Abstract
Background
Participation restrictions following traumatic brain injury are associated with executive function (EF) deficits (EFDs). The subacute recovery phase’s specific characteristics (enhanced brain plasticity and impaired self-awareness) and contextual factors (inpatient setting) warrant adjusting cognitive rehabilitation protocols. The Intervention of Participation and Executive Functions (I-PEX) was designed to improve EFDs during subacute inpatient rehabilitation.
Objective
To investigate the I-PEX’s preliminary efficacy to improve EFDs during the performance of complex daily activities and enhance self-awareness, cognitive self-efficacy, participation, and quality of life postdischarge.
Methods
A pilot pre-, post-, and follow-up double-blind randomized controlled trial with 25 participants randomly allocated to the I-PEX (n = 13) or treatment-as-usual (n = 12) group. Cognitive assessments were administered pre- and postintervention, and quality of life and participation questionnaires 1-month postdischarge. Data analysis included repeated measures analysis of variance mixed design and independent t-tests, extracting effect sizes.
Results
Significant group-by-time interaction effect with a medium effect size was found for the primary outcome measure; EFs manifested in complex daily activities, indicating a larger improvement for the experimental group. The group effect was not significant. The experimental group’s mean delta score (pre–post improvement) was significantly higher (1.75 ± 2.89; t(23) = 2.52, P = .019), with a large effect size (d = 1.012, 95% confidence interval [0.166-1.840]). We found no significant group and interaction effects for EFs, self-awareness, and cognitive self-efficacy or no significant differences in participation or quality of life postdischarge.
Conclusions
Results provide initial evidence for the I-PEX efficacy in treating EFDs in the subacute phase and could help determine effect size for future studies.
Clinical Trial Registry Number:
ClinicalTrial.gov NCT04292925.
Keywords
Introduction
Traumatic brain injury (TBI) frequently results in lifelong disability, especially among young adults.1,2 One common reason for long-term disability and participation restrictions is executive function (EF) deficits (EFDs).3,4 Post-TBI, problems with EFs, such as monitoring, initiation, planning, organization, cognitive flexibility, and working memory, have been associated with participation restrictions.5,6 They affect the ability to return to pre-injury participation in complex daily activities, including employment, leisure, social, and instrumental activities of daily living (IADL).7,8
Post-TBI recovery is roughly divided into acute, subacute, and chronic phases; a lengthy rehabilitation process is often required. 9 Currently, cognitive rehabilitation for post-TBI EFDs integrates approaches addressing multiple EF aspects, metacognitive knowledge, and self-regulating strategies. 10 Results appear enhanced when patients have some level of self-awareness.11,12 Cicerone et al 10 recommend metacognitive-strategy training as a practice standard in post-TBI subacute rehabilitation. Jeffay et al 11 recommend conducting it at least 6 months post-TBI, likely due to the limited research examining EF interventions in the subacute phase.13-16 Both recommend strategies that focus on functional outcomes personally relevant to patients.
The subacute recovery phase typically ranges between 3 and 6 months postinjury. 17 Developing and examining novel intervention protocols’ efficacy in the subacute phase are warranted in light of 2 characteristics distinguishing this phase and affecting cognitive rehabilitation: enhanced brain plasticity and high rates of impaired self-awareness.18,19 Brain plasticity in this phase combines spontaneous and rehabilitation-induced plasticity.20,21 This, and the high-intensity treatment usually used during the subacute phase, may present an opportunity to improve cognitive impairments (EFDs).22,23 Impaired self-awareness may negatively affect patients’ task engagement, cognitive effort, and commitment to goal attainment, compromising their ability to participate actively in and benefit from cognitive rehabilitation.18,24
In the subacute recovery phase, the inpatient setting can significantly affect the cognitive treatment process. During inpatient rehabilitation—in and outside treatment sessions—patients engage in activities similar to community-based activities but with different scales and complexities. These activities include IADL (eg, making cafeteria purchases, arriving on time for treatments), leisure–social (eg, maintaining relationships with other patients), and multiple open-ended, meaningful activities. We suggest inpatient participation to describe this multidimensional participation. Similar to the International Classification of Functioning, Disability and Health (ICF), 25 highlighting interrelationships between body functions and participation, we suggest a reciprocal relationship between inpatient participation and EFs: Each reflects and affects the other. Therefore, we hypothesize that inpatient participation can be used to train EFs and become a short-term cognitive-rehabilitation goal.
A recent cohort study found that increasing contextualized (real-life context) treatment activities during inpatient rehabilitation can improve rehabilitation outcomes, specifically, postdischarge community-based participation. 22 This suggests that incorporating inpatient participation as central to intervention protocols explicitly for inpatients in the subacute phase may improve long-term outcomes. Another subacute-phase aspect found to improve outcomes and encourage recovery is increasing the proportion of challenging EF-targeted treatments. 23 Given the nature of brain plasticity at this phase, these results indicate that better rehabilitation outcomes might be achieved with treatment protocols targeting EFs and systematically and progressively challenging the patient.
The Intervention of Participation and Executive Functions (I-PEX) is a novel, client-centered intervention protocol developed and designed for the unique needs and characteristics of the subacute phase post-TBI. This contextualized treatment protocol systematically targets EFs with inpatient participation at its core. Considering the ICF model, 25 the I-PEX focuses on interrelationships between EFs and participation in complex daily activities, specifically, how EFs are applied in daily participation in the current context. The I-PEX aims to improve underlying cognitive impairment (EFDs), gradually reconstruct self-awareness, and promote inpatient participation. It may have the potential to improve cognitive rehabilitation outcomes and better prepare patients for broader community-based participation postdischarge. It is based on the retraining model, a theoretical model developed for cognitive treatment during the subacute phase. 26 The model emphasizes training underlying cognitive components to improve and develop the cognitive process rather than circumvent the deficit. It stresses increasing the patient’s awareness of the improved abilities and strategy use. The task difficulty increases according to the patient’s improving abilities while considering timing, functional potential, and rehabilitation dynamics.
This pilot randomized controlled trial (RCT) aimed primarily to investigate the I-PEX’s preliminary efficacy for improving EFDs in performing complex daily activities by adults hospitalized in inpatient rehabilitation facilities post-TBI. Secondary aims were to evaluate its efficacy in enhancing EFs, self-awareness, and cognitive self-efficacy; examine its efficacy in promoting participation and quality of life 1-month postdischarge; and collect data for future RCTs to calculate sample size.
Methods
Study Design
This pre–post double-blind (assessors/participants) pilot RCT was conducted in an inpatient rehabilitation medical center. Participation and quality of life measures were administered 1-month postdischarge. The Loewenstein Rehabilitation Medical Center Review Board (#0028-18-LOE) and the University of Haifa Ethics Committee (#161/19) approved the study protocol. All participants provided written informed consent.
Participants
The study sample comprised 25 participants hospitalized in an inpatient rehabilitation medical center. The experimental group (n = 13) received treatment using the I-PEX only; the control group (n = 12) received treatment-as-usual only. Inclusion criteria were a physician’s diagnosis of moderate-to-severe TBI confirmed by imaging (per medical records); age 18 to 70 years; able to understand assessment-tool instructions; at least 1 functional upper extremity (ie, could use hand for daily activities like eating), determined by their occupational therapist; preserved basic cognitive abilities, determined by 3 domains of the Loewenstein Occupational Therapy Cognitive Assessment: orientation (scoring 6+/8), visual perception (3+/4), and spatial perception (2+/4); and intact or corrected vision. Participants with histories of epilepsy, drug use, or psychiatric or neurologic disorders affecting cognitive function were excluded.
Procedures and Randomization
The medical records of each patient admitted to the TBI department were screened for preliminary eligibility. Patients meeting the moderate-to-severe TBI and age criteria but no exclusion criteria were randomized to either I-PEX or occupational therapy treatment-as-usual. Allocation was concealed (sequentially numbered, sealed envelopes). An independent researcher, uninvolved in the interventions or data collection, conducted randomization using blocks of 4 (ratio 1:1).
The TBI rehabilitation department commonly assigns a specific clinician to each patient upon admission. Because 3 department clinicians were trained on the I-PEX and treatment-as-usual, and 4 provided only treatment-as-usual, we randomized before the baseline assessment (after preliminary eligibility) to ensure patients could be assigned to a specific clinician based on their study group. Thus, we avoided needing to change clinicians after enrolling the patients. Then, participants underwent the baseline cognitive assessment, including performance-based and neuropsychological assessments and self-awareness and self-efficacy questionnaires. Assessors were experienced occupational therapists working in rehabilitation facility departments other than the TBI department; all were unfamiliar with the participants. Before data collection, the first author provided the assessors with 3 to 4 training sessions (1 hour each) on administering the assessments.
Following baseline assessment, patients (blinded to their group allocation) underwent individualized treatment sessions (45 minutes each) 3 to 5 times a week for 3 to 6 weeks according to their group protocol (variations due to coronavirus disease-2019 [COVID-19] restrictions). Each received only 1 cognitive treatment—I-PEX or treatment-as-usual; all received care (eg, occupational therapy not cognition-focused, physical therapy, or speech–language therapy) per the department’s standard practice. Patients retook the baseline assessments and questionnaires postintervention and completed participation and quality of life questionnaires via telephone 1-month postdischarge. Demographic and diagnostic data were collected from medical records and self-reports.
Contamination was minimized by ensuring clinicians administering treatment-as-usual were unfamiliar with the IPEX and carefully examining treatment adherence by the clinicians administering the IPEX.
Interventions
Intervention of Participation and Executive Functions
The I-PEX comprises independent modules for 5 EF areas that influence daily participation but are typically impaired post-TBI 27 : initiation, cognitive flexibility, monitoring, planning, and problem-solving.6,28 Initiation, cognitive flexibility, and monitoring are core EF components thought to be the basis of higher-level planning and problem-solving.29,30 The I-PEX addresses 1 EF at a time based on the clinician’s identification of which is most affecting the patient’s daily functioning. Each module is systematically structured to facilitate the patient’s gradual learning and awareness of concepts related to the specific EF. It also promotes self-awareness while incorporating metacognitive strategy elements, such as self-monitoring and feedback, into future performance. 11 Patients come to understand how the EF manifests in daily participation, when and why it is needed, recognizing when it is applied during a task, strategies assisting its execution, and applying this knowledge in inpatient participation.
The I-PEX provides a structure for the treatment process and each module and session. The treatment-process structure is based on 3 learning stages described in several learning theories (acquiring, consolidating, and overlearning knowledge) 31 and 3 training phases (initial acquisition, mastery and generalization, and maintenance) Sohlberg and Turkstra 32 described. Each treatment session focuses on self-generating strategies targeting the EF component.26,33 Key features for promoting self-awareness include comparing the patient’s predictions to actual performance, error detection, and corrections.32,34
Each specific module’s treatment process consists of 3 learning stages, each requiring several sessions, depending on the patient’s learning ability: (1) foundation—learn basic concepts related to the specific EF and its role in daily activities; (2) assimilation—practice and acquire the EF skill in structured daily activities; and (3) implementation—learn to transfer skills acquired in the first 2 stages to inpatient participation activities within the rehabilitation facility.
Each session follows a consistent structure in all 3 stages, promoting gradual, systematic learning of EF concepts and their roles in inpatient, home, and community participation. The first part involves an EF-focused discussion to define and relate the EF to everyday functions and familiar activities currently and before the injury. The second part, task performance, includes pre-, task-, and postperformance analyses. The clinician and patient discuss the task and its instructions and demands—how the specific EF would manifest in the task, appropriate strategies for efficient performance, and so forth. The patient performs the task, followed by a postperformance analysis. The clinician and patient summarize and discuss, for example, challenges, the EF’s role in the performance, and whether the strategies helped. The third part is an EF–participation discussion to connect learned EF content with everyday functions in the rehabilitation facility, home, and community.
In each stage, the discussion, analysis, and task complexity are adapted to the patient’s independence, awareness, and understanding in performing the task. The clinician must actively observe the performance throughout each treatment session to identify factors such as learning patterns, strategies used, and understanding during the discourse.
The I-PEX is designed to allow maximum flexibility. Because post-TBI patients vary greatly in EFDs and learning abilities, clinicians can adapt the I-PEX contents and progress rate to each patient’s condition and learning capacity. An intervention manual describing the treatment process structure through the learning stages guides clinicians.
Treatment-As-Usual
Standard treatment in the TBI department is mainly a decontextualized intervention adjusted to each patient’s abilities. Aiming to strengthen cognitive function, considered the basis for performing real-life activities, 22 it emphasizes remedial improvement of EF components overall rather than individual EFs. It includes didactic tasks (eg, computerized and pen-and-paper exercises) in a structured environment. It also focuses on cognitive strategies during pen-and-paper tasks simulating community-based functional activities (eg, organizing a daily schedule according to a predetermined chore list or managing a household budget according to defined constraints). During treatment sessions, the clinician provides feedback and mediation to promote effective task performance, raise awareness, and instruct on transferring strategies to daily life.
The I-PEX differs from treatment-as-usual in its consistent, systematic intervention structure focusing on 1 module at a time but integrating them all as the treatment progresses. It addresses how EFs guide the performance of complex daily activities and inpatient participation.
Measures
Psychometric properties of measures used in this study are provided in the Supplemental Material. Primary and secondary outcomes were administered pre- and post-intervention.
Primary Outcome
The Multiple Errands Test-Hospital Version (MET-HV) 35 assessed EFs as they manifest in complex daily activities. This performance-based assessment evaluates the effect of EFDs on everyday functioning in a hospital environment. It consists of tasks like running errands or obtaining information in a hospital setting. The final score (0-36) is based on errors in 4 categories—inefficiencies, rule breaks, interpretation failures, and task failures; lower scores indicate better performance. 36 Completion time is measured in minutes from when the assessor instructs the participant to begin to when the participant says they completed the task. There is evidence for its reliability and validity for adults with acquired brain injury 35 ; different versions were used as outcome measures in previous studies examining the effectiveness of EF interventions.13,37,38
Secondary Outcomes
The WebNeuro, a web-based computerized neuropsychological test battery, assessed EFs. 39 Subtests were the Digit Span for working memory (number of digits sequence); Trail-Making Test for cognitive flexibility and inhibition (completion time); Maze Test for monitoring, planning, and problem-solving (completion time, learning time, and number of errors); and Go-No-Go for inhibition (number of errors). The assessment battery scores are raw and standardized z-scores adjusted for age and gender.
The Self-Perceptions in Rehabilitation Questionnaire (SPIRQ) 40 assessed self-awareness in rehabilitation. Its 20 items are divided into 4 mini-scales: current-self, future-self, self-in-rehabilitation, and emotional reactions. Each item is rated on a 6-point Likert scale from 1 (strongly disagree) to 6 (strongly agree). Higher scores reflect greater awareness. Test–retest reliability and internal consistency were acceptable. 40
The Cognitive Self-Efficacy Questionnaire II 41 assessed cognitive self-efficacy for managing cognitive symptoms. Items are rated on a 10-point scale. Mean scores range from 0 to 10; higher scores indicate better cognitive self-efficacy for managing cognitive symptoms.
A satisfaction questionnaire was developed for this study to assess participants’ satisfaction with the intervention. It comprises 13 questions using a Likert scale from 1 (completely unsatisfied) to 5 (completely satisfied). Mean scores were calculated and used.
1-Month Postdischarge Outcomes
The Mayo-Portland Adaptability Inventory-4 (MAPAI-4) 42 participation index assessed participation 1-month postdischarge. It contains 8 items with ratings from 0 (no problem) to 4 (assistance required). T-scores were generated according to the questionnaire manual’s instructions 43 ; lower t-scores indicate better outcomes.
The Quality of Life after Brain Injury questionnaire (QOLIBRI) 44 assessed quality of life 1-month postdischarge. This questionnaire comprises 37 items generating 6 scores: 4 satisfaction (cognition, self, daily life, and autonomy and social relationships) and 2 bothered (emotions and physical problems) scales. Its overall scale comprises 6 items assessing overall life satisfaction. Each item’s response is scored from 1 (not at all) to 5 (very). The sum of all items is converted to a percentage: 0 represents the lowest possible, and 100 represents the best possible health-related quality of life.45,46
Screening Measure
The TBI department administers the Loewenstein Occupational Therapy Cognitive Assessment to all patients upon admission to screen their cognitive abilities. This measure was designed to provide a preliminary cognitive profile for patients with acquired brain injury. 26 We used its orientation, visual, and spatial perception cognitive domains to screen for the TBI group. Its psychometric properties were established in various populations. 47
Basic Functional Ability
The Functional Independence Measure is commonly used in TBI departments to assess basic ADL in inpatient rehabilitation programs. It comprises 18 items (13 motor/5 cognitive). Each item’s score ranges from 1 to 7, and final (sum of items) scores range from 18 to 126; higher scores indicate more independence. 48 It is a valid measure of functional independence for individuals with TBI. 49
Statistical Analysis
We examined data normality with Shapiro–Wilk tests, skewness, and kurtosis; most variables distributed normally. Combining these findings with evidence of analysis of variance (ANOVA)’s robustness when the sample size was about 12 in each group, we used IBM SPSS (version 25) parametric statistical tests relating mostly to effect sizes. 50 Because this was a preliminary pilot study, we did not calculate power. 51 Participant characteristics in both groups were summarized using mean, standard deviation, or percentages. We performed mixed-design, repeated-measures ANOVA with group as the between-subject and time as the within-subjects factor to examine the IPEX’s preliminary efficacy in primary and secondary outcomes. We calculated partial eta squares (ηp2) and interpreted them as small (0.02-0.13), moderate (0.13-0.26), and large (>0.26) effect sizes. 52 For significant interaction effects, we performed additional analysis of within- and between-group comparisons using paired and independent t-tests, respectively, extracting Cohen’s effect size (d) and 95% confidence interval (CI). We interpreted d values as large (>0.80), medium (0.50-0.20), or small (<0.20) effects. 53
When preintervention assessment data were available, we used intention-to-treat analysis for patients who did not complete the study’s protocol. To do so we imputed the last observed data. 54
Results
Participant Characteristics
The Consolidated Standards of Reporting Trials diagram (Figure 1) shows the participant flow from screening to postdischarge follow-up. Between June 2020 and May 2022, 268 adults consecutively admitted to the inpatient rehabilitation facility with TBI diagnoses were screened; 32 were assessed as eligible, and 7 declined to participate. Twenty-five were randomized to the experimental (n = 13) or control (n = 12) groups.
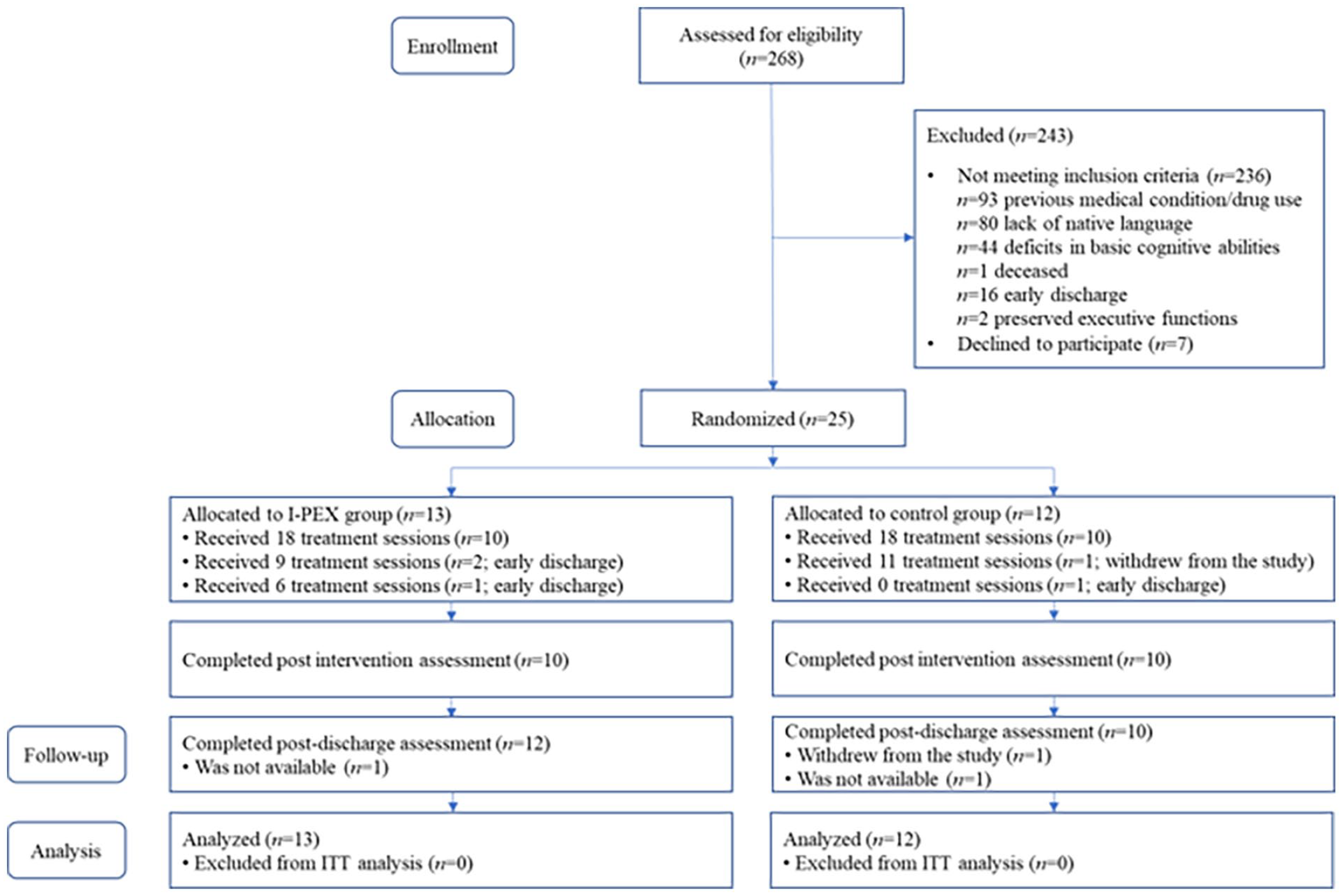
CONSORT diagram.
Table 1 presents demographic data, injury-related characteristics, and number of treatment sessions by group. No significant between-group differences were found in the demographic or injury-related data, number of treatments, or primary and secondary outcome baseline measures.
Participant Demographics and Characterization.
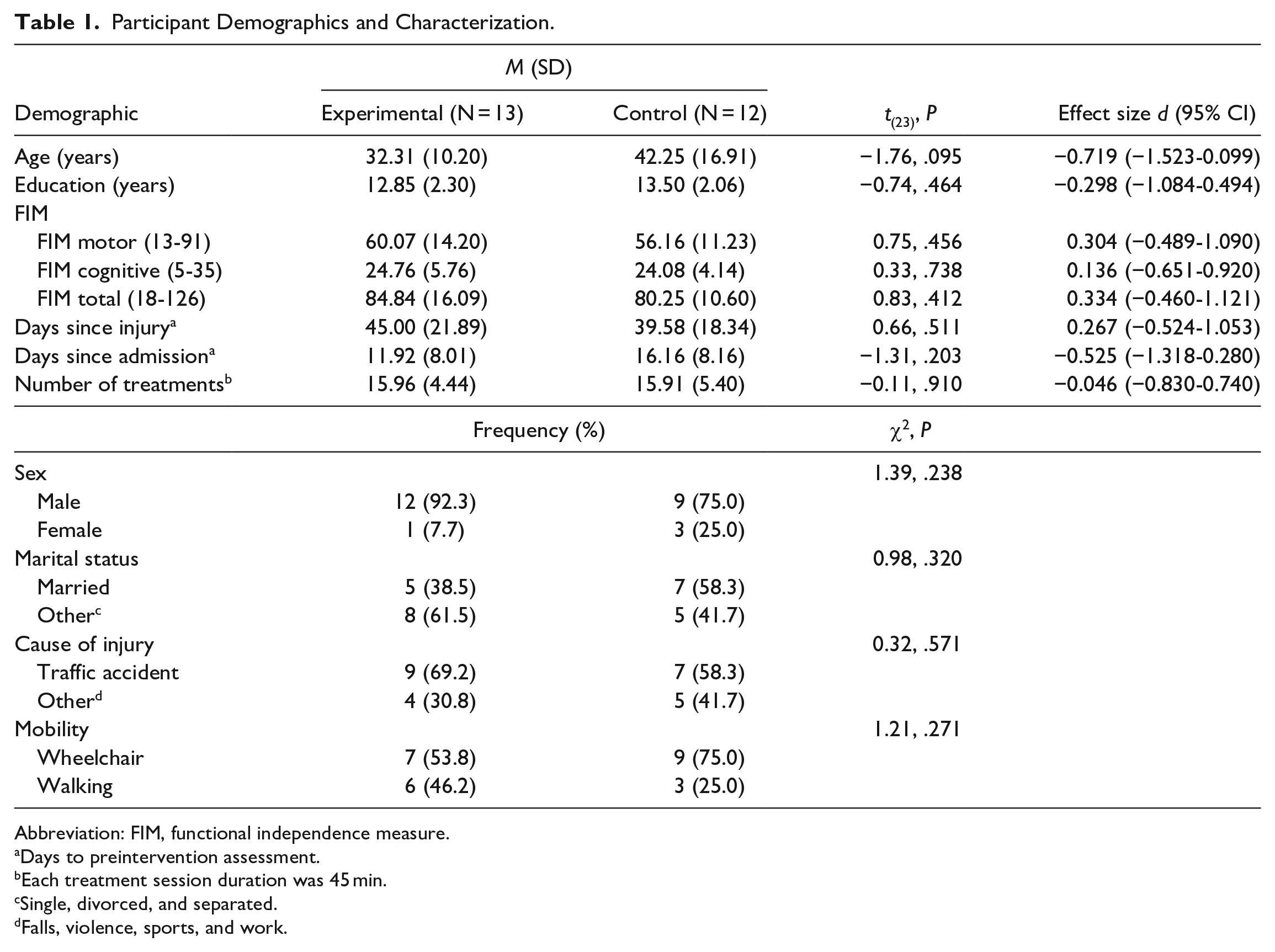
Abbreviation: FIM, functional independence measure.
Days to preintervention assessment.
Each treatment session duration was 45 min.
Single, divorced, and separated.
Falls, violence, sports, and work.
Intervention Fidelity
Clinicians administering the I-PEX received an intervention manual developed for the study describing the treatment course over the 3 learning stages. It included guidelines for each EF module, specifying instructions and examples for task selection, pre- and post performance discussion, and mediation level and type. The first and second authors (I-PEX developers) consulted when necessary and monitored each session to ensure adherence (each module’s stage/session structure). Clinicians administering the control group intervention were unfamiliar with the I-PEX and provided treatment-as-usual.
Intervention Effects
Primary Outcome
Table 2 presents the MET-HV results. Significant time and group effects by time interaction were found for the final score, but the group effect was not significant. The effect size was medium (ηp2 = .218) for group-by-time interaction. Between-group differences were analyzed using t-tests for the mean delta score (pre–post) to clarify the interactions. The experimental group’s mean delta score (5.30 ± 3.99) was significantly higher than the control group’s (1.75 ± 2.89; t(23) = 2.52, P = .019), with large effect size (d = 1.012, 95% CI [0.166-1.840]). Paired t-tests (within group analysis) showed that the experimental group significantly improved, t(12) = 4.78, P < .001, with large effect size (d = 1.328, 95% CI [0.558-2.069]); no significant improvement was found in the control group, t(11) = 2.09, P = .06, d = 0.604, 95% [CI −0.025-1.211]). A significant time effect was found in completion time, with large effect size (ηp2 = .273), but group and group-by-time interaction effects were not significant with small effect sizes.
Descriptive Statistics and Results of Within- and Between-Parametric Group Comparisons Pre- and Postintervention.
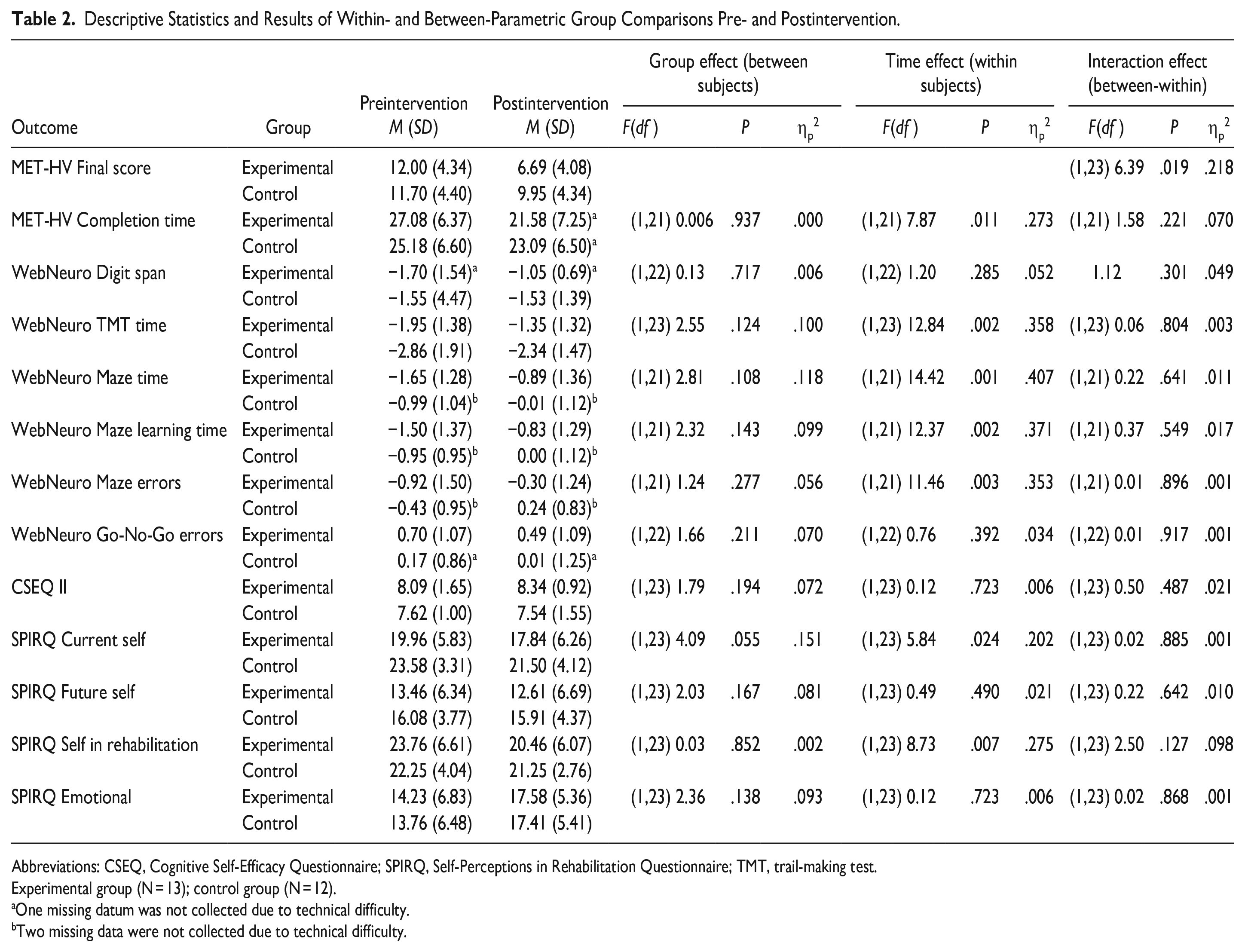
Abbreviations: CSEQ, Cognitive Self-Efficacy Questionnaire; SPIRQ, Self-Perceptions in Rehabilitation Questionnaire; TMT, trail-making test.
Experimental group (N = 13); control group (N = 12).
One missing datum was not collected due to technical difficulty.
Two missing data were not collected due to technical difficulty.
Secondary Outcome
Table 2 also presents secondary outcomes. No significant group or group-by-time interactions were found for any of the secondary outcomes. In the WebNeuro, significant time effects with large effect sizes were found in the Trail-Making Test completion time (ηp2 = .358), Maze Test completion (ηp2 = .407) and learning (ηp2 = .371) times, and number of errors (ηp2 = .353). In the SPIRQ, significant effects of time were found only in the current-self mini-scale with medium effect size (ηp2 = .202) and self-in-rehabilitation mini-scale with large effect size (ηp2 = .275).
Mean scores on the intervention-satisfaction questionnaire (only those who remained until the end of the study) were 3.91 (±1.20) for the experimental group and 4.10 (±0.67) for the control group, with no significant between-group differences, t(20) = −0.44, P = .662, and small effect size (d = −0.190, 95% CI [−1.029-0.654]).
Table 3 presents the MAPAI-4 participation index, QOLIBRI scores, and between-group analysis. There were no significant between-group differences despite low-to-medium effect sizes in favor of the experimental group.
Descriptive Statistics and Results of Between-Parametric Group Comparisons Postdischarge.
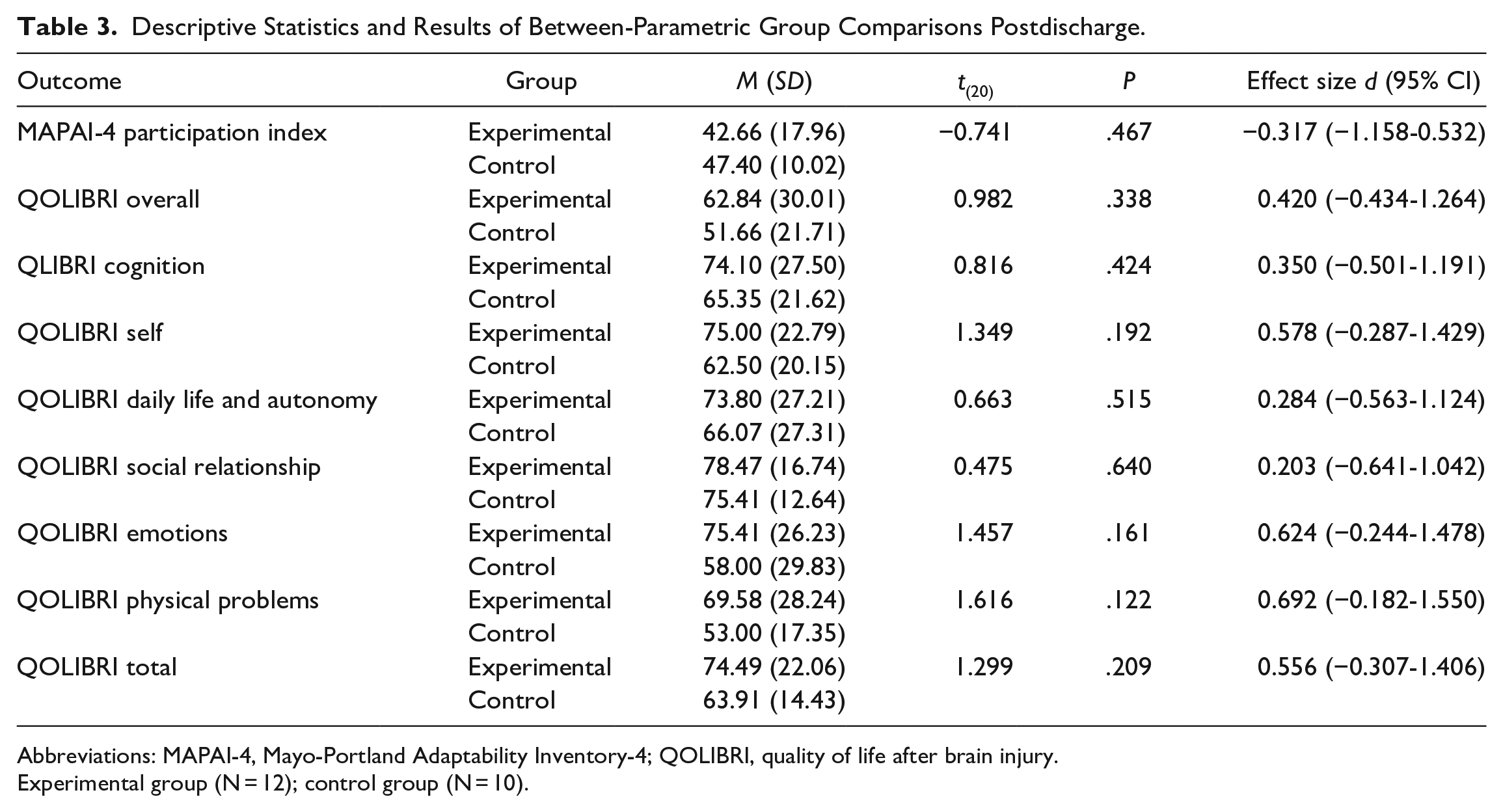
Abbreviations: MAPAI-4, Mayo-Portland Adaptability Inventory-4; QOLIBRI, quality of life after brain injury.
Experimental group (N = 12); control group (N = 10).
Discussion
This study examined the preliminary efficacy of the I-PEX, a novel intervention protocol for treating EFDs during the inpatient rehabilitation subacute recovery phase post-TBI. A significant group-by-time interaction was found in the primary outcome (MET-HV final score), indicating larger improvement in the experimental group with a medium effect size. No significant between-group or interaction effects were found for any secondary outcome. Within-group analyses showed significant improvement in both groups’ EFs and some self-awareness aspects.
The MET-HV final-score results might suggest initial evidence that the I-PEX could improve EFs manifested while performing complex daily tasks. As expected, MET-HV completion time did not improve following the I-PEX intervention, given that processing speed is not an aim of the I-PEX. Nevertheless, it might suggest that performance was more effective postintervention because participants performed better within the same timeframe. The MET was recently reviewed as having limited test–retest reliability. 55 However, studies using different MET versions as outcome measures concluded that improvements indicated generalization, transfer, and increased self-monitoring.13,37,38
The nature of the I-PEX may explain the improvements seen in our study’s experimental group. This contextualized treatment systematically uses inpatient participation in complex daily activities like scheduling and attending appointments with the social worker, arriving in the treatment room during free time, and working independently. Establishing consistent links between treatment content and everyday function in each session may encourage transfer and generalization. These results could also indicate that by contextualizing training of various EFs, the I-PEX might provide patients with a base to improve performance in complex daily tasks. Attention and visuospatial memory may have improved spontaneously during the study, possibly contributing to the MET-HV final score. 56
The neuropsychological battery results indicate that both groups improved in underlying EFs (eg, monitoring, cognitive flexibility, and problem-solving) postintervention, with spontaneous recovery possibly contributing to both groups’ gains.20,21 The I-PEX addresses each EF component, implements component-specific strategies, and systematically constructs the learning process with inpatient and actual participation tasks. Perhaps, in so doing, the I-PEX promoted the patients’ underlying EFs and ability to perform complex daily activities, consequently improving daily function. 57 The experimental group’s improved performance of complex daily activities and neuropsychological tests corresponds with previous studies.58,59 The control group’s improved EFs might be attributed to the treatment-as-usual emphasizing remedial improvement and training cognitive strategies. Unlike the experimental group, the control group’s improvement was not apparent when performing complex daily activities.
There are 3 possible explanations for why the between-group differences were less pronounced. First, spontaneous plasticity might have contributed to both groups’ demonstrated improvement. Future studies should consider imaging to explore spontaneous and rehabilitation-induced plasticity’s contribution to improved EFs and daily function following cognitive intervention. Second, the I-PEX is structured so that clinicians identify the EFD most affecting daily participation, which may be challenging because the 5 EFs may not be functionally separable. Being a preliminary study and the first time clinicians used the I-PEX for cognitive treatment may have contributed to the experimental group not seeing greater improvements. Future studies could refine the I-PEX manual instructions by examining clinicians’ experiences and reasoning in choosing an EF module. Third, both groups received cognitive treatment from experienced clinicians. The rehabilitation facility has used the treatment-as-usual for many years; it is based on theoretical knowledge and vast clinical experience in cognitive rehabilitation. Although it is mainly decontextualized, the clinicians instruct participants in transferring the learned content to daily life, the foundation of occupational therapy practice. Therefore, treatment-as-usual was probably effective, leading to less pronounced between-group differences.
Neither group’s self-awareness or cognitive self-efficacy improved, consistent with previous studies examining cognitive intervention effects on post-TBI self-awareness.60,61 This result might be due to the outcome measures we chose for examining the I-PEX efficacy in promoting self-awareness and cognitive self-efficacy—participant-only self-reports. In retrospect, these measures may have generated inaccurate responses. The higher proportion of impaired self-awareness during the subacute phase 20 may have led participants to overestimate their preintervention abilities but be more realistic postintervention, ultimately resulting in similar scores. Further, these questionnaires measure general—not task-specific—self-awareness and cognitive self-efficacy and rely on patients’ metacognitive knowledge. Self-awareness (metacognition) improves with time (first year postinjury). 19 Thus, interventions during the subacute phase may not improve metacognition, especially in a short-period study like this. Future studies might consider measuring self-awareness (specifically, online awareness) pre- and postintervention and at follow-up with more objective assessment tools and perhaps clinicians’ and caregivers’ knowledge rather than solely patients’ self-reports. 62
Postdischarge health-related quality of life and participation did not significantly differ between groups. Both group scores fell within normal ranges,45,63 respectively indicating medium and small effect sizes in favor of the I-PEX group. Systematically incorporating daily participation in the context of inpatient rehabilitation possibly promoted better reintegration into daily participation postdischarge, manifesting as health-related quality of life within norms. The lack of significant differences between the groups might have resulted partly from the small sample and from examining health-related quality of life and postdischarge participation with questionnaires administered 1-month postdischarge—possibly insufficient time to understand the injury’s consequences fully. Future research should consider evaluating participants’ actual performance, administering questionnaires to caregivers, and using larger sample sizes and longer postdischarge time intervals.
Limitations
This study’s small sample size limits generalizing results. The size was based on 2 reasons: First, recruitment occurred during the COVID-19 pandemic’s lockdowns, restricting admission to patients requiring complex care, usually with more severe injuries. Patients who might have otherwise been eligible to participate were not admitted or discharged early. Second, the study demands dictated strict inclusion criteria (eg, maintaining attention for up to 60 minutes for pre- and postassessments) that are strenuous for patients in early recovery phases. Thus, the study did not include patients who would have otherwise been suitable for I-PEX treatment.
The next development-process step to strengthen the I-PEX further, theoretically and clinically, is examining the feasibility of the I-PEX in clinical settings. This step focuses on therapists’ and patients’ experiences and perceptions of the I-PEX and developing methods to enhance self-awareness. The results of this pilot RCT may be used for sample size calculations for future full-scale RCTs. Further exploration of the theoretical underpinnings of the I-PEX is an additional future direction.
We lacked information about TBI severity (eg, Glasgow Coma Scale) and loss-of-consciousness duration due to technical issues in information transfer between the acute hospital and rehabilitation facility. Other limitations were using the MET-HV as a primary outcome measure, which has limited evidence regarding its test–retest reliability, and we did not establish its inter-rate reliability in the current study. Additionally, we did not perform power calculations because this was a preliminary pilot study. We did not account for other treatments that might affect cognition during the study, such as speech–language therapy. Finally, we did not examine treatment benefits in other daily function areas, such as leisure or ADL.
Conclusions
This pilot RCT provides initial findings supporting the I-PEX’s feasibility and efficacy in treating EFDs manifested while performing complex daily tasks in the subacute phase post-TBI. Treatment using the I-PEX could potentially improve underlying EFDs, possibly enhancing daily function. The high satisfaction rate suggests that participants viewed the intervention as contributing to their rehabilitation and everyday function. Plausibly, systematically integrating inpatient participation into the intervention increases patient engagement in the rehabilitation process. It relates directly to their daily participation, making the treatment more tangible. The I-PEX usability, including efficacy for long-term community-based participation post-TBI and other brain injuries, needs further examination. This study’s moderate effect size can be considered when planning future studies to assess the effectiveness of the I-PEX.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241231529 – Supplemental material for Efficacy of Intervention of Participation and Executive Functions (I-PEX) for Adults Following Traumatic Brain Injury: A Preliminary Pilot Randomized Controlled Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683241231529 for Efficacy of Intervention of Participation and Executive Functions (I-PEX) for Adults Following Traumatic Brain Injury: A Preliminary Pilot Randomized Controlled Trial by Rotem Eliav, Yael Nadler Tzadok, Shir Segal-Rotenberg and Rachel Kizony in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Author Contributions
Rotem Eliav: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Project administration; Visualization; Writing—original draft; and Writing—review & editing. Yael Nadler Tzadok: Conceptualization; Visualization; and Writing—original draft. Shir Segal-Rotenberg: Data curation and Investigation. Rachel Kizony: Formal analysis; Methodology; Supervision; Writing—original draft; and Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Loewenstein Rehabilitation Medical Center under grant number KM600010303.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
