Abstract
The first Stroke Recovery and Rehabilitation Roundtable established a game changing set of new standards for stroke recovery research. Common language and definitions were required to develop an agreed framework spanning the four working groups: translation of basic science, biomarkers of stroke recovery, measurement in clinical trials and intervention development and reporting. This paper outlines the working definitions established by our group and an agreed vision for accelerating progress in stroke recovery research.
Introduction
The first Stroke Recovery and Rehabilitation Roundtable (SRRR) was convened with the aim to move rehabilitation research forward. 1 Working collectively across four initial priority areas, we reviewed, discussed, and attempted to achieve consensus on key recommendations in each of the areas of translation of basic science, 2 biomarkers of stroke recovery, 3 measurement in clinical trials 4 and intervention development and reporting. 5 Agreed definitions were a priority. Definitions within stroke recovery research are particularly complex given both the extended time window over which research, clinical interventions and recovery take place; and the multi-disciplinary, multi-faceted nature of the field. This paper outlines the working definitions established by our group that underpinned the scope and methodologies of each of the four groups. Agreed priority areas for accelerating progress in stroke recovery research are highlighted as a way forward for the field. These were developed following comprehensive discussions at the first SRRR roundtable meeting convened in Philadelphia, 2016.
A major point of agreement of the SRRR expert group was to focus on progress of stroke recovery research in the next decade and beyond. ‘Rehabilitation’ as a blanket term for all therapy-based interventions post-stroke was considered problematic, vague and an impediment to progress. Rehabilitation reflects a process of care, while recovery reflects the extent to which body structure and functions, as well as activities, have returned to their pre-stroke state. With that, the term ‘recovery’ can be represented in two ways: (1) the change (mostly improvement) of a given outcome that is achieved by an individual between two (or more) timepoints, or (2) the mechanism underlying this improvement in terms of behavioural restitution or compensation strategies.6,7 We used the definition of rehabilitation developed by the British Society of Rehabilitation Medicine, 8 “a process of active change by which a person who has become disabled acquires the knowledge and skills needed for optimum physical, psychological and social function.” Stroke rehabilitation is most often delivered by a multidisciplinary team, defined by the World Health Organisation (WHO) 9 to encompass the coordinated delivery of intervention(s) provided by two or more disciplines in conjunction with medical professionals. This team aims to improve patient symptoms and maximise functional independence and participation (social integration) using a holistic biopsychosocial model, as defined by the International Classification of Functioning Disability (ICF). 9
Recovery
The motor system has been studied more than any other in stroke recovery research, as such this was the focus of most dialogue within the SRRR. While many of the principles of recovery emerging from research conducted on the motor system likely extend to non-motor systems, differences exist in the organisation of brain systems. In discussing stroke recovery, acknowledging that any improvement in any domain of the ICF can be viewed as a sign of ongoing recovery is important. For research, understanding the processes that underpin how recovery is achieved during stroke rehabilitation is of utmost value. An understanding that distinguishes between behavioural restitution and use of compensation strategies will further direct how we should train stroke patients to regain the ability to complete meaningful tasks and how we should design interventions, including technology applications for stroke such as rehabilitation robotics.
Behavioral restitution or true recovery
Behavioral restitution has been defined as a return towards more normal patterns of motor control with the impaired effector (a body part such as a hand or foot that interacts with an object or the environment) and reflects the process toward “true recovery.”10,11 True recovery defines the return of some or all of the normal repertoire of behaviors that was available before injury. Neural repair is required for true recovery. Although rarely complete after stroke, some degree of true recovery is nearly always achieved. 12 For the motor system, recovery is best measured with kinematics, 4 and for the language system, a test of speech or language production may be the optimal measure. 13 The development of stroke treatments administered after the hyperacute period of early damage and brain cell death that restore normal function, thereby promoting true recovery, remains an aspirational goal yet to be realized across functional domains.
Compensation
A patient’s ability to accomplish a goal through substitution with a new approach rather than using their normal pre-stroke behavioral repertoire constitutes compensation. This behavior does not require neural repair, but may require learning. Compensation may be seen in all functional domains. In the motor domain, compensation strategies employ the use of intact muscles, joints and effectors in the affected limb, to accomplish the desired task or goal.10,11 In the language system, compensation may refer to the use of an augmentative and alternative communication device, including a communication board. At present, researchers commonly test interventions that allow or promote compensation, rather than behavioral restitution, in order to improve a patient’s safety and quality of life. This approach is compounded by the choice of an outcome measure, which is unable to distinguish between the two, so that the potential mechanism of an intervention remains opaque.
Spontaneous biological recovery
In animals, this term refers to improvements in recovery of behavior in the absence of a specific, targeted treatment and occurs during a time-sensitive window that begins early after stroke and slowly tapers off.6,11,14 In human stroke survivors, a similar period of heightened recovery of behaviors occur early post-stroke with little or no active treatment. 15 The duration of the window varies across neural systems, e.g. weeks to months post stroke for arm movement, 16 but longer (weeks to years) for other systems, such as language. 13 There is literature pertaining to motor,17–20 visuospatial neglect, 21 and language22,23 systems; data for other neural systems exist but are sparser, highlighting research priorities for the field. Most stroke survivors exhibit spontaneous recovery, progressing through characteristic stages. 24 Proportional recovery rules suggesting that the degree and rate of recovery are strongly predictable post stroke have been proposed in a number of domains (e.g. in upper limb recovery,19,20 visuospatial neglect 21 and language functions.22,25) However, a substantial group of patients do not fit such proportional recovery rules. Our challenge is to study spontaneous recovery, to understand its biological basis, to determine if we can identify recovery phenotypes in order to select patients for interventions, 26 and to use this knowledge to guide the development of interventions that boost behavioral recovery beyond that which occurs spontaneously. Additional definitions that are key for the field of stroke rehabilitation and recovery are contained throughout this document and in Appendix 1.
Timeline of stroke recovery
A further challenge for our field is determining the optimal timing to implement interventions focused on recovery and repair.1,6,27,28 As a first step, we needed to agree on a common framework—underpinned by what we know about the biology of recovery— for defining what is meant by “acute,” “sub-acute,” and “chronic.”6,29 These terms are often used in recovery research without adequate definition. Building on previous work by Dobkin and Carmichael, 28 we developed the framework shown in Figure 1. The framework is strongly informed by pre-clinical research in animal models of stroke,30–33 as well as individuals with stroke,18,27,34 particularly from studies of the motor system. This framework should be updated as more knowledge is acquired. Figure 1 outlines the timing (hours, days, months) of several important biological processes in ischaemic 35 and haemorrhagic 36 stroke, as well as the temporal terms (hyper-acute, acute, early and late sub-acute, chronic) across the first six months post-stroke and beyond. The possibility for behavioral changes even years post-stroke is recognized. However, the current understanding of brain repair processes suggests that the majority of behavioral recovery, and the rapid changes occur in the first weeks-to-months post stroke for most people. This time perspective represents an important treatment target to maximize the potential of restorative interventions.
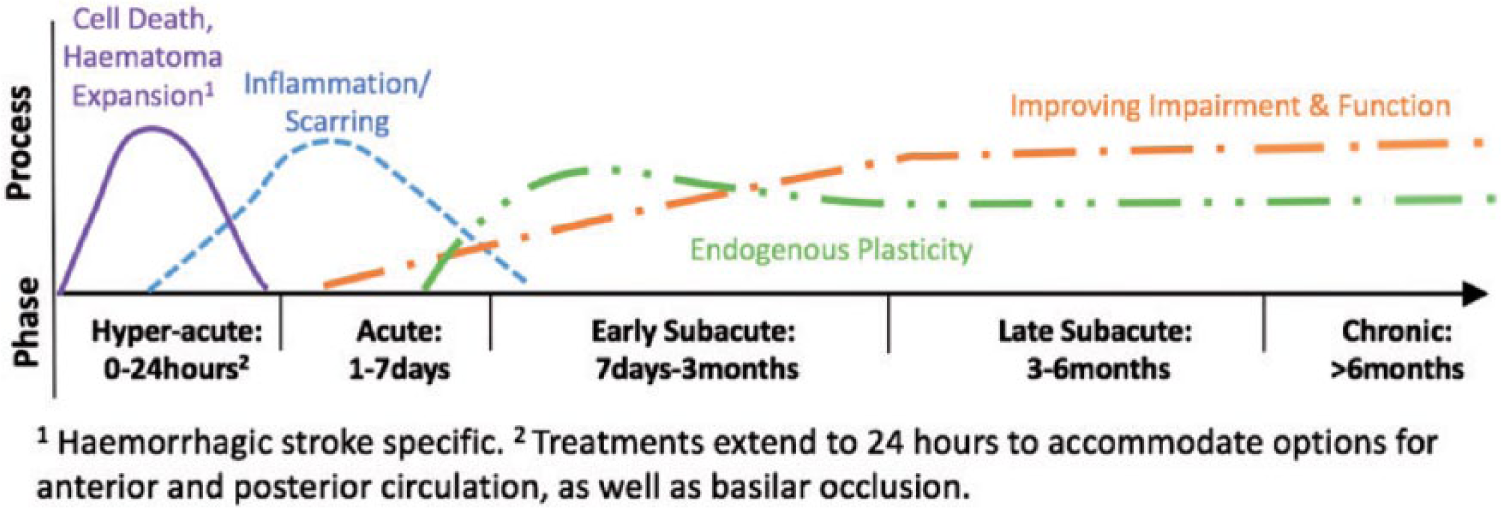
Framework that encapsulates definitions of critical timepoints post stroke that link to the currently known biology of recovery.
The convention proposed for recovery research is that treatments commenced within a week of stroke onset should be classed as “acute.” Relatively, few recovery trials have initiated restorative treatments within this post-stroke phase (for reviews see37,38). The first week until the first month post-stroke (acute and early sub-acute) is a critical time for neural plasticity6,30,39 and should be a target for recovery trials, with some uncertainty about how early and how intensively to start training.37,40 Importantly, we strongly recommend that in all recovery and rehabilitation research, the time from stroke onset is gathered and reported. The start and end of any intervention(s), experimental or standard of care, as well as timing of outcome and follow-up assessment should also be reported. Using this framework, the SRRR groups provide recommendations, e.g. the measurement group recommend core measures to be included in every trial of stroke recovery and rehabilitation; 4 the biomarker group provide recommendations about the timing and type of data acquisition. 3
The way forward
As the body of research in stroke recovery and rehabilitation continues to grow, we will increasingly see interventions specifically developed with the aspiration to target true recovery rather than compensation. Finding breakthrough treatments is critical and has the potential to set the stroke recovery research field on a radically new path. One only needs to look at the transformational effect of thrombolysis and endovascular thrombectomy on acute stroke outcomes, research funding in this area, and importantly, on health service delivery, to understand the importance of breakthrough treatments in recovery. A number of key themes for future research and collaboration emerged from the SRRR discussions are briefly outlined below.
As a group, the SRRR participants are committed to progressing these themes. We hope that researchers, clinicians and academics working or interested in the field of stroke recovery, together with funding bodies and journal editors, will join us in pursuing and promoting the goals outlined here and in our recommendation papers.2–5
Footnotes
Appendix
Additional Definitions That Are Key for the Field of Stroke Rehabilitation and Recovery.

Acknowledgements
We would like to acknowledge Professor Julie Bernhardt for convening this first Stroke Recovery and Rehabilitation Roundtable (SRRR), Ms Katie Lafferty for moderating discussions at SRRR and Dr Karen Borschmann for project management. We thank everyone who contributed to the SRRR meeting: Myzoon Ali, Jovita Balcaitiene, Michele Basso, Francesca Bosetti, Marian C. Brady, Leeanne M. Carey, Alexandre R. Carter, Daofen Chen, Leonid Churilov, Andrew N. Clarkson, Leonardo G. Cohen, David A. Copland, Numa Dancause, Catherine M. Dean, Janice J. Eng, Coralie English, Amanda J. Farrin, Cynthia Felix, Rebecca J. Fisher, Anne Forster, Mary Elizabeth Harriman, Tammy C. Hoffman, Heidi Johansen-Berg, Jukka Jolkkonen, Theresa A. Jones, Mary Teena Joy, Katie Lafferty, Peter Langhorne, Natasha A. Lannin, Alex P. Leff, Elizabeth A. Lynch, Jane M. Maguire, Louise D. McCullough, Timothy H. Murphy, Michael Nilsson, Kathryn A. Radford, Charlotte Rosso, Gustavo Saposnik, Martin E. Schwab, Cathy M. Stinear, Katharina S. Sunnerhagen, Erwin E. H. van Wegen, Marion F. Walker, Caroline L. Watkins, Tadeusz Wieloch, Carolee Winstein.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JB was supported by NHMRC (1058635); KSH was supported by NHMRC (1088449) and Micheal Smith Foundation for Health Research (MSFHR) (15980); SLW was supported by National Institutes of Health (NIH) (1U10NS086607); LAB was supported by the Canada Research Chairs and MSFHR (CI-SCH-01796); DC was supported by the HSF of Canada and CPSR; SCC was supported by a grant from NIH (K24 HD074722). We acknowledge the following organisations for their financial support of the meeting: National Health and Medical Research Centre (NHMRC) Centre of Research Excellence in Stroke Rehabilitation & Brain Recovery (Australia), Heart and Stroke Foundation (HSF) of Canada, Heart and Stroke Foundation Canadian Partnership for Stroke Recovery (CPSR). An unrestricted educational grant was provided by Ipsen Pharma. The Florey Institute of Neuroscience and Mental Health acknowledges support from the Victorian Government, in particular funding from the Operational Infrastructure Support Grant.
