Abstract
Walking is a complex motor behavior with a special relevance in clinical neurology. Many neurological diseases, such as Parkinson’s disease and stroke, are characterized by gait disorders whose neurofunctional correlates are poorly investigated. Indeed, the analysis of real walking with the standard neuroimaging techniques poses strong challenges, and only a few studies on motor imagery or walking observation have been performed so far. Functional near-infrared spectroscopy (fNIRS) is becoming an important research tool to assess functional activity in neurological populations or for special tasks, such as walking, because it allows investigating brain hemodynamic activity in an ecological setting, without strong immobility constraints. A systematic review following PRISMA guidelines was conducted on the fNIRS-based examination of gait disorders. Twelve of the initial yield of 489 articles have been included in this review. The lesson learnt from these studies suggest that oxy-hemoglobin levels within the prefrontal and premotor cortices are more sensitive to compensation strategies reflecting postural control and restoration of gait disorders. Although this field of study is in its relative infancy, the evidence provided encourages the translation of fNIRS in clinical practice, as it offers a unique opportunity to explore in depth the activity of the cortical motor system during real walking in neurological patients. We also discuss to what extent fNIRS may be applied for assessing the effectiveness of rehabilitation programs.
Introduction
Walking is one of the most fundamental motor functions in humans,1-3 often impaired in some focal neurological conditions (ie, stroke), or neurodegenerative diseases, such as Parkinson’s disease (PD). 4 Worldwide almost two thirds of people over 70 years old suffer from gait disorders, and because of the progressively ageing population, an increasing pressure on health care systems is expected in the coming years. 5
Although the physiological basis of walking is well understood, pathophysiological mechanisms in neurological patients have been poorly described. This is caused by the difficulty to assess in vivo neuronal processes during overt movements.
During the past 20 years, functional magnetic resonance imaging (fMRI) has been the preferred instrument to investigate mechanisms underlying movement control 6 as well as movement disorders. 7 fMRI allows measuring the blood oxygenation level-dependent (BOLD) signal that, relying on variations in deoxy-hemoglobin (deoxyHb) concentrations, provides an indirect measure of functional activity of the human brain. 8 Patterns of activation/deactivation and connectivity across brain regions can be detected with a very high spatial resolution for both cortical and subcortical structures. This technique, however, is characterized by severe limitations and constraints about motion artifacts and only small movements are allowed inside the scanner. This entails dramatic compromises on the experimental design and on the inclusion/exclusion criteria. Multiple solutions have been attempted to overcome such limitations. For instance, many neuroimaging studies have been performed on the motor imagery,9,10 but imaging can be different from subject to subject, 11 and imagined walking and actual walking engage different brain networks. 12 Other authors have suggested the application of virtual reality, 13 and there have been a few attempts to allow an almost real-walking sequence while scanning with fMRI.14,15 Additional opportunities to investigate the mechanisms sustaining walking control include the use of surrogate tasks in the scanner as proxy of walking tasks, 16 or to “freeze” brain activations during walking using positron emission tomography (PET) radiotracers, which allow the retrospective identification of activation patterns, albeit with some uncertainties and low spatial and temporal resolution. 12
Therefore, until now there has not been an ecological way to noninvasively assess neurophysiological correlates of walking processes in gait disorders.
Functional near-infrared spectroscopy (fNIRS) is becoming an important research tool to assess functional activity in special populations (neurological and psychiatric patients) 17 or for special tasks.18-21 fNIRS is a noninvasive optical imaging technique that, similarly to fMRI, measures the hemodynamic response to infer the underlying neural activity. Optical imaging is based on near-infrared (650-1000 nm) light propagation into scattering tissues and its absorption by 2 major chromophores in the brain, oxy-hemoglobin (oxyHb) and deoxyHb, which show specific absorption spectra depending on the wavelength of the photons. 22 Typically, an fNIRS apparatus is composed of a light source that is coupled to the participant’s head via either light-emitting diodes (LEDs) or through fiber-optical bundles with a detector that receives the light after it has been scattered through the tissue. A variation of the optical density of the photons measured by detectors depends on the absorption of the biological tissues (Figure 1A). Using more than one wavelength and applying the modified Beer-Lambert law, it is possible to infer on the changes of oxyHb and deoxyHb concentrations. 23 fNIRS has a number of definite advantages compared to fMRI, its major competitor: (a) it does not pose immobility constrains, 25 (b) is portable, 26 (c) allows recording during real walking, 27 (d) allows long-lasting recordings, (e) it does not produce any noise, (f) it makes possible the investigation of brain activity during sleep, 28 (f) it allows to obtain a richer picture of the neurovascular coupling as it measures changes in both oxyHb and deoxyHb concentration with high temporal resolution (up to milliseconds). High temporal resolution is usually not mandatory for the investigation of the hemodynamic response whose dynamic takes at least 3 to 5 seconds, but it can be useful for the study of transient hemodynamic activity like the initial dip 29 or to detect subtle temporal variations in the latency of the hemodynamic response across different experimental conditions.19,21,30 The major drawback of fNIRS in comparison to fMRI is its lower spatial resolution (few centimeters under the skull) and its lack of sensitivity to subcortical regions.18,19 However, this might be considered a minor limitation, as there is a large body of evidence suggesting that (a) cortical mechanisms take place in walking, 31 (b) the organization of the motor system is distributed along large brain regions, 32 and (c) the function of subcortical structures is mirrored in the cerebral cortex. 33
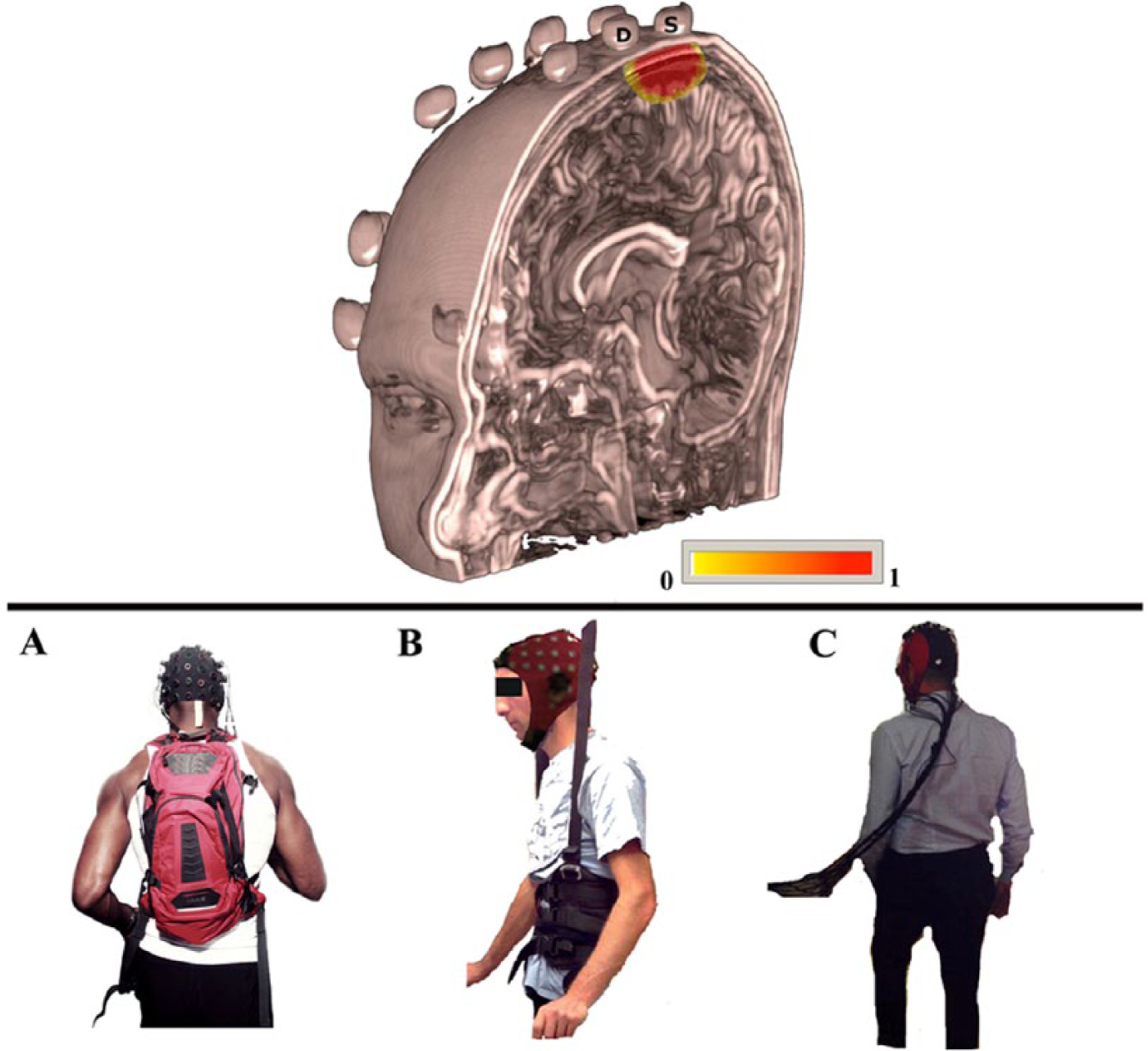
Illustration of penetration depth of near-infrared light into the tissue in a probe configuration used to investigate motor performances during walking task (upper row). The picture shows brain reconstruction from a high-resolution anatomical MRI. The spheres placed over the skull correspond to vitamin E capsules employed during the MRI to mark the positions of the optodes and to allow the coregistration of the individual anatomy together with the optode position. In this illustration, only the photons propagation from one source (S) to one detector (D) have been simulated. The yellow-red scale indicates the degree of sensitivity 74 for the considered source-detector pair to the head/brain structures. (A, B, and C) Lower row: Examples of fNIRS experimental device used for assessing brain activity during real walking tasks. These fNIRS approaches included either commercial device, such as (A) wireless portable fNIRS system (NIRx; Germany) or support systems for treadmill walking activity with body weight support 24 (B) or with free movement range (C).
Overall, fNIRS has proved to allow a reliable investigation of walking functions in healthy young and elder subjects, 34 while in gait disorders this functional brain imaging technique is emerging as a promising area of research that would help to better characterize in vivo pathophysiological mechanisms underlying dynamic postural control. For this reason, we aimed at providing an overview of this recent literature deducing whether there is enough evidence to suggest a broader application of this technique in a clinical context.
Methods
Information Sources, Search Strategy, and Eligibility Criteria
This systematic review was conducted following the PRISMA guidelines. 35 To perform a systematic literature search, we selected studies browsing different databases (PubMed, Medline, Web of Science, Scopus) combining 3 search fields (“fNIRS,” “walking” and “gait” or “movement” “disorders”), from inception to September 30, 2016. In order to ensure that at least one of the terms could be found in the results, the search fields were connected with “AND.” All terms in one search field were linked with the conjunction “OR.” Search string was structured as follows: “fNIRS” OR “functional near-infrared spectroscopy” - AND - “gait” OR “walk” OR “walking” OR “postural control” OR “cycling” - AND - “movement disorders” OR “gait disorders.” To increase the likelihood that all the relevant studies were identified, further articles were included from hand search, starting from the reference list of previously retrieved articles. Articles were considered for inclusion only if (a) they enrolled subjects with the following neurological disorders: PD or Parkinsonisms (as defined by the UK Brain Bank criteria 36 ) and stroke; (b) they described experimental protocol that included a real walking task, cycling or postural control task; and (c) they used fNIRS system with more than 2 channels or measurement points. Articles were excluded if they considered subjects with history of other neurological or psychiatric disorders such as Tourette’s disorder or essential tremor. The literature search yielded a total of 489 results. After removing all duplicates, 111 articles were screened and considered eligible for the review. The full texts of these articles were reviewed and the inclusion criteria were examined. Finally, only 12 studies were included in quantitative synthesis. The approach is presented in Figure 2.
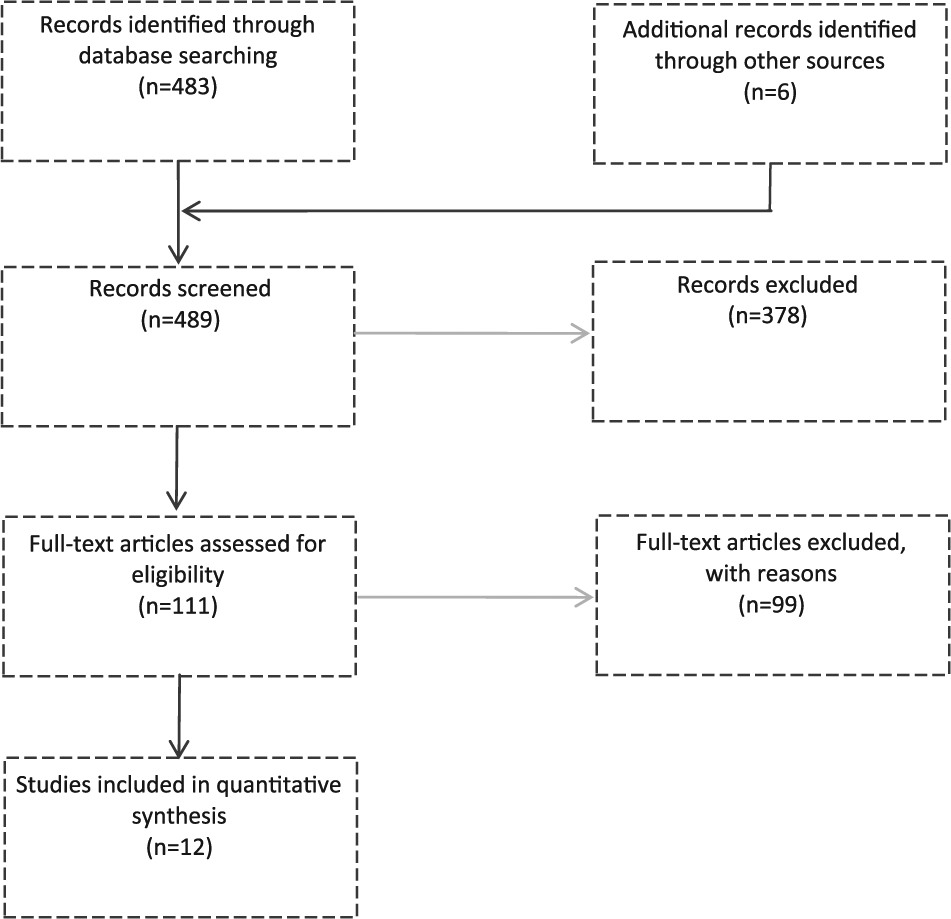
Flow chart of the search strategy.
With the aim to focus our research on the application of fNIRS to patients with specific gait disorders (ie, stroke and PD) during real walking, postural control, or cycling task, we summarized and discussed each study, contextualizing its results along the existing knowledge (and the usefulness of this neuroimaging method as well) in the neurological realm. For this purpose and for facilitating comparisons, we summarized all relevant clinical and neuroimaging findings in Table 1.
Results
Three articles37-39 were excluded for the limited number of measurement channels (2-channels37,38 or 1-channel 39 ). Twelve articles satisfied our search criteria and were included in this review. Eight articles applied fNIRS for investigating walking-related functional activity in patients with stroke, whereas 4 recent articles studied PD patients (Table 1).
fNIRS Applications in Gait Disorders.
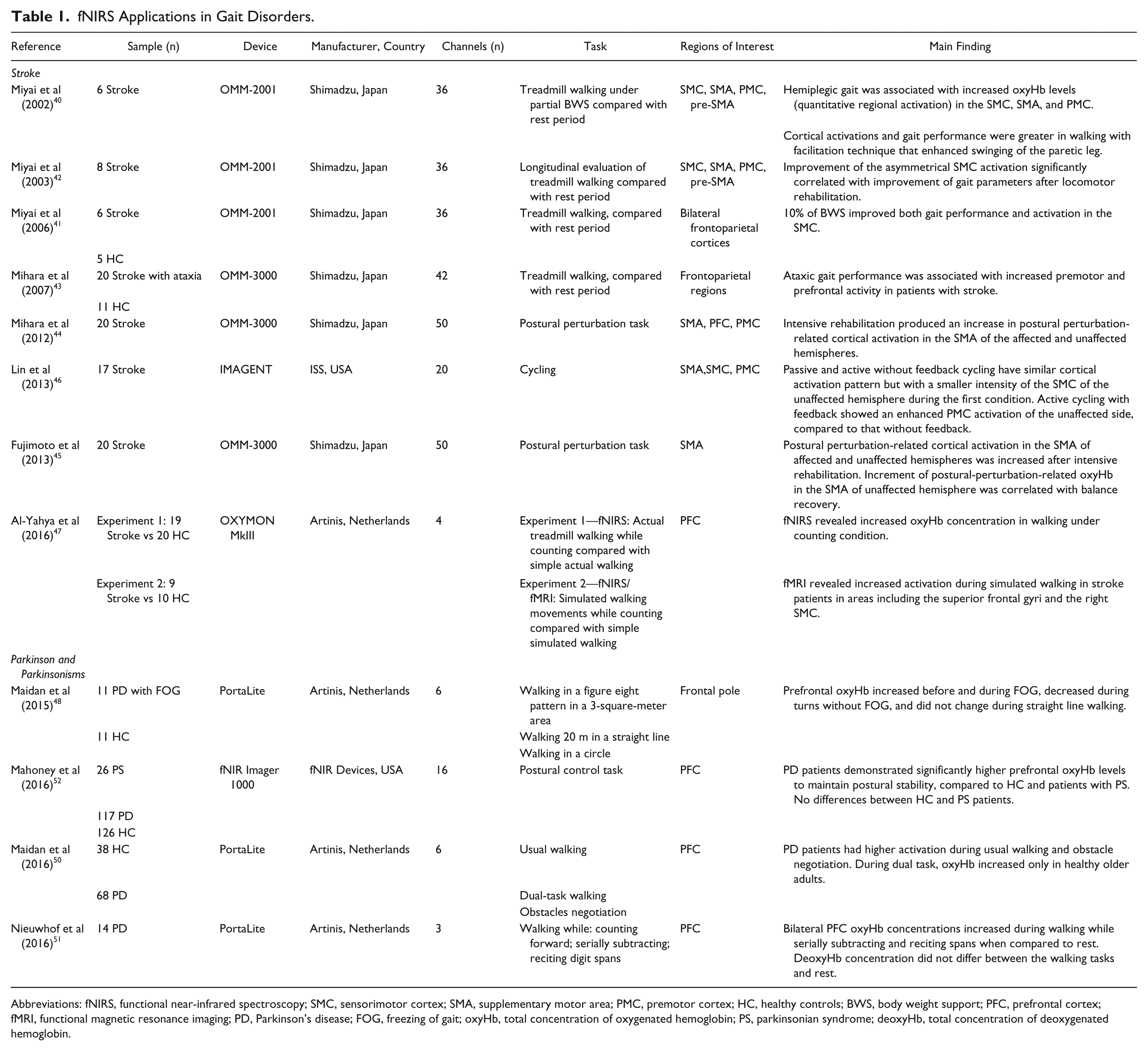
Abbreviations: fNIRS, functional near-infrared spectroscopy; SMC, sensorimotor cortex; SMA, supplementary motor area; PMC, premotor cortex; HC, healthy controls; BWS, body weight support; PFC, prefrontal cortex; fMRI, functional magnetic resonance imaging; PD, Parkinson’s disease; FOG, freezing of gait; oxyHb, total concentration of oxygenated hemoglobin; PS, parkinsonian syndrome; deoxyHb, total concentration of deoxygenated hemoglobin.
fNIRS Applied on Gait Disorders: Stroke
Miyai and colleagues 40 were the first to investigate cortical activation during hemiplegic gait in 6 nonambulatory stroke patients. Experimental protocol required a treadmill walking (0.2 km/h), alternated with rest every 30 seconds, for 4 repetitions. This task was performed under partial body weight support (BWS) in 2 different rehabilitative conditions: walking with mechanical assistance in swinging the paretic leg control (CON) and walking with a facilitation technique that enhanced swinging of the paretic leg (FT). The asymmetry in hemispheric activations was assessed by computing the laterality index (LI) corresponding to (oxyHb in the affected hemisphere − oxyHb in the unaffected hemisphere)/(oxyHb in the affected hemisphere + oxyHb in the unaffected hemisphere). Authors found that gait performance was associated with higher activation of the primary sensorimotor cortex (SMC) in the unaffected hemisphere. Instead, activation of the premotor cortex (PMC) in the affected hemisphere was revealed during hemiplegic gait. Overall, cortical activations and gait performance were greater in walking with FT than with CON. In a second experiment, these authors 41 extended their investigation to the effects of BWS comparing 6 stroke patients with mild to moderate hemiparesis, and 5 age-matched healthy controls (HC). They found a tight association between the activity of the SMC with improvement in gait performance modulated by BWS. In HC, BWS increased overall activation but did not modify gait parameters and there was no correlation between gait parameters and the SMC activation. A follow-up study from the same group 41 evaluated brain changes as a consequence of long-term (2 months) locomotor rehabilitation program. Before rehabilitation, hemiparetic gait was associated with increased oxyHb levels in the SMC, PMC, and supplementary motor area (SMA), which was greater in the unaffected hemisphere rather than in the affected hemisphere. After rehabilitation treatment, the authors found significant improvement in gait performance associated with increased LI in the SMC activation and enhanced activity of the PMC in the affected hemisphere.
Mihara et al 43 investigated cortical activations during walking in patients with infratentorial stroke. Twelve patients with predominant ataxia without severe hemiparesis and 11 age-matched HC were required to walk on a treadmill. Patients with stroke walked slowly, for a shorter time and wore parachute jackets attached to a BWS apparatus to be prevented from falling. Resting-state fNIRS data were obtained from 6 seconds just before the gait period, whereas the gait data were evaluated from 2 phases of the gait period: acceleration and steady phases. In controls, cortical activations in the lateral and medial prefrontal cortex (PFC) during the acceleration phase tended to be attenuated during the steady phase of the gait period, while they were sustained throughout the gait period in ataxic patients. The same group 44 performed another fNIRS experiment in order to investigate the cortical activation induced by external postural perturbation in 20 hemiplegic stroke patients. As a balance control task, they used a combination of brisk forward and backward translations of a platform at a velocity of up to 0.5 m/s with displacements of 4 cm without any prior cues. Group analysis showed significant task-related increases in oxyHb signals after postural perturbation in dorsal PFC and PMC, as well as in the parietal cortex of the unaffected hemisphere. The hyperactivity in the prefrontal cortex was associated with balance ability measured with the Berg Balance Scale.
With the similar experimental protocol and the same fNIRS apparatus, Fujimoto et al 45 performed a longitudinal fNIRS study in 20 stroke patients to evaluate brain changes in postural control after intensive inpatient rehabilitation. Results revealed that intensive rehabilitation increased the SMA activation bilaterally, and notably, the enhancement of the unaffected hemisphere significantly correlated with balance recovery.
Since cycling exercise is a commonly used paradigm for restoring locomotion rhythm in stroke patients, Lin et al 46 used a 20-channel fNIRS instrument to investigate cortical control in subcortical stroke patients during 3 cycling conditions: (a) active cycling with visual speed feedback, (b) active cycling without visual speed feedback, and (c) motor-driven passive cycling. Results revealed that passive and active cycling produced similar cortical activation patterns on SMC, SMA, and PMC, suggesting that passive cycling may be beneficial in facilitating motor recovery after stroke. In addition, active cycling yielded larger activation of the SMC in the unaffected side than passive cycling.
Finally, Al-Yahya et al 47 investigated the relationship between the PFC functional activity and gait measures in chronic stroke walking under single-task (ST) and dual-task (DT) conditions (ie, walking while simultaneously performing a cognitive task). In a first experiment, 19 stroke patients and 20 controls performed walking on a treadmill at self-selected speed. DT-walking was associated with stronger PFC activation compared with ST-walking, for both groups and hemispheres. In a second experiment, in a subset of patients chronic stroke (n = 9) and HC (n = 10), fNIRS and fMRI were acquired simultaneously during ST and DT simulated walking. The 2 tasks were: counting backward and reciprocal feet movement. Participants were administered 4 different conditions: (a) counting only, (b) feet movement only, (c) simultaneous counting and feet movement, and (d) rest. Stroke patients showed a significantly increased PFC activity compared with controls, for both ST and DT walking. fMRI analysis confirmed an increased activation of the PFC.
fNIRS Applied on Gait Disorders: Parkinson’s Disease
Moving to the neurodegenerative realm, Maidan et al 48 were the first in evaluating brain activity of freezing of gait (FOG) in PD using fNIRS. FOG is a unique and disturbing gait disorder usually observed in PD patients with unknown cerebral pathophysiology.13,49 HC and PD patients performed different walking tasks known to provoke FOG, including anticipated and unanticipated turns. PD patients were investigated during their “ON” medication state (typically 2 hours after anti-parkinsonian medication intake). Changes in the prefrontal oxyHb level during straight line walking of 9 seconds were used to compare turns with and without FOG. Sustained oxyHb increases were seen about 3 seconds before turns with FOG. In contrast, oxyHb levels did not change before turns without FOG and decreased during the turns. In HC no oxyHb changes were observed. Overall, FOG during anticipated turns is associated with increased frontal activation starting a few seconds before the FOG itself. In a more recent study, Maidan et al 50 investigated the role of the frontal lobe during DT walking and obstacle negotiation. Thirty-eight HC and 68 PD in ON phase performed 3 walking tasks: (a) usual walking, (b) DT walking, and (c) obstacles negotiation. Gait and frontal activation were assessed during the 3 tasks, using accelerometers and fNIRS, respectively. PD patients had higher activation during usual walking. During DT, oxyHb increased only in elder HC. Finally, during obstacle negotiation, oxyHb increased in patients with PD and tended to increase in older HC.
In order to evaluate the reliability of the PFC activity as a biomarker of gait control in PD, Nieuwhof et al 51 examined the feasibility of a portable wireless fNIRS device. Five repetitions of 3 tasks were included in the experimental protocol: walking on an electronic roll-up walkway while (a) counting forward, (b) serially subtracting, and (c) reciting digit spans. Increased oxyHb concentration in the PFC was detected in walking performed during high demanding cognitive task.
Finally, fNIRS activity in the PFC was also explored by Mahoney et al 52 to compare neural activation patterns of postural stability in 3 groups: (a) 26 patients with Parkinsonian syndromes (PS), (b) 117 patients with mild parkinsonian signs (MPS), and (c) 126 elder HC. OxyHb levels were recorded during a 10-second postural control task from 16 fNIRS channels covering the PFC. PS patients required a higher PFC oxygenation levels compared to HC and PD patients to maintain postural stability. Otherwise, no difference was detected between HC and PD patients.
Discussion
In 2015, Hamacher et al 34 made the first attempt to summarize the current status of knowledge gained using electroencephalogram (EEG), PET, fMRI, and fNIRS on brain activity during gait (both real and imaged). Overall, neuroimaging literature has mainly attempted to understand the nature of cortical compensation strategies required to ensure gait task solving, highlighting the need to apply this technology in gait disorders, which represents a potential field where fNIRS technology may establish a pivotal role.
For this reason, the focus of this review is centered on this specific topic where the emerging concepts are that (a) changes in oxyHb levels of the PFC and PMC are more sensitive to compensation strategies reflecting postural control and restoration of gait disorders; (b) the activation of the unaffected hemisphere is crucial in gait control, mainly in stroke patients; and (c) the evaluation of treatments improving gait by means of fNIRS are completely lacking in PD patients.
What fNIRS Teaches Us About Gait Control in Neurological Patients
The first lesson of fNIRS imaging speaks about neurofunctional differences between HC and stroke patients during walking tasks. Overall, in healthy individuals it has been demonstrated that the presence of compensatory activities in the prefrontal and premotor cortices as a response to task complexity (ie, walking backward 53 ) or aging, 34 whereas the lack of SMC activation has been interpreted in terms of higher control of ongoing gait mostly depending upon subcortical automatisms. 54
In patients with stroke both PFC and bilateral frontoparietal cortices are involved in real bipedal walking, as well as in active cycling, 46 suggesting the presence of widespread compensatory responses.40-44 Altered cortical activations during impaired gait might be described considering the different roles of the infra- and supratentorial structures in locomotor control. 55 The first, cortical regions being appointed to control the automated locomotion and the muscle tones during gait, while the cerebellobasal ganglia pathways involved in the adjustment of locomotor performance to altered environment.56,57 After stroke, the improvement of locomotor outcome supported by rehabilitation depends on the activity and plastic reorganization of multiple brain areas (PFC and PMC). 58
On the other hand, imaging literature on PD-related gait disorders provided a complicated puzzle to assemble. Basically, pathophysiological mechanisms underlying FOG in PD patients are a matter of debate. For instance, Lenka et al 59 and Canu et al, 60 evaluating fMRI resting-state activity, provided completely different patterns of functional changes. The former described a reduced connectivity between parietal and somatosensory cortex, whereas the latter 60 a more consistent decrease of functional connectivity between the SMC and SMA. fNIRS, performed using different walking tasks, highlights the role of the PFC either during postural control of PD patients or during the emergency of FOG symptoms. 51 Mahoney et al 52 were the first in studying patients with Parkinsonian syndromes, where an increase of PFC oxyHb was associated with improvement in postural instability. Furthermore, Maidan et al 50 described the hyperactivity of the Brodmann’s Area 10 (a prefrontal region involved in the motor planning) occurring before FOG during anticipated turns. This finding was in agreement with the changes in theta frequency band shown in recent EEG studies, 61 which unveiled association between motor-planning deficits and FOG. Although fNIRS has never been applied to evaluate the effects of treatments for improving gait in PD, these findings have the merit to suggest new nonpharmacological approaches. Indeed, following evidence provided by Maidan et al 50 and Nieuwhof et al, 51 it has been proposed that increasing cognitive resources (by means of rehabilitation protocols) to counteract executive control deficits in FOG patients might extend the efficiency on walking ability.
Methodological Considerations, Limitations, and Challenges
In a recent opinion paper on fNIRS application for the study of the neural control of gait, Perrey 62 suggests fNIRS as a very promising tool, with real advantages especially when, as it happens for walking and gait control, it is necessary to measure under ecologically compatible conditions. Perrey, 62 however, also warns about the lack of validated pipelines for the analysis of fNIRS during movement and the special care that should be devoted to disentangle signal originating in the brain from the one related to more superficial layers (skin, skull, and other tissues) and movements, both very prominent during walking and gait control. In our experience, fNIRS clinical application requires a careful personalization of the acquisition, with an a priori definition of the cortical region(s) and network of interest and the a priori evaluation of fNIRS sensitivity to cerebral cortex as compared to the effect of other tissues.18-21 This approach is especially suitable with those systems that allow to freely position the optodes on the patient’s skin and useful when optodes are directly glued on the patient’s skin. 25 This solution has been validated in a clinical context and has showed a marked reduction of the movement artifacts.20,25
This approach also allows taking into account, at least to some extent, fNIRS sensitivity to the extracerebral and superficial hemodynamic activity, which contaminates the optical signal by systemic oscillations that can dilute or bias the task-related cortical hemodynamic response. 63 Even if signal averaging over several observations usually allows isolating brain activity from physiological trends, this limitation becomes stronger for investigation of real gait, as it can induce a systemic variation in blood pressure and in skin/scalp activity and hamper the reliability of the results. It is worth noting that none of the studies listed in Table 1 apply a correction method to mitigate the effects of surface hemodynamic activity. In this respect short-separation channels seem to be very promising as they allow regressing the physiological noise out while selectively preserving cortical activity. 64
Furthermore, as noted in a recent work, 19 classical wired fNIRS instruments are usually unwieldy: cables may reduce the freedom of participants’ movement and head and/or cable oscillations can generate motion artifacts elicited by the decoupling between the optodes and the scalp. In this regard, wearable multi-channel fNIRS systems 65 can attenuate such drawbacks, and they should be considered as the most convenient instrumentations for neurorehabilitation, particularly for PD patients. Notably, the number of portable and/or wireless systems is constantly increasing, 66 and in the forthcoming future fiber-less diffuse optical tomography systems 67 might further enrich the flexibility of optical imaging.
Future Perspectives
The results on the cortical control of gait gathered so far with fNIRS encourage a further effort to face the significant amount of methodological works that still need to be performed in order to improve the accuracy of this technique. fNIRS has allowed the direct mapping of the cortical regions involved in gait and walking control and provided useful information that might be exploited to tailor the rehabilitation therapy in a clinical setting. In the near future, fNIRS may become an innovative tool for brain-computer interface applications. 68 For instance, fNIRS signals (possibly combined with EEG) could be exploited to stimulate the patient’s muscles to walk, control orthosis and exoskeletons. 69 This is coming to our fingertips, thanks to the recent development of new light and wireless fNIRS systems. 70 Such systems might become wearable in the near future and could be integrated with exoskeletons to improve patients’ movement and to foster multiple walking modalities in virtual reality training environments.
Conclusions
We summarize that fNIRS applied to gait disorders revealed the presence of different compensatory responses targeting the prefrontal/premotor cortex as main regions. Although this represents a new field of study, the evidence provided so far encourages the translation of this fNIRS-based behavioral tool in clinical practice either for assessing the effectiveness of rehabilitation treatments or for evaluating neurofunctional correlates of gait disorders in ecologically valid conditions.71-73
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by MIUR (Ministero Universita’ e Ricerca; PON 03 PE 00009) grants to Prof Aldo Quattrone.
