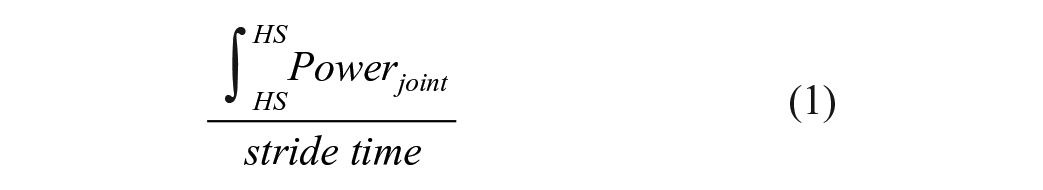Abstract
Background and Purpose. Previous studies suggest that individuals poststroke can achieve substantial gains in walking function following high-intensity locomotor training (LT). Recent findings also indicate practice of variable stepping tasks targeting locomotor deficits can mitigate selected impairments underlying reduced walking speeds. The goal of this study was to investigate alterations in locomotor biomechanics following 3 different LT paradigms. Methods. This secondary analysis of a randomized trial recruited individuals 18 to 85 years old and >6 months poststroke. We compared changes in spatiotemporal, joint kinematics, and kinetics following up to 30 sessions of high-intensity (>70% heart rate reserve [HRR]) LT of variable tasks targeting paretic limb and balance impairments (high-variable, HV), high-intensity LT focused only on forward walking (high-forward, HF), or low-intensity LT (<40% HRR) of variable tasks (low-variable, LV). Sagittal spatiotemporal and joint kinematics, and concentric joint powers were compared between groups. Regressions and principal component analyses were conducted to evaluate relative contributions or importance of biomechanical changes to between and within groups. Results. Biomechanical data were available on 50 participants who could walk ≥0.1 m/s on a motorized treadmill. Significant differences in spatiotemporal parameters, kinematic consistency, and kinetics were observed between HV and HF versus LV. Resultant principal component analyses were characterized by paretic powers and kinematic consistency following HV, while HF and LV were characterized by nonparetic powers. Conclusion. High-intensity LT results in greater changes in kinematics and kinetics as compared with lower-intensity interventions. The results may suggest greater paretic-limb contributions with high-intensity variable stepping training that targets specific biomechanical deficits. Clinical Trial Registration. https://clinicaltrials.gov/ Unique Identifier: NCT02507466
Keywords
Introduction
Restoration of locomotor function poststroke is a primary goal of rehabilitation, although reduced walking speeds and abnormal gait patterns often persist for years postinjury. Previous studies suggest specific locomotor training (LT) parameters, including the amount and cardiovascular intensity of task-specific (stepping) practice, can enhance walking speeds.1,2 Additional research also demonstrated that LT focused on practice of variable stepping tasks directed toward the primary biomechanical subcomponents of upright locomotion that contribute to forward progression (limb swing, propulsion, stance control, or dynamic stability3-6) results in greater gains in gait speed and endurance than typical interventions.7,8 A recent study 9 indicated that high-intensity LT performed in variable contexts with focus on these biomechanical subcomponents, but without attempts to normalize kinematics, resulted in gains in gait speed or dynamic stability as compared with variable, low-intensity LT, or high-intensity LT with limited variability, respectively.
Despite these gains, the biomechanical strategies used by patients poststroke to achieve faster speeds can be concerning for clinicians and patients. For example, asymmetrical limb-loading or altered kinematic patterns are often used to compensate for paretic-limb deficits, which can be energetically inefficient 10 and precipitate musculoskeletal injury. 11 Traditional rehabilitation theories12,13 discourage use of compensatory patterns during rehabilitation, and conventional interventions often focus on minimizing neurological deficits (i.e. strength and balance), and normalizing kinematics using therapist assistance 14 or robotic assistance. 15 Such therapeutic activities are thought to enhance neurological recovery, defined as restoration of previous neurological function to allow movement patterns similar to able-bodied individuals. 16 However, the efficacy of these strategies to improve specific measures of function or neurological recovery are limited. Nonetheless, a major concern of performing high-intensity LT without focus on kinematic patterns is that such practice may result in and reinforce compensatory strategies with repeated training,12,13 or alternative movement strategies in presence of residual neurological recovery.
An alternative theory is that practice of challenging stepping tasks that require altered volitional commands may result in neuromuscular adaptations that can contribute to enhanced functional and neurological recovery. A large body of literature indicates that individuals with or without neurological injury adapt their neuromuscular strategies in response to various biomechanical or environmental demands that perturb their typical movement patterns.17,18 For example, split-belt treadmill walking paradigms have been utilized in individuals with step length asymmetry poststroke to induce perturbations that increase this asymmetry (ie, magnify errors), although immediate and long-term adaptations result in improved spatiotemporal symmetry with removal of the perturbation.19-21 However, patients poststroke demonstrate deficits beyond gait asymmetry that contribute to reduced locomotor function, including difficulty with propulsion, limb swing, stance control, and dynamic stability. 7 Pilot studies of high-intensity LT in variable contexts that attempt to address these deficits during practice of challenging stepping tasks has resulted in gains in paretic and nonparetic limb kinematics and kinetics that could be characterized as indicator of recovery and compensation.7,22 Whether such training elicits differential changes in movement strategies as compared with other LT strategies, including interventions not focused on specific biomechanical subcomponents of walking or practice at lower intensities, is not clear.
The goal of this study was to investigate training-induced changes in locomotor kinematics and kinetics following 3 different LT paradigms. In this secondary analysis from a previous randomized clinical trial, 9 we compared changes in spatiotemporal, kinematic, and kinetic variables during walking trials following up to 30 sessions of high-intensity variable (HV) training, low-intensity training in variable contexts (low-variable, LV), or high-intensity training focused only on forward walking (high-forward, HF). Gains were analyzed between groups using standard analyses of variance (ANOVAs) and within groups using principal component (PC) analyses to ascertain the subset of biomechanical variables that best explain the variance of in each training group. With differences in gains in gait speeds with high-intensity training, we anticipated greater improvements in kinetics following either HV or HF versus LV due to greater neuromuscular activation strategies required during high-intensity training. Furthermore, given the attention to specific biomechanical deficits targeted during variable training paradigms, we hypothesized greater changes in specific measures of paretic-limb kinematics and kinetics following HV versus HF training. The results from this study may provide insight into whether attention to specific biomechanical deficits during training may ameliorate abnormal movement strategies in patients poststroke.
Methods
Participants
Individuals with chronic (>6 months) hemiparesis poststroke were recruited, with specific inclusion criteria as follows: 18 to 85 years old; lower-extremity Fugl-Meyer <34; overground self-selected velocity (SSV) <1.0 m/s; and medical clearance to participate. Exclusion criteria included presence of lower extremity contractures that significantly limited range of motion, significant osteoporosis, cardiovascular, respiratory, or metabolic instability, inability to ambulate >150 feet prior to stroke, previous history of peripheral or central nervous system injury, and inability to adhere to study requirements. An additional requirement for evaluation of gait biomechanics during graded treadmill (TM) testing was the ability to walk for ≥1 minute at 0.1 m/s with the use of handrails as needed. All participants gave written informed consent and all procedures were approved by the local institutional review board.
Experimental Intervention
Participants were randomized to receive up to 30 one-hour sessions of either HV, HF, or LV training over 2 months, with up to 40 minutes of stepping per session. Primary training goals for all groups were to (1) maximize the amount of successful stepping practice, (2) achieve targeted cardiovascular intensities, and (3) increase difficulty of walking tasks as tolerated. Targeted heart rate (HR) ranges were determined using age-predicted maximum [208 − (0.7 × age)], 23 with HV or HF targeting 70% to 80% HR reserve, and LV using 30% to 40% HR reserve. 9
For HV training, sessions were divided into ~10-minute bouts, including speed-dependent TM training, skill-dependent TM training, overground training, and stair climbing. Speed-dependent TM training was performed with an overhead harness system in case of loss of balance, with goals to increase speeds to reach targeted intensities. Criteria for successful stepping included positive bilateral step lengths, minimal limb collapse, and maintaining upright posture in the sagittal and frontal planes. 7 Skill-dependent TM training was performed by applying perturbations to challenge postural stability, propulsion, and limb swing, and included walking in multiple directions, over inclines and obstacles, with resisted propulsion provided with elastic bands, weighted vests, and leg weights on the paretic limb with limited handrail use as tolerated, or reduced handrail use or body weight support and advance with multi-directional walking to challenge postural stability. Two to 5 different tasks/perturbations were randomly alternated and repeated within 10-minute bouts, with difficulty increased as tolerated. Attention was directed toward task completion rather than normalizing kinematic strategies. Overground training focused on achieving fastest possible speeds or performing variable tasks as described above, with use of a gait belt or overhead mobile or rail suspension system. Stair climbing was performed over static or rotating stairs (Stairmaster) using reciprocal gait patterns and progression to faster speeds and reduced handrail use, use of leg weights or weighted vests to target biomechanical deficits. If specific tasks were not practiced during individual sessions, subsequent sessions focused on missed tasks. For the HF paradigm, intensity was also set to 70% to 80% HR reserve, although was limited only to forward walking on a TM or overground. Task difficulty was increased by increasing walking speeds within targeted intensities. For the LV paradigm, training sessions were similar to HV training described above but with targeted intensities set to 30% to 40% HR reserve. 9 Across all groups, participants performed 1800 to 3500 steps/sessions (HF > HV > LV, P < .01) over 32 to 38 minutes/session (LV > HV and HF, P < .01) over 25 to 29 sessions (P = .79). 9
Data Collection
Participants performed both overground testing of SSV (ProtoKinetics LLCA) and graded TM assessments at baseline (BSL) and posttraining (POST). Graded TM testing was performed on a motorized TM with speeds starting at 0.1 m/s for 1 minute and increased in 0.1 m/s increments every minute with simultaneous 12-lead electrocardiography and oxygen consumption measures. Testing speed was increased until ACSM (American College of Sports Medicine) criteria for test termination was reached, including significant electrocardiogram abnormalities, evidence of gait instability, or the participant refused to continue. The fastest TM speed that participants could walk for 1 min was considered peak TM speed.
Biomechanical data at BSL and POST were collected on an instrumented split-belt motorized TM with speed adjusted to participants’ peak speed achieved during testing. During POST, participants also walked at TM speeds matched to peak speeds achieved at BSL (MATCH) to control for differences in speed. Data were collected for 30 seconds, beginning ~15 seconds after the start of the test to allow for accommodation. The split-belt TM was embedded with bilateral 6-degree-of-freedom force plates (Bertec Corporation) and surrounded by an 8-camera motion capture system (Motion Analysis Corporation). Thirty-two reflective markers were placed on bilateral lower limbs using a modified Cleveland Clinic marker set, and kinematic and kinetic data were sampled at 100 Hz.
Data Analysis
Marker and force data were processed using Cortex software (Motion Analysis Corp), and further analyzed using custom software in Visual3D (C-Motion Inc) and MATLAB (Mathworks). A bilateral 6-degree-of-freedom model of each subject’s lower limbs was created from marker data during static standing. Lower limb inertial properties were estimated based on the subject model and anthropometric measurements of limb positions and joint centers. Marker data were filtered using a low-pass, second-order Butterworth filter (10 Hz). Sagittal joint angles were calculated from the transformations between model segments. Sagittal joint moments were calculated from inverse dynamics using low-pass filtered ground reaction force data (second-order Butterworth at 20 Hz) and joint angle measurements. Sagittal joint powers were calculated as the product of joint moments and angular velocities. Kinetic data were normalized to body weight, and stance was identified as the period when vertical forces >10 N. Kinematic and kinetic data were further normalized to percentage gait cycle (%GC) and average step cycle profiles were created for all complete steps.
Specific kinematic and kinetic variables of interest included specific spatiotemporal measures that describe the walking patterns, and those that may estimate patterns of recovery vs compensation. Spatiotemporal measures of interest included cadence and stride length, as well as paretic single limb stance (%SLS) and step length asymmetry (SLA; paretic /nonparetic step length). Joint kinematic variables included sagittal hip, knee and ankle angles, including peak flexion and extension, total range of motion (ROM), and hip-knee joint coordination, defined by hip-knee phase plots and quantified in terms of stride-to-stride consistency across multiple gait cycles using the average coefficient of correspondence (ACC). The ACC ranges between 0 and 1, with higher values indicating a greater consistency in hip-knee joint coordination during walking.24,25 In general, we suggest that greater changes in ROM and hip-knee ACCs in the nonparetic limb may be estimates of neurological recovery, whereas patterns of greater change in the nonparetic limb would be characteristic of compensation.
Kinetic metrics of interest included average positive sagittal ankle, knee, and hip powers, which were calculated by integrating joint power within the stride only when values were positive.
Powers were averaged over specific portions of the GC consistent with peak power generation (ie, concentric or positive power) profiles observed during walking. Figure 1 depicts a single-subject example of paretic limb sagittal joint powers, with both positive and negative powers identified by others previously.22,26,27 As the present study used averaged versus peak joint powers, portions of the gait cycle were identified that encompassed the peaks identified, and powers were averaged over this range.22,27 The specific portions of the GC chosen were slightly larger (greater range of %GC) than would be expected in individuals without neurological injury given the variability in neuromuscular coordination of patients poststroke demonstrated previously. Importantly, only positive powers were averaged during those portions (negative values were not averaged with positive values). Specific phases included positive ankle powers during 40% to 80% of the GC (A2), associated with concentric plantar flexor activity for propulsion. For knee joints, powers were calculated from 10% to 50% GC, which is typically associated with knee extension during the primary loading throughout most of single limb stance (K2). Positive hip joint powers were calculated in 2 separate bins including 0-30% of the GC associated with hip extensor activity following initial contact (H1) and during 50% to 100% of the GC associated with hip flexor activity prior to and during swing (H3). Other power absorption (negative) phases (ie, A1, K1, K3-K4, H2, Figure 1) are not included in this analysis. In this analysis, greater gains in joint powers in the paretic vs non-paretic limbs would be indicative of patterns of recovery versus compensation.
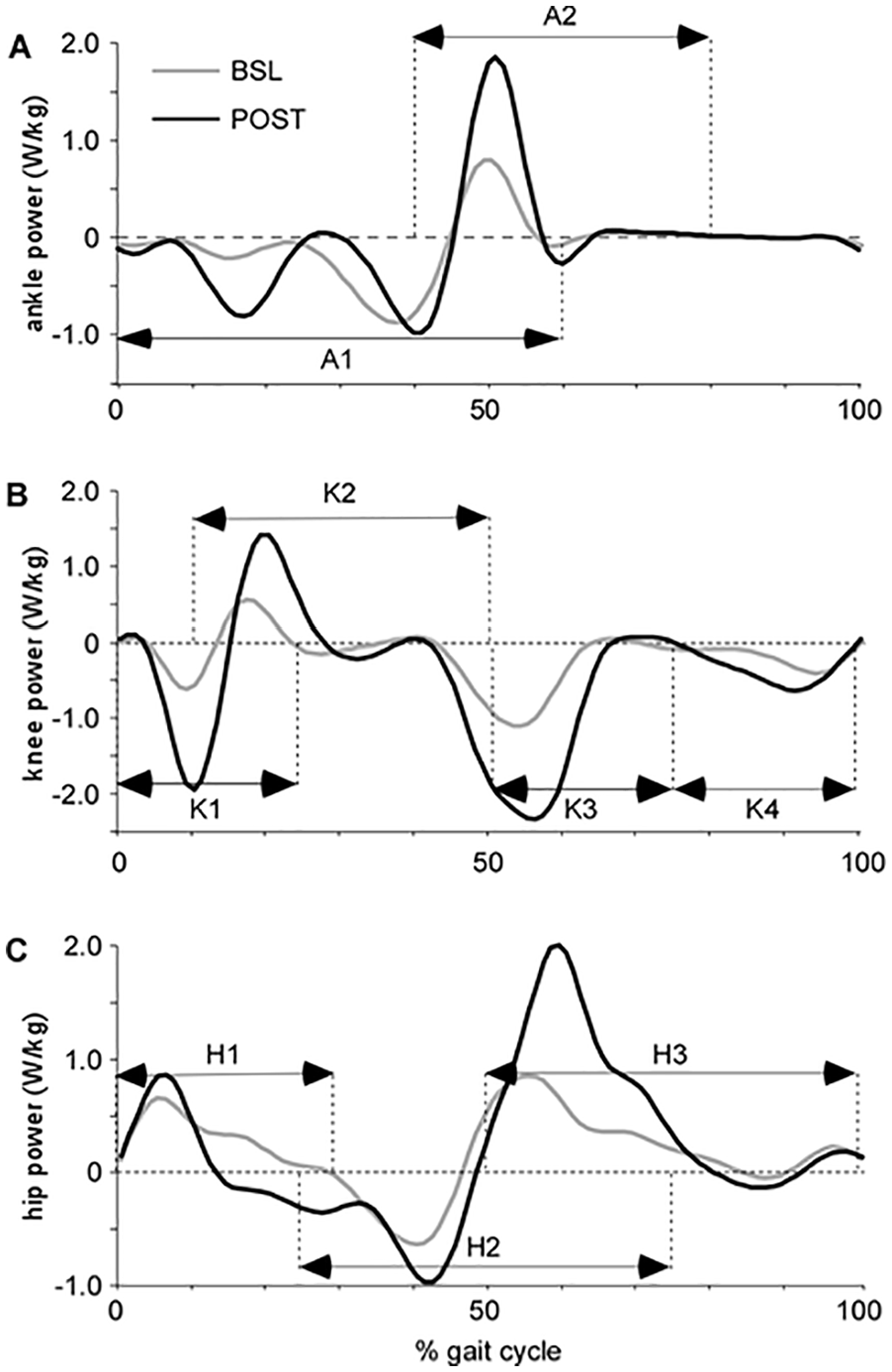
Single subject data for paretic hip, knee, and ankle powers throughout the gait cycle at both baseline (BSL; gray) and posttraining (POST; black). The designated portions of the gait cycle (A1-2, K1-4, H1-3) are identified to indicate approximate gait cycle phases when peak joint powers are observed.
Statistical Analysis
Data are presented as mean ± standard deviation (SD) throughout the text and tables. All variables were checked for normality using the Shapiro-Wilk test, presented as mean (SD) with analyses conducted using SPSS (v22, IBM Corp). One-way ANOVAs and chi-square analyses were utilized to evaluate BSL differences in demographics, clinical characteristics, and gait biomechanics between groups. Two separate 1-way ANOVA analyses were then conducted to compare training-induced changes between groups, including differences in changes at peak TM at BSL and POST and differences in changes at POST with speeds matched to BSL peak speeds (MATCH). Significance was set at α = .05, with post hoc Bonferroni comparisons.
Stepwise multiple linear regression analyses were used to investigate relationships between changes (Δ) in SSV and peak TM speed from BSL to POST and changes in specific variables of joint kinematics and kinetics across all participants combined as opposed to independent training groups given their smaller sample sizes. Spatiotemporal variables were omitted due to their known contributions to changes in speed and correlation with other kinematic/kinetic parameters.
Considering the inherent interdependency between gait variables, PC analyses were applied to 16 specific gait variables (joint kinematics and positive powers) as a method to extract the primary features (ie, reduce the dimensions) of the training-induced changes with smaller sample sizes. 28 PC analyses were performed separately for each training paradigm to discern potential contributions of biomechanical variables accounting for changes observed with each training paradigm. For each LT group, participants and variables were arranged in a matrix with gait variables as columns and participants (observations) as rows. The PCs were retained according to the scree plot which delineates individual contribution of each PC to explain the overall variance. The goal was that all the retained PCs together explained ≥80% of the overall variance. To examine the validity and robustness of the calculated PCs, a bootstrapping procedure was conducted of each LT group with 1000 iterations on a random subset of 80% of participants in that group. The factor loadings (the correlation between original gait variables and each PC) and component score coefficients (the contribution or weight of original variables to form each PC) were then cross-validated using Pearson’s correlations (r) and significance values. To limit the redundancy of contributing gait variables across the orthogonal PCs and in light of bootstrapping technique to reassure robustness of the executed PCs, a significant threshold of P < .01 was used to determine significance (vs r values 24 ), that is, variables with significant (P < .01) contributions to the first 3 PCs were reported.
Results
Fifty participants (17 LV, 15 HF, 18 HV) with valid kinematic data were included (Table 1) with no between-group differences in BSL demographics or clinical characteristics. Data from 40 of 90 participants were lost due to loss of marker placement during TM testing (n = 22), termination of study participation without POST biomechanical assessments (n = 2), inability to walk at least 0.1 m/s at BSL (n = 5), or transition of laboratory location (n = 11). Eight participants were unable to accurately place each limb on separate TM belts, resulting in 42 participants with valid kinetic data (15 LV, 14 HF and 13 HV). Baseline peak TM speeds within each group were nearly identical to speeds in the full clinical trial. 9 Comparisons of other demographic and clinical characteristics indicate minimal differences between groups, except for body weight (P = .02) with differences in gender (P = .06) and duration poststroke (P = .15) approaching significance.
Demographic and Baseline Characteristics.
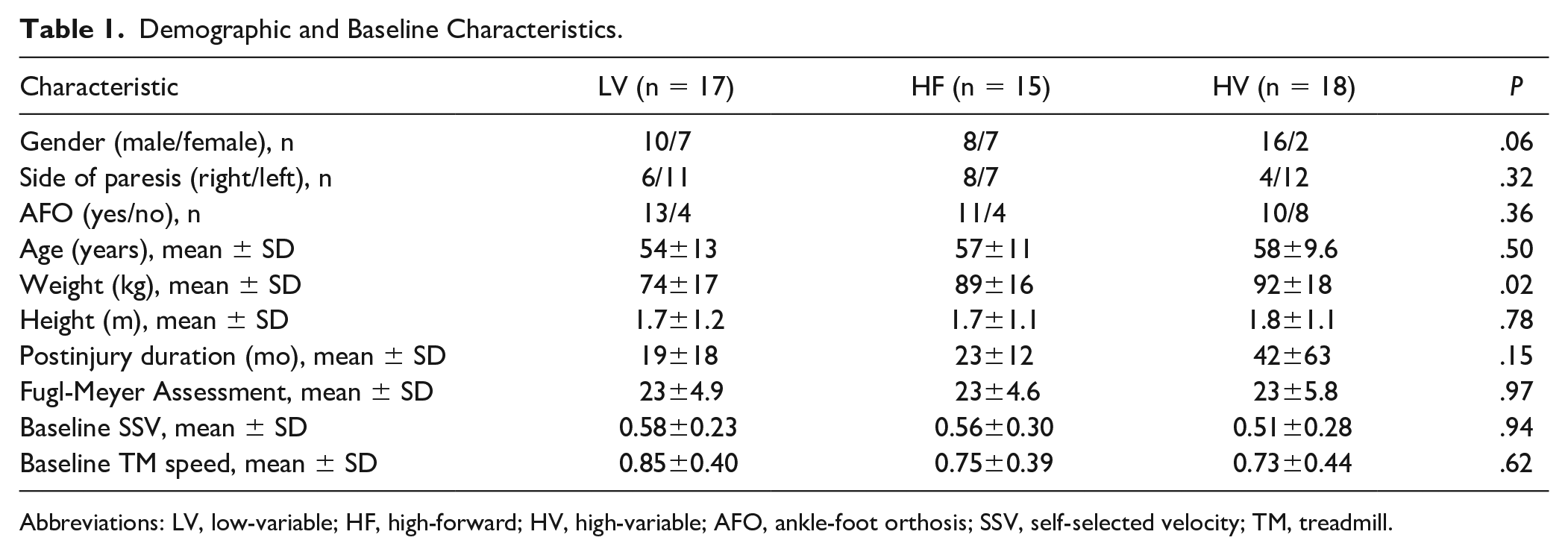
Abbreviations: LV, low-variable; HF, high-forward; HV, high-variable; AFO, ankle-foot orthosis; SSV, self-selected velocity; TM, treadmill.
Changes in Gait Kinematics and Kinetics
Changes in both SSV (HV: 0.14 ± 0.14 m/s; HF: 0.18 ± 0.10 m/s; LV: 0.02 ± 0.06 m/s) and peak TM speed (HV:0.41 ± 0.10 m/s; HF: 0.44 ± 0.17 m/s; LV: 0.11 ± 0.13 m/s) were significantly different between groups favoring HF and HV over LV (both Ps < .01). Speed-related differences were reflected by consistent differences in cadence and stride lengths (Table 2, both Ps < .01). All groups demonstrated improvements in temporal symmetry (ie, % paretic single-limb stance) or step length asymmetry (SLA), with no significant differences between groups. For MATCH comparisons, there were no differences in spatiotemporal parameters (Supplemental Table).
Comparison of Kinematic and Kinetic Variables at BSL and POST as Mean ± SD. a
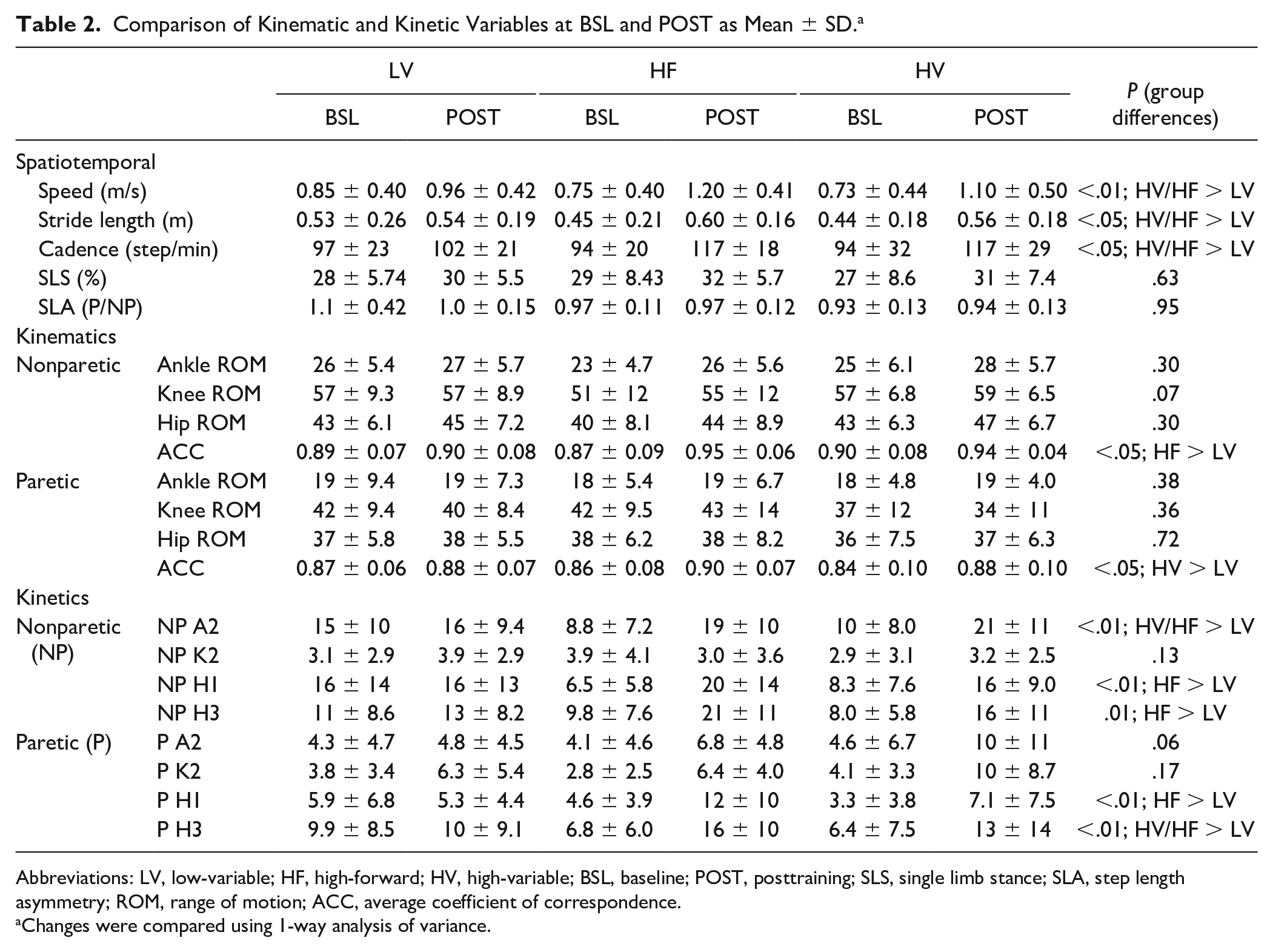
Abbreviations: LV, low-variable; HF, high-forward; HV, high-variable; BSL, baseline; POST, posttraining; SLS, single limb stance; SLA, step length asymmetry; ROM, range of motion; ACC, average coefficient of correspondence.
Changes were compared using 1-way analysis of variance.
Evaluation of joint kinematic and kinetic variables at BSL peak TM speeds revealed no differences between groups. Training-induced changes in kinematics indicated limited differences except for changes in intralimb hip-knee consistency (ie, ACC) in paretic and nonparetic limbs (both Ps < .05; Table 2), with differences in nonparetic hip-knee ACC between HF versus LV (Δ = 0.08, P < .05) and paretic hip-knee ACC between HV versus LV approaching significance (Δ = 0.05, P = .05). For MATCH comparison, few differences were observed (Supplementary Table) except for nonparetic hip-knee ACC (HF > HV; P < .05), and nonparetic hip ROM (HV and LV > HF; P < .05).
For kinetic variables, several training-induced changes were different between groups (Table 2). Results from ANOVAs and post hoc comparisons revealed significant differences in bilateral hip power at initial contact (H1) and terminal stance/preswing (H3; P < .01), and nonparetic ankle power (A2; P < .001) following HF versus LV paradigms. Nonparetic ankle propulsive powers were also increased following HV versus LV training (A2; P < .05), with additional differences in paretic hip power (H3; P < .05). Substantial differences in paretic A2 powers observed following HV and LV training were not significantly different (P = .06). For MATCH comparisons, only differences in paretic knee power were significant following HV versus LV (K2; P < .05; Supplementary Table).
Association Between Gait Biomechanics and Locomotor Function
Stepwise, multiple linear regression analyses estimated the relative contributions of training-induced changes in kinematic and kinetic variables to improvements in walking speeds (ΔSSV and Δpeak TM speed). For all participants, peak TM speeds were positively associated with nonparetic kinetic and kinematic variables, explaining up to 64% of the variance. Conversely, ΔSSV were positively related to paretic ankle power with contributions of nonparetic hip power, also accounting for 47% of the variance.
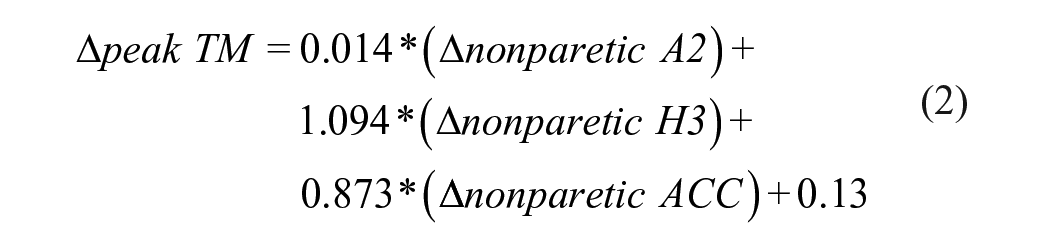

Subsequently, PC analyses was utilized to extract the primary features of the training-induced changes in biomechanical variables (joint kinematics and kinetics) within each group. Analyses of the 16 joint kinematic and kinetic variables revealed 5 robustly reproducible PCs that accounted for >80% of the variance in each training group. The first three PCs explained >50% of the variance and are discussed further (Figure 2). For LV training, PC1 was characterized by paretic ankle ROM and hip power (H1 and H3), as well as non-paretic H3, PC2 was defined by nonparetic hip-knee ACC and paretic ankle power (A2), and the third PC was defined by nonparetic hip power (H1). For HF, the first 3 PCs were organized differently; PC1 was characterized by nonparetic ACC and A2, and paretic knee (K2) and hip (H3) power, PC2 was defined by bilateral H1, and PC3 was characterized by nonparetic hip ROM and H3. For HV, all PCs were from the paretic limb; the first PC focused on paretic H1 and H3 as well as ankle ROM and PC2 was defined by paretic A2, with no specific gait variables with significant correlations to PC3.
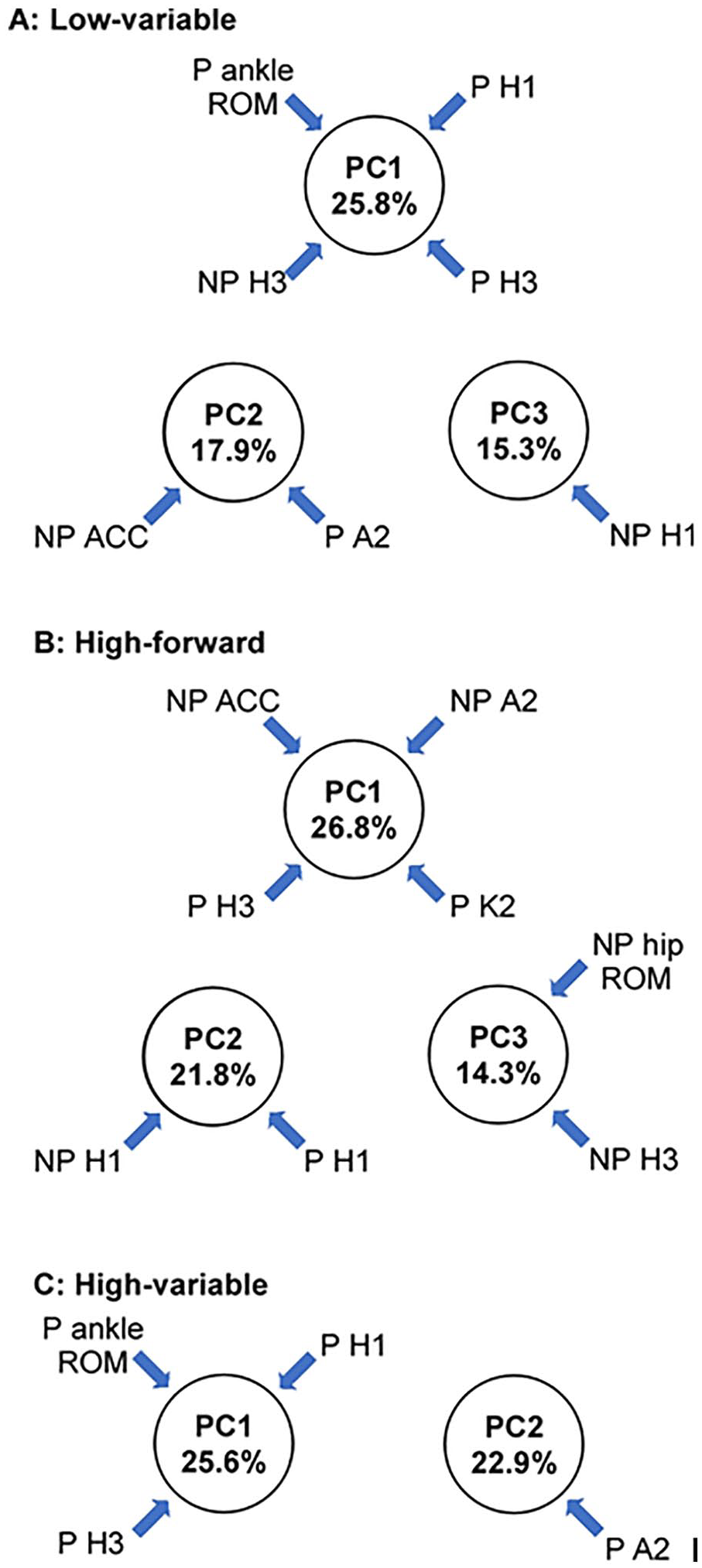
Principal component analyses for each training subgroup, delineating which biomechanical variables contribute to each principal component in (A) LV, (B) HF, and (C) HV. For each training group, the first 3 principle components and the variables that contribute to them are indicated. LV, low-variable; HF, high-forward; HV, high-variable; ROM, range of motion; ACC, average coefficient of correspondence; P, paretic; NP, nonparetic.
Discussion
The present study detailed changes in locomotor kinematics and kinetics following 3 different LT paradigms in participants poststroke, revealing consistent differences in TM speed, stride length and cadence between high- versus low-intensity training. Between-group comparisons of hip-knee ACCs and joint powers indicated more consistent differences between HF and LV in the nonparetic limb, whereas differences between HV and LV appear to favor more paretic limb variables. Subsequent PC analyses revealed PCs loaded on paretic limb kinetic variables and hip-knee kinematic consistency with HV training. Conversely, PCs in HF and LV were characterized by changes in both nonparetic and paretic limb hip-knee ACCs and specific kinetic variables. The combined findings could provide some insight into patterns of compensation and recovery in patients poststroke with specific locomotor interventions.
Previous studies detailing differential changes in joint kinematic and kinetics following various training paradigms provided to individuals poststroke are scarce.25,29-31 While a primary limitation of this study is the small sample size, particularly within each training group, significant between-group differences were nonetheless observed for many variables. Differences in spatiotemporal patterns and kinematic consistency between high- versus low-intensity groups are consistent with previous 25 and preliminary studies22,30 and reinforce the notion that high-intensity training does not entrain abnormal kinematic patterns. Rather, such training appears to facilitate more “normal” movement strategies. Analyses of joint powers further suggest the specific methods for delivering high-intensity training may influence neuromuscular strategies. The PC analyses indicate greater loading of paretic limb changes with HV, which may be indicative improve neurological recovery underlying gains in walking function. Furthermore, greater loading of bilateral limb changes in HF suggest patterns of recovery and compensation underlying locomotor improvements.
The potential significance of these findings may be 2-fold. First, strategies that focus on providing large amounts of stepping practice during forward walking at high intensity may result in gains in locomotor function, although improvements may be more dependent on both “normal” and compensatory strategies. 22 A separate conclusion is that HV training that focused on paretic limb deficits resulted in greater recovery of neuromuscular strategies used prior to injury indicative of neurological recovery. Importantly, compensatory patterns are also observed in all groups (Table 2) consistent with previous results. 22 In specific patients with substantial distal impairments that require ankle-foot orthoses, such compensations will be necessary. Nonetheless, the combined findings suggest attention to specific biomechanical locomotor deficits may influence patterns of neurological and functional recovery in patients poststroke. Namely, attention to specific locomotor deficits that contribute to forward progression (ie, propulsion, limb swing) appears to elicit gains in those biomechanical deficits with HV training. Furthermore, greater gains in dynamic stability were observed following HV, where very little changes in balance are observed with HF training. 9 The combined findings emphasize the potential significance of performing stepping training in variable context, and further highlight the importance of specificity of training to elicit desired motor outcomes poststroke.
Additional limitations to the present study include the inability to include additional participants from the training study due to foot placement during testing, or those unable to walk at least 0.1 m/s on the treadmill at BSL. These constraints are related to those with significant impairments in locomotor function and whether these findings can be applied to nonambulatory patients early poststroke are not clear. Furthermore, regression analyses on training subgroups were not possible because of sample sizes, and the use of PC analyses partly overcomes this issue, 28 although alternative research suggests otherwise. 32 Additional work is needed to confirm these findings across larger, more diverse patient populations.
Conclusions
Providing stepping training at higher intensities resulted in significant gains in spatiotemporal parameters, kinematic consistency, and power generation as compared to lower intensity activities. Post hoc comparisons and PC analyses suggest greater paretic limb coordination and joint powers were observed following high-intensity training in variable contexts, whereas changes following high-intensity training targeting only walking forward resulted in greater trend of bilateral improvements. The data support the hypotheses that focused attention to paretic limb deficits during variable stepping tasks at high intensities can result in improved paretic kinematics and kinetics indicative of recovery versus compensation.
Supplemental Material
Supplementary_Table – Supplemental material for Locomotor Kinematics and Kinetics Following High-Intensity Stepping Training in Variable Contexts Poststroke
Supplemental material, Supplementary_Table for Locomotor Kinematics and Kinetics Following High-Intensity Stepping Training in Variable Contexts Poststroke by Marzieh M. Ardestani, Christopher E. Henderson, Gordhan Mahtani, Mark Connolly and T. George Hornby in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by NIDILRR-H133B031127 and NIH-NINDS-NS079751.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.