Abstract
Background. Growing evidences suggest that brain-derived neurotrophic factor/tropomyosin receptor kinase B (BDNF/TrkB) plays a key role in the regulation of hippocampal synaptic plasticity in a prenatal stress (PNS) rat model. Repetitive transcranial magnetic stimulation (rTMS) is currently being acknowledged to affect attention and memory in both preclinical and clinical studies, although the mechanism is still unclear. Objective. The current study aimed to explore whether a whole brain rTMS (5 Hz, 14 days) could ameliorate cognitive dysfunction–induced PNS in male offspring, and examine if the positive effect of rTMS was associated with the BDNF/TrkB signaling in the hippocampus. Methods. The rats were randomly divided into 5 groups: CON, PNS, PNS + rTMS, PNS + rTMS + DMSO (dimethyl sulfoxide), and PNS + rTMS + K252a. Spatial cognition was evaluated by using Morris water maze test. Following behavioral assessment, both paired-pulse facilitation and long-term potentiation were recorded from Schaffer collaterals to CA1 region in the hippocampus. Synaptic, apoptotic, and BDNF/TrkB signaling proteins were measured by Western blot. Results. PNS-exposed offspring exhibited cognitive deficits, long-term potentiation inhibition in the hippocampus, the decrease of synaptic and BDNF/TrkB signaling proteins expression, apoptosis, and reduced number of cells in the CA1 region. Five-hertz rTMS significantly alleviated the PNS-induced abnormalities. However, the effect of rTMS was antagonized by intracerebroventricular infusion of K252a (a TrkB inhibitor). Conclusions. The findings suggest that 5-Hz rTMS significantly improves the impairment of spatial cognition and hippocampal synaptic plasticity, which is possibly associated with the activation of BDNF/TrkB signaling.
Keywords
Introduction
Prenatal stress (PNS) causes widespread deleterious effects on the development of offspring and thus induces cognitive deficits.1,2 Increasing evidences show that spatial learning and memory loss is a severe consequence of PNS. In fact, the data, obtained from pregnant women exposed to stressful environment, showed that the possibility of their offspring to behavioral and emotional impairment was increased. 3 Moreover, animal studies showed that PNS-induced synaptic plasticity abnormality in hippocampus, neurons loss, and behavioral changes in the offspring. 4 One of our previous studies showed that PNS induced cognitive deficits and synaptic plasticity damages in adolescent male offspring. 5 Accumulating evidences suggest that brain-derived neurotrophic factor (BDNF), as a putative biological marker, has been involved in the depressive disorder. In animals subjected to chronic stress6,7 and forced swimming, 8 BDNF levels were depressed in the hippocampus. Antidepressants may relieve the symptoms during pregnancy, but little information is available on the developmental consequences for the offspring.9,10
Repetitive transcranial magnetic stimulation (rTMS) is currently being acknowledged to affect attention and memory in both preclinical and clinical investigations.11,12 Indeed, 1-Hz or 10-Hz rTMS could improve the cognitive performance of depressed patients. 13 Furthermore, one of our previous studies reported that 5-Hz rTMS effectively facilitated spatial cognition and synaptic plasticity associated with the level of BDNF in normal Wistar rats. 14 However, there has been still a profound lack of knowledge concerning the putative effects of rTMS on cognitive deficits and the underlying molecular mechanism.
BDNF and its specific membrane-bound receptor TrkB, a member of the tropomyosin receptor kinase (Trk) family, are considered as important upstream regulators of synaptic plasticity in brain. 15 BDNF binding to TrkB triggers autophosphorylation of tyrosine residue in its intracellular domain, which consequently activates 1 of 3 major signaling pathways involving in mitogen-activated protein kinase (MAPK), phosphatidylinositol 3-kinase (PI3K), and phospholipase Cg (PLC-g). 16 It is well-known that stress decreases the level of BDNF/TrkB/CREB signaling,17,18 which probably weakens the transcription of BDNF and Bcl-2. Blocking BDNF signaling pathway may activate apoptosis pathways. 19 Interestingly, previous studies showed that rTMS improved BDNF/TrkB signaling both in vivo and in vitro, and the transcription factor cAMP response element binding protein (CREB) was phosphorylated by intracellular kinases in response to a vast range of physiological and pathological stimuli.20,21
In the present study, we hypothesized that rTMS efficiently alleviated the impairment of the hippocampal synaptic plasticity and spatial cognition through regulating BDNF/TrkB signaling. Accordingly, we examined the effects of 5-Hz rTMS on prenatal restraint stress-induced cognitive dysfunction in male offspring. To investigate the regulatory mechanisms of rTMS on synaptic plasticity, a TrkB inhibitor K252a was employed in the offspring. The Morris water maze (MWM) was performed to evaluate the spatial cognition. Afterward, the long-term potentiation (LTP) and paired-pulse facilitation (PPF) from the hippocampal Schaffer collaterals to CA1 region were recorded. In addition, BDNF/TrkB signaling-related protein expression and apoptosis level were measured by Western blot assay. And synapse-related protein expression was detected by the immunofluorescence staining and Western blot assay in the hippocampus. Moreover, the hematoxylin-eosin (HE) staining was used to detect the number of cells in the CA1 region. To our knowledge, this is the first evidence that rTMS significantly improves the cognitive deficits and synaptic plasticity damages via activating BDNF/TrkB signaling pathway.
Material and Methods
Animals
Ten pregnant Wistar rats were purchased from the Laboratory Animal Center of the Academy of Military Medical Science of People’s Liberation Army and then housed with standard laboratory conditions (24°C ± 2°C room temperature, 12-hour light/dark cycle with lights on at 7:00
Experimental Design and PNS Procedure
The PNS procedure was the same as for our previous study. 5 From Days 15 to 21 of gestation (Figure 1A), 8 pregnant rats were used to establish the PNS model, in which the pregnant rats were restrained in a transparent plastic cylinder (64 mm in diameter) for 45 minutes, 3 times a day. The other 2 pregnant rats were defined as control. The pregnant animals were housed with 2 individuals in 1 cage to avoid the effect of social isolation for rats. And then they were housed separately until day 19 of gestation to wait for natural birth. After birth, all the offspring were reserved together with their biological mothers until weaning. No more than 4 pups from same litter were used. Only male pups were randomly used and housed with 4 to 6 individuals in 1 cage. The male offspring were divided into 5 groups: CON (n = 8), PNS (n = 8), PNS + rTMS (n = 8), PNS + rTMS + DMSO (dimethyl sulfoxide) (n = 6), and PNS + rTMS + K252a (n = 6).
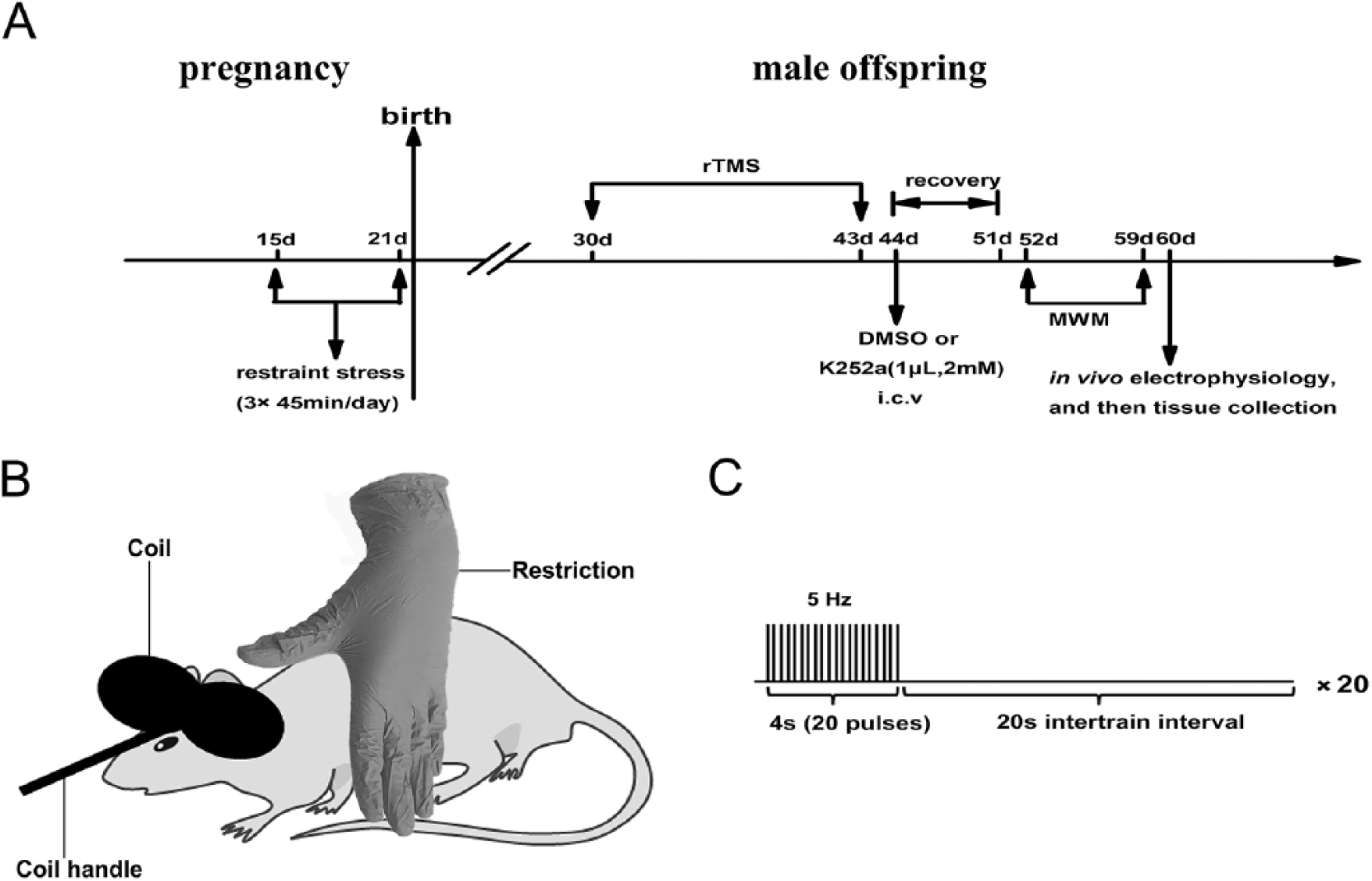
(A) Experimental procedure and schedule. (B) Diagram of rat stimulation. (C) Diagrammatic sketch of rTMS (repetitive transcranial magnetic stimulation) parameter pattern.
Repetitive Transcranial magnetic stimulation procedure
The rTMS was applied on postnatal male offspring for 30 days (Figure 1A). The details of rTMS stimulation parameters and pattern are shown in Figure 1B and C. The protocol is provided in the supplementary material.
Stereotaxic Surgery
After the 14 days of rTMS (Figure 1A), animals in either the PNS + rTMS + DMSO group or the PNS + rTMS + K252a group were anesthetized and then were placed in a stereotaxic frame, and DMSO (1 µL) or K252a (2 mM, 1 µL) was injected into lateral ventricles (0.9 mm posterior to the bregma, 1.5 mm lateral to midline, 3.3 mm ventral below the dura) of the brain at a flow rate of 0.5 µL/min using a 10-µL Hamilton syringe. 22 After the injection, the needle was kept in place for 5 minutes before it was slowly withdrawn.
Morris Water Maze Test
Seven days after the surgery, the MWM test was performed to examine the spatial cognition. It consists of both initial training (IT) and space exploring tests (SET). The IT stage includes 4 test days, 2 sessions for each day, and 4 trials for each session. Trials are randomly started in drop zones from 4 start locations (S, E, NE, and SW). In each trial, the animals were allowed to swim for 60 seconds to find the hidden platform in the quadrant NW. Rats that failed to find the platform were guided to the platform and stayed for about 10 seconds.
Afterward, it was performed in the SET stage using 1 trial without the platform after the last session of the IT stage approximately 24 hours later.
The detailed protocols are provided in the Supplementary Materials.
In Vivo Electrophysiological Recordings
In vivo recordings were made 24 hours after the MWM test. Briefly, rats were anesthetized with urethane (1.2 g/kg body weight; supplemental doses of 0.2-0.8 g/kg when needed) by intraperitoneal injection, and then they were placed in a stereotaxic frame (SN-3, Narishige, Japan) for a surgery.
To determine whether the stimulating and recording electrodes were in the accurate sites, input/output (I/O) curve was determined using a range of stimulation intensities from 0.1 to 1 mA. The stimulating intensity (range 0.3-0.5 mA) that could evoke a response of 70% of its maximum amplitude was chosen to deliver in PPF, baseline, LTP field excitatory postsynaptic potential (fEPSP) recordings.
The detailed protocols are provided in the Supplementary Material.
Antibodies
Supplementary Table 1 lists the antibodies used in the study.
Total Proteins Extract and Western Blot Assay
The rats in 5 groups were decapitated immediately after electrophysiological recording (n = 4 in each group). The hippocampus was promptly separated, stored at −80°C, ground and lysed in 400 µL lysis buffer, which contained a protease inhibitor cocktail (1:100 dilutions).
The detailed protocols are provided in the supplementary material.
Hematoxylin-Eosin and Immunofluorescence Stainings
After the brain was isolated from the sacrificed rat, it was quickly embedded in OCT (optimal cutting temperature) compound for tissue sectioning. After that, 20-µm thick coronal sections were immediately made using a freezing microtome. Fixation was performed after slicing procedure by ethanol for 15 minutes. The detail of the protocol about HE and immunofluorescence staining is provided in the supplementary material.
Data and Statistical Analysis
All data were presented as mean ± standard error of the mean (SEM). The data normality was tested by Shapiro-Wilk test for each group. For group comparisons of normally distributed data (P > .05), we used 1-way analysis of variance (ANOVA) or 2-way repeated-measures ANOVA. Post hoc statistics were obtained using Bonferonni’s multiple comparison test. For non-Gaussian distributions (P ≤ .05), we used Kruskal-Wallis test. All analyses were performed using SPSS 21.0 software, and the significance level was set at .05.
Results
rTMS Significantly Ameliorated Cognitive Impairments in PNS Male Offspring
The MWM test was performed to estimate the effect of rTMS on spatial learning and memory in the PNS male offspring. During the IT stage, it could be seen that the escape latencies were decreased with training (Figure 2A). It showed that there were significant differences of day × group interaction (F(12, 93) = 2.078, P < .01), day (F(3, 29) = 106.105, P < .001), and group (F(4, 31) = 9.849, P < .001). The escape latencies were significantly increased in the PNS group compared with that in the CON group (day 2, P < .001; day 3, P < .001; day 4, P < .001; Figure 2A), and shorter in the PNS + rTMS group compared with that in the PNS group (day 2, P < .001; day 3, P < .001; day 4, P < .001; Figure 2A). There was no significant difference of escape latencies between the PNS + rTMS group and the PNS + rTMS + DMSO group (Figure 2A). After treatment with K252a, the latencies were significantly increased compared with that in the PNS + rTMS + DMSO group (day 4, P < .05; Figure 2A). There was a similar tendency for the path length compared with that of the escape latency (Figure 2B). Furthermore, there was no significant difference of swimming speed in all groups (Figure 2C, P > .05).
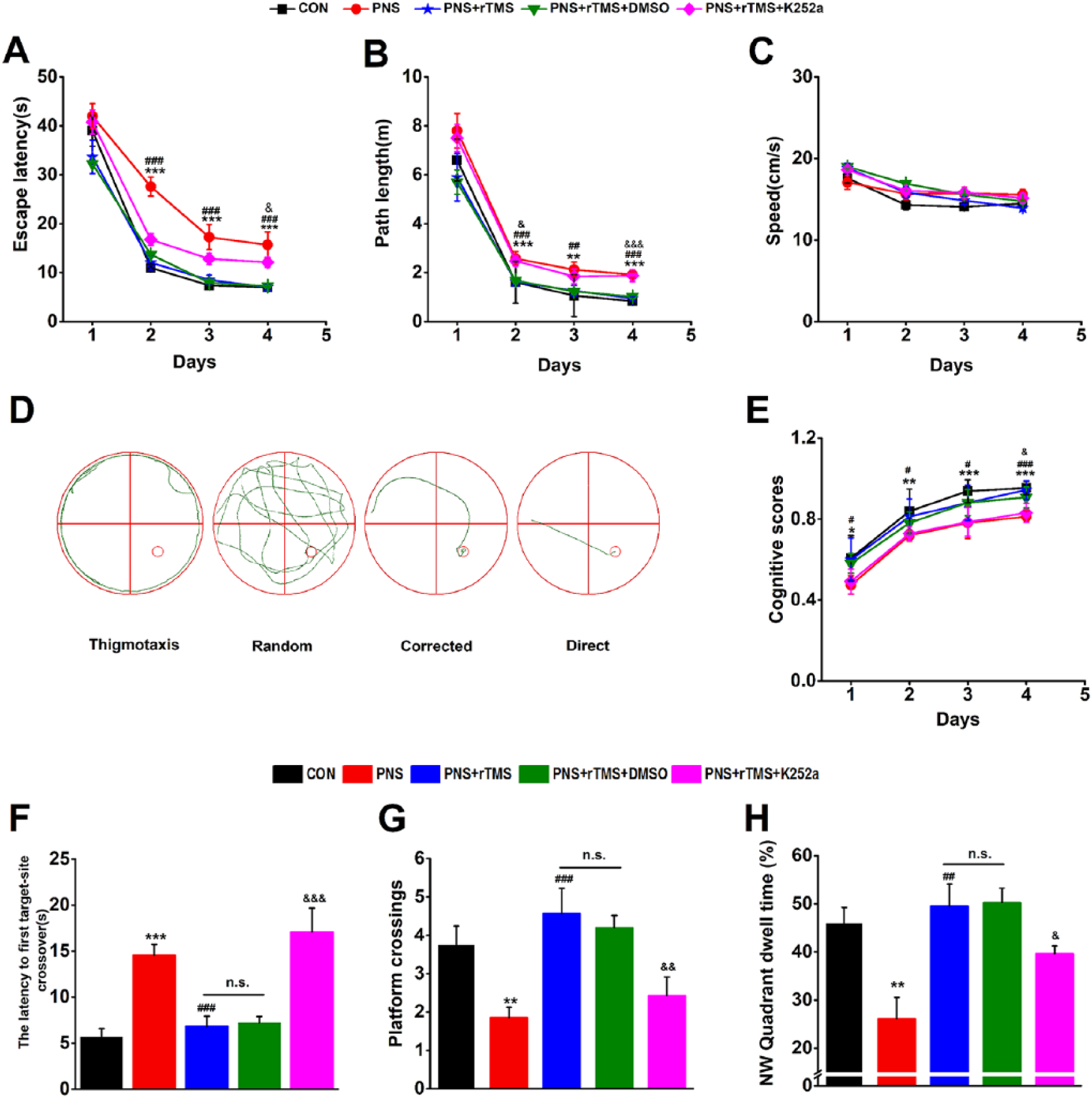
Performances of male offspring in the Morris water maze (MWM) test in the 5 groups. (A) Mean escape latency was calculated for each day in the IT (initial training) stage. (B) Mean path length was calculated for each day in the IT stage. (C) Mean swimming speed was calculated for each day in the IT stage. (D) Examples of search strategies in the IT stage. (E) The cognitive scores of search strategies in the IT stage. (F) The latency to first target-site crossover in the SET (space exploring tests) stage. (G) Mean number of platform area crossings in the SET stage. (H) Mean percentage of time spent in target quadrant (NW) in the SET stage.
The typical traces and statistical results of cognitive scores in spatial strategy are shown in Figure 2D and E, respectively. There were significant differences of day × group interaction (F(12, 93) = 0.228, P < .01; Figure 2E), day (F(3, 29) = 221.20, P < .001; Figure 2E), and group (F(4, 31) = 11.683, P < .001; Figure 2E). The cognitive scores were decreased in the PNS group compared with that in the CON group (day 1, P < .05; day 2, P < .01; day 3 and day 4, P < .001), and significantly increased in the PNS + rTMS group compared with that in the PNS group (day 1, day 2, and day 3, P < .05; day 4, P < .001). There was no significant difference of the scores between the PNS + rTMS group and the PNS + rTMS + DMSO group. After treatment with K252a, they were reduced compared with the PNS + rTMS + DMSO group (day 4, P < .01).
In the SET stage, the reference memory was evaluated. There were statistical differences of the latency to first target site crossover among the groups (F(4, 35) = 13.051, P < .001; Figure 2F). Post hoc Bonferonni’s multiple-comparison test showed that the latency was longer in the PNS group than that in the CON group (P < .001), and shorter in the PNS + rTMS group than that in the PNS group (P < .001). There was no statistical difference of the latency between the PNS + rTMS group and the PNS + rTMS + DMSO group. After treatment with K252a, the latency became longer in the PNS + rTMS + DMSO group than that in the PNS + rTMS group (P < .001). Kruskal-Wallis test showed that there were statistical differences of the platform crossings (Figure 2G; F(4, 34) = 20.110, P < .001) and the target quadrant dwell time among the groups (Figure 2H; F(4, 34) = 15.120, P < .001). Kruskal-Wallis multiple comparison showed that the number of platform crossings (P < .01) and the target quadrant dwell time (P < .01) were significantly decreased in the PNS group compared with that in the CON group. They were significantly increased in the PNS + rTMS group compared with that in the PNS group (P < .001 and P < .01). There was no significant difference of them between the PNS + rTMS group and the PNS + rTMS + DMSO group. After treatment with K252a, they were statistically decreased compared with that in the PNS + rTMS + DMSO group (P < .01 and P < .05).
rTMS Significantly Enhanced Hippocampal LTP in PNS Male Offspring
In the electrophysiological experiment, the fEPSPs were recorded in the hippocampal CA1 area through stimulating Schaffer collaterals. The inset in Figure 3A shows an example of I/O curve. The amplitude was increased as stimulating intensity strengthened. After paired pulses were applied to the hippocampus Schaffer collaterals, the PPF appeared with the second fEPSP that was obviously larger than the first one all the times (Figure 3B). There was no significant interaction of different interstimulus interval (ISI) × group interaction (F(4, 20) = 0.234, P > .05) and group (F(4, 20) = 1.551, P > .05). Figure 3B inset shows an example of PPF.
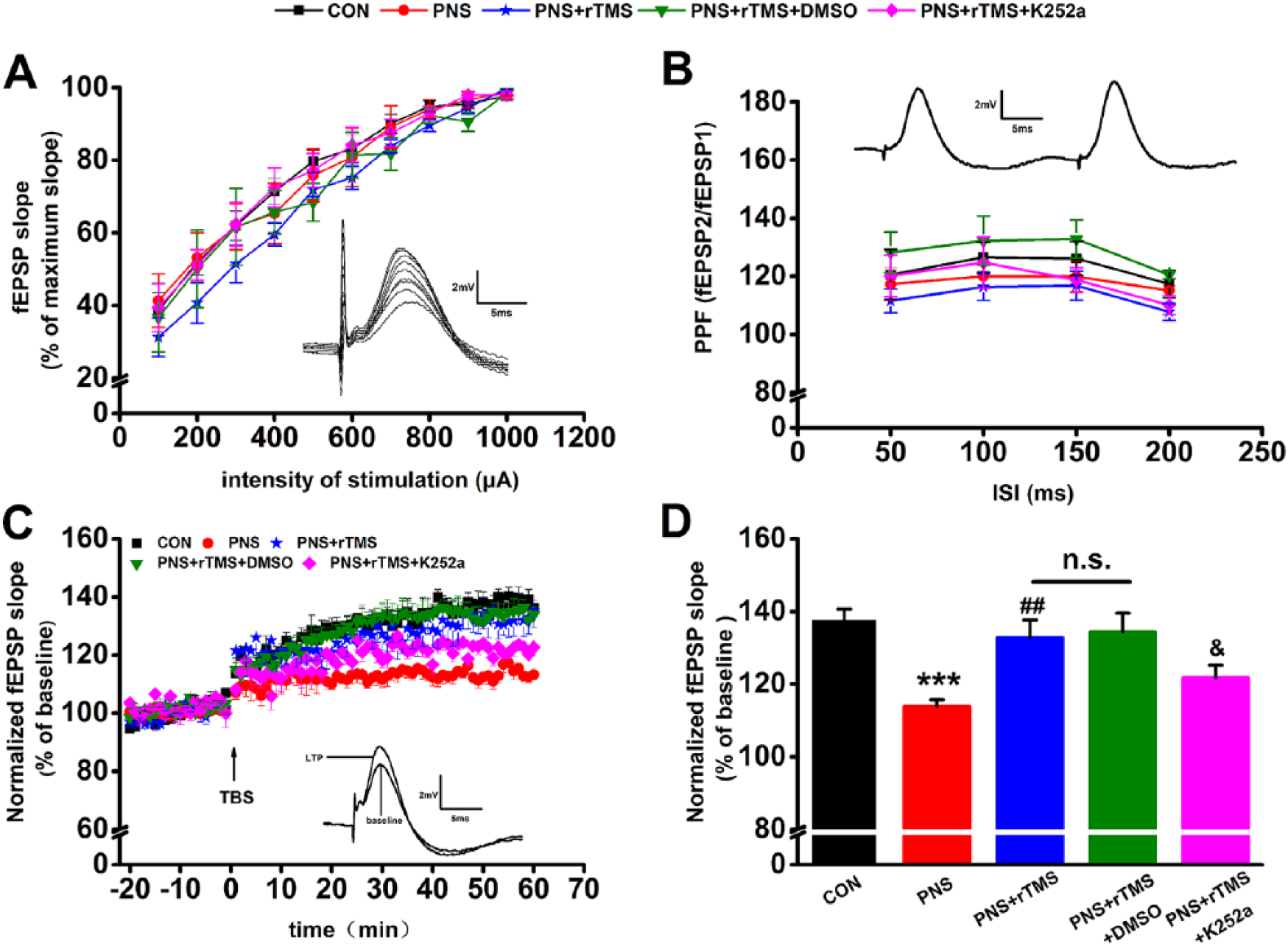
In vivo electrophysiological recordings from Schaffer collaterals to the hippocampal CA1 region. (A) I/O (input/output) curve was determined using a range of stimulation intensities from 0.1 to 1 mA. The inset shows an example of I/O curve. (B) PPF (paired-pulse facilitation), a form of short-term plasticity, was measured and expressed as the ratio of fEPSP2 to fEPSP1 (field excitatory postsynaptic potentials). The inset shows an example of PPF. (C) The changes of time coursing of fEPSP slopes in the 5 groups. The first 20 minutes of evoked responses were normalized and used as the baseline responses of LTP (long-term potentiation). The inset shows an example of fEPSP at baseline TBS (theta burst stimulation) and LTP in each group. (D) Magnitude of LTP was determined as responses between 50 and 60 minutes after the TBS.
In the LTP assay, the fEPSPs slopes recorded for 1 hour were increased immediately following the TBS (theta burst stimulation). The time course of fEPSPs slopes that has been normalized to the 20-minute baseline period is presented in Figure 3C (left). The inset in Figure 3C represents an example of fEPSPs at both the baseline and LTP of a rat in the CON group. Results from the last 10 minutes revealed that there were significant differences of LTP between the groups (F(4, 35) = 8.028, P < .001; Figure 3D). Furthermore, post hoc Bonferonni’s multiple-comparison test showed that the mean fEPSPs slopes were much smaller in the PNS group compared with that in the CON group (P < .001); however, they were significantly enhanced by rTMS in the PNS + rTMS group (P < .01). There was no significant difference of LTP between the PNS + rTMS group and the PNS + rTMS + DMSO group. After treatment with K252a, the mean fEPSPs slopes were decreased compared with the PNS + rTMS + DMSO group (P < .05; Figure 3D).
rTMS Increased BDNF/TrkB Signaling in the Hippocampus of PNS Male Offspring
The BDNF including its receptor TrkB and downstream p-CREB and CREB was measured by using Western blot assay in the hippocampus. The bands were distinguished by corresponding antibodies (Figure 4A). It was found that there were statistical differences of the above protein levels among the 5 groups (BDNF, F(4, 13) = 7.607, P < .001; TrkB, F(4, 13) = 4.807, P < .05; p-CREB/CREB, F(4, 13) = 11.196, P < .001; Figure 4B). The PNS significantly decreased BDNF (P < .05; Figure 4B), TrkB (P < .05; Figure 4C), and p-CREB/CREB (P < .001; Figure 4D) proteins in the PNS group compared with that in the CON group. However, all of these indexes were significantly increased in the PNS + rTMS group compared with that in the PNS group (BDNF, P < .05; TrkB, P < .01; p-CREB/CREB, P < .001). There was no significant difference of the indexes between the PNS + rTMS group and the PNS + rTMS + DMSO group. Interestingly, the expressions of BDNF (P < .001), TrkB (P < .01), and p-CREB/CREB (P < .001) were reduced in the PNS + rTMS + K252a group compared with that in the PNS + rTMS + DMSO group.
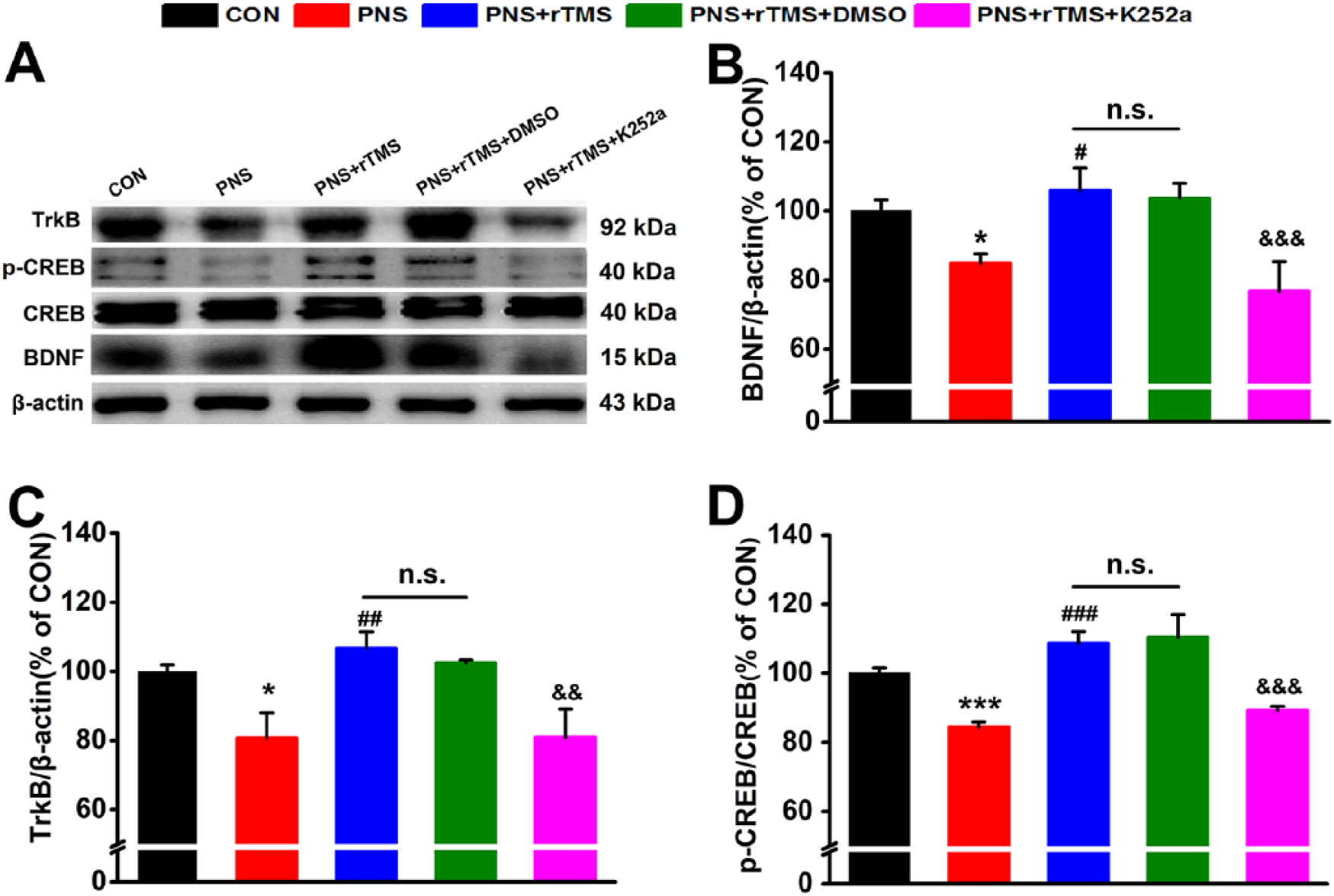
The expression of brain-derived neurotrophic factor (BDNF), tropomyosin receptor kinase B (TrkB), and cAMP response element binding protein (p-CREB/CREB) in the hippocampus. (A) Results are immunoblots from single representative experiments. (B) The expression values of the BDNF. (C) The expression values of the TrkB. (D) The expression values of the p-CREB/CREB were normalized with β-actin value.
rTMS Increased the Expression of NR2A and NR2B in the Hippocampus of PNS Male Offspring by Activating the BDNF/TrkB Signaling
The synaptic-related markers synaptophysin (SYP), NR2A, and NR2B were detected in the hippocampus. Prominent bands were identified by antibodies respectively (Figure 5A). It was found that there were statistical differences of the NR2A (F(4, 14) = 7.607, P < .001) and NR2B (F(4, 14) = 4.807, P < .05) protein levels among the 5 groups. However, there was no significant difference of the SYP protein level in the 5 groups (P > .05; Figure 5B). The PNS significantly decreased NR2A (P < .05; Figure 5C) and NR2B (P < .001; Figure 5D) proteins in the PNS group compared with that in the CON group. Both NR2A and NR2B were significantly increased in the PNS + rTMS group compared with that in the PNS group (NR2A, P < .01; NR2B, P < .001; Figure 5C and D). Similarly, there was no significant difference of the indexes between the PNS + rTMS group and the PNS + rTMS + DMSO group (Figure 5C and D). After treatment with K252a, the expression of NR2A (P < .01) and NR2B (P < .001) was reduced compared with that in the PNS + rTMS + DMSO group (Figure 5C and D). In addition, immunofluorescence staining test showed that there were similar results about the expressions of both NR2A and NR2B in these 5 groups in the hippocampal CA1 region (Figure 5E).
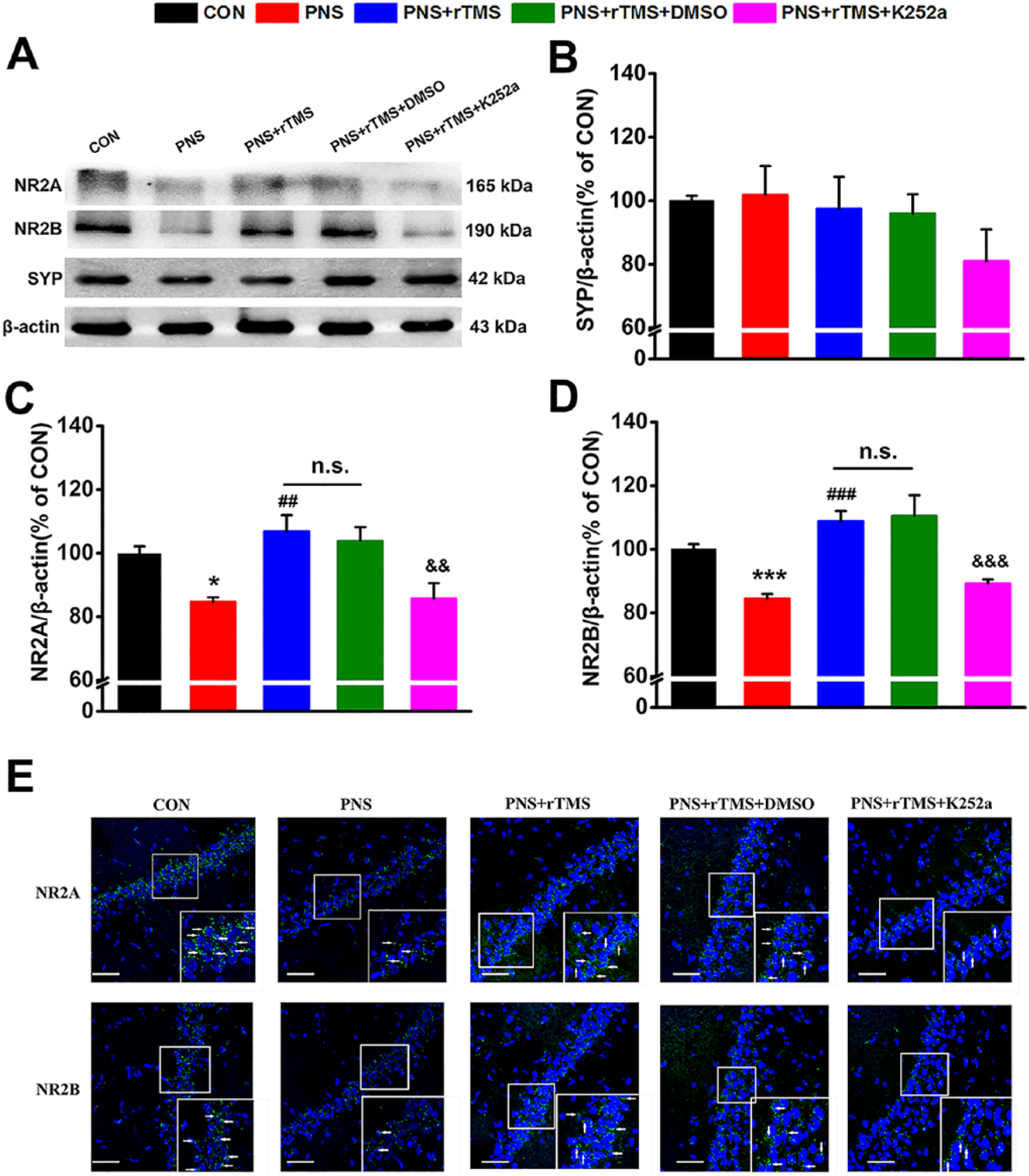
The expression of SYP (synaptophysin), NR2A, and NR2B in the hippocampus in 5 groups. (A) Results are immunoblots from single representative experiments. (B) The expression values of the SYP. (C) The expression values of the NR2A. (D) The expression values of the NR2B were normalized with β-actin value. (E) The expressions of NR2A and NR2B identified by immunofluorescence staining in the hippocampal CA1 region. White arrows denote protein expression. Scale bar: 50 µm.
rTMS Prevented the Apoptosis in the Hippocampus of PNS Male Offspring by Activating the BDNF/TrkB Signaling
The number of cells in the CA1 region was evaluated by HE staining (Figure 6A). As shown in Figure 6B, they were calculated in the hippocampal CA1 region. There were statistical differences of the cells number among the 5 groups (F(4, 14) = 23.212, P < .001; Figure 6B). The PNS significantly decreased the number of cells in the PNS group compared with that in the CON group (P < .001; Figure 6B), and it was significantly increased in the PNS + rTMS group compared with that in the PNS group (P < .01; Figure 6B). There was no significant difference of the number between the PNS + rTMS group and the PNS + rTMS + DMSO group (Figure 6B). After treatment with K252a, The number of cells in the hippocampal CA1 region was reduced in the PNS + rTMS + K252a group compared with that in the PNS + rTMS + DMSO group (P < .05; Figure 6B).
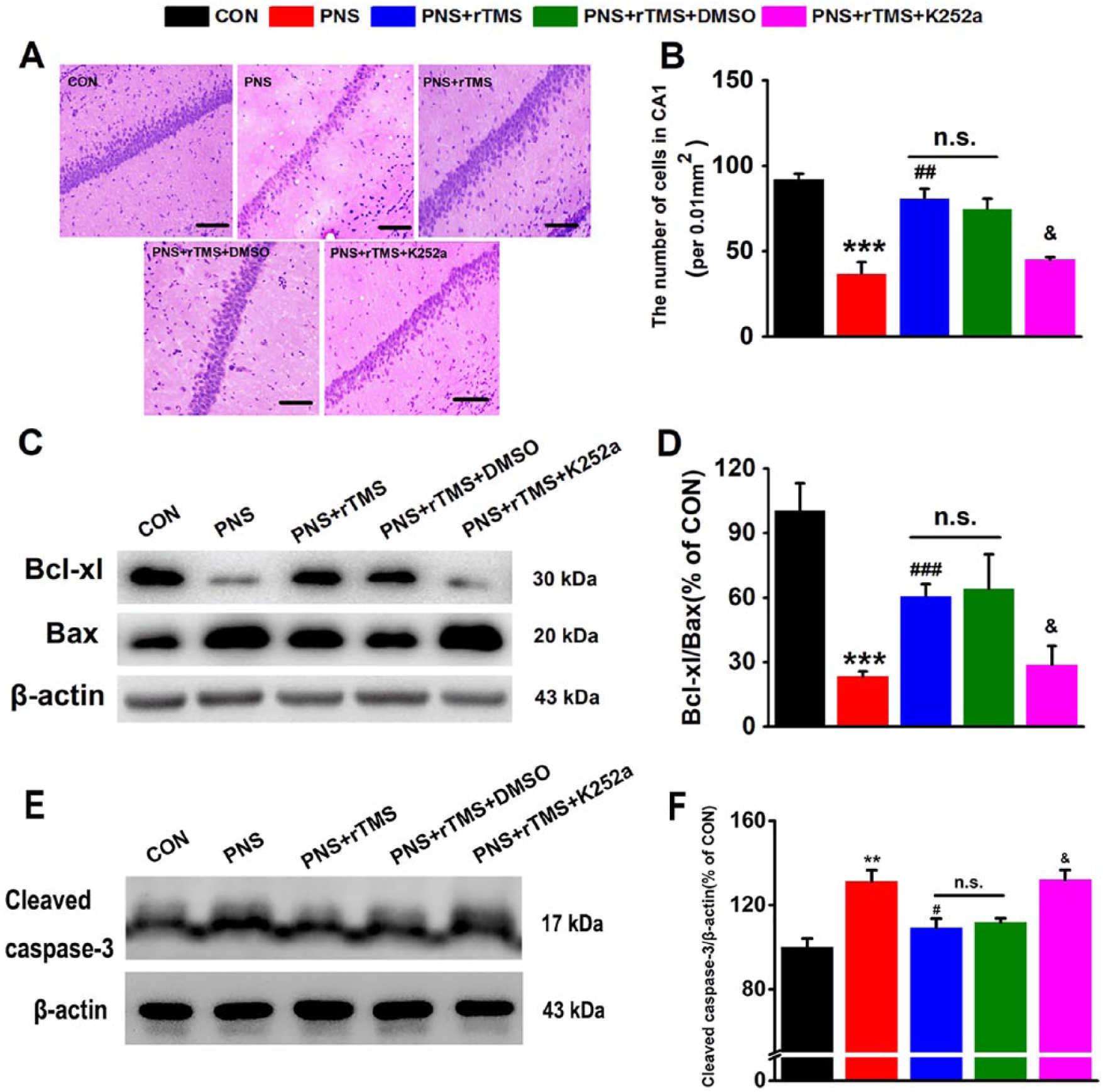
The level of apoptosis in the hippocampus in the 5 groups. (A) Photomicrograph of hematoxylin-eosin (HE) staining. Scale bar: 50 µm. (B) Comparison of the number of cells between the 5 groups. (C) Results are immunoblots from single representative experiment. (D) The expression values of the Bcl-xl/Bax. (E) Results are immunoblots from single representative experiment. (F) The values of cleaved casepase-3/β-actin expression.
The neuronal apoptosis was subsequently evaluated by measuring the levels of Bcl-xl/Bax and cleaved caspase-3. As shown in Figure 6C and E, prominent bands were identified by antibodies. It was found that there were statistical differences of Bcl-xl/Bax (F(4, 14) = 27.065, P < .001; Figure 6D) levels and cleaved caspase-3 (F(4, 14) = 10.111, P < .001; Figure 6F) among the 5 groups. There was a significant decrease of Bcl-xl/Bax ratio (P < .001) and an increase of cleaved caspase-3 (P < .01) in the PNS group compared with that in the CON group (Figure 6D and F). Furthermore, there was a significant increase of Bcl-xl/Bax ratio (P < .001; Figure 6D) and a decrease of cleaved caspase-3 (P < .05; Figure 6F) in the PNS + rTMS group compared with that in the PNS group. After treatment with K252a, the Bcl-xl/Bax ratio was significantly reduced (P < .05; Figure 6D) and the cleaved caspase-3 was statistically increased (P < .05; Figure 6F) in the PNS + rTMS + K252a group compared with that in the PNS + rTMS + DMSO group. However, there was no significant difference of either Bcl-xl/Bax ratio or cleaved caspase-3 between the PNS + rTMS group and the PNS + rTMS + DMSO group.
Discussion
During the past 2 decades, TMS has rapidly become a valuable method to noninvasively regulate and intervene the brain function. 23 In addition, rTMS is able to change and modulate activity beyond the stimulation period. 24 Therefore, rTMS has therapeutic potential in clinical and basic neuroscience research, but the long-lasting influence mechanism remains unclear. The present study provides the primary evidence that rTMS significantly alleviates cognitive deficits in the PNS offspring by promoting BDNF/TrkB signaling. The results showed that the PNS caused cognitive damages and synaptic plasticity impairments in the male offspring, and neuronal number decrease in the hippocampus. These PNS-induced behavioral and cellular deficits were well improved by rTMS. Interestingly, these beneficial effects were significantly blocked by intracerebroventricular (i.c.v.) infusion of K252a. Taken together, our results suggest that PNS causes synaptic plasticity damage and the reduction of the number of neurons in the male offspring hippocampus. Moreover, BDNF/TrkB signaling presumably plays an important role in the neuroprotective effect of rTMS.
There are several rTMS protocols with different lasting effects. Variables influencing the effects include stimulus frequency, stimulus intensity, and the total number of stimuli.25-27 For example, a previous study showed that high-frequency stimulation (>1 Hz) produced facilitatory after effects, whereas low-frequency stimulation (<1 Hz) induced inhibitory effects in the brain. 28 In the present study, we used the rTMS protocols similar to that of our previous study. 14 Interestingly, there are some evidences that PNS exerts a sex-specific effect. PNS resulted in a significant increase of BDNF expression in male offspring, but no effect in female offspring. 29 Female animals usually displayed less anxiety-like behaviors than males. 30 Moreover, male offspring were more sensitive to the PNS in cognition.31,32 In this study, only male offspring were employed, therefore, the effect of rTMS on female offspring needs to be explored in future studies. Moreover, PNS induced learning deficits and inhibited neurogenesis of dentate granule cells in both juvenile and adult offspring. 33 The differences in stressor type or exposure time might cause contrasting outcomes in offspring’s behavior test. PNS from day 15 to day 21 of gestation produced learning deficits.34,35 On the contrary, however, single and intense PNS enhanced learning performance in adolescent offspring. 36 In our study, pregnant rats were restrained thrice daily for 45 minutes from day 15 to day 21 of gestation, in which the above protocol could strongly impair learning and memory in offspring.
The MWM test was carried out to measure spatial learning and memory. The MWM performance has been strongly correlated with LTP and NMDA (N-methyl-
Long-term potentiation of synaptic transmission in the hippocampus is the synaptic basis of learning and memory in vertebrates. 39 And the hippocampal CA1 area is a more suitable location to look for learning-specific changes in excitatory postsynaptic potential (EPSP).40,41 Accordingly, the LTP from the hippocampal Schaffer collaterals to CA1 region was recorded. A higher fEPSP slope is commonly associated with more effective synaptic transmission and better learning and memory. In this study, rTMS significantly alleviated the impairment of LTP in the PNS + rTMS group compared with that in the PNS group; however, the beneficial effects were efficiently blocked by i.c.v. infusion of K252a. Interestingly, the above results are closely associated with the behavioral performance in offspring. PPF is a short-lasting (no more than 1 second) increase in the second evoked population excitatory postsynaptic potential when it follows shortly after the first, and thought to have a presynaptic locus in the hippocampus. 42 Moreover, SYP is an important presynaptic structural protein involved in synaptic vesicle formation and exocytosis, which is closely connected with synaptic plasticity.43,44 LTP is expressed both pre- and postsynaptically depending on the initial neurotransmitter release, but not regulated by the initial release probability within its first hour. 45 Our data showed that there were no significant differences of both PPF and SYP expression between those groups, suggesting that the effect of PNS on synaptic transmission and plasticity was feasibly mediated by postsynaptic mechanisms rather than presynaptic mechanisms. The NR2A and NR2B receptors are thought to be able to generate a persistent increase in synaptic strength. The data show that PNS decreases the LTP in offspring, and the effect is associated to the significant reduction of both NR2A and NR2B protein levels in the hippocampus, which is in line with those issued data. 46 However, a previous study showed that PNS did not change NR2A and NR2B in the hippocampus. 47 Overall, there is a general approval that a deficiency in NMDA receptors could impair LTP. After rTMS treatment, rats in the PNS group restored the levels of both NR2A and NR2B in the hippocampus, while the beneficial effects were blocked by i.c.v. infusion of K252a. It was reported that spatial learning could increase the glutamate release, implying that behavioral tasks probably enhanced the synaptic plasticity. 38
rTMS induces the increase of BDNF level 48 and enhances the synaptic plasticity 49 in the brain. BDNF binding to TrkB triggers autophosphorylation of tyrosine residue, and then leads to the phosphorylation of CREB, which is blocked by K252a as an inhibitor of receptor tyrosine kinases. 49 The TrkB produces 2 different types of BDNF receptor by alternatively splicing. The first type is the full-length TrkB (~135-145 kDa) and the second one is the truncated TrkB (~90 kDa). In the hippocampus, there is a pronounced increase in the level of truncated TrkB and little change in the level of the full-length TrkB accompanied by developmental processes. Meanwhile, truncated TrkB is co-expressed with catalytically active Trks in certain neuronal populations, including hippocampal pyramidal neurons, dentate granule cells, and neocortical neurons. 50 In the present study, the truncated TrkB was identified in the male offspring hippocampus. The inhibition of p-CREB decreased the transcription of the BDNF regulated by the Ca2+ influx. 51 NMDA activation can induce Ca2+ influx, then trigger a phase of CREB phosphorylation mediated by a family of kinases, such as PKA and CaMKII, which is important to the development and synaptic plasticity.52,53 CREB-knockout mice have deficiencies in spatial learning and memory, and the LTP is markedly attenuated in the hippocampus. 54 In addition, the long-lasting BDNF-induced enhancement of LTP was blocked by K252a in the normal adult hippocampus. 55 Indeed, K252a did not affect the LTP in the brain slices of adult normal hippocampus. 56 In the present study, the K252a was injected 24 hours after the end of the rTMS to modulate the BDNF-TrKB signaling in the PNS + rTMS group. Our data show that rTMS significantly improves the impairment of the hippocampal synaptic plasticity and spatial cognition possibly associated with regulating BDNF/TrkB signaling. Moreover, we do understand that the regulation of BDNF/TrkB signaling with K252a is only one of the underlying mechanisms. Further studies are required to determine the mechanism of BDNF/TrkB signaling in PNS offspring. It means that the K252a does not actually block the rTMS itself but rather affects the maintenance of rTMS.
Bcl-2 and its related family member Bcl-xl inhibit both caspase-dependent and caspase-independent apoptosis. The balance between Bcl-2 and Bax plays a key role in apoptosis and is regarded as an important target responsible for the treatment of psychiatric disorders. 4 Meanwhile, blocking the BDNF-TrkB signaling by K252a may decrease the transcription of prosurvival factors including Bcl-2, and then activate apoptosis pathway. Several previous studies showed that stress decreased the expression of antiapoptosis proteins, such as Bcl-2 and BAG-1, and increased the apoptosis proteins, such as Bax and caspase-3.57,58 In this study, PNS reduced the number of cells and Bcl-xl/Bax ratio, and increased the cleaved caspase-3 in the hippocampus; however, the effect was reversed by using rTMS treatment. Furthermore, the effect of rTMS treatment was effectively weakened by K252a, suggesting that rTMS prevented the reduced cells that were associated with BDNF/TrkB signaling. However, since K252a could induce cell apoptosis, 59 the less number of cells is likely to be a mixture of less production and greater loss.
The present study revealed the effect of rTMS on spatial cognition in PNS male offspring, as well as the possible molecular mechanism of rTMS. However, we do understand that there is only one stimulation pattern of rTMS used in the study, which is possibly not enough to draw a solid conclusion.
Conclusions
Our results suggest that 5-Hz rTMS significantly improves the impairment of hippocampal synaptic plasticity and prevents the reduced cells, which was possibly associated with activating BDNF/TrkB signaling pathway, and further ameliorates the spatial cognitive deficits induced by PNS in the male offspring.
Supplemental Material
Supplementary_Materials – Supplemental material for rTMS Ameliorates Prenatal Stress–Induced Cognitive Deficits in Male-Offspring Rats Associated With BDNF/TrkB Signaling Pathway
Supplemental material, Supplementary_Materials for rTMS Ameliorates Prenatal Stress–Induced Cognitive Deficits in Male-Offspring Rats Associated With BDNF/TrkB Signaling Pathway by Yingchun Shang, Xin Wang, Fangjuan Li, Tao Yin, Jianhai Zhang and Tao Zhang in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
TZ and TY conceived and designed the experiment.
YS, XW, and FL performed the experiments.
YS, XW, and JHZ analyzed the data.
YS and TZ wrote the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (31771148 to TZ; 81127003 to TY; 61633010 to JZ), and 111 Project (B08011 to TZ).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
