Abstract
Objective
When a stroke damages the corticospinal tract (CST), it has been hypothesized that the motor system switches to using the corticoreticulospinal tract (CRST) resulting in abnormal arm synergies. Is use of these tracts mutually exclusive, or can the motor system spontaneously switch between them depending on the type of movement it wants to make? If the motor system can share control at will, then people with a rudimentary ability to make dexterous movements should be able to perform synergistic arm movements as well.
Methods
We analyzed clinical assessments of 319 persons’ abilities to perform “out-of-synergy” and “in-synergy” arm movements after chronic stroke using the Upper Extremity Fugl-Meyer (UEFM) scale.
Results
We identified a moderate range of arm impairment (UEFM = ~30-40) where subjects had a rudimentary ability to make out-of-synergy (~23%-50% on the out-of-synergy score) and dexterous hand movements (~3-10 blocks on Box and Blocks Test). Below this range persons could perform in-synergy but not out-of-synergy or dexterous movements. In the moderate range, however, scoring better on out-of-synergy movements correlated with scoring worse on in-synergy movements (P = .001, r ≈ −0.6).
Conclusion
Rudimentary dexterity corresponded with reduced ability to move the arm in-synergy. This finding supports the idea that CST and CRST compete and has implications for rehabilitation therapy.
Introduction
A hallmark of hemiparetic stroke is the loss of fine motor control in the contralesional arm and hand that is characterized by grouped movement patterns known as synergies. 1 Attempts to lift the arm against gravity result in a flexor synergy pattern characterized by shoulder flexion and abduction, and flexion in the elbow, wrist, and fingers together as a unit. Attempts to reach the opposite knee result in an extensor synergy movement pattern, characterized by shoulder adduction/internal rotation, extension of the elbow and wrist, and finger flexion/adduction. These patterns were documented by Twitchell 2 in the 1950s and formed the basis of one of the most widely used assessments of upper extremity impairment, the Upper Extremity Fugl-Meyer (UEFM) Assessment. 3 To obtain a UEFM score, a trained evaluator grades the subject’s ability to move the arm within the flexion and extension synergies, followed by their ability to make movements out of these synergies.
More recently, sensor-based studies supported the existence of abnormal synergies by showing that the arm joint torques that persons with hemiparesis can produce are constrained in a way consistent with the abnormal synergies.4-7 For example, when a person with hemiparesis produces a shoulder abduction torque (part of the flexion synergy), they exhibit a substantially decreased ability to generate an elbow extension torque (part of the extension synergy). Unimpaired subjects do not show this constraint and are able to more independently control arm joint torques.4,5,7
What causes this abnormal muscle coupling? A leading hypothesis supported by the work of Dewald et al5,6,8-12 and others13-15 is that abnormal synergies arise because the motor system recruits the contralesional corticoreticulospinal tract (CRST) to control arm movement, thereby circumventing the damaged corticospinal tract (CST). Following damage to the CST, the reticulospinal tract (RST) can still be accessed through contralesional motor areas.
Anatomical and neurophysiological data suggest that this is done primarily through the premotor cortex (PM) and supplementary motor area (SMA),16-18 potentially forming a compensatory backup system.15,19,20 Evidence for use of the CRST after stroke is still mostly indirect but is accumulating and includes (1) CRST diffusely innervates spinal segments in a way consistent with flexion and extension synergies 21 ; in contrast, CST exhibits a more focal innervation suitable to support of independent arm joint torques.15,18,22 (2) The contralesional hemisphere exhibits increased activity in people with more severe arm paresis.18,20,23 (3) Motor-evoked potentials evoked by stimulating the contralesional hemisphere are increased in people with hemiparesis.24,25 (4) Involuntary grasping activity correlates with increased activation in contralesional but not ipsilesional cortical motor areas, suggesting that this gross hand movement may be mediated by CRST. 18 (5) Adding the action of lifting the arm against gravity to the action of opening the hand also increases cortical activity in ipsilateral (contralesional) motor areas and decreases activity in contralateral (ipsilesional) motor areas, again suggesting that abduction of the arm may be mediated by CRST. 18 (6) Diffusor tensor imaging (DTI) metrics of patients with severe CST injury indicate disruption of the CST but increased integrity of RST compared with control subjects.26,27 (7) DTI also indicates that CRST integrity increases with increasing arm impairment after stoke, while CST integrity decreases. 20
Recruitment of the CRST pathway may thus be an adaptive strategy that enables persons with severe CST damage to achieve a greater level of movement ability, albeit movement ability constrained to occur in abnormal synergy patterns. That is, switching to use of the CRST may preserve gross motor control at the cost of more fine, individuated motor control. But what if a stroke does not fully disable the CST? Can the motor system spontaneously change back to using the CRST when the CST would be a better substrate for the specific type of movement it wishes to make? Or is use of each tract mutually exclusive at any given level of impairment? As shown in Figure 1, we propose these possibilities as 2 conceptual models—a “Shared” and an “Exclusive” model—for how the CST and CRST might be used in motor recovery after stroke.
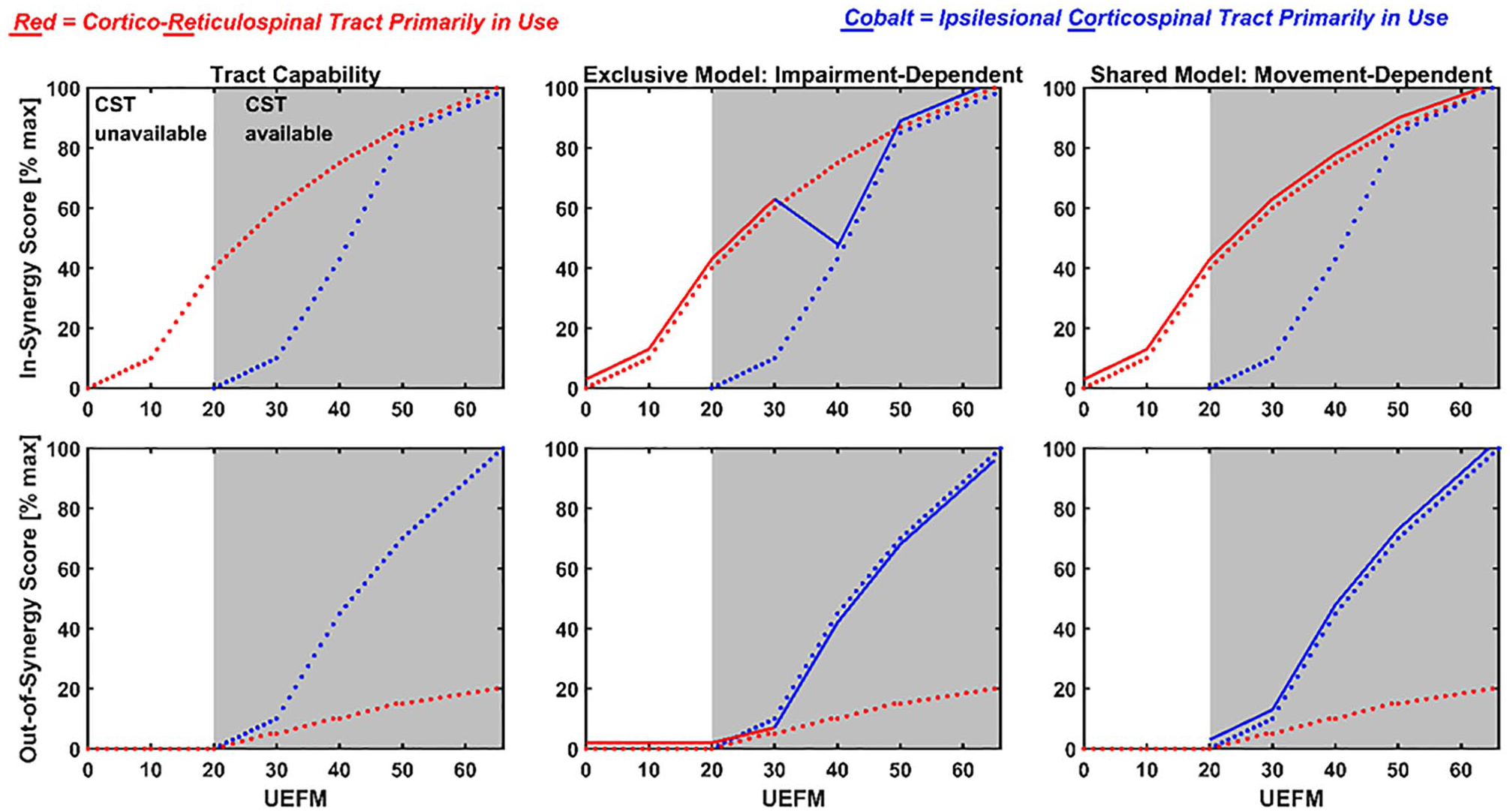
Two conceptual models (Exclusive and Shared models) of how the motor system could use the corticoreticulospinal tract (CRST) and ipsilesional corticospinal tract (CST) to control arm movement after stroke. For all graphs, the x-axis represents impairment, quantified using the Upper Extremity Fugl-Meyer (UEFM) score. Lower UEFM scores indicate more severe impairment and higher scores less severe impairment. Left column: Tract capabilities. As a basis for both conceptual models, these 2 graphs plot the theoretical capability of the CRST and CST to perform 2 different classes of movement as a function of impairment level. The classes are: in synergy (top graph) and out of synergy (bottom graph). CRST is well-structured to mediate in-synergy movements (red dotted line, top graph) because of its more diffuse pattern of termination onto the flexor and extensor motoneuronal pools. It thus outperforms CST (cobalt dotted line, top graph), which must individually activate muscles to create a multiple-muscle synergy, which in turn requires that the parts of the CST responsible for activating each target muscle be preserved, and that these parts be preserved in sufficient quantity that the CST can sufficiently recruit those muscles. We further assume that both CRST and CST capability depends on the level of anatomical damage to the tract, and thus tract capability increases with increasing UEFM for both tracts. CST is better structured to mediate out-of-synergy movements, an ability that emerges in the range UEFM [20-30] (bottom left graph). Middle column: Exclusive model. The dotted lines reproduce the capabilities from the left column graphs. The solid lines indicate by their color which tract is being used in the model. For the Exclusive model, the motor system chooses and sticks with one tract to perform all movements, although which tract it chooses depends on the UEFM score of the person. At low UEFM scores (UEFM < 30), the motor system chooses the CRST (red) to control both in-synergy (top middle graph) and out-of-synergy (bottom middle graph) movements. At higher UEFM scores (UEFM > 30), it chooses CST (cobalt) to control both types of movements. As a result, at a moderate impairment level (UEFM ~30-40), the Exclusive model predicts a decrease in the in-synergy task score as out-of-synergy task score increases. Thus, in the Exclusive model, we expect a negative correlation between the in-synergy and out-of-synergy scores in a moderate UEFM range. Right column: Shared model. In this model, the motor system chooses which pathway to use based on the type of movement it wishes to perform, at any impairment level. We reasoned that if the motor system can use either pathway, it will choose CSRT (red) for in-synergy movement and CST for out-of-synergy (cobalt) movement. Thus, scores for both types of movements should monotonically increase over the UEFM range, following tract capability. In sum, we assert that we can distinguish the Exclusive model from the Shared model by testing if there is a dip in UEFM score for in-synergy movement capability, causing a negative correlation with out-of-synergy movement capability.
There is some evidence that the CST and CRST can work together. For example, the finding of activation in both the ipsilesional and contralesional motor cortices during arm movement after stroke28,29 is consistent with this idea. Also, a recent experiment examining the startReact, 30 an involuntary release of a planned movement via the startle reflex thought to reflect CRST involvement, also supports this idea. In unimpaired individuals, startReact manifested during gross hand grasp but not individuated finger movements, suggesting that the CRST is called into play for one type but not the other type of hand movement.
Here, we reasoned that if the motor system can share control tracts to control different movements, then people with a stroke that preserves some CST function should be able to perform both in-synergy and out-of-synergy movements, and these abilities would follow similar recovery trends. That is, they should be able to use the CRST when they want to perform an in-synergy movement (Figure 1, right column, top graph), and use the CST when they want to perform an out-of-synergy movement (Figure 1, right column, bottom graph). If sharing is not possible, we expected that some people who use a damaged CST would, for some range of impairment, perform relatively worse on in-synergy type arm movements since a damaged CST would theoretically be worse than an intact CRST at mediating in-synergy movements (Figure 1, middle column, top graph).
Methods
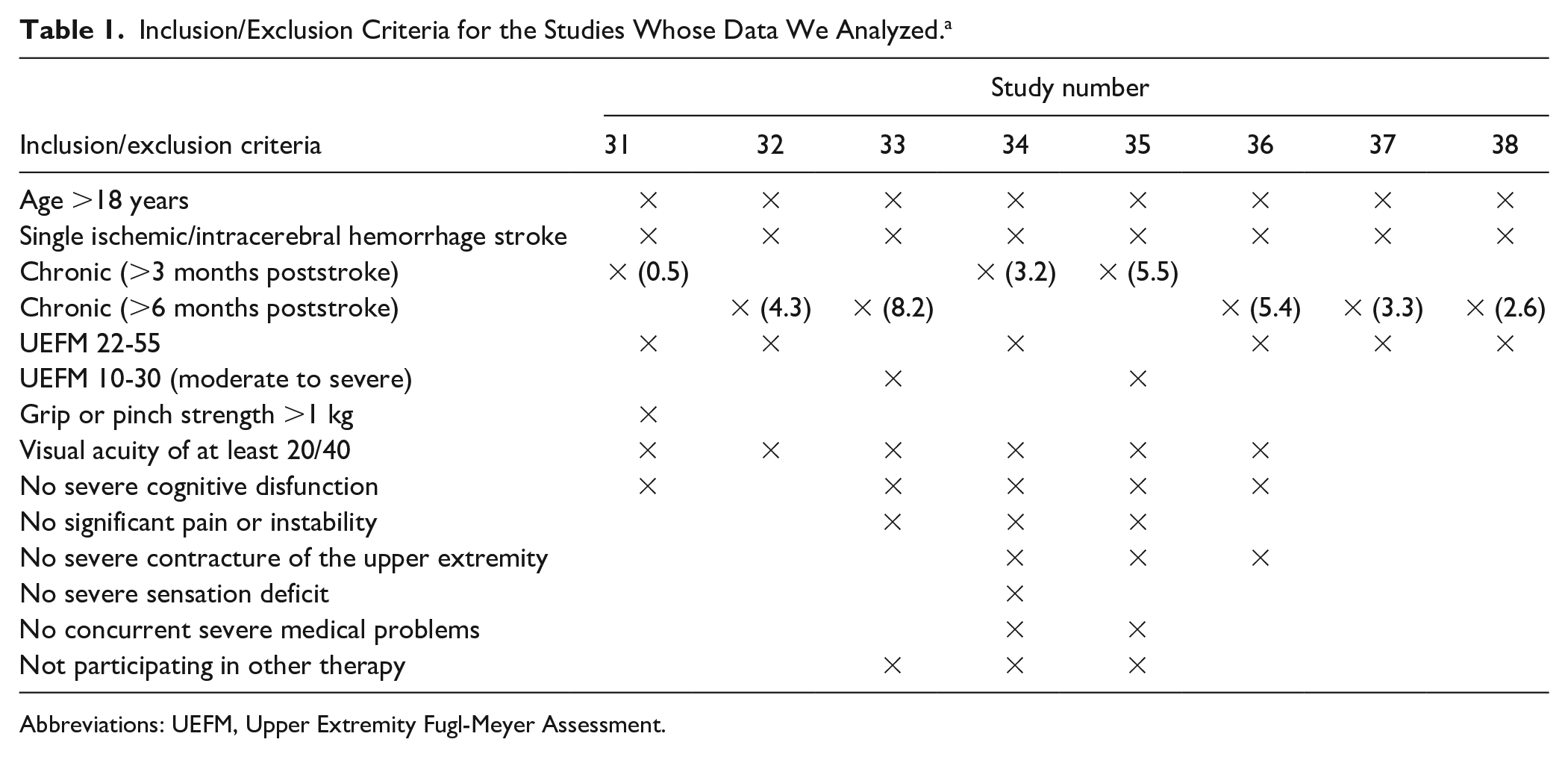
We used UEFM measurements from 319 persons with a chronic stroke reported in several movement training studies (Table 1) at University of California Irvine.31-38 In these studies, 3 licensed, trained physical therapists with more than 15 years of experience performed the UEFM test using a standardized scoring methodology described previously. 39 The interrater difference in UEFM scoring, as quantified in the scoring methodology study, as well as in other studies40,41 is approximately constant across UEFM score and has a zero-mean normal distribution with a standard deviation of ~2. Thus, it is unlikely that inconsistencies in UEFM scoring methodology caused the patterns we describe below.
Inclusion/Exclusion Criteria for the Studies Whose Data We Analyzed.a
Abbreviations: UEFM, Upper Extremity Fugl-Meyer Assessment.
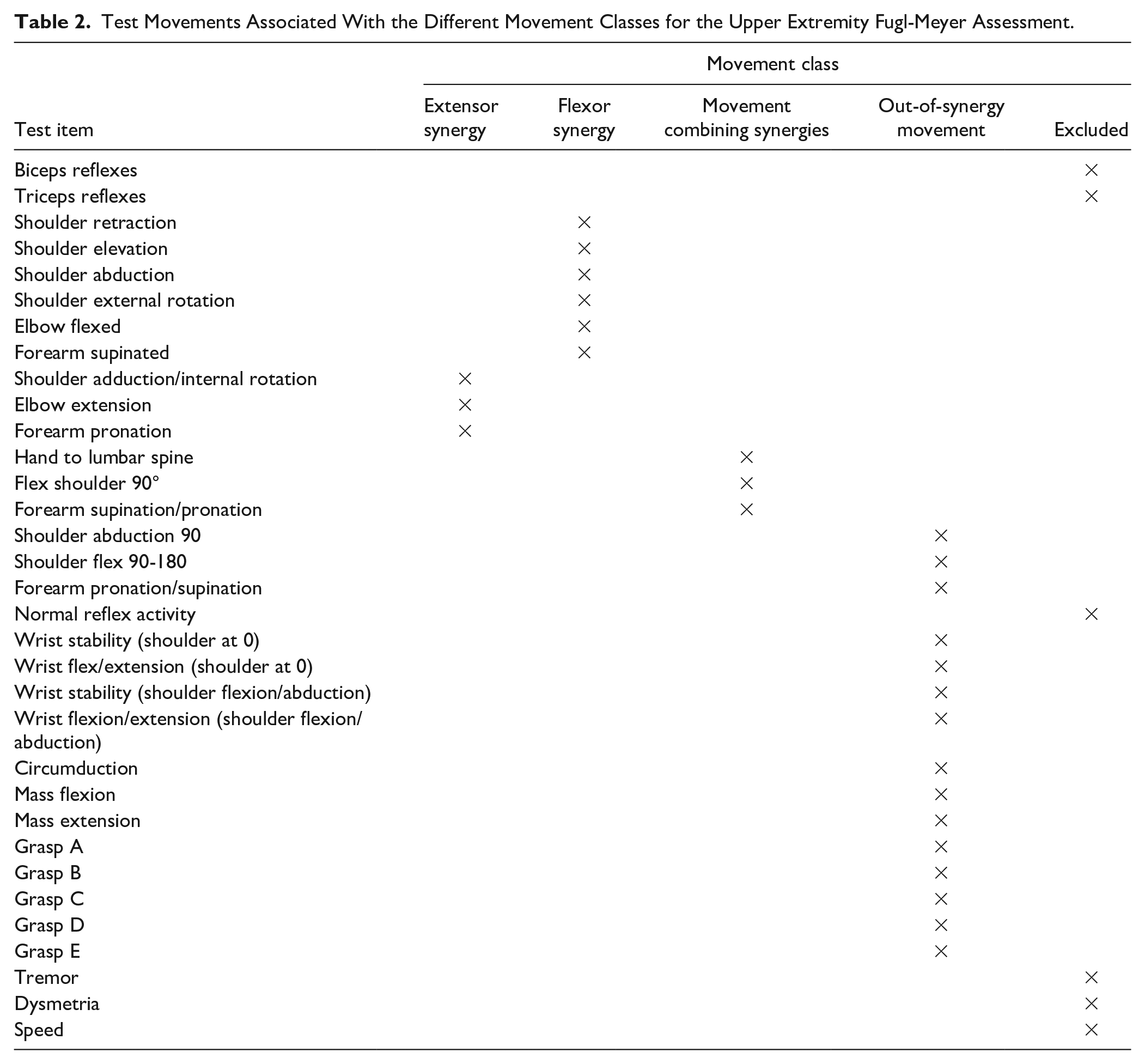
We grouped subjects by scores into 11 bins: 10-14 (n = 20 subjects), 15-19 (15), 20-24 (23), 25-29 (15), 30-34 (35), 35-39 (18), 40-44 (25), 45-49 (17), 50-54 (29), 55-59 (84), 60-65 (38). Consistent with the way the UEFM scale is normally organized, we grouped UEFM test items into four classes: extensor synergy (3 items), flexor synergy (6 items), movement combining synergies (3 items), and out-of-synergy movement (15 items) (Table 2). We ignored the 3 reflex test items, since all subjects scored a 2 on the first 2 reflex items. The third reflex item—“Normal Reflex Activity of Biceps and Triceps” is evaluated only if the subject scores a total score of 6 on the “Volitional Movement No Synergy Dependence” task (Shoulder Abduction 90, Shoulder Flex 90-180, and Forearm Pro/Supination items) and therefore was not scored for most subjects. We also ignored the score on the finger-to-nose task (Tremor, Dysmetria, and Speed items) due to the unique scoring instructions. A subject who cannot move their arm will score a 2 for Tremor and Dysmetria since they are not observed, which is different from the scoring instructions for other items, where a 0 score is given for the inability to perform the task. Furthermore, Pandian et al 42 found no correlation between the above items and other items of the UEFM scale (except of a moderate correlation between “Normal Reflex Activity” and “stability of the wrist”), suggesting that these items lack interrelationship.
Test Movements Associated With the Different Movement Classes for the Upper Extremity Fugl-Meyer Assessment.
We calculated the fraction of subjects who scored 0, 1, or 2 on each of the remaining 27 items for each bin of total UEFM score. For each movement class, we averaged these fractions across the items belonging to that class. Note that a score of 0 corresponds roughly to the concept “can’t perform,” 1 “can partially perform,” and 2 “can successfully perform,” with the criteria for assigning a score defined specifically for each test item so that trained raters can achieve a high level of consistency.3,39,43
To compare fractions of subjects who scored 0, 1, or 2 between bins, we calculated the difference in fraction for each item between bins and grouped them into the previously defined movement classes. Next, we compared between-bin differences for the 4 classes using nonparametric Mann-Whitney-Wilcoxon U test.
44
We also tested for correlation between the total subscore for each movement class for the individuals’ scores using the nonparametric Spearman’s correlation coefficient and a sliding window of 10 points on the UEFM score to select subgroups of the 319 subjects for which to calculate the correlation. The total subscore for each movement class range was as follows: (0-12) for the flexor synergy, (0-6) for the extensor synergy, (0-6) for the combing synergies, and (0-30) for the out-of-synergy movement. We moved the sliding window over the range of the UEFM score, starting at F0 = 10. Each window included all subjects in the data set who had a UEFM score in the range of:
Results
As the total UEFM score increased, the fraction of subjects unable to perform the various classes of movements (i.e., scored 0 on them) generally decreased, while the fraction of subjects who could perform the movement classes (i.e., scored 1 or 2) generally increased, for all 4 classes of test items (i.e., extensor synergy, flexor synergy, movement combining synergies, out-of-synergy movement, Figure 2). The fraction of subjects who could partially perform the flexor synergy (i.e., scored 1) was the first to increase, resulting in a greater fraction of subjects who scored 1 in the UEFM range [10-20] for flexor versus extensor synergy (McNemar mid-P test 46 = .03). This increase in flexor synergy was followed by an increase in the fraction who could perform the extensor synergy, movement combining synergies, and then out-of-synergy movement (Figure 2B).
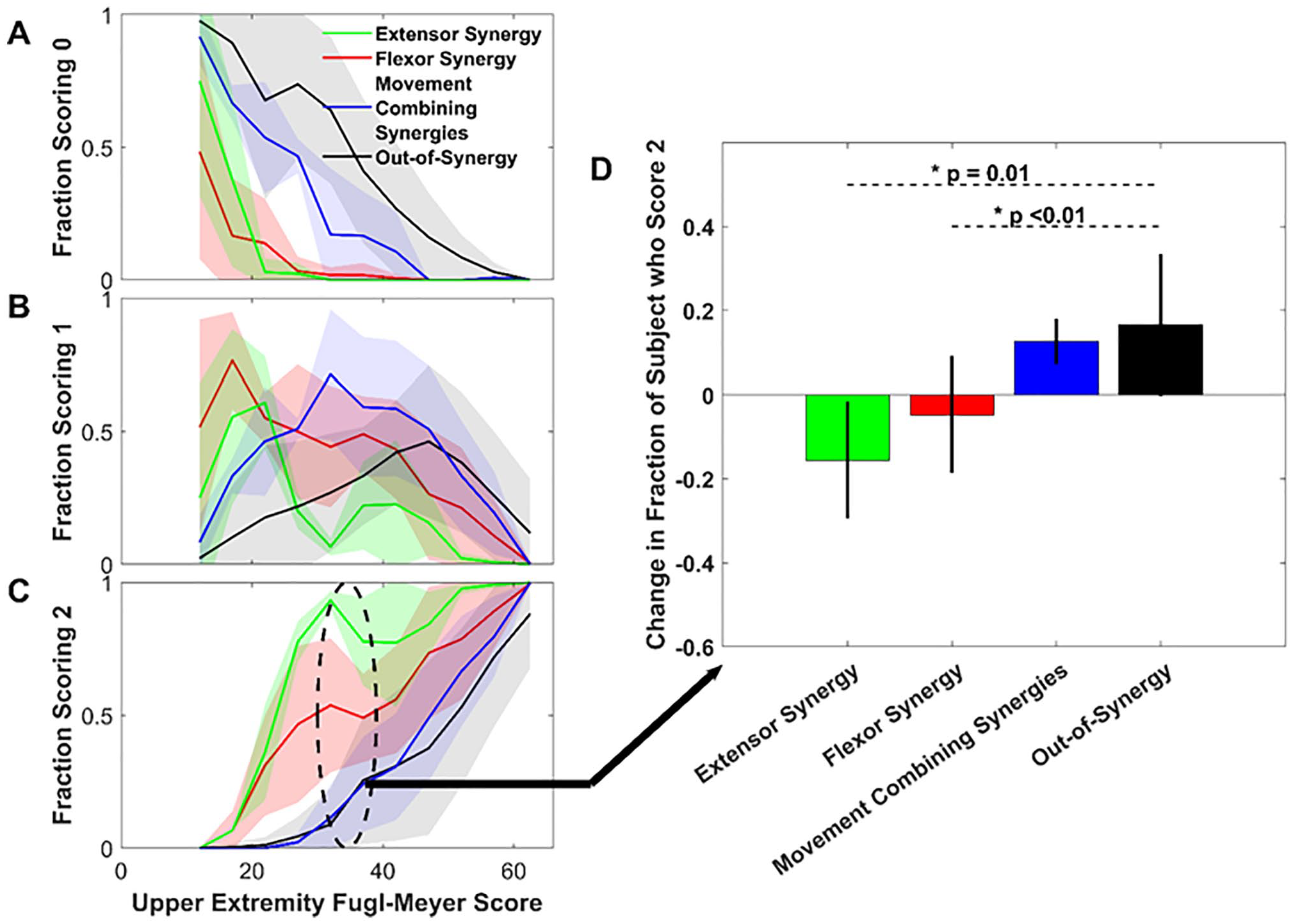
The fraction of subjects who scored 0 (A), 1 (B), or 2 (C), averaged across Upper Extremity Fugl-Meyer Assessment (UEFM) test items corresponding to four classes of movement. Colored area represents ± 1 standard deviation (D) The mean difference in fraction of subjects in bin 35-39 who scored 2 on items compared to the fraction in bin 30-34, separated by movement class. Bars show mean ± 1 SD; * indicates significant difference between movement classes (U test).
There was an exception to the general trend of increasing rate of recovery across movement classes as UEFM increased, however. The average fraction of subjects (across items in the movement class) that was able to fully perform the flexion and extension synergies did not increase as quickly as UEFM score increased into the range 35-39 (Figure 2C). This range is also where the fraction of subjects able to perform out-of-synergy movement started to more substantially increase. To analyze whether this relative drop in recovery rate was statistically significant, we calculated the change in fraction scoring a “2” between bins [30-34] and [35-39] for the four different classes (Figure 2D). There was a significant difference between bins in change in fraction of subjects who scored a “2” on the flexor synergy class (Δ = −0.047; P = .01, U test) and extensor synergy class (Δ = −0.156; P < .01) when compared with the between-bin change in fraction of scoring 2 on the out-of-synergy movements evaluated for the same bins.
The question arose as to whether this relative drop in in-synergy recovery rate compared with out-of-synergy recovery rate in this range also manifested for individual subjects. We calculated the correlation coefficient between the total subscore for each movement class using a sliding window of 10 points on the UEFM score to select subgroups of the 319 subjects for which to calculate the correlation (Figure 3). Moving the sliding window resulted in 46 correlation calculations (mean sample size across correlations = 63 ± 29 SD subjects; minimum sample size = 40 subjects in the UEFM range [17, 28], maximum sample size = 130 subjects in the UEFM range [53, 64]). We used a significance level corresponding to an unweighted Bonferroni correction (i.e., P = .0011). For subjects in the moderate range, but not subjects in the severe or mild ranges, the summed subscore they achieved on out-of-synergy test elements was negatively correlated with the summed subscore on in-synergy elements, for both the flexion (P < .0011, r ranging from −0.7 to −0.5 in UEFM range [27-44]) and extension synergy (P < .0011, r ranging from −0.5 to −0.6 in the UEFM range [32-40]) (Figure 3A and B). A significant positive correlation was found at the UEFM range [15-18] for the extensor synergy and out-of-synergy movement (P < .0011, r = 0.65). A significant (P < .0011) positive correlation was found between movement combining synergy and out-of-synergy movements in the UEFM range [25-30] (r = 0.48-0.63) and [57-60] (r = 0.4).
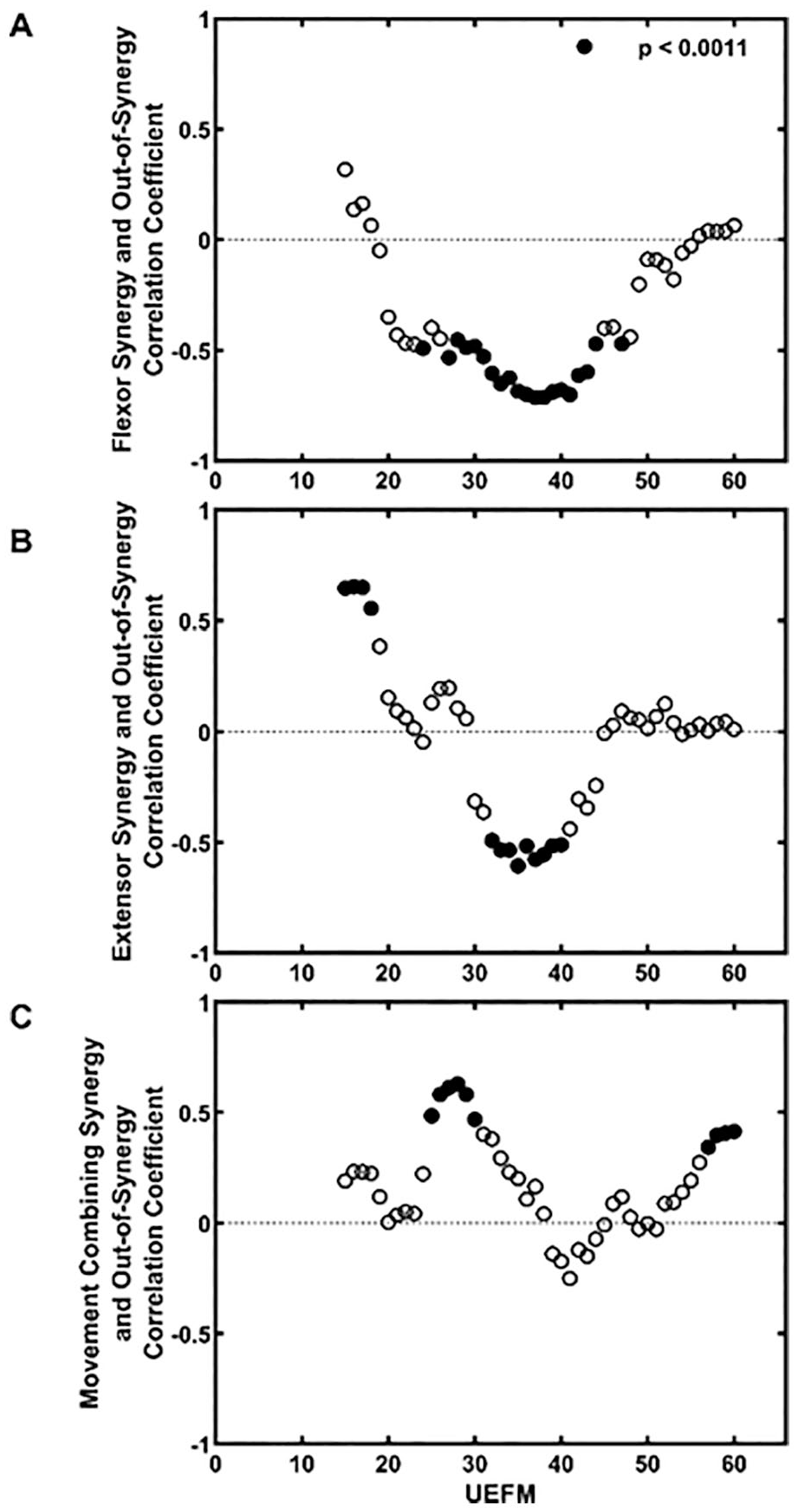
Correlation analysis of the different classes of movements, as a function of Upper Extremity Fugl-Meyer Assessment (UEFM) score. We calculated correlation coefficients using a sliding window of 10 points on the UEFM score to select subgroups of the 319 subjects for which to calculate the correlation. Filled-in points indicate statistical significance (unweighted Bonferroni correction 45 : α = .05/(N = 46) = 1.1e-3, where N = 46 is the number of the total performed correlation analyses over the UEFM range [10-65]). For subjects in the moderate range, but not subjects in the severe or mild ranges, the summed subscore they achieved on out-of-synergy test elements was negatively correlated with the summed subscore on in-synergy elements, for both the flexion and extension synergies.
As described in the Introduction, the ability to move out of synergy is hypothesized to rely on the CST. Another function of CST is mediating dexterous hand movement.30,47 To understand when the ability to move the hand emerges as a function of UEFM score, we analyzed dexterous hand items (mass extension and grasps A-E, n = 6) compared with the arm items (and gross flexor grasp, n = 21) (Figure 4A). Similar to the synergy-based analysis, the fraction of subjects who could score 2 on the arm items was nonzero in the lower range of the UEFM, while the fraction who could score 2 on the dexterous hand items was near zero. Thus, the ability to perform arm movements emerged before the ability to perform dexterous hand movements. The fraction of subjects who could score 2 on the dexterous hand items began to increase above zero at an UEFM score of about 22, with an increase in this tendency at an UEFM score about 34. As a second measure of hand function, we plotted each subject’s Box and Blocks Test (BBT) score against the UEFM score, for the subjects for whom we had these data available as well as data we scanned from Platz et al, 41 resulting in 140 subjects in total (Figure 4B). The BBT is an assessment for unilateral manual dexterity for which a subject is required to grasp, transport, and release as many blocks as possible in 60 seconds. The pattern was similar to the pattern for UEFM hand items: BBT score increased above zero around UEFM = 22, with a further increase around UEFM = 34. Thus, hand dexterity, presumably mediated by CST function, emerged in the moderate range of impairment, similar to the ability to move the arm out-of-synergy.
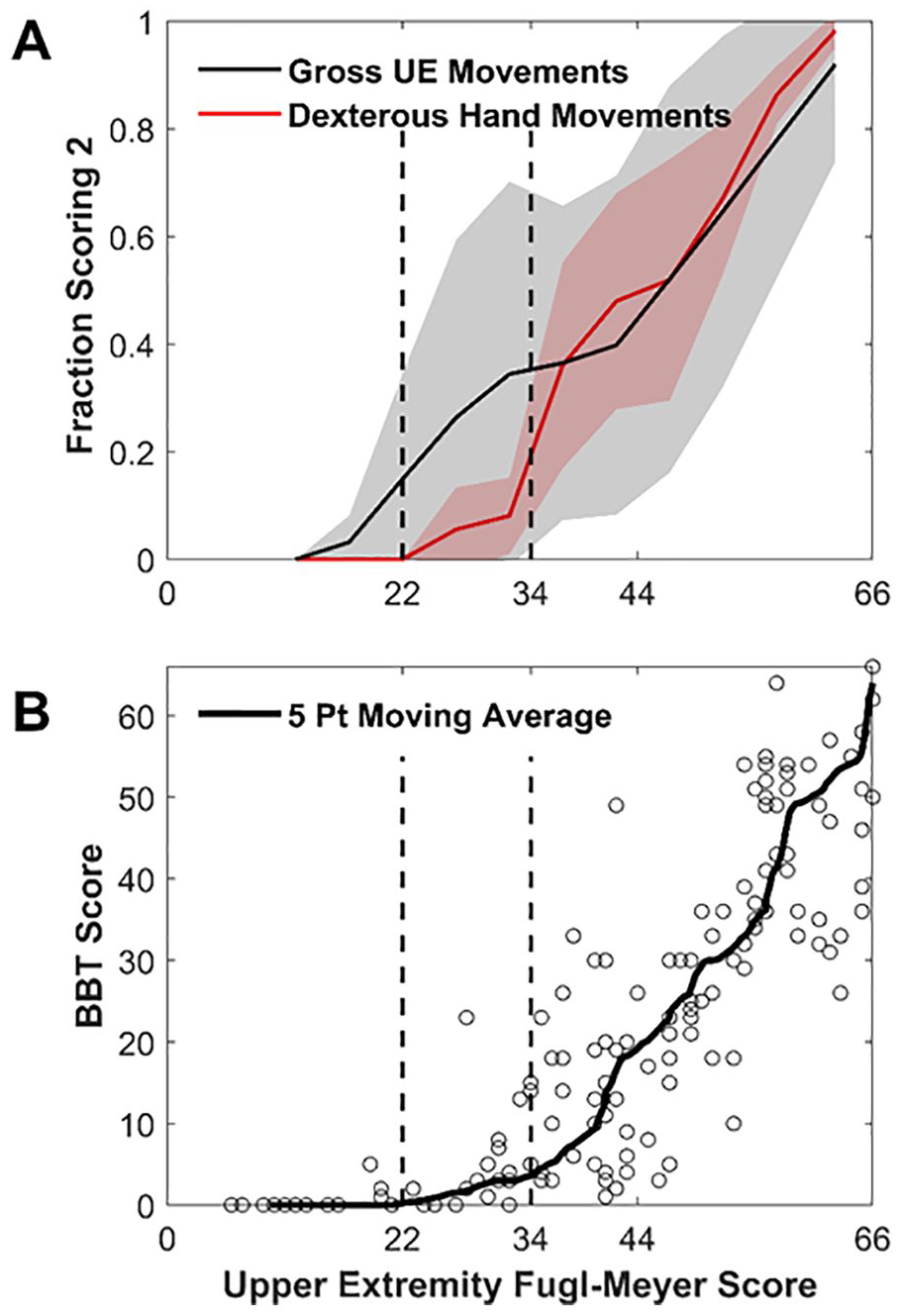
Analysis of gross upper extremity and dexterous hand movement ability as a function of Upper Extremity Fugl-Meyer Assessment (UEFM) score. (A) Fraction of subjects who scored a 2 on all UEFM arm items (and gross flexor grasp, black line) or dexterous hand items as a function of total impairment level. Colored area represents ± 1 standard deviation. The fraction of subjects who could well-perform dexterous movements exceeded zero when the total UEFM score was greater than 22, with a steeper increase in hand movement ability for subjects in the range [34-44] (B) The Box and Blocks Test (BBT) score as a function of total impairment level. Subjects with UEFM greater than 22 were able to grasp and release blocks on the BBT. An increase in the slope of the BBT score is again noticeable around UEFM = 34.
Finally, returning to an earlier observation, the ability to perform the flexor synergy partially recovered before the extensor synergy for severely impaired individuals (UEFM [10-20]). However, in an UEFM range of [20-40], the fraction of subjects who scored 2 on the extensor synergy significantly exceeded the fraction scoring 2 on the flexor synergy (McNemar mid-P < .01). To gain insight into what drove this reversal, we examined the individual test items associated with the extensor and flexor and synergies. The fraction of people scoring well on the extensor items was relatively consistent across items, producing a low variance. The fraction scoring well on the flexor items exhibited a larger variance. We found that part of the cause of this variance was that, for subjects in the UEFM range [30-34], the fraction who could fully perform each of these test items was greater than 0.5 except for the Forearm Supinated item, which was 0.06. When we removed the Forearm Supinated item, the fraction scoring 2 on the rest of the items grew from 0.54 to 0.64 and the variance decreased, more closely mirroring the extensor synergy recovery, but still not fully.
Moreover, for the UEFM bins of interest identified in the initial group-wise analysis above (ie, bins [30-34] and [35-39]), we observed that the change in fraction of subjects who fully performed the Forearm Supinated item was positive, while for all other flexor synergy items the change was negative. Taking all items for the flexor synergy together, the absolute drop between these bins was not significant (P = .44). However, when we removed the Forearm Supinated item from this class, the drop in UEFM fraction became significant (P = .01). Thus, the fraction of subjects who could perform the modified flexor synergy movement class dropped temporarily as recovery progressed. For the extensor synergy the absolute drop between those bins was not significant (P = .11).
Discussion
We first identified and compared 2 groups of people using archived data from several studies that performed UEFM assessments on 319 persons who had experienced a stroke at least three months previously. The first group (UEFM [30-34]) was rated as more impaired than the second group (UEFM [35-39]) based on UEFM score. Members of this first group had little to no hand dexterity and struggled to move the arm in a coordinated way (i.e., in “out-of-synergy” patterns). However, they were relatively better at performing arm movements “in-synergy.” These findings are consistent with the idea that this group relied on the CRST pathway. The second group exhibited some return of hand dexterity and some ability to move the arm out-of-synergy, consistent with the idea that this group relied on the CST pathway. In this UEFM range, the rate of recovery of the synergy classes was significantly less than the rate of recovery of the out-of-synergy class. When we further analyzed this observation, we found a negative correlation between in-synergy scores and out-of-synergy scores, but only for participants with UEFM scores falling in a moderate range (UEFM [~30-40]). This trade-off was not observed for groups of individuals who were rated as more or less impaired. A correlate to the presence of the trade-off was whether the group had rudimentary ability to move out of synergy (defined as scoring ~23%-50% on the out-of-synergy score) and rudimentary hand dexterity (defined as scoring ~3-10 on the BBT), both thought to be markers of CST use.
One way to interpret these findings is that individuals who rely on a severely damaged CST struggle to effectively use the CRST, consistent with the Exclusive model of Figure 1. That is, there is some form of competition between the tracts, and it is not possible to share control between the tracts based on the type of movement (i.e., in-synergy or out-of-synergy) that one wishes to perform. Stated more precisely, we hypothesize that individuals in the chronic phase after a stroke with an UEFM score in a moderate range typically rely on “weak” residual CST for upper extremity movement control. They are worse at performing abnormal synergies than more impaired subjects because their “weak” CST is not as competent as the more impaired subjects’ CRST at generating those movements.
Although it is sometimes criticized because it is a coarse, summed scale, the UEFM is one of the most widely used measures of upper extremity impairment in poststroke hemiparesis.3,39,48,49 It has well-established reliability and validity across different stroke recovery time points48,49 and is associated with corticospinal tract integrity. 50 A recent high-resolution structural magnetic resonance imaging study 20 of both the brainstem and the cervical spinal cord found a correlation between motor impairment severity, assessed with the UEFM, and decreases in ipsilesional corticospinal tract integrity and increases in contralesional medial reticulospinal tract integrity. Hence, analysis of UEFM data can divulge changes in the use of motor pathways after stroke.
The group analysis of UEFM scores presented here also supported several established concepts of stroke recovery. One concept is that recovery proceeds in a pattern from movement in-synergy, to movement combining synergy, to movement out-of-synergy, a concept that mirrors the relative ordering of the fraction of subjects who can do each of these movement classes (Figure 2C). Another concept is that arm movement ability recovers before dexterous hand movement ability, which is supported by the data in Figure 4A. One more concept is that there is a bias toward flexion movements for more severely impaired persons.1,2,51 Flexor bias is consistent with the higher fraction of the more-impaired subjects (UEFM [10-20]) who scored 1 on the flexor synergy compared to the extensor synergy (Figure 2B). This flexor bias is congruous with the use of CRST by these individuals, as it has been shown in both nonhuman primates 14 and in recent human magnetic resonance imaging studies 20 that CRST recruitment is associated with flexion synergy expression.
However, even though a flexor bias was apparent at a severe level of impairment, extensor activity recovered more quickly as a function of UEFM score as UEFM score increased above 20. We found that part of the explanation related to UEFM flexor synergy score properties: a single flexor synergy item—the Forearm Supinated item—which was more resistant to recovery than the other flexor and extensor synergy items. The flexor synergy score thus appears to contain an item that may be better associated with CST function, and this miscategorization might account for some of the apparent “lag” in flexor recovery in this range. However, it does not fully account for the lag, and thus there may indeed be changing rates of flexor and extensor synergy recovery that depend on impairment level, perhaps arising from specific properties of components of the CRST.
Limitations
This study has several limitations that suggest directions for future research. First, while sensitive and reliable, the UEFM is a coarse scale, having only 3 levels of grading for each test movement. We moderated this limitation by examining data from a large group of subjects allowing us to generate a continuous measure of recovery for each test item (i.e., the fraction of subjects in a range of UEFM scores able to score 0, 1, or 2 as well as the summed subscores for different categories of movement). However, the results presented here should be tested using finer resolution measures of arm motor control, such as multijoint force production capability, quantifying more precisely the ability to move in synergy and out of synergy.
Another limitation is that we had no imaging information about stroke location, including, importantly, the amount of damage to the CST; we instead relied on behavioral inference. Furthermore, we had no imaging or neurophysiological data that confirmed our behavioral inferences about how our subjects’ motor systems used CST or CRST. Future studies should combine behavioral measures with imaging and neurophysiological measures to test the competition hypothesis suggested here. Evidence for increased involvement of the CRST following stroke, including what we present here, is still mostly indirect, and requires further anatomical and neurophysiological confirmation.
Another pathway that may play a role in recovery is contralesional uncrossed CST. We did not consider a possible role for uncrossed CST here in part because its role in recovery is still uncertain as reviewed in Zaaimi et al. 52 For example, in one study, 53 synaptic responses measured in ipsilateral motor neurons innervating hand and forearm muscles evoked by cortical stimulation in both lesioned and control adult macaque monkeys were rare and weak. While we framed the interpretation of our results in terms of the putative roles of the ipsilesional CST and CRST in upper extremity recovery, we should mention another possible contributor to the emergence of abnormal synergies. Yao et al 54 found that, for unimpaired persons or persons with moderate impairment after stroke, the overlap in cortical activation in bilateral sensorimotor cortices (premotor, supplementary motor, primary motor and primary somatosensory cortices on the contralateral and ipsilateral hemispheres) associated with either shoulder or elbow motor tasks decreased during motor preparation, but it increased in individuals with severe stroke. Additionally, for unimpaired persons, the cortical activity was restricted to cortices on the contralateral hemisphere, whereas for moderate to severely impaired persons there was a scattered bilateral activity. Thus, increase in cortical overlap and increased use of the CRST may be connected and both may contribute to the appearances of the abnormal synergies in more severely impaired persons. The decrease in cortical overlap found for the moderately impaired group might suggest that different residual cortical resources can be used for different motor tasks resulting in the increased ability to control individual joints (out-of-synergy movement) and increased dexterity. However, it seemingly cannot explain the decrease in the ability to perform in-synergy movements. If the use of different cortical resources can be modulated, it is unclear why several resources cannot be recruited to perform the synergies. Future research should combine brain imaging of the cortex and brainstem with finer resolution assessment of arm and hand movement ability to test these concepts.
Clinical Implications
We recently published an analysis of the “rule breakers” or “nonfitters” of the proportional recovery rule. 55 We showed that a special group of these nonfitters (22 < UEFM < 40) responded better to intensive movement training in the chronic stage following stroke. Our current analysis suggests that this group is at least partially comprised of individuals who switch to using a “weak” CST, thereby exhibiting some return of hand dexterity and some ability to move the arm out of synergy. If so, a clinical implication is that there may be people with a “weak” CST who have not made the “switch” to using it. In such cases, it would seem logical that rehabilitative movement training should focus on helping patients explore away from using the CRST toward using the CST. This could presumably be accomplished by presenting tasks that require the CST, thereby promoting switching. Even though patients may experience a degradation in the ability to perform some arm movements as they attempt this switch, in the long run, achieving the switch to using the CST may help them achieve better upper extremity recovery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by 2R01HD062744 from the National Center for Medical Rehabilitation Research at the National Institutes of Health (NIH), and by GM-055246 from the Initiative for Maximizing Student Development (MBRS-IMSD) program at NIH.
