Abstract
Background. In a randomized control trial conducted in patients with Parkinson’s disease, a treadmill training program combined with virtual reality that targeted motor and cognitive aspects of safe ambulation led to fewer falls, compared with treadmill training alone. Objective. To investigate if the 2 types of training differentially affected prefrontal activation and if this might explain differences in fall rates after the intervention. Methods. Sixty-four patients with Parkinson’s disease were randomized into the treadmill training arm (n = 34, mean age 73.1 ± 1.1 years, 64% men, disease duration 9.7 ± 1.0 years) or treadmill training with virtual reality arm (n = 30, mean age 70.1 ± 1.3 years, 71% men, disease duration 8.9 ± 1.1 years). Prefrontal activation during usual, dual-task, and obstacle negotiation walking was assessed before and after 6 weeks of training, using a functional near-infrared spectroscopy system. Results. Treadmill training with and without virtual reality reduced prefrontal activation during walking (P < .001), with specific interactions related to training arm (P = .01), lateralization (P = .05), and walking condition (P = .001). For example, among the subjects who trained with treadmill training alone, prefrontal activation during dual-task walking and obstacle negotiation increased after training, while in the combined training arm, activation decreased. Conclusions. Prefrontal activation during usual and during more challenging walking conditions can be altered in response to 2 different types of training. The addition of a cognitive training component to a treadmill exercise program apparently modifies the effects of the training on the magnitude and lateralization of prefrontal activation and on falls, extending the understanding of the plasticity of the brain in PD.
Introduction
A striking feature of Parkinson’s disease (PD) is the decreased automaticity that stems from dopamine deficiency in the striatum. A recent model suggests that because of this reduced automaticity, patients with PD recruit additional brain networks, specifically cognitive prefrontal areas, even during simple tasks, as a form of compensation. 1 However, this cognitive compensatory mechanism may increase the risk of falls.2,3 When walking demands exceed prefrontal capacity, for example, under complex walking conditions such as dual-tasking and obstacle negotiation,4-7 the system may fail and a fall occurs. 1 Indeed, increased prefrontal activation during walking predicts future falls in highly functioning older adults. 8 Therefore, one can speculate that an intervention that improves components in the frontal-striatal circuitry may have a direct impact on the ability to walk in complex situations and reduce fall risk.
Exercise programs such as walking on a treadmill improve the gait of patients with PD.9,10 However, the effect of these unimodal interventions on cognition and complex walking performance is less pronounced. This led to the development of a multimodal treadmill training program augmented by virtual reality (TT + VR) that targets both motor and cognitive aspects of gait simultaneously, providing training in a stimulating environment that promotes obstacle negotiation.10,11 A recent randomized control trial found that among patients with PD, TT + VR reduced fall frequency, more than that seen in response to treadmill training (TT) alone and that this effect persisted after adjusting for disease severity. 12 The TT + VR training was designed to address deficits related to dual-task and obstacle negotiation, tasks that are related to prefrontal activity.3,6
Previous work showed that healthy adults increase prefrontal activity during complex walking conditions, as compared with the low complexity of usual walking.6,7 In contrast, patients with PD have higher levels of prefrontal activation already during usual walking, diminishing the ability to further recruit this brain area during more difficult tasks. 3 Consistent with the model noted above, this behavior may account for the lower performance during challenging walking conditions. Therefore, exercise may affect prefrontal cortex recruitment in 2 ways concurrently or separately: (1) increases the efficiency of neural control, which reduces activation in order to produce the same (or higher) level of usual walk performance, and (2) extends capacity, that is, the maximal ability to activate the prefrontal cortex as task difficulty increases.13-16
Evidence for neuroplasticity after exercise in patients with PD shows decreased corticomotor excitability, 17 increased grey matter volume, 18 and increased connectivity between cortical-subcortical regions after exercise.18,19 In a recent study, we showed that exercise can affect brain plasticity in mode-specific ways; however, these findings represent brain activation during motor imagery of walking that was performed while lying supine in the magnetic resonance imaging (MRI) machine. 20 Therefore, in the present study, we take one step forward to study actual walking. We use functional near-infrared spectroscopy (fNIRS), a tool that allows direct assessment of activation during actual walking, to assess the effects of training on prefrontal activation, and more specifically the effects of motor-cognitive training, and the different effects compared with motor training alone. We aimed to evaluate (1) the effects of gait training on prefrontal activation, (2) the effects of a simultaneous motor-cognitive training on prefrontal activation versus motor training alone, and (3) the relationship between training effects and fall risk in people with PD. We hypothesized that both training forms will reduce prefrontal activation, reflecting enhanced automaticity, but that TT + VR will reduce activation to a greater extent than the TT, mainly during more complex walking conditions.
Methods
Participants
This study was a substudy of the V-TIME project.11,12 It included only patients with PD from 2 sites, Tel Aviv Medical Centre in Israel and Radboud University Medical Centre in the Netherlands. A convenient sample of 34 patients with PD who participated in the TT training and 30 patients with PD who participated in the TT + VR training was included in this fNIRS substudy. Inclusion criteria were (1) diagnosed with PD, as defined by the UK Brain Bank criteria; (2) 60 to 90 years of age; (3) in Hoehn and Yahr stage II-III; (4) able to walk at least 5 minutes unassisted; and (5) taking any anti-Parkinsonian medication. Participants were excluded if they had psychiatric comorbidity, clinical diagnosis of dementia or other clinically significant cognitive impairment (Mini Mental State Exam score <24), a history of neurological disorder that could affect their performance, any orthopedic problems that may affect their gait, or had unstable medical condition including cardiovascular instability. 11 Recruitment was performed via reaching out to outpatient and related clinics of the geriatric medicine and neurology departments of Tel Aviv Medical Centre (n = 47) and Radboud University Medical Centre, Nijmegen, the Netherlands (n = 17). No differences in patient characteristics and assessments were found between the 2 sites. The study was approved by local ethical committees and was performed according to the principles of the Declaration of Helsinki. All participants gave their written informed consent prior to participation.
Experimental Design
A complete description of the study design can be found in Mirelman et al. 11 Briefly, after undergoing screening to confirm eligibility, participants were randomized to 1 of 2 arms of the study (TT + VR or TT) and then performed pretraining assessment that included tests of gait and prefrontal activation and cognitive evaluation. Participants in both training arms practiced 3 sessions per week for 6 weeks for approximately 45 minutes in each session. Training consisted of walking on a treadmill in both groups, while the TT + VR group was also required to negotiate virtual obstacles presented to them on a screen in front of the treadmill. Posttraining assessment was scheduled within 1 week from last training session. One single assessor in each site, blinded to training arm allocation, performed all assessments in the practical, self-reported on-medication state, at about the same time of day. The fNIRS signal processing was carried out by a blinded researcher at the Tel Aviv Medical Center.
The pre- and Posttraining Assessment Protocol
Participant characteristics and general physical and cognitive performance measures were obtained. Gait and prefrontal activation were assessed during 3 walking tasks: (1) usual walking, (2) walking while serially subtracting 3 seconds from a given 3-digit number (dual task), and (3) walking while negotiating obstacles (size of 30 cm width × 20 cm depth × 10 cm height). Each walking task was performed 5 times and each walk included 20 seconds of standing quietly, walking for 30 seconds in a 30-meter walkway, and standing quietly for another 20 seconds. These 20 seconds were sufficient to ensure return to baseline levels and to rule out possible effects of previous tasks. Rest periods were given between trials based on the participant’s needs. The cognitive evaluation consisted of the Mini Mental State Examination (MMSE) to assess global cognitive function, 21 and a computerized neuropsychological test battery that included several tests such as Go-no-go, Stroop, catch game, and long- and short-term verbal memory tasks. The software generated from these tests an index score for attention, executive function, visual spatial processing, and a general cognitive score (Mindstreams, NeuroTrax Corp, Israel). 22 In addition, the motor part of the Unified Parkinson’s Disease Rating Scale (UPDRS) was assessed to determine disease severity. 23
Functional Near-Infrared Spectroscopy
As described earlier,3,7,24 changes in oxygenated hemoglobin (HbO2) concentration in the prefrontal cortex were measured with the PortaLite fNIRS system (Artinis Medical Systems, Elst, the Netherlands) while performing the different walking tasks. HbO2 levels were selected as they were found to be more reliable and sensitive to changes in cerebral blood flow.25,26 Changes in deoxygenated hemoglobin were used to remove artifacts from the signal using the filter correlation–based signal improvement (CBSI).27-29 The system uses near-infrared light, which is transmitted at 2 wavelengths, 760 nm and 850 nm. Data was sampled with a frequency of 10 Hz. Two probes were positioned (by the same assessor in all evaluations) at a height of 15% of the nasion-inion distance from nasion and at 7% of the head circumference to the left and right from midline. In a previous study, MRI scans (T1 sequence) were performed to confirm that these locations target left and right Brodmann’s areas 10, that is, the dorsolateral and anterior prefrontal cortex (PFC).30,31
The PortaLite has 3 transmitters and 1 receiver, with transmitter-receiver distances of 30, 35, and 40 mm. A bandpass filter with frequencies of 0.01 to 0.14 Hz was used to reduce physiological noise such as heart beat (high-frequency noise ≥0.15 Hz) and signal drift (low frequency noise <0.01 Hz). To remove motion artifacts, a wavelet filter was used,27,28 followed by CBSI.27-29 HbO2 concentration signals of the 3 channels of each probe were then averaged, resulting in an HbO2 signal for the left and right PFC. For each trial, the average concentration of HbO2 during walking and during the 5 seconds of quiet standing just before the start of walking (referred to as the baseline) were calculated. Consistent with previous studies of fNIRS signals, each baseline concentration was subtracted from the average concentration during task performance to evaluate the relative change in HbO2 concentration during specific tasks.6,7,25,32-34 All trials were averaged per task, resulting in 2 HbO2 concentrations for each task (left and right PFC). HbO2 levels were stable across the 5 trials of walking in each condition, showing a consistent standard error of 0.073 to 0.079.
Behavioral Task Performance
As described previously,3,7 gait was measured using an electronic walkway with pressure sensors embedded in a carpet. The walkway was connected to a personal computer using PKMAS software (ProtoKinetics, Havertown, PA) for processing and data storing. In order to characterize the performance of usual walk, dual-task walk, and obstacle negotiation, 2 key aspects of walking that have been associated with PD pathology, that is, gait speed and stride length, were studied. 35 The gait changes reflect the performance change after training while the fNIRS findings provide support for brain plasticity. All motor and cognitive assessments pre- and posttraining were conducted at the same week of the fNIRS examination. In contrast, fall frequency posttraining reflects the number of falls occurred during the 6 months after finishing the training program.11,12
Statistical Analyses
Means and standard errors were calculated for all dependent variables. Differences in levels of HbO2 pre- and posttraining (time effect), between TT and TT + VR (training arm effect), between right and left prefrontal cortex (laterality effect), and between conditions (ie, usual walk, dual-task walk, and obstacle negotiation, condition effect) were analyzed using a linear mixed model (“linear mixed model 1”), including the interactions between time, training arm, laterality, and conditions effects. The factors age, gender, UPDRS-motor, gait speed pretraining, and global cognitive score pretraining were included to control for possible differences between training arms at baseline. To better examine the differences between training arms, we reduced degrees of freedom by conducting a second linear mixed model (“linear mixed model 2”) that compared the delta of HbO2 (post minus pre training) in each condition, while controlling for HbO2 level pretraining due to possible differences between training arms. For linear mixed models on HbO2 concentrations, random intercepts were allowed between participants since it improved model estimations. For all models, a covariance matrix was set for variance components. Post hoc test P values were Bonferroni corrected for multiple comparisons.
Changes in behavioral measurements such as gait speed, stride length, number of falls, and executive function after training were assessed using a third linear mix model (“linear mixed model 3”) while controlling for age, gender, and UPDRS-motor, factors that can mediate changes in performance. Random intercepts were allowed between participants since it improved model estimations, and a covariance matrix was set for variance components. Table 1 summarizes the 3 statistical models that were used. The association between prefrontal activation and fall rate was explored using Spearman correlation coefficients. Statistical significance was assumed when the P value was less than or equal to .05. Statistical analysis was performed using SPSS for Windows version 22.
Statistics Models.

Abbreviations: UPDRS, Unified Parkinson’s Disease Rating Scale; HbO2, oxygenated hemoglobin.
Results
Participants
As shown in Table 2, at baseline participants in both training arms were similar with respect to age, gender, disease duration, disease severity, MMSE, attention, executive function, visual spatial function, gait speed, and stride length.
Patient Characteristics Pretraining.
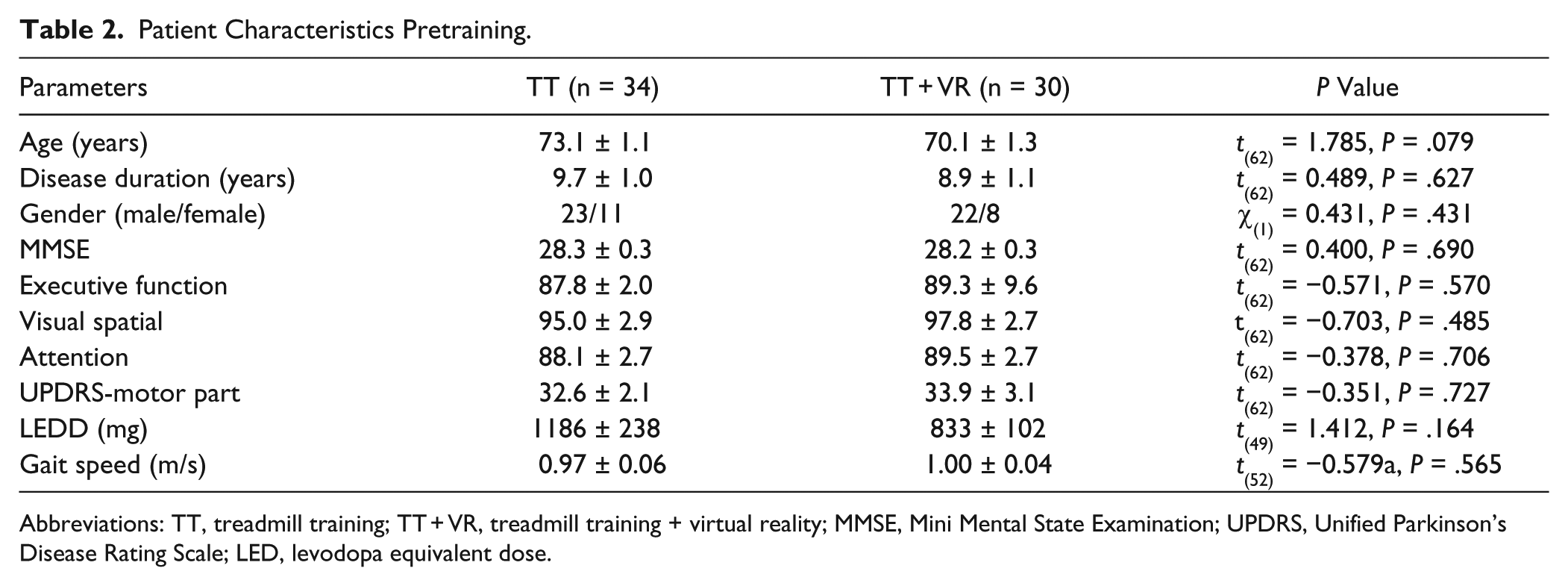
Abbreviations: TT, treadmill training; TT + VR, treadmill training + virtual reality; MMSE, Mini Mental State Examination; UPDRS, Unified Parkinson’s Disease Rating Scale; LED, levodopa equivalent dose.
Effects of Exercise Training on Prefrontal Activation
The linear mixed model 1 analysis revealed that treadmill training with and without virtual reality reduced prefrontal activation during walking in patients with PD (Time effect, F(1,562) = 14.123, P < .001). However, this effect was different between training arms (Time * Arm interaction, F(1,562) = 6.618, P = .010); patients in the TT + VR arm had a larger decrease than patients in the TT arm. Furthermore, changes in activation differed between the right and left prefrontal cortex (Time * Laterality interaction, F(1,557) = 3.790, P = .050), with the left prefrontal cortex presenting larger decreases than the right prefrontal cortex. In addition, prefrontal activation was different between the 3 walking conditions (Condition effect, F(2,557) = 7.136, P = .001), with complex walking conditions demonstrating higher activation than usual walking. No interaction with time was observed.
More specifically, the linear mixed model 2 analysis showed that patients in the TT + VR arm reduced prefrontal activation to a greater extent than patients in the TT alone (Arm effect, F(1,50) = 4.114, P = .048). However, as shown in Figure 1, these differences between training arms were specific to the right prefrontal cortex during complex walking conditions. While in the TT arm, HbO2 during dual task and obstacle negotiation increased after training, in the TT + VR arm HbO2 decreased after training (dual task: F = 4.327, P = .043; obstacle negotiation: F = 5.670, P = .021). No differences between training arms were found in the right prefrontal cortex during usual walking (F = 0.337, P = .564). In contrast to the right prefrontal cortex, both training arms showed reduced activation in the left prefrontal cortex after training (Laterality effect, F(1,248) = 5.787, P = .017). No differences in prefrontal activation due to affected side were found between the arms (P = .400). Reduced activation is presented as negative values in Figure 1.
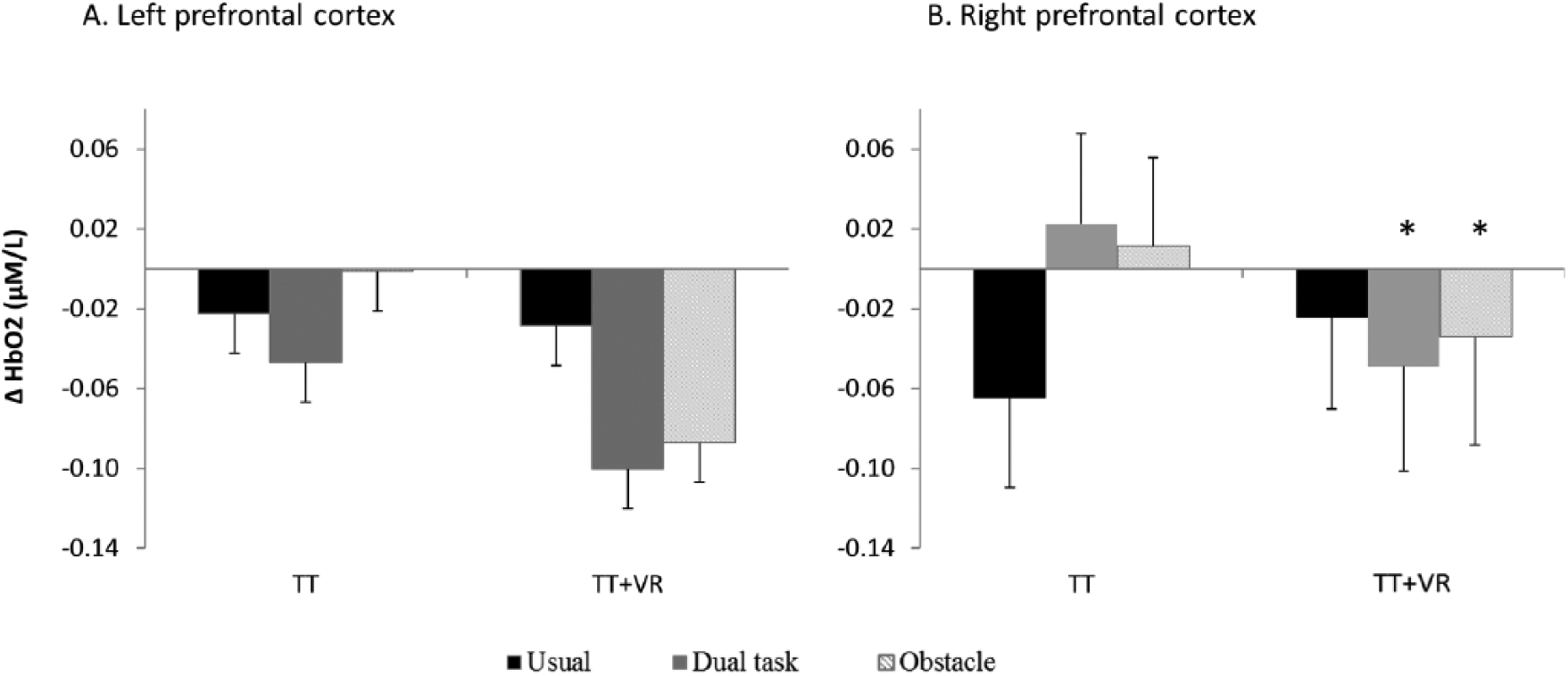
Change in left and right HbO2 level after exercise training in each training arm. A training-specific response is seen for the right prefrontal cortex, while for the left prefrontal cortex, both arms had a similar effect, reducing activation in all 3 conditions. The “*” indicates a significant difference between training arms.
Effects of Training on Behavioral Measures
Figure 2 presents the changes in gait performance, number of falls, and executive function after training in each of the training arms. Patients in both training arms showed improvements in gait measurements such as gait speed and stride length. Gait speed and stride length increased during usual walking (gait speed: F(1,53) = 6.178, P = .016; stride length: F(1,56) = 11.705, P = .001) and dual task walking (gait speed: F(1,54) = 4.159, P = .046; stride length: F(1,43) = 3.961, P = .053; Figure 2A). In addition, the number of falls 6 months postintervention was lower (F(1,98) = 4.386, P = .039; Figure 2B), and executive function scores increased after training (F(1,62) = 4.809, P = .032; Figure 2C). Although no significant differences between training arms were found for these behavioral outcomes, as shown in Figure 2, subjects in the TT + VR arm tended to have a greater reduction in number of falls and a larger increase in gait speed and executive function score, consistent with the findings in the overall study. 12 In addition, as presented in Figure 3, across both training arms a higher number of falls after training was significantly correlated with higher left prefrontal activation during usual walking after training (r = .312, P = .031), but not with right prefrontal activation (r = .110; P = .458).
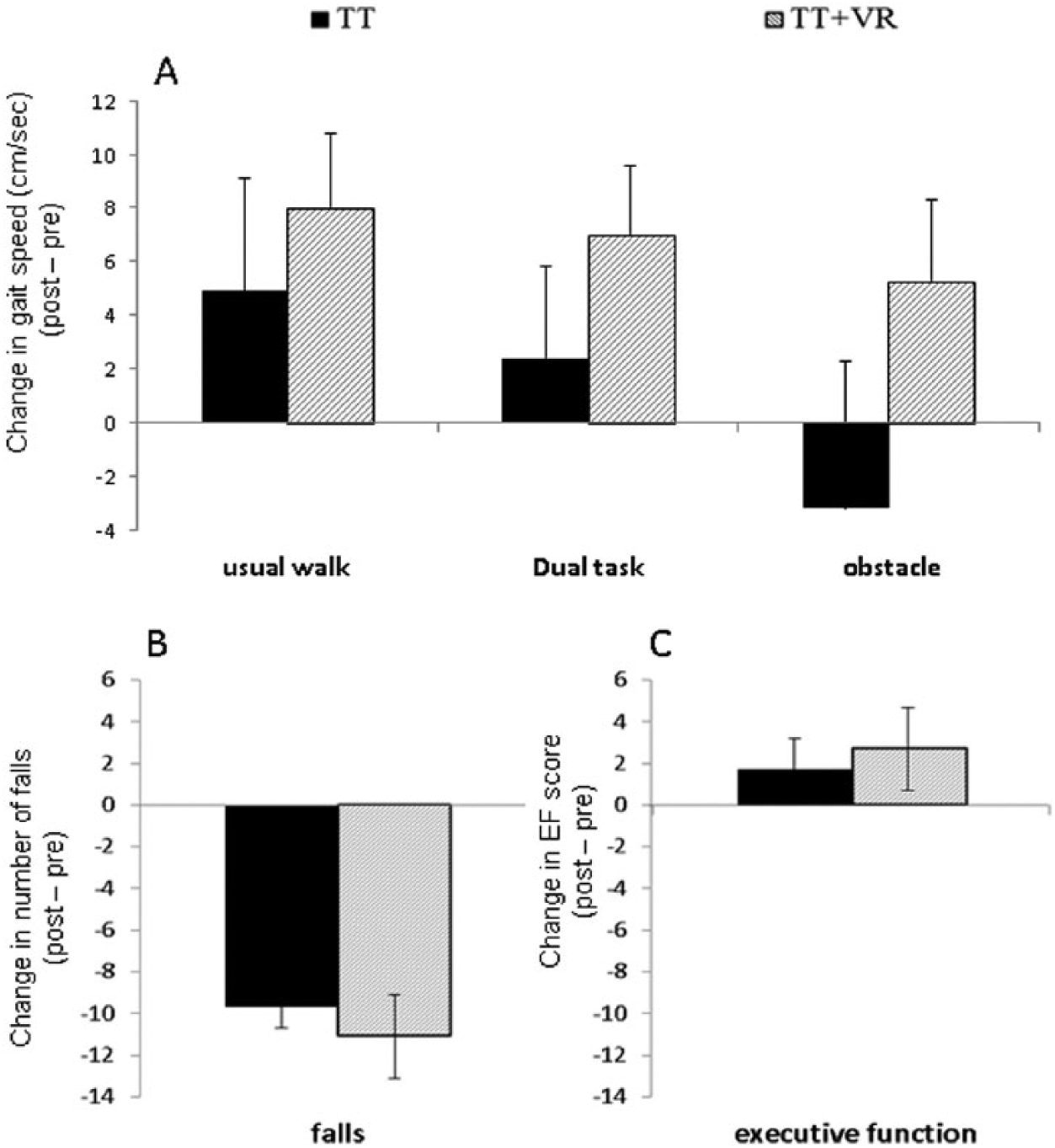
Effects of exercise training on behavioral measures in each training arm. Both training arms presented significant improvements in (A) gait speed during usual walk and dual task, (B) fall rates, and (C) executive function score after training. However, the TT + VR arm tended to improve more than the TT arm.
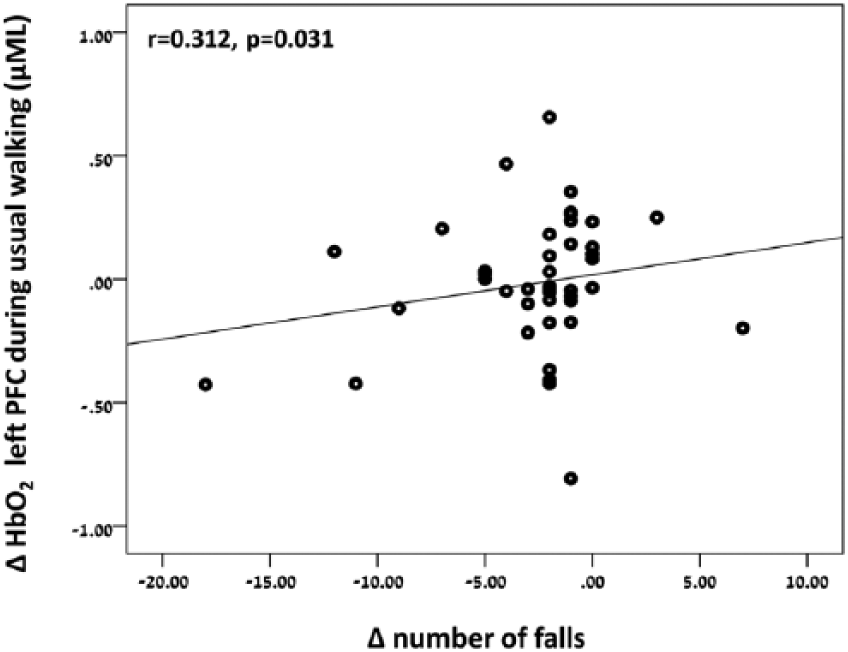
Correlation between the change in HbO2 after training (post minus pre training) in left prefrontal cortex during usual walking, and change in number of falls after training (6 months posttraining minus pre training).
Discussion
We investigated the changes in prefrontal activation after motor-cognitive training and motor training alone in patients with PD to better understand the role of prefrontal cortex in falls in PD, and the impact of different forms of exercise on brain function. Four main findings were observed: (1) gait training reduced prefrontal activation during usual walking and complex walking conditions, (2) TT + VR reduced prefrontal activation to a greater extent than TT alone, (3) different patterns of change between training arms were observed in the right prefrontal cortex during complex walking conditions, and (4) lower number of falls after training was associated with reduced left prefrontal activation in both training arms. Taken together, these findings suggest that motor-cognitive training may lead to a more efficient recruitment of the prefrontal cortex during the complex walking conditions that we encounter in everyday life.
Effects of Exercise on Prefrontal Activation
Accumulating evidence supports the idea that in aging and PD walking is less automated and requires reliance on cognitive resources to maintain performance.1,36 Increased activation of the prefrontal cortex with aging and further with PD during usual walking underscores the compensatory role of prefrontal cortex during walking.3,6 Our findings showing decreased prefrontal activation after training suggest that gait training improves automaticity of walking, which, in turn, lowers the reliance on cognitive resources during usual walking in patients with PD. These robust changes of an 18% reduction in prefrontal activation after training during real usual walking are consistent with our findings from an fMRI study, 20 and a large body of evidence that demonstrates that this form of exercise can enhance the walking pattern of patients with PD by enhancing the utilization of the striatal-thalamic-cortical-motor circuit responsible for automaticity of walking.9,37 As indicated from animal and human models, exercise can, to some degree, enhance neuroplasticity that promotes angiogenesis (growth of new blood vessels), neurogenesis (new functional neurons), and synaptogenesis (new synapses), all processes that improve efficiency and may contribute to reducing the required level of activation.38,39
In line with previous studies, pretraining prefrontal activation in patients with PD increased during usual walking and stayed at high levels of activation during the more complex walking conditions as dual tasking and obstacle negotiation, compared with healthy older adults, indicating high reliance on cognitive resources during different walking tasks. The reduced levels of activation observed after training, even in the challenging conditions, suggests that this is a reflection of better utilization of motor networks, more similar to that observed in healthy young and older adults.3,6 In addition, it reflects a more efficient activation and increased functional reserve capacity observed as higher executive function scores after training, which enabled better walking performance during these complex conditions. The reduced prefrontal activation during these challenging conditions may provide a possible explanation to the lower fall rates observed in both arms after training.
Interestingly, different effects of training were observed between the 2 training arms, with the TT + VR arm demonstrating a greater reduction in prefrontal activation than the TT arm. This difference was mainly observed in the right prefrontal cortex during complex walking conditions of dual tasking and obstacle negotiation. The TT + VR intervention promoted training of obstacle negotiation that included many repetitions of step modification in different situations and environments, components that are important to facilitate motor learning. In addition, patients in the TT + VR arm walked in a virtual environment that included different distractors that required divided attention, an important component in dual tasking. The use of virtual reality in treadmill exercise enhanced cognitive engagement by providing feedback, attentional demands, and motivation. Similarly, task-specific exercise such as obstacle negotiation requires greater motor preparatory processing, motor control, and set shifting, all key components ascribed to the prefrontal cortex. 40 Our findings suggest that the task-specific exercise provided by the virtual reality resulted in experience-dependent neuroplasticity in the prefrontal cortex, which may have increased efficiency and resulted in further reduced activation. Previous work showed that these changes in prefrontal activation are not simply the result of changes in cerebral blood flow due to a different intensity of training. 41
Lateralized Effects of Training
Interestingly, the training arms showed an opposite pattern. Patients in the TT arm showed increased activation in the right prefrontal cortex during complex walking conditions after training, while patients in the TT + VR arm demonstrated decreased activation after training. Specific roles have been attributed to the right and left sides of the prefrontal cortex. The left prefrontal cortex has been associated with learning of new movement sequences or improvement in motor performance through repeated practice.42,43 In addition, damage to left frontal regions showed disrupted coordination of movement that correlated with a disruption in accuracy of movement execution.43,44 Both training arms provided repeated practice of walking that emphasized coordination and accuracy of movements. We speculate that the reduced activation in left prefrontal cortex observed after training in both training arms may reflect a more efficient activation which reduced utilization of cognitive compensatory mechanisms.
Several lines of evidence demonstrate that right frontal regions play a dominant role in maintaining inhibitory control 45 and executing action modification,43,44 both necessary functions to perform walking while dual tasking and obstacle negotiation. In addition, the right prefrontal cortex has been implicated in sustaining actions that depend on directed attention,43,44 a function necessary for performing adequate dual tasking and obstacle negotiation. According to the specific roles attributed to right prefrontal cortex, it seems that the TT + VR improved efficiency of the right prefrontal cortex or reduced the need for right prefrontal compensation, both resulting in decreased activation after training. In contrast, the TT training alone did not require or challenge divided attention, inhibitory control, or step modification during training. As such, the effect on the right prefrontal cortex was not as pronounced in the TT arm as in the TT + VR arm.
On the other hand, reduced number of falls after training was correlated with decreased activation in the left prefrontal cortex during usual walking. This finding may explain the effect of training on fall rates in both training arms and may indicate that increased prefrontal activation already during usual walking is one of the key factors contributing to falls. As such, our results provide evidence that targeting prefrontal cortex via different interventions is important to reduce fall risk.
Taken together, our findings point at lateralization of training effects on prefrontal cortex, which depends on the specifics of exercise training properties. These results are consistent with the HAROLD model, 46 which suggests changes in lateralization of prefrontal activity during tasks that involve cognition with aging. These changes in prefrontal lateralization reflect a functional compensation strategy with aging.46,47 It can also explain previous findings in patients with PD that showed similar activation in both right and left prefrontal cortex during walking tasks. 3 Based on the HAROLD model, one can suggest that the prefrontal lateralization found after training provides evidence to the reduced use of compensatory activation in the prefrontal cortex and a more normalized pattern of prefrontal recruitment. The association between the reduced activation and reduced fall rate provides further reinforces that the neural effect observed has clinical utility. 8
Limitations and Future Directions
A main limitation of the present study is that the fNIRS system that was used included only 2 probes that were located specifically on prefrontal cortex (Brodman area 10), which is considered part of the dorsolateral prefrontal cortex (DLPFC). Having 2 probes placed only on the forehead limits our ability to show that the observed pattern of activation is specific to prefrontal cortex. As such, these conclusions should be taken cautiously and further study is needed. Although the location of the fNIRS probes had been confirmed with MRI scan, the fNIRS has lower spatial resolution than fMRI. In addition, exercise has been shown to increase cerebral blood flow. This may influence the fNIRS results that measure oxygenated hemoglobin concentration. However, the fact that both training arms included treadmill training at the same level of intensity rules out this possibility. While the reduction in fall rates was significantly correlated to activation in the left PFC across both groups, no significant correlations were found between behavioral measures and PFC activation between the training arms. However, this study was a substudy and was not powered to show changes in behavioral measures. These results may help estimate sample sizes in future studies. In addition, future work should explore longer term retention effects of motor cognitive interventions and include the evaluation of training effects on broader brain regions.
Conclusions
These intriguing findings provide new evidence on the potential of prefrontal cortex plasticity after exercise, even in the presence of PD. The results also have important implications for clinical practice and rehabilitation. Providing a combined motor cognitive training intervention may result in specific changes in prefrontal activation patterns that enhance functional abilities, reduce falls and fall risk, which in turn could potentially slow down deterioration in patients with PD. In addition, the ability to quantify changes in the fNIRS measures in response to exercise in the present study adds to the growing use of fNIRS as a neuroimaging tool, providing encouraging evidence to the translation of fNIRS in neurorehabilitation. 48
Footnotes
Acknowledgements
We would like to thank all the participants and partners in the V-TIME project including the Institute for Aging and Health, University of Newcastle (UNEW); Department of Neurosciences, Universita Degli Studi Di Genova (UNIGE); Department of Rehabilitation Sciences, Katholieke Universiteit Leuven (KULeuven); University of Sassari in Sardinia (UNISS); Inition 3D Technologies (INITION); Advanced Drug Development Services (ADDS); and Beacon Tech Limited (BTL) for their contribution.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AM, JMH, and NG have a patent Virtual Reality for movement disorders and/or treatment pending. All other authors have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded in part by the European Commission (FP7 Project V-TIME-278169).
