Abstract
Introduction
Patients with Parkinson disease (PD) are unable to generate adequate amplitude of movement and maintain internal gait rhythm because of a dopaminergic deficiency in the nigrostriatal pathway. 1 This results in an abnormal gait pattern characterized by reduced gait speed, shortened stride length, and a longer double-limb support phase. 2 Despite optimal medical and surgical therapies, patients with PD develop progressive gait disturbances and disability.2-4 Therefore, improving walking ability is a primary goal in PD rehabilitation. 5
Physiotherapy (PT) aimed at enabling patients to maintain their maximum level of activity and independence is often prescribed, 6 but with the exception of rhythmic cueing strategies, 7 treatments for gait have modest benefit. 6 Electromechanical devices and treadmill training (TT) may improve walking ability in PD.8,9 Animal models of PD suggested that training programs based on exercise may increase neuroprotection and neurorestoration. 10
As to the issue of motor learning, sequential movements such as walking are acquired through a process of implicit learning becoming automatic with practice.11,12 In PD, implicit learning impairment emerges with progression of the disease, whereas explicit learning is impaired in the early stage of illness as a consequence of early deficits in attention and working memory. 12 Task-specific paradigms may improve the retention of motor skill learning in adults with PD. 13 Miyai and colleagues evaluated the short- and long-term effects of a 4-week body weight–supported TT in patients with PD, reporting greater improvements in motor performance and walking ability compared with conventional PT.14,15 In addition, TT without body weight support has been reported to better improve walking ability, lower-extremity tasks, and dynamic balance than conventional PT in PD8,16,17 even after a single session. 5 Moreover, it has been observed that specific exercise programs based on incremental speed-dependent TT may improve walking ability and reduce postural instability in PD. 18
A pilot study that aimed to evaluate the potential effects of TT with robot assistance on reducing of freezing of gait (FOG) reported promising results for FOG symptoms and walking ability. 19 To date, no randomized controlled trial (RCT) has been carried out to primarily analyze the effects of robot-assisted gait training (RAGT) on walking ability in people with PD.
The main aim of this RCT was to evaluate whether a rehabilitation program consisting of RAGT could be more effective than a conventional PT program for improving walking ability (ie, gait speed and walking capacity) in patients with PD. The secondary aim was to examine how RAGT could influence spatiotemporal gait parameters and whether this could have a positive impact on fatigue and severity of disease in PD.
Methods
This RCT was performed in the Neurological Rehabilitation Unit of the Azienda Ospedaliera-Universitaria Integrata of Verona, Italy. Inclusion criteria were confirmed idiopathic PD, Hoehn and Yahr stage of 2.5 or 3, 20 and Mini-Mental State Examination score >23. 21 Exclusion criteria were severe dyskinesias or “on-off” phases, change of PD medication during the study, deficits of somatic sensation in the lower limbs, vestibular disorders or paroxysmal vertigo, other neurological or orthopedic conditions involving the lower limbs, and cardiovascular comorbidity.
All participants were outpatients and gave their informed consent for participation in the study. The study was carried out according to the Declaration of Helsinki and was approved by the local ethics committee.
Prior to testing, participants were divided into 2 groups according to a simple randomization scheme 22 using a randomization list locked in a desk drawer accessible only to the principal investigator. During the study, participants were instructed to take their PD medications regularly and were tested and trained 1 to 2.5 hours after taking their morning dose. Participants did not perform any type of rehabilitation in the 3 months before the study nor did they undergo any form of PT other than that scheduled in the study protocol.
Treatment Procedures
Each patient underwent a training program consisting of twelve 45-minute sessions (including rest periods), 3 days a week (Monday, Wednesday, and Friday) for 4 consecutive weeks.
Robotic stepper training device (RST) group
Patients allocated to the RST group performed RAGT by means of the Gait Trainer GT1 (Reha-Stim, Berlin, Germany), one of several commercially available robotic-assistive devices.23-36 The GT1 is a static suspension system consisting of 2 motor-driven footplates positioned on 2 bars that provide a robot-assisted propulsion by means of a planetary gear system, simulating stance and swing with a ratio of 60% to 40% between the 2 phases. 23 No previous treatment protocol has been proposed for this population. We defined our protocol according to our clinical experience, the GT1 characteristics (speed up to 2 km/h), and the TT protocols in the literature that suggest a progressive reduction of body weight support combined with an increase of gait speed.14,15,37 In our view, the rationale for supporting body weight was to increase safety and compliance with the RAGT. In this study, the step length was individually defined according to the evaluation performed with the GAITRite system. A step length of 48 cm (maximum GT1 step length) was chosen for patients with a step length >48 cm. Each training session consisted of 3 parts (each one lasting 10 minutes), with a 5-minute rest after each of them. First, we trained patients at 20% of body weight supported and a speed of 1 km/h for 10 minutes; then, at 10% of body weight supported and a speed of 1.3 km/h for 10 minutes; and finally, at 0% of body weight supported and a speed of 1.6 km/h for 10 minutes. Patients were instructed to “help” the GT1 gait-like movement during training. Any patient unable to maintain the chosen pace was excluded from the study.
Physiotherapy group
Patients allocated to the PT group performed a PT program, including active joint mobilization and conventional gait training. Each treatment session consisted of 2 parts with a 5-minute rest between them. First, patients performed active joint mobilization of the lower limbs (hip, knee, and ankle) in the supine and prone positions (10 repetitions of 6 exercises) for 10 minutes (5 minutes per position). Then, they performed conventional gait therapy based on the proprioceptive neuromuscular facilitation (PNF) concept for 30 minutes.5,38 Among the PNF techniques, we facilitated pelvic motion to improve control of the pelvis as a “key point of control” for maintaining a gait pattern.5,38 Conventional gait therapy consisted of 10 minutes each of rhythmic initiation, slow reversal, and agonistic reversal exercises applied to the pelvic region.5,38 The time spent in PNF was equal to that spent in RAGT. The same trained therapist treated all the patients in this group and standardized the duration of each part of the treatment.
Testing and Intervention
Patients were evaluated before (T0), immediately after (T1; primary end point), and 1 month after the end of treatment (T2). All patients were evaluated by the same examiner (an experienced internal co-worker) who was blinded to treatment allocation.
Primary outcomes
Primary outcomes were the 10-m walking test (10MWT) 39 and the 6-minute walking test (6MWT). 40 The 10MWT has a test-retest reliability of 0.90 in this population 41 and was selected as a measure of gait speed. 39 We required patients to walk on a flat hard floor at their fastest speed for 10 m (the distance was defined by 2 lines on the floor). To minimize acceleration and deceleration, patients were positioned 1 m behind the start line and were instructed to walk until 1 m past the finish line. The evaluator was positioned at the finish line and recorded time with a handheld stop watch. Patients were not allowed to use walking aids. Walking capacity was assessed using the 6MWT. 40 Test-retest reliability is 0.90 in this population 41 and is aimed at evaluating submaximal endurance. 40 Participants were required to walk indoors along a marked distance (1 lap, 40 m) at their maximum speed for 6 minutes 40 without walking aids.
Secondary outcomes
The GAITRite system (CIR Systems Inc, Havertown, Pennsylvania) was used to evaluate spatiotemporal gait parameters.42,43 To avoid acceleration and deceleration, patients were asked to ambulate along the 7.66-m GAITRite electronic walkway at their fastest speed on a 12-m course. Three trials of this task were conducted, and their mean was calculated. Stride length, coefficient of variation of stride time [(Standard deviation of stride time/Mean stride time) × 100], cadence, and ratio between single- and double-support duration were evaluated. The Parkinson’s Fatigue Scale (PFS) was used to assess fatigue.
44
It is a 16-item scale with item response options ranging from 1 (
Sample Calculation and Statistical Analysis
A prestudy power calculation estimated that 18 participants would provide 90% power to detect a difference of 4.3 m/s in the 10MWT between the 2 groups.
46
We used the independent samples
Results
A total of 41 participants (Table 1 and Figure 1) were recruited from among 74 outpatients consecutively admitted to our Neurological Rehabilitation Unit from November 2009 to January 2011. Of these, 21 patients were allocated to the RST group and 20 to the PT group Figure 1). Because of lack of cooperation, 3 patients in the RST group and 2 patients in the PT group withdrew. According to the step length on the GT1 for the RST group at T0 (mean = 45.11 cm; SD = 7.46 cm), the mean number of gait cycles per minute performed in each session of RAGT was 76.97 (SD = 21.80) in the first part (20% body weight support; speed = 1 km/h), 100.15 (SD = 28.36) in the second part (10% body weight support; speed = 1.3 km/h), and 123.34 (SD = 34.92) in the third part (0% body weight support; speed = 1.6 km/h). No adverse events occurred during the study.
Demographic and Clinical Features of Patients
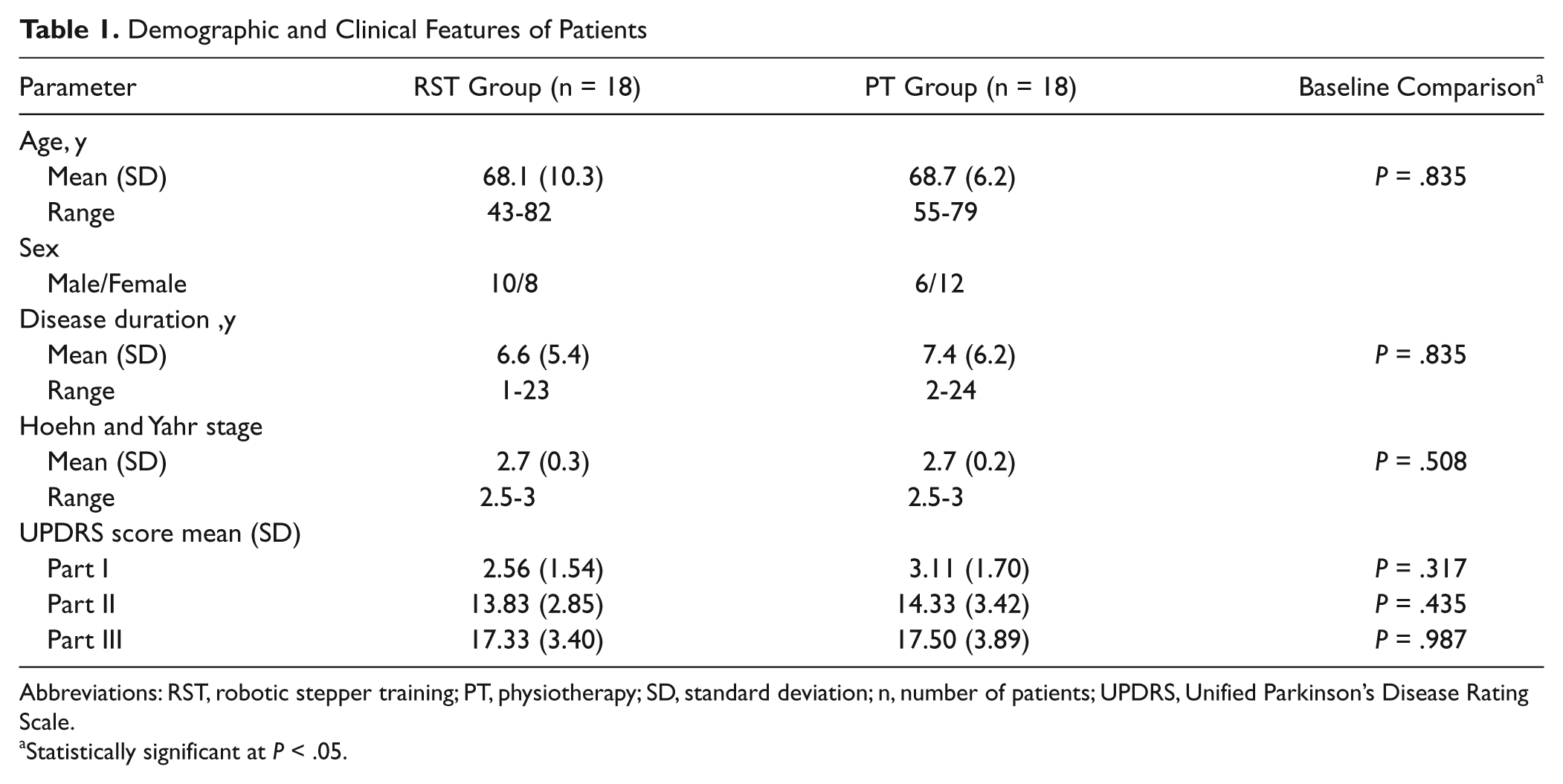
Abbreviations: RST, robotic stepper training; PT, physiotherapy; SD, standard deviation; n, number of patients; UPDRS, Unified Parkinson’s Disease Rating Scale.
Statistically significant at
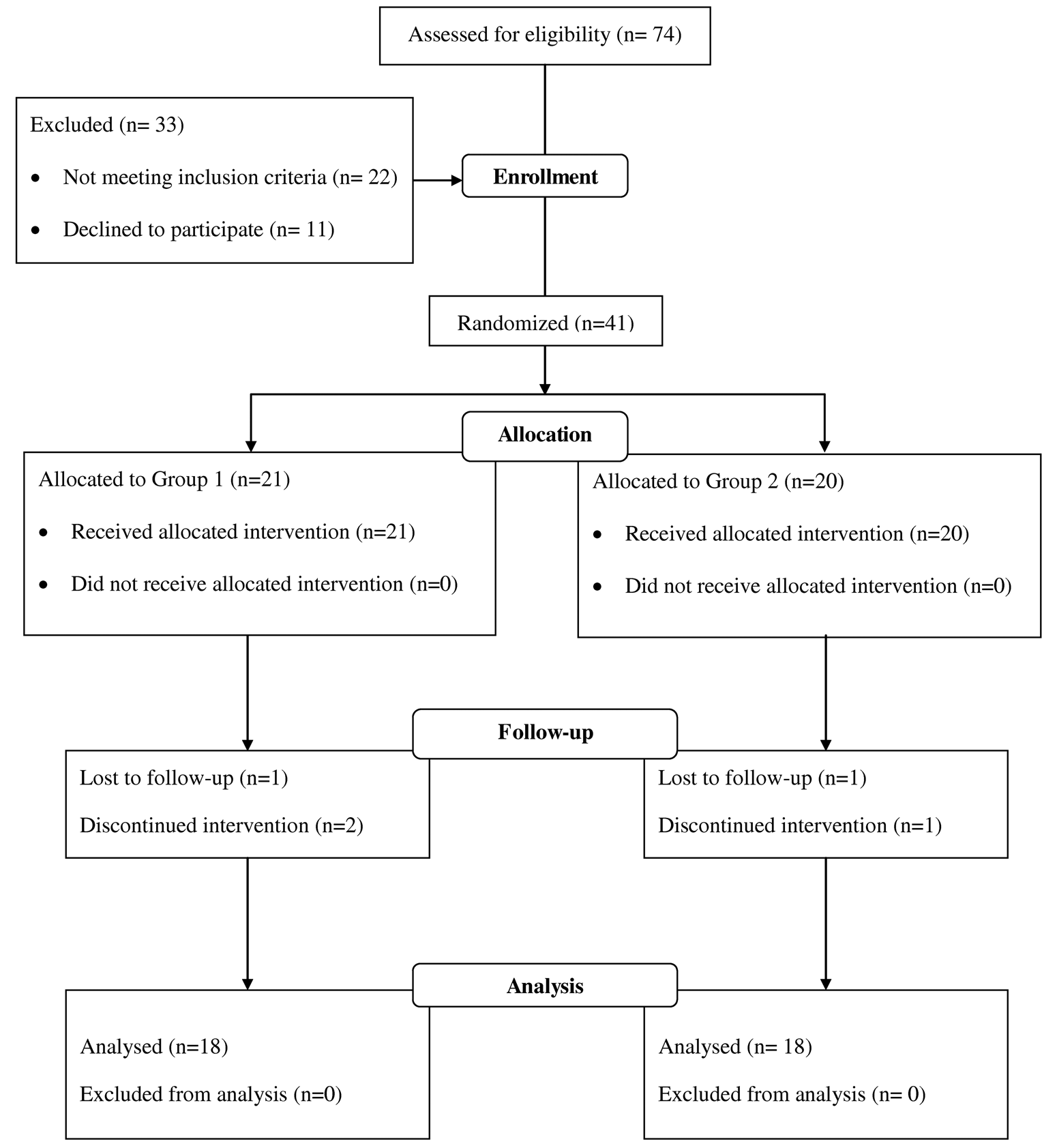
Flow diagram of the study.
Baseline
Multiple separate independent samples
Patients’ Performance in All Outcome Measures
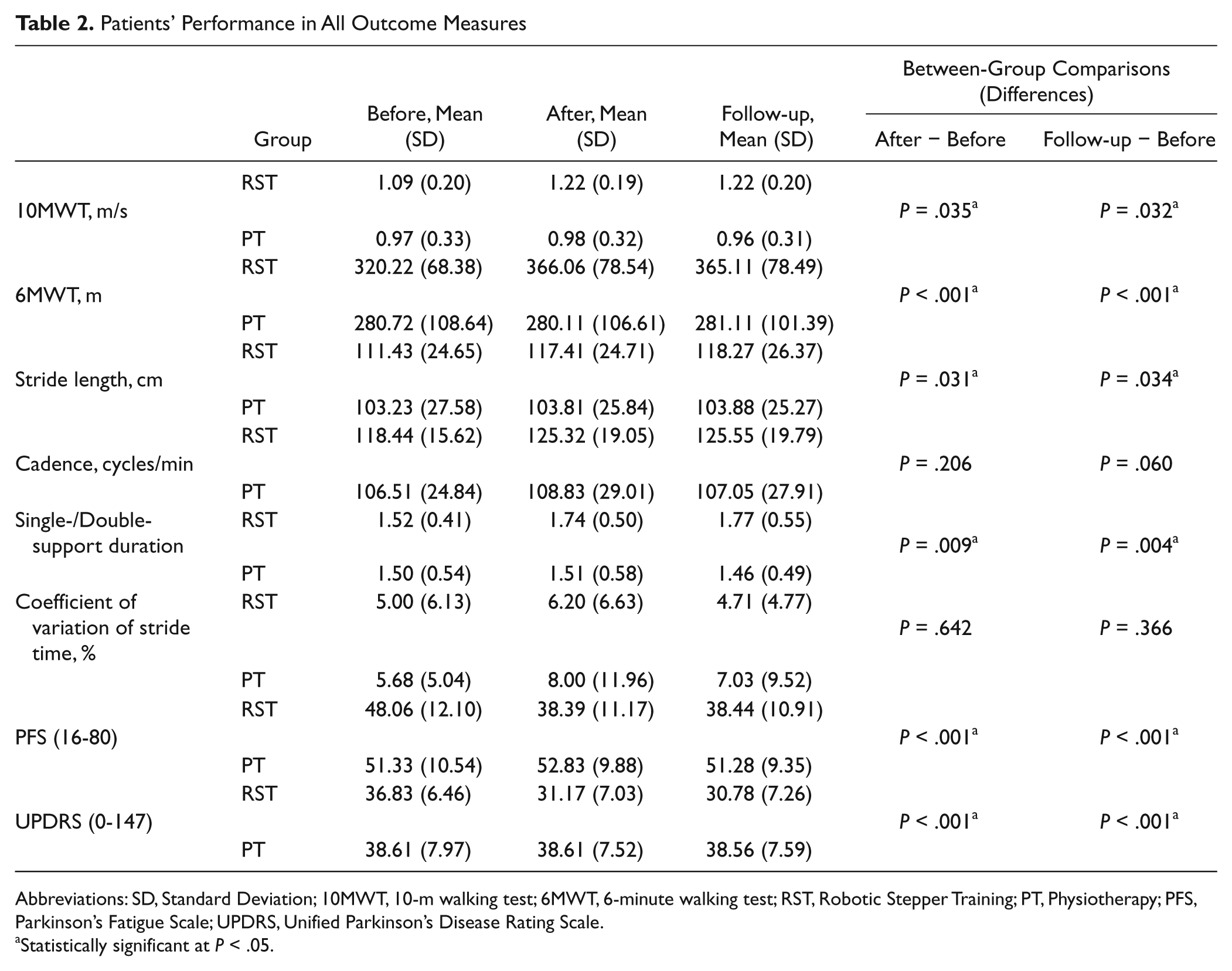
Abbreviations: SD, Standard Deviation; 10MWT, 10-m walking test; 6MWT, 6-minute walking test; RST, Robotic Stepper Training; PT, Physiotherapy; PFS, Parkinson’s Fatigue Scale; UPDRS, Unified Parkinson’s Disease Rating Scale.
Statistically significant at
Effects of Training on Gait Speed and Walking Capacity
For the 10MWT, a significant main effect was revealed for treatment (
For the 6MWT, a significant main effect was revealed for treatment (
Effects of Training on Gait
As for the stride length, a significant main effect was seen for treatment (
For cadence, no significant main effect was revealed for treatment. Within-group multiple comparisons showed significant changes from T0 to T1 (
For the ratio between single- and double-support duration, a significant main effect was revealed for treatment (
Effects of Training on PFS and UPDRS
Between-group comparisons showed that patients in the RST group performed significantly better (
Discussion
The aim of this study was to evaluate whether RAGT could be more effective than a conventional PT program in improving walking ability in patients with PD. Our results showed that patients with PD who underwent RAGT improved gait speed, walking capacity, cadence, and fatigue better than those who took part in a conventional PT program that did not emphasize walking practice. It should be underscored that the PT group had lower baseline scores (albeit not significantly different) than the RST group. The change in the UPDRS was not clinically important. 48
Gait hypokinesia is a major impairment limiting walking capacity in people with mild to moderate PD. 49 Previous studies have shown that gait speed can be enhanced by increasing cadence, stride length, or both.50-52 The improvement of gait speed, promoted by RAGT in this study, results mainly from changes in stride length. Furthermore, the ratio between single- and double-support duration showed significant increases in patients who underwent RAGT. Based on these findings, we argue that RAGT could effectively act on gait pattern generation in PD. This is relevant, from a rehabilitative point of view, considering not only that gait speed is the largest and only independent contributor to walking capacity in PD but also that interventions aimed at improving walking capacity in PD should target sustaining walking at fast speeds. 49 Moreover, it has been reported that people with PD who routinely use lower walking velocities deteriorate over time, further reducing their walking capacity. 49 Our findings show that RAGT significantly improves walking capacity when compared with a conventional PT program in patients with PD. This is further confirmed by the significant reduction in fatigue, as shown by the PFS. However, it should be noted that the statistically significant improvement in gait parameters obtained by the RST group (0.12 m/s for the 10MWT and 45.84 m for the 6MWT at T1) might not be considered as clinically significant. In fact, a recent study described as clinically significant changes of 0.25 m/s for the 10MWT and 82 m for the 6MWT in patients with PD. 41 On the other hand, we should underscore that changes of 0.10 m/s for the 10MWT and 50 m for the 6MWT are considered as a minimally clinically meaningful effect in older adults. 53
A challenging question is to understand why RAGT proved to be more effective in PD. It is plausible that several repetitions of gait-like movements could act as an external proprioceptive cue by setting the walking pattern and reinforcing the neuronal circuits that contribute to gait pacing. Moreover, our patients were instructed to “help” the GT1 movement during training, and this could have enhanced the cueing effect of RAGT by focusing the patient’s attention on the imposed external pace. 54 Robotic training could have also enhanced the automating of motor control by stimulating the central pattern generators through a greater activation of hip extensors as compared with the PNF approach.50,55 Another possible explanation may be found in the reconditioning effect of RAGT. Reduced physical activity has been reported to be associated with reduced walking capacity in PD. 50 Our patients experienced improved walking ability and reduced fatigue after RAGT probably because of the augmented physical activity induced by active robotic training compared with less walking during PT. Moreover, the effects of deconditioning appeared to be negligible for up to 1 month, probably because our patients were able to maintain condition by engaging in more extended movements in their everyday life after RAGT. This is in keeping with previous findings about high-intensity resistance training in PD, which described the persistence of lower-body muscle strength gains for up to 4 weeks after treatment.56,57
In this study, patients in the PT group performed active joint mobilization and conventional gait therapy based on the PNF concept with an amount of treatment time similar to those in the RST group but failed to show any significant improvement in walking ability. In a recent review about evidence-based PT in people with PD, Morris and colleagues 58 described 3 key elements for the management of gait disorder training based on compensatory strategies to bypass the defective basal ganglia and strategies to improve motor learning and performance through practice; management of secondary musculoskeletal and cardiorespiratory sequelae that occurs as a result of deconditioning, age, and comorbidities; and promotion of physical activity and fall education. A possible explanation of the lack of treatment effect in the PT group is that exercises were performed lying in bed, and therefore, patients received very little input suitable for improving motor strategies or physical activity. On the other hand, patients in the RST group performed a high intensity training with a large number of repetitions of gait cycles per minute. Taking into account the scant effects of PNF on Parkinsonian gait, adequate future studies with matched-dose therapies are needed to further compare the effectiveness of PT procedures with that of robotic devices for gait rehabilitation in PD. One previous pilot study of 4 patients demonstrated a reduction of FOG by repetitive robot-assisted TT (Lokomat, Hocoma, Zurich, Switzerland). 20 Authors also reported an increase in gait speed and stride length as secondary outcomes. 20
An important limitation of the present study is the nonsignificant (but substantial) difference for the primary measurements of outcome at baseline, which might be a potential confounder for understanding the added value of robotic training. Another limitation is the lack of a follow-up assessment at 3 or more months after training and the lack of assessment of balance. Moreover, we did not test participants “off” medication and thus cannot draw conclusions about the contribution of RAGT in the unmedicated state.
Future studies should determine the frequency, duration, and other important aspects of RAGT parameters, such as speed and need for body weight support, that might be most effective and compare RAGT with TT or another active gait intervention to improve walking ability in patients with PD.
Footnotes
Acknowledgements
The authors would like to thank Stefano Tamburin and Patrizia Ianes for their technical assistance in the preparation of the manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
