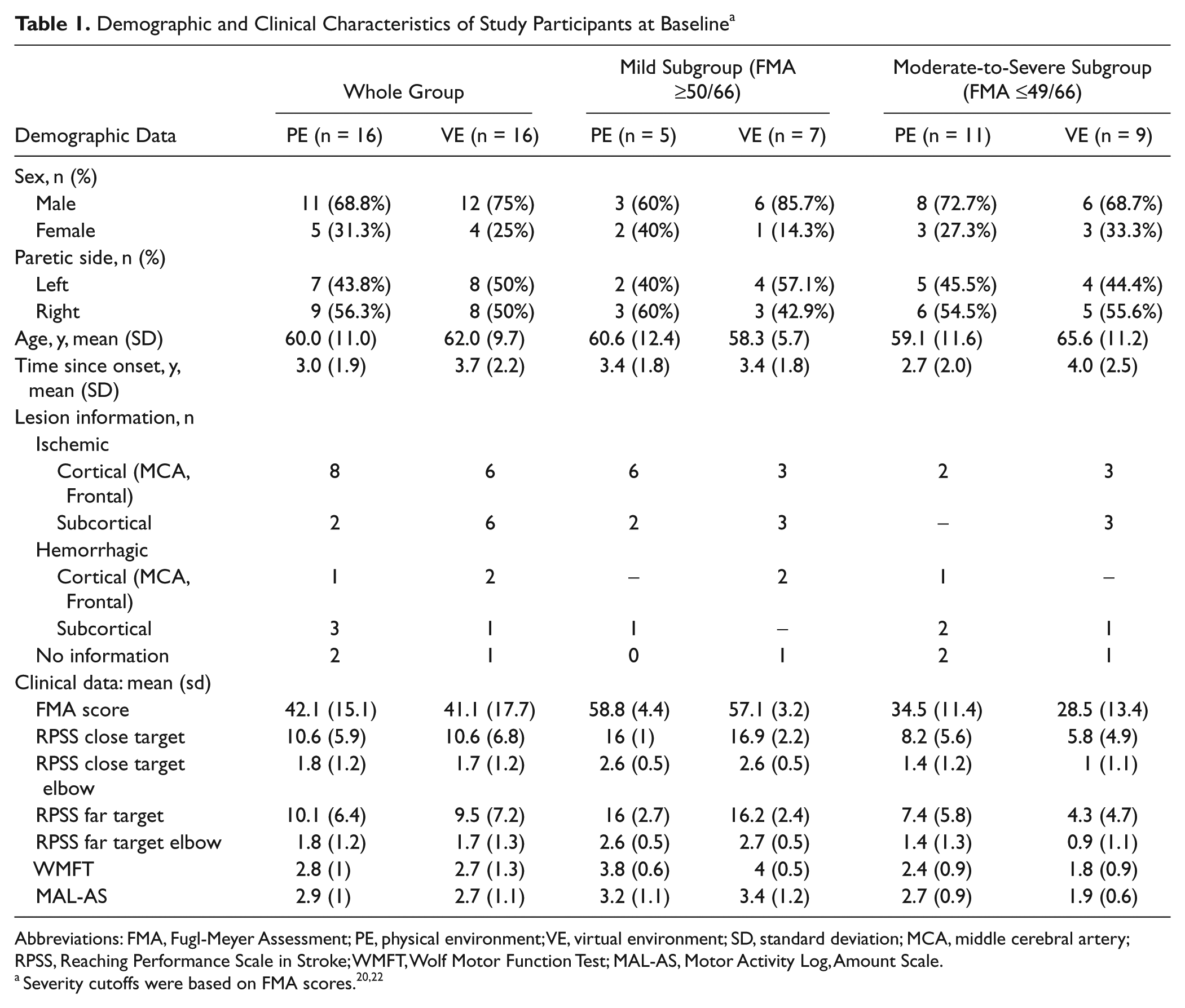
Abbreviations: FMA, Fugl-Meyer Assessment; PE, physical environment; VE, virtual environment; SD, standard deviation; MCA, middle cerebral artery; RPSS, Reaching Performance Scale in Stroke; WMFT, Wolf Motor Function Test; MAL-AS, Motor Activity Log, Amount Scale.
Severity cutoffs were based on FMA scores. 20,22
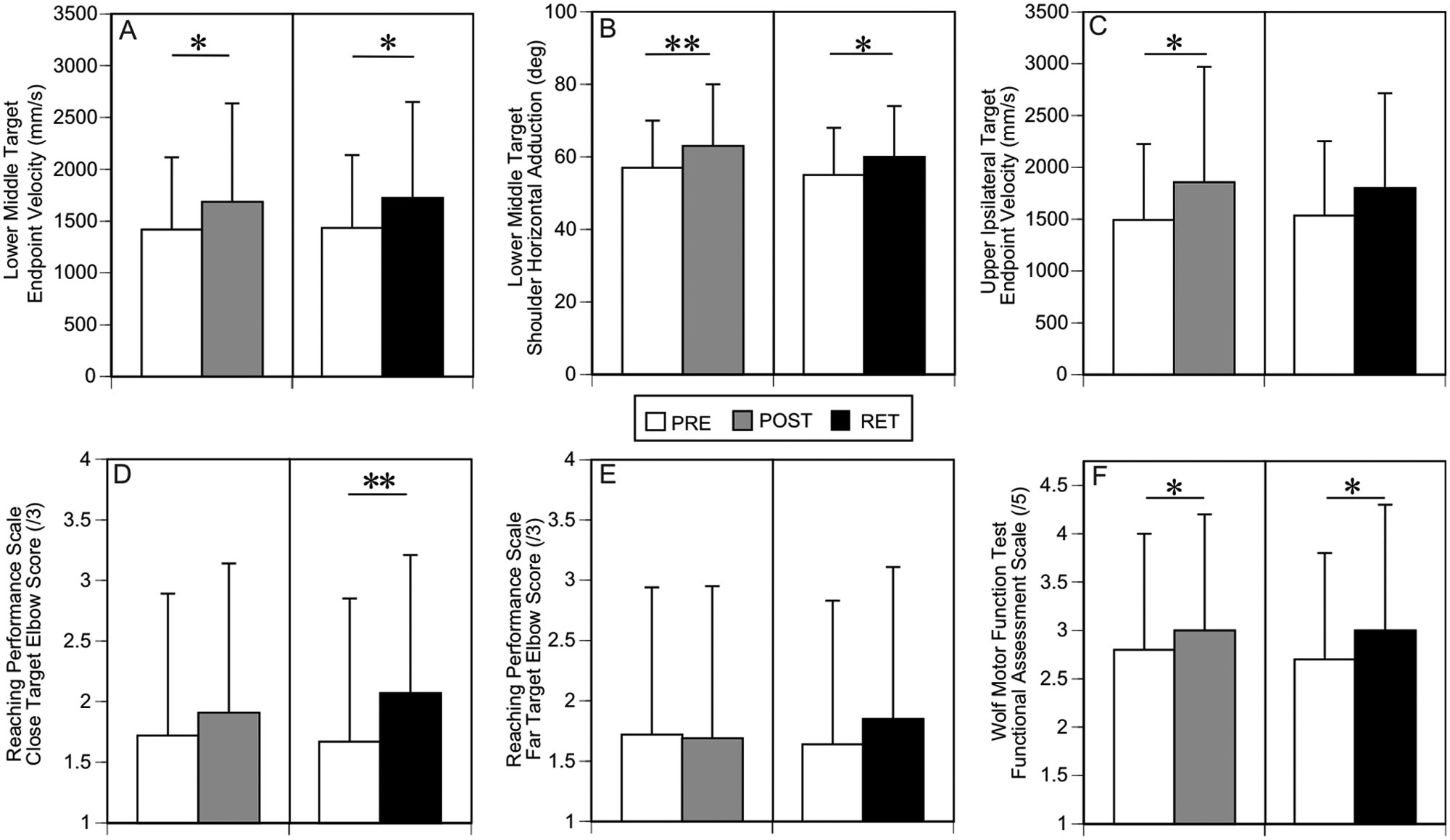
Effect of time on outcomes: Post hoc test results for the overall effects of time from PRE to POST and PRE to RET for (A) end point velocity and (B) shoulder horizontal adduction for lower-middle target, (C) end point velocity for upper-ipsilateral target, (D, E) Reaching Performance Scale for stroke elbow subscale scores for close (D) and far (E) targets, and Wolf Motor Function Test Functional Assessment Scale mean scores (F). Data are overall mean (SD) values across both groups and training environments. Asterisks indicate significance: *
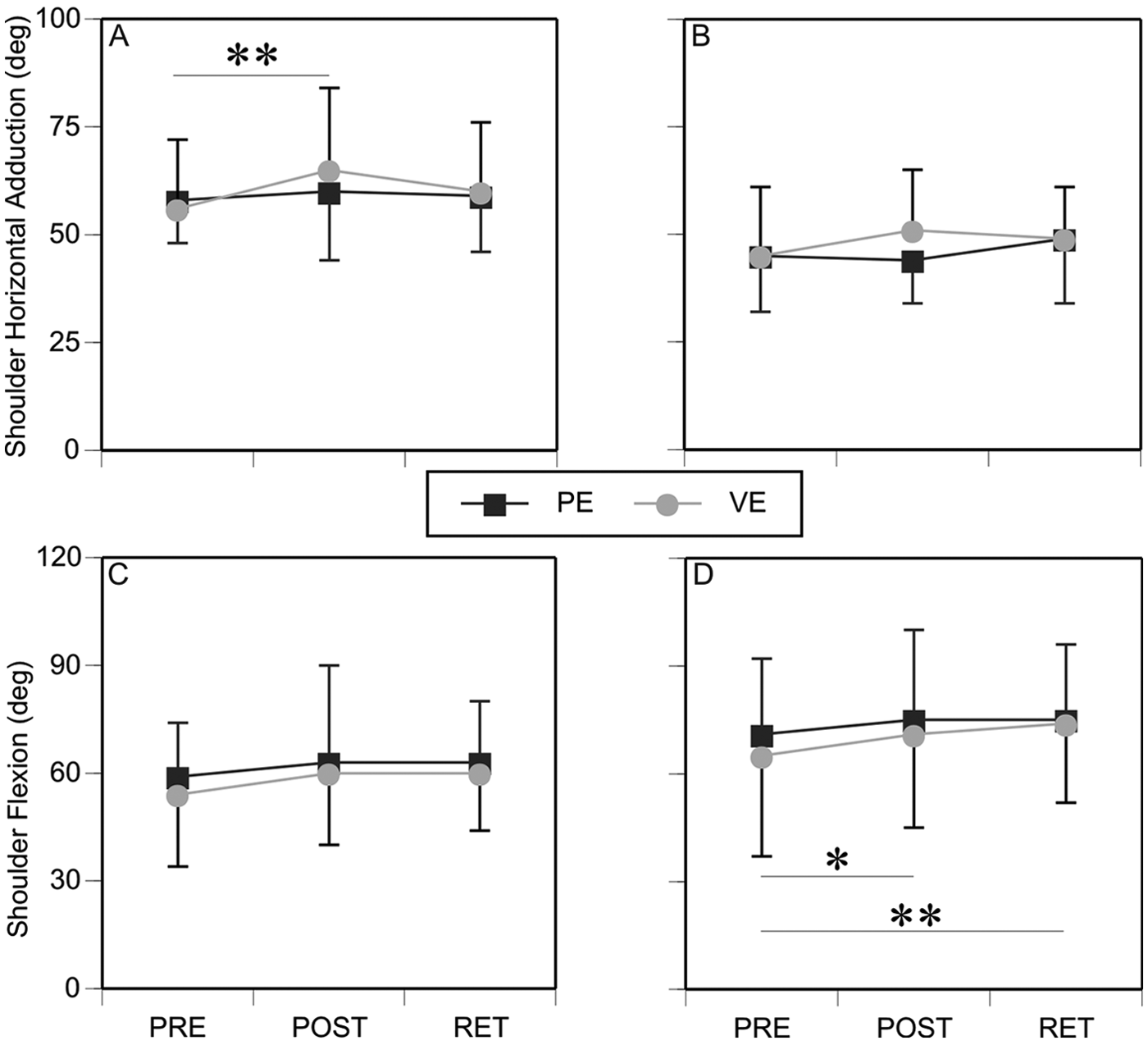
Effects of training environment on outcomes: Mean (SD) values for shoulder horizontal adduction and shoulder flexion for lower-middle target (A, C) and upper-ipsilateral target (B, D) for the 2 groups training either in PE or VE at PRE, POST, and RET. Asterisks indicate significance: all significance refers only to the VE; *
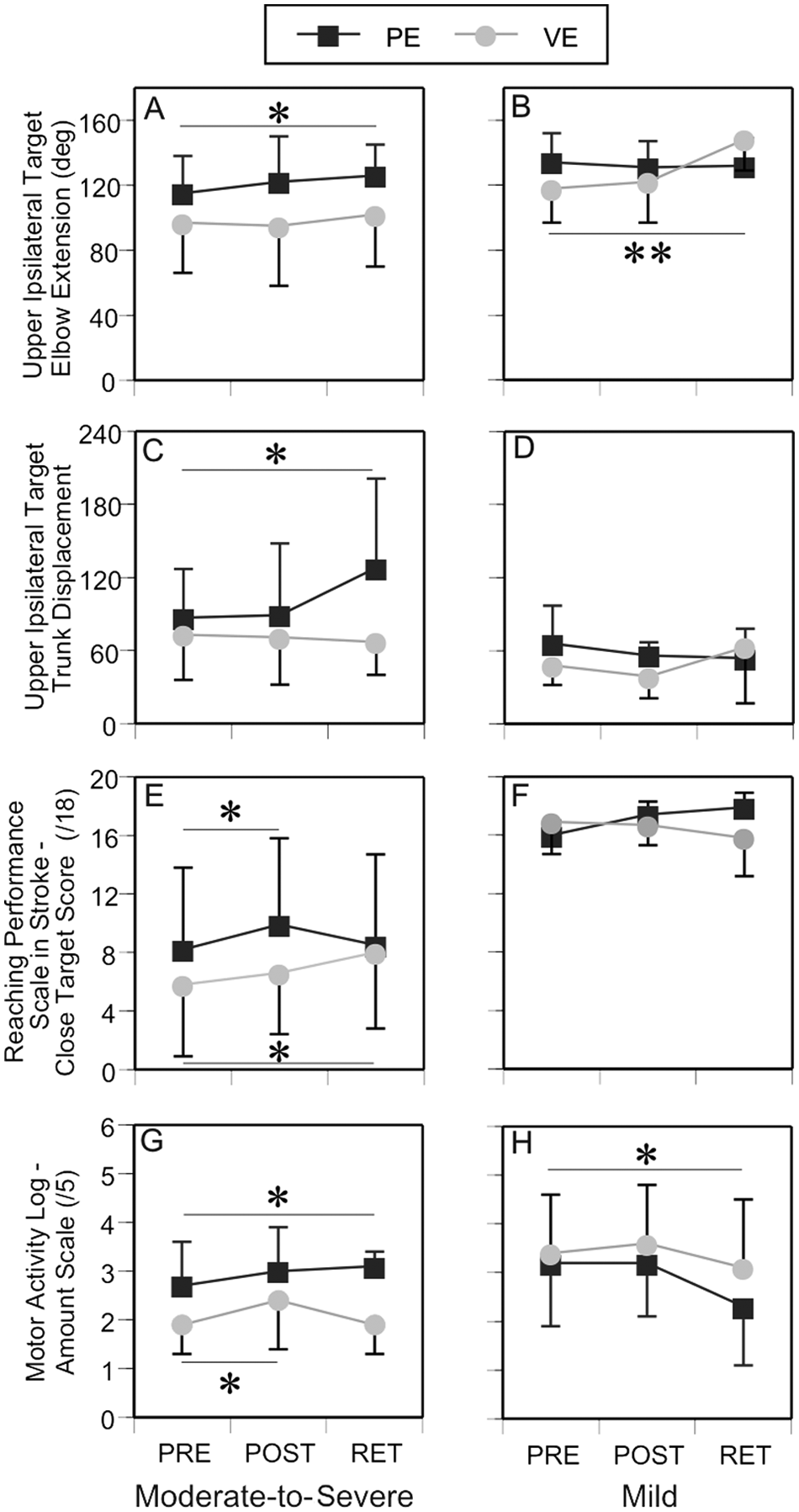
Effects of impairment severity on outcomes: Mean (SD) values for elbow extension (A, B) and trunk displacement (C, D) for pointing movements to upper-ipsilateral target, Reaching Performance Scale for Stroke—close target total scores (E, F) and Motor Activity Log—Amount Scale (G, H) scores for the moderate-to-severe and mild subgroups training either in PE or VE at PRE, POST, and RET. Asterisks indicate significance. Significance for the PE is indicated above the symbol and for VE below the symbol: *
Effect of Time
Overall, participants increased endpoint velocity (
For clinical outcomes, participants increased elbow extension during a reach-to-grasp task (RPSSelbow close target:
Effect of Training Environment and Environment by Time
Although there was no overall effect of environment, Environment × Time interactions were found for 2 kinematic variables. ShHor (
Effect of Impairment Severity
Among kinematic variables, elbow extension (
For clinical measures, performance on the reach-to-grasp task (RPSSclose total,
In the mild group, whereas no changes were noted in VE, those training in PE decreased MAL-AS sores at RET by 0.9 points (
Motivation Questionnaire Results
Intrinsic Motivation Task Evaluation Questionnaire scores indicated that participants enjoyed training in the 2 environments equally and considered the experiences useful and important. Participants felt more competent practicing movements in PE compared with VE (χ2 = 6.77,
Discussion
Effects of practice in VE compared with PE on arm motor impairment and activity levels were addressed. Training environments were matched by practice intensity, frequency, and feedback. They only differed in that the VE provided a more colorful interface, additional visual feedback about target acquisition, and a game score. Both groups improved arm motor impairment measures (increased end point velocity: both targets; ShHor: LM target only) and clinical impairment scores (RPSSelbow subscales) and activity levels (WMFT-FAS).
Improvements in both groups over time can be attributed to practice intensity. Repetition intensity (72 trials/session) was greater than twice that used (32) in typical stroke rehabilitation sessions. 36 Studies have shown better motor outcomes with more intensive practice regardless of the treatment medium (e.g., robotics versus conventional therapy17). Potential mechanisms of better outcomes with intensive training include higher levels of neurotrophic factors like brain derived neurotrophic factor (BDNF) 37 known to contribute to motor learning after stroke.
The number of trials (n = 72) was considered intensive based on findings that learning became asymptotic after a larger number of trials (average 36) when compared with controls performing the same task (average 15). 21 Generally, motor learning studies can include as many as 200 to 800 repetitions. A recent study 38 showed that those with stroke could perform an average of 332 repetitions of tasks involving graded movements of reaching and grasping, manipulating and releasing objects in a 1-hour session without fatigue. However, as in our previous study, although participants showed behavioral changes, concomitant neuronal plasticity changes were not assessed. However, our results allow us to conclude that at least 72 repetitions per session may be necessary for the nervous system to find motor solutions based on redundancy,39,40 leading to improvements in movement patterns at the behavioral level.
Because the effect of practice intensity has been well documented, we intentionally controlled for this factor as well as for the type and frequency of feedback. Our goal was to identify whether the additional attributes of VE (motivation, interactivity) offered any advantage to motor learning in stroke. Motivation and interactivity of the VE were enhanced by the added visual effects and game score that enabled participants to track success. Despite both groups having the same practice intensity and feedback, the VE group had better target-specific changes in ranges of ShHor for LM and shoulder flexion for UI targets. These changes resulted in successful pointing to LM and UI targets, respectively, because of their placement in the arm work space. Similar results were reported by Mirelman et al 41 for the lower limb in a single-blind RCT. Compared with ankle training using a robotic device, dose-matched practice on a robotic device coupled with a VE resulted in greater improvements in gait velocity and walking distance (on the 6-minute walk test) and number of steps in the community (measured using accelerometers). Changes were retained at follow-up. Combined with our findings, this provides encouraging evidence of the advantage of using VEs in upper- and lower-limb rehabilitation poststroke.
Recent systematic reviews stress the importance of providing salient feedback about arm movement quality for improving motor learning42,43 and motivation. We provided feedback about both motor performance (KR—velocity, precision) and movement patterns (KP—trunk displacement). Both groups used KR similarly as indicated by the overall increase in velocity (Figures 3A and 3C). However, KP may have been used differently in each environment. In VE, KP resulted in increased elbow extension without compensatory trunk movement in the mild subgroup. Conversely, in PE, an improvement in elbow extension was accompanied by increased trunk displacement in the moderate-to-severe group, considered maladaptive or compensatory. 44 This suggests that VE may be a more effective medium for providing feedback, especially trunk movement KP.
Participants may have been more aware in VE that successful task performance depended on the movement pattern used. This awareness may have engaged them to use more cognitive effort 45 and better motor planning to increase the essential ranges of arm motion required for successful pointing. Indeed, previous research using VEs in those with chronic stroke 46 has reported better performance in the trail-making test, an assessment of task switching and visual attention. The lower stress levels felt by VE participants may have added to their enjoyment and ability to concentrate on the movement patterns used. In contrast, the higher levels of perceived competence of the PE group may have led this group to believe that they were already proficient at performing the task, so that they placed less importance on using more optimal movement patterns.
Task practice improved performance of everyday life activities in both environments. However, the improvement in the MAL-AS reached a meaningful clinically significant level (change of 0.5 32 ) only in the moderate-to-severe subgroup training in the VE.
The lack of change in clinical outcomes in the mild group may partly be attributable to ceiling effects of the measures used. Clinical measures may also not be sensitive enough to detect deficits identified using kinematic analysis, especially in those with mild hemiparesis. In a previous study, changes in upper-limb movement patterns of 33% to 300% were not reflected in Fugl-Meyer scores in a group of patients with mild poststroke hemiparesis (FMA ≥ 50). 22 However, this will have to be examined separately for each outcome measure.
Limitations
Although we found significant between-group differences, these were small for most outcomes. One reason could be that the VE, though ecologically valid, was especially designed to differ from the PE only in terms of the feedback delivery medium. This was intentionally done to isolate and compare only the effects of the feedback delivery medium on changes in arm motor impairment and activity levels. The differences found between environments in some aspects of motivation cannot be attributed specifically to different parts of the VE (ie, opening scene animation vs interactive pointing scene). Although we found changes on the RPSS total score and elbow subscale, the absence of an established minimal detectable change or the minimum clinically important difference value limits a broader interpretation of these results. Apart from a few items on the MAL, we did not assess participation or quality of life. Inclusion of these measures in future studies will provide more information about the effects of VE training on these domains.
Implications for Neurorehabilitation
VEs that are custom designed according to the needs of the individual have the potential to increase patient engagement by making therapy more fun and interesting. At the same time, such applications allow clinicians to adapt difficulty levels and activities according to patient preferences and rehabilitation goals. Our results suggest that there is additional value in using a VE as a training environment to enhance arm motor recovery, especially in the chronic poststroke stage. This supports the use of VE as a means to increase the amount of rehabilitation-specific exercise time—specifically, for targeted upper-limb tasks.
Conclusion
Dose-matched practice in VE resulted in arm motor recovery at the behavioral level 47 compared with PE in chronic poststroke patients. This may be attributable to more efficient use of feedback in VE. Our results corroborate earlier findings that even patients with mild upper-limb impairment have motor deficits that are not always identifiable using common clinical measures22,44 and that they have the potential for further motor recovery (eg, elbow extension range) if they are given appropriate task practice and feedback.
Footnotes
Acknowledgements
We would like to acknowledge the individuals who participated in our study and thank Ruth Dannenbaum, Rhona Guberek, and Christian Beaudoin for their assistance with patient recruitment and technical support.
Authors’ Note
Clinical trial registration number: ACTRN12611000858998.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Heart and Stroke Foundation of Canada (HSFC) and in part by the Physiotherapy Foundation of Canada. MFL holds a Canada Research Chair in Motor Recovery and Rehabilitation. HS is a career scientist with Ministry of Long Term Health and Care, Ontario. SKS is supported by Focus on Stroke Doctoral Research award (Canadian Institutes of Health Research, HSFC, Canadian Stroke Network). CBL is supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil.
