Abstract
Background. The extent to which the upper-limb flexor synergy constrains or compensates for arm motor impairment during reaching is controversial. This synergy can be quantified with a minimal marker set describing movements of the arm-plane. Objectives. To determine whether and how (a) upper-limb flexor synergy in patients with chronic stroke contributes to reaching movements to different arm workspace locations and (b) reaching deficits can be characterized by arm-plane motion. Methods. Sixteen post-stroke and 8 healthy control subjects made unrestrained reaching movements to targets located in ipsilateral, central, and contralateral arm workspaces. Arm-plane, arm, and trunk motion, and their temporal and spatial linkages were analyzed. Results. Individuals with moderate/severe stroke used greater arm-plane movement and compensatory trunk movement compared to those with mild stroke and control subjects. Arm-plane and trunk movements were more temporally coupled in stroke compared with controls. Reaching accuracy was related to different segment and joint combinations for each target and group: arm-plane movement in controls and mild stroke subjects, and trunk and elbow movements in moderate/severe stroke subjects. Arm-plane movement increased with time since stroke and when combined with trunk rotation, discriminated between different subject groups for reaching the central and contralateral targets. Trunk movement and arm-plane angle during target reaches predicted the subject group. Conclusions. The upper-limb flexor synergy was used adaptively for reaching accuracy by patients with mild, but not moderate/severe stroke. The flexor synergy, as parameterized by the amount of arm-plane motion, can be used by clinicians to identify levels of motor recovery in patients with stroke.
Introduction
Deficits in upper-limb (UL) movement after stroke persist in a large proportion of patients well past the acute and subacute stages. 1 Recovery of UL movement may continue into the chronic phase due to the large potential for neuroplasticity in the central nervous system.2,3 It has recently been emphasized that recovery of UL movement requires therapeutic efforts that focus on decreasing motor impairment. 4 Quantitative movement analysis methods have emerged as a means of identifying kinematic patterns that discriminate between UL functional improvements due to true motor recovery and those due to compensatory or substitutive movement patterns.5-10 Specifically, true motor recovery at the kinematic level is defined as the reappearance of typical movement patterns and sequences used before stroke for performance of a task, while compensation is defined as the use of additional or alternate kinematic patterns during task performance. 11
A common motor compensation in patients with chronic stroke is excessive trunk displacement.5,6,12 The amount of compensatory trunk displacement used during a sagittal reaching task has been related to the motor deficit severity (ie, greater severity, more compensatory movement used). Furthermore, when recruited, trunk movement onset occurs before or simultaneously with arm movement onset and continues after the end of arm movement offset, similar to the pattern of arm-trunk recruitment in healthy subjects for trunk-assisted reaching.6,13 This suggests that the trunk is an integral part of the reaching pattern and is not simply recruited in response to limitations in arm movement extent (ie, recruited sequentially). Recovery of isolated reaching ability after training programs incorporating different forms of trunk restraint or feedback on excessive trunk use has been characterized by decreased compensatory trunk movement and improved UL kinematics.13,14
To maximize UL motor recovery, however, it may not be sufficient to focus only on minimizing single-axis trunk compensations. Motor compensations during reaching have also been described in terms of excessive axial trunk rotations15,16 and scapular movements. 17 Another movement feature described in patients with stroke is the flexor synergy,18,19 an abnormal synergistic shoulder and elbow coupling that permits the patient to lift the arm (as a compensation for a deficit in isolated shoulder flexion), while at the same time tends to bring the hand back toward the body. However, it is unclear to what extent the pathological flexor synergy is adaptive for reaches into different parts of the arm workspace by patients with different levels of stroke severity.
In the healthy nervous system, nonpathological kinematic synergies have been described that are based on the ability to produce abundant joint movement combinations to accomplish the same motor goal (ie, “motor equivalence”).20-24 These kinematic synergies are highly adaptive for activities such as reaching, pointing, and hand function. In contrast to adaptive kinematic synergies described in the healthy system, the pathological flexor synergy is an abnormal linkage between arm joints resulting in limitations in performing task-relevant movement that may have reduced adaptability.16,25-27 Information about the extent to which the damaged nervous system preserves kinematic adaptability is important for therapists to determine the potential for motor recovery after stroke and to design personalized treatment programs to recover specific motor elements related to reaching ability.
Based on the principle of motor equivalence, we hypothesized that the damaged nervous system may retain the ability to reorganize trunk and arm degrees of freedom (DoFs) in order to compensate for deficits in arm reaching extent. We characterized the arm flexor synergy using an arm-plane measure28,29 to capture the covariance of the whole arm motion rather than individual DoFs separately.
We predicted that if the arm-plane represents a fixed synergy between different shoulder DoFs regardless of the task, then additional compensatory DoFs would be recruited for reaching. In contrast, if the synergy could be modified according to the task, then it would be adaptive and contribute to reaching to some extent.
Methods
Participants
All subjects were right-hand dominant. They included 16 subjects with left-hemispheric stroke (13 males, age 65.2 ± 9.8 years) and 8 healthy age-matched controls (3 males, age 58.6 ± 7.0 years) with no other neurological, sensorimotor, or orthopedic impairments. Participants with stroke had sustained a single stroke >2 months previously, an arm recovery stage score ≥3/7 on the Chedoke-McMaster Stroke Assessment, 30 no lateral neglect, apraxia, or major cognitive deficits according to standard clinical assessments, could understand instructions and perform the required movements. Individuals with brainstem or cerebellar stroke were excluded. Participants signed informed consent forms approved by the Helsinki Committee (Sheba Medical Center–Israeli Health Ministry, 20060315-HT4666).
For the stroke group, UL impairment was measured with the Fugl-Meyer Arm Assessment (FMA). 31 Maximal scores of 56 points did not include 10 points for reflex testing. Subjects were stratified according to impairment severity based on FMA scores 32 where 50 to 56 points indicated mild motor impairment (n = 7) and 24 to 49 points indicated moderate-to-severe impairment (n = 9; Table 1).
Demographic Data of All Subjects and Clinical Measures of Subjects With Stroke. a
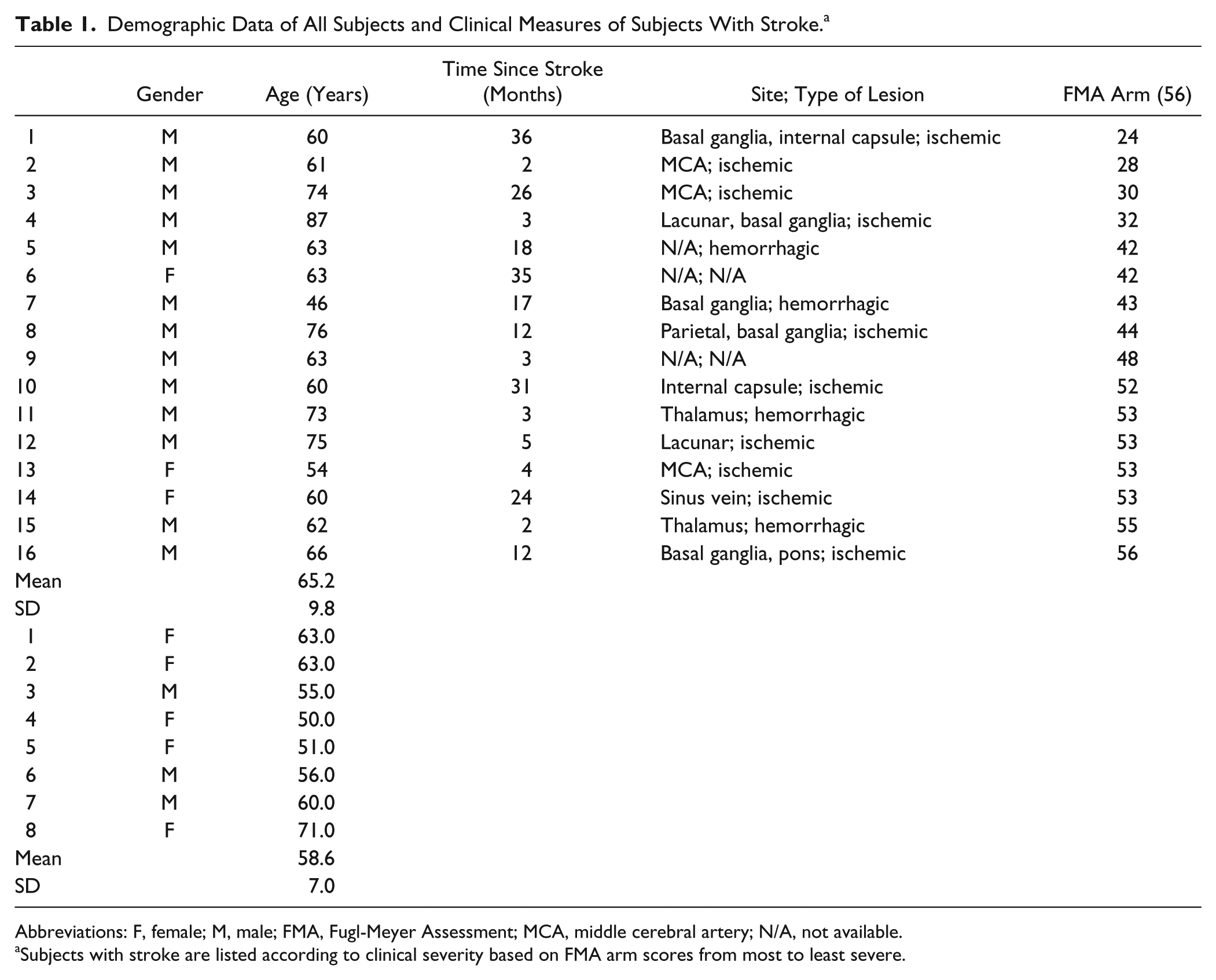
Abbreviations: F, female; M, male; FMA, Fugl-Meyer Assessment; MCA, middle cerebral artery; N/A, not available.
Subjects with stroke are listed according to clinical severity based on FMA arm scores from most to least severe.
Experimental Procedure
Subjects sat on a chair with feet supported and their hand resting on a 100 mm wide × 150 mm long starting platform on a table adjusted to elbow height. The chair had a back support that did not restrict trunk movements. Three targets (30 mm × 30 mm squares) were suspended on transparent wires in front of the subject. Target 2 (central) was placed just beyond the fully extended arm length (measured from acromion to radial stylus process) and aligned with the affected shoulder at eye level (150 mm above the acromion). Targets 1 and 3 were located 150 mm lower and 300 mm to the left (1, contralateral) and right (3, ipsilateral) of the central target. Subjects were instructed to point to specific targets without touching them when prompted by prerecorded verbal cues. Two sets of 21 trials (7 trials per target, fixed random order) were recorded for a total of 42 movements per subject. Several familiarization trials were performed prior to recording to avoid learning effects.
Data were recorded from 3 passive reflective markers paced over the lateral acromion, lateral epicondyle, and wrist styloid process using an arm Cartesian coordinate frame (Figure 1A). Trunk displacement was measured by three orthogonal markers placed on a rigid exoskeleton attached to the subject’s back and extending above the head to avoid marker occlusion. The exoskeleton was not affected by shoulder or head movement. These markers were used to establish a body-fixed trunk Cartesian coordinate frame (Figure 1B). Data were recorded with a 3-camera optoelectronic system (7s, 100 Hz, ProReflex, Qualisys, Göteborg, Sweden) and analyzed offline (Matlab v.7, MathWorks, Natick, MA, USA).
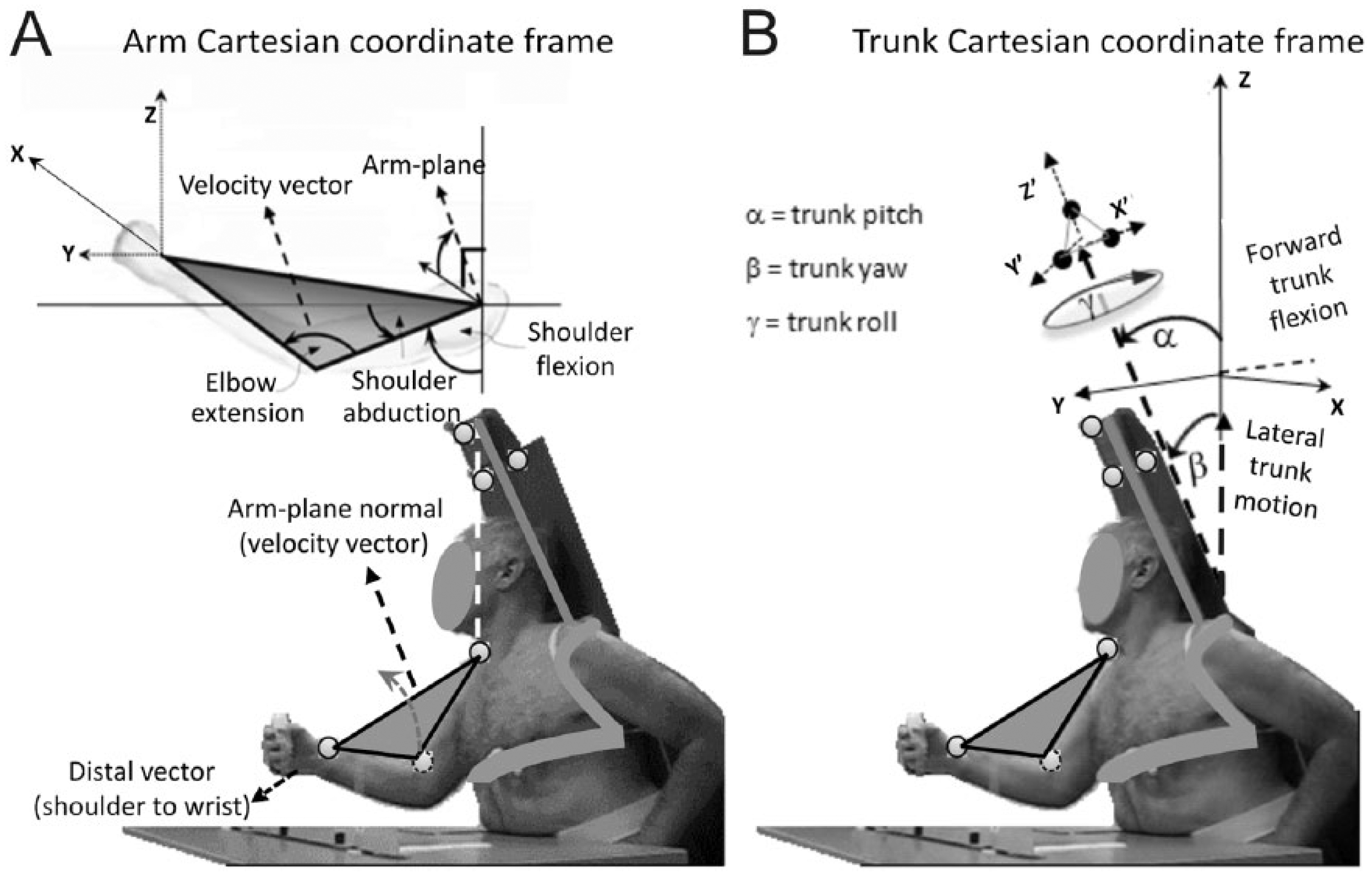
Experimental setup, subject and marker positions as well as arm (A) and trunk (B) Cartesian coordinate frames for data analysis. (A) The arm-plane angle is formed by 3 markers placed on the shoulder, elbow, and wrist and is the complement of the angle between the arm-plane normal and the vertical. Arm-plane motion is illustrated in the upper panel. (B) Markers to track trunk motion are placed on a posterior extension attached to the trunk of the subject, to avoid data loss from marker occlusion. Definitions of trunk angles within the trunk coordinate frame are illustrated in the top panel.
Data Analysis
Hand movement onset and offset were defined as the times at which the wrist marker velocity exceeded and remained above, or decreased and remained below 10% of peak sagittal velocity for 0.01 seconds. Trunk (average of 3 markers) and arm-plane movement onset and offset were similarly determined. Movement trajectories were filtered (5 Hz Wiener filter) prior to velocity computation.
The arm-plane measure was used to quantify the UL flexor synergy since it describes a kinematic variable of reduced dimensionality with a known meaning (Figure 1A). The arm-plane angle captures the kinematics of all 3 shoulder DoFs (rotation, flexion-extension, and adduction-abduction) and can be used to represent whole arm motion in conjunction with the wrist-shoulder vector.28,29 This approach, derived from Screw Theory, applies to systems that rotate and translate simultaneously (during reaching, the shoulder rotates while the elbow extends) and has previously been used to describe dynamics of human motion, 33 cervical conditions, 34 knee rotations,35,36 and upper arm motion.37,38 Arm-plane direction was represented by the normal vector formed by the cross-product between arm segment direction vectors (Figure 1A). To capture plane direction with respect to the body coordinate system, we defined the arm-plane angle as the complement of the angle between the arm-plane normal and the vertical trunk direction, calculated in a shoulder-centered coordinate system. In this system, a greater complement angle represents a higher elbow elevation. Arm-plane angular velocity was determined by the change in arm-plane angle over time.
Movement quality was described by 5 spatial kinematic variables (distance, range), one temporal variable, and movement error. Distance variables characterized sagittal (forward motion positive) and lateral (rightward motion positive) trunk displacement (millimeters). Range variables characterized trunk, elbow, and arm-plane angular movement (degrees). Trunk rotation range was defined about the vertical axis (rotation—left rotation positive) as the Euclidean (angular) distance between onset and offset angles. For trunk with respect to arm-plane (onset) time, positive values denote that the trunk started moving before the arm-plane. Ranges of elbow extension and arm-plane angle motion and total (radial) endpoint error, that is, Euclidean distance between the final endpoint position and target, were also calculated. Although subjects were not specifically instructed to perform precise motion, total error indicated performance quality.
Statistical Analyses
Statistical analyses were done using SPSS (version 20.0.0, IBM, Armonk, NY, USA) with significance of P < .05. For regression analyses, P values of <.05 and >.1 were used for inclusion or rejection, respectively. Two-way mixed-design analyses of variance were done independently for each variable with target direction (3 levels: contralateral, central, and ipsilateral) as the within-subject factor and subject group (3 levels: control, mild stroke, moderate/severe stroke) as the between-subject factor. Relative effect sizes were quantified by partial η2 values. Simple correlations were done with 2-tailed Pearson tests if normality conditions were met. Linear regression analyses identified variables contributing to the total error for each direction and group (9 models) and included kinematic and clinical data. Multinomial logistic regression analysis assessed within-group differences between variables and was used to predict subject group assignment based on movements made to each target (3 analyses). Multinomial logistic regression is a multiple logistic regression analysis with more than 2 categories (in our case 3: control, mild, and moderate/severe). Thus, for each target direction, analysis was broken into a series of binary, pairwise comparisons. 39 For all analyses, backward rather than forward elimination was used whenever possible since this procedure more likely retains predictors that would have significant effects when other variables are kept constant (suppressor effects). 39 Forward elimination was used when backward analysis led to computational singularities. For linear regression analyses, both regression coefficients (bs) and normalized regression coefficients (βs) were computed. Regression coefficients were normalized with respect to the variable (factor) size to facilitate comparison between relative effects of different variables.
Results
Reaches made by subjects with stroke were qualitatively slower, less stereotypical, and less smooth (Figure 2) compared with controls. Average sagittal trunk displacement was less than 29 mm (Figure 3A, horizontal dotted lines) for all target directions in controls but was 3.3 to 5.9 times greater in the mild stroke group and 6.5 to 12.2 times greater in the moderate/severe stroke group. As previously reported, 40 subjects with moderate/severe stroke used substantially more trunk sagittal displacement (group: F2, 21 = 5.4, P = .013, Figure 3A) than all other groups and this was greatest for ipsilateral reaches (target: F2, 42 = 22.4, P < .001). For trunk lateral displacement, there was an interaction between group and target direction (F4, 42 = 7.4, P < .001; Figure 3B). While displacement was negligible and similar between targets for controls (2.0-11.1 mm), subjects with moderate/severe stroke used greater leftward trunk displacement (73.6 mm) for contralateral target reaches and rightward trunk displacement (94.3 mm) for ipsilateral target reaches (P < .001). The greatest amount of trunk rotation was used by all groups for reaches toward the contralateral target (F2, 42 = 83.7, P < .001, Figure 3C), and subjects with stroke used greater trunk rotation than controls for all targets (F2, 21 = 9.0, P = .001). Subjects with moderate/severe stroke used less elbow extension than all other subjects (group: F2, 21 = 7.3, P = .004, Figure 3D) and the least amount of elbow extension was used by all groups for ipsilateral reaches (target: F2, 42 = 25.3, P < .001). The arm-plane angle (Figure 3E) for all groups was lowest for ipsilateral reaches (F2, 42 = 10.4, P < .001) and highest for participants with moderate/severe stroke for all 3 target directions (F2, 21 = 3.4, P = .05). For all groups, average endpoint errors (Figure 3F) toward the central target were highest (F2, 42 = 54.3, P < .001).
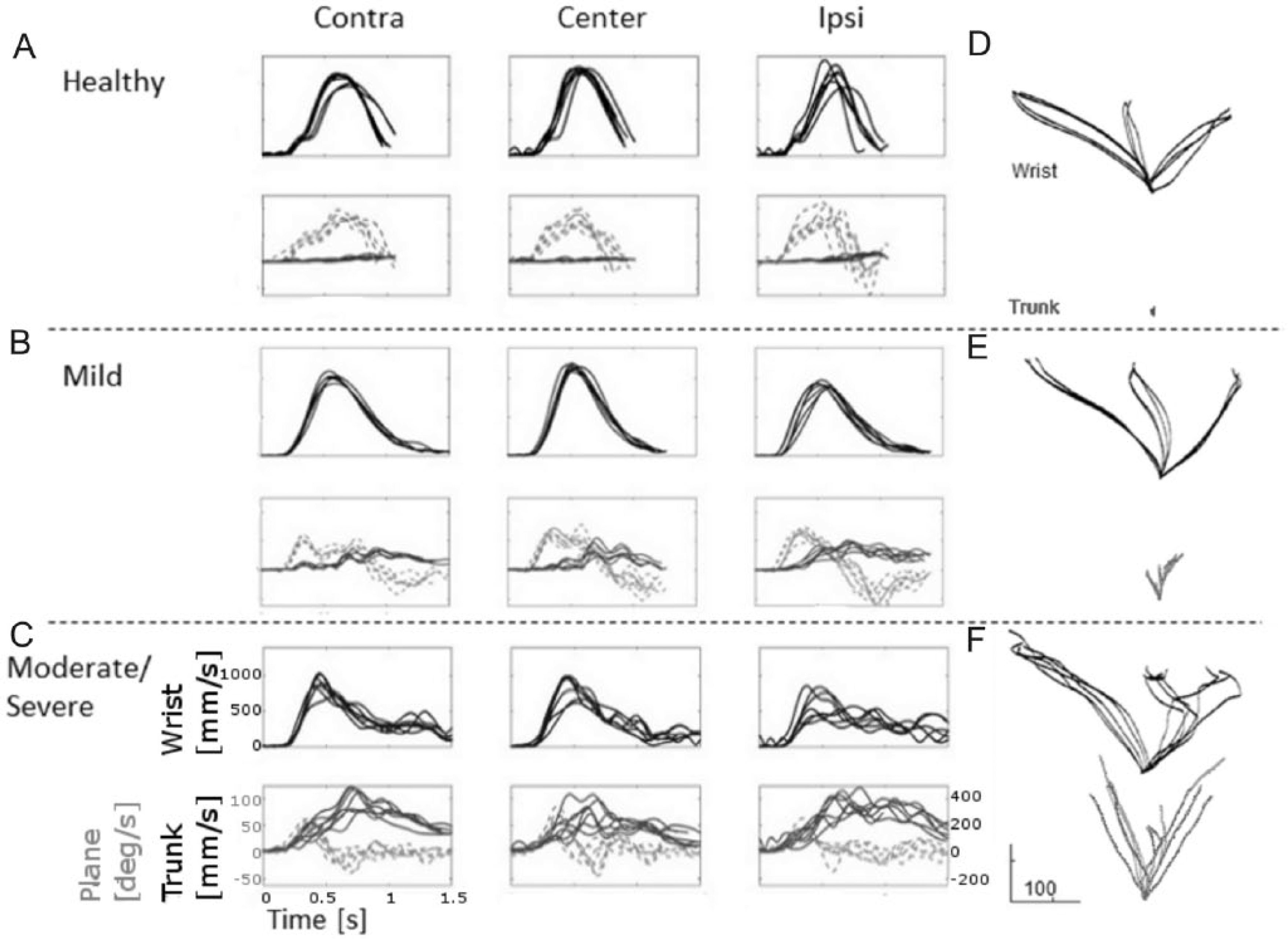
Examples of 3 movements to each target (Contra, contralateral; Center, central; Ipsi, ipsilateral) for one representative subject in each group (A, healthy control subject; B, subject with mild stroke; C, subject with moderate/severe stroke). First row in each panel shows wrist velocity profiles and second row shows trunk (solid lines) and arm-plane (dashed lines) movement superimposed. (D-F) Wrist (black lines) and trunk (gray lines) trajectories of reaches to 3 targets projected on the horizontal plane for representative control subject and subjects with mild and moderate/severe stroke, respectively.
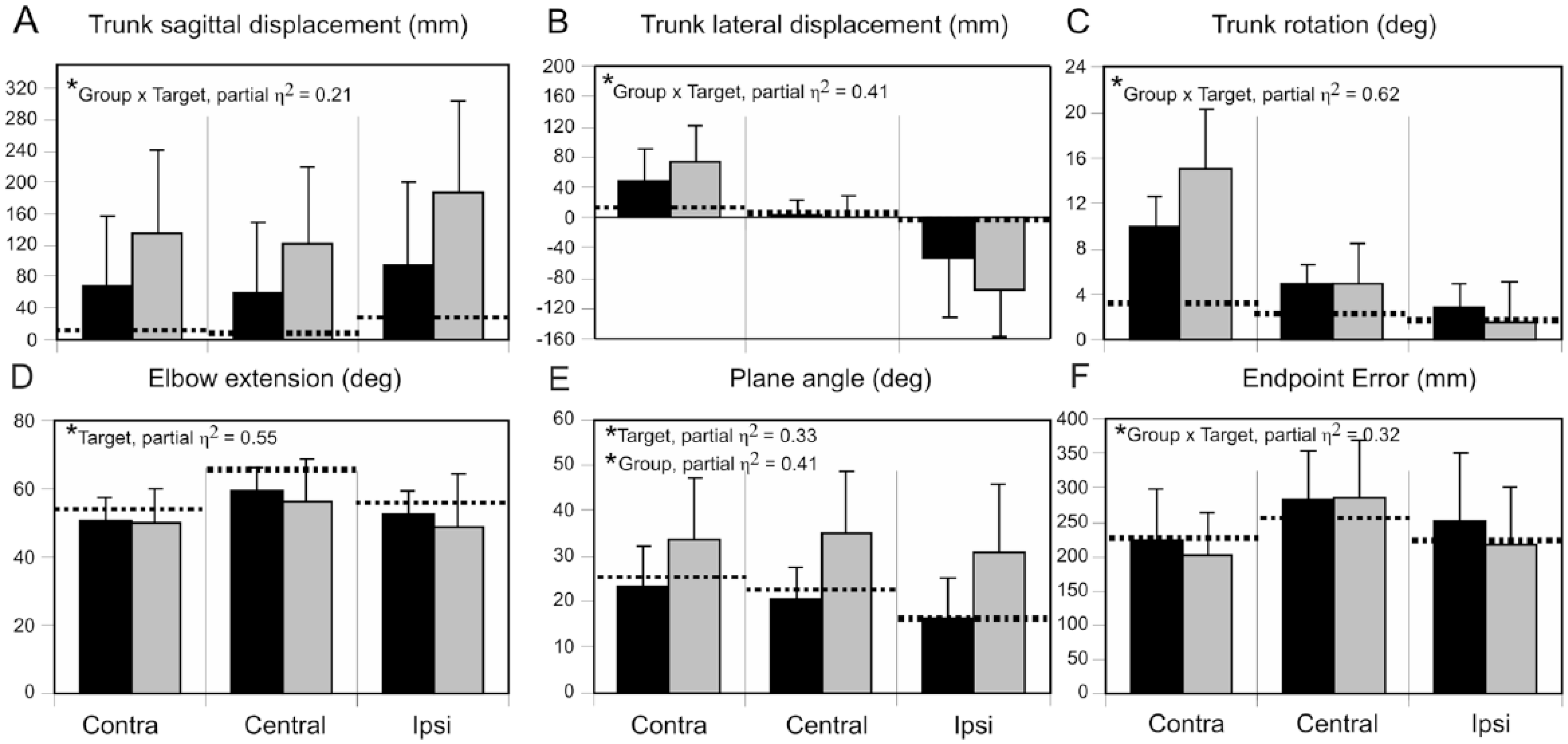
Histograms of group mean and standard deviation data for sagittal (A) and lateral (B) trunk displacement, trunk rotation angle (C), elbow extension angle (D), arm-plane angle (E), and endpoint error (F). Data for subjects with mild (black bars) and moderate/severe stroke (gray bars) are compared with means for the healthy control group (horizontal dashed lines). In each panel, data are presented for reaches to the contralateral (Contra), central, and ipsilateral (Ipsi) targets. Effect sizes (partial η2 values) are shown on upper left sides of each panel.
Temporal Coordination
Trunk movement started earlier than arm-plane movement in controls (47.2 ± 55.9 ms) while it occurred later or simultaneously with the arm-plane in mild (−32.3 ± 67.7 ms) and moderate/severe stroke groups (−82.2 ± 69.9 ms; F2, 21 = 11.8, P < .001). Onsets differed between controls and each stroke group (control–moderate/severe P < .001, control–mild P < .05).
Correlations With Time Since Stroke
For the mild stroke group, time since stroke negatively correlated with elbow extension range (ipsilateral r = −0.95, P < .002; central r = −0.95, P < .02; contralateral r = −0.94, P < .003). For the moderate/severe group, there was a positive correlation between time since stroke and arm-plane angle for the ipsilateral (r = 0.79, P < .02) and, marginally, for the central (r = 0.62, P = .07) target. However, for both groups, time since stroke was not correlated with FMA or endpoint error.
Factors Contributing to Endpoint Error
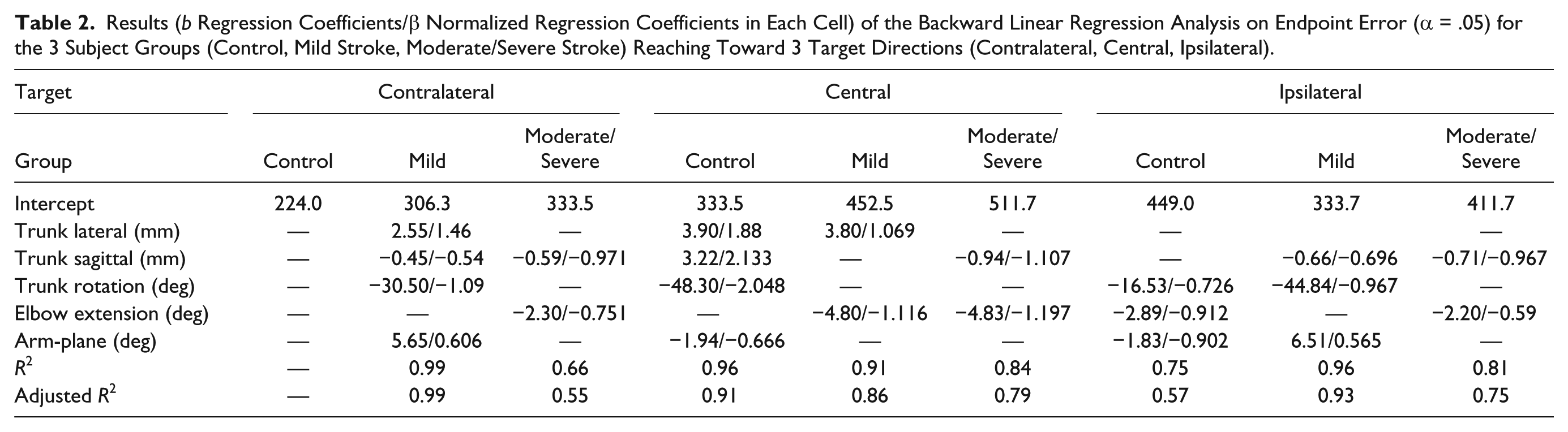
For controls, trunk, elbow, and arm-plane spatial variables partially explained endpoint error (adjusted R2 = 0.57) for reaches to ipsilateral but not contralateral targets (Table 2). For the Central target, all variables except for elbow extension range explained the error (adjusted R2 = 0.91). For the moderate/severe stroke group, the same 2 spatial variables (trunk sagittal distance and elbow extension range) explained the error to a moderate extent (adjusted R2 = 0.55-0.79) for all targets. For the mild group, the error was explained by different patterns of spatial variables for each target with relatively high adjusted R2 values (0.86-0.99). In this group, the arm-plane was one factor explaining contralateral and ipsilateral target errors.
Results (b Regression Coefficients/β Normalized Regression Coefficients in Each Cell) of the Backward Linear Regression Analysis on Endpoint Error (α = .05) for the 3 Subject Groups (Control, Mild Stroke, Moderate/Severe Stroke) Reaching Toward 3 Target Directions (Contralateral, Central, Ipsilateral).
Kinematic Classification of Individuals Into Groups Based on Severity
Trunk rotation and arm-plane angle predicted the subject group for each individual with an overall accuracy of 87.5% for contralateral and 79.2% for central targets, with high prediction rates for each group (Table 3). For the ipsilateral target, backward elimination led to computational singularities for the multinomial regression analysis, and thus, forward elimination was used. For this target, the model only included trunk lateral displacement with an overall prediction accuracy of 70.8%. The model did less well predicting group assignment for subjects with mild stroke, with only 3 out of 7 individuals classified correctly while 2 were incorrectly classified as control, and 2 were incorrectly classified as moderate/severe. Control and moderate/severe subjects were correctly classified with 87.5% and 77.8% accuracy, respectively.
Results of the Multinomial Regression Analysis for Group (α = .05) for Each Target Direction (Contralateral, Contra; Central, and Ipsilateral, Ipsi): Model Parameters (b) for Each Pairwise Group Comparison (Top) and Classification Tables (Bottom). a
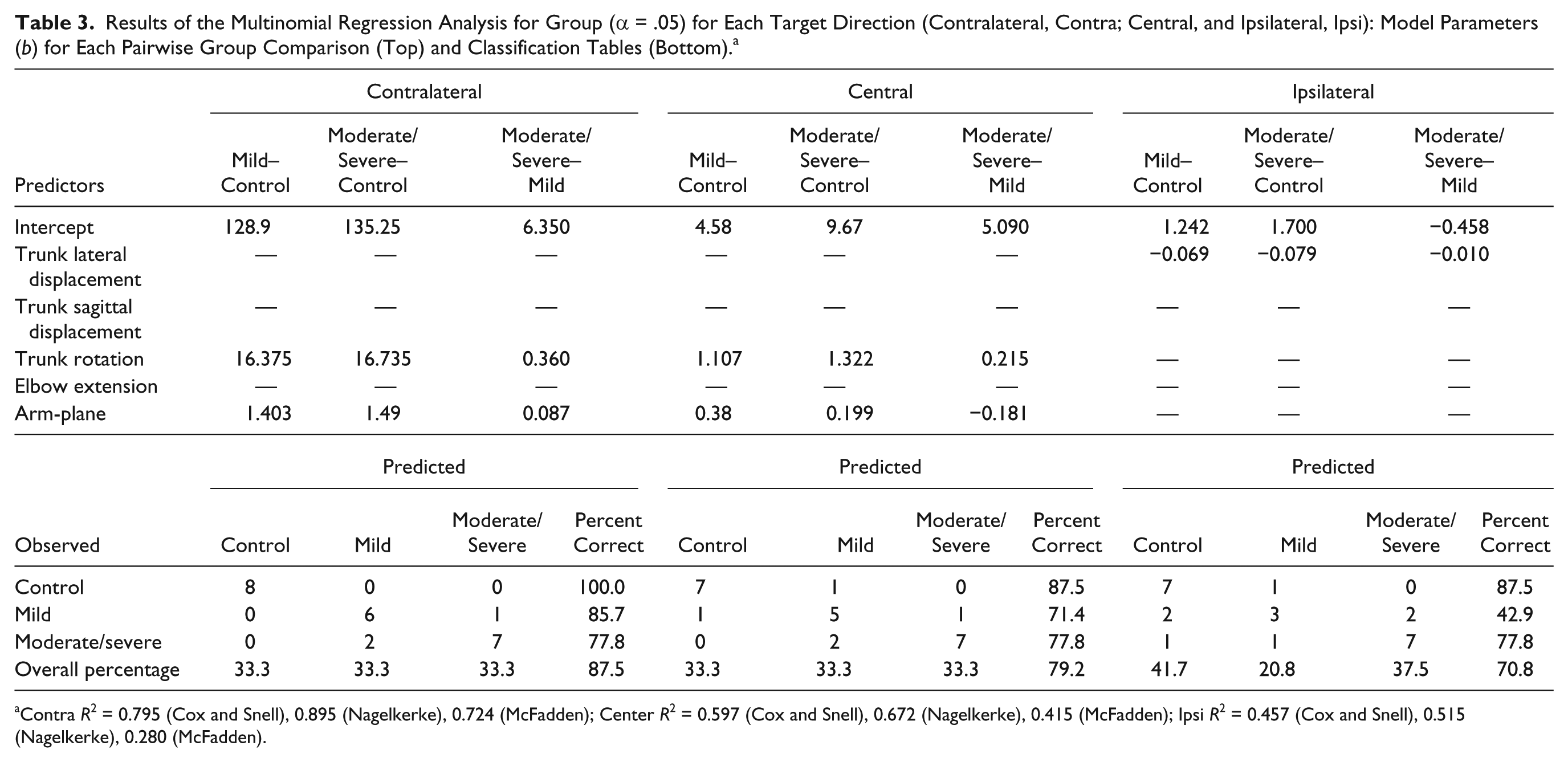
Contra R2 = 0.795 (Cox and Snell), 0.895 (Nagelkerke), 0.724 (McFadden); Center R2 = 0.597 (Cox and Snell), 0.672 (Nagelkerke), 0.415 (McFadden); Ipsi R2 = 0.457 (Cox and Snell), 0.515 (Nagelkerke), 0.280 (McFadden).
Discussion
Temporal and spatial characteristics of trunk movements and their relationship to arm-plane motion in individuals with chronic stroke of different levels of severity were compared with healthy controls. In addition to describing individual joint kinematics, arm motion was quantified based on rotation of the arm-plane relative to trunk motion. The advantage of the global arm-plane parameterization of shoulder movement is that it provides a means to evaluate arm and trunk covariance during reaching and may be used to describe the behavior of the pathological flexor synergy.
Pathological UL synergies used by patients with stroke when reaching into different parts of the workspace25,41 or performing a reach-to-grasp movement 16 have been well described. A synergy is considered pathological when the motion described by it is fixed or invariant. Hence, such synergies reduce the available number of DoFs, resulting in a greater reduction in the kinematic abundance of the system42,43 beyond that provided by task-specific movement synergies described in the healthy nervous system. For example, Shaikh et al 27 found that patients with moderate-to-severe stroke had deficits in adapting shoulder horizontal adduction/abduction and elbow extension/flexion coupling to preserve hand path invariance during a reaching task, but this ability was preserved in patients with mild stroke. Sukal et al 41 and Ellis et al 25 described a reduction in arm workspace with increasing shoulder abduction since the increasing abduction torque was coupled with greater elbow flexion in the pathological flexor synergy. Similarly, we found that patients with moderate-to-severe hemiparesis did not adapt UL joint rotations for reaching in 3 target directions. In contrast, mild stroke and healthy groups could use different combinations. This suggests that the UL synergy did not contribute to precise reaching movements in the moderate-to-severe stroke group. However, arm-plane movement could be used in a similar way to healthy controls in the mild stroke group. A distinction is made between the flexor synergy and the arm-plane. The arm-plane describes the covariance of movements of different DoFs of the shoulder. Since shoulder abduction is often a component of sagittal reaching in healthy subjects, arm-plane motion can be used to characterize this natural covariance. In addition, the arm-plane characterizes the shoulder motion component of the flexor synergy in patients with stroke.
Indeed, participants with stroke used different compensatory trunk movements for reaches in the 3 directions (eg, lateral and sagittal trunk displacement for ipsilateral target; lateral trunk displacement and rotation for contralateral target; sagittal trunk displacement and rotation for central target), as well as greater arm-plane rotation, all of which were more prominent in the moderate/severe stroke group. Previous studies identified only 2 elements of the UL flexor synergy (shoulder abduction, elbow flexion) affecting reaching 25 and reach-to-grasp 16 movement. Reisman and Scholz 26 studied a larger number of arm and trunk DoFs and described how the UL flexor synergy contributed to hand movement from the beginning to the end of the reach in a small group of subjects with mild-to-moderate stroke using principal component analysis (PCA). They concluded that patients with mild-to-moderate stroke retained the feature of a synergy that is related to error compensation.24,26 In other words, despite the presence of abnormal joint coupling, individuals with mild-to-moderate stroke could coordinate joint rotations to some extent during reaching to minimize undesirable movements of the hand path (hand path error compensation). Our results support this finding in a larger cohort of patients but suggest that the preservation of the adaptive features of a synergy, as defined by Latash et al, 42 are only present in patients with mild hemiparesis.
Trunk Compensations for Deficits in Arm Reaching Extent
All stroke subjects used trunk movement adaptively to compensate for limitations in arm reaching extent. Our previous study showed that patients with stroke used less shoulder horizontal adduction for reaches toward the central and ipsilateral targets. 38 While control and mild stroke groups had similar final plane-angles, the moderate/severe group had higher plane-angles for all target directions. Higher elbow positioning may have been used to adduct the endpoint across the body (contralateral direction). However, this adaptive behavior fell short of bringing the hand close to the target, necessitating additional compensatory trunk involvement, that is, increasing trunk roll. The same strategy was used by this group for reaches to the central target.
Stroke subjects who were unable to fully extend the elbow also used excessive trunk forward displacement to bring their hand closer to all 3 targets. This compensation has been described previously for sagittally located targets with the degree of trunk forward displacement varying with the severity of the motor impairment.5,12 Increasing trunk pitch lowers hand height. Subjects with mild and moderate-to-severe stroke used the arm differently to overcome limitations in elbow extension and hand height. Those with mild stroke used arm-plane movement adaptively to partially compensate hand lowering by elevating the arm into abduction as reflected by higher arm-plane angles for reaches to the ipsilateral and contralateral targets. However, those with moderate-to-severe hemiparesis used the same strategy for all 3 targets, demonstrating a lack of adaptability of the arm for assisting forward hand displacement. Furthermore, Ellis et al 25 suggested that pathological shoulder abduction and elbow flexion coupling limits forward reaching distance resulting in the incorporation of compensatory trunk movements as the hand reaches forward. However, our results and those of previous studies 6 have shown that the trunk and arm-plane recruitment begins before or simultaneously with hand movement. This suggests that trunk recruitment is an integral part of the reaching synergy and is incorporated into the reaching synergy in a feedforward manner rather than in response to a limitation in arm displacement.
Arm-Plane Versus Principal Component Analysis and Uncontrolled Manifold Approaches
Characterization of arm-plane motion, recorded with a minimal marker set, enabled us to identify temporal and spatial linkages between whole arm and trunk movement in control and stroke subjects reaching in three target directions. The analysis provided adequate information about the UL movement synergy based on recording data from only 3 markers instead of a much more elaborate data collection and analysis process required to capture movements of seven DoFs of the arm. This minimal approach is deterministic rather than stochastic, whereby system mechanics are known and related. Thus, we describe 3-dimensional arm motion in terms of whole arm rotation as if the nervous system was concerned with the combined effect of changing arm posture, and not with the effect of individual joint rotations. PCA, on the other hand, uses dependencies between variables for projecting high-dimensional data into a lower-dimensional form while controlling for the amount of information loss. Principal components reflect patterns of data covariance that may have many different causes. Thus, care should be taken in attaching meaning to them. 44 Like PCA, the uncontrolled manifold approach is also based on statistical features arising from joint rotation variance, 45 except that assumptions are made a priori and later tested about the directions in which the variance is maximized. Thus, this approach has also been criticized as being somewhat biased. 46 Rather than using statistical methods, we opted to reduce data dimensionality using the arm-plane angle, which is a variable with a known meaning. The plane angle includes all three shoulder DoFs (rotation, flexion-extension, adduction-abduction) involved in reaching, and therefore, more directly represents a functional movement parameter.
Clinical Implications
The amount of shoulder abduction alone has previously been identified as a marker of post-stroke recovery. For example, less shoulder abduction during reaching predicted motor function outcome on the Wolf Motor Function Test. 47 In our study, amounts of arm-plane movement combined with trunk rotation when pointing to central and contralateral targets discriminated between different subject groups and between patients with different impairment severity. A recent review concluded that impairment severity is the primary determinant of loss of UL function at nearly all time points after stroke. 48 Our results suggest that 2 kinematic features, the amount of arm-plane movement and compensatory trunk displacement, are sufficient to identify patient-specific motor deficits to be targeted in impairment-based rehabilitation. Furthermore, these 2 kinematic features can be easily observed and quantified in clinical settings without sophisticated motion-analysis equipment. Kinematic features can be quantified numerically using the Reaching Performance Scale for Stroke, 49 a clinical scale that allows clinicians to identify shoulder and trunk compensations during reach-to-grasp movements. In addition, to verify whether a movement pattern is compensatory, a clinician could restrict the trunk movement and observe whether the patient is able to extend the arm further.
We also found in our cross-sectional correlational analysis that the greater the chronicity in the mild stroke group, the less the individuals used elbow extension for reaching. In addition, individuals with more chronic moderate/severe stroke used higher arm-plane angles. These results imply that motor compensations may increase with time since stroke but confirmation would have to be obtained from longitudinal studies.
Differences in the use of motor compensations may be related to lesion location and volume. We did not find any relationships between kinematic features of movement and lesion location. A limitation of this study is that we did not have access to lesion volume data to analyze this potential relationship.
Conclusions
Our results suggest that the damaged poststroke system seeks to use the most effective movement strategies available to it, characterized by excessive shoulder movement to raise hand height and compensatory trunk displacement to advance the hand. This shoulder strategy is most effective in patients with mild UL motor impairment who preserve some degree of motor adaptability. However, arm-plane movement is less adaptable in patients with more severe impairment and engenders the use of more compensatory movement strategies. The advantage of using adaptive synergies is that functional tasks can still be accomplished despite limitations in independent joint movements. The disadvantage is that once established, adaptive strategies may be difficult to break down in attempts to recover fractionated movement patterns and may be considered as learned bad-use. 50 Using clinical observation of 2 movement components, arm-plane and trunk movement, clinicians can identify and quantify compensatory motor strategies, which may be targeted in rehabilitation to improve UL motor recovery.
Footnotes
Acknowledgements
The authors wish to thank Dr Valeri Goussev from the Centre for Interdisciplinary Research in Rehabilitation of Greater Montreal. The authors also thank Prof Patrice (Tamar) Weiss and Dr Harold Weingarden from the Sheba Medical Center, Israel, for their assistance in Helsinki Ethics Approval and for facilitating the data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MFL holds a Tier 1 Canada Research Chair in Motor Recovery and Rehabilitation. Supported by REPAR-FRSQ (Quebec) and the Israel-France Research Networks Program in Neuroscience and Robotics (SB, DGL).
