Abstract
Introduction. It is well documented that freezing of gait (FoG) episodes occur in situations that are mentally challenging, such as dual tasks, consistent with less automatic control of gait in people with Parkinson disease (PD) and FoG. However, most physical rehabilitation does not include such challenges. The purpose was to determine (1) feasibility of a cognitively challenging Agility Boot Camp–Cognitive (ABC-C) program and (2) effects of this intervention on FoG, dual-task cost, balance, executive function, and functional connectivity. Methods. A total of 46 people with PD and FoG enrolled in this randomized crossover trial. Each participant had 6 weeks of ABC-C and Education interventions. Outcome measures were the following: FoG, perceived and objective measures; dual-task cost on gait; balance; executive function; and right supplementary motor area (SMA)–pedunculopontine nucleus (PPN) functional connectivity. Effect sizes were calculated. Results. ABC-C had high compliance (90%), with a 24% dropout rate. Improvements after exercise, revealed by moderate and large effect sizes, were observed for subject perception of FoG after exercise, dual-task cost on gait speed, balance, cognition (Scales for Outcomes in Parkinson’s disease–Cognition), and SMA-PPN connectivity. Conclusions. The ABC-C for people with PD and FoG is a feasible exercise program that has potential to improve FoG, balance, dual-task cost, executive function, and brain connectivity. The study provided effect sizes to help design future studies with more participants and longer duration to fully determine the potential to improve FoG.
Background
The majority of patients with Parkinson disease (PD) eventually develop freezing of gait (FoG), an intermittent failure to initiate or maintain walking. Specifically, 26% of people with mild PD and 80% of those with severe PD are affected by FoG, and it is one of the most common reasons for falls and dependence.1,2 FoG leads to overall loss of mobility, increased social isolation, and decreased quality of life. 3 There is currently no consensus on the brain mechanism underlying FoG and no good treatment for this troubling feature of PD. 3
There is no singular brain region associated with FoG. Cortical, subcortical, limbic, and heteromodal systems (eg, attention and default networks) have been associated with FoG.4-6 For example, studies have reported white matter loss associated with FoG that includes both motor and nonmotor regions, such as striato-frontal tracts and connections of the cerebellum with the subthalamic nucleus and with the pedunculopontine nucleus (PPN). 7 Abnormalities in functional connectivity between the PPN and frontal/visual cortices, cerebellum, and corticopontine and pallidal-sensorimotor pathways in those who freeze compared with those who do not have also been reported.7,8
Our group has previously demonstrated that people with PD and FoG have decreased structural connectivity and increased functional connectivity between the right PPN and the right supplementary motor area (SMA) and right subthalamic nucleus compared with people without FoG or age-matched control participants.9,10 In fact, the stronger the functional connectivity between the right SMA and PPN, the worse the FoG (quantitatively measured during a turning in place task), suggesting that increased SMA-PPN connectivity may be a maladaptive compensation for loss of network structure. 9 This right-hemisphere pathway is part of both the cognitive inhibition network and the locomotor network, consistent with greater executive inhibition deficits in people with FoG compared with those without FoG.9-13
People with PD and FoG also have increased prefrontal cortex activity during walking and turning compared with people with PD without FoG, consistent with a loss of automaticity.11,12 FoG episodes occur in situations that are mentally challenging, such as dual tasks, 13 also consistent with less automatic control of gait in PD with FoG. 14 For example, dual-task cost on gait speed and stride length as well as step initiation is larger in people who freeze. 15 Dual-task interference in people who freeze is related to (1) larger right than left white matter structural connectivity of the SMA and PPN and (2) executive inhibition impairments.16-19 Thus, dual-task cost in people who freeze has been linked to abnormalities in executive inhibition-locomotor brain networks, suggesting that interventions targeting dual-task cost may also improve FoG.
We adapted our original Agility Boot Camp (ABC) program to incorporate additional executive-function tasks during mobility challenges known to be challenging for people with PD, now called Agility Boot Camp–Cognitive (ABC-C).18,20,21 We recently showed that rehabilitation using the ABC training for PD resulted in improvements in mobility such as turning, gait speed, and balance in people with PD.21,22 However, the original ABC and current physical therapy interventions might have limited success for people with FoG because the interventions do not directly address deficits related to executive function, specifically response inhibition, set-switching, and dual-task function. This is a substudy focusing only on people with PD who have FoG—part of a larger clinical trial to improve balance, gait, and cognitive function in people with mild to moderate PD. 20
The purpose of this proof-of-concept study was to determine (1) feasibility of the ABC-C program and (2) the effects of this intervention on FoG, gait automaticity (dual-task cost), balance, executive function, and functional connectivity between the PPN and SMA.
Methods
Participants and Study Design
Patients were eligible if they had mild to moderate severity of idiopathic PD (Hoehn and Yahr Levels II-III and FoG, defined as >0 on the New Freezing of Gait Questionnaire [NFOG-Q]). 23 Additional inclusion criteria required that participants be (1) between 50 and 90 years old; (2) without major musculoskeletal or peripheral or central nervous system disorders (other than PD) that could significantly affect their balance and gait; (3) without recent changes in medication and excessive use of alcohol or recreational drugs; (4) without history of structural brain disease, active epilepsy, stroke, or dementia that would interfere with ability to follow intervention and testing procedures; (5) able to stand or walk for 2 minutes without an assistive device; (6) without a medical condition that precludes exercise; and (7) without claustrophobia, severe tremor, or any health history (ie, implanted devices, deep brain stimulation) that would put the patient at risk near the magnetic resonance imaging (MRI) scanner. Here, we considered only the participants with PD and FoG, who were part of 2 clinical trials registered at ClinicalTrials.gov (NCT02236286 and NCT02231073 20 ). The Veterans Affairs Portland Health Care System (VAPORHCS) and Oregon Health & Science University (OHSU) ethics committees approved the study, and it was compliant with the Declaration of Helsinki. The protocol was approved by the OHSU/VAPORHCS joint Institutional Review Board (IRB No. 8979 and IRB No. 4131).
This was a randomized, single-blinded, crossover design, exercise trial. Each participant crossed over to the second intervention after 6 weeks of the first intervention and midpoint assessment the following week (see Figure 1). Final assessment was completed the week following the 6 weeks of the second intervention. Both interventions were designed to have the same quantity of intervention and were delivered by the same 2 trainers for all sessions. Participants completed the same battery of tests at baseline, midpoint, and final assessments (Figure 1). All other nonstudy interventions (ie, medication and other exercise) were kept as stable as possible and any changes monitored. The researchers who performed baseline, midpoint, and final assessments remained blinded to group assignment throughout the duration of the study. Additionally, internal regulatory reliability assessments between raters were performed; all sessions were also videotaped, and only experienced testers were used for the clinical ratings.
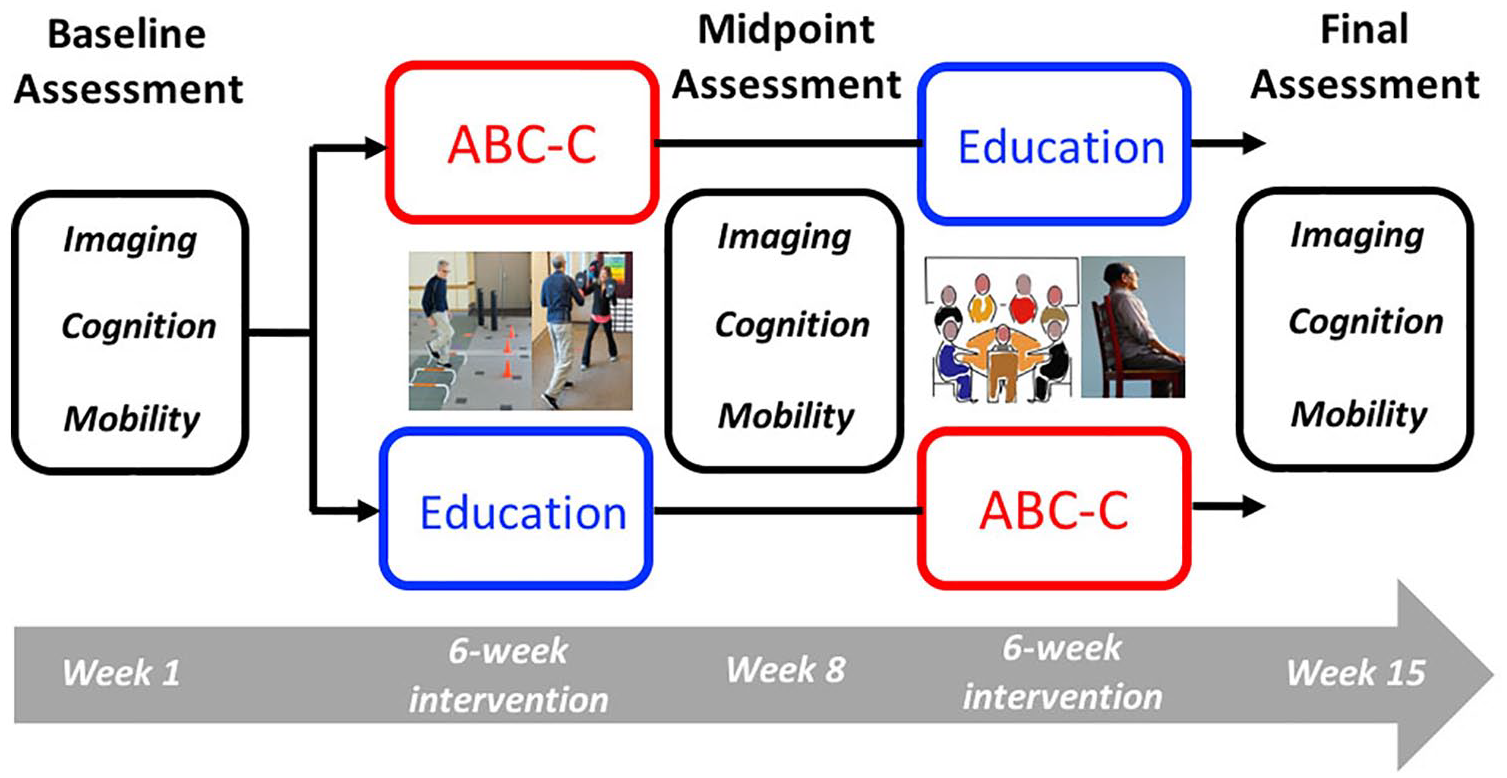
Study design.
Participants were randomly assigned, per a centralized database (REDCap), to either education first or exercise first after passing the phone screening procedure. Randomization was implemented by an independent statistician using a block size method. The certified exercise trainer, who was unblinded to group assignment, notified the participants of their group and schedule by phone. Participants were consented and enrolled immediately before baseline assessment.
Exercise
People participated in 80-minute, group (2-6 per group) exercise sessions 3 days per week for 6 weeks. The exercise protocol is an adaptation of our ABC exercise program for PD published previously.18,20,21 Briefly, the exercises are designed as a circuit and challenge cognitive-mobility skills known to be impaired in PD. Stations included (1) gait training, (2) functional skill training 24 (3) agility obstacle course, (4) lunges, (5) boxing, and (6) adaptive tai chi. 25 Each activity was chosen for its inherent focus on multidirectional movements, dynamic postural transitions, axial mobility, big movements, and whole-body motor sequencing. Each activity was progressed with cognitive challenges (see online Appendix 1 and Peterson et al 18 ). Participants engaged in each station for 10 to 20 minutes, with rest periods in between stations, as needed. Each participant progressed from beginning to intermediate to advanced stages at each station at their own pace for both motor and cognitive challenges (online Appendix 1). Because people with PD are known to have difficulty with self-initiated movements, we deliberately focused on movements without external cues. The exercise class structure and progressions were led by a trained and experienced exercise trainer with oversight from a licensed physical therapist. Trained research assistants stood nearby and assisted participants who self-reported frequent falls or had observed instability during the first exercise session.
Education
The education program was a chronic disease education program developed by our research team to teach patients with PD how to live better with their chronic condition. Briefly, it included content and discussion of topics on developing a care team, sleep, nutrition, mood, medication, and stress management. Classes consisted of the same group of people (2 to 6 per class) meeting with the same exercise trainer for 80-minute sessions, once a week, for 6 weeks. To match the dose of the education intervention with the exercise intervention, participants were also given relaxation recordings to be used at home 5 days per week for 30 minutes for an overall education dose of 240 min/wk, similar to the exercise dose.
Compliance was recorded for both exercise and education at each session. Progression was tracked for each of the 6 stations, with each progression accounting for a point (online Appendix 1). Additionally, participants stated their level of perceived exertion (0-10 scale) after each exercise session. The level of self-reported exertion was recorded to determine the level of challenge of the program and to determine if people were progressively challenged during the exercise over time. We did not use this for analysis, but only to determine if people with FoG could increase exertion in this type of exercise program. For education, participants recorded compliance for the relaxation sessions in a logbook, and the instructor record attendance at each weekly education session.
Procedures and Outcome Measures
The larger protocol and mobility assessment are described in the protocol publication of this clinical trial. 20 All assessments were scored in the practical off-levodopa state (12 hours withdrawal of anti-Parkinson medication). Imaging and cognitive tests were assessed one day, and clinical, patient-reported, and mobility outcomes were assessed on a second day. Each participant had 8 inertial sensors (APDM, Inc, Portland, OR), worn over the sternum, lumbar spine, and bilaterally on the wrists, shins, and feet while completing the walking, turning, and mini Balance Evaluation System Test (mini-BESTest) trials. As part of the larger study, participants completed several other objective tests of balance, mobility, and cognition 20 but were not analyzed as part of this smaller project.
The primary outcome measures for this study were the following: perceived FoG using the NFOG-Q 23 and a quantitative FoG ratio. 26 The NFOG-Q is a self-assessment of a person’s experience with FoG. The FoG ratio is an objective measure of FoG severity extracted from the anteroposterior acceleration sensed by 2 inertial sensors on the shins while turning in place 360° to the right and then to the left, for 1 minute. The FoG ratio was calculated as the square of the power in the 3- to 8-Hz band (trembling freeze band) divided by the square of the power in the 0.5- to 3-Hz band (stepping movement band). 26
The dual-task cost on gait speed and stride length was calculated as well as the cognitive dual-task cost. 18 In both single task (ST) and dual task (DT) conditions, participants were instructed to walk at a comfortable pace back and forth continuously between 2 lines 7.6 m apart. In the DT condition, participants were instructed to walk while reciting every other letter of the alphabet. The ST condition was always completed before the DT condition, and the ST condition was always completed after performing the cognitive test for 1 minute while seated. To evaluate the performance on the cognitive task, the number of responses and the number of errors were recorded for both the seated and DT walking conditions. Accuracy was calculated as the percentage difference (total error) in the walking compared with the sitting condition.
Balance impairments were measured with the mini-BESTest, a sensitive measure of dynamic balance, commonly used to quantify balance impairments in people with PD. 27 This is a 14-item clinical test (maximum score of 28) that measures 4 domains of balance, with each item scored 0 to 2, with higher scores indicating better performance.
Measures of cognition included the Scales for Outcomes in Parkinson’s Disease–Cognition (SCOPA-COG) and tests targeting inhibition and set-shifting. The SCOPA-COG consists of 10 items divided into 4 domains: memory, attention, executive function, and visuospatial function. 28 Scores range from 0 to 43, with higher scores reflecting better performance. The inhibition/set-shifting domain of executive function was assessed with the Trail Making Test Ratio B/A (TMT B/A), 29 Flankers reaction time in incongruent/congruent conditions, and Stroop tests (ratio of naming colors/incongruent colors-words). 30
A subset of 13 participants were measured at all 3 intervention time points with resting state functional MRI (rsfMRI), an imaging method that evaluates regional brain area relationships that exist in a resting state.6,8,9 Using this method, we measured right SMA-PPN connectivity. Our previous publication showed that the functional connectivity between SMA and PPN was abnormally high in people with PD with FoG compared with those without FoG or control participants. So we used the same regions of interest in the SMA and PPN to investigate whether resting state functional connectivity can be reduced by our ABC-C program. 9
Briefly, a 10-mm sphere was created at the peak coordinates of the SMA on the midline (x = 0, y = −7, z = 55). 31 A 6-mm sphere was created bilaterally for the PPN (x = ±6, y = −30, z = −19).10,32 The mean time series from the SMA BOLD signal at rest was correlated with the mean time series from each of the PPN regions of interest using the Pearson coefficient of correlation (r) for each participant. The procedures for the rsfMRI were previously described, and details are in online Appendix 2.9,33
Statistical Analysis
Feasibility was assessed by dropouts, adverse events, compliance, and progression in this challenging agility program. We hypothesized that the exercise intervention would result in larger improvements in freezing, dual-task cost, balance, executive function, and brain connectivity than the placebo, control education intervention. Because we had 3 observations for each participant (baseline, midpoint, and final; Figure 1), we calculated the change caused by the intervention as Midpoint − Baseline for the first intervention and Final − Midpoint for the second intervention. The effect of exercise and education interventions were reported as the standardized response mean 34 (SRM) for each measure. The SRM, which was calculated as the mean change between before and after each intervention period divided by the SD of the change, 34 and the 95% CI were reported. 35 Generally, an SRM value of 0.20 represents a small, 0.50 a moderate, and 0.80 a large effect of the intervention. 35 To assess whether the change in FoG measures was associated with the baseline FoG ratio, we used the Pearson correlation coefficient (in cases of skewness of the data, we used a log-transform).
Finally, as an exploratory analysis (see online Appendix 4), we used a linear mixed-model design that included an indicator of intervention effects (education vs exercise), order effects (exercise or education first), and period effects (sequence, education-exercise or exercise-education, differences) to determine whether the “difference in change” differed between exercise and education (significance threshold of 0.05). The intervention term reflected the main focus of the study: to determine whether the effects of exercise differed from the effects of education. A random-effects term was included for participants. All statistical analyses were performed in MATLAB R2018b (The Mathworks Inc, Natick MA) using the Statistics and Machine Learning Toolbox.
Results
Feasibility of ABC-C
A total of 108 potential people with PD and FoG were screened (Figure 2). Among them, 62 were not included for the following reasons: 37 were excluded—comorbidities (28), deep brain stimulation (8), recent medication change (1)—and 25 declined to participate. We enrolled 46 participants with FoG, with 25 randomly assigned to the exercise first group and 21 to the education first group (Figure 2). Clinical characteristics did not differ between randomized groups, except for a higher Montreal Cognitive Assessment (MoCA) in the exercise-first group (P = .04; Table 1). The subset of 13 people with MRI were on average 69.3 ± 9.5 years old, with Movement Disorder Society–Unified Parkinson’s Disease Rating Scale Part III score of 50.6 ± 17.6, postural instability-gait disturbance (PIDG) of 8.1 ± 4.0, Hoehn and Yahr score of 2.7 ± 0.09, and MoCA score of 25.5 ± 4.4. Dropouts during interventions included people who discontinued the intervention (7) or were lost to follow up (1; Figure 2). Overall, 17% of people dropped out over the course of the study. Specifically, 24% dropped out during the exercise portion, whereas 9% dropped out during the education session. Participants dropped out for the following reasons: 2 sustained injuries outside of classes (low-back injury and sciatica), 1 had a heart attack outside of classes, 1 had a medical procedure, and 1 had a urinary tract infection. One participant fell during their fourth exercise class, resulting in a hip fracture, and 1 participant did not want to commute to classes anymore. In addition, 1 participant was lost to follow-up and could not be contacted. The only serious adverse event was the fall with hip fracture. This adverse event occurred during the gait station when emphasis was placed on taking long strides. The participant was spotted by a research assistant but fell while making a turn. There were also 4 minor adverse events that did not result in dropouts: 3 fell during the exercise class (no medical intervention required) and 1 fell getting out of their car. No participant reported medication changes or major life, health, or activity changes. We asked all participants to come to testing in the practical off state, at least 12 hours since their last dosage of medications. Only 2 people came in for testing after taking their medications that morning, so they were sent home and rescheduled for another day that week.
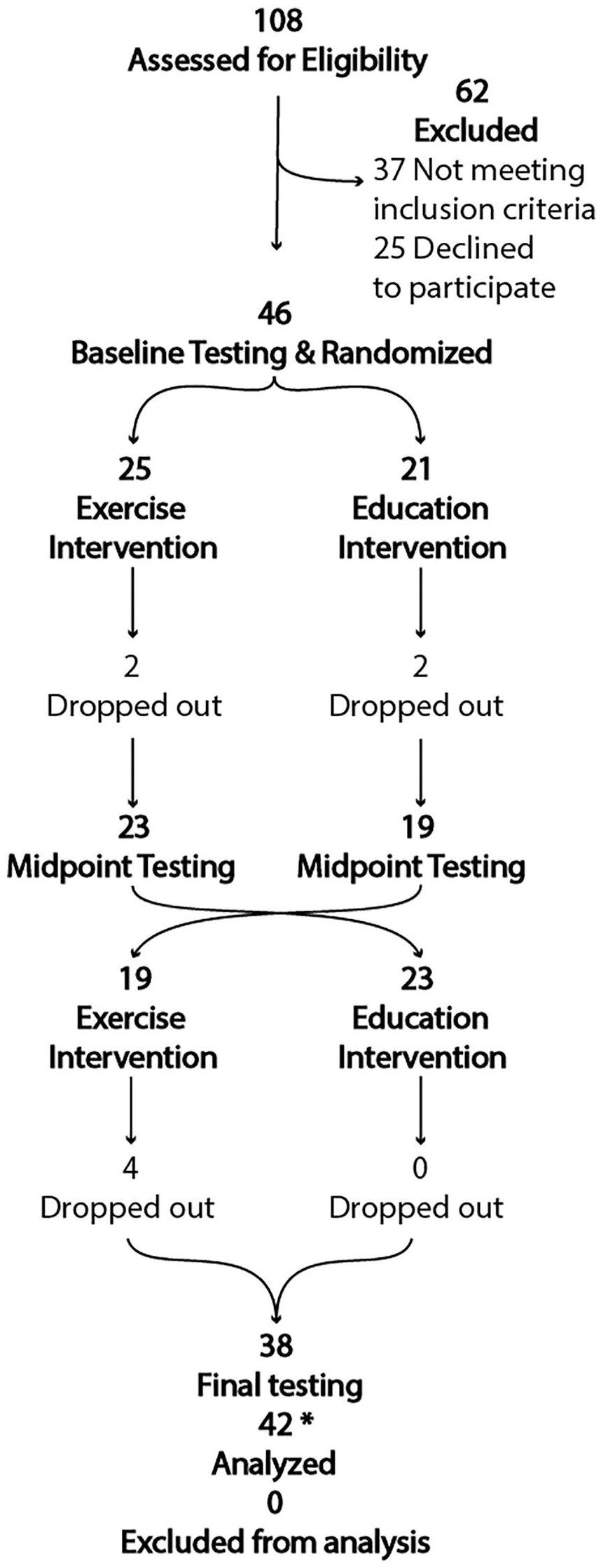
Consort diagram.
Participant Characteristics.
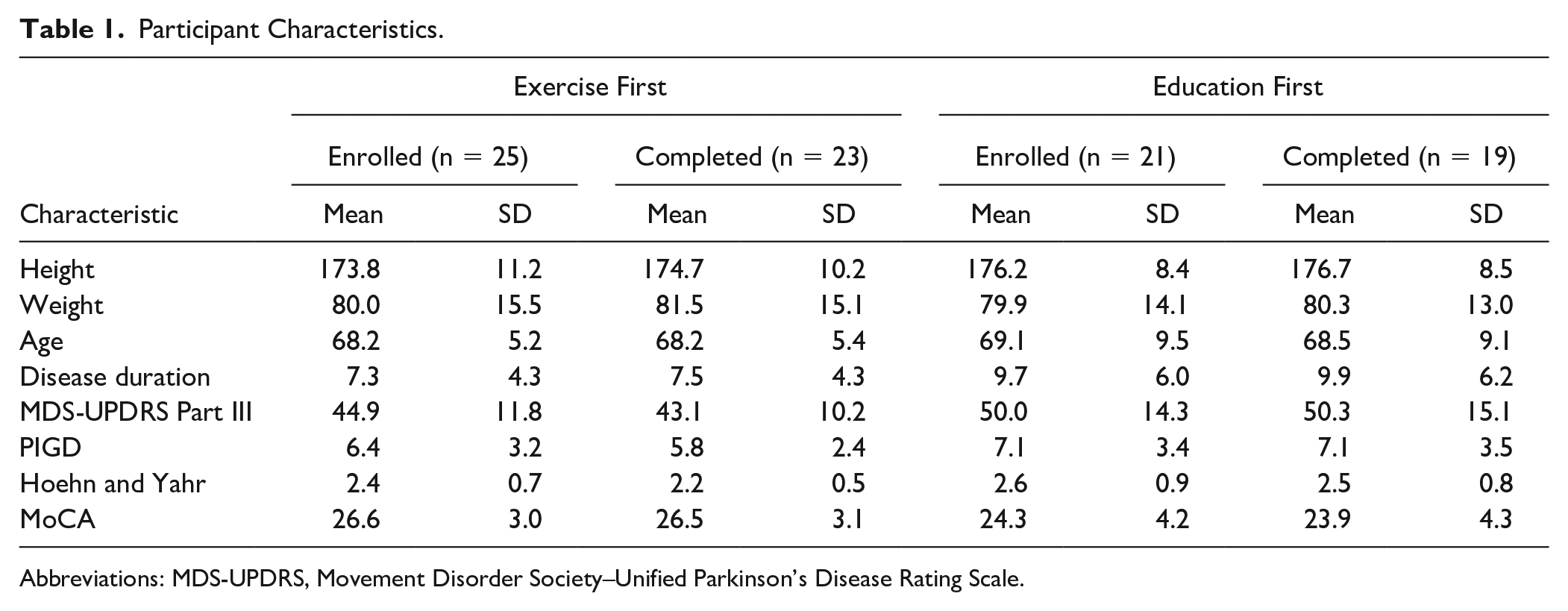
Abbreviations: MDS-UPDRS, Movement Disorder Society–Unified Parkinson’s Disease Rating Scale.
The exercise session compliance was 90%, whereas the education session compliance was 80%, including both group class attendance and home relaxation practice (94% and 66%, respectively). Progression in mobility challenge, as documented by the exercise trainer, had a starting average of 34% maximal difficulty the first week and incrementally increased to 67% of the possible points for progression. Cognitive progressions averaged from 33% to 85% possible difficulty level between the first and final weeks. For the perceived exertion scale of 0 to 10, participants increased from, on average, 4.3 to 6.9 over 6 weeks. No recorded changes in medications or exercise routines were reported during the study.
Freezing of Gait Severity
The NFOG-Q showed a moderate effect size after exercise (SRMExercise = 0.42 and SRMEducation = −0.12; for the P value, see online Appendix 4; Figure 3A and Table 2). The FoG ratio showed small effect size (SRMExercise = 0.04 and SRMEducation = 0.23). However, the change in FoG ratio was significantly associated with the baseline FoG ratio. Higher (worse) FoG ratios at baseline were associated with larger improvements with exercise (r = −0.41; P = .01) but not with education (see online Appendix 3 for regression figures).
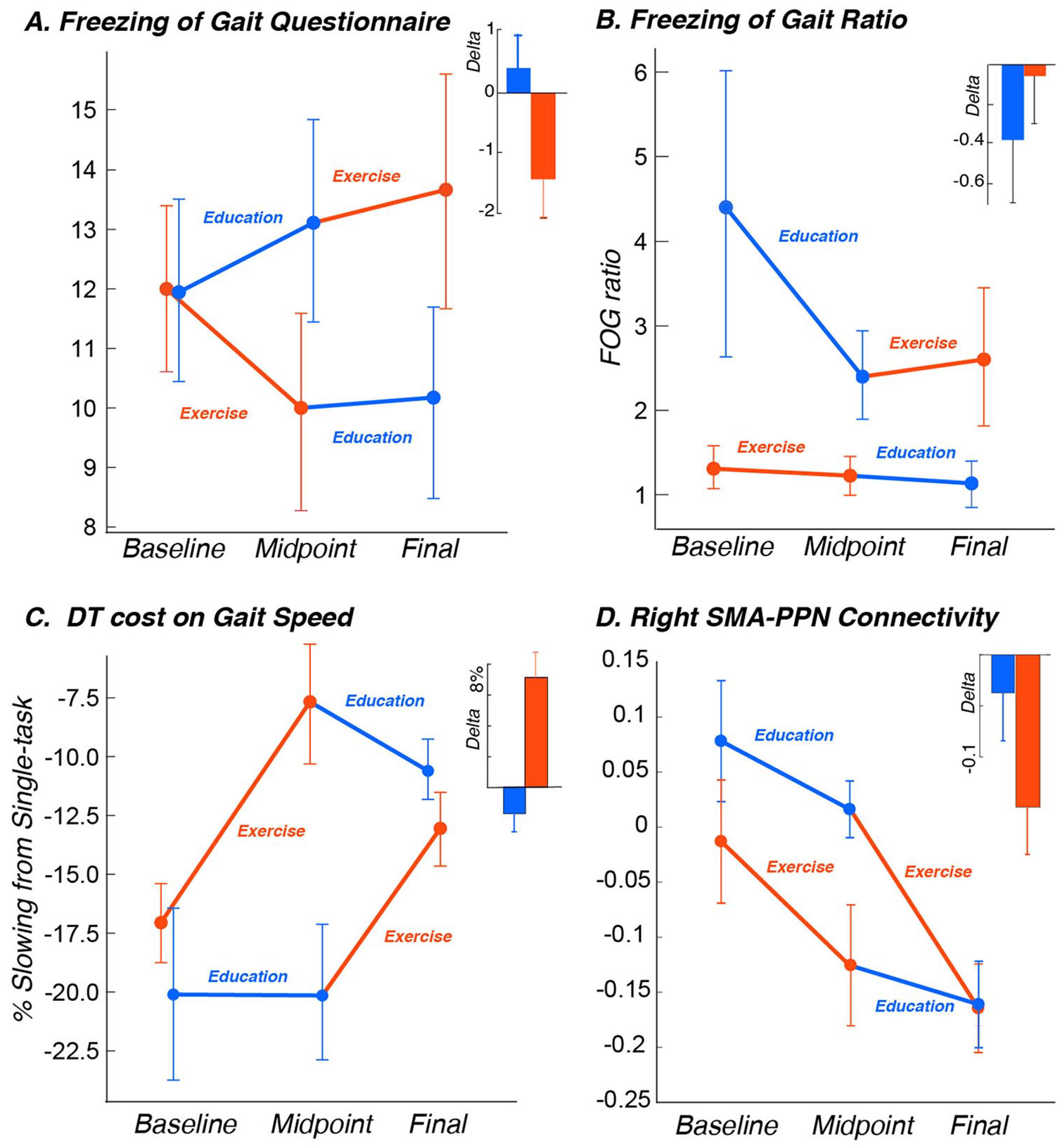
Mean (and SEM) over the 3 time points in the 2 groups (exercise first and education first) and bar graphs (mean ± SEM) of the δ for (A) NFOG-Q, (B) FoG ratio, (C) dual-task cost on gait speed, and (D) SMA-PPN connectivity.
Mean and SD of the δ for Each Outcome Measure. a
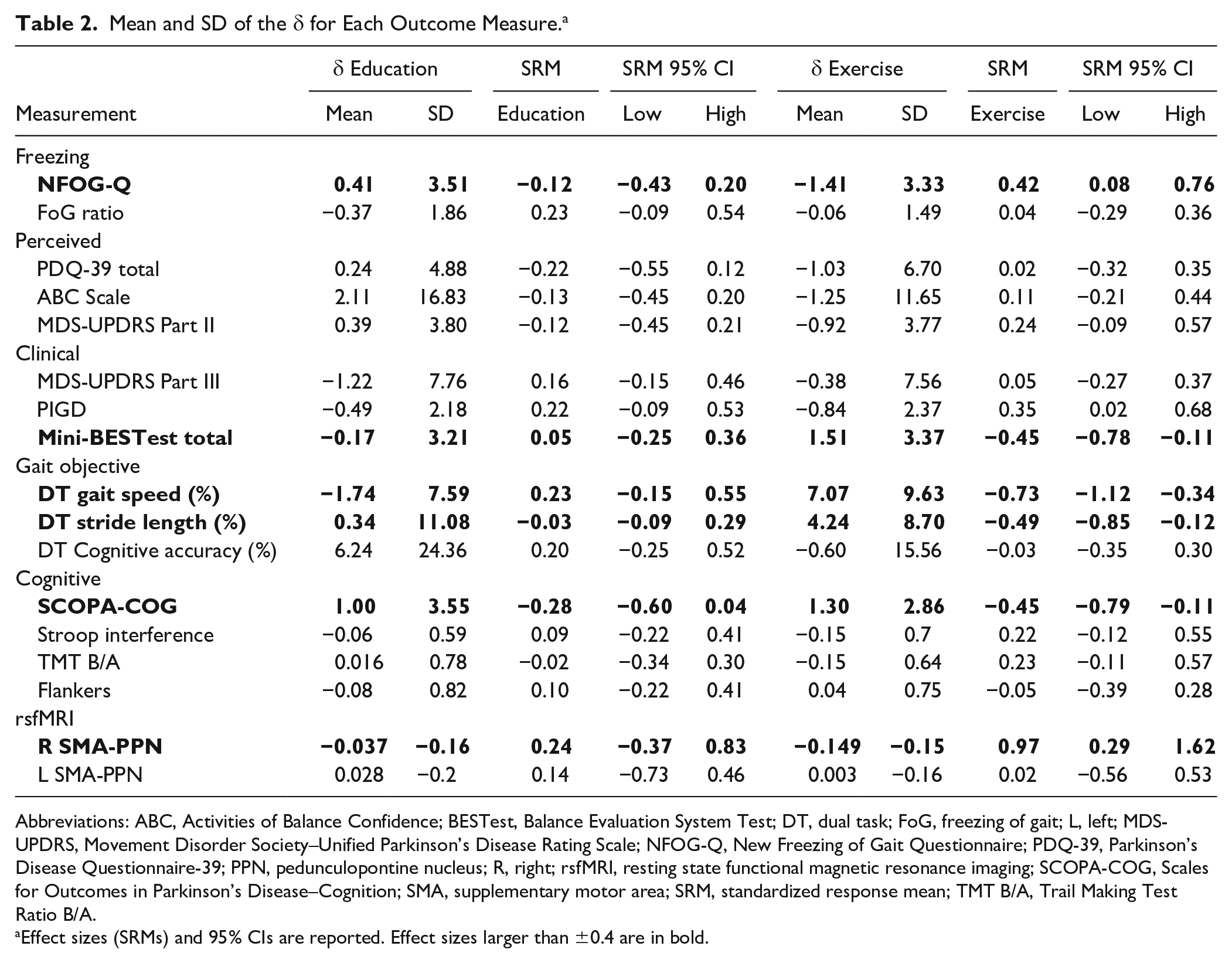
Abbreviations: ABC, Activities of Balance Confidence; BESTest, Balance Evaluation System Test; DT, dual task; FoG, freezing of gait; L, left; MDS-UPDRS, Movement Disorder Society–Unified Parkinson’s Disease Rating Scale; NFOG-Q, New Freezing of Gait Questionnaire; PDQ-39, Parkinson’s Disease Questionnaire-39; PPN, pedunculopontine nucleus; R, right; rsfMRI, resting state functional magnetic resonance imaging; SCOPA-COG, Scales for Outcomes in Parkinson’s Disease–Cognition; SMA, supplementary motor area; SRM, standardized response mean; TMT B/A, Trail Making Test Ratio B/A.
Effect sizes (SRMs) and 95% CIs are reported. Effect sizes larger than ±0.4 are in bold.
Dual-Task Cost on Gait
The dual-task cost on gait speed showed a large effect size after exercise but not after education (SRMExercise = −0.73 and SRMEducation = 0.23); see Figure 3C. The dual-task cost on stride length showed a moderate effect size after exercise (SRMExercise = −0.49 and SRMEducation = −0.03). Finally, the effect sizes on the accuracy of the cognitive task while walking were small (details are given in Table 2).
Balance Impairments
The overall mini-BESTest showed a moderate effect size after exercise but not after education (SRMExercise = −0.45 and SRMEducation = 0.05; details are given in Table 2).
Executive Inhibition Function
The SCOPA-COG showed a moderate effect size after exercise (SRMExercise = −0.45 and SRMEducation = 0.28), whereas the TMT B/A, the Stroop interference score, and the Flankers only showed small effect sizes after exercise as well as after education (details are given in Table 2).
Right SMA-PPN Connectivity
The right SMA-PPN showed a large effect size only after exercise (SRMExercise = 0.97 and SRMEducation = 0.24); see Figure 3D. Specifically, after exercise, but not after education, the connectivity between the right SMA and PPN was reduced in the 13 participants who had imaging. The left SMA-PPN showed very small effect sizes with the interventions (Figure 3D and Table 2).
Discussion
The main finding from this study was that a small-group, agility exercise intervention with cognitive challenges, ABC-C, is a feasible rehabilitation intervention for people with PD who have FoG. The study has provided effect sizes for FoG, dual-task cost, balance, executive inhibition, and right SMA-PPN connectivity, which can be used to power future clinical trials.
Freezing of Gait
This is the first exercise study to examine if an objective measure of FoG could be improved. We found some improvement in perceived FoG but no change in the objective measure of FoG. Although a strength of the study was in assessing people with PD off their dopaminergic medication, the perception of FoG severity may come from the recollection of on periods at home. Similarly, a recent article showed that cognitive training, twice a week for 7 weeks, was effective in improving the NFOG-Q but only when assessed in the on state. 36 Additionally, we found that those with the most severe FoG, measured objectively, improved with the ABC-C exercise (online Appendix 3). Specifically, the worse the FoG ratio was at baseline, the larger the improvement in FoG ratio after exercise, but not after education. However, we cannot identify the number of patients who improved because we do not have a threshold severity for the objective measure. Future studies should determine the clinically important difference for both the NFoG-Q and the FoG ratio in the same patients.
Previously a handful of small rehabilitation studies focused on improving FoG have had mixed results. A study of a bike-walking device reported reduced FoG in some, but not all, people with FoG. 37 Likewise, a study of 20 people with PD practicing dual tasking on a virtual-reality device while stepping in place on a balance platform reported some improvements in dual-task cognitive and gait measures in people with FoG but not with the FoG-Q. 38 Of note, these studies did not have a placebo or control intervention group. Although some positive results have been shown for the effects of dance versus no intervention on balance in people with FoG,39,40 a change in FoG, itself, was not significant in 2 recent meta-analyses on the topic. 33 A recent review 41 on robot-assisted gait training for FoG detailed 4 studies showing beneficial results on FoG, but the studies were primarily of low quality.
Dual-Task Gait
The effect of ABC-C on dual-task cost on gait speed showed a large effect size (−0.73). The improvement in dual-task cost on gait in people with FoG in our study is consistent with a growing body of evidence that dual-task function can be improved with training and practice in people with PD as well as healthy elderly controls.42-44 A short (nonsystematic) review recently highlighted the importance of training, rather than avoiding, dual-task situations and recommended the need for larger controlled trials. 45 Subsequently, a large, well-controlled trial assessed the effects of dual-task training in a group of 121 people with PD and found that regardless of how the dual-task training was administered, people with PD improved in other, unique dual-task situations, thereby suggesting that dual-task performance can be transferred to daily life situations. 45 In this large study, however, the subgroup of people with FoG also improved in dual-task cost on gait measures after training, but the improvements were not retained because they were in those without FoG. 45
Balance Impairments
The mini-BESTest score improved a small to moderate amount (SRM = −0.45) with exercise compared with the control education intervention. This improvement was primarily related to improvement in the anticipatory postural adjustment items in the mini-BESTest (SRM = −0.45), which may be expected because participants focused on increasing the size and speed of mobility but practiced neither postural responses nor standing balance. Anticipatory postural adjustments are known to be more abnormal in people with PD who have FoG compared with those without, especially under dual-task conditions. 46
Executive Function
Apart from a moderate effect size in the general SCOPA-COG test, the other executive tests (TMT, Flankers, and Stroop) only showed small effect sizes. It is unclear whether potential improvements did not transfer to the paper/computer cognitive tests or whether the tests were not sensitive enough to detect small changes over 6 weeks of practice. In fact, participants were able to progress in the difficulty of their cognitive challenges during the agility training, as detailed in online Appendix 1. Two earlier systematic reviews on the topic of exercise reducing cognitive dysfunction in PD47,48 reported a handful of studies suggesting improved cognition after exercise but also reported evidence of reporting and selection bias; variability on dosing, timing, and intensity of the exercise; and overall varying quality of studies. However, both these systematic reviews suggested that although the evidence was not strong, there were positive trends worth future trials given the impact of cognitive dysfunction on people with PD, their caregivers, and the costs of care. 49
Brain Connectivity
The change in neural connectivity between SMA and PPN, although with only 13 people, showed the largest SRM at 0.97. A power analysis showed that we would achieve 80% power to detect this difference in a similar crossover trial with 34 participants (17 in each group) and 90% power with 46 participants. Here, we focused specifically on 2 regions of interest because our previous studies demonstrated reduced structural connections as well as increased functional connectivity between right SMA and PPN in patients who froze compared with those who did not or control participants.9,10 Our a priori hypothesis was that the abnormally increased functional connectivity between right-sided SMA and PPN in people with FoG was maladaptive and could be reduced with exercise, associated with a reduction in FoG. A larger study should determine whether these changes in connectivity between specific brain regions are associated with the extent of FoG reduction or improvement of dual-task cost.
Previous studies have also shown changes in brain functional connectivity and functional imaging with exercise in people with PD50-54 but not specifically in people with FoG and not in the brain circuitry we investigated here. For example, forced-rate, lower-extremity pedaling over 8 weeks showed significantly increased functional connectivity between the primary motor cortex and the ipsilateral thalamus that was retained for 4 weeks after exercise in people with PD.50,55 This change in functional connectivity was correlated with change in pedaling rate, providing a plausible mechanism for efficacy of high-rate exercise for bradykinesia. Results of our mobility-cognitive agility training program also agree with other studies in people with PD looking at the effects of dual-task training on brain activity. A study comparing treadmill training versus treadmill training while using virtual reality to avoid obstacles, considered to be a dual-task condition, showed different effects on brain activity after each intervention. Specifically, brain activity using fMRI mental imagery showed that only the group who practiced walking with virtual reality showed lower activation in frontal brain regions after training, 53 consistent with more automatic, less cortical control. However, this study did not measure functional network connectivity or consider FoG or cognitive changes.
The current study has several limitations, including the small sample size—specifically, only 13 individuals with rsfMRI before and after both interventions. Although we consider it a strength that we tested in the off levodopa state to better reflect FoG, future studies should consider testing participants both off and on levodopa. Another limitation is measuring FoG with a questionnaire and a turning task in the laboratory. Because FoG is very context dependent, quantifying FoG under natural, daily-living situations with continuous monitoring may provide more sensitive measures of change. Heterogeneous group exercise classes may not be as effective as grouping people with PD according to disease severity or freezing status. Although an effort was made to match the ABC-C exercise and control education groups in minutes per week of intervention, some of the education protocol involved relaxation practice at home; so although the dose might match between conditions, the overall contact time with staff was not equal and compliance was lower with the meditation at home. Exercise required 3 in-person visits per week, whereas education required just 1 visit per week, which may have contributed to the higher dropouts during exercise. The lack of a washout period may have been a limitation, and this may have influenced the variability of the results. However, we did not expect a strong aftereffect after such a short intervention. A 6-week intervention for complex gait and balance disorders in a chronic disease may be too short. In fact, reduction in falls with exercise in PD appears to require at least 6 months of exercise. 56 Finally, although our overall dropout rate was 17%, most of the dropouts occurred during the exercise portion of the study. Although the exercise-first group had lower MoCA scores than the education-first group, we did not find any relationship regarding lower cognition relating to more dropouts. Specifically, the 2 who dropped out from exercise first had a mean MoCA score of 27.5, and the 6 who dropped out from the education-first group had a mean MoCA score of 25.2. The mean of the people who did not drop out was very similar; exercise first = 26.5 and education first = 23.9.
In conclusion, the ABC-C for people with PD and FoG is a practical, feasible group exercise program that has potential to improve balance, dual-task cost, and brain connectivity. Whether the ABC-C is effective for FoG in particular is still not clear, and perhaps a longer trial (ie, 6 months) and/or added cueing-based exercise57,58 is needed. Overall, more work with longer, larger trials is needed to determine the potential for rehabilitation to improve FoG in Parkinson disease.
Supplemental Material
Appendix2 – Supplemental material for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease
Supplemental material, Appendix2 for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease by Laurie A. King, Martina Mancini, Katrijn Smulders, Graham Harker, Jodi A. Lapidus, Katrina Ramsey, Patricia Carlson-Kuhta, Brett W. Fling, John G. Nutt, Daniel S. Peterson and Fay B. Horak in Neurorehabilitation and Neural Repair
Supplemental Material
Appendix3 – Supplemental material for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease
Supplemental material, Appendix3 for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease by Laurie A. King, Martina Mancini, Katrijn Smulders, Graham Harker, Jodi A. Lapidus, Katrina Ramsey, Patricia Carlson-Kuhta, Brett W. Fling, John G. Nutt, Daniel S. Peterson and Fay B. Horak in Neurorehabilitation and Neural Repair
Supplemental Material
Appendix_1 – Supplemental material for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease
Supplemental material, Appendix_1 for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease by Laurie A. King, Martina Mancini, Katrijn Smulders, Graham Harker, Jodi A. Lapidus, Katrina Ramsey, Patricia Carlson-Kuhta, Brett W. Fling, John G. Nutt, Daniel S. Peterson and Fay B. Horak in Neurorehabilitation and Neural Repair
Supplemental Material
appendix_4 – Supplemental material for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease
Supplemental material, appendix_4 for Cognitively Challenging Agility Boot Camp Program for Freezing of Gait in Parkinson Disease by Laurie A. King, Martina Mancini, Katrijn Smulders, Graham Harker, Jodi A. Lapidus, Katrina Ramsey, Patricia Carlson-Kuhta, Brett W. Fling, John G. Nutt, Daniel S. Peterson and Fay B. Horak in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
Thank you to Fuzhong Li who provided a short adaptation of his “Tai Ji Quan: Moving for Better Balance” program and to Dr. Becky Farley who provided a short adaptation of her PWR!Moves® program for the Agility Boot Camp. We also thank Julie Carter for her help in designing the education classes; Natassja Pal, and Peter Martin for help with recruitment and data collection; Peter Fino and Carolin Curtze for help in data collection; and Nancy Nelson and Nancy Barlow for help in running the classes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Horak has an equity interest in APDM, a company that may have a commercial interest in the results of this study. This potential conflict of interest has been reviewed and managed by the Research and Development Committee at the VA Portland Health Care System and Oregon Health and Science University. They have put in place a plan to help ensure that this research study is not affected by the financial interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by (1) Department of Veterans Affairs Grant No. I01 RX001075 (FBH), (2) NIH: NIA Grant No. 2R01AG006457 (FBH), (3) Collins Trust Grant (BWF), and (4) the Oregon Clinical and Translational Research Institute (OCTRI), NIH Grant No. UL1 RR024140.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
