Abstract
Background. An increasing number of studies have indicated that the ipsilesional arm may be impaired after stroke. There is, however, a lack of knowledge whether ipsilesional deficits influence movement performance during purposeful daily tasks. Objective. The aim of this study was to investigate whether, and to what extent, movement impairments are present while performing an ipsilesional upper extremity task during the first 3 months after stroke. Methods. Movement kinematics describing movement time, smoothness, velocity, strategy, and pattern were captured during a standardized drinking task in 40 persons with first-ever stroke and 20 controls. Kinematics were measured early and at 3 months poststroke, and sensorimotor impairment was assessed with Fugl-Meyer Assessment in stroke. Results. Half of the ipsilesional kinematics showed significant deficits early after stroke compared to controls, and the stroke severity had a significant impact on the kinematics. Movements of the ipsilesional arm were slower, less smooth, demonstrated prolonged relative time in deceleration, and increased arm abduction during drinking. Kinematics improved over time and reached a level comparable with controls at 3 months, except for angular velocity of the elbow and deceleration time in reaching for those with more severe motor impairment. Conclusions. This study demonstrates that movements of the ipsilesional arm, during a purposeful daily task, are impaired after stroke. These deficits are more prominent early after stroke and when the motor impairment is more severe. In clinical studies and praxis, the use of less-affected arm as a reference may underestimate the level of impairment and extent of recovery.
Introduction
At stroke onset, function of the contralesional upper extremity, generally considered as the affected side, has been reported to be impaired in approximately 50% to 75% of stroke patients,1-3 and approximately 30% do not regain full motor recovery.2,4,5 Motor deficits of the ipsilesional side, generally considered as the nonaffected side, have been investigated less and the impact of these deficits is to large extent unknown.
Several bilateral neural mechanisms may be behind the dysfunction in the ipsilesional upper extremity. Approximately 10% to 15% of the corticospinal pathways from cortex to end muscles run uncrossed through the spinal cord and therefore can also affect the function of the ipsilesional arm.6-9 Damage in one hemisphere also disturbs the neural processing between the hemispheres.7,10 The primary motor and lateral premotor cortex are closely connected to each other through corpus callosum composing a single cortical unit, which plays an important part in learning and execution of voluntary movements.6-11 In planning and execution of targeted unilateral actions, activity in both hemispheres has been reported.11,12 During more demanding and complex tasks, both hemispheres are even more involved, and damage in one will affect the global task performance.13-15 In stroke rehabilitation, little focus has been targeted toward the ipsilesional side, 16 and particularly little is known how possible deficits in the ipsilesional side may affect functioning in daily activities. 10
Ipsilesional impairments are generally more subtle compared to the contralesional side, and usually not detected with traditional clinical rating scales.14,17,18 Increasing number of studies have, however, reported ipsilesional deficits after stroke using timed dexterity, coordination, and accuracy tests.13,16,19-22 Neglecting a possible decreased function of the ipsilesional side after stroke will also have consequences for clinical evaluations. In several clinical assessments, such as the Fugl-Meyer Assessment and the Action Research Arm Test, the less-affected upper extremity is used as a reference when the quality of movement performance is compared to the less-affected arm in order to achieve the highest scores on an item. This means that the scores of the affected side may become overestimated. 16
In order to capture and specify the impairments of the ipsilesional upper extremity more closely, kinematic movement analysis may be used.23-27 In addition to temporal measures, kinematics will provide information on movement quality, through measures such as movement smoothness and joint angles. It also allows measurement of movements during purposeful daily tasks, which may offer an ecologically valid analysis of existing deficits.24,28,29 Kinematics have been used in several studies of the ipsilesional discrete or reciprocal aiming,12,30-32 but there are a limited number of studies investigating movement deficits during functional reaching including object manipulation.17,25,33 Since movement kinematics are sensitive to task constraints, its goal, and how the object will be used,34,35 it is important to investigate ipsilesional movement performance also during purposeful daily tasks.
The purpose of this study was to investigate whether, and to what extent, movement performance of the ipsilesional upper extremity, measured using kinematic movement analysis of a purposeful daily task during the first 3 months after stroke, is impaired compared to controls, and whether the severity of stroke impairment has an impact on movement performance.
Methods
Participants
Participants included in the current study were extracted from a larger longitudinal prospective observational study, Stroke Arm Longitudinal Study at the Gothenburg University (SALGOT; ClinicalTrials.gov NCT01115348). 36 In SALGOT, during an 18-month period consecutively, every person with first-ever stroke who was admitted to the largest of 3 acute stroke units at the Sahlgrenska University Hospital, Gothenburg, Sweden, was screened. Inclusion criteria for the current study were the following: ischemic or hemorrhagic stroke, age above 18 years, impaired upper extremity function 3 days poststroke, verified as less than 66 points on the Fugl-Meyer Assessment for Upper Extremity, and a kinematic measurement performed within the first 4 weeks, and a follow-up measurement available at 3 months poststroke. The exclusion criteria were the following: upper extremity injury or condition prior to stroke limiting the functional use of the extremity, severe multi-impairment or diminished physical condition prior to stroke, short life expectancy, and non-Swedish speaking. The flowchart of the inclusion process is shown in Figure 1. In the SALGOT, the kinematic data of the ipsilesional arm performance were only available from the measurements where the participants were able to complete the drinking task with their contralesional, more-affected arm. Four participants with ischemic stroke showed minor infarctional changes on the contralesional hemisphere based on the computed tomography or magnetic resonance imaging records and were therefore discarded from the analyses. Thus, 40 persons with stroke were included in the analyses. All assessments were performed, after a joint training period, by 3 experienced physiotherapists who were not otherwise involved in the care of the persons. All participants followed a standard rehabilitation plan and routine. 36
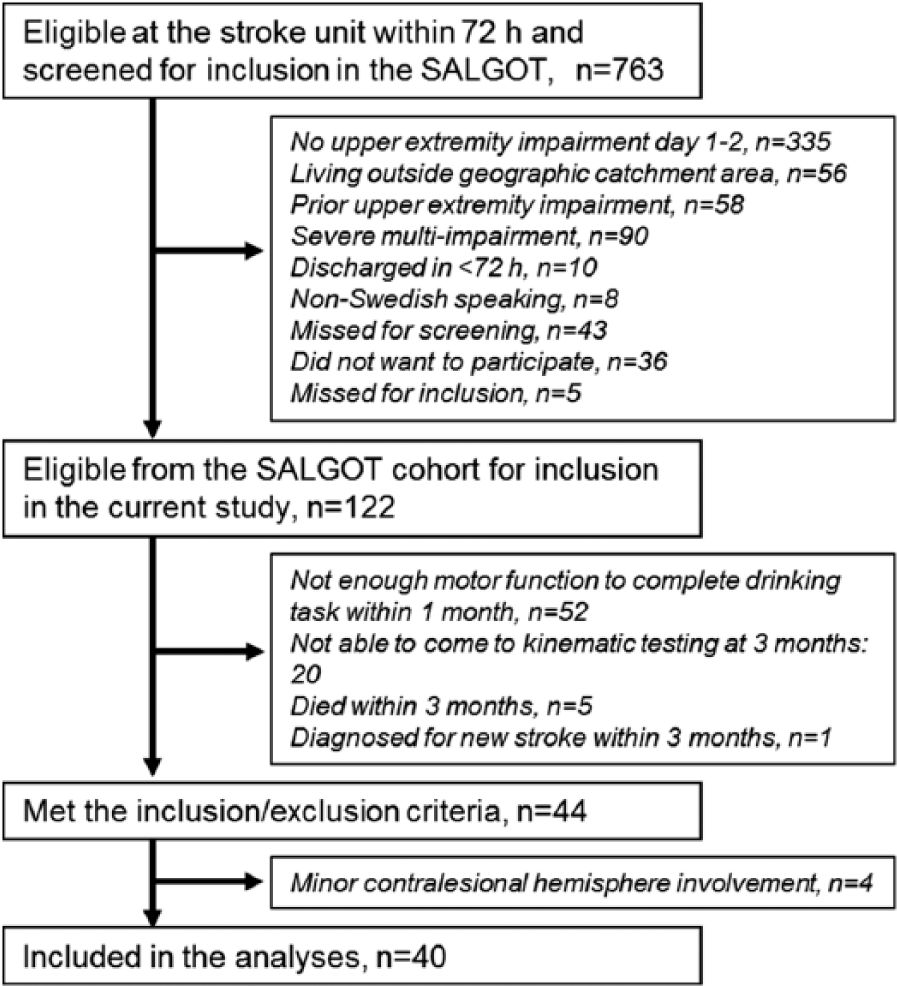
Flowchart over the inclusion process. SALGOT, Stroke Arm Longitudinal Study at Gothenburg University.
Twenty controls, with commensurable age and gender distribution, were included as the reference group. Controls were included if they did not report any musculoskeletal or neurological problems affecting the arm function. 24 The ethical approval was provided by the Regional Ethical Review Board in Gothenburg, Sweden (318-04, 225-08), and oral and written informed consent was obtained from all participants.
Kinematic Movement Analysis of the Drinking Task
For the 3-dimensional motion analysis, 5 optoelectronic cameras were used (ProReflex, MCU240 Hz, Qualisys AB, Sweden). From each camera infrared light was reflected by 9 markers that were placed on the third metacarpophalangeal joint, styloid process of ulna, and lateral elbow epicondyle on the tested side; on the middle part of acromion bilaterally; on the upper part of sternum and the notch between eyebrows; and on the upper and lower edges of the drinking glass. 24 The captured data were transferred to Matlab software (Mathworks Inc, Natick, CA) for custom-made analysis and filtered with 6-Hz second-order Butterworth filter in both forward and reverse directions, resulting in a zero-phase distortion and fourth-order filtering. 24
The drinking task consisted of 5 phases: reaching and grasping the glass, transport of the glass to mouth, drinking, transport of the glass back to the table, and returning the hand to the initial position.24,37 A standardized testing protocol with good test-retest reliability was used.24,37 The drinking task aimed to imitate a natural unconstrained performance similar to a real-life situation. Participants were sitting in a height-adjustable chair with their back against the chair’s back, having the upper arm in neutral adducted position with approximately 90° flexion at elbow and the palm of the hand resting at the table and wrist line parallel to the edge of the table. In the midline of the body (approximately 80% of the arm’s length), a glass (hard plastic cup, diameter 7 cm, height 9.5 cm) with 100 mL water, was placed at a distance of 30 cm from the table edge. The task was performed with each arm, starting with the ipsilesional arm, without any further constraint at a comfortable self-selected speed. To ensure full understanding of the instructions and to get familiarized with the task, 1 to 2 trials were performed before recording. The drinking task was done for 5 times with each arm, and the 3 middle trials were used to calculate an average value for each kinematic variable. The extra trials were only used in few measurements when the data of the 3 trials were incomplete.
Kinematic Variables
The kinematic variables used in this study were movement time (MT), peak velocity of the hand in reaching (PV), peak angular velocity of the elbow extension in reaching (PAVE), smoothness, percentage of time to peak hand velocity (TPV%) and to first velocity peak in reaching (T1P%), trunk displacement (TD), maximal joint angles for shoulder abduction, and elbow extension (Figure 2). The selection of kinematic variables was based on earlier studies of the contralesional arm, with emphasis to include clinically relevant measures with good reliability, validity, and responsiveness that describe both temporal and spatial aspects of movement performance.24,37-42
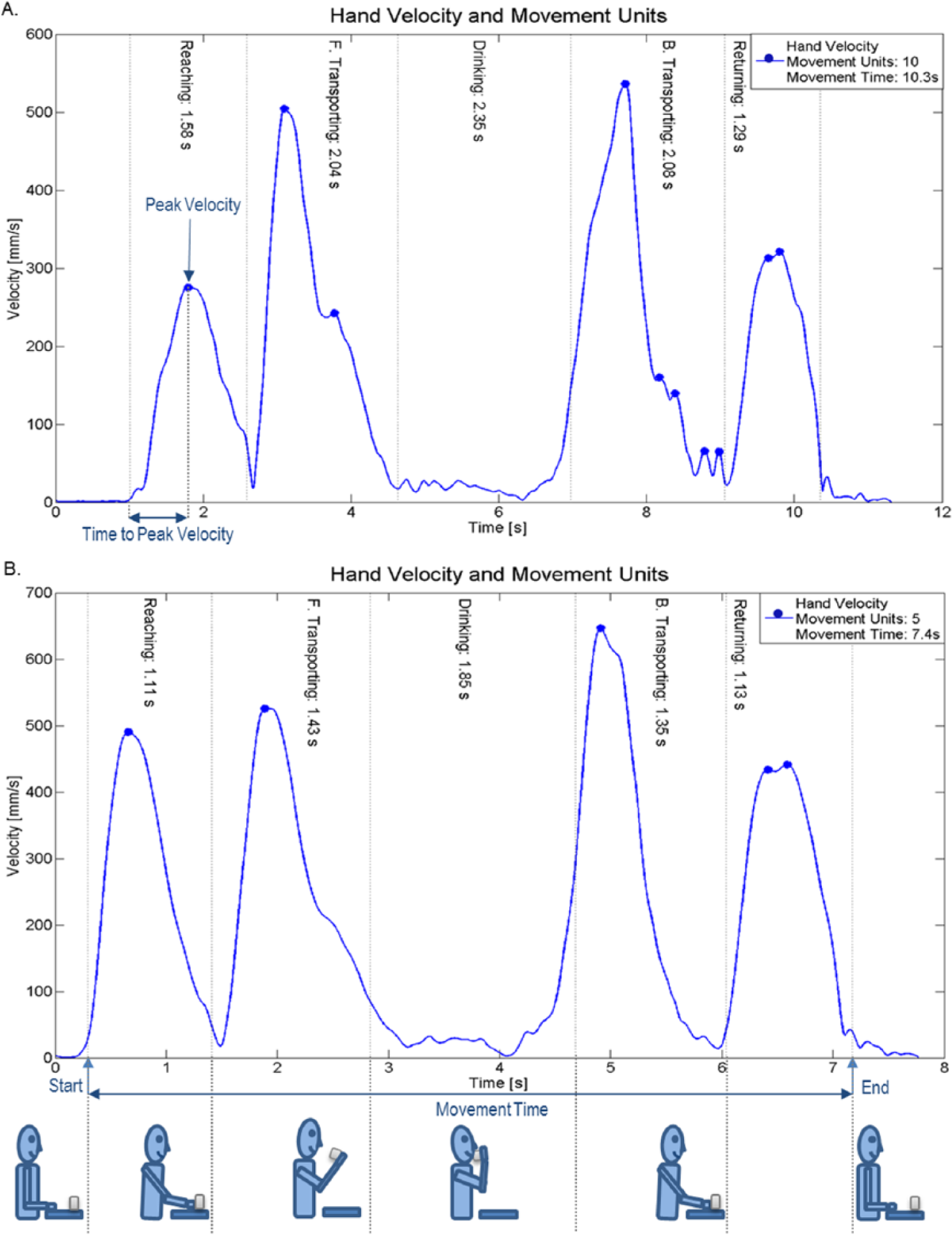
Velocity profile of the drinking task for the ipsilesional upper extremity in a participant: (A) early after stroke, (B) 3 months poststroke. Animation of the drinking phases and schematic description of kinematic measures of movement time, number of movement units (smoothness), peak velocity, and time to peak velocity are embedded in the figure.
Movement time was calculated from the start to the end of the movement. The start was defined as time when the hand marker exceeded 2% of the maximum velocity in the reaching phase; and the end was defined as time when the hand velocity was less than 2% of the maximum velocity in the returning phase. 24 The same cutoff of 2% was used for all movement phases. Movement smoothness, quantified as number of movement units, was obtained separately for reaching and forward transport (NMU 1), back transport and returning (NMU 2), and summed for 4 movement phases (NMU total). To define a movement unit, the tangential velocity profile of the hand was searched for local minima and maxima. A difference between a minimum and the next maximum velocity value that exceeded the amplitude limit of 20 mm/s signified a velocity peak. The time between 2 subsequent peaks had to be at least 150 ms.24,43 These peaks signify repeated accelerations and decelerations during movements and reflect efficiency of movements. 44
Joint angles were calculated for maximal elbow extension in reaching phase, and for maximal arm abduction in reaching and drinking phase. The elbow angle was determined by the angle between the vectors joining elbow and wrist markers and the elbow and shoulder markers, and the shoulder angle by the angle between the vectors joining the shoulder and elbow markers and the vertical vector from the shoulder marker toward the hip. Trunk displacement was defined as the maximal forward displacement of the thorax marker from the initial position and was computed for the entire drinking task. 24
Clinical Assessments and Procedures
The Fugl-Meyer Assessment for Upper Extremity (FMA-UE), a stroke-specific performance based impairment scale, was used to assess the sensorimotor function. 45 FMA-UE includes 33 items divided into 4 subscales (arm, wrist, hand, and coordination) scored on a 3-level ordinal scale. The scoring is based on the ability to perform isolated movements within and out of the synergy patterns. The maximum score of 66 corresponds to unimpaired motor function. FMA-UE is often used to classify the severity of stroke impairment, 46 and the scale demonstrate excellent reliability and validity.45,47 The moderate upper extremity impairment was defined as FMA-UE score between 32 and 57, and the mild impairment between 58 and 66 of the contralesional arm.24,48 The FMA-UE sensation (0-12), passive range of motion and pain (0-24), were also assessed. 45 Spasticity in elbow and wrist was assessed with Modified Ashworth Scale. 49 Type of ischemic stroke was classified according to the TOAST classification, 50 and localization was classified with the Oxfordshire Classification. 51
Statistical Analysis
Statistical analyses were performed using the IBM SPSS 22 (SPSS Inc, Chicago, IL). The level of significance was set to P < .05 (2-tailed). Parametric statistics were used for normally and nonparametric statistics for nonnormally distributed variables.
Differences between the right and left sides in controls were tested with paired sample t test or Wilcoxon signed rank test. The 1-way between-groups analyses of variance (ANOVA) with planned comparisons were used to study differences in kinematic measures between the stroke participants and controls. In variables where a significant difference was identified between the right and left arms in controls, the 1-way between-groups ANOVA and post hoc testing with Bonferroni correction was used to test the differences in stroke separately against the left and right arms in the reference group. For variables with nonnormal distribution, Kruskal-Wallis test and Mann-Whitney U test for post hoc testing with a more stringent significance level (P < .017) was employed.
The impact of the motor impairment (moderate, mild, or no impairment) and the influence of the tested side (right or left arm) on the kinematic measures were analyzed by using the 2-way between-groups ANOVA. Post hoc testing with Bonferroni correction was used for variables demonstrating significant differences between groups. In variables demonstrating a significant interaction with the tested side the differences were analyzed separately for right and left arms in controls. Analyses were conducted separately for early and at 3 months poststroke. For variables with nonnormal distribution, the same procedure as described for 1-way ANOVA described above was used. To interpret the strength of difference between groups, the effect size (ES) statistics of partial eta squared (η 2 ) were used, where the effect size larger than 0.14 indicates a large effect. 52 Subsequently, differences in kinematic variables over time, from early to 3 months poststroke, were analyzed using paired-samples t test or Wilcoxon sign rank test.
Preliminary power analysis (statistical power of 0.8, α = .05) based on the available data on persons with mild stroke impairment and controls 24 indicated that a sample size between 10 and 13 participants in each group was needed to show difference in measure of smoothness, movement time, and trunk displacement. Because the deficits of the ipsilesional side could be even smaller compared to those with mild impairment, the optimal sample size should probably be larger than 11. Therefore, the available sample size of 40 persons with stroke extracted from the SALGOT cohort was considered to be adequate.
Results
Demographic data and clinical characteristics of 40 persons with stroke are displayed in Table 1. The control participants (mean age 58.1 ± 9.4 years, 11 men, 9 women) apart from one (bimanual) were right hand dominant. Age (P = .06), sex (P = .78), and hand dominance (P = .78) were not significantly different between participants with stroke and controls, but persons with moderate impairment due to stroke were older compared to controls (P = .01). Kinematics of the ipsilesional upper extremity are displayed in Table 2. There were no significant differences in kinematics between right and left upper extremity in controls except for peak hand velocity and arm abduction.
Demographic and Clinical Characteristics for Persons With Stroke at Baseline Along With Sensorimotor Impairment at 3 Months Post Stroke.
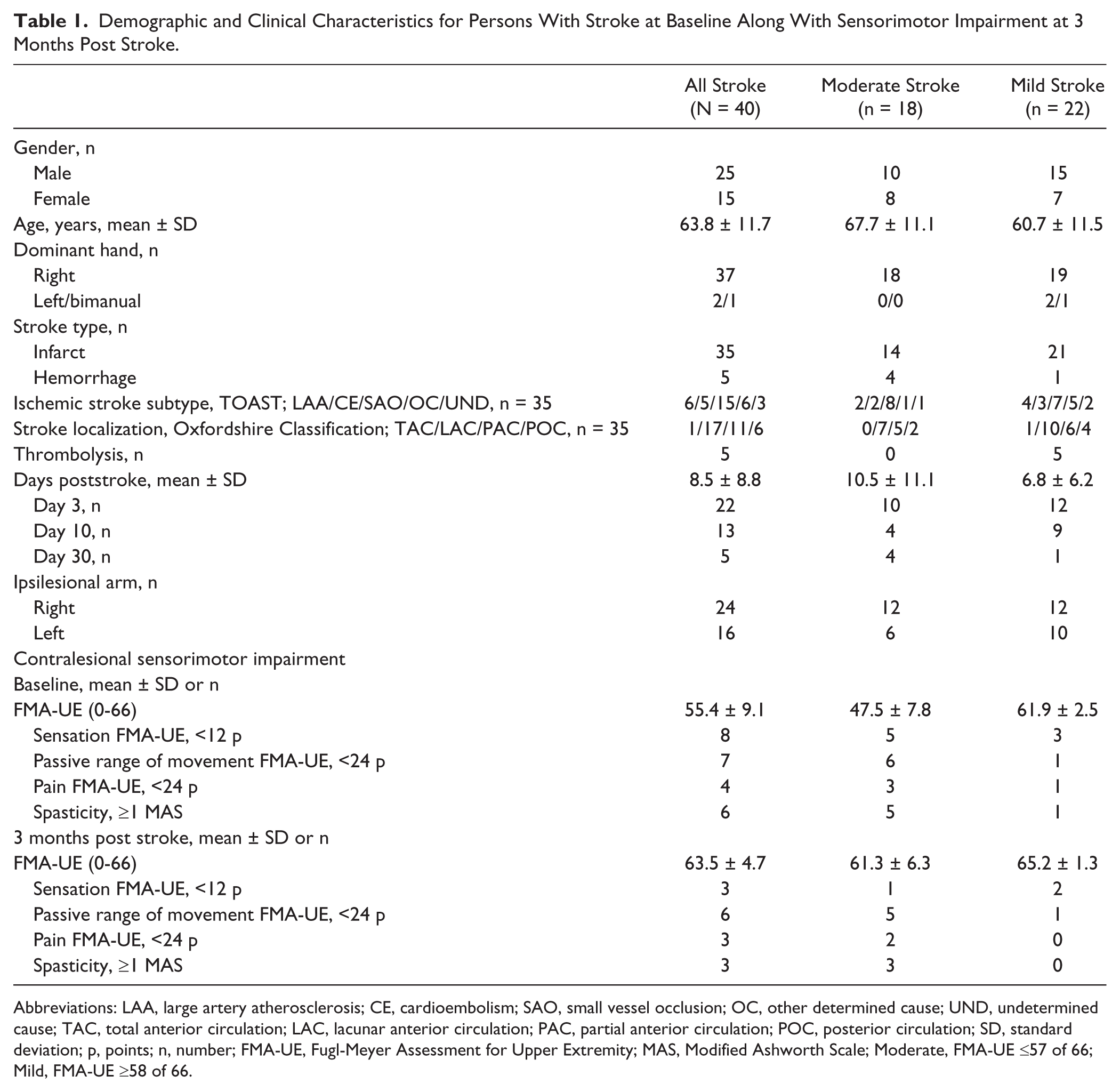
Abbreviations: LAA, large artery atherosclerosis; CE, cardioembolism; SAO, small vessel occlusion; OC, other determined cause; UND, undetermined cause; TAC, total anterior circulation; LAC, lacunar anterior circulation; PAC, partial anterior circulation; POC, posterior circulation; SD, standard deviation; p, points; n, number; FMA-UE, Fugl-Meyer Assessment for Upper Extremity; MAS, Modified Ashworth Scale; Moderate, FMA-UE ≤57 of 66; Mild, FMA-UE ≥58 of 66.
Kinematic Measures (Mean ± SD) of the Ipsilesional Upper Extremity in Persons With Stroke and in Healthy Controls.
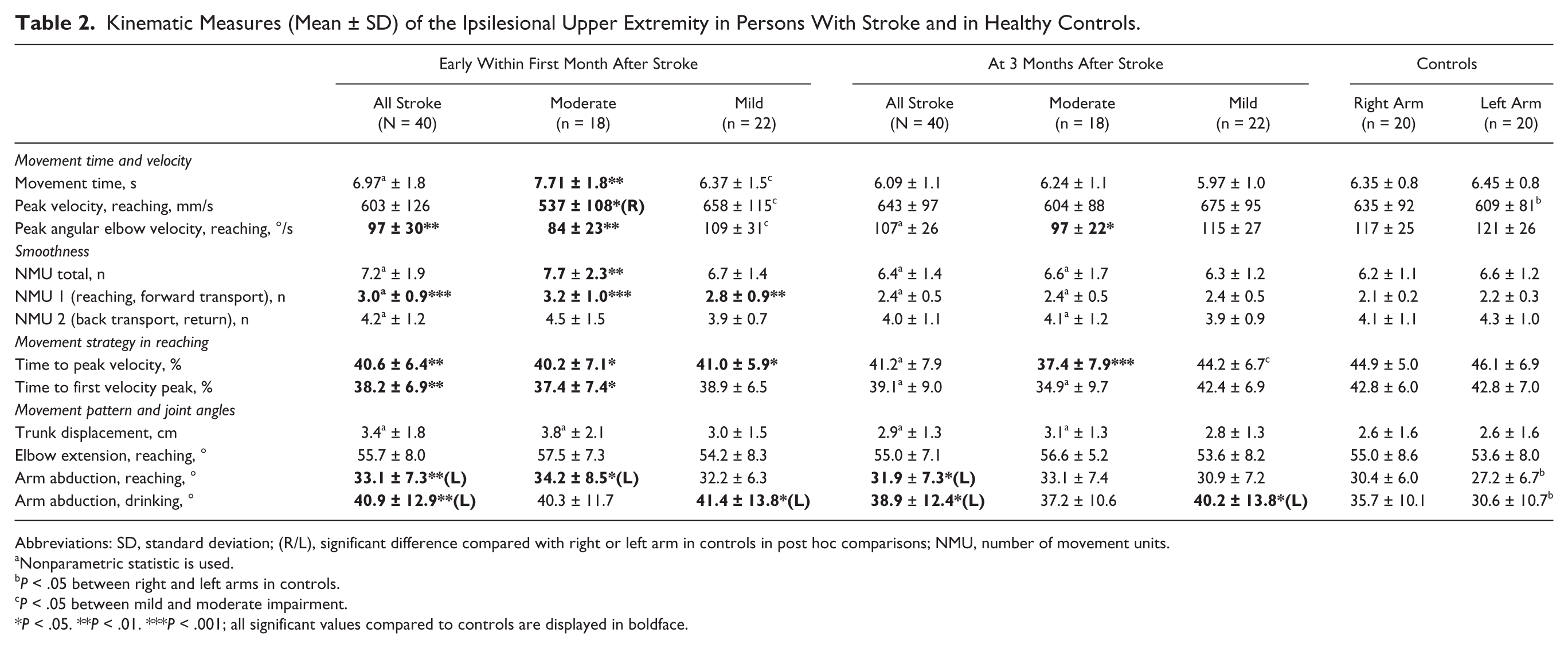
Abbreviations: SD, standard deviation; (R/L), significant difference compared with right or left arm in controls in post hoc comparisons; NMU, number of movement units.
Nonparametric statistic is used.
P < .05 between right and left arms in controls.
P < .05 between mild and moderate impairment.
P < .05. **P < .01. ***P < .001; all significant values compared to controls are displayed in boldface.
Ipsilesional Movement Performance Early After Stroke
Significant differences between the entire stroke group and controls were detected in 6 of the 12 kinematic measures early after stroke. Movements of the ipsilesional arm showed a lower maximum velocity of the elbow extension during reaching, larger number of movement units in reaching and forward transport, indicating decreased movement smoothness. The relative time to the first (T1P%) and maximal velocity peak (TPV%) was reached earlier, indicating that relatively longer time was spent for deceleration during reaching. Also a larger arm abduction angle during reaching and drinking was observed (Table 2).
The upper extremity impairment level had a significant impact on ipsilesional movement performance early after stroke. In the subgroup with moderate impairment, 8 out of 12 kinematic measures were significantly different from controls (Table 2). The effect sizes (partial eta squared) for the differences were large in movement time (MT, 0.14), smoothness (NMU 1, 0.24), elbow angular velocity (PAVE, 0.18). A significant interaction effect between the impairment level and tested side was detected for the arm abduction, and post hoc analysis demonstrated significant difference only compared to the left arm in controls.
In the subgroup with mild impairment, significant differences compared to controls were found in 3 measures: movement smoothness (NMU 1), TPV% and arm abduction during drinking, the latter was however significant only in comparison with the left arm in controls and the main effect was influenced by the interaction with the tested side (Table 2). In addition, significant differences were found in measures of MT, PV, and PAVE between the moderate and mild impairment subgroup.
Ipsilesional Movement Performances 3 Months After Stroke
In the entire stroke group, significant differences could still be found in arm abduction at 3 months after stroke. These differences were, however, small and only significant in comparisons with the left arm in controls.
The subgroup with moderate impairment demonstrated a slower elbow angular velocity (PAVE) and a longer relative deceleration phase in reaching (TPV%) compared to the controls (Table 2). The impact of impairment level effect was large for the TPV% (η2 = 0.23). In the subgroup with mild impairment, a significantly larger abduction angle in drinking phase was detected, but this difference was only present when compared with the left arm in controls and the main effect was influenced by the interaction of the tested side.
Change in Ipsilesional Kinematics Over Time
A significant improvement over time was found for 5 out of 12 kinematic measures both in entire and moderate impairment subgroup (Table 2). Movements of the ipsilesional side were faster (MT, P < .001; PV, P < .05; PAVE, P < .01) and smoother (NMU total, P < .01; NMU1, P < .001) 3 months poststroke compared to the initial measurement (Figure 2). In the subgroup of mild impairment, significant improvement was also seen in movement smoothness (NMU 1, P < .05).
Discussion
Results from the current study extend previous research and show that movement deficits of the ipsilesional upper extremity after stroke are also present during a purposeful daily task, such as drinking from a glass. Both speed related kinematics as well as measures related to movement quality and movement pattern were impaired particularly early after stroke. Reduced movement performance was more prominent in those with moderate stroke impairment compared to mild impairment, both early and at 3 months after stroke. Movement kinematics improved over time, and reached a level comparable with controls at 3 months poststroke. In the subgroup of moderate stroke impairment, however, remaining deficits in angular velocity of elbow extension along with a longer relative deceleration phase in reaching could still be seen after 3 months.
Several underlying neural mechanisms, such as noncrossing and interhemispheric motor pathways as well as diaschisis on remote but connected areas of the brain lesion, may explain why the ipsilesional deficits, as seen in the current study, where most manifest early after stroke and associated with the severity of stroke.8,9,22,26,53 Functional imaging studies have also shown increased bilateral hemispheric activity during more complex tasks. 22 Thus, in activities such as drinking, the movement performance may to larger extent rely on bilateral brain activation. In the paretic arm, the movement smoothness during grasping has found to be associated with bilateral brain activity. 54 This indicates that certain tasks and movement parameters, described as more complex and requiring continuous sensory feedback, may be more or less dependent on bilateral hemispheric activation. However, further research on hemispheric lateralization as well as specificity is needed to further illuminate these mechanisms.
The ipsilesional upper extremity performance has been investigated in a previous study, with comparable setup of a reach-to-grasp task in a subacute phase after stroke.17,25 Similarly to our study, it reported that smoothness of reaching was reduced early after stroke and that a significant improvement was seen within the first 3 months poststroke. At 3 months, the smoothness was comparable to healthy controls in the current study, but still decreased in the study of Metrot et al. 17 This discrepancy may be caused by the existing differences on the impairment severity among the participants (mean FMA-UE scores of 42 contra 64 in the current study). In the current study, the smoothness demonstrated the strongest effect size early after stroke in both moderate and mild impairment subgroups. Smoothness was also improved over time in all groups. Previous kinematic studies of the contralesional arm have also demonstrated that the smoothness is one of the key measures for quantifying and evaluating movement performance after stroke.24,38,39 Jerky grasping movements of the paretic arm 6 weeks poststroke have been associated with elevated recruitment of both ipsilesional and contralesional sensorimotor areas, which indicates that these secondary areas may be important in error correction process. 54 Together, these findings imply that smoothness seems to have relevance not only for contralesional but also for ipsilesional arm performance after stroke.
Deficits and improvements during the first 3 months after stroke have also been reported for movement time and velocity during ipsilesional reaching.17,25,30,33 In the current study, only the subgroup with moderate stroke impairment showed initial deficits and significant improvements in these measures. This is reasonable when taking account that the previous study 17 included persons with a higher motor impairment level, in comparison with the current study. Kinematic measures related to movement pattern, such as trunk displacement and elbow extension, did not differ significantly from controls in the present study. Although significant differences were found in arm abduction angle, these differences were relatively small and difficult to interpret due the interaction effect of the tested side. Based on these findings, it can be suggested that, particularly in those with more severe motor impairment, the ipsilesional deficits lay mainly in measures related to speed (MT, PV, PAVE), movement smoothness (NMU), and strategy (TPV%, T1P%). These deficits may be difficult to detect at first sight, which can explain why the ipsilesional impairments are often not detected during clinical evaluation.
It has been discussed whether the side of the stroke lesion would influence the deficits observed on the ipsilesional arm; however, the literature is not in unison.33,55-57 Previous studies have reported differences in arm kinematics due to the lesion side in movement trajectory modulation and positional error at the end of the movement.57,58 On the other hand, movement time, peak velocity, acceleration, and smoothness, as also used in the current study, have not shown to be influenced by the side of the lesion.17,58 In the current study, the influence of the tested side was not studied directly, but taken into account in the analyses. The tested side did not have an impact on the results except for 2 measures, the peak velocity and arm abduction. This finding was, however, mainly influenced by the difference found between the right and left arms’ performance in controls.
In the stroke group, the kinematic testing of drinking task was performed several times (2-4) during the first 3 months poststroke. Controls were only tested once. This possible study design bias can probably not be counted for training effect, but a familiarity with the testing session and possible underlying wish to get a better result compared to earlier testing cannot be ruled out.
The strength of the current study is the use of an objective and sensitive method of kinematics, which allows differentiation between movement deficits such as speed, smoothness, and movement pattern.25,59 This together with a relatively large sample size retrieved from a larger unselected study sample (SALGOT) and that almost 90% of participants were tested as early as within 10 days after stroke is unique and improves the external validity of the results. To be included early after the onset of stroke is valuable and offers a possibility to evaluate the motor impairment and activity limitation on the ipsilesional side before the natural recovery and training effect will have an impact on the movement performance. As the movement kinematics are highly dependent on the task constraints, the use of a functional daily task, such as drinking, adds valuable ecologically valid knowledge regarding motor functioning of the ipsilesional arm. A limitation of the current study is that only those who were able to perform the drinking task with their more-affected arm were included. Thus, the findings from the current study can only be generalized to persons with mild and moderate motor impairment after stroke in the subacute stage. On the other hand, it could be expected that the ipsilesional arm would be even more affected in the group with severe motor impairment. The mean age of the controls was lower compared to persons with moderate stroke impairment (P = .01), which may have contributed to found differences in kinematics in this subgroup. On the other hand, the magnitude of differences in kinematics was largest in this subgroup, but a study with a larger and better matched control group would be warranted.
Clinical Interpretation and Implications
Findings from this study suggest that in clinical studies and evaluations, the use of less-affected upper extremity as a control or reference may underestimate the level of impairment and extent of recovery. The ipsilesional upper extremity should be considered as the less-affected upper extremity and be part of the physiotherapy assessment of motor function after stroke. Future research should also include people with more severe impairment and investigate which motor control parameters and underlying mechanism are influencing movement performance in ipsilesional upper extremity, so that these deficits could be directly targeted in the rehabilitation process.
Conclusions
Results of the current study demonstrate that movement performance of the ipsilesional upper extremity is affected after stroke even while performing purposeful daily activities. The ipsilesional deficits are influenced by the degree of the initial motor impairment of the more-affected upper extremity. These deficits are also more prominent early after stroke and will reach a level comparable with healthy controls after 3 months poststroke. In clinical evaluations, the less-affected upper extremity should not unreservedly be used as a reference to the more-affected side.
Footnotes
Acknowledgements
The authors thank the study participants and Hanna Persson and Anna Danielsson for their help in data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Local Research and Development Board for Gothenburg and Södra Bohuslän, The National Stroke Association in Sweden, JB Wennerström’s Foundation, Hjalmar Svensson’s Research Foundation, Promobilia, Norrbacka Eugenia Foundation, Swedish Heart and Lung Foundation, and Swedish Brain Foundation.
