Abstract
Background. Repetitive facilitative exercise (RFE), a combination of high repetition rate and neurofacilitation, is a recently developed approach to the rehabilitation of stroke-related limb impairment. Preliminary investigations have been encouraging, but a randomized controlled evaluation has yet to be performed. Objectives. To compare the efficacy of RFE with that of conventional rehabilitation in adults with subacute stroke. Methods. A total of 52 adults with stroke-related upper-limb impairment (Brunnstrom stage ≥III) of 3 to 13 weeks’ duration participated in this randomized, controlled, observer-blinded trial. Participants were randomized into 2 groups and received treatment on a 4-week, 40 min/d, 5 d/wk schedule. Those assigned to RFE received 100 standardized movements of at least 5 joints of their affected upper extremity, whereas those in the control group participated in a conventional upper-extremity rehabilitation program. Primary and secondary outcomes (improvement in group Action Research Arm Test [ARAT] and Fugl-Meyer Arm [FMA] scores, respectively) were assessed at the end of training. Results. In all, 49 participants (26 receiving RFE) completed the trial. ARAT and FMA scores at baseline were 19 ± 21 and 39 ± 21 (mean ± standard deviation). Evaluation at the trial’s completion revealed significantly larger improvements in the RFE group than in the control group in both ARAT (F = 7.52; P = .009) and FMA (F = 5.98; P = .019) scores. Conclusions. These findings suggest that RFE may be more effective than conventional rehabilitation in lessening impairment and improving upper-limb motor function during the subacute phase of stroke.
Introduction
Stroke is a leading cause of disability and leaves about two-thirds of its survivors with significant long-term impairments. 1 Its economic, individual, and societal costs are huge, with monetary costs having been estimated to amount to as much as 69 billion dollars per year in the United States alone. 2 A number of approaches have been developed over the years to improve rehabilitation of limb function following stroke. Unfortunately, the benefits of all approaches, particularly in the upper extremity, are far more limited than we would like.3-5
A number of issues appear central to the success of a rehabilitation program. Prominent among these is the intensity of a program, with a number of recent systematic reviews demonstrating that increases in either the time devoted to or number of repetitions involved in an intervention are associated with improved functional outcome.1,6-8 The benefits of a high intensity program are well established, and although no clear guidelines exist regarding the best levels of intensity in practice, the need for its incorporation into a therapy program is widely accepted.9,10
The nature of the therapy delivered also appears to be important: repetitive task-oriented training may be particularly promising. 8 Nevertheless, although task-related training appears to be beneficial, its performance in a manner that permits a high rate of repetition can be difficult. 6 As a result, the nature of an optimal therapy program remains elusive. In fact, approaches as varied as constrained use,11,12 robotic assistance,13,14 bilateral arm training,15,16 mirror therapy,17,18 strength training,19,20 and task-related/virtual reality techniques21,22 have all been shown to be capable of improving motor function following stroke.1,10 Thus, while there is strong support for the benefits of task- and context-specific approaches,1,10 it is clear that intensity is important regardless of the nature of the training.
These factors provoke the idea that a new way of marrying a high level of intensity with other training techniques may lead to a more effective approach to stroke rehabilitation. One possibility that intrigued us was the combination of high repetition rate with neurofacilitation techniques, because the latter not only lend themselves to repetition, but it has also been suggested that the repetition may be effective in lessening synergistic movement patterns.23,24 We, therefore, developed a repetitive facilitative exercise (RFE) approach that, when applied to the upper extremities, involves repetitive training of stereotyped flexion and extension movements in combination with a modification of conventional neurofacilitation approaches. 24 More specifically, the therapist harnesses muscle spindle stretch and skin-generated reflexes to assist the patient’s efforts to move an affected joint.
Our preliminary work demonstrated that this approach may improve the Brunnstrom stage 25 of the affected upper limb—and thereby motor control and joint movement—more effectively than conventional rehabilitation approaches. 24 However, the magnitude of RFE’s benefits relative to conventional therapy, particularly with respect to more functional measures, such as the ability to manipulate objects, remains unknown.
Given this, we conducted a randomized, controlled, blinded trial to test the hypothesis that a 4-week, 5 d/wk RFE regimen is more effective than a similarly intense conventional rehabilitation program in the rehabilitation of stroke-related upper-extremity impairment. The study’s primary and secondary outcomes were, respectively, improvement in motor function as measured by the Action Research Arm Test (ARAT) 26 and improvement in motor control as measured by the Fugl-Meyer Arm Motor Scale (FMA). 27
Methods
Design
This 4-week, 2-center, randomized, observer-blinded trial involved a group of participants with subacute stroke who were randomized to either an RFE or a conventional rehabilitation program (see Figure 1). Randomization was accomplished in a centralized manner using a computer-generated random number that did not take a participant’s location into account. Evaluators were blinded to a participant’s treatment assignment, and participants were unaware of the study’s hypotheses. Outcome measures were assessed at the trial’s initiation, midpoint, and 4-week conclusion.
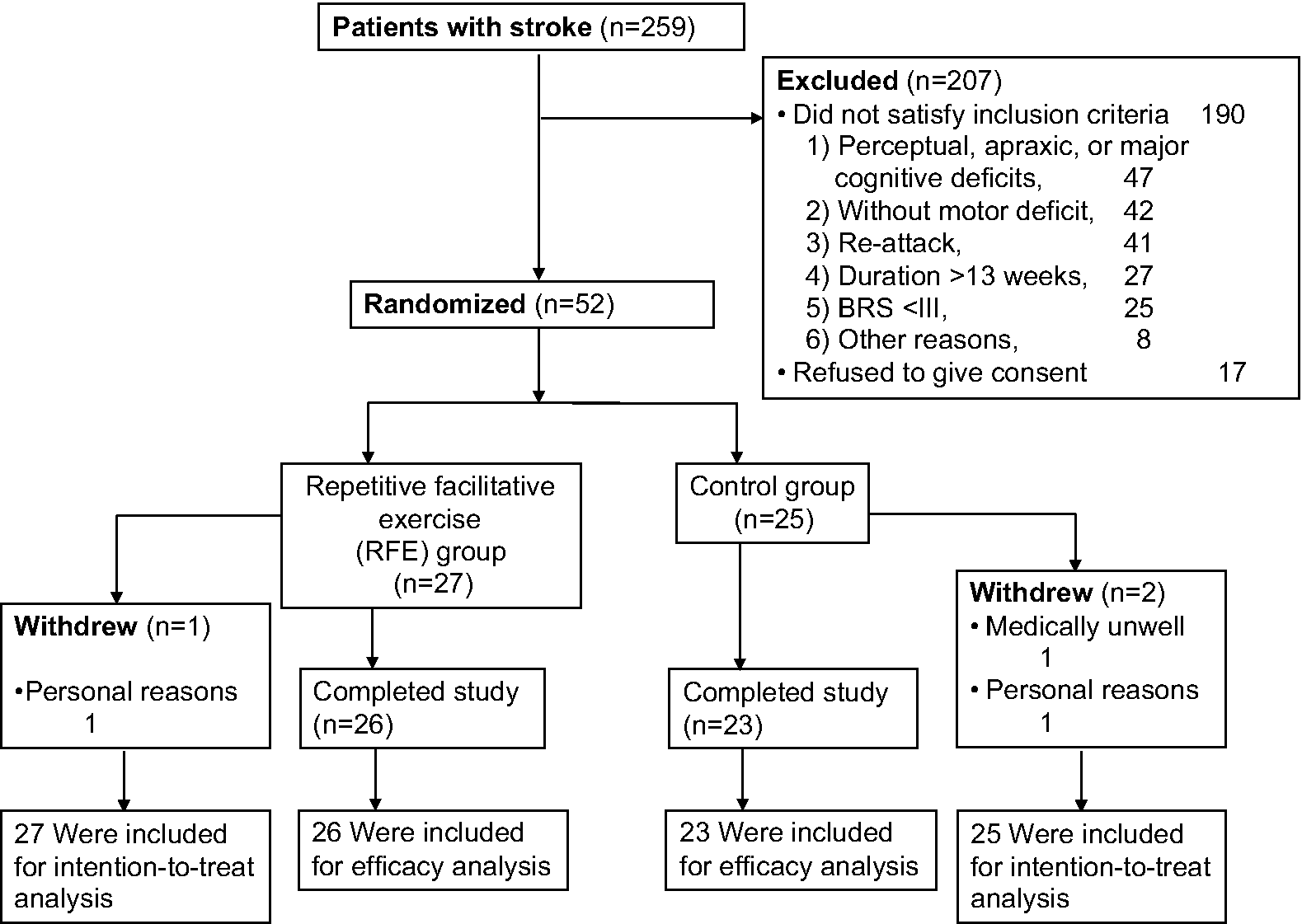
Study recruitment and randomization. Abbreviations: BRS, Brunnstrom stage of the proximal upper-limb; Duration, elapsed time following stroke.
Participants
Participants were recruited between October 2008 and March 2010 from 2 Japanese inpatient rehabilitation centers. Inclusion criteria were as follows: (1) a new, single, computed tomography–confirmed stroke of 3 to 13 weeks’ duration; (2) Brunnstrom proximal upper-limb stage ≥III 25 ; and (3) the ability to follow simple directions. Exclusion criteria were the following: (1) arm contractures/ pain; (2) preexisting upper-extremity impairment; (3) cerebellar lesion; (4) unstable medical status; (5) perceptual, apraxic, or cognitive deficits that would prevent adequate study participation; or (6) inability to provide informed consent. The study was approved by the ethics committees of the study sites and was registered in the Kagoshima University Clinical Trial Registry (20-114). Written informed consent was obtained from each patient before inclusion.
Intervention
All participants, regardless of their group assignment, underwent training in 40-minute sessions on a 4-week, 5-d/wk schedule. In addition, all performed 30 min/d of dexterity-related training—that is, object-related reach-to-grasp training—immediately after each treatment session. They also continued their participation in a standard inpatient rehabilitation program that included activities of daily living, physical therapy, mobility training, and speech therapy.
Rigorous efforts were made to maintain trial purity and blinding. In particular, treatment was limited to a group of 11 skilled occupational (10) and physical (1) therapists who were restricted to provide treatment to participants in 1 of the 2 groups. Additional measures taken to maintain training purity included the following: (1) therapists confirming their appropriate assignment to RFE or conventional therapy before the assigned start of each session, (2) minimalization of treatment discrepancies with intensive training and strict adherence to a detailed treatment manual, and (3) maintenance of fixed therapist/patient assignments throughout the study.
RFE group
The overall aspects of the RFE program have been described previously24,28,29 and are, therefore, only summarized here. Briefly, repetitive facilitative techniques were used to elicit movement of the shoulder, elbow, wrist, and fingers in a manner designed to minimize synergistic movements. Treatment involved rapid passive stretching of the muscles of the targeted joints in conjunction with tapping and rubbing the skin to assist in the generation of a contraction. Participants were directed to concentrate on generating movement on the joint being treated while avoiding contraction of nontargeted muscles. Therapists provided verbal directions with commands such as “bend/straighten” or “one, two, three” (Figure 2). Participant efforts were supplemented as necessary to achieve a full range of motion (ROM).
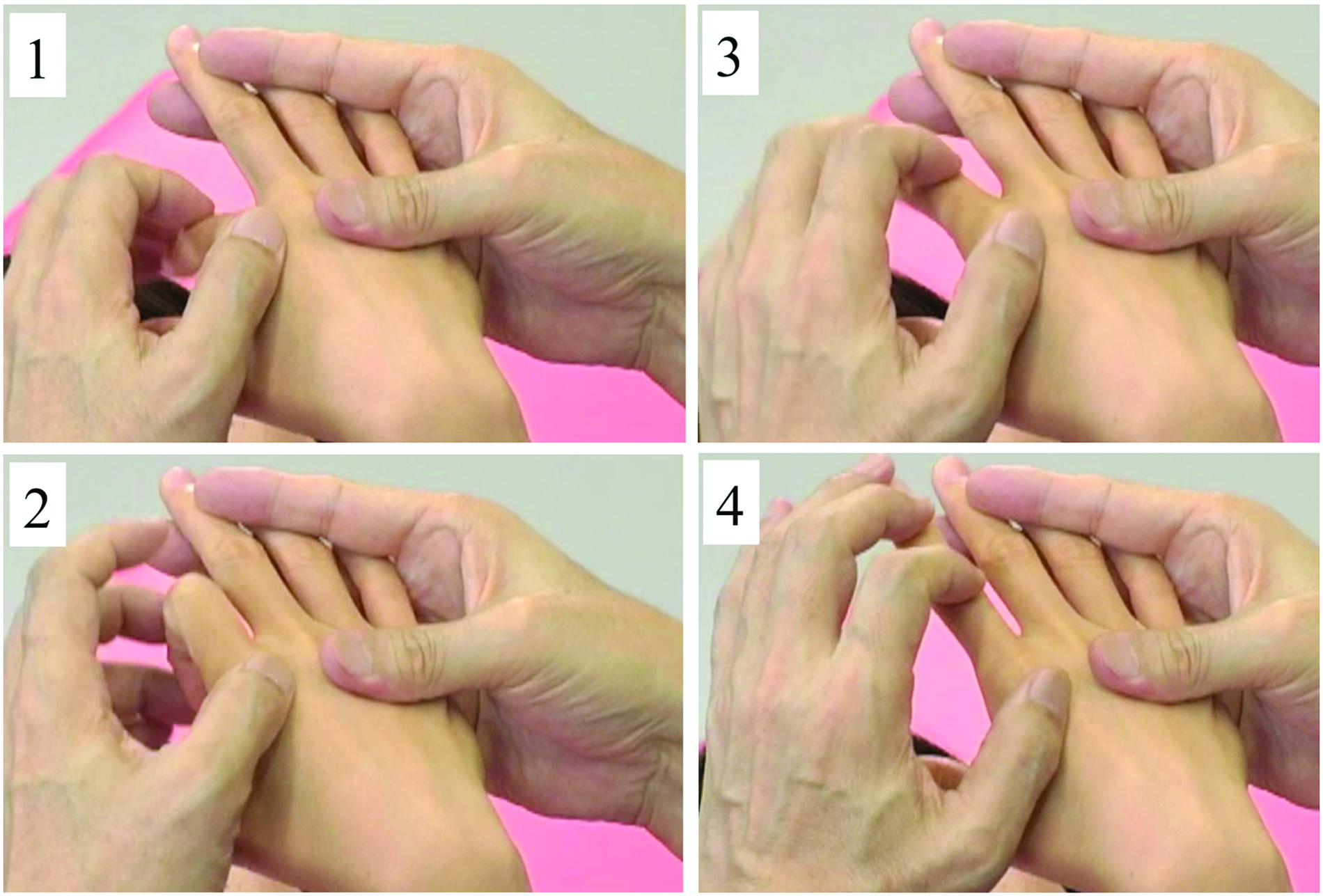
An example of repetitive facilitative exercise (RFE) to elicit a right second finger extension: (1) The patient’s wrist is placed in flexion while the patient is in the supine position. To induce extension of only the second finger, the therapist holds the third, fourth, and fifth fingers motionless; (2) the second finger is quickly flexed by the therapist immediately before instructing the patient to extend it; (3) the therapist’s index finger then pushes the proximal phalanx forward to facilitate the finger’s extension; (4) during extension, the therapist maintains his or her fourth fingertip loosely on the patient’s fingernail to assist the paretic finger in obtaining a full range of motion. This pattern is repeated in 2 sets of 50 repetitions separated by a 1- to 2-minute rest period.
A total of 8 specific patterns were used: (1) shoulder flexion with the elbow bent at 90° in the supine position; (2) shoulder horizontal extension/flexion in the supine position with the elbow ranging in flexion from about 70° to 110°; (3) shoulder flexion/adduction/external rotation with flexion of the elbow and forearm supination accompanied by wrist flexion, finger flexion, and shoulder extension/abduction/internal rotation while extending the elbow and pronating the forearm accompanied by wrist dorsiflexion and finger extension in the supine position; (4) shoulder flexion/abduction/external rotation with elbow extension accompanied by wrist dorsiflexion and finger extension; (5) forearm supination/pronation with 90° elbow flexion in the sitting position; (6) wrist dorsiflexion and forearm pronation with extension of the fingers in the supine position; (7) finger extension with wrist flexion in the supine position (Figure 2); and (8) finger extension/flexion with wrist flexion in the sitting position.
Training involved a minimum of 5 and maximum of 8 patterns, with the number determined according to the participant’s level of motor impairment. Principles for selection and treatment included the following: (1) an emphasis on the proximal musculature, given its role in stabilizing distal movements; (2) gradual increases in movement difficulty with a goal of maintaining movement purity and avoiding synergy; and (3) consideration of the importance/nature of the movement (eg, the first and second fingers were trained separately and the third and fourth together to facilitate the acquisition of pinch).
Exercises were performed as 2 sets of 50 repetitions with an intervening 1- to 2-minute rest period. Thus, a total of 500 to 800 repetitions were performed during each session.
Each RFE session concluded with 30 minutes of dexterity-related training involving activities such as reaching-to-grasp blocks or pegs of differing sizes with the affected upper limb. Assistance with an unaffected limb was permitted.
Control group
Participants in the control group underwent a conventional upper-extremity rehabilitation program on the same 4-week, 5-d/wk, 40-min/session schedule as those receiving RFE. Sessions consisted of: (1) ROM exercises; (2) passive, assistive, active, and progressive resistive exercise; (3) the use of skateboards or weighted sanders; and (4) pinching or grasping blocks of various sizes. Each session concluded with an additional 30 minutes of the same dexterity-focused training activities as with the RFE group.
Strong efforts were made to ensure that the control and intervention groups were similar in terms of their time spent in their therapy sessions. Therapists in both groups recorded therapy duration (time), content, and compliance in detail for each session. The duration, frequency, and type of therapy provided to the control group were chosen to be consistent with that generally provided to stroke patients with this level of motor impairment.
Outcome Measures
The ARAT 26 served as the primary outcome measure. This instrument is designed to evaluate upper-extremity strength, dexterity, and coordination. It consists of 19 items divided into 4 subscales: grasp, grip, pinch, and gross movement in the horizontal and vertical planes. Subscales are scored on a 4-point ordinal scale from 0 (can perform no part of the test) to 3 (performs normally). The maximum score is 57. Reliability, validity, and responsiveness of the ARAT are well established in patients with stroke. 30
The secondary outcome measure, FMA, 27 was used to assess motor impairment. FMA has a maximum score of 66, which indicates optimal recovery and has psychometric properties that have been shown to be satisfactory in stroke patients. 30 Adverse effects of treatment were assessed through patient questioning. Two trained and experienced therapists who had no other contact with the study served as blinded evaluators for the study.
Statistical Analysis
A target sample size of 25 patients per group was calculated to give 80% power (α = .05, 2-tailed test) to detect a mean difference of 6 points in ARAT score, assuming that the study would have a drop-out rate of 5% to 10% and that its findings would have a standard deviation of 7 points. A between-group difference in ARAT scores of 6 points was targeted because the minimal clinically important difference of the ARAT is 5.7. 31
Baseline characteristics were compared using unpaired t tests for continuous variables and χ2 tests for frequencies. The primary analysis was an efficacy analysis that involved all patients who completed the study without missing a therapy or evaluation session. We also performed an intention-to-treat analysis in which the last-observation-carried-forward method was used for dealing with missing data. Change scores were calculated by subtracting baseline from outcome scores at 2 and 4 weeks.
General linear models, including multiple regressions within a repeated-measures analysis of covariance (ANCOVA) design, were used to compare improvement between the 2 groups at 2 and 4 weeks. Change scores between 0 and 2 weeks and between 0 and 4 weeks on the ARAT total, ARAT sub scores (grasp, grip, pinch, and gross movement), and FMA scores were the dependent variables (factor “time” in the analysis). Group (control, RFE) was the independent variable (factor “group”). To take into consideration nonhomogeneity between the 2 groups, pretreatment value of the same measure, time since stroke onset, study center (2 levels), age, sex, lateralization of the event, stroke type, and stroke location were included as covariates. A statistical interaction between group and time served to determine the efficacy of the RFE on the outcome measures. The primary end point for the trial was the change in the ARAT total score between the initiation and conclusion of the trial. Two-tailed tests were used. We considered P values less than .05 to be statistically significant.
Results are presented as mean and standard deviation, standard error, or 95% confidence limits, as appropriate. To index the magnitude of group differences in performance, the effect size η2 = Sum of squares (SS)between/SStotal was calculated for the ARAT total, ARAT subscales, and FMA scores. 32 The value of η2 represents the magnitude of the variability in the dependent variable (factor time) that can be explained (accounted for) by the independent variable (factor group). SPSS (version 19.0 for Mac OS X) was used for statistical analysis.
Results
Participant Characteristics
Figure 1 illustrates the process of recruitment. A total of 259 candidates were screened between October 2008 and March 2010. Of these, 190 did not meet the inclusion criteria, and an additional 17 declined to participate. The remaining 52 were enrolled in the study with 27 randomized to the RFE treatment group (14 from 1 of the 2 participating centers and 13 from the other) and 25 to the control treatment (12 from the first center and 13 from the other). There were 3 dropouts within the first week; 1 from each group as a result of their request for non–study-related reasons and 1 from the control group as a result of non–study-related medical issues. The remaining 49 participants successfully completed all assigned treatments (ie, participated throughout the full session) and evaluation sessions. Thus, data from 49 participants were available for the efficacy analysis. Data from all 52 initially randomized participants were used for the intention-to-treat analysis (Figure 1).
No adverse treatment effects were noted. The 2 groups did not differ significantly in terms of demographic characteristics at entry—age, sex, stroke duration, and stroke location—or baseline measures of ARAT and FMA scores (Table 1). There were no significant differences in demographics between those at the 2 participating centers or between those who dropped out and the overall sample.
Baseline Characteristics of Participants (n = 49) a
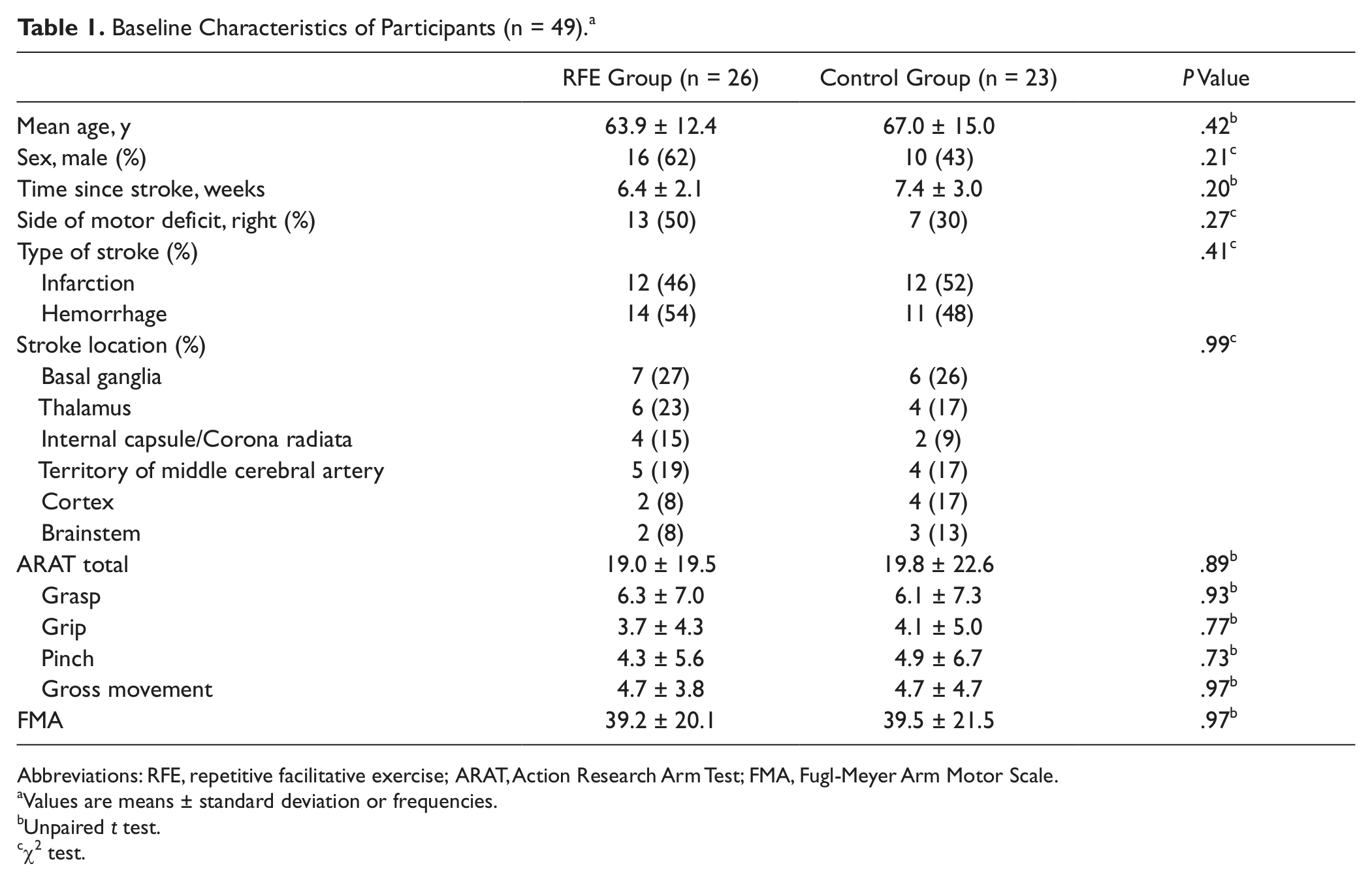
Abbreviations: RFE, repetitive facilitative exercise; ARAT, Action Research Arm Test; FMA, Fugl-Meyer Arm Motor Scale.
Values are means ± standard deviation or frequencies.
Unpaired t test.
χ2 test.
Efficacy and Intention-to-Treat Analyses
Action Research Arm Test
Efficacy (Table 2) and intention-to-treat (Table 3) analyses are presented as change scores relative to baseline at the 2-week midpoint and 4-week conclusion. The tables also display results of the repeated-measures ANCOVA that tested the effects of RFE relative to control treatment.
Changes in Outcome Measures at the Midpoint and End of Treatment: Efficacy Analysis (n = 49) a
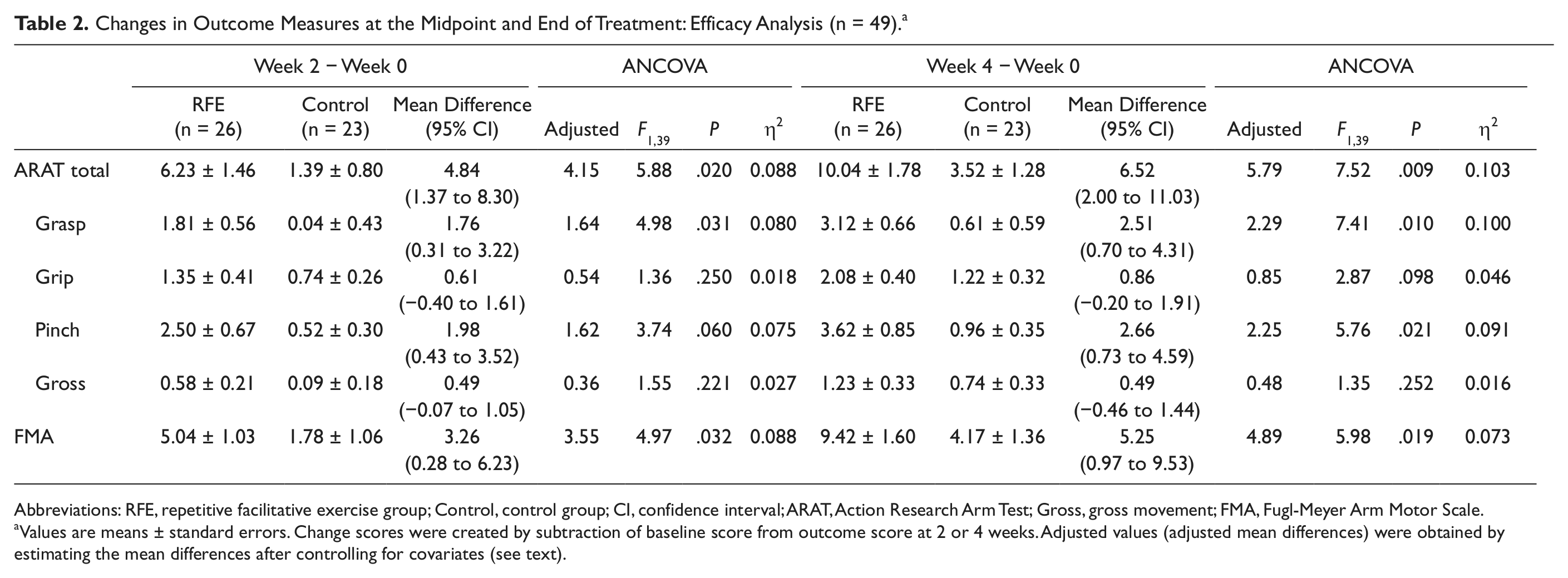
Abbreviations: RFE, repetitive facilitative exercise group; Control, control group; CI, confidence interval; ARAT, Action Research Arm Test; Gross, gross movement; FMA, Fugl-Meyer Arm Motor Scale.
Values are means ± standard errors. Change scores were created by subtraction of baseline score from outcome score at 2 or 4 weeks. Adjusted values (adjusted mean differences) were obtained by estimating the mean differences after controlling for covariates (see text).
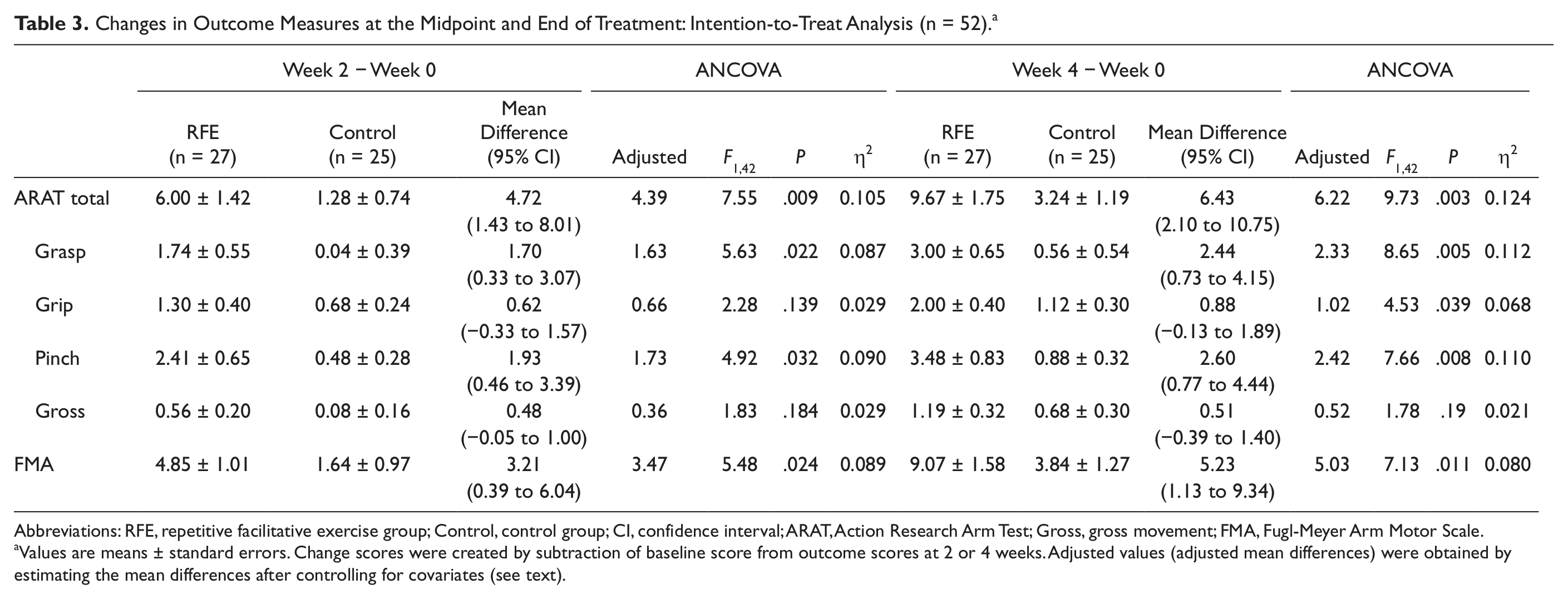
Changes in Outcome Measures at the Midpoint and End of Treatment: Intention-to-Treat Analysis (n = 52) a
Abbreviations: RFE, repetitive facilitative exercise group; Control, control group; CI, confidence interval; ARAT, Action Research Arm Test; Gross, gross movement; FMA, Fugl-Meyer Arm Motor Scale.
Values are means ± standard errors. Change scores were created by subtraction of baseline score from outcome scores at 2 or 4 weeks. Adjusted values (adjusted mean differences) were obtained by estimating the mean differences after controlling for covariates (see text).
In the efficacy analysis, the RFE group displayed a crude mean increase of 10.04 points at the end of training on the study’s primary outcome variable, the ARAT, relative to a 3.52 improvement in the control group (Table 2). The crude difference between the 2 groups was 6.52 points (95% confidence interval [CI] = 2.00-11.03); the adjusted difference was 5.79 points. There were significant group and time interactions in the ARAT after 2 (F = 5.88: P = .02) and 4 (F = 7.52; P = .009) weeks of the intervention (Table 2). A similar pattern was seen in the intention-to-treat analysis (Table 3), in that the difference in improvement between the 2 groups in the intention-to-treat analysis favored the RFE group by 6.43 points (crude difference; 95% CI = 2.10-10.75; adjusted difference = 6.22 points) and showed a significant interaction between group and time (F = 9.73; P = .003).
Two of the 4 ARAT subscales, grasp and pinch, displayed significant interactions between group and time after 4 weeks of intervention (F = 7.41, P = .01 and F = 5.76, P = .021, respectively). Significant differences, however, were not found in the other 2 subscales (Table 2). Results from intention-to-treat analysis revealed that 3 of the 4 subscales—grasp, grip, and pinch—favored RFE after 4 weeks of intervention (F = 8.65, P = .005; F = 4.53, P = .039; and F = 7.66, P = .008, respectively) but not in gross movement (Table 3).
Fugl-Meyer Arm Motor Scale
Efficacy analysis revealed that differences in the change scores of the 2 groups on the FMA again favored RFE at both the study’s midpoint (crude difference 3.26 points [95% CI = 0.28-6.23]; adjusted difference = 3.55 points) and 4-week conclusion (crude difference 5.25 points [95% CI = 0.97-9.53]; adjusted difference = 4.89 points). There were significant interactions between group and time (Table 2) at both week 2 (F = 4.97; P = .032) and week 4 (F = 5.98; P = .019). Intention-to-treat analysis also revealed significant interactions between group and time (Table 3) at both time points (F = 5.48, P = .024; F = 7.13, P = .011, respectively).
Discussion
This study represents the first randomized controlled evaluation of RFE, a combination of a high dose (high frequency) of repetitions and neurofacilitation techniques, compared with a duration-matched conventional rehabilitation program in the treatment of stroke-related upper-extremity impairment. The findings are intriguing, in that, at least for those with subacute stroke, RFE demonstrated both statistically and clinically significant benefits over conventional rehabilitation on the trial’s primary outcome variable (ARAT). It is also encouraging that the findings on ARAT, which is designed to measure dexterity and function, were mirrored by the study’s secondary outcome variable, the Brunnstrom-oriented FMA, which was chosen as a measure of motor control.
Overall, the results of this study support and extend the findings of our earlier exploratory work. 24 More specifically, the improvements in object manipulation, as evaluated with ARAT in this investigation, are present but more pronounced than those found in that earlier work. 24 The magnitude of the lessening of motor impairment (as measured by the FMA) in the RFE group found in this trial is also consistent with that found in earlier work. 24 It is also encouraging that no adverse effects were noted with the use of RFE either in terms of patient complaints or increased muscle tone (data not shown).
A number of investigations and meta-analyses failed to show evidence of efficacy using neurophysiological approaches for motor recovery after stroke.1,10,33 Given these reports, it might appear surprising that the neurophysiologically based RFE intervention proved more effective than a conventional rehabilitation program. We believe that the answer lies in 2 factors: RFE’s use of an unusually large number of repetitions and differences in the nature of its application from traditional neurofacilitation approaches.
Role of High Rate of Repetition
This study was designed to ensure that the participants in the RFE and control groups received equal amounts of therapy time because it is known that differing amounts of participation can affect outcomes. 34 However, as noted above, while the participants entered into the conventional therapy group received what most would consider to be an intensive therapy program, it is true that those assigned to RFE received far more active-assisted movements/sessions than occur in traditional approaches. 33
Basic science studies suggest that repetition of voluntary movement is important for recovery.35-37 In this regard, it is pertinent that RFE uses repetitive movements of increasing difficulty in a manner similar to that of other successful upper-extremity studies.37,38 The first, the Arm BASIS Training trial, which resulted in improved motor control following stroke, provides support for the RFE approach, in that it also emphasized avoidance of synergistic patterns and used a similar paradigm of systematic repetitive voluntary/assisted movements throughout a patient’s full ROM.39,40 A difference from our approach, however, is that Arm Basis training, unlike RFE, does not use stretch reflexes.
Additional support for the benefits of high repetition rates may come from another randomized controlled trial that compared Bi-Manu-Track with neuromuscular electrical stimulation, which found that the high-repetition Bi- Manu-Track method was associated with larger improvements in patients with stroke-associated upper-extremity impairments. 13
Differences in Type of Intervention
RFE uses neurofacilitation techniques, in conjunction with therapist’s verbal direction and physical assistance, to assist the patient’s efforts in moving an affected joint. 24 In contrast, the control treatment did not use neurofacilitation. Although traditional neurofacilitation techniques seek to normalize muscle tone and posture,41-43 RFE differs from those approaches in a number of ways. Three in particular come to mind. First, unlike traditional neurofacilitation approaches, RFE uses tactile and stretch facilitation in conjunction with specific joint positioning to generate targeted movements. Second, RFE places greater importance on proximal movements than is true for more traditional neurofacilitation approaches. 24 Third, RFE tailors specific approaches for individual digits (see Figure 2), which may contribute to the RFE group’s improvement in the grasp and pinch subscales of the overall ARAT score.
In summary, it remains unclear whether the improvements associated with the application of RFE in our study were a result of the facilitative technique itself, the high number of repetitions, or a combination of the 2. Further studies are required to investigate the mechanisms that form the basis of RFE’s effects on motor recovery.
Clinical Applicability of RFE
Stroke rehabilitation is an active and evolving area of research, and a variety of approaches, ranging from constrained use11,12 and robots13,14 to various forms of stimulation,44-49 are in various stages of development. All have strengths and weaknesses. Some are invasive, and many require specialized equipment. RFE, while sharing a requirement for intense therapist involvement, has the advantages of requiring no equipment and being able to be performed by any therapist who has mastered the techniques basic to their training. More research is needed, but RFE could conceivably be integrated with other approaches to achieve further improvement in its capabilities.
Limitations
Among the strengths of our study are its rigid adherence to a carefully designed protocol, use of validated outcome measures, and a high completion rate among participants. Among its weaknesses are a lack of follow-up and the inability, thus far, to assess the sustainability of its findings. Similarly, mixtures of stroke location and type as well as varying levels of impairment and its restriction of those in the subacute stages of stroke render generalization to other situations difficult. Further research will require an examination of whether the improvements in function obtained by RFE translate into improved activities of daily living performance and “real world arm use” during functional activities.
Conclusion
RFE, at least during the subacute phase of stroke, may be more effective than conventional rehabilitation in lessening stroke-associated impairment and improving upper-limb motor function. The optimal regimen of RFE and its long-term benefits remain to be determined.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
