Abstract
Background: In the chronic phase after stroke, cortical excitability differs between the cerebral hemispheres; the magnitude of this asymmetry depends on degree of motor impairment. It is unclear whether these asymmetries also affect capacity for plasticity in corticospinal tract excitability or whether hemispheric differences in plasticity are related to chronic sensorimotor impairment. Methods: Response to paired associative stimulation (PAS) was assessed bilaterally in 22 individuals with chronic hemiparesis. Corticospinal excitability was measured as the area under the motor-evoked potential (MEP) recruitment curve (AUC) at baseline, 5 minutes, and 30 minutes post-PAS. Percentage change in contralesional AUC was calculated and correlated with paretic motor and somatosensory impairment scores. Results: PAS induced a significant increase in AUC in the contralesional hemisphere (P = .041); in the ipsilesional hemisphere, there was no significant effect of PAS (P = .073). Contralesional AUC showed significantly greater change in individuals without an ipsilesional MEP (P = .029). Percentage change in contralesional AUC between baseline and 5 m post-PAS correlated significantly with FM score (r = −0.443; P = .039) and monofilament thresholds (r = 0.444, P = .044). Discussion: There are differential responses to PAS within each cerebral hemisphere. Contralesional plasticity was increased in individuals with more severe hemiparesis, indicated by both the absence of an ipsilesional MEP and a greater degree of motor and somatosensory impairment. These data support a body of research showing compensatory changes in the contralesional hemisphere after stroke; new therapies for individuals with chronic stroke could exploit contralesional plasticity to help restore function.
Introduction
After a stroke resulting in upper-limb impairment, cortical excitability shifts in both the ipsilesional and contralesional sensorimotor cortices, reflecting underlying neurobiological processes of recovery (for review see: Auriat et al 1 ). In the chronic phase of stroke, the ipsilesional hemisphere shows decreased cortical excitability2,3 and decreased interhemispheric inhibition, 4 whereas the contralesional hemisphere shows normal or increased cortical excitability2,3,5 and increased interhemispheric inhibition to the ipsilesional hemisphere.6,7 The functional integrity of the ipsilesional corticospinal tract, assessed by presence or absence of an ipsilesional motor-evoked potential (MEP), is a further predictor of increases in the activity of the contralesional hemisphere.8-10 Yet the consequences of these neurophysiological characteristics for motor outcomes after stroke are not well understood. The observation that contralesional excitability is higher in individuals with poorer upper-limb outcomes led to the development of the interhemispheric competition model (IHCM), which theorizes that increased contralesional excitability results in increased inhibition from the contralesional hemisphere to the ipsilesional hemisphere, thus hindering the excitability of ipsilesional motor regions.11,12 Recent data, however, suggest that the nature of hemispheric interactions after stroke is more complex than accounted for in the IHCM. Specifically, when individuals with chronic stroke are stratified by severity, individuals with more severe paresis show less interhemispheric inhibition from the contralesional hemisphere to the ipsilesional hemisphere. 13 The IHCM further fails to predict response to repetitive transcranial magnetic stimulation (TMS) interventions in individuals with severe upper-limb impairment; suppressing the activity of the contralesional hemisphere results in poorer upper-limb function in individuals with severe paresis, but benefits individuals with mild paresis.14-16 Increased contralesional excitability may, therefore, represent a compensatory mechanism of cortical plasticity in individuals with greater injury to descending ipsilesional motor tracts.12,17
Neurophysiological measures of cortical excitability may relate to the capacity for neuroplasticity in the sensorimotor cortex. However, single- and paired-pulse TMS measures of corticospinal tract (CST) excitability, which have been used to establish the IHCM, do not directly assess capacity for neuroplasticity. Paired associative stimulation (PAS) is an experimental technique that involves delivery of an electrical stimulation over a peripheral nerve, followed by a single TMS pulse over the motor cortex, such that the peripheral volley arrives at the primary motor cortex simultaneously with the delivery of the TMS pulse. 18 PAS can transiently increase corticospinal excitability and is a noninvasive model of long-term potentiation (LTP)-like plasticity. 19 In otherwise healthy adults, PAS effects are potentiated by an acute exercise bout20,21 and decrease with aging, 22 providing useful information about cortical neuroplasticity in humans. Little is known about how the effects of PAS are altered by stroke. Previous work by Castel-Lacanel et al23,24 has shown that the effects of PAS can be elicited in the paretic extensor carpi radialis (ECR) in chronic stroke 23 but decrease in the paretic ECR between the subacute and chronic stages of recovery. 24 These previous studies provide a proof-of-concept for the use of PAS in chronic stroke populations, but they did not assess plasticity in the contralesional motor cortex and further did not consider the impact of sensorimotor impairment on PAS effects.
In the present study, we evaluated PAS effects on corticospinal excitability in a cohort of individuals with chronic stroke. PAS was performed bilaterally to evaluate contralesional and ipsilesional corticospinal plasticity as well as to evaluate whether the degree of change in corticospinal excitability was related to motor and somatosensory impairment in the paretic limb. We hypothesized that ipsilesional cortical plasticity would be decreased in individuals with chronic hemiparesis, and we further hypothesized that the potential for cortical plasticity would relate to motor and somatosensory impairment in the upper limb.
Methods
Participants
We recruited individuals in the chronic phase of first-time ischemic stroke between the ages of 45 and 85 years. Individuals were recruited by convenience sampling from the community and local postings. Participants were excluded if they had a history of seizure/epilepsy, a major psychiatric diagnosis, neurodegenerative disorder, substance abuse, or head trauma. Informed consent was obtained for each participant in accordance with the Declaration of Helsinki. The University of British Columbia research ethics boards approved all aspects of the study protocol.
Upper-Limb Assessments
To assess motor impairment, we used the upper-extremity portion of the Fugl-Meyer (FM) assessment. 25 The FM is scored from 0 to 66, with higher scores indicating less motor impairment. To assess motor function, we used the Wolf Motor Function Task (WMFT) mean rate score. 26 The WMFT consists of 15 movement items; performance is indexed by time to complete the task. Three trials of each task were performed and performance times averaged. Average performance time was then used to calculate a projected mean rate per minute of task completion as follows: Task rate = 60 (s)/Performance time (s). If an individual was unable to complete a task within 120 s, then a score of 0 was assigned for that task. The average task rate of performance was averaged across all 15 tasks for a total mean rate score, with higher numbers indicating better motor function. To assess somatosensory function, we used the Semmes-Weinstein monofilament test,27,28 testing perceptual thresholds at 4 sites on each arm (dorsum, thenar, and hypothenar regions of the hand and dorsal forearm). Sensation threshold was determined by summing perceptual thresholds on each testing site for a total score out of a possible 26.6, with higher values indicating greater somatosensory impairment.
Experimental Procedure
TMS procedures were conducted for each participant within 1 cerebral hemisphere (ipsilesional/contralesional) per testing session, with a minimum 24-hour period between testing sessions.
Electromyography (EMG)
Surface EMG data were collected with 3-cm diameter circular surface recording electrodes (Covidien, Mansfield, MA) placed over the muscle belly of the ECR. The EMG signal was collected with LabChart software (LabChart 7.0). EMG data were preamplified (1000×) and band pass filtered at 10 to 1000 Hz with a Powerlab system and bioamplifiers (AD Instruments, Colorado Springs, CO). Data were recorded from 100 ms prior to 400 ms post–TMS delivery.
Peripheral Nerve Stimulation
Square-wave pulses of 0.2 ms duration were delivered at the wrist with a constant current stimulator (DS7A; Digitimer, Hertfordshire, UK). The bar electrode was placed over the radial nerve, approximately 1 inch proximal to the carpus. Immediately before TMS procedures, the lowest stimulation intensity to evoke a just-visible twitch in the ECR muscle was determined (motor threshold); bar electrode stimulation was delivered at 150% of motor threshold during PAS stimulation.
Transcranial Magnetic Stimulation
TMS was performed according to recently updated safety guidelines, 29 with a Magstim figure-of-eight coil (Magstim 70 mm P/N 9790; Magstim, Carmarthenshire, UK) and a Magstim 2002 stimulator (Magstim Co, UK). The optimal ECR cortical hotspot was localized and registered using Brainsight neuronavigation software (Rogue Resolutions, Montréal, Québec, Canada) to maintain accurate positioning of the TMS coil for all subsequent stimulation delivery. Resting motor threshold (RMT) was determined by finding the lowest percentage of maximum stimulator output (%MSO) that elicited a MEP of at least 50 µV in 5 out of 10 consecutive trials in the relaxed contralateral ECR. 30 Active motor threshold (AMT) was determined by finding the lowest %MSO that elicited a MEP of at least 200 µV in 5 out of 10 consecutive trials in the contralateral ECR while holding an isometric contraction at 20% of maximum voluntary contraction, 30 guided by visual feedback from a digitized force output reading from a handgrip dynamometer (AD Instruments, Colorado Springs, CO). During all TMS data collection, the background EMG signal was monitored continuously on LabChart to ensure that the ECR remained quiescent. If background EMG was visible, the experimenter added TMS pulses until the planned number of trials (for MEP recruitment curves (RCs) and paired pulse measures) was completed with the ECR muscle at rest; trials with visible background activity or experimental artifacts were later deleted manually by a single rater (JKF), which accounted for <0.02% of trials.
Paired Associative Stimulation
PAS was performed by delivering a peripheral afferent volley using a bar electrode placed over the radial nerve with 0.2-ms pulses delivered at 150% motor threshold, 25 ms prior to delivering a TMS pulse over the ECR hotspot at 130% RMT. 31 Pulses were delivered at 0.25 Hz for 30 minutes, for a total of 450 stimulations. 20 As attention has been shown to influence the effects of PAS, 31 participants were instructed to focus on the sensation of the electrical stimulation occurring at their wrist. To gauge participants’ degree of attention and to refocus attention to the wrist, cutaneous stimuli were delivered once every ~3 minutes at the bar electrode on the wrist asynchronously with PAS stimulations. Participants were asked to indicate when they felt the asynchronous peripheral stimulation; if the participant failed to indicate when an asynchronous stimulus was delivered, the experimenter would instruct the participant to guide attention back to the wrist.
The PAS protocol was modeled after previous published PAS methods used in the abductor pollicis brevis in healthy controls.20,31,32 Our protocol was also similar to a PAS protocol used in the paretic ECR in individuals with chronic stroke, which used 30 minutes of PAS stimulation with a 25-ms interstimulus interval (ISI).23,24 However, this past work applied PAS stimuli at 0.1 Hz and delivered ~30% of the total number of PAS stimulations as compared with our study protocol. The dosage of PAS stimulation applied in the current study replicates previous PAS protocols performed in healthy adults. 20
PAS effects persist for up to 30 to 60 minutes post-PAS in healthy adults 18 ; to evaluate the time course of PAS-related effects in individuals with chronic stroke, we collected TMS outcome measures (RCs and paired-pulse) at 3 time points: immediately before PAS (baseline), then at 5 minutes post-PAS, and 30 minutes post-PAS. TMS outcome measures took ~10 minutes to collect at each timepoint.
Recruitment Curves
MEP RCs were collected by applying single-pulse TMS over the ECR hotspot while at rest, at a rate of 0.1 to 0.2 Hz, with 20% variance between pulses. A total of 48 stimuli were delivered at intensities ranging from 90% to 140% RMT in 10% increments, with 8 stimuli at each intensity. The order of each TMS output increment was randomized prior to collection and held constant for each individual experimental session. The outcome measure of the RC was area under the curve (AUC), which was calculated using a trapezoidal integral method as in previous work.21,33
Paired Pulse
Intracortical facilitation (ICF) and short-interval intracortical inhibition (SICI) data were collected according to previously described procedures.34,35 For both ICF and SICI, a subthreshold conditioning stimulus (CS) was delivered at 90% AMT, followed by a suprathreshold test stimulus (TS). The TS was delivered at the %MSO required to consistently evoke an MEP of ~1 mV in the relaxed ECR. For individuals in whom the ECR MEP amplitude did not reach 1 mV, the TS was delivered at the %MSO beyond which MEP amplitudes no longer increased (mean amplitude nonparetic ECR: 907 µV, standard error of the mean [SEM]: 94.74; paretic ECR: 347 µV, SEM: 48.83). The ISI of the CS and TS for SICI and ICF were set at 2 and 12 ms, respectively. For both ICF and SICI, 15 unconditioned stimuli were delivered at the TS intensity, followed by 15 paired-pulse stimulations (CS + TS) of ICF or SICI. ICF and SICI were calculated as a ratio of the average CS amplitude to the average TS amplitude and multiplied by 100 to obtain a percentage of cortical facilitation and inhibition. Values greater than 100% were considered to represent facilitation in ICF, and values smaller than 100% were considered to represent greater inhibition in SICI. The CS was held constant at 90%AMT for each session, but the TS was adjusted so that the MEP amplitude elicited was ~1 mV across each testing block.
Statistical Analyses
Statistical analyses were conducted using SPSS software (SPSS V23) and Statistica 13. For all variables, assumptions of normality were confirmed by the Shapiro-Wilk test, and nonnormal data were log transformed where necessary. Significance was set at P ≤.05 for all analyses.
One-way repeated-measures analysis of variance (ANOVAs) were performed to assess hemispheric differences in somatosensory impairment, RMT, baseline AUC, SICI, and ICF. To evaluate whether PAS induced a significant change in AUC, SICI, and ICF, we performed 1-way ANOVAs with within-subjects factor Time (baseline, 5 minutes, 30 minutes). Because of the expected difference in baseline MEP amplitudes between the paretic and nonparetic limbs and in order to include data from individuals who lack an ipsilesional MEP, 1-way ANOVAs were performed separately within each cerebral hemisphere (ipsilesional/contralesional). An additional analysis was performed to evaluate whether the absence of an ipsilesional CST, a prognostic indicator of motor recovery, 36 influences PAS effects on contralesional AUC. The group was divided by ipsilesional MEP status (presence [MEP+] vs absence [MEP−]), and a 2-way mixed-model ANOVA was performed with within-subject factor Time (preintervention, 5 minutes, 30 minutes) and between-subject factor MEP Status (MEP+/ MEP−). Post hoc analyses using Tukey’s honestly significant difference were performed when appropriate.
Associations With Sensorimotor Outcomes
To assess how the degree of change in cortical excitability in response to PAS relates to sensorimotor outcomes in the paretic limb, percentage change in AUC was calculated across time points that showed a significant difference in response to PAS (ΔBaseline − 5-minute AUC). ΔBaseline − 5-minute AUC was calculated as follows: [(5-minute AUC − Baseline AUC)/Baseline AUC] × 100. Bivariate correlational analyses (Pearson’s r for normally distributed data or Spearman’s r for nonnormally distributed data) were conducted to assess relationships between measures of sensorimotor outcomes in the paretic limb (FM, WMFT, and sensory threshold) and changes in cortical excitability (ΔBaseline − 5-minute AUC).
Results
Baseline Measures
A total of 22 individuals in the chronic phase of stroke participated in the study (means ± SD: age, 69 ± 6 years; poststroke duration, 103 ± 73 months; FM score, 40 ± 22; WMFT paretic limb, 35 ± 26; WMFT nonparetic, 54 ± 14). See Table 1 for participant characteristics. Sensory thresholds were significantly higher in the paretic limb, indicating greater cutaneous somatosensory impairment in the paretic limb (F1,20 = 10.299; P = .004). We were unable to elicit an ipsilesional MEP in 7 participants. In those in whom we elicited ipsilesional MEPs (n = 15), baseline RMT was significantly higher (F1,14 = 6.406; P = .024) and AUC was significantly lower (F1,14 = 10.542; P = .006) in the paretic limb. One-way ANOVA showed no hemispheric differences in ICF (F1,14 = 1.1258; P = .300) or SICI (F1,14 = 1.036; P = .326) at baseline.
Participant Characteristics.
Abbreviations: F, female; FM, Fugl-Meyer; M, male; PSD, post stroke duration (months); WMFT, Wolf Motor Function Task.
Paired Associative Stimulation
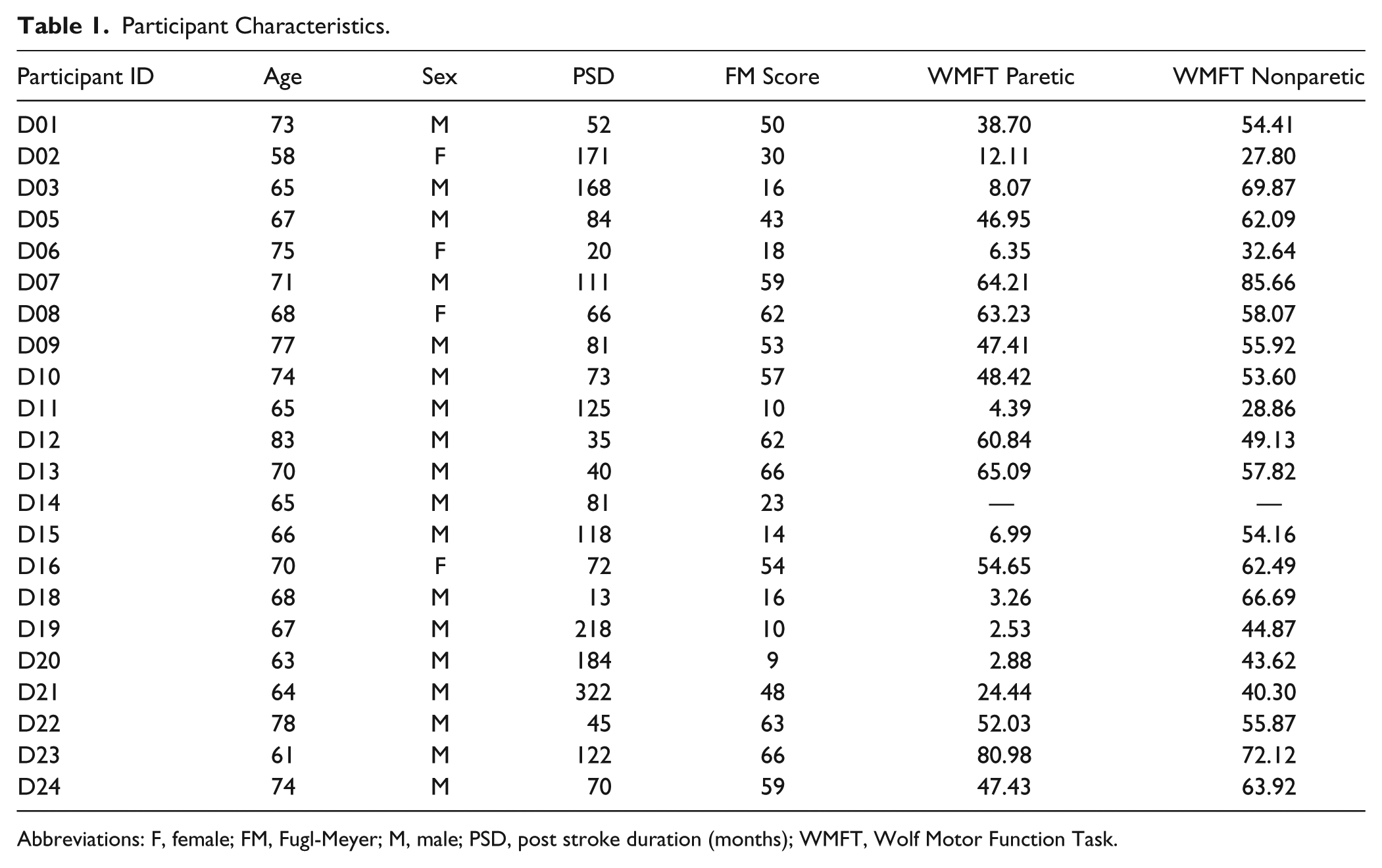
Mean data across all participants displaying the contralesional and ipsilesional AUC before and after PAS are shown in Figures 1A and 1B, respectively. One-way ANOVA revealed that PAS induced a significant effect of Time in the contralesional hemisphere (F2,42 = 3.464; P = .041). Post hoc analyses revealed that AUC was significantly greater at 5 minutes post-PAS compared with baseline (P = .042), with no other differences. In the ipsilesional hemisphere, 1-way ANOVA resulted in a trend toward significance for the effect of Time on AUC (F2,28 = 2.876; P = .073).
Response to paired associative stimulation (PAS), measured by area under the curve (AUC) of recruitment curves conducted immediately prior to PAS (baseline) and at 5 and 30 minutes post-PAS. Individual data are represented by the light gray lines; mean data are represented by the black lines. A. PAS induced a significant increase in area under the curve (AUC) between baseline and 5 minutes post-PAS in the contralesional hemisphere. B. There was no significant change in AUC in response to PAS in the ipsilesional hemisphere. Error bars are standard error of the mean. *P < .05.
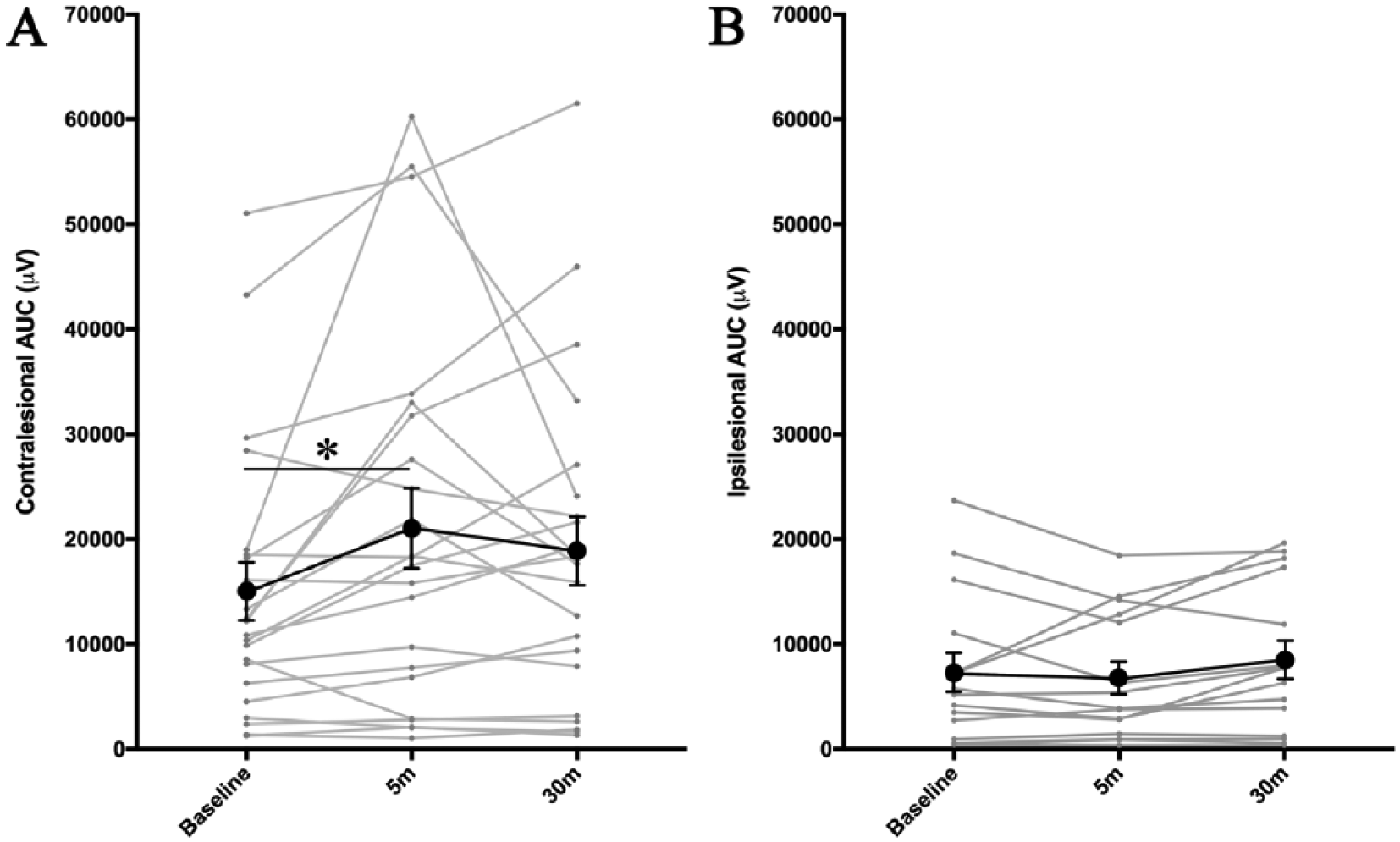
We further examined the impact of ipsilesional MEP status (presence or absence of an ipsilesional MEP; MEP+/−) on contralesional AUC; mean results of this analysis are presented in Figure 2. Two-way ANOVA revealed a significant main effect of Time on contralesional AUC (F2,40 = 7.516; P = .002), with no significant main effect of MEP status (F1,20 = 0.335; P = .569), and a significant Time × MEP Status interaction (F2,40 = 3.853; P = .029). Post hoc contrasts showed a significant difference between baseline and 5-minute AUC in the MEP− group only (P = .004), with no significant baseline and 5-minute difference in the MEP+ group (P = .484), and no difference in baseline data between MEP+/MEP- groups (P = .999).
The impact of ipsilesional motor-evoked potential (MEP) status on contralesional response to paired associative stimulation (PAS). Data presented are area under the curve (AUC) in the contralesional hemisphere, split based on the presence (gray squares) or absence (black circles) of an ipsilesional MEP. PAS induced a significant increase in contralesional AUC amplitude only in individuals without an ipsilesional MEP (MEP−). Error bars are standard error of the mean. **P < .01.
Paired Pulse
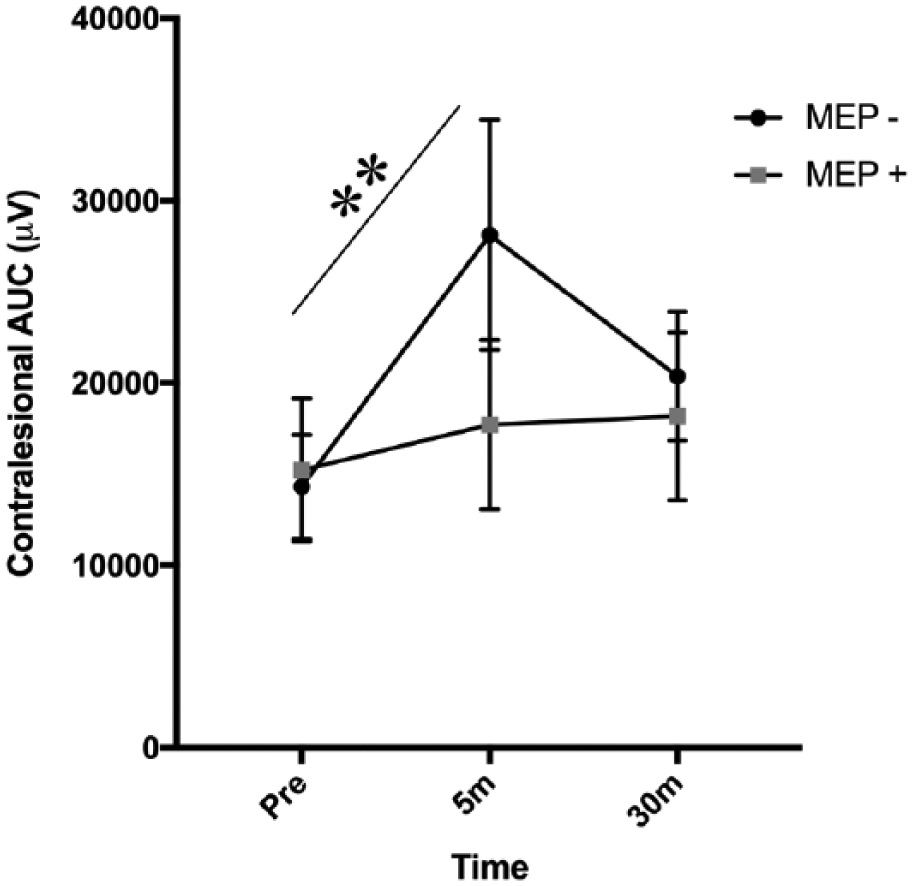
Mean data across all participants displaying ICF and SICI before and after PAS are shown in Figures 3A and 3B, respectively. One-way ANOVA in the contralesional hemisphere showed no effect of Time on ICF (F2,40 = 0.160; P = .853), or SICI (F2,40 = 1.243; P = .299). Similarly, 1-way ANOVA in the ipsilesional hemisphere showed no effect of Time on ICF (F2,28 = 1.116; P = .342) or SICI (F2,28 = 0.367; P = .696).
Changes in (A) intracortical facilitation (ICF) and (B) intracortical inhibition (SICI) in response to paired associative stimulation (PAS). There was no significant change in ICF or SICI in response to PAS in either cerebral hemisphere. Error bars are standard error of the mean.
Relationships Between Sensorimotor Outcomes and Response to PAS
Results from correlations between ΔBaseline and 5-minute AUC, and sensorimotor impairment scores are visualized in Figure 4. Contralesional ΔBaseline − 5-minute AUC correlated negatively with FM score (Figure 4A: r = −0.443; P = .039) and correlated positively with sensory threshold in the paretic limb (Figure 4B: r = 0.444; P = .044). Contralesional ΔBaseline − 5-minute AUC did not relate to WMFT in the paretic limb (r = −0.317; P = .097).
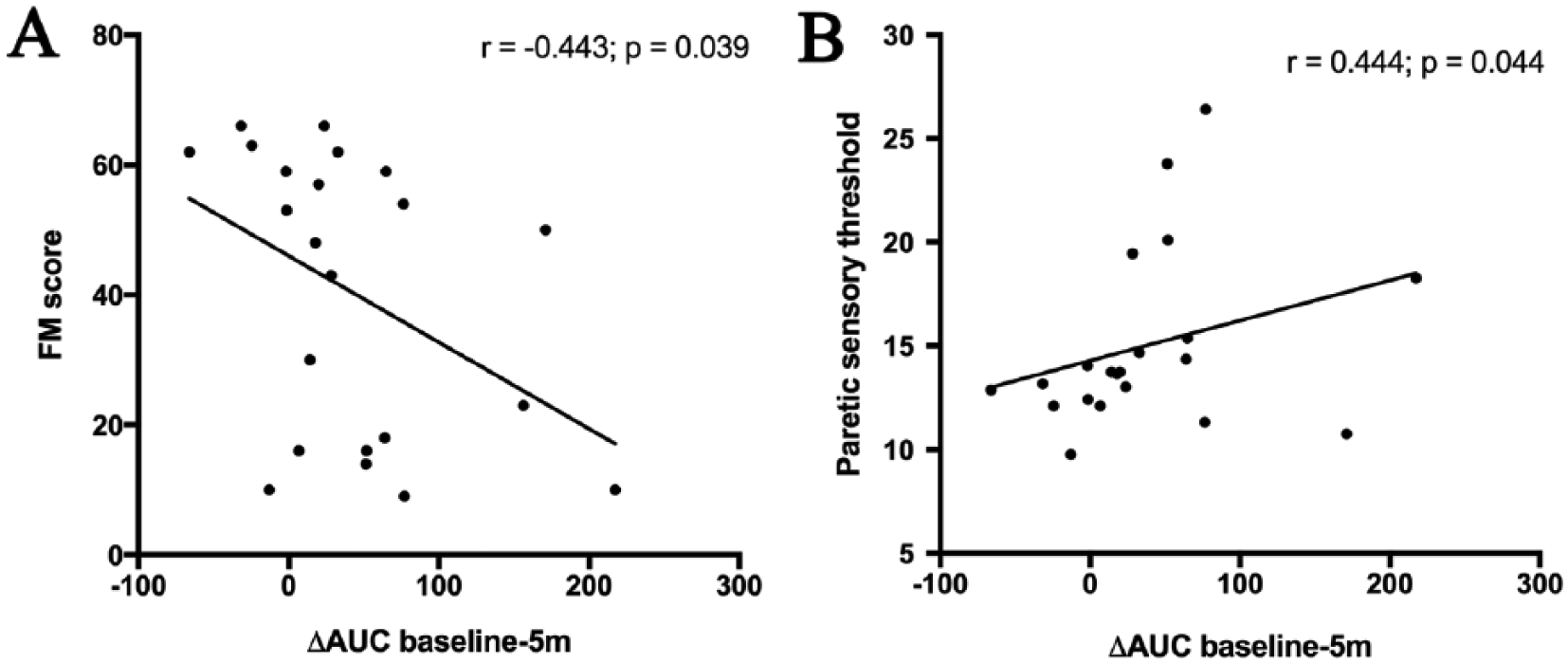
Correlations between sensorimotor impairment in the paretic limb and degree of change in contralesional area under the curve (AUC) between baseline and 5 minutes post–paired associative stimulation. A. Motor impairment, assessed by Fugl-Meyer score (FM) negatively correlated with the degree of contralesional plasticity, indicating that individuals with more severe motor impairment showed greater contralesional plasticity. B. Paretic sensory threshold, assessed by summed perception thresholds of graded monofilaments, positively correlated with degree of contralesional plasticity, indicating that individuals with more severe somatosensory impairment showed greater contralesional plasticity.
Discussion
Capacity for neuroplasticity is a fundamental mechanism underlying response to rehabilitation after stroke. In the present study, we used PAS bilaterally to assess neuroplastic capacity in corticospinal tract excitability in individuals with chronic hemiparesis.
There was a significant effect of PAS in the contralesional hemisphere, causing an increase in corticospinal tract excitability. There was no significant effect of PAS in the ipsilesional hemisphere. Furthermore, the degree of change in contralesional corticospinal excitability varied as a function of severity of upper-limb impairment, with individuals with more severe impairment showing greater contralesional corticospinal plasticity than more mildly affected individuals. Our findings suggest a compensatory increase in contralesional plasticity in individuals with greater damage to descending ipsilesional tracts.
LTP is proposed to underlie improvements in motor function in response to rehabilitation after stroke. 37 Accordingly, there has been a large research effort to understand how neuroplasticity relates to motor outcomes after stroke. 38 LTP after stroke has primarily been tested in vitro in animal models 37 ; methods to noninvasively assess LTP-like plasticity in humans are more limited. PAS shares cardinal features with Hebbian models of neural plasticity 39 ; PAS effects are topographically specific, 18 are timing 40 and intensity 41 dependent, and are mediated by N-methyl D-aspartate (NMDA)-receptor activation. 42 Of the available noninvasive brain stimulation techniques designed to modulate cortical excitability, PAS has the strongest mechanistic evidence as a model of LTP-like plasticity. 19 Additionally, PAS relies on associative polysynaptic plasticity from the relay of information from the somatosensory to the motor cortex, 18 modeling proposed mechanisms of sensorimotor integration and plasticity in motor learning.43,44 PAS, therefore, has significant potential to increase our understanding of the capacity for cortical remodeling after stroke; however, previous research had not examined PAS effects bilaterally in individuals with chronic stroke.23,24
Contralesional Hemisphere Plasticity
Across our sample, there was a significant increase in contralesional excitability in response to PAS. However, in the current study, when the sample was subdivided based on the presence of an ipsilesional MEP, we saw that the effect of PAS in the contralesional hemisphere was driven by individuals who did not have an ipsilesional MEP. Importantly, there were no differences in corticospinal excitability between MEP+/− groups at baseline; therefore, the significant increase in contralesional excitability in response to PAS in the MEP− group reflects an increased capacity for contralesional LTP-like plasticity, rather than reflecting increased contralesional excitability overall. Presence or absence of an ipsilesional MEP is a marker that has been shown to predict upper-limb motor outcomes in chronic stroke. 36 Absence of an ipsilesional MEP has previously been associated with increased contralesional excitability9,10 and contralesional sensorimotor cortical thickness 8 in chronic stroke. The current data complement these previous findings, suggesting that an increased potential for LTP-like plasticity mediates increases in contralesional cortical excitability or thickness. Rodent models have found that there is an increase in NMDA receptor activity45,46 and synaptic branching 47 in the contralesional hemisphere after a focal lesion. The converging evidence from rodent and human work suggests that the contralesional hemisphere is a site for compensatory increases in plasticity in response to stroke damage. Our findings provide novel data to a body of evidence from neuroimaging3,48,49 and neurophysiology6,13,50 work, suggesting that the dynamics of cortical excitability and plasticity depend on the severity of chronic motor impairment.
Contralesional plasticity also related to clinical measures of motor and somatosensory impairment in the paretic limb, with both greater motor and somatosensory impairment relating to a larger increase in contralesional corticospinal excitability in response to PAS. As the effects of PAS are dependent on the transmission of a peripheral somatosensory volley to the primary sensorimotor cortex, this technique provides a model of sensorimotor integration as a driver of cortical plasticity. Somatosensory impairment has not been as widely investigated as motor impairment in the stroke literature. 51 Somatosensory impairment is likely an essential component to understanding upper-limb motor outcomes; the integrity of the somatosensory system predicts upper-limb recovery 52 and predicts response to repetitive TMS in chronic stroke. 53 Our data provide evidence that the integrity of the somatosensory system, detected by monofilament thresholds, is also related to compensatory increases in contralesional plasticity. These findings emphasize the importance of considering sensorimotor integration in understanding poststroke motor outcomes because the integrity of sensory signaling might be key for plasticity-induced effects from existing therapies.
Ipsilesional Hemisphere Plasticity
There was no significant increase in corticospinal excitability in response to PAS in the ipsilesional hemisphere. It is well documented that peri-infarct tissue shows increased plasticity in the acute phase after stroke, 38 but ipsilesional plasticity may evolve over the course of recovery into the chronic stage. Work from Castel-Lacanal et al 24 showed that PAS effects in the ipsilesional hemisphere decrease from the subacute to the chronic stage of stroke recovery. Our data are congruent with these previous findings, suggesting that ipsilesional plasticity may be altered or attenuated in chronic stroke. In the chronic phase of stroke, motor recovery plateaus, but the capacity for motor learning54,55 remains; our data may indicate that the contralesional hemisphere is a site of plasticity in response to motor learning in individuals with chronic stroke; however, this speculation warrants confirmation through studies that include behavioral measures of motor learning. Premotor regions in the ipsilesional hemisphere are another candidate site of cortical remodeling in severe stroke, 56 and activity in the ipsilesional premotor and prefrontal regions have previously been linked to motor learning in individuals with chronic stroke. 57 It is important to note that our data on ipsilesional plasticity come only from individuals who have ipsilesional MEPs evoked from M1, and thus, the interpretation of our results in the ipsilesional hemisphere should be limited to individuals who have less complete injury of descending motor tracts. Future research should consider how impairment severity interacts with plasticity and motor learning in contralesional and nonprimary motor regions.
Consistent with previous work with PAS in healthy adults,58,59 we observed considerable individual variability in response to PAS, with some individuals showing a decrease in corticospinal excitability in response to PAS. A limitation of our data is that we did not collect sensory-evoked potentials (SEPs) to adjust the delivery of our TMS pulse to the timing of the ascending somatosensory volley. 60 SEP latencies may be delayed in the ipsilesional hemisphere in individuals with chronic stroke. 61 Delayed SEP latencies could affect PAS if the N20 SEP component is delayed to such a degree that it occurs after the TMS pulse is delivered because this would evoke a decrease in MEP amplitude, mimicking long-term depression (LTD)-like plasticity. 60 PAS protocols designed to induce LTD-like plasticity commonly use a N20 5-ms ISI.60,62 Previous EEG work in individuals with chronic stroke noted an average 2-ms delay in N20 latency, 61 which would not be sufficient to induce LTD-like effects in the ipsilesional hemisphere. Therefore, we are confident that a 25-ms ISI was appropriate for PAS in the ipsilesional hemisphere. However, because we did not collect SEP data on our participants, the possibility the PAS ISI was set to a nonoptimal interval for some study participants cannot be fully excluded. Future research using PAS in individuals with chronic stroke should specifically tailor PAS protocols to the SEP N20 latency within each cerebral hemisphere. Other sources of individual variability in response to PAS could include age, 63 fitness-level, 64 and brain-derived neurotrophic factor polymorphism.65,66 Although consideration of these factors was beyond the scope of the present study, they may provide interesting avenues of future exploration in relationship to motor outcome poststroke.
Relationships With Paired Pulse Measures
Paired-pulse TMS assesses the excitability of intracortical networks. 34 Previous work in individuals with chronic stroke has observed alterations in SICI 67 and ICF, 68 such that SICI is increased and ICF is decreased in the ipsilesional hemisphere, 69 although these results are not consistent across all individuals.67,68 Our data did not reveal any hemispheric differences in intracortical excitability in chronic stroke at baseline, in contrast to previous studies in this population.67-69 SICI relies on γ-aminobutyric acid (GABAA) receptor–mediated inhibition 70 ; the mechanisms of ICF are largely unknown but are thought to rely on glutamatergic signaling. 71 As GABA-mediated inhibition has been shown to decrease LTP-like plasticity,72,73 SICI is a candidate measure that may relate to response to PAS. 58 However, because there was no significant difference in SICI between hemispheres, it is possible that GABA-mediated inhibition was not altered to an extent to influence our PAS protocol in our sample of those with chronic stroke. Furthermore, we observed no effect of PAS on intracortical circuits, which is consistent with previous studies in healthy controls.21,42,74,75 It appears that changes in corticospinal excitability induced by PAS are not related to modulation of ICF or SICI in individuals with chronic stroke.
Conclusions
The current study addresses a critical gap in the chronic stroke literature, comprehensively assessing ipsilesional and contralesional neuroplasticity-like response to PAS in individuals with chronic stroke. Our findings support a bimodal balance-recovery model,12,76 which posits that individuals with a loss of structural reserve in descending ipsilesional pathways show a compensatory reliance on the contralesional hemisphere.12,15 In the present study, structural reserve was indexed both by the severity of clinical measures and the presence or absence of an ipsilesional MEP; these measures all related to a larger degree of change in contralesional corticospinal tract excitability in response to PAS.
Understanding how the potential for cortical plasticity is altered in chronic stroke has important implications for developing effective therapies in this population. There is significant interindividual variability in response to noninvasive brain stimulation protocols, 77 which can be understood in consideration of changes in underlying physiology in response to different degrees of injury. Repetitive TMS stimulation of the contralesional hemisphere disrupts paretic limb function in individuals with severe stroke.78,79 Furthermore, the structural integrity of the ipsilesional CST predicts response to noninvasive brain stimulation interventions; low ipsilesional CST integrity is associated with a negative response to contralesional inhibitory rTMS.15,80 The congruence of the data suggests that contralesional homotopic regions serve as a compensatory site of functional remodeling for individuals with more complete injury to descending ipsilesional motor pathways. This concept is described by the bimodal-balance model, which argues for variance in physiological adaptations in response to brain injury, depending on the extent of stroke damage.12,76 Our data support this model by providing evidence for increased capacity for neuroplasticity in the contralesional motor cortex in individuals with severe chronic stroke compared with individuals with mild stroke. Future research should examine how hemispheric responses to PAS relate to motor learning in the paretic limb and whether severity of limb impairment modulates these effects in order to further elucidate how cortical remodeling after stroke varies as a function of upper-limb impairment.
Footnotes
Acknowledgements
We are grateful to Dr Cameron Mang for his input on experimental protocol design and Dr Sue Peters for her assistance with motor function data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
