Abstract
Introduction
Cognitive deficits are among the most common and invalidating consequences of traumatic brain injury (TBI), which may significantly interfere with the individual’s social and vocational outcomes.1,2 This wide range of cognitive disturbances is caused by damage to white-matter connections, due to diffuse axonal injury (DAI). 3 As a consequence of DAI, a wide network of areas, subserving different cognitive functions, might be compromised, so that the domains of attention,4-6 memory, 7 and executive functioning 8 are quite consistently impaired. Although in the first postinjury phase a period of spontaneous reorganization occurs, due to the resolution of acute neurological events resulting in a partial motor and cognitive recovery, 9 many deficits are still present in the chronic phase of injury. Many authors have emphasized the importance of cognitive rehabilitation to reduce the cognitive and behavioral consequences of TBI, thus increasing patients’ autonomy and quality of life.10,11
Cognitive rehabilitation is defined as a systematic, functionally oriented service of therapeutic activities based on assessment and understanding of the patient’s cerebral and behavioral deficits. It is not limited to restorative interventions, directly aimed at intervening on the cognitive disturbances caused by the brain damage, but it may also comprehend the use of compensatory processes by establishing alternative patterns of cognitive activity (through compensatory cognitive mechanisms for impaired neurologic systems) or creating new patterns of activity through external support devices (eg, prosthesis or adaptive aids), which, even if not specifically focused on the direct modification of cognitive impairments, are aimed at improving the patient’s overall level of functioning and his/her quality of life.12,13 The common element of these rehabilitation approaches is neural plasticity: the brain is able to reorganize and relearn those functions that were lost after an acquired brain lesion, by promoting the correction of maladaptive plasticity and leading to more functional neural growth state.14-19 This mechanism is remodeled by behavioral experience: the brain can be altered by a wide variety of events and stimuli across its lifespan. 3 Even though there many studies in the literature that report the effectiveness of rehabilitation in promoting brain restoration,20-23 only a few of them focused on the neural modifications induced by cognitive treatment. This might result in a lack of relevant information, as these behavioral and cognitive evaluations alone cannot provide a complete understanding of the causal links between injuries, their location, the subsequent deterioration in structural connectivity, and the functional deficits experienced by patients in their daily lives. 24 Neuroimaging and neurophysiological techniques could allow a better assessment and diagnosis, as well as an understanding of the neural mechanisms that accompany cognitive improvements (or lack of improvements). Among the primary techniques that have been employed to investigate the neural modifications of TBI there are electroencephalography (EEG) (including event-related potentials [ERPs]) and structural or functional magnetic resonance imaging (MRI and fMRI, respectively). MRI and fMRI provide researchers with an effective and noninvasive method to examine brain changes following treatment interventions.1,3,13,18 Similarly, ERPs and oscillatory activity from the human EEG give relevant information about the severity of injury and its impact on neuronal pathways, including their efficiency in conducting signals from the peripheral to the central nervous system (CNS), the ability of CNS structures to process sensory input, and the ability of specific sensory systems to perceive and integrate stimuli. 25 However, even if many studies discussed all the possible applications of neuroimaging and neurophysiological instruments for diagnostic and prognostic purpose,25-33 as far as we know there are no review studies focusing on the neuroplastic changes induced by cognitive training in TBI individuals. The main purpose of this work was to broadly examine the literature on the structural, functional, and neurophysiological modifications of cognitive treatments in chronic TBI subjects. A review study in this field may give a better understanding of how a chronically and traumatically injured brain is reshaped by targeted stimulation and which networks are predominantly activated in this process. In this way, it may be possible to share common standpoints arising from all the studies in question, in order to gain further clinical implications.
Methods
Studies investigating both functional and structural neural modifications induced by cognitive training in TBI subjects were included in this review. In particular, we included every form of neural reorganization induced by external, repeated, and targeted stimulation leading to a more adaptive cognitive or behavioral outcome. Both types of research on restorative and compensative rehabilitation were considered. The following inclusion criteria were used: studies carried out on TBI diagnosed patients, an absence of any other previous cerebrovascular/cerebral accident or neurodegenerative disease, and absence of other concomitant psychiatric illnesses or drug addiction.
Furthermore, inclusion criteria recommended participants being between the ages of 18 and 65 years, being in the chronic phase of injury, and undergoing cognitive training. Finally, pre- and post-training neuroimaging examinations were required.
The following study types were included: group studies, pilot studies, single cases, and meta-analysis and reviews.
Search Strategy
Studies were identified from the results of the following electronic databases searched: PubMed, ISI Web of Knowledge, Scopus, Psychinfo, and Cochrane.
Search terms for studies published between 1985 and 2016 on functional neuroplasticity induced by cognitive rehabilitation included cognitive training (or rehabilitation); and/or traumatic brain injury (or brain injury) combined with functional magnetic resonance (or fMRI); or event-related potentials; or electroencephalography (or EEG); or neural restoration; or plasticity; or remediation.
With regard to research on structural changes, the following keywords were entered: cognitive training (or rehabilitation) and/or traumatic brain injury (or brain injury) combined with structural changes (or structural modifications); or computed tomography (CT); or morphometry; or diffusion tensor imaging; or magnetic resonance, in articles published between 1985 and 2016.
Forty-five published articles were initially identified (35 related to functional changes and 10 on structural modifications). Articles were reviewed to remove those that did not meet at least one of the aforementioned inclusion criteria. So 34 studies were excluded (25 on functional changes and 10 on structural changes). With regard to the studies on functional neuroplasticity, the following articles were ruled out: 1 article reporting fMRI acquisition only in the pretraining session 32 ; 1 study protocol 34 ; 4 studies referring to individuals with mixed etiology35-38; 4 studies including patients in the acute phase of recovery13,39-41; 2 articles focusing on the neural changes induced by motor rehabilitation42,43; 13 articles (5 descriptive articles and 8 reviews) that did not specifically focus on the effects of cognitive training in TBI subjects.24,25,33,44-53
None of the articles on structural changes following cognitive training was included. Six articles (5 reviews and 1 descriptive article) were ruled out because they were too general or because they did not specifically focus on the effects of cognitive training in TBI subjects2,54-58; 1 article was excluded because it just analyzed the correlations between cognitive performance and CT findings in TBI subjects, without considering the rehabilitation process, 59 and 1 study was removed because it was about subjects in the acute phase of recovery. 60 Finally, 2 studies were excluded because they did not use posttraining neuroimaging examinations.30,31 In light of such analysis, 11 studies were found to meet the inclusion criteria: 7 fMRI studies (2 single-case studies1,61 and 5 group studies11,19,62-64), 3 articles with evoked readiness potentials (2 single-case studies,65,66 and 1 group study 67 ), and one EEG study 28 (see Table 1 and Figure 1).
Summary of Study Findings.
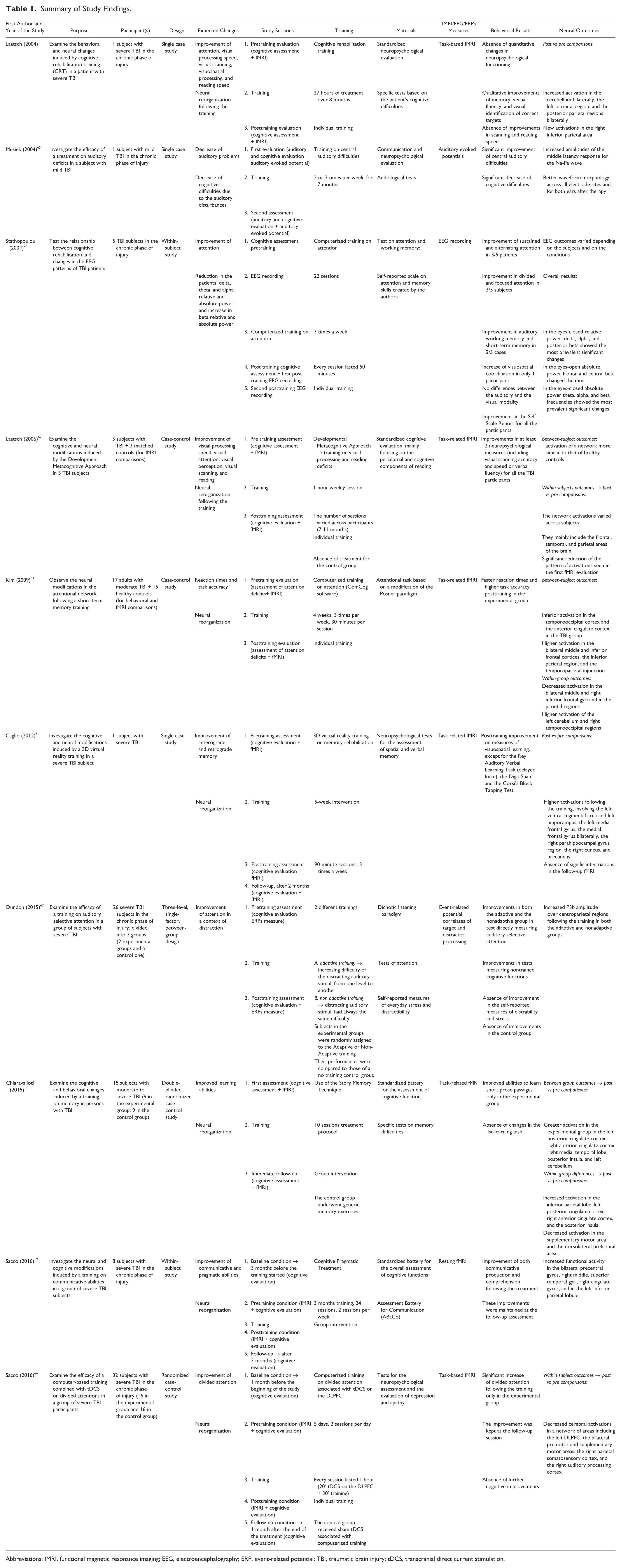
Abbreviations: fMRI, functional magnetic resonance imaging; EEG, electroencephalography; ERP, event-related potential; TBI, traumatic brain injury; tDCS, transcranial direct current stimulation.
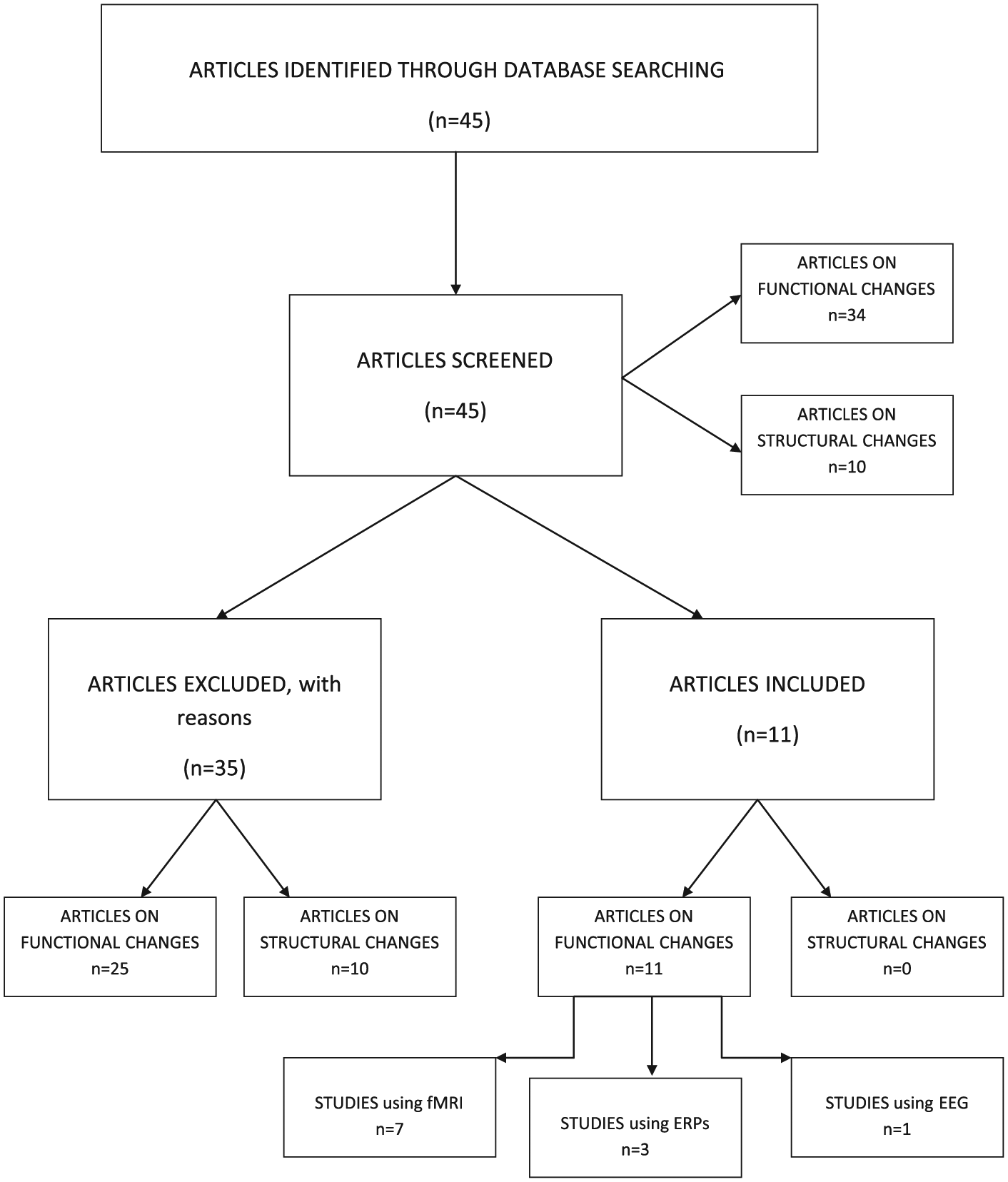
Flowchart of the studies retrieved from the literature search.
Results
The aim of this review was to collect all the studies focusing on the neural and neurophysiological modifications induced by cognitive training in TBI subjects. Eleven studies were found. Among these, none focused on the structural changes induced by cognitive rehabilitation. This may likely be due to the fact that this review dealt only with TBI individuals in the chronic phase of injury: in this case, short-lasting cognitive training (a few months or weeks) could not be sufficient to induce relevant modifications of brain structures. However, these issues deserves further analysis.
Attention Abilities
Mild to severe disturbances of attention are frequently reported after TBI and constitute one of the main barriers to social and vocational reintegration. 28 In a first fMRI study, Laatsch and colleagues 1 analyzed the neural and cognitive sequelae of a training on visual search and reading abilities in a subject with severe TBI. In spite of the absence of significant behavioral changes, the authors found a qualitative improvement in most of the trained abilities. Such an enhancement was accompanied by a neural reorganization, probably due to the augmented attention and accuracy during the task: neuroimaging results pointed out an increased activation in the cerebellum and in the posterior parietal regions bilaterally, as well as in the left occipital region. Furthermore, new activations in the right inferior parietal area were found.
The same results were replicated in another fMRI study by the same research group. 62 Once again, the 3 TBI participants underwent training sessions on attention, visual scanning, and reading abilities. Their cognitive improvements were accompanied by a significant change in the neural activation pattern that, although varying across subjects, mainly included the frontal, temporal, and parietal areas of the brain. Post- versus precomparisons highlighted a significant reduction in the pattern of activations seen in the first fMRI evaluation, leading to the involvement of a network that seemed to be more similar to that of healthy controls. Consistent with their previous outcomes, 1 the authors attributed these neural modulations to a higher ability to perform the attention tasks.
The findings of this study are in line with those of another fMRI experiment 64 on the behavioral and neural modifications induced by cognitive training in TBI subjects. In their sham-controlled study, the authors evaluated the efficacy of 10 tDCS (transcranial direct current stimulation) sessions combined with a computerized program in enhancing the divided attention abilities of TBI participants. Similarly to the findings by Laatsch et al, 62 posttreatment neuroimaging results highlighted a more functional and specific neural reorganization, resulting in a decreased activation in the areas subserving attention tasks, such as the left dorsolateral prefrontal cortex, the bilateral premotor and supplementary motor areas, the right parietal somatosensory cortex, and the right auditory processing cortex. These areas are involved in dual task processing in healthy subjects, 68 since they oversee the attention allocation, so reflecting the higher executive demand required in divided versus selective attention tasks.
A training-induced redistribution of attention network resources was also found in the study carried out by Kim et al on the efficacy of a computerized training on attention and short-term memory in a group of TBI subjects. 63 The fMRI outcomes highlighted an increased activation in the anterior cyngulate cortex, precuneus, and cerebellum. Interestingly, this study reported an improvement in attention performance accompanied by a diminished frontal activation. According to the authors, these outcomes suggest that intensive training may lead to a better and more automatic task execution that, in turn, results in a reduced activation in the areas normally engaged in the attention processes.
Other studies used neurophysiological measures to focus on the cerebral modifications induced by attention treatments. In an EEG study, Stathopoulou and Lubar 28 analyzed the changes in cerebral waves following attention training in a group of 5 TBI subjects. Converging evidence 69 suggests that higher attention deficits result in increased delta, theta, and alpha power, and decreased beta bands. So cognitive treatment usually aims at reducing the patients’ delta, theta, and alpha relative and absolute power, normally involved in restful situations, and increasing beta bands, usually active in cognitive tasks. Stathopoulou and Lubar 28 partially confirmed these hypotheses, even in the presence of improved attention for all the participants. The neurophysiological outcomes strongly varied among participants and seemed to be related to factors other than cognitive performance. The authors proposed that, in the presence of severe brain lesions, such as those caused by diffuse axonal damage, there may be multiple factors able to influence the neurophysiological brain responses, such as the area of brain injury, the time of injury, and age and gender.
More recently, Dundon et al 67 used ERPs to examine the efficacy of a treatment on auditory selective attention in a group of subjects with severe TBI. Improvements in auditory attention were obtained and were accompanied by increased P300, which most likely reflected the increased ability of the participants to focus on the target information, despite the distracting stimuli. According to many studies in the literature,70,71 an increased P300 signal in correspondence to the temporoparietal areas may index working memory and attentional operations necessary to process the target information under adverse noisy conditions.
Another ERPs study was conducted by Musiek et al. 65 This single case study was mainly focused on the treatment of auditory deficits in a subject with mild TBI. Nevertheless, the presence of concomitant cognitive deficits, especially involving attention, was acknowledged. This was according to the hypothesis that, even if most of the attention difficulties are caused by a damage in the frontal and parietal networks of the CNS, an inferior amount could be the consequence of a primary damage in the auditory system. Posttraining results confirmed this theory: when the auditory deficits diminished, the related cognitive disturbances also decreased. The neurophysiological correlates of these modifications included the following: increased amplitudes of the middle latency response for the Na-Pa wave, accompanied by a better waveform morphology across all electrode sites for both ears.
Also Pachalska et al. 66 used event-related potentials to assess the functional changes induced by rehabilitation programs in a TBI patient. Interestingly, in this study the traditional cognitive training was associated with EEG-neurofeedback and noninvasive brain stimulation: in the first part of the treatment a relative beta training was applied to activate the hypofunctioning areas of the frontal lobe, markedly involved in cognitive and behavioral control. The second part of the rehabilitation consisted in the application of 40 rTMS (repetitive transcranial magnetic stimulation) sessions, associated with cognitive and behavioral treatment. Posttraining results revealed a better recovery of attention, memory, and executive functions following rTMS treatment, while lower improvements were noticed after the use of neurofeedback. Similarly, the most statistically reliable changes in physiological parameters, including improvement of the P300 NOGO component, were shown after the rTMS program. On the contrary, the use of neurofeedback was not sufficient to induce significant neurophysiological changes.
Memory Abilities
In the same way as attention, memory difficulties need to be clinically treated, given their pivotal role in the patient’s recovery. The rehabilitation of mnestic difficulties, especially in subjects with frontal lesions and diffuse axonal damage, focuses mainly on abnormalities in the strategic aspects of memory, which are closely connected to executive functioning and prefrontal cortex. 32 This can be seen in the fMRI study by Chiaravalloti et al. 11 In their case-control experiment, the authors evaluated the efficacy of the Story Memory Technique, a training focusing on the use of strategies to facilitate new learning, in 9 subjects with severe TBI and mnestic deficits. Behavioral results showed increased learning abilities only in the experimental group, while control participants who underwent generic memory exercises did not improve their performance. Neurally speaking, posttraining outcomes highlighted a greater activation in the experimental group during performance of a memory task in the left posterior cingulate cortex, right anterior cingulate cortex, right medial temporal lobe, posterior insula, and left cerebellum. On the other hand, the activation of the supplementary motor area and the dorsolateral prefrontal area diminished after the training. Similarly to Kim and colleagues, 63 Chiaravalloti et al’s findings also suggest that, in response to rehabilitation, the brain displays an ability to reorganize more efficiently. This may result in a less diffuse pattern of cortical activity in certain areas, likely to be due to a diminished cognitive effort in performing the task in question, and in a redistribution of neural activities within other areas, such as in the cingulate cortex, the insula and the medial temporal lobe.
The role of the medial temporal lobe has been also explored by Caglio et al. 61 In their single case study the authors assessed the efficacy of a virtual reality training in reducing the memory deficits of a subject presenting with severe TBI. They hypothesized that improved mnestic abilities should be accompanied by increased activity in the medial temporal lobe, whose structures are fundamental for declarative or long-term memory. 72 In line with such a hypothesis, posttraining outcomes revealed that the improvement in visual-spatial memory learning was accompanied by an increased metabolism in the left hippocampus and right parahippocampal cortex, indicating, once again, that cognitive training may lead to a more functional neural modulation.
Communicative and Pragmatic Abilities
Besides attention and memory problems, another invalidating consequence of TBI is communicative and pragmatics difficulties. Conversations in TBI individuals are less interesting, less appropriate, more confused, and inefficient than those of non–brain-injured controls. 73 Studies analyzing the efficacy of cognitive treatment for the improvement of communicative abilities in severely brain injured subjects include that carried out by Sacco et al. 19 In their work, the authors investigated the behavioral and neural modifications induced by cognitive pragmatic treatment in a group of TBI participants. 74 Interestingly, the neuroimaging measure used in this study did not consist of task-related fMRI, as in the previous studies: a resting state paradigm was used to investigate neural modification occurring after rehabilitation sessions. The advantage of using resting state magnetic resonance is that, in contrast to task-based fMRI, it provides a complete identification of the brain network connectivity.3,50 Study findings showed that the improvement in communicative skills was accompanied by an increase in functional activity in a network of areas usually involved in pragmatic competence. These mostly included the bilateral precentral gyri, the right middle and superior temporal gyri, the right cingulate gyrus, and the left inferior parietal lobule. The increase in functional connectivity observed in this study is consistent with previous studies, suggesting a functional network remodeling after treatment, 50 caused by a compensatory mechanism leading to a more effective and functional information processing.
Discussion
This article includes a review of all the studies on the effectiveness of cognitive training carried out on TBI individuals, assessed through both cognitive tasks and neuroimaging or neurophysiological measures. Since the findings reported here involved patients with a wide range of severity of TBI and resultant cognitive disturbances, and given the different methods used to investigate the cerebral patterns of posttreatment activation, it is actually difficult to provide a coherent summary of results. However, some general trends emerged. With regard to the studies on neurophysiological modifications, a more functional response of the brain through cognitive training was described. Although these studies focused on different aspects, like sensorial modifications, 65 attention,28,67 or more generic cognitive and behavioral outcomes, 66 they all demonstrated that rehabilitation is useful in reducing the physiological disturbances due to diffuse brain injuries, such as reduced cortical signal to noise, disruption of the oscillatory rhythm, and increased performance variability. 26 In particular, Dundon et al 67 and Pachalska et al 66 showed that intensive cognitive training lead to increased amplitude of the P300 waves, index of more functional processing of the target stimulus: a decreased P300 component often observed in TBI survivors has been considered as an indicator of reduced attention processing, 75 which also persists a long time after the injury. Therefore, significant improvements in its amplitude may indicate a more functional and appropriate response to external stimuli.
The outcomes of the neuroimaging studies also highlighted the fact that cerebral activation may be significantly modified by cognitive rehabilitation, although most of this research was limited to very small sample sizes and was lacking in controls. 11 In general terms, cognitive rehabilitation seems to lead to a readjustment of the maladaptive mechanisms involved in the recovery process.62,63,76 Into the bargain, according to the “negative plasticity” framework by Mahncke et al 77 and Tomaszczyk et al, 10 a series of self-reinforcing and dysfunctional mechanisms are activated after acquired brain injury. This is probably due to a combination of reduced schedules of activity, a reduced quality of sensory-perceptual processing, and weakened neuromodulatory control. Taken together, these factors increase reliance on simplified cognitive processing, which, in turn, leads to “negative learning.” As a consequence, maladaptive brain changes emerge in performing more complex tasks, thus resulting in a reduced cognitive task performance. 78
A clear example of this process is the hyperactivation frequently reported after an episode of TBI, 79 where areas of overactivity are thought to reflect the unsuccessful efforts of a vicarial neuronal system to repair the damage found in other areas.32,46,63,80 As a result, the patient needs additional resources to solve the task executed in a less automatic way. A number of studies in the literature81,82 suggest that individuals with TBI may need to take on more extensive and distributed brain resources, especially in response to challenging conditions, in order to match the behavioral performance of control participants. The causes of this complex phenomenon may be different and vary substantially among subjects: it has been explained as brain reorganization,83,84 neural compensation, degeneracy, or poor regulation of neural resources. The findings of some studies in the literature85-87 interpreted this hyperactivation as a form of compensation, which facilitates task performance so that, in the absence of compensatory neural recruitment, a failure to perform or a poorer task performance would occur. Alternatively, it has been explained as a form of poor regulation of sources, leading to irregular or randomized involvement of various areas during task execution.88,89 Distinguishing among all these possible explanations may significantly improve the understanding of how neural activity is modified in performance and recovery after TBI. More work has to be carried out in correlating neuroimaging and behavioral results. In response to this, cognitive reorganization following rehabilitation occurs when a patient uses a different set of cognitive processes to perform the same task because she/he has learned a new cognitive procedure. Neurally speaking, this is reflected in a change in the task-specific neuronal architecture, which takes place during learning or relearning processes in the damaged brain, thus reducing dysfunctional connections. 46 In some of the studies considered in this review,1,11,61,63 successful functional recovery was accompanied by reduced patterns of activations, which could be interpreted as the result of reweighting of interactions within an existing network. 18
However, in the case of TBI, it seems somewhat limiting to describe functional recovery following cognitive treatment as the only result of a reduction of dysfunctional hyperactivations. Rather, it would seem more proper to refer to it as a redistribution of the neural network 63 where, in parallel to a reduction of the areas previously hyperactivated during the task, there may be an increased involvement of other neural circuits, usually active in healthy subjects. To this end, Caglio et al 61 described an increased metabolism in the left hippocampus and right parahippocampal cortex during a verbal task after a computerized training on spatial memory. In a similar vein, Chiaravalloti et al 11 reported that, in parallel with a decreased activation in the supplementary motor and dorsolateral prefrontal area, there was an increased activation of the left posterior cingulate cortex, right anterior cingulate cortex, and posterior insula. A higher activation of similar circuits, described as part of the default mode network, has also been reported by Sacco and colleagues in their article on communication, 19 thus showing that this network may be modified by cognitive rehabilitation, with consequent changes in functional connectivity. Although none of the studies analyzed in this review have specifically focused on the latter aspect, it is now widely established that a defective connectivity may significantly condition the cognitive performance of brain injured subjects50,90-92: diffuse axonal damage (DAI), typical of moderate to severe TBI, may cause axons fragmentation and tearing,93,94 leading to a diminished or interrupted integration of information processing across spatially distinct brain regions. Taking into account all of these aspects, the marked variability that emerges from the studies considered here, showing complex and sometimes contradictory patterns of functional network abnormalities and network readjustment after cognitive rehabilitation, is not surprising. The same extreme variability has been described by Laatsch et al, 62 who found highly heterogeneous fMRI results even among the 3 TBI patients participating in their cognitive training. According to the authors, studies on persons who have suffered TBI, besides producing interesting results, also impose uncontrollable factors, such as a modified functional connectivity, that may influence both neuropsychological results and fMRI activation patterns.
A final point of discussion is related to the role of noninvasive brain stimulation (NIBS) in cognitive rehabilitation. Since noninvasive and painless NIBS techniques, mainly associated with mild adverse effects, can modulate cortical excitability focally, they have been increasingly used in the clinical setting. 10 Both studies reported in this review64,66 highlighted the efficacy of NIBS methods in enhancing the outcomes of cognitive rehabilitation, also in the presence of more complex neurological and cognitive sequelae. In particular, the study by Pachalska et al 66 demonstrated the greater efficacy of rTMS in reducing higher order cognitive difficulties in severely brain-injured subjects. Similarly, according to the results obtained by Sacco et al, 64 only the combination of tDCS and cognitive training was able to induce significant improvements in divided attention in TBI participants. On the contrary, the cognitive treatment alone was not sufficient to improve the performance of controls. These findings are in line with other studies,38,77 highlighting the key role played by NIBS tools in increasing neuroplasticity and functional outcomes following brain injury.
As previously claimed, a number of limitations could have influenced the interpretation of the literature’s results. First, the limited number of studies make it difficult to clearly and definitively interpret the data, especially if we consider that most of them focused on different aspects of cognition and used different rehabilitation tools and tasks. Thus, the variability of the cognitive functions investigated could be considered as one of the main restraints of this article: all the studies focused on various aspects of memory, attention, or communication, so it was very difficult to find a common activation pattern among them. A further remarkable limitation of this study is that most of the analyzed research included a small number of participants, and in many cases, a control group was missing. What made the data interpretation even more difficult was that many studies included only single cases; therefore, it was harder to generalize the outcomes. These aspects have been further complicated by the etiology of the participants included in the review: in spite of its evident social impact and consequences, TBI is always linked to a misinterpretation of data, due to its broad range of neural and clinical consequences. Despite these limitations, the studies reported here demonstrate the efficacy of cognitive training in promoting neural reorganization following acquired brain injury. This in turn diminishes the risk of creating a spiral of negative neuroplastic changes deriving from the disuse of previously healthy neural structures leading to accelerated aging, atrophy, and white matter alterations, which are frequently reported in the chronic stages of moderate-severe TBI.
Footnotes
Acknowledgements
We thank Christian Jennings for his careful language revision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by local research funds (ex60%) managed by University of Turin, Department of Psychology, year 2016.
