Abstract
Introduction
Meaningful recovery of arm and hand function after stroke is a challenge for stroke survivors, as well as therapists.1-3 Over the past decade, our understanding of requirements for recovery 4 and the number of interventions designed to promote recovery 5 have increased. Nonetheless, many stroke survivors continue to have severe upper limb disability early after stroke, which persists in the long term. 1 While intensive and repetitive task-oriented practice is considered important for functional recovery, 5 stroke survivors with little or no underlying movement have difficulty engaging in such practice. 6 Consequently, training interventions specifically designed to enable stroke survivors with severe paresis to undertake task-oriented practice have emerged.7,8 SMART Arm training is one such intervention. 9
The SMART Arm consists of a mechanical device interfaced with an interactive computer-training program that is combined with outcome-triggered electrical stimulation (OT-stim). It is designed to enable a stroke survivor with little or no muscle activity to reach in a straight-line path (Figure 1). The key mechanical component is a “track” to which the hand is attached. Initially, the track makes reaching easier by minimizing resistance to movement and reducing mechanical degrees of freedom to be controlled. As the capability of the stroke survivor progresses, demands are increased through incremental additions of load, track inclination, elevation and orientation, or relaxation of constraints (eg, release of hand from splint to enable grasping). To enhance motor learning, the interactive computer-training program provides instruction, visual and auditory feedback, and progressive modification of training parameters (eg, tapering of feedback). OT-stim is used to make reaching possible when a stroke survivor presents initially with negligible voluntary muscle activity. As the stroke survivor attempts to reach, and achieves the designated “outcome” (eg, their initial maximum active reach distance), electrical stimulation (ES) is triggered to augment reaching through full range. As capacity for independent movement increases, the “outcome” (ie, distance reached) required to trigger the assisting ES is incrementally increased.
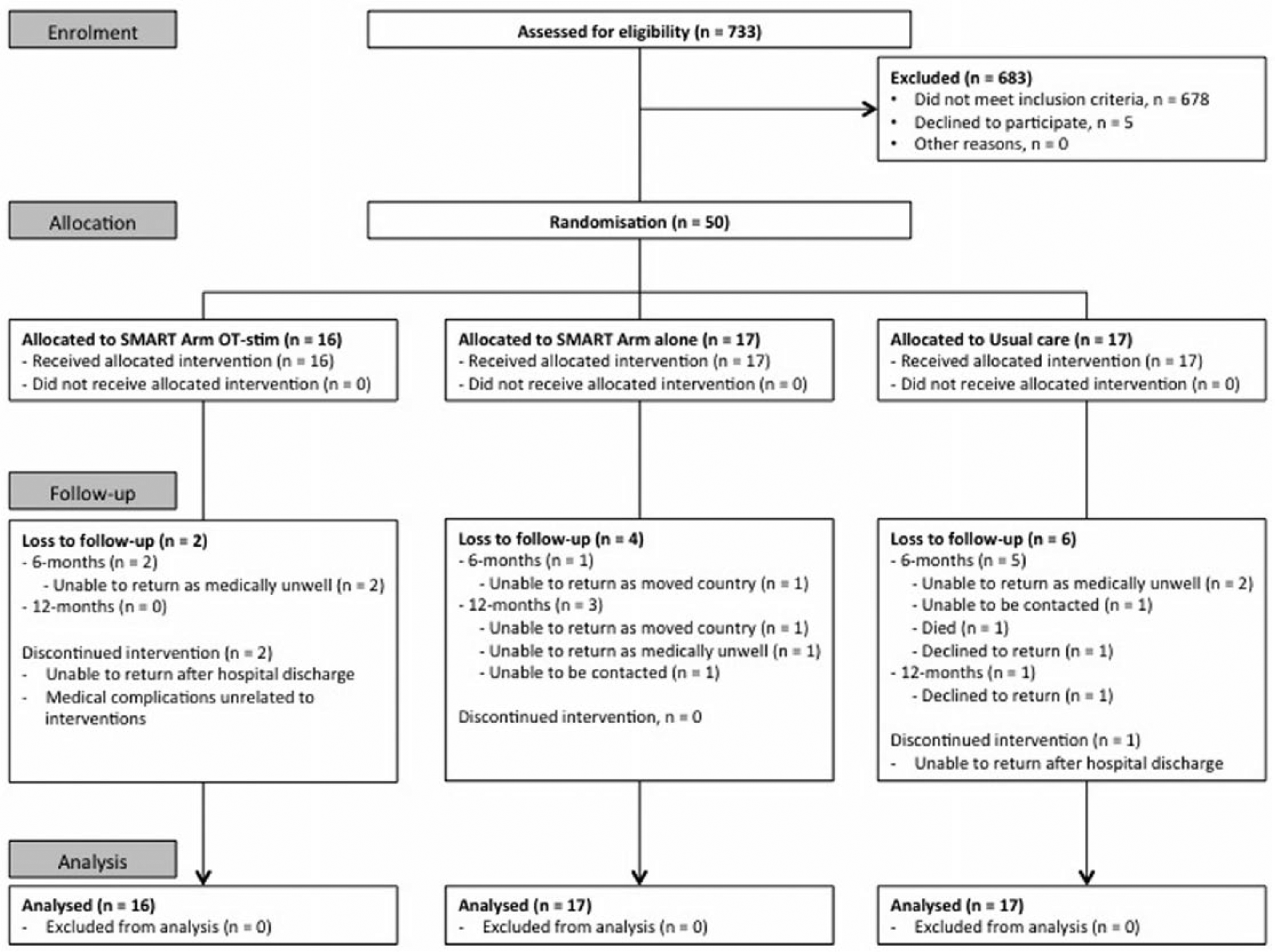
Participant flowchart.
The effectiveness of SMART Arm training has been demonstrated in 2 studies: a randomized controlled trial (RCT) in the community 9 and a pilot RCT in an inpatient setting. 10 Accordingly, changes in muscle activation patterns 11 and increases in corticospinal reactivity, 12 that have accompanied increases in capacity to perform reaching movements, have exhibited some of the characteristics of “true” recovery. 13 The feasibility of SMART Arm training in the home with minimal supervision has also been demonstrated in a single case study. 14
The ultimate challenge for stroke survivors with severe upper limb paresis is to bridge the gap between no movement, and use of the arm in everyday tasks. To maximize the likelihood of this occurring, rehabilitation is typically offered in the first 3 to 6 months following stroke, when the brain is considered most responsive to rehabilitation15-17 and when there are fewer secondary impairments, such as contractures 18 or pain. 19 Ideally, the stroke survivor also has the opportunity to use their arm in everyday tasks relevant to their goals and lifestyle. 20 Consistent with this rationale, the purpose of the present trial was to determine the efficacy of SMART Arm training early after stroke when used in combination with usual therapy. Our primary hypothesis was that SMART Arm training with or without OT-stim, combined with usual therapy—when compared with usual therapy alone—would lead to greater improvement in arm function in stroke survivors with severe upper limb disability undergoing inpatient rehabilitation. Our secondary hypothesis was that those who receive SMART Arm training with usual therapy—compared with those who received usual therapy alone, would achieve greater improvement in arm and hand function, fewer adaptive changes, and greater use of the arm in everyday tasks after training and at 6- and 12-month follow-up.
Methods
Design
A prospective, multicenter, assessor-blinded, 3-group, parallel-randomized controlled trial was undertaken: recruitment February 2010 to December 2012; follow-ups until December 2013. Princess Alexandra Hospital Ethics Committee (Clearance ID: 2008-046) and The University of Queensland Medical Research Ethics Committee (Clearance ID: 2007001628) approved the study protocol, which was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants or their legal guardian. The protocol for this study was registered with the Australian and New Zealand Clinical Trials Registry (ACTRN12608000457347). Detailed description of study methods can be found in the published study protocol. 21
Participants
Consecutively admitted patients at trial sites were screened for eligibility by an independent research assistant. Trial sites included the Acute Stroke Unit or Geriatric and Rehabilitation Unit at Princess Alexandra Hospital and Queen Elizabeth II Jubilee Hospital in Brisbane, Australia. Patients were included if they were adult stroke survivors (≥18 years) with a primary diagnosis of first-ever unilateral stroke less than 4 months prior confirmed radiologically (computed tomography or magnetic resonance imaging) or clinically by the consulting physician; had severe upper limb disability demonstrated by a score of 3/6 or less on the Motor Assessment Scale (MAS) item 6 (upper arm function) 22 ; and able to follow single stage commands. Patients were excluded if deemed medically unstable by a medical officer; had upper limb comorbidities that could limit function (eg, arthritis); inability to tolerate (eg, hypersensitivity) or contraindication to (eg, pacemaker) cutaneous ES.
Randomization Protocol
Physiotherapists and occupational therapists identified potential participants and notified the trial screener. After consent and initial assessment, participants were randomly allocated to 1 of 3 dose-matched groups (2 intervention, 1 control), using random permuted blocks of 4 or 6 in a 1:1 ratio. The allocation sequence was prepared using a computer-generated random number sequence by an offsite investigator, who was not involved in recruitment, intervention or data collection. A set of consecutively numbered, sealed, opaque envelopes containing allocations was centrally generated for each hospital.
Intervention
Participants were randomly allocated to 60 min/day, 5 days a week for 4 weeks of (1) SMART Arm with OT-stim and usual therapy, (2) SMART Arm alone and usual therapy, or (3) usual therapy. Volume of training was based on “usual” time reportedly spent on upper limb therapy at each site. SMART Arm training was delivered in 1 session by 1 of 5 physiotherapists or occupational therapists, trained in operation of the SMART Arm. “Usual therapy” was delivered and recorded in logbooks in a second session by hospital physiotherapy and occupational therapy staff. If a SMART Arm or usual therapy session (eg, due to illness) was missed, additional days were added to ensure participants had the opportunity to engage in 20 days of therapy.
The 2 SMART Arm groups participated in SMART Arm training according to the established training protocol. 10 Training sessions began with the participant seated in a chair beside the SMART Arm, restrained by a harness to restrict compensatory trunk movement. The affected arm was positioned in 90° of elbow flexion, forearm mid pronation-supination, and wrist mid extension-flexion in the functional position. The hand was placed in a splint connected to a manipulandum that was mounted on a linear slide that served to constrain movement to one plane and reduce resistance to movement. The slide was instrumented to measure displacement, for the purpose of providing continuous visual feedback to the participant. When prompted by an audible tone, the participant attempted to push their hand along the linear track to reach a preset goal. This goal was a horizontal target line superimposed on the visual display at a position that corresponded with the participant’s maximum passive reaching distance established at session commencement. Participants were asked to perform a consistent minimum number of repetitions: 60 per day for week 1 and 80 per day for weeks 2 through 4. This dose was established in previous SMART Arm trials.9,10 At the discretion of SMART Arm trainers, progression of training parameters occurred once consistent performance of reaching was achieved. Progressions recorded included repetitions, track inclination, degree of shoulder external rotation, hand position, load, visual and auditory instruction, and feedback, (therapist) instruction and supervision. To ensure intervention fidelity and consistency between trainers, 1 researcher (KSH) monitored participant training logbooks for dose and progression of training and accordingly, mentored trainers.
Participants allocated to SMART Arm–stim received stimulation to lateral head of triceps via an Empi 300PV unit (St Paul, MN, USA) to augment full-range reaching. Stimulation parameters consisted of a 1-second ascending ramp of 200-μs pulse width biphasic stimulation at 35 Hz applied at a constant level. Stimulation was initiated once the participant reached their previously recorded “personal best” displacement and ceased once they achieved the goal or after 8 seconds, a time period considered adequate to complete the task. Stimulation intensity was the maximum tolerated by the participant. When required, participants receiving SMART Arm alone were assisted, with therapist hands-on support, to augment full range reaching consistent with usual therapy.
Usual therapy, which has been previously reported, 10 included physiotherapy, occupational therapy, and therapy assistant time, and involved a mix of one-on-one and group therapy sessions. Content of sessions was not standardized and included passive (eg, stretching, cyclic ES), active (eg, strengthening, modified task-oriented practice) and activities of daily living (eg, dressing) interventions.
Outcome Measures
All participants were assessed at 4 time points: within 3 days prior to intervention commencement (baseline, 0 weeks) and after intervention completion (4 weeks), and within 2 weeks of 26- and 52-week follow-up. The primary outcome was postintervention MAS item 6 (MAS6), upper arm function, measured on a scale from 0 to 6 with a higher score equating to better function. This measure has established reliability and validity within a stroke population 22 and sensitive to change in people with severe upper limb disability undertaking rehabilitation, 23 and after SMART Arm training.9,10,14
Secondary clinical outcome measures included impairment, activity, and participation measures. Impairment measures included strength of lateral head of triceps brachii using Medical Research Council (MRC) manual muscle testing with ratings from 0 to 5; resistance to passive elbow extension using the modified Ashworth Scale 24 ; and joint tenderness on passive movement of the hemiplegic shoulder using Ritchie Articular Index (RAI). 25 Arm activity was measured using MAS items 6, 7, and 8 total score and Rasch Partial Credit Model. 26 Participation measures consisted of 2 self-report measures: Stroke Impact Scale (SIS) 27 and Motor Activity Log-28 (MAL). 28 Secondary laboratory measures, included kinematics and kinetics of reaching and are to be reported separately.
Demographic information collated from medical records included age, stroke onset date, stroke type (ischemic or hemorrhagic) and stroke medical intervention (eg, thrombolysis). Clinical measures to quantify presence of multiple impairments at baseline included the Cognitive Linguistic Quick Test 29 ; upper limb sensation using the Nottingham Sensory Assessment 30 ; and motor function using the MAL-defined range of motion measure, 28 which included voluntary finger extension as a marker of a recovery potential. 31 The SAFE Score was calculated retrospectively from the MAL-defined range of motion measure.
Sample Size
A priori power calculation was undertaken to determine sample size. Based on previous findings,9,10 mean improvement in MAS6 posttraining of 1.8 (SD 2) in the usual therapy group, 2.91 (SD 2) in SMART Arm alone group, and 3.91 (SD 2) in SMART Arm with OT-stim group were estimated. To achieve 80% power, significance of 0.05 with pairwise comparisons and allowing for a 15% dropout rate, 25 subjects were required per group, totaling 75.
Analysis
Statistical analysis was performed using the intention-to-treat principle with a 2-tailed α-level of .05 (IBM SPSS v22.0, IBM Corp, Armonk, NY, USA). Descriptive statistics were used to ensure comparability between groups at baseline, and to test assumptions concerning use of parametric statistics. To determine differences in outcome between groups, generalized linear mixed effects models were used for interval data and mixed effects models for continuous data. Group, time, and interaction between group and time were used as fixed factors and participants as a random factor. For the primary outcome measure, comparison was made between groups for change in MAS6 scores between baseline (0 weeks) and posttraining (4 weeks) and 95% confidence intervals for mean differences for pairwise comparisons were calculated. A 1-point (15%) change was adopted as a clinically important difference.32,33 Post hoc within-group
Results
A total of 733 individuals with stroke were admitted and assessed for eligibility with 50 stroke survivors with severe arm paresis (MAS6 < 4) consenting to participate. Recruitment ceased prior to reaching the estimated sample size of 75 as system resources could not support the study beyond the 3-year recruitment period. Random allocation resulted in 16 participants in the SMART Arm OT-stim group, 17 in the SMART Arm alone group, and 17 the usual therapy group. Attrition rate was 6% after training, 22% at 26 weeks and 30% at 52 weeks. Flow of participants is presented in Figure 1. At baseline there was heterogeneity across the sample, nonetheless demographic and clinical characteristics of each group were similar (Table 1) with evidence of severe and multiple impairments of motor, sensory and cognitive-linguistic function across all 3 groups. According to a SAFE score of <4, all participants had severe upper limb impairment and most participants (78%) lacked voluntary finger extension with no difference between groups.
Participant Characteristics at Baseline.
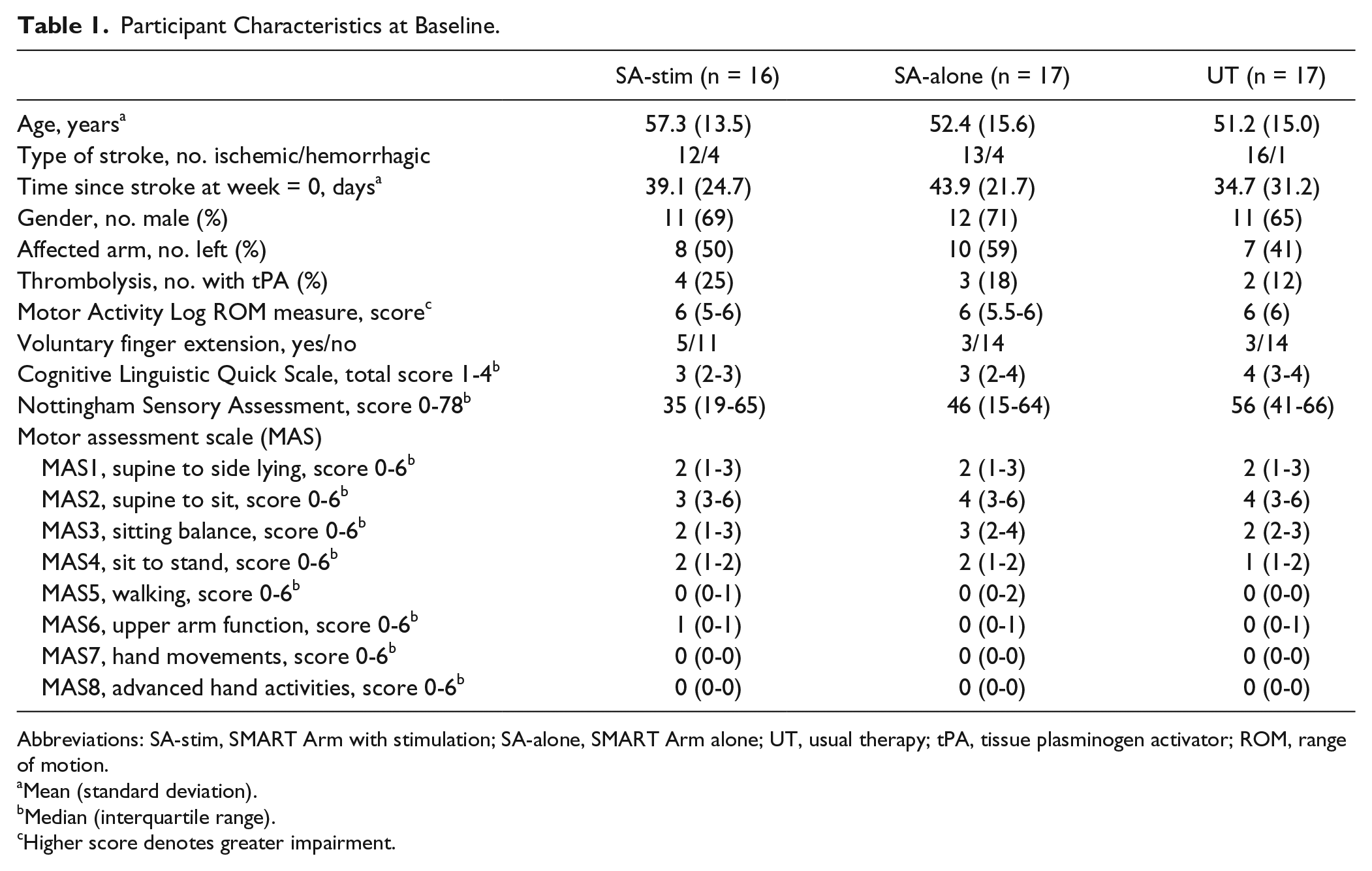
Abbreviations: SA-stim, SMART Arm with stimulation; SA-alone, SMART Arm alone; UT, usual therapy; tPA, tissue plasminogen activator; ROM, range of motion.
Mean (standard deviation).
Median (interquartile range).
Higher score denotes greater impairment.
Compliance with training was high, with 90% of participants completing 85% or more training sessions, with no differences between groups. On average, usual therapy consisted of 46 minutes of individual and 11 minutes of group therapy per participant, per day with a higher dose (29 vs 17 minutes,
Improvement in arm function (MAS6) was not significantly different between groups immediately posttraining, or at 26- or 52-week follow-up (time × group) (
Median (Interquartile Range) or Mean (SD) at Each Time Point by Group.
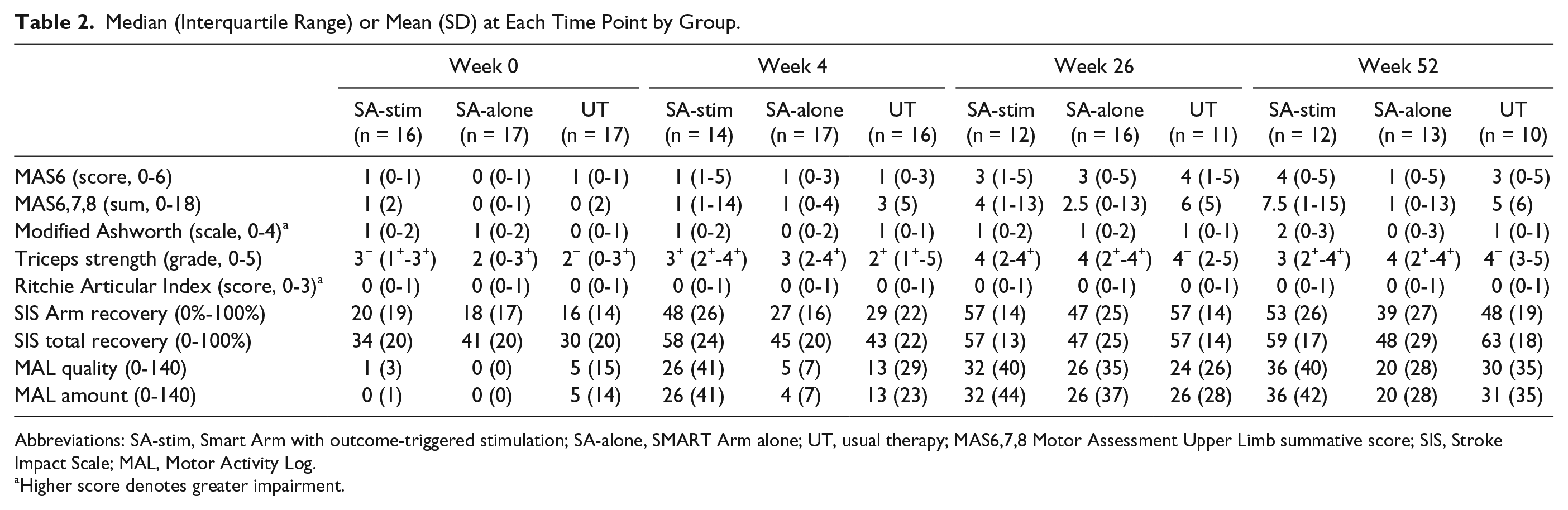
Abbreviations: SA-stim, Smart Arm with outcome-triggered stimulation; SA-alone, SMART Arm alone; UT, usual therapy; MAS6,7,8 Motor Assessment Upper Limb summative score; SIS, Stroke Impact Scale; MAL, Motor Activity Log.
Higher score denotes greater impairment.
Within-Group Differences (Means and SD) and Between-Group Differences (Mean Difference and 95% Confidence Intervals).
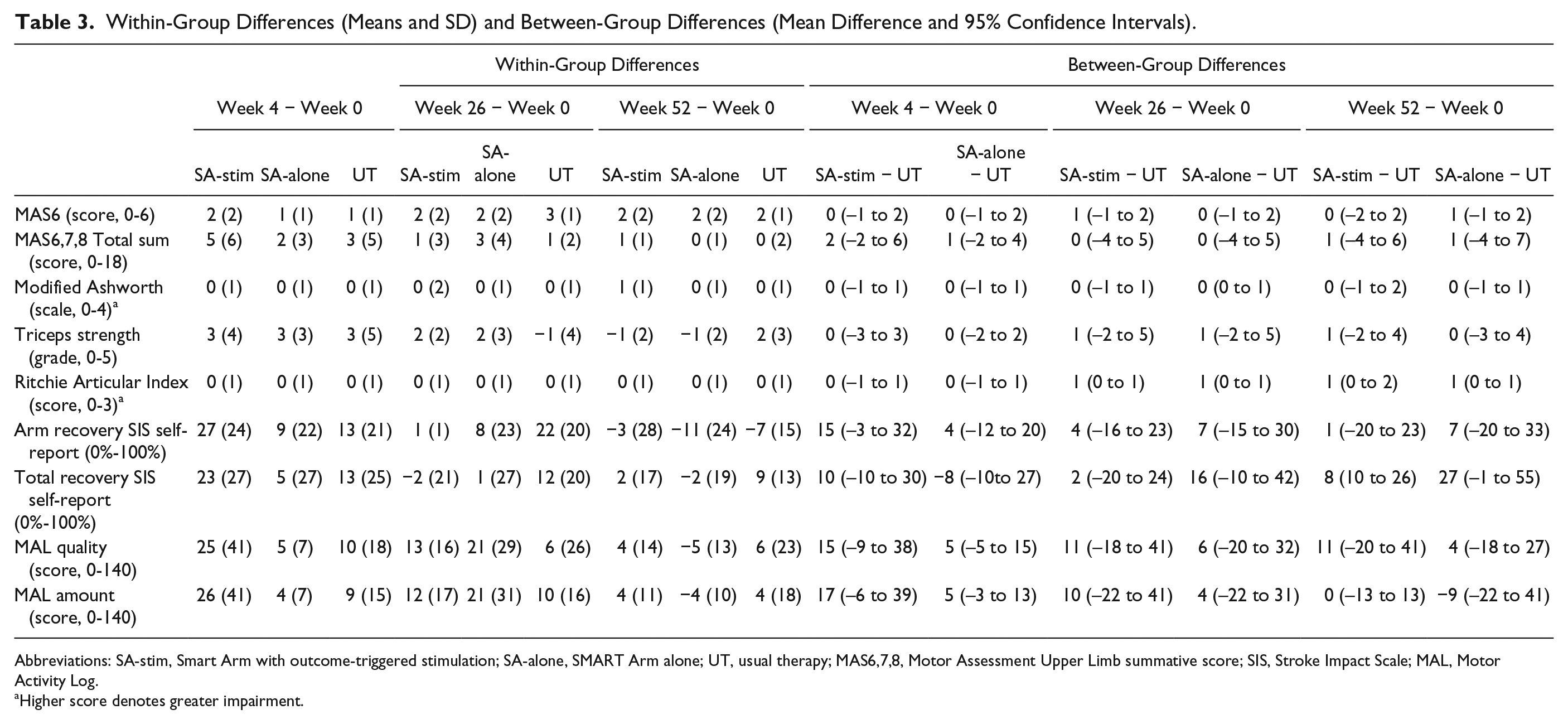
Abbreviations: SA-stim, Smart Arm with outcome-triggered stimulation; SA-alone, SMART Arm alone; UT, usual therapy; MAS6,7,8, Motor Assessment Upper Limb summative score; SIS, Stroke Impact Scale; MAL, Motor Activity Log.
Higher score denotes greater impairment.
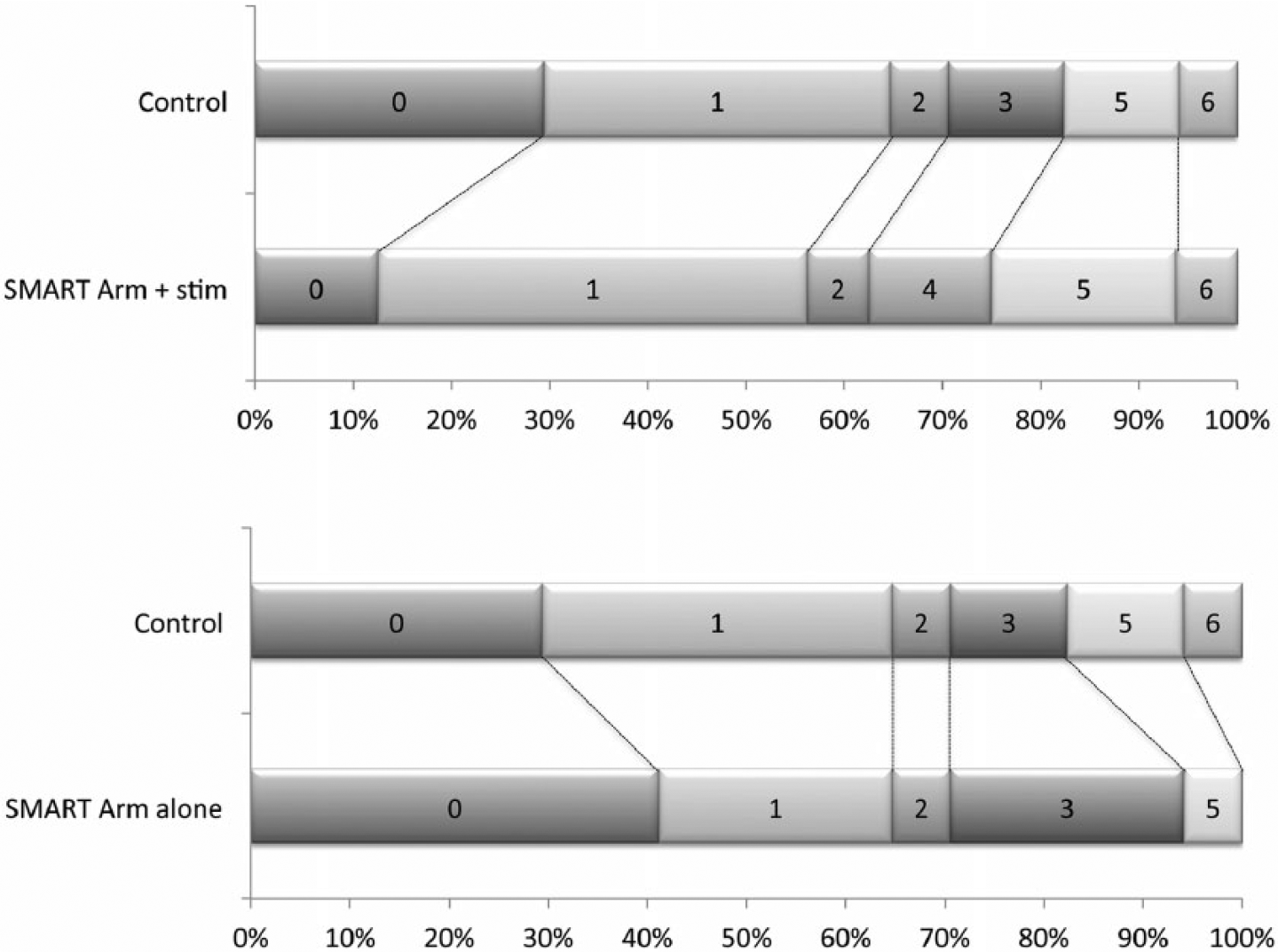
Comparison between control and intervention groups on MAS Item 6 score at post-training.
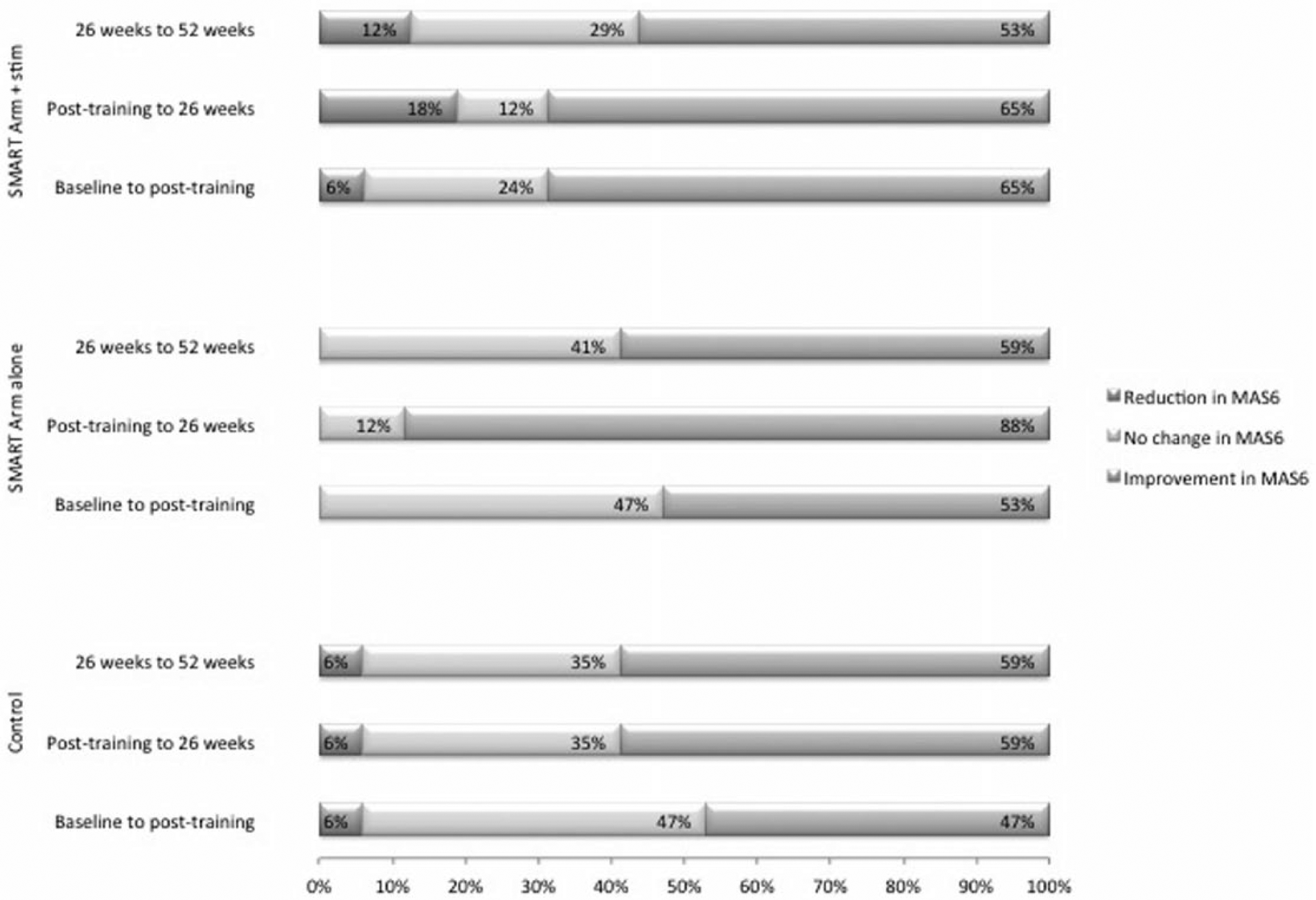
Proportion of participants who achieved an improvement, no change or reduction in scores on the Motor Assessment Scale item 6 from baseline to posttraining, from posttraining to 26 weeks, and from 26 to 52 weeks.
Improvement in arm function within all 3 intervention groups over time was statistically
Secondary outcome measures (Tables 2 and 3) were not significantly different between intervention groups. All 3 groups showed a statistically and clinically significant improvement over time for triceps muscle strength (
Discussion
This study assessed with respect to usual therapy alone, the effect on arm function of SMART Arm training, when used with and without OT-stim in combination with usual therapy, in stroke survivors with severe upper limb disability participating in inpatient rehabilitation. All groups exhibited higher levels of function following the training period, yet contrary to our hypothesis there were no differences in the degree of change between groups.
The RCT design that required random allocation of participants to groups may have been suboptimal for the individual. For example, OT-stim group participants trained with ES for the 4 full weeks. During this time, some participants were able to reach without requiring stimulation. Unnecessary stimulation could have reduced the challenge for participants, and level of engagement and motivation required to progress optimally. 36 Similarly, SMART Arm alone group participants with negligible movement may have taken longer to learn to reach than if they had first used ES to augment reaching. Observed patterns of improvement lend support to this proposition. Odds of SMART Arm OT-stim group participants making a moderate to large improvement were greatest post-training, but not sustained when posttraining practice occurred without ES augmentation, revealed at 6-month follow-up. In contrast, those who trained without ES were more likely to make a moderate to large improvement at 6-months posttraining than immediately after training. A plausible explanation is that SMART Arm alone group participants were forced to “solve the movement problem” 37 to reach successfully during SMART Arm training, and could subsequently apply this same strategy to practice of everyday tasks after training. 38 The salient point is that even though the SMART Arm interactive training program is designed to provide for each stroke survivor a level of challenge that is tailored to their current capability, 39 the RCT training protocol possibly limited the degree to which this objective was achieved. Furthermore, without a clear contrast in training between the two SMART Arm groups, it is possible that benefits of training were determined more by the extent to which the intervention matched participants’ “challenge point,” rather than by their allocated intervention.
Our previous studies demonstrated benefits of SMART Arm training,9,10,14 yet the present study showed no additional benefit over and above usual therapy. While the estimated sample size was not achieved due to recruitment delays, insufficient power (type II error) is an unlikely explanation, as there was no apparent trend toward group differences. To determine how to gain the “best effect,” training characteristics that optimize exploitation of recovery potential, and individual characteristics of stroke survivors that determine the likely degree of response, need to be considered.
In this current study, 20 hours of training over 4 weeks for stroke survivors with severe paresis was almost certainly too little practice, over too short a time period, to shift the recovery profile, above the spontaneous recovery profile.13,40-42 There is increasing evidence that the greatest opportunity for motor recovery after stroke occurs during the unique “sensitive period,” in which time-enhanced spontaneous as well as intervention-mediated plasticity exists. 13 As the “sensitive period” is presumed to begin within 2 weeks after stroke and to continue for approximately 3 months,40,42,43 greater spontaneous recovery may have occurred for participants who were earlier poststroke. For the full potential benefit of augmented recovery to be realized, training would have needed to commence within 2 weeks rather than on average, 39 days (SD 26) poststroke and continued for up to 3 months, rather. Additionally, although comparable across the 3 intervention groups, variability observed in time poststroke could indicate that a larger sample size was required to demonstrate a significant difference between groups. 31 Future research would need to control for time poststroke to mitigate the potential influence of this factor on study findings.
With regard to training intensity, optimal dose is unknown.44,45 A recent systematic review suggests that a dose at least triple of what is “usually provided” is required to demonstrate a therapeutic benefit. 41 In practical terms, the challenge is for stroke survivors to commence intensive practice within an acute setting, where many diagnostic and therapeutic interventions compete for their time, and to continue practice during the time in which inpatient and outpatient rehabilitation is typically available.
Usual therapy (received by all participants) was of a high volume in this current study compared to previous studies. 35 No consistent effort was made, however, to relate goals of usual therapy to SMART Arm training, limiting potential for carryover for those who received both. 46 In contrast, in a previous study when SMART Arm training occurred in the home environment, 14 the participant could directly link SMART Arm training with purposeful real world use 47 and make highly meaningful gains for the effort. 4 To tease out active ingredients 45 that may be critical when SMART Arm training is used in combination with usual therapy, a secondary analysis of training data is currently underway.
Participants in this current study were comparable between groups in terms of time since stroke, severity of upper limb paresis and presence of cognitive, linguistic, and/or sensory impairments. Presence of voluntary finger extension at baseline was uncommon, highlighting limitations of this measure as a marker of recovery potential in a severely impaired cohort.
31
It is not known whether groups were comparable in terms of neurobiological reserve, as measures of residual brain function (eg, motor-evoked potentials) or structure (eg, corticospinal tract asymmetry) were not obtained.
48
Use of measures of brain reserve in future would allow prognostic stratification of participants, which in turn, may permit identification of stroke survivors with the greatest potential to achieve clinically meaningful improvements in arm function through SMART Arm training. For those with more limited neurobiological reserve, strategies to exploit their potential for
Two alternative interventions that enable stroke survivors with severe paresis to engage in upper limb practice are robotic therapy and ES.50-52 Similar to SMART Arm, evidence of effectiveness of these interventions above usual therapy has been variable. A recent RCT 8 comparing robotic therapy and ES with usual therapy demonstrated no difference between groups while earlier systematic reviews demonstrated improvements in arm impairment following robotic therapy 53 and arm activity following ES. 54 Participants in the current study had more severe motor impairments than participants typically selected for robotic therapy and ES studies,7,53,54 making comparison between interventions difficult. Encouragingly, all participants in the current study engaged in repetitive upper limb practice, despite presence of severe and multiple impairments across sensory, cognitive, and/or linguistic domains. Contrary to oft-held views,40,55 participants were able to demonstrate improvements in arm function that were maintained at 12-month follow-up. 56
Implications
To
To
Footnotes
Acknowledgements
The research team thank the following organizations and their staff for assistance with the project: Acute Stroke Unit and Geriatric and Rehabilitation Unit at Princess Alexandra Hospital; Geriatric and Rehabilitation Service at Queen Elizabeth II Jubilee Hospital; therapists who provided the intervention (Claire Lynch, Robyn Lamont, Joanne Mak); and who completed blinded assessments (Katrina Kemp, Dr Brenda Ocampo, Nampech Wuthapanich, Jessica Whiting), individuals who contributed to the construction of the SMART Arm device (Dr Stephen Wilson and Mr Russell Gee) and the laboratory data collection equipment (Dr Craig Tokono and Dr Christoph Szubski).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RN Barker, RG Carson, KS Hayward, SG Brauer, and D Lloyd are inventors of the SMART Arm and are currently involved in commercialization of the SMART Arm device with SMART Arm Pty Ltd but do not receive financial compensation. Their respective university commercialization trusts are represented on the Board of SMART Arm Pty Ltd.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a National Health & Medical Research Council Project Grant 2008-2011 (project ID: 511241). KS Hayward was funded by a National Heart Foundation of Australia and National Stroke Foundation of Australia Postgraduate Biomedical Scholarship (PB 09B 4847) and an Australia Postgraduate Award scholarship. SG Brauer, RG Carson, D Lloyd, and RN Barker received no personal financial support for this project.
