Abstract
Kinematic redundancy of the human body provides abundant movement patterns to accomplish the same motor goals (motor equivalence). Compensatory movement patterns such as excessive trunk displacement in stroke subjects during reaching can be viewed as a consequence of the motor equivalent process to accomplish a task despite limited available ranges in some joints. However, despite compensations, the ability to adapt reaching performance when perturbations occur may still be limited when condition-specific changes of joint angles are required. We addressed this hypothesis in individuals with and without stroke for reaching a target placed beyond arm reach in standing while flexing the hips (free-hip condition). In randomly selected trials, hip flexion was unexpectedly blocked, forcing subjects to take a step (blocked-hip condition). In additional trials, subjects took an intentional step while reaching the target (intentional-step condition). In blocked-hip trials, healthy subjects maintained smooth and precise endpoint trajectories by adapting temporal and spatial interjoint coordination to neutralize the effect of the perturbation. However, the ability to produce motor equivalent solutions was reduced in subjects with stroke, evidenced by substantial overshoot errors in endpoint position, reduced movement smoothness and less adaptive elbow-shoulder interjoint coordination. Movement adaptability was more limited in stroke subjects who used more compensatory movements for unperturbed reaching. Results suggest that subjects with mild-to-moderate stroke only partially adapted arm joint movements to maintain reaching performance. Therapeutic efforts to enhance the ability of individuals with stroke to find a larger number of task-relevant motor solutions (adaptability) may improve upper limb recovery.
Introduction
Reaching tasks repetitively performed in daily life can be accomplished by different combinations of joint rotations (i.e., degrees of freedom, DFs) or movement patterns due to the kinematic abundance of the human body, 1 a property known as motor equivalence. 2 Understanding how the capacity for motor equivalence is impacted by stroke may provide insights into mechanisms of motor deficits as well as how motor recovery is expected to occur.
An example of motor equivalence is the ability to use different combinations of elbow and shoulder movements to reach a stationary target with or without additional trunk movement. When trunk movement was unexpectedly arrested by an electromagnet during seated reaches to a target, healthy individuals showed adaptive changes in arm-trunk interjoint coordination, leading to an invariant endpoint trajectory and preservation of accuracy.3,4 Healthy individuals could also maintain reaching accuracy despite expected and/or unexpected perturbations that challenged postural stability during forward reaching from standing.5,6 This was accomplished by flexibly adapting elbow and shoulder movement in response to perturbations. The ability to maintain the endpoint position in space under this perturbation paradigm has been characterized by a gain coefficient (g) ranging from 0 to 1. 7 A gain of 1 indicated that the effect of the perturbation (eg, intentional trunk displacement or unexpected trunk arrest) on hand position was fully neutralized by changing arm joint angles, resulting in maintenance of the same hand position, while a gain of 0 indicated no arm joint adaptive changes, resulting in changes in hand position due to trunk movement. Whether trunk movement was intentionally involved or unexpectedly blocked, healthy individuals could adapt to expected or unexpected perturbations, and preserve high gains (0.85-0.87).7-9 Individuals with unilateral vestibular deficits and Parkinson disease had lower gains (0.47-0.67), indicating reduced movement adaptability.8-10
For reaching tasks, kinematic abundance is reduced in individuals with stroke due to decreased active arm joint ranges 11 and reduced redundancy, 12 restricting how joint movements combine to accomplish a task. This often results in the system finding alternative solutions to accomplish reaching tasks such as the recruitment of other, less desirable DFs (ie, compensatory movement). For example, some individuals with stroke recruit excessive trunk and/or shoulder movement during reaching and pointing tasks to compensate for reduced elbow extension and/or disrupted elbow-shoulder interjoint coordination.13-15 Compensations have been characterized by an increased arm-plane angle,16,17 altered slope of elbow-shoulder angle-angle diagrams, 14 reduced temporal elbow-shoulder coupling, 18 and excessive trunk displacement.19,20 Compensatory movements during reaching can be viewed as the system using its remaining kinematic abundance to perform the task, despite deficits in some DFs.
However, it is unknown to what extent the altered kinematic system preserves the ability to find motor equivalent solutions when unexpected movement perturbations occur. Reduced movement adaptability after stroke has been reported for tasks involving reaching from sitting.21-23 For example, when trunk movement was unexpectedly arrested by an electromagnet during seated reaches to a beyond-reach target in individuals with stroke, adaptations in elbow-shoulder interjoint coordination patterns were delayed resulting in a divergence of the hand trajectory from its intended path. 23 While most studies about movement adaptability focus on reaching from sitting, more DFs are available for tasks involving reaching from standing, as commonly occurs in daily life. Since the ability to adapt movement patterns to perturbations may be reduced because of decreased redundancy, individuals relying on compensatory movements (eg, excessive trunk displacement) may have reduced movement adaptability even in standing despite the availability of additional DFs. The study objective was to determine the adaptive capacity of the limited redundant system during whole-body reaching in subjects with stroke. We hypothesized that healthy individuals would maintain endpoint accuracy and trajectory smoothness regardless of unexpected/intentional perturbations by changing movement patterns in a condition-specific manner (ie, motor equivalence). However, this capacity would be reduced in individuals with stroke, and related to their level of stroke severity. Preliminary results have appeared in abstract form. 24
Methods
Participants
Nineteen right-handed adults with stroke (age = 62 ± 8 years) and 12 right-handed healthy adults (age = 65 ± 10 years) participated after signing consent forms approved by the Centre for Interdisciplinary Research in Rehabilitation of Greater Montreal Ethics Committee. Subjects with stroke >6 months previously and unilateral hemiparesis (≥3/7 on Chedoke-McMaster Stroke Assessment Arm Scale 25 ) were included. Individuals with multiple strokes, cognitive impairment (Montreal Cognitive Assessment 26 <20/30), inability to stand independently, absence of upper limb (UL) and/or lower limb (LL) proprioception (Fugl-Meyer Assessment, FMA 27 ), UL pain, marked apraxia or visuospatial neglect were excluded. Controls were excluded if they had a history of neurological disorders or physical conditions affecting task performance.
Clinical Evaluation
Motor and sensory impairments and balance in stroke subjects were evaluated with valid and reliable scales by an experienced clinician (Table 1). UL (maximum 66) and LL (maximum 28) motor impairment were assessed with FMA (voluntary movement, passive range of motion, reflex function). An UL-FMA cutoff score of 50/66 distinguished between mild and moderate subgroups. 28 UL sensation (light touch, proprioception) was measured with FMA (maximum 12). Elbow flexor spasticity was evaluated with the Composite Spasticity Index (CSI; test-retest reliability, intraclass correlation coefficient [ICC] = 0.87-0.97)29,30 in which scores of 0 to 9, 10 to 12, and 13 to 16 represent mild, moderate, and severe spasticity respectively. 31 Six aspects of balance were assessed with the Brief Balance Evaluation Systems Test (B-BESTest, reliability, ICC = 0.87-0.99).32,33
Demographic Characteristics (Arranged by Decreasing UL-FMA Scores).
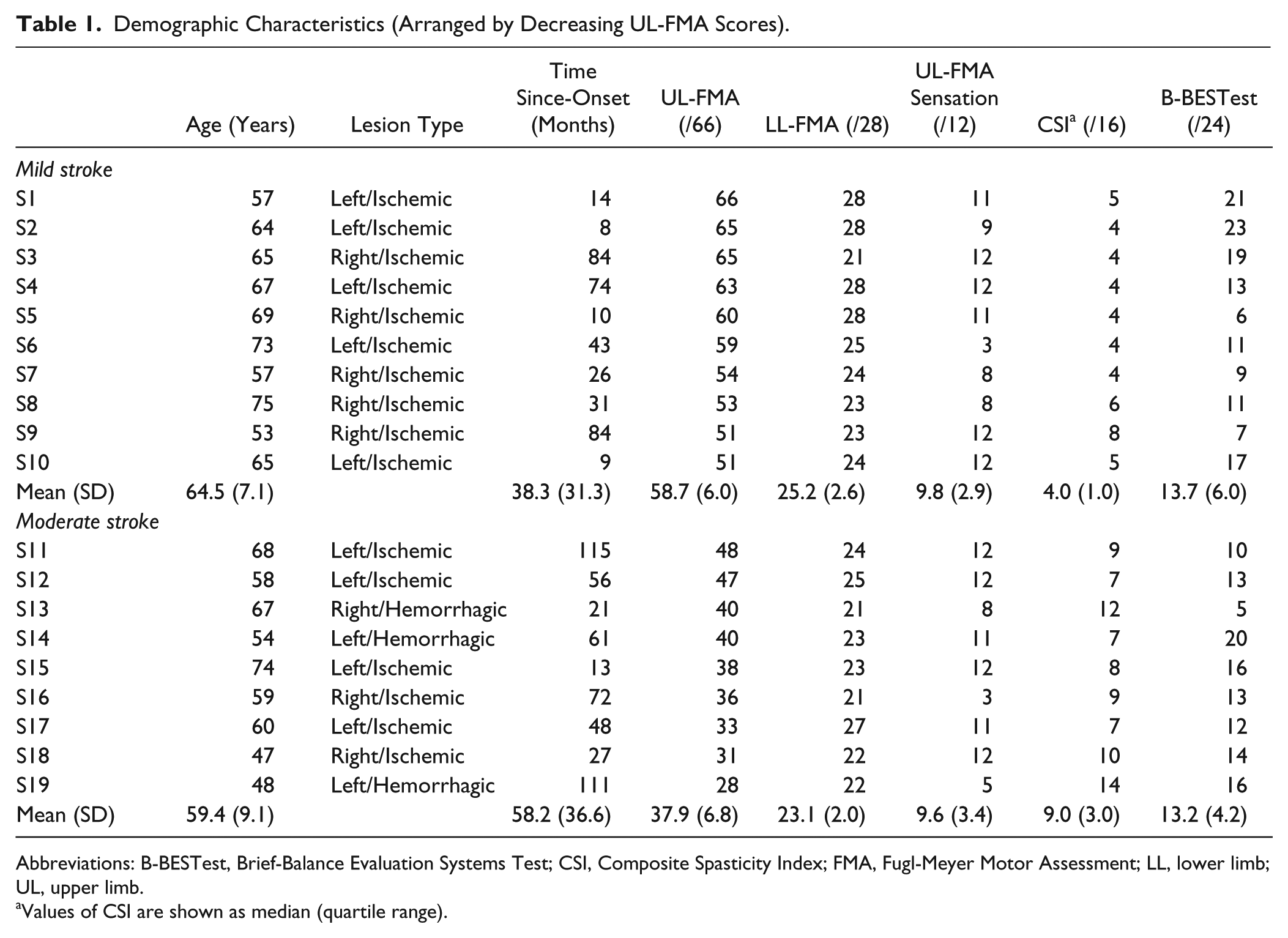
Abbreviations: B-BESTest, Brief-Balance Evaluation Systems Test; CSI, Composite Spasticity Index; FMA, Fugl-Meyer Motor Assessment; LL, lower limb; UL, upper limb.
Values of CSI are shown as median (quartile range).
Pointing Task
The more-affected (stroke) or nondominant (control) arms were tested. Subjects stood with feet shoulder-width apart and wore a ceiling-mounted safety harness to prevent falling (Figure 1A). The target (light-emitting diode; LED) was placed at 130% arm length, measured from the acromion to the index fingertip (endpoint) with the arm extended, in the mid-sagittal plane at anterior superior iliac spine (ASIS) height (Figure 1A). The endpoint starting position was standardized at the upper edge of the ipsilateral greater trochanter.
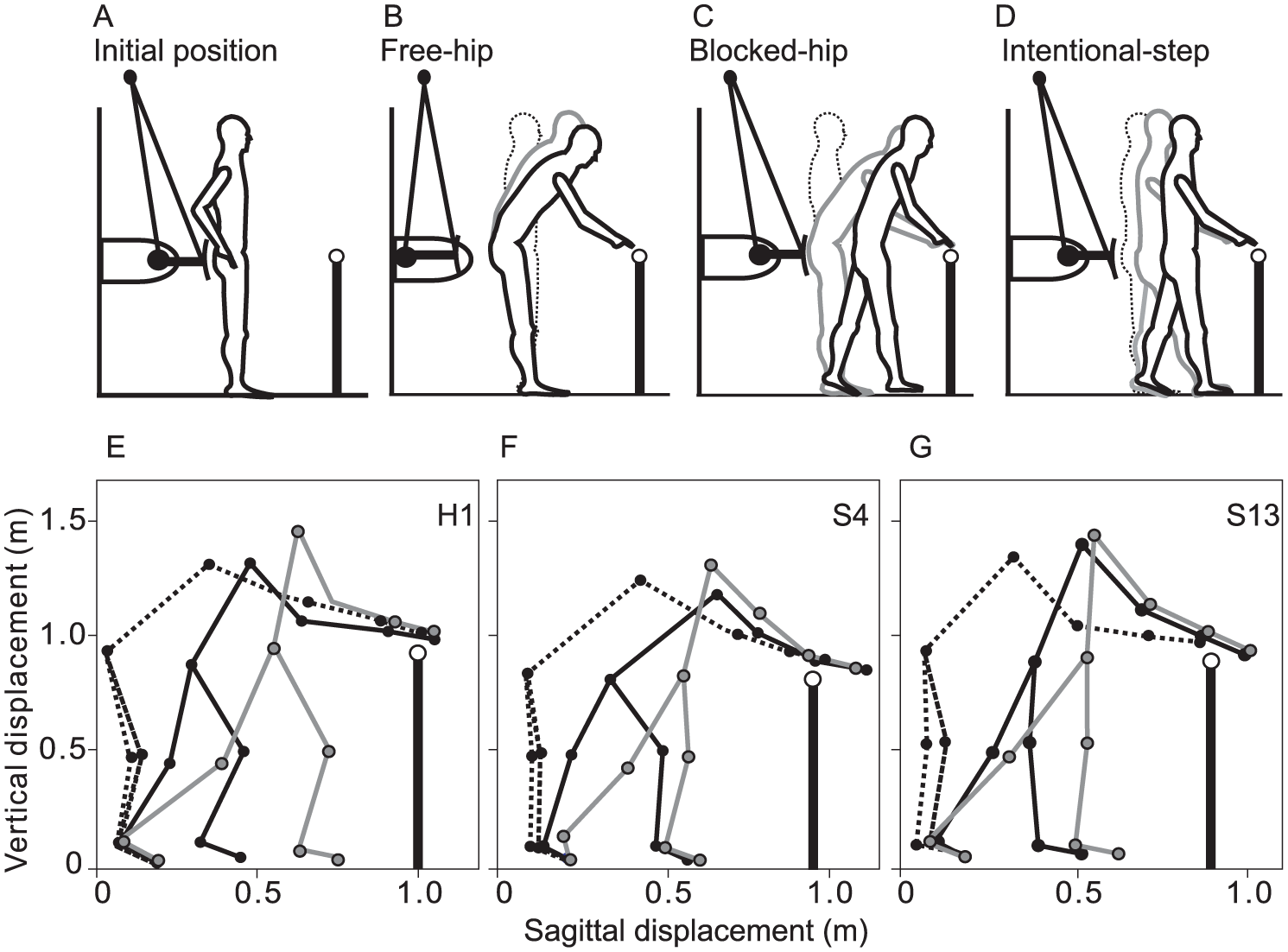
Experimental conditions and examples of whole-body displacements. (A) Subjects reached to a remembered target (open circle) placed beyond the arm’s reach with the eyes closed. (B) In most trials, no perturbation was applied due to retraction of a board that prevented hip flexion (free-hip condition). (C) In 30% of randomly chosen trials, hip flexion was unexpectedly prevented by locking the board with an electromagnetic device, forcing subjects to take a step to prevent falling (blocked-hip condition). (D) In another condition, no perturbation occurred but subjects took an intentional step during reaching (intentional-step condition). (E) In the healthy subject, endpoint position was similar in all 3 conditions. The elbow was extended in the free-hip condition (black dashed line), while it was more flexed in the blocked-hip (black solid line) and intentional-step (gray line) conditions. (F) In the mild-stroke subject, some endpoint overshoot error occurred due to stepping in the blocked-hip and intentional-step conditions. The elbow was extended in the free-hip condition (black dashed line). (G) In the moderate-stroke subject, a substantial endpoint overshoot error occurred due to uncompensated stepping in the blocked-hip and intentional-step conditions. The elbow was flexed in the free-hip condition, and did not change across conditions.
Subjects reached to the remembered target with eyes closed on hearing a GO signal (beep) as fast and accurately as possible (Figure 1B). Since the target was placed beyond arm length, to reach the target, subjects had to lean the trunk forward by flexing the hips that resulted in a backward buttock motion (hip strategy, Figure 1B). This motion involved slight knee and ankle flexion and subjects were instructed to avoid excessive knee flexion.34,35
A perturbation device consisting of a height-adjustable electromagnet attached to the wall behind the subject, was initially locked to a ferromagnetic plate on a concave-shaped board behind the subject. In the initial position, there was a ~1 cm gap between the buttocks and board (Figure 1A). In 70% of trials (35 of 65 trials), the electromagnet was switched off simultaneously with the GO signal and the board swung backward without obstructing buttock movement (free-hip condition; Figure 1B). In the remaining 30% of trials, the electromagnet remained locked preventing backward buttock motion. This forced a stepping reaction to regain balance (blocked-hip condition; Figure 1C). Perturbed and non-perturbed trials were randomly assigned. In an additional 15 trials, subjects took an intentional forward step (intentional-step condition, Figure 1D) to reach the same remembered target. The intentional-step condition controlled for the influence of prediction on movement adaptability and for ensuring that subjects could perform accurate whole-body reaching while stepping. Subjects received no instructions about step timing or magnitude to allow natural behavior to occur. Prior to recording, subjects practiced reaching in each condition (7-10 trials per condition, random) with eyes closed while feedback on reaching accuracy was provided at trial end by subjects visually checking final endpoint position deviations. Since absolute reaching accuracy was not a main outcome measure, practice was only done to ensure that subjects understood the task. The task was performed without extended practice, visual or haptic feedback to avoid learning effects. Lack of visual and haptic information also ensured that movement corrections were not made. Rest periods of 5 minutes were allowed after 15 to 20 trials to avoid fatigue.
Data Recording and Analysis
Marker movements were recorded for 7 seconds per trial at 100 Hz using a 3-dimensional motion capture system (Optotrak, NDI, Canada). Infrared LEDs were positioned on the index fingertip (endpoint), ulnar head (wrist), lateral epicondyle (elbow), mid-sternum (trunk) and bilaterally on acromions (shoulder), ASIS (hip), patellae (knee), and lateral malleoli (foot).
Data were interpolated (third-order spline function) and filtered with a low-pass 10 Hz Butterworth filter. When trunk movement is involved in forward reaching, trunk movement usually continues after the end of the endpoint movement. 36 Thus, movement onsets and offsets were determined when endpoint and ipsilateral acromion tangential velocity respectively exceeded or fell below 10% of their peak values for >50 ms. In each trial, timing of all kinematic events was determined relative to endpoint movement onset.
The primary outcome was the condition-specific gain, and secondary outcomes were endpoint performance variables (trajectory straightness, smoothness) and movement quality variables (arm and trunk angles, interjoint coordination) as defined in Levin et al. 37 Since endpoint displacement can be accomplished in different ways (eg, by movements of only arm joints or by a combination of arm/trunk movements), characterization of joint/trunk kinematics describes movement quality.
Endpoint Movement Measures
Changes in reaching accuracy and smoothness during blocked-hip (or intentional-step) conditions compared with the free-hip condition indicated the extent of spatial and temporal adaptation to perturbation. The arm-trunk gain (g) provides an index of spatial adaptability since it measures the influence of whole-body displacement associated with taking a step on hand position. Gain was calculated as the differences in the final endpoint (d) and hip (h) marker sagittal positions between free-hip and blocked-hip or intentional-step conditions:
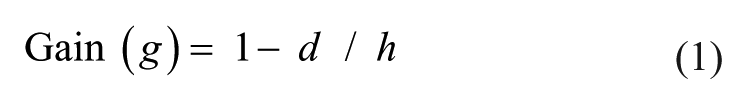
In particular, g = 1 if d = 0 (ie, full adaptation), and g = 0 if d = h (ie, no adaptation). Trajectory straightness was evaluated with the index of curvature (IC) defined as the ratio of total endpoint displacement to the shortest distance between initial and final positions. IC = 1 and IC = 1.57 for a straight and semicircular path, respectively (π/2). Trajectory smoothness was evaluated by the number of movement units (NMUs) defined as the number of peaks in the endpoint tangential velocity profile between endpoint movement onset and offset, where fewer NMUs indicate smoother reaching. 38
Joint Kinematic Measures
Condition-specific changes in elbow flexion, shoulder flexion, hip flexion and trunk rotation angles at movement offset were computed. The elbow angle was determined by the dot product of vectors defined by wrist, elbow and shoulder markers, where 0° indicated full elbow extension. Shoulder flexion was computed as the angle between the vertical projection through the sternal marker and the vector between the right shoulder and elbow markers, where the arm alongside the body was 0°. Hip flexion angle was computed as the angle between the vertical projection through the hip marker and the vector between the ipsilateral shoulder and hip markers, where hip angle in upright standing was 0°. Trunk yaw angle was derived by the plane formed by the trunk and both acromial markers in room coordinates, where initial position was 0°.
Temporal and spatial interjoint coordination between adjacent joint angles (elbow flexion–shoulder flexion; shoulder flexion–hip flexion) in the Free-hip condition was evaluated by the cross-correlation coefficient at zero-time lag 39 and the slope of the angle-angle diagram, 13 respectively. For cross-correlation, a value closer to 1.0 denotes a stronger correlation and indicates that motions of the 2 joints occur in the same direction and are tightly coupled in time. Angle-angle diagrams were generated for each joint pair, and the slope of the linear regression was estimated. A slope of ±1.0 indicated that both joints moved the same amount while slopes >1.0 or <1.0 indicated that the movement involved predominantly one joint. 13
Stepping Measures
Step-onset timing and step-length were computed for blocked-hip and intentional-step conditions. Step-onset was defined as the time when the tangential velocity of the ankle marker on the stepping leg exceeded 10% of peak values for >50 ms. Step-length was the distance of the ankle marker on the stepping leg from its initial to the final foot position. Data analysis was done with custom-made programs (Matlab 2014a, MathWorks, Natick, MA).
Statistical Analysis
Data normality and variance homogeneity between groups were verified using Shapiro-Wilk and Levene’s tests. Mauchly’s test for sphericity and Greenhouse-Geisser DFs adjustments were used for non-spherical data. All outcomes except for IC, cross-correlation coefficients, angle-angle slopes and CSI scores were normally distributed with variance homogeneity. Two-way mixed model analyses of variance and paired t tests with Bonferroni corrections estimated pairwise differences between groups (healthy, mild stroke, moderate stroke) and conditions (free-hip, blocked-hip, intentional-step) for gains, NMUs and joint angles. For IC, Friedman and Wilcoxon signed-rank tests with Bonferroni corrections estimated pairwise differences between conditions. Mann-Whitney U and Hodges-Lehmann tests estimated differences in IC between healthy and stroke subjects and their 95% confidence intervals (CIs). Kruskal-Wallis and Mann-Whitney U tests with Bonferroni corrections estimated group differences for measures of temporal and spatial interjoint coordination. Effect sizes (ESs) were estimated for all statistically significant comparisons, using Cohen’s d for parametric data and by dividing the standardized test statistic with the square root of the sample size for nonparametric data. 40 To determine relationships between movement adaptability and compensatory movement patterns with stroke recovery, endpoint performance measures (gains, IC, NMUs) in blocked-hip and intentional-step conditions, and movement quality measures (elbow-shoulder interjoint coordination, elbow flexion, trunk rotations) in the free-hip condition were correlated with clinical impairment scales (FMA, CSI, BESTest) using Pearson and Spearman coefficients. Analyses were done with SPSS version 21 (IBM Corp, Armonk, NY) using initial significance levels of P < .05.
Results
Endpoint Measures
Healthy Subjects
Examples of whole-body displacements and endpoint velocity profiles in one healthy and two stroke subjects are shown in Figures 1E-G and 2. In the healthy subject (Figures 1E and 2A and D), final endpoint position, endpoint trajectory, and velocity profiles were similar across conditions. For the whole group, despite substantial hip displacement (h = 34.0 ± 7.6 cm), differences (d) between mean final endpoint positions of perturbed and free-hip conditions were small (blocked-hip condition: 6.9 ± 4.6cm; intentional-step condition: 8.4 ± 4.9cm), resulting in high gains (Table 2). Gains were greater for the intentional-step compared with the blocked-hip condition (95% CI = [0.03, 0.13], ES = 1.2). Endpoint movement speeds were similar and endpoint trajectories were almost straight in the free-hip (IC = 1.10 [0.07]), blocked-hip (IC = 1.08 [0.04]), and intentional-step (IC = 1.06 [0.03]) conditions. Trajectory curvatures were similar between blocked-hip and free-hip conditions, while they were 4% straighter in intentional-step compared with free-hip conditions (95% CI = [0.06, 0.01], ES = 0.83, χ2 = 8.167, P = .019; Table 2). Although there were 1 to 2 NMUs in all conditions (Figure 2D), movements were less smooth in the blocked-hip compared with the free-hip condition by 33% (95% CI = [0.05, 0.64], ES = 0.98, F2, 60.0 = 31.044, P < .001; Table 2).
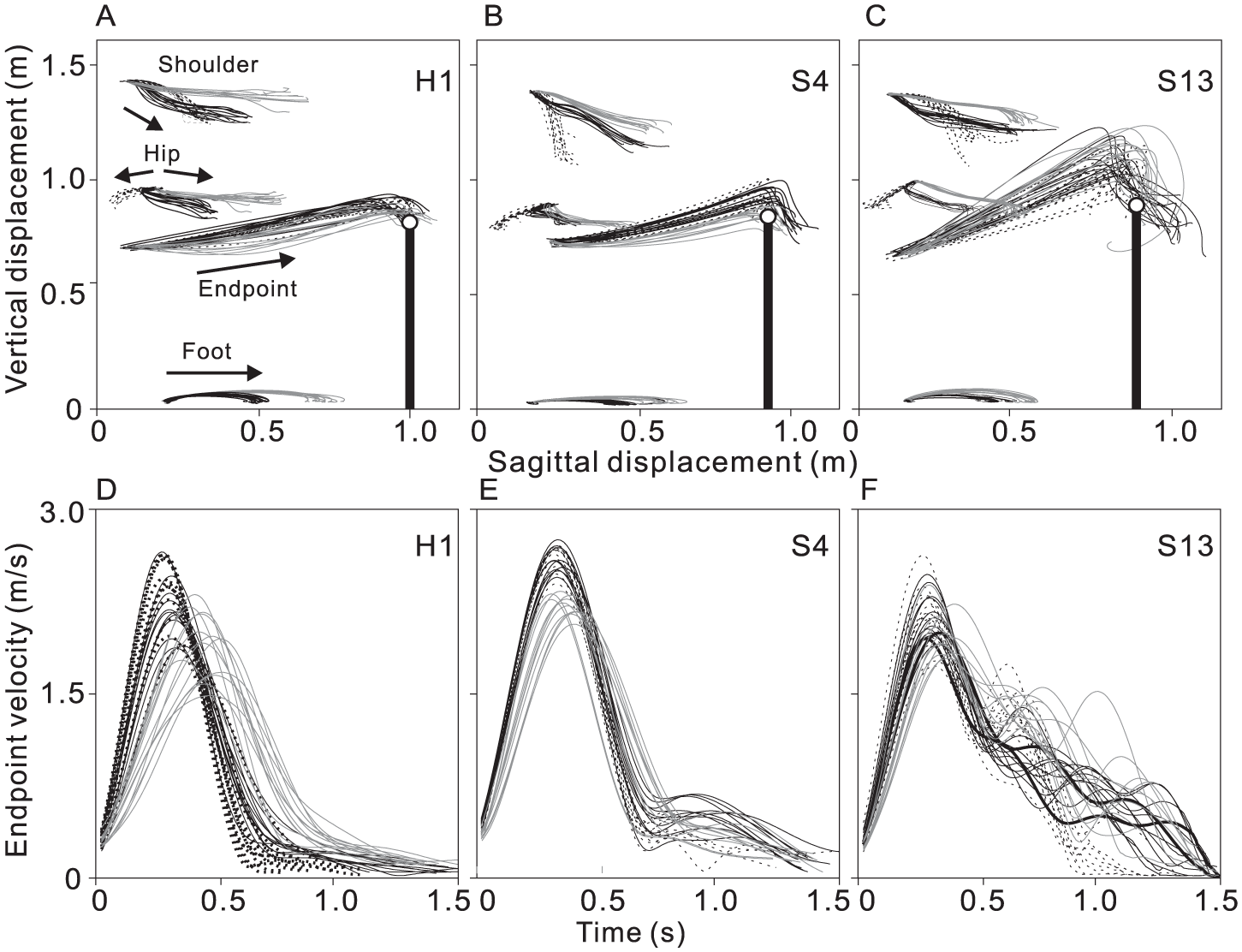
Examples of segment trajectories and endpoint velocity profiles during whole-body reaching in healthy and stroke subjects. (A) In the healthy subject, the endpoint position relative to the target (open circle on top of vertical line) was similar in all 3 conditions, despite substantial whole-body displacement occurring in blocked-hip (black solid lines) and intentional-step (gray lines) conditions, resulting in high gains in both conditions for this subject (blocked-hip, 0.81; intentional step: 0.82). (B) In the subject with mild stroke (upper limb Fugl-Meyer Motor Assessment [UL-FMA] = 63), some overshoot error occurred due to whole-body displacement in blocked-hip (19.1 cm) and intentional-step (6.3 cm) conditions, resulting in lower gains (blocked-hip, 0.45; intentional-step, 0.79) compared with the healthy subject. (C) In the subject with moderate stroke (UL-FMA = 40), the trajectory was more curved and a substantial overshoot error occurred due to whole-body displacement in the blocked-hip (24.2 cm) and intentional-step (14.4 cm) conditions, resulting in lowers gain than healthy subjects (blocked-hip, 0.35; intentional-step, 0.59). (D) Velocity profiles of each of the 3 subjects for each experimental condition. In the healthy subject, the endpoint velocity profile was bell-shaped in all 3 conditions. (E) In the subject with mild stroke, the velocity profile was bell-shaped in the free-hip (black dashed lines) and intentional-step condition (gray lines), while a second peak occurred in the blocked-hip condition (black solid lines). (F) In the subject with moderate stroke, a second peak occurred in the free-hip condition (black dashed lines), and additional peaks occurred in the blocked-hip condition (black solid lines).
Comparison of Kinematic Data From Control, Mild-Stroke, and Moderate-Stroke Groups in 3 Conditions. a
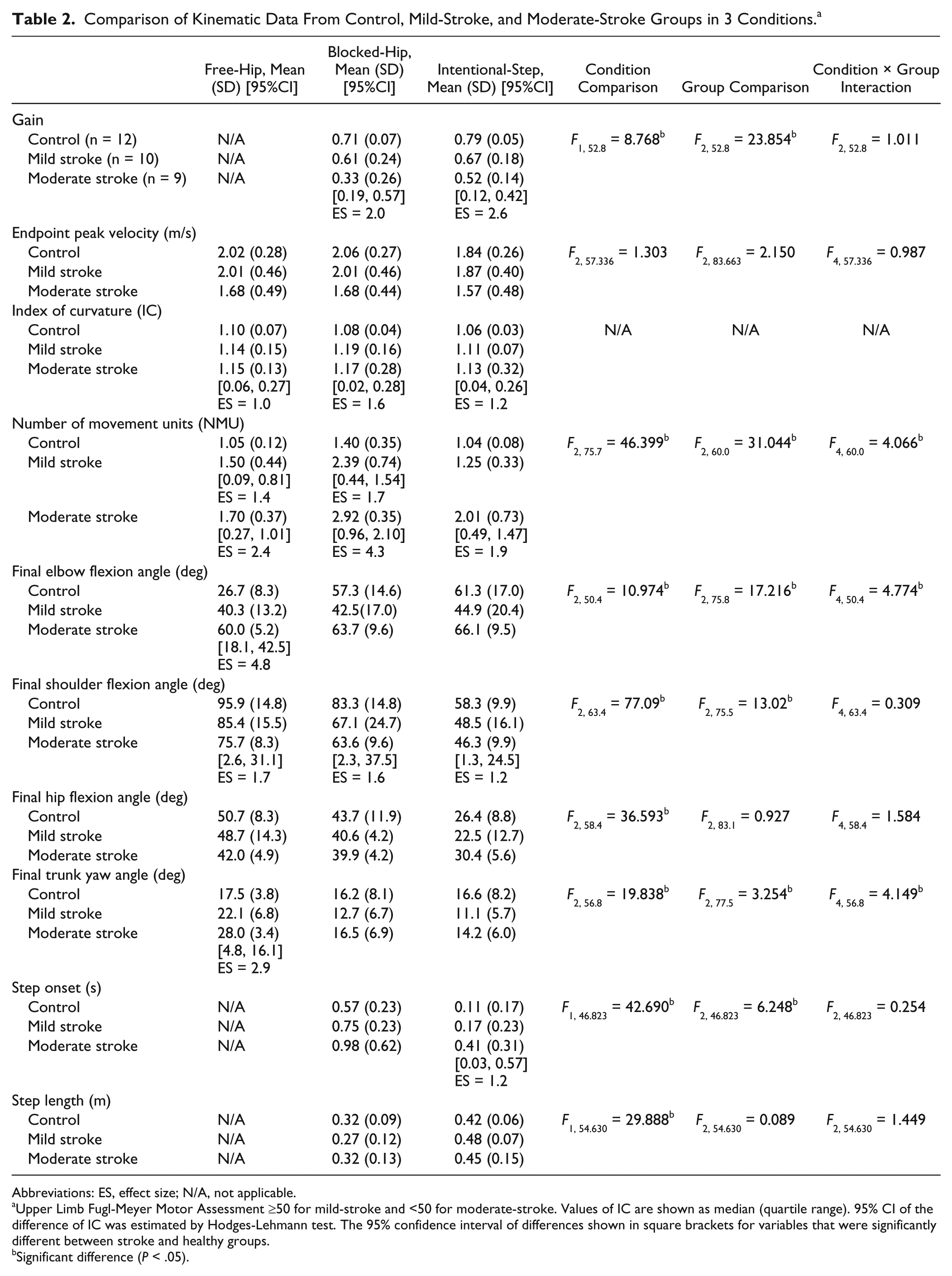
Abbreviations: ES, effect size; N/A, not applicable.
Upper Limb Fugl-Meyer Motor Assessment ≥50 for mild-stroke and <50 for moderate-stroke. Values of IC are shown as median (quartile range). 95% CI of the difference of IC was estimated by Hodges-Lehmann test. The 95% confidence interval of differences shown in square brackets for variables that were significantly different between stroke and healthy groups.
Significant difference (P < .05).
Stroke Subjects
Figures 1 and 2 show examples of whole-body reaching in subjects with mild (Figures 1F and 2B) and moderate (Figures 1G and 2C) stroke. Final endpoint positions, endpoint trajectories and velocity profiles differed across conditions. Group differences (d) between mean final endpoint positions of free-hip compared with both blocked-hip (mild, 6.9 ± 5.8 cm, 95% CI = [1.9, 15.7], ES = 1.1; moderate, 13.1 ± 8.7 cm, 95% CI = [3.3, 22.9], ES = 3.4) and intentional-step (mild, 11.5 ± 7.1 cm, 95% CI = [−6.1, 15.3], ES = 1.8; moderate, 15.9 ± 6.7 cm, 95% CI = [8.6, 23.3], ES = 4.2) conditions were large, resulting in low to moderate gains (Table 2). However, hip displacements were similar to controls (h = 35.9 ± 12.9 cm). Gains for the intentional-step condition were greater than those for the blocked-hip condition for the moderate but not for the mild group (Table 2). Endpoint velocity profiles had multiple peaks in all conditions (less smooth), with more NMUs in the blocked-hip compared with the free-hip condition by 59% (95% CI = [0.15, 1.62], ES = 2.0, P = .001) and 73% (95% CI = [0.77, 1.68], ES = 3.3, P < .001), respectively, for mild- and moderate-stroke subjects (Table 2).
Comparisons Between Stroke and Healthy Subjects
Compared with healthy subjects, gains were lower in subjects with stroke (F2, 52.8 = 23.854, P < .001), particularly in those with moderate stroke in both blocked-hip (95% CI = [0.19, 0.57], ES = 2.0) and intentional-step conditions (95% CI = [0.12, 0.42], ES = 2.6). Endpoint smoothness was reduced in stroke compared with healthy subjects (F2, 75.7 = 46.399, P < .001; Table 2). Condition-specific differences in NMUs were greater in both mild- and moderate-stroke compared with controls (group × condition interaction: F4, 60.0 = 4.066, P < .006).
Joint Kinematic Measures
Healthy Subjects
Free-hip reaching involved elbow extension, hip flexion, and shoulder flexion (Figure 3A and D). The elbow joint was more flexed by 30.6° (95% CI = [17.8, 43.4], ES = 1.9) in the blocked-hip and by 34.6° (95% CI = [20.0, 49.2], ES = 2.2) in the intentional-step condition at movement offset compared with the free-hip condition (F2, 50.4 = 10.974, P < .001; Table 2). Mean trunk yaw angles were small and similar across conditions (Table 2).
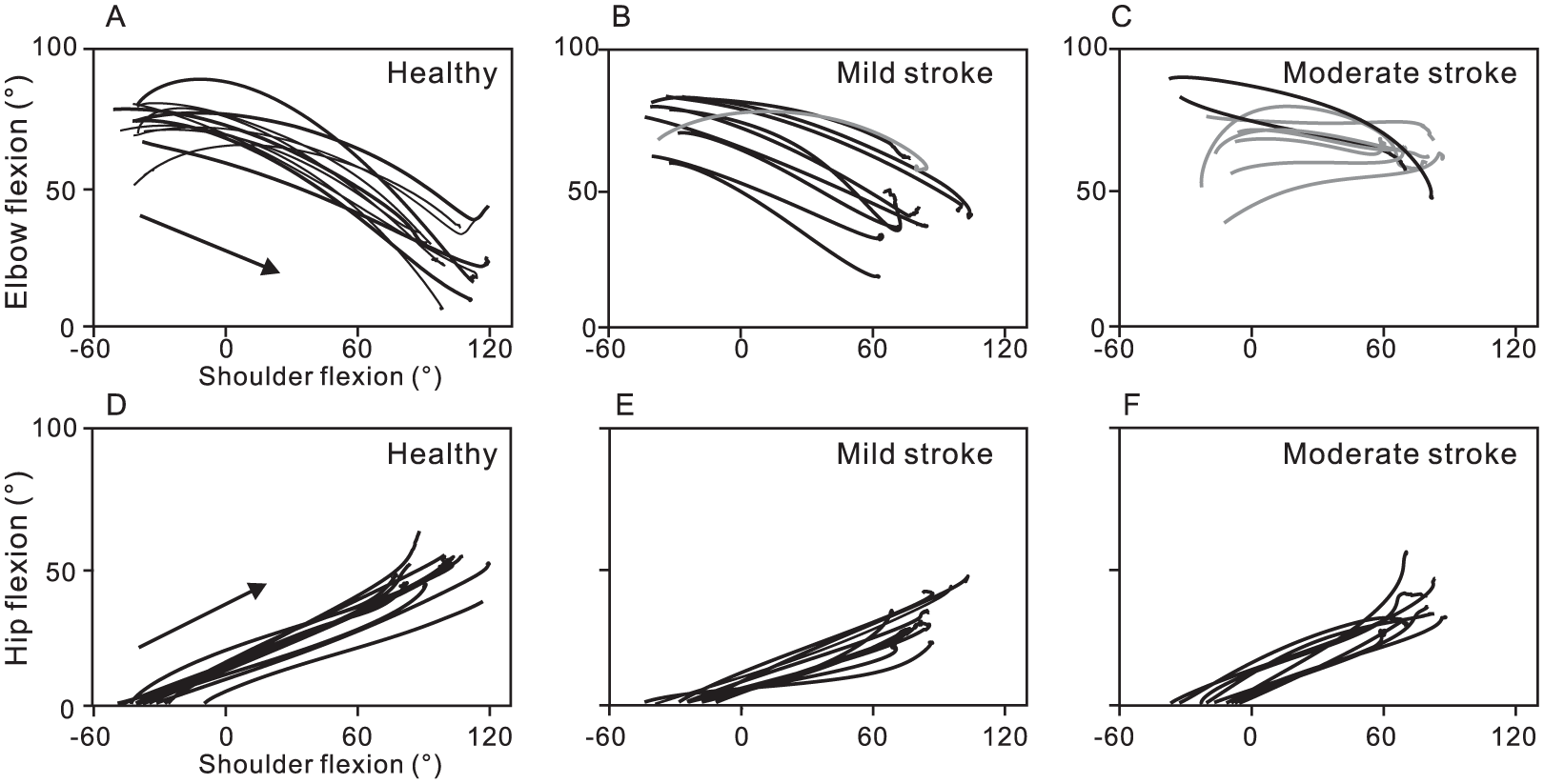
Examples of interjoint coordination between the elbow, shoulder and hip joints. (A) Mean elbow-shoulder interjoint coordination in the free-hip condition for each subject. In healthy subjects, elbow extension occurred together with shoulder flexion. (B) In subjects with mild stroke, elbow-shoulder interjoint coordination was similar to healthy subjects, except for 1 subject (S10; fugl-Meyer Motor Assessment [FMA] = 51, gray line) who used less elbow extension. (C) In subjects with moderate stroke, elbow flexion occurred together with shoulder flexion in 7 out of 9 subjects (gray lines). (D-F) Hip-shoulder interjoint coordination in the free-hip condition. Hip flexion occurred together with shoulder flexion in all 3 subject groups.
Stroke Subjects
In 8 stroke subjects, movement patterns differed from healthy subjects (Figure 3, gray lines) such that elbow flexion occurred together with shoulder and hip flexion in the free-hip condition. For all subjects, while elbow angles were similar across conditions, trunk yaw angles were smaller in blocked-hip (95% CI = [4.2, 18.8], ES = 1.4) and intentional-step conditions (95% CI = [7.2, 20.3], ES = 1.7), compared with the free-hip condition (F2, 56.8 = 19.838, P < .001; Table 2). Step-timing and length were not correlated with interjoint coordination in either blocked-hip (step-timing, r = −0.19, P = .43; step-length, r = 0.30, P = .22) or intentional-step (step-timing, r = −0.02, P = .94; step-length, r = −0.12, P = .63) conditions.
Comparisons Between Stroke and Healthy Subjects
Compared with healthy subjects, at the final position, elbow flexion (95% CI = [18.1, 42.5], ES = 4.8) and trunk yaw (95% CI = [4.8, 16.1], ES = 2.9) angles were greater in moderate-stroke subjects in the free-hip condition. Medians [quartile range] of elbow-shoulder cross-correlation coefficients at zero-lag were −0.96 [0.04] and −0.90 [0.30] in healthy and mild-stroke subjects, respectively, while they were lower −0.35 [0.85] in the moderate-stroke group (95% CI = [0.03, 0.84], ES = 1.2, P = .003). Similarly, slopes of elbow/shoulder diagrams were similar between controls (−0.32 [0.11]) and mild-stroke (−0.32 [0.18]) subjects, but were lower in subjects with moderate-stroke (−0.04 [0.24]; 95% CI = [0.09, 0.35], ES = 1.0, P = .003). In contrast, cross-correlation coefficients and slopes between shoulder and hip flexion in perturbed reaching were similar in all groups (cross-correlations: healthy, 0.99 [0.02]; mild stroke, 0.98 [0.04]; moderate stroke, 0.98 [0.02]; slopes: healthy, 0.41 [0.09]; mild stroke, 0.52 [0.22]; moderate stroke, 0.56 [0.12]).
Condition-specific changes (group × condition interaction) in elbow flexion angles were smaller in both stroke groups compared with healthy subjects (F4, 50.4 = 4.774, P = .002), but were greater for trunk yaw angles (F4, 56.8 = 4.149; P = .002).
Gains
Gains for the blocked-hip condition in stroke subjects who demonstrated altered elbow-shoulder interjoint coordination patterns in unperturbed reaching were lower (gains = 0.31 ± 0.16; Figure 3B and C, gray lines) than those for subjects who had coordination patterns similar to healthy subjects (gains = 0.59 ± 0.24; black lines in Figure 3B and C; 95% CI = [0.02, 0.39], ES = 1.2, P = .006).
In all groups, step-onset occurred earlier in the intentional-step compared with the blocked-hip condition (healthy, 95% CI = [0.42, 0.71], ES = 2.0, P < .001; mild stroke, 95% CI = [0.59, 0.91], ES = 2.6, P < .001; moderate stroke, 95% CI = [0.50, 1.46], ES = 0.6, P = .032) and step-length was longer (healthy, 95% CI = [0.04, 0.17], ES = 1.1, P = .004; mild stroke, 95% CI = [0.12, 0.31], ES = 1.8, P < .001; moderate-stroke, 95% CI = [0.01, 0.27], ES = 1.0, P < .001). In subjects with moderate stroke, step-onset in the intentional-step condition was delayed compared with healthy subjects (95% CI = [0.03, 0.57], ES = 1.2, P < .007). However, gains in both conditions were not correlated with step-onset (blocked-hip condition, r = 0.03, P = .90; intentional-step condition, r = −0.25, P = .31) or length (blocked-hip condition, r = −0.33, P = .17; intentional-step condition, r = 0.02, P = .92) in subjects with stroke.
Greater use of compensations during unperturbed reaching in subjects with stroke was correlated with lower gains in the blocked-hip condition (trunk rotation vs gain, r = −0.44, P = .007; elbow flexion vs gain, r = −0.57, P < .001).
Relationship of Kinematic Outcomes to Clinical Scores
Stroke subjects with greater UL impairment had lower gains (blocked-hip, UL-FMA: r = 0.49, P = .017, Figure 4A; intentional-step: r = 0.46, P = .023), more curved trajectories (CSI, blocked-hip: r = 0.44, P = .030; intentional-step: r = 0.46, P = .024) and less smooth trajectories (blocked-hip: UL-FMA: r = −0.61, P = .003; intentional-step: r = −0.68, P = .001). Greater clinical severity was also associated with greater elbow flexion (UL-FMA, r = −0.65, P = .001; Figure 4B) and trunk rotation (UL-FMA: r = −0.45, P = .028; Figure 4B) at movement offset, as well as altered temporal (UL-FMA, r = −0.62, P = .002; CSI, r = 0.63, P = .002) and spatial (UL-FMA, r = −0.67, P = .001; CSI, r = 0.62, P = .001) elbow-shoulder interjoint coordination in unperturbed reaching. Balance scores (B-BESTest) and sensation were not correlated with any performance or movement quality measures.
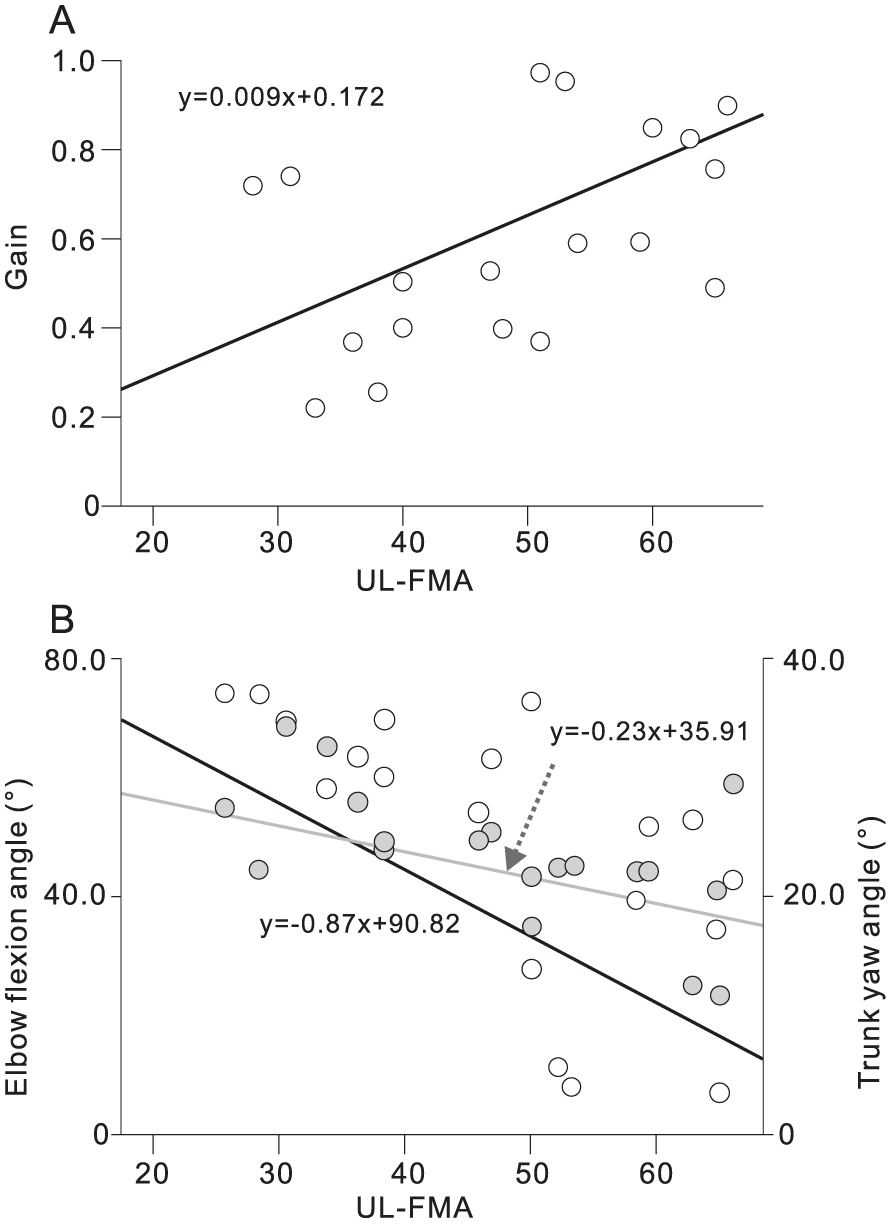
Correlations with clinical scores in subjects with stroke. (A) Upper limb Fugl-Meyer Motor assessment (UL-FMA) scores were significantly correlated with (A) gains in the blocked-hip condition (r = 0.49, P = .017); (B) final elbow flexion angle (white circles, r = −0.65, P = .001) and final trunk yaw angle (gray circles, r = −0.45, P = .028) in the blocked-hip condition.
Discussion
We investigated movement adaptability (ie, capacity of motor equivalence) of whole-body reaching in stroke subjects by testing their ability to maintain reaching performance (final endpoint position, trajectory curvature and movement smoothness) despite substantial changes in the number of DFs and challenges to postural stability elicited by reactive or voluntary stepping. Similar to results in younger adults,6,41 healthy older subjects maintained reaching performance even when postural stability was challenged and stepping occurred, by adaptively changing their interjoint coordination, while more impaired stroke subjects did not.
Previous studies have suggested that referent control underlies motor equivalence (eg, invariant reaching performance).3,6,42 Specifically, movement occurs to minimize the difference between an actual and a centrally specified referent body configuration (RC) that defines the posture at which all muscles can reach their activity minima. To produce reaching, the system changes RC to produce a referent endpoint trajectory (RT) and consequently, an actual endpoint trajectory toward the target. Trial-to-trial variations in changes in RC and in history-dependent neuromuscular properties are responsible for small intertrial variations in interjoint coordination and reaching trajectories,41,43 justifying the expression that “movement does not repeat itself.” 44
Our results are consistent with the suggestion that changes in RC elicited by perturbations contain components preventing changes in the referent endpoint trajectory. This allows healthy subjects to preserve the endpoint trajectory by altering joint rotations. 6 However, individuals with stroke, in particular those with moderate stroke, had reduced movement adaptability, evidenced by lower gains and altered trajectory curvatures and smoothness (Figures 2 and 3). Deficits in specification of RC or RT may be responsible for reduced motor equivalence observed in stroke subjects. These deficits may also be affected by impairments in proprioception, spatial memory, muscle force, tone, and balance.
The low gains in stroke subjects indicate that changes in movement patterns occurring in response to perturbations were incomplete. The capacity to use motor abundance to stabilize endpoint trajectories during reaching from sitting is decreased in subjects with mild-to-moderate stroke.12,22,45 In our study, healthy subjects flexibly changed elbow, shoulder and hip joint angles to neutralize the influence of perturbations on reaching performance. However, while elbow angles in stroke subjects were similar across conditions, large condition-specific changes occurred concomitantly in trunk rotation angles. This suggests that stroke subjects used different adaptive movement patterns than healthy subjects to neutralize perturbation effects. Similarly, in a previous study, stroke subjects used less elbow and more trunk movement compared to healthy subjects during UL obstacle avoidance. 46 These results are consistent with the observation of reduced ability to flexibility change elbow and shoulder joint angles in subjects with stroke during reaching from sitting.16,47
Our study provides new information about the relationship between compensatory movement patterns and adaptability of reaching after unexpected perturbations. We showed that subjects with altered elbow-shoulder interjoint coordination in unperturbed reaching had significantly reduced gains compared with those who had coordination patterns similar to healthy subjects. Furthermore, excessive trunk rotation and reduced elbow extension during unperturbed reaching was significantly correlated with reduced gains in stroke subjects. These findings suggest that compensatory movement patterns may limit the capacity of using the system’s available motor abundancy during whole-body reaching, reducing motor adaptability to environmental constraints.
Generalizability of results is limited for individuals with more severe or more acute stroke and affected by the small sample size. Our findings may not be applicable to activities involving different DFs, such as locomotion or reaches to targets at different locations.48,49 Results may not be applicable to stroke subjects with severe proprioceptive and/or balance deficits. It is also possible that results may have been influenced by the side and type of stroke as well as hand dominance. Furthermore, although the task was performed without vision, individuals with stroke may demonstrate better movement adaptability when visually guided corrections are permitted. Future studies may address these questions.
Implications
Compensatory movements after stroke may limit the capacity for motor equivalence in response to perturbations. During task practice to enhance UL functional ability (eg, task-oriented training, constraint-induced therapy), individuals with stroke may incorporate motor solutions that include unwanted compensations such as trunk displacement, altered arm plane angle, and shoulder elevation. For individuals with good recovery potential (reappearance of elemental motor patterns present prior to stroke 37 ), trunk restraint and/or provision of explicit/implicit feedback in combination with task-oriented training may enhance the capacity for motor adaptability.
Footnotes
Acknowledgements
Thanks are extended to Anatol G. Feldman for his comments on the manuscript, Rhona Guberek for subject recruitment and clinical assessment, Melanie Baniña for help in data collection and to all the participants who volunteered for the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Y.T. was supported by a Richard and Edith Strauss Musculoskeletal Doctoral Fellowship awarded by McGill University. A.A.M. received a doctoral fellowship from Heart and Stroke Foundation of Canada. M.F.L. holds a Tier 1 Canada Research Chair in Motor Recovery and Rehabilitation.
