Abstract
Background. The residual hemiparesis after stroke results in a unilateral reduction in propulsive force during gait. Prior work has suggested the presence of a propulsive reserve in the paretic limb. Objective. The purpose of this study was to quantify the paretic propulsive reserve in individuals poststroke and to determine the biomechanical mechanism underlying the generation of additional paretic propulsive limb force. Methods. Ten individuals with chronic hemiparesis poststroke walked on a treadmill against an impeding force (ascending 0% to 10% body weight [BW], in 2.5% BW increments, followed by descending 10% to 0% BW, also in 2.5% BW increments) applied to the body’s center of mass. The resulting propulsive forces were measured bilaterally and compared between impeding force levels. We then assessed potential mechanisms (trailing limb angle and plantarflexion moment) underlying the changes in propulsion. Results. Overall, peak paretic propulsive force increased by 92% and the paretic propulsive impulse increased by 225%, resulting in a significant increase in the paretic limb’s contribution to propulsion. Participants continued to produce increased paretic propulsion on removal of the impeding force. The trailing limb angle contributed significantly to the increase in paretic propulsion, whereas the plantarflexion moment did not. Conclusions. Participants exhibited a robust propulsive reserve on the paretic limb, suggesting that there is untapped potential that can be exploited through rehabilitation to improve gait recovery. The increase in propulsive symmetry indicates that a greater response was observed by the paretic limb rather than increased compensation by the nonparetic limb.
Introduction
Individuals with chronic hemiparesis following stroke are limited by slow, asymmetrical gait, and limited endurance. 1 Slow gait speed is due, in part, to the inability to generate adequate propulsive force with the paretic limb,2,3 which is essential for propelling the body forward during gait.4,5 Although reductions in paretic propulsion are often reported,2,6 there is evidence that individuals recovering from stroke contain a reserve in propulsive forces during gait. For example, the existence of a robust walking speed reserve7-9 and voluntary muscle activation deficits10,11 both suggest that greater limb propulsion may be available. In fact, greater paretic propulsion has been demonstrated during a long-step walking task with the nonparetic leg, 12 while walking faster, 13 and while walking with visual feedback of paretic propulsive forces. 14
Propulsive reserves are well known to occur in unimpaired older adults, 15 but unlike unimpaired older adults, the presence of unilateral deficits following stroke indicates that the nonparetic limb may simply compensate for the reduced paretic propulsion.2,13 Indeed, the nonparetic limb produces net positive work on the body’s center of mass (COM) to compensate for the net negative work produced by the paretic limb. 6 Furthermore, the asymmetry in propulsive forces observed at self-selected walking speeds2,6 does not change when individuals poststroke walk at faster speeds. 16 Instead, the nonparetic limb appears to produce greater propulsive forces to allow for the transition from comfortable to fast walking speeds. 13 Nevertheless, recent evidence is suggestive of a propulsive reserve in individuals poststroke.12,17 Understanding the ability of the paretic limb to increase propulsive force volitionally is therefore important for promoting gait recovery poststroke.
Propulsion during the latter half of stance is heavily influenced by limb mechanics. 18 Specifically, the force generated by the plantarflexors, and manifest as an ankle plantarflexion moment, supplies the majority of the limb force during the propulsive phase of gait.19,20 Additionally, the angle at which the limb force is applied influences the force’s effect on the body’s COM. 21 For example, a large ankle plantarflexion moment provided with the leg directly below the pelvis will simply propel the body up, without generating any forward acceleration. The effective angle of the horizontal force can therefore be simplified as the trailing limb angle (TLA). 18 The fact that both TLA 22 and propulsion 23 increase with faster gait speeds, however, confounds the question of how TLA influences propulsion in the absence of a change in gait speed.24,25
The purpose of this study was to seek the magnitude of a propulsive reserve in individuals poststroke and to determine the mechanism underlying the generation of any additional paretic propulsive limb force. We hypothesized that individuals poststroke would exhibit the ability to produce greater propulsion with their paretic limb in response to a restraining force to the body’s COM. 26 Furthermore, we anticipated that the paretic limb would exhibit both a greater TLA and plantarflexion moment to produce the increase in paretic propulsion.18,24
Methods
Participants
We recruited 10 individuals (see Table 1) with chronic hemiparesis (time poststroke 105 ± 127 months) to participate in this study. Participants were included if they had a unilateral brain lesion due to stroke; ability to walk ⩾5 minutes on a treadmill with only handrail assist for balance; and the ability to walk ⩾10 m overground without an assistive device. Potential participants were excluded if they required an ankle-foot orthosis for ambulation, or self-reported any musculoskeletal, cardiorespiratory, metabolic, or other neurological disorder that could interfere with gait. All participants signed an informed consent form approved by the Institutional Review Board of the University of North Carolina at Chapel Hill.
Participant Demographics.
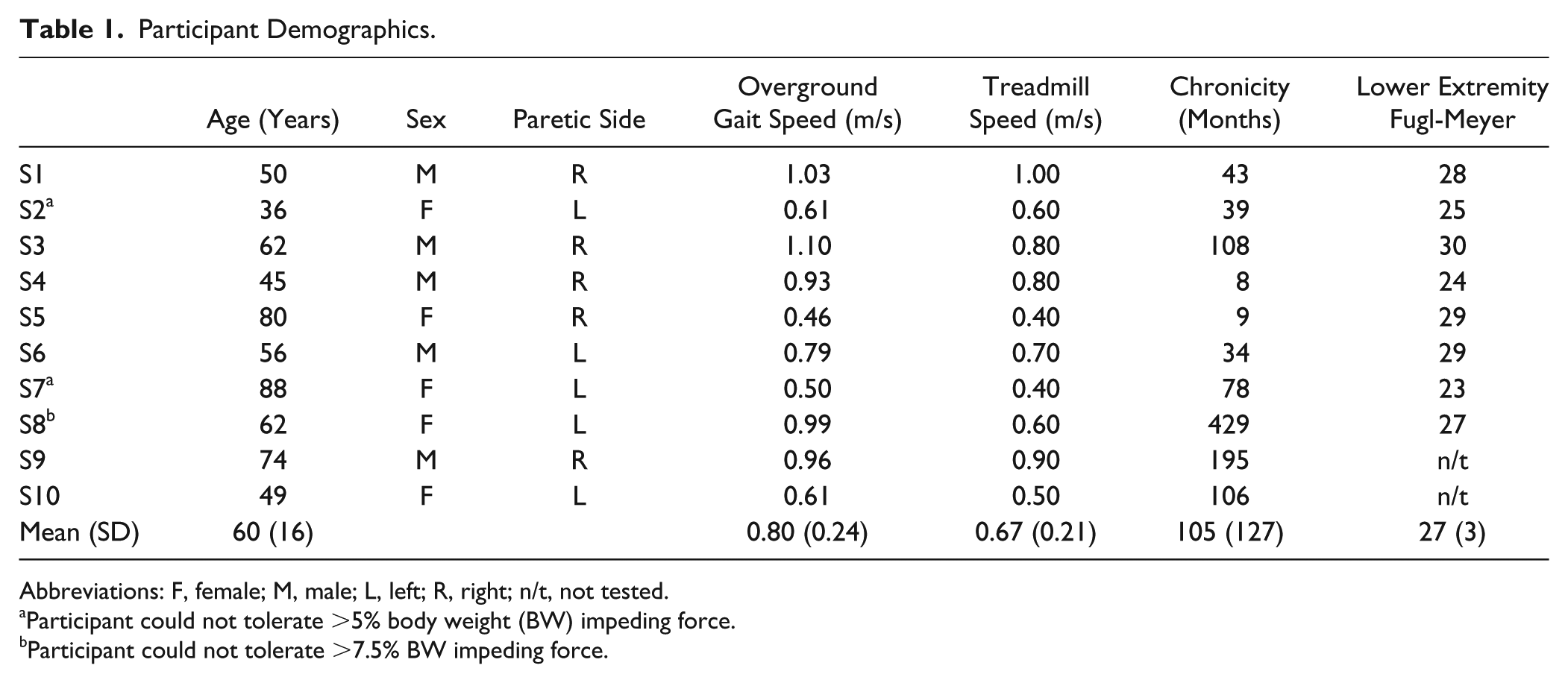
Abbreviations: F, female; M, male; L, left; R, right; n/t, not tested.
Participant could not tolerate >5% body weight (BW) impeding force.
Participant could not tolerate >7.5% BW impeding force.
Data Collection
Prior to data collection, each participant’s sensorimotor coordination was evaluated with the lower extremity portion of the Fugl-Meyer assessment. 27 Comfortable overground walking speed was measured as the average of 2 passes on a 20-foot pressure sensitive walkway (Zeno; ProtoKinetics, Havertown, PA). 28 Participants completed all subsequent testing on an instrumented dual-belt treadmill (Bertec, Worthington, OH) while walking at their comfortable treadmill walking speed. We attempted to have participants walk at their comfortable overground speed; however, the physical challenge associated with testing prohibited this in most cases. While walking on the treadmill, all participants wore a safety harness that was attached overhead. Although the harness was available to prevent a fall, no body weight support was provided. Participants were allowed to use a side handrail, if needed.
While walking at a constant speed, a posterior restraining force was applied to the pelvis, which is thought to approximate the body’s COM. 26 The restraining force was provided via a stretched elastic band (TheraTubing, Theraband, Akron, OH) attached to a standard gait belt positioned around the pelvis. We monitored the magnitude of the restraining force using a load cell (MLP-150; Transducer Technique, Temecula, CA) to ensure consistent loads. Participants were instructed to resist the force by maintaining their position in the middle of the treadmill. We applied loads in an ascending stepwise manner, from 0% to 10% of the participant’s body weight (BW), in 2.5% increments (Figure 1). At each force level, the restraining force was applied for ~20 seconds. No rest breaks were provided during testing. The pull-force was then removed as a descending step function using the same loads.
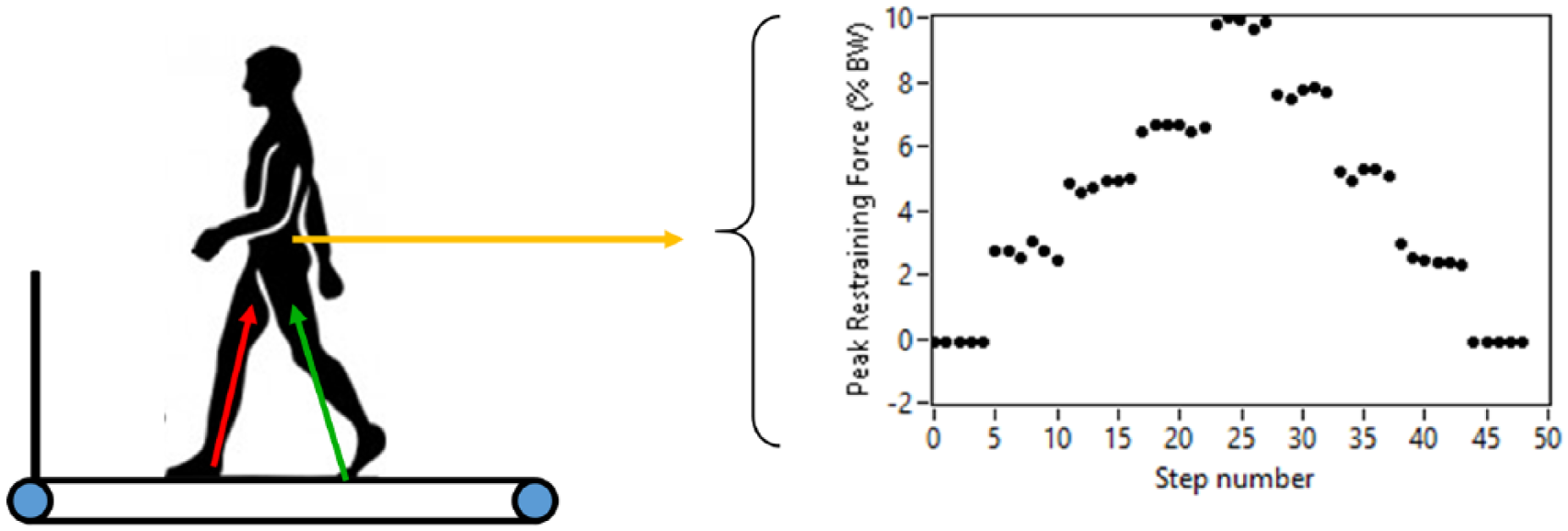
Schematic representation of testing setup. An elastic therapy band was used to provide a posterior restraining force. The restraining force was applied in a stepwise manner from 0% to 10% body weight (BW) in 2.5% BW increments and then decreased in the reverse manner. Concurrently, we recorded ground reaction forces to compute paretic and nonparetic propulsive forces.
Prior to walking, 14-mm retroreflective markers were placed on both limbs and the pelvis, as described previously.23,29 While walking, we recorded the 3-dimensional positions of each marker using an 8-camera motion capture system (Vicon, Los Angeles, CA) recording at 120 Hz. Concurrently, ground reaction forces and the pull force were recorded at 1200 Hz.
Data Management and Analysis
Marker trajectories and ground reaction forces were low-pass filtered with a fourth-order, 6- and 20-Hz Butterworth filter, respectively. Joint moments were computed using inverse dynamics (Visual3D; C-Motion, Germantown, MD). Custom software (Labview, National Instruments, Austin, TX) was used to calculate the average pull force throughout the gait cycle, the peak paretic propulsion, the propulsive impulse, as well as the plantarflexion moment and the TLA at peak propulsion for each step. The TLA was computed as the angle formed by a vertical line and a line connecting the pelvis COM and the center of pressure. Peak propulsion was measured as the peak of the anteriorly directed ground reaction force and the propulsive impulse is the integral of the anteriorly directed ground reaction force. The paretic limb’s contribution to propulsion was calculated as a ratio of the paretic limb’s propulsive force divided by the sum of the paretic and non-paretic limb’s force. 2 Thus, a value of 0.5 represents symmetric propulsion. Because both the peak propulsion and the propulsive impulse provide different information, 30 the ratio was calculated separately using both measures. Finally, we computed cadence as the inverse of step time, and calculated paretic and nonparetic step lengths as the anteroposterior distance between the heels at heel strike. Within each subject, each variable was then averaged separately at each force level on the ascending section as well as the descending section.
Data were analyzed using SPSS (v24; IBM Corp, Armonk, NY). Data were checked for normality using the Shapiro-Wilk test. We then used a repeated-measures analysis of variance (repeated for pulling force magnitude) to determine differences in each outcome measure between the different magnitudes of pull force. When we observed significant main effects, we carried out several planned post hoc comparisons using paired samples t tests. This approach allowed us to retain data from all subjects, including three participants who could not tolerate the 10% BW impeding force. Specifically, we compared the initial 0% condition (ie, baseline) to each value on the ascending section, as well as compared each matched condition from the ascending and descending condition (eg, initial 0% compared with final 0%; ascending 2.5% compared with descending 2.5%, and so on). Next, we sought to determine the mechanical mechanisms underlying the paretic propulsion reserve (eg, difference between initial 0% BW and highest tolerated impeding force conditions) and any residual paretic propulsion on removal of the impeding force (eg, initial 0% BW and final 0% BW impeding force conditions). Specifically, we performed Pearson correlation analyses to determine how the changes in plantarflexion moment and TLA related to the changes in peak propulsion of the paretic limb.
Results
Three of the 10 subjects were unable to tolerate 10% BW impeding force. Two participants ascended to 5% BW, and 1 participant ascended to 7.5% BW before descending back to 0% BW. Those subjects are noted in Table 1. The actual average impeding force provided throughout the gait cycle was 2.3% ± 0.4%, 4.5% ± 0.4%, 7.0% ± 0.4%, and 9.1% ± 0.3% BW for the ascending section (ie, 2.5%, 5.0%, 7.5%, and 10.0% BW) conditions. The only difference between the ascending and descending sections was observed at the 5% BW condition (4.9% ± 0.6% BW on the descending section; P = .006; d = 1.12).
Propulsive Mechanics
Both the paretic (P < .001;
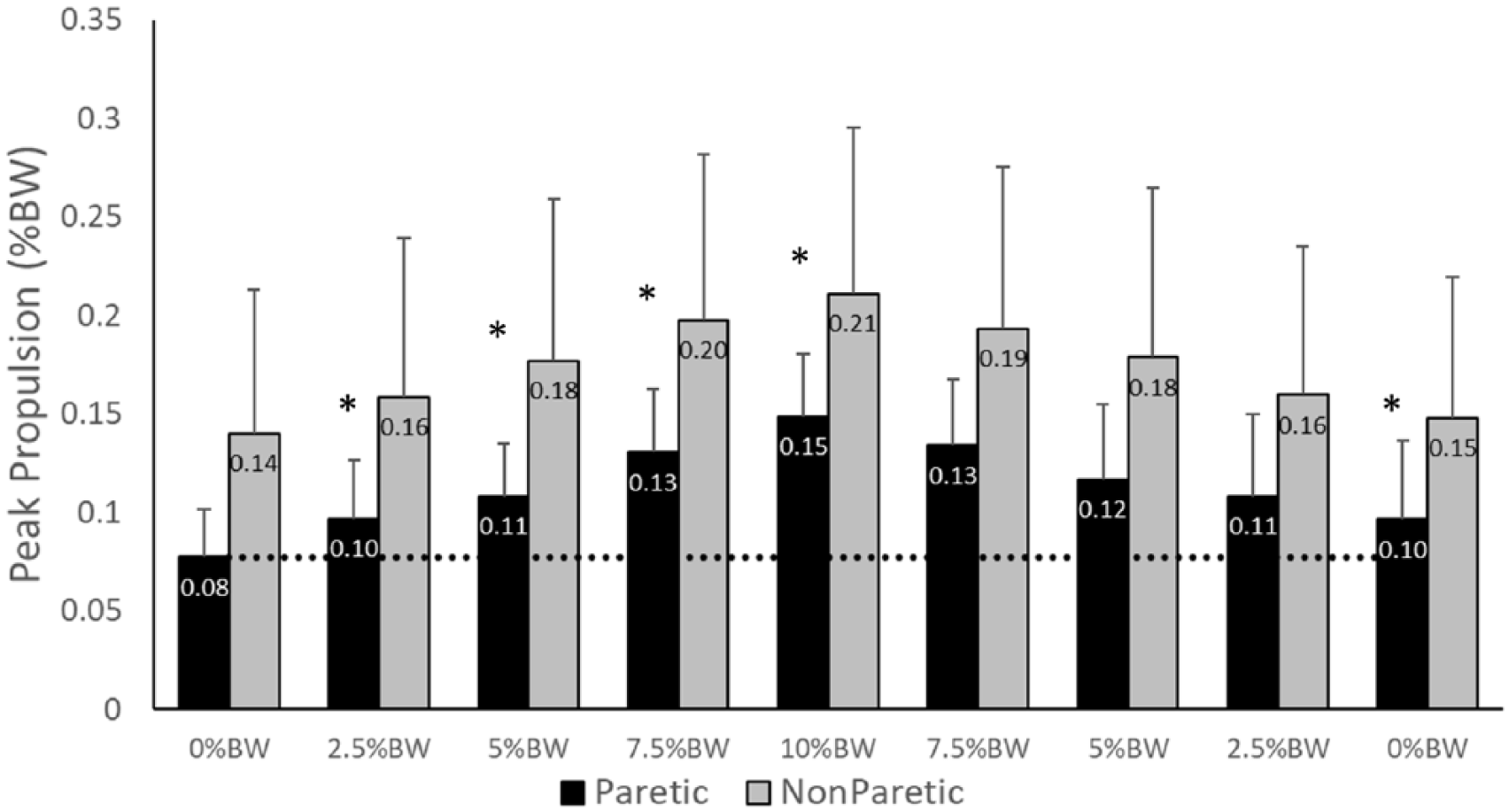
Peak propulsion forces from the paretic (black) and nonparetic (gray) limbs. Note the significant increase in propulsive force on both the paretic and nonparetic limbs in response to an impeding force. The propulsive force produced by the paretic limb remained elevated during the final 0% body weight (BW) condition. Values represent the group mean and SD. *Indicates a significant difference from the initial 0% BW condition at the P < .05 level.
The paretic (P < .001;
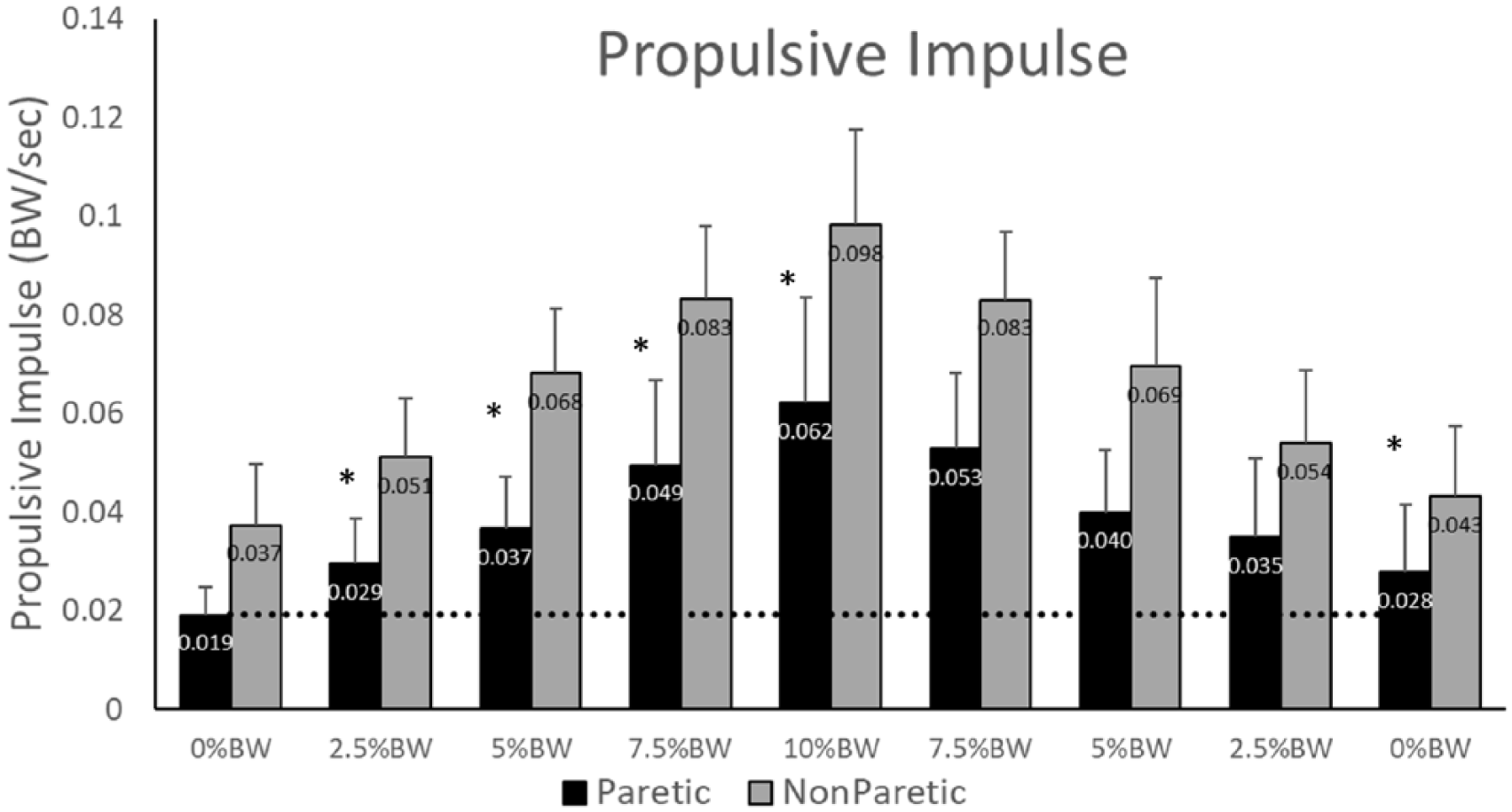
Propulsive integral for the paretic (black) and nonparetic (gray) limbs. Note the significant increase in integral on both the paretic and nonparetic limbs in response to the impeding force. The propulsive integral produced by the paretic limb remained elevated during the final 0% body weight (BW) condition. Values represent the group mean and SD. *Indicates a significant difference from the initial 0% BW condition at the P < .05 level.
Propulsive Asymmetry
The paretic propulsion ratio based on peak values was significantly influenced by the impeding force (P = .042;
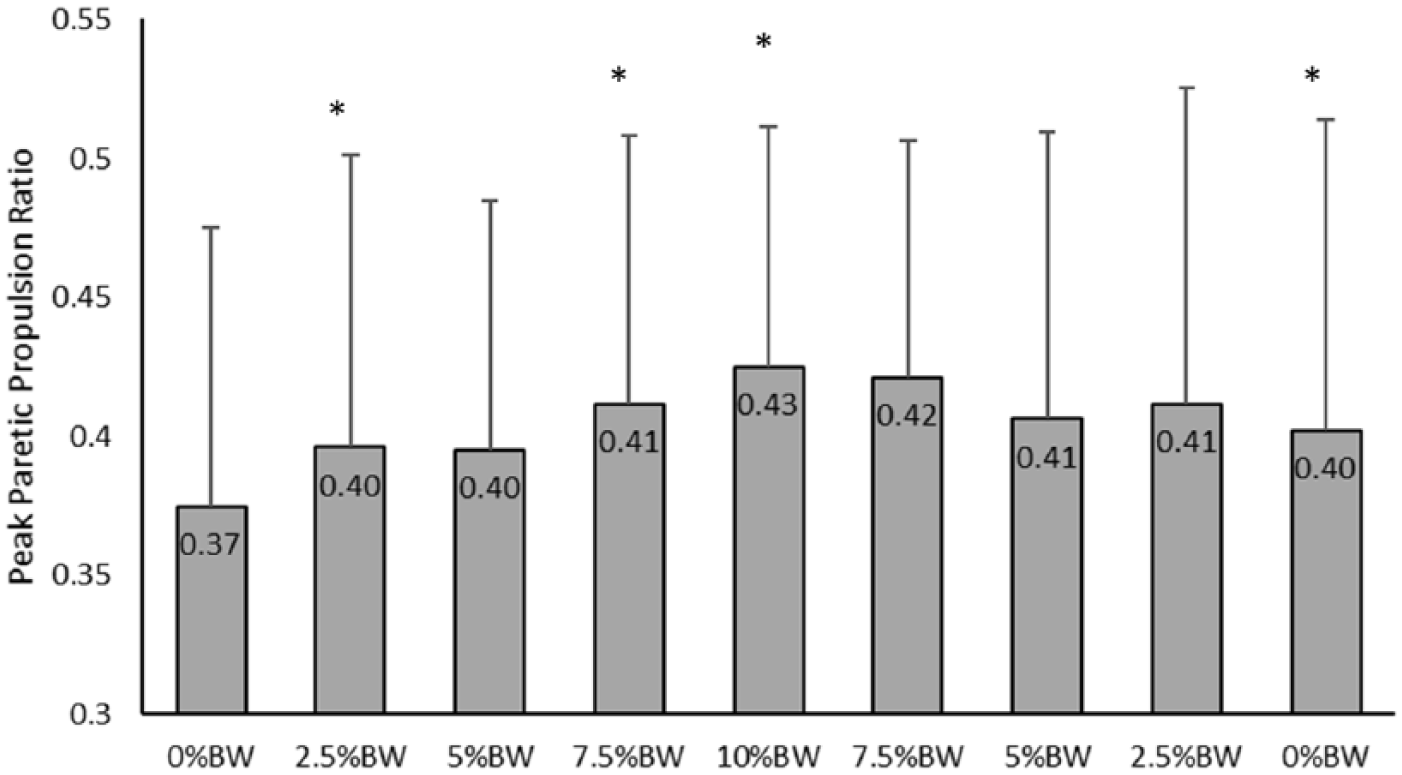
Paretic contribution to propulsion presented as a ratio of peak paretic propulsion to the sum of paretic and nonparetic propulsive peaks. Values represent group mean and SD. *Indicates a significant difference from the initial 0% body weight (BW) condition at the P < .05 level.
Mechanical Contributions to Propulsive Reserve
We performed regression analyses to provide insight into the mechanisms (eg, TLA and plantarflexion moment) underlying the increased propulsive peaks observed during the 10% BW impeding force and the final 0% BW impeding force conditions. In both cases, the increased TLA was significantly related to the increased paretic propulsive peak. Specifically, 76% of the variance in the increased peak paretic propulsion during the 10% BW impeding force condition was attributed to the change in the TLA (P = .011; R2 = 0.760; Figure 5). Likewise, the TLA explained 61% of the variance in peak paretic propulsion change between the initial and final 0% BW conditions (P = .013; R2 = 0.612). In both cases, the change in the paretic limb’s ankle plantarflexion moment did not correlate to change in peak paretic propulsion (P > .062).
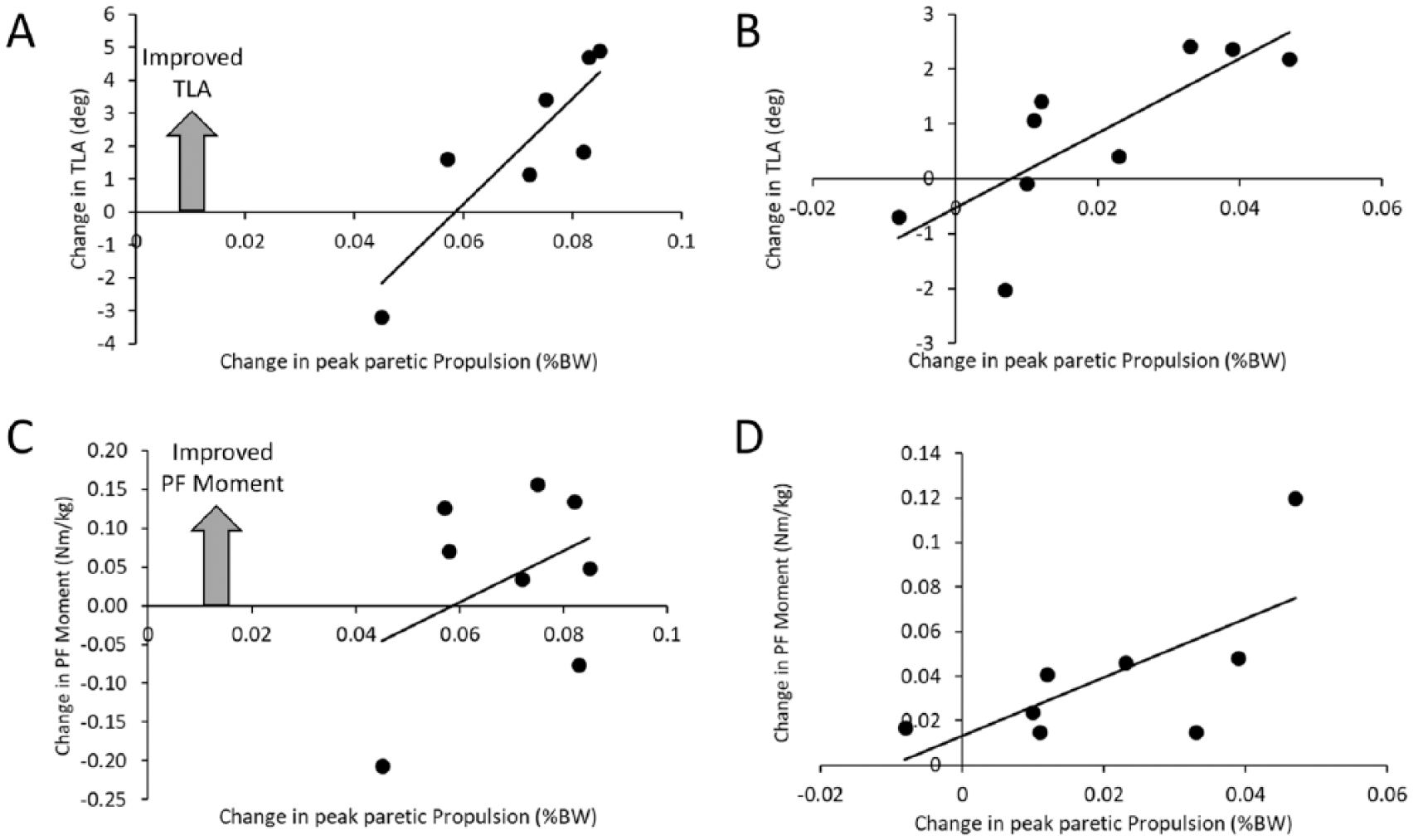
The relationship between the change in peak paretic propulsion from the initial 0% body weight (BW) condition and the 10% BW condition (A and C) with the change in trailing limb angle (TLA) (A) and plantarflexor (PF) moment (C). The change in propulsive force noted at the final 0% BW condition (B and D) was plotted with the change in TLA (B) and PF moment (D). Note that the change in the TLA is significantly correlated with the change in peak paretic propulsion, whereas the change in PF moment was not.
We observed that the step lengths of the paretic limb were affected by the impeding force (P = .007;
Discussion
Our hypothesis that participants with chronic hemiparesis would exhibit a propulsive reserve on the paretic limb was supported by these data. In particular, we observed a robust increase in paretic propulsive forces (eg, 92% increase) and this increased force persisted on removal of the impeding force. Additionally, our hypothesis that mechanical contributions could explain the paretic propulsive reserve were only partially supported by these data. The observed increase in paretic propulsion was attributed to an increase in the TLA, with no significant contribution from the change in ankle plantarflexor moment. We have contributed to the growing body of literature supportive of an underutilized paretic propulsive force in individuals poststroke, along with providing accompanying insight into the mechanical mechanism underlying the manner by which participants were able to exploit the reserve.
We have quantified the presence of a robust and underused propulsive reserve in the paretic limb of individuals recovering from stroke. This exciting finding suggests that the propulsive reserve can be exploited using rehabilitative techniques to augment gait for individuals following stroke. These data build on prior work that was suggestive of a propulsive reserve, 31 to clearly document the individual response of the paretic and nonparetic limbs to an impeding force. Of importance, is that the increased propulsive response occurred immediately and without training. Prior work has demonstrated increased propulsion by the paretic limb after weeks of training, 13 or has been confounded by changing gait speed, which is closely related to propulsive forces. 23 That participants could elicit such an immediate response, without training, suggests that the available propulsive force can be activated volitionally. In particular, prior work has shown that both individuals poststroke 14 and unimpaired older adults 15 can manipulate propulsive limb forces using visual feedback. Our findings provide further support for the ability of individuals poststroke to augment paretic propulsive forces.
The additional propulsive force that our subjects were able to access was maintained following removal of the impeding force. Notably, we observed that during the final testing condition, participants retained significantly greater propulsive force with the paretic limb, yielding greater propulsive symmetry. It is entirely possible that the retained propulsive force is the result of locomotor adaptation; however, there are several notable differences between our findings and those of classic gait adaptation studies. First, the period of exposure to each impeding force was relatively short (~20 seconds). Most locomotor adaptation paradigms employee a 15- to 30-minute exposure to the altered environment,32-34 which is substantially longer then what we exposed our participants to. Second, gait-related aftereffects are typically washed out within a few steps.32,33,35 Instead, it appears that our participants continued to produce greater propulsion forces for the duration of the 20-second condition, beyond what we would typically associate with an adaptive aftereffect. This suggests that the participants were gaining some benefit from the increase in anteriorly directed force production, although it is not clear if this benefit was perceived either implicitly or explicitly by the participants. Additionally, we only recorded the final condition for 20 seconds and therefore, we are unaware of the duration of the increased propulsive force. Finally, all testing was performed on the treadmill and therefore we are unaware of whether the additional propulsive force transfers to the overground environment. 36
Although both the paretic and nonparetic limbs increased propulsive force, the paretic limb increased to a greater extent than the nonparetic limb. This is surprising, given the deficits encountered. Mechanical gait asymmetries are common following stroke2,6,19 and have been related to hemiparetic severity. 2 Given the ability of the nonparetic limb to compensate for the weakened paretic limb2,13 it is notable that the paretic limb is able to adjust to produce a greater component of the body’s propulsive force. Interventions that encourage greater contributions from the paretic limb allow for greater recovery of function, rather than continued compensation by the stronger nonparetic limb.
Despite prior work suggesting that the magnitude of paretic propulsive forces can be manipulated by both the TLA and plantarflexion moment,18,24 our data suggest that changing the plantarflexion moment did not contribute to an increase in the propulsive force. Although Figure 5D seems to indicate a tendency for the change in plantarflexion moment to contribute to the change in propulsion, this finding was driven by one outlier. Instead, we observed that participants increased the TLA, which led to the observed increase in propulsion. Loss of hip extension, a critical component of the TLA, is often reduced during gait after stroke.37,38 Although this may be due to restricted joint motion 39 there may also be contributions related to disrupted selective motor control 40 and muscle weakness. 41 Nevertheless, participants implicitly chose to increase the TLA, rather than the plantarflexion moment to provide the additional needed paretic propulsion. We can speculate that participants did not have the ability to further increase the plantarflexion moment volitionally.19,42 Consequently, the primary means to increase propulsion was through the TLA. 18 The interaction between TLA and plantarflexion moment in modulating propulsion has important implications for rehabilitation. Given the weakness in distal plantarflexor muscles,42,43 attempts to provide additional plantarflexion moments have been undertaken.44,45 These efforts have attained only moderate success, perhaps due to the inability to increase TLA concurrently. 46 Clearly, if the TLA remains reduced or decreases further, the result will be an ineffective pushoff force by the plantarflexors. Thus, solely targeting plantarflexor function appears to be ineffective. 44 Instead, attempts to concurrently increase the plantarflexor moment and TLA have shown greater success. 47
There are several limitations associated with this work. Notably, we tested a small number of participants for this study. Although others have used comparable sample sizes to ask related questions,12,14,15 our small sample size could limit generalizability. This is particularly true in that our participants were fairly high-functioning. Given the inherent challenge associated with the impeding force, we felt safer recruiting faster walkers. We recognize that this may have influenced our results as slower walkers may not demonstrate the same capacity to increase paretic propulsive force, or they may use a different method of increase paretic propulsion. In the future, the presence of a paretic propulsive reserve should be calculated in slower walkers as different mechanisms may underlie propulsive changes.
In summary, we observed a robust propulsive reserve in the paretic limb of individuals recovering from stroke. This additional force was elicited by increasing the TLA of the paretic leg. Importantly, the additional force persisted at the end of testing and yielded more symmetric propulsion. These findings have important implications for rehabilitation as they are suggestive of an untapped reserve that can be exploited through novel interventions14,15 that could enhance walking recovery.
Footnotes
Acknowledgements
The authors acknowledge Yunna Sinskey, MD, for assistance with data collection.
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported by the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health, through Grant Award Number UL1TR002489.
