Abstract
Background
The recovery of motor function and independence in activities of daily living (ADL) following stroke can be significantly improved with multidisciplinary rehabilitation. 1 Recovery is often partial, so attempts have been made to enhance the beneficial effect of rehabilitation with pharmacological agents. Most studies have tried dexamphetamine. 2 In early inpatient rehabilitation the effect of amphetamine in combination with physiotherapy continues to be controversial. A Cochrane review of studies on 287 patients suggested that motor function and ADL may improve through added amphetamine treatment by a relative change in motor assessment scores using assessments, including the Fugl-Meyer Motor Score, the Rivermead Motor Assessment Score, and the Lindmark Motor Assessment Chart. 3 It can be hypothesized that those study results are related to small numbers of participants and variations across trials, including treatment onset, duration, frequency, and dexamphetamine dosage, as well as less than optimal methodological quality of early studies (randomization procedures, assessment grading levels, measurement frequency, and blinding).4,5 The present study addressed many of these concerns, although the number of subjects was modest.
Dexamphetamine increases the release of dopamine and noradrenalin in the brain, which can positively influence mechanisms of plasticity and repair.6,7 We hypothesized that the combination of dexamphetamine and physiotherapy (experimental group, EG) within 60 days of onset of stroke, and selected intervention parameters during inpatient rehabilitation, will further improve motor function and ADL compared with a control group (CG) receiving placebo and physiotherapy.
Methods
The study was designed as a randomized, double-blind, placebo-controlled trial with repeated measurements. Patient recruitment started in winter 2001. The study was completed after the last 12-month follow-up assessment in summer 2006.
Patients following stroke who were admitted to one center for inpatient rehabilitation were assessed for study eligibility. Patients were included if they fulfilled the following criteria: first-ever ischemic cerebral infarction 14 to 60 days before intervention, patients had to have motor impairments of arm, hand, leg, and foot which were assessed with a CSMA score of one to five. Only one out of the arm, hand, leg, or foot subscale was allowed to be scored with level six, impairment in accordance with location of infarction found in computed tomography/magnetic resonance imaging, and older than 18 years. Exclusion criteria were additional terminal, psychiatric, or severe neurological diseases; unstable cardiac arrhythmia (including atrial fibrillation, supraventricular tachycardia); uncontrolled arterial hypertension (systolic >160 mm Hg and/or diastolic >100 mm Hg); global aphasia; dementia; neglect; treatment with an anticonvulsant, anxiolytic, neuroleptic, or α-adrenergic agonist and antagonists. Figure 1 shows the study flow chart. The study was approved by the responsible ethics committee and registered at clinicaltrials.gov trial register: NCT00572767.

Study flow chart.
After giving written informed consent, patients were randomly assigned to EG or CG using a computer-generated randomization list (ratio 1:1). Randomization was concealed in an envelope. All patients and participating study staff (physician, nurses, therapists, assessors, and pharmacist) were blinded to the patient’s group allocation. The envelopes were opened after the 12-month follow-up assessment of the last patient. Both prescribed tablets (dexamphetamine, placebo) were equal in shape, color, and taste.
Outcome Measure
Patients were assessed using the CMSA by 1 of the 4 blinded assessors.8,9 CMSA was administered twice during baseline (B1 at 11 days and B2 at 4 days before intervention), once a week during the 5-week treatment phase (S1-S5) and during follow-up at 1 week (F1), 6 months (F2), and 12 months (F3) after the intervention.
The CMSA evaluates ADL level (disability score = primary outcome measure) and motor function (impairment score = secondary outcome measure).8,9 The ADL subscale includes gross motor function and a walking index (1 =
All blinded assessors completed training sessions as recommended by Gowland et al. 11 Interrater reliability was evaluated during 4 patient video-rating sessions by calculating the intraclass correlation coefficient (ICC). The ICC for the ADL subscale was 0.96 (ranged between 0.78 subscale leg to 1.00 subscales postural control and arm). The mean ICC over all subscales was 0.94.
Intervention
Patients in EG received dexamphetamine sulfa, 10 mg (G. Streuli & Co AG) Tuesdays and Thursdays for 5 weeks, 1 to 3 hours prior to a 1-hour physiotherapy session. Patients in CG received placebo tablets on the same days.
Physiotherapy Treatment
Treating therapists were blinded to the patients’ group allocation. For both groups, the everyday physiotherapy sessions followed principles of the Bobath concept and motor learning.12-14 Physiotherapy included training on balance control, mobility, transfers, ADL, and controlled limb movements. Therapy objectives were based on the patient’s individual impairments. All patients also received a standard multidisciplinary inpatient rehabilitation program including occupational therapy, speech and language therapy, and, if required, hydrotherapy and massages. After discharge, patients received outpatient physiotherapy once or twice a week for 30 to 45 minutes during the follow-up period.
Statistics
At B2, homogeneity between groups was tested regarding CMSA disability and impairment score, and patient characteristics according to Table 1 using independent
Patient Characteristics at Baseline 1
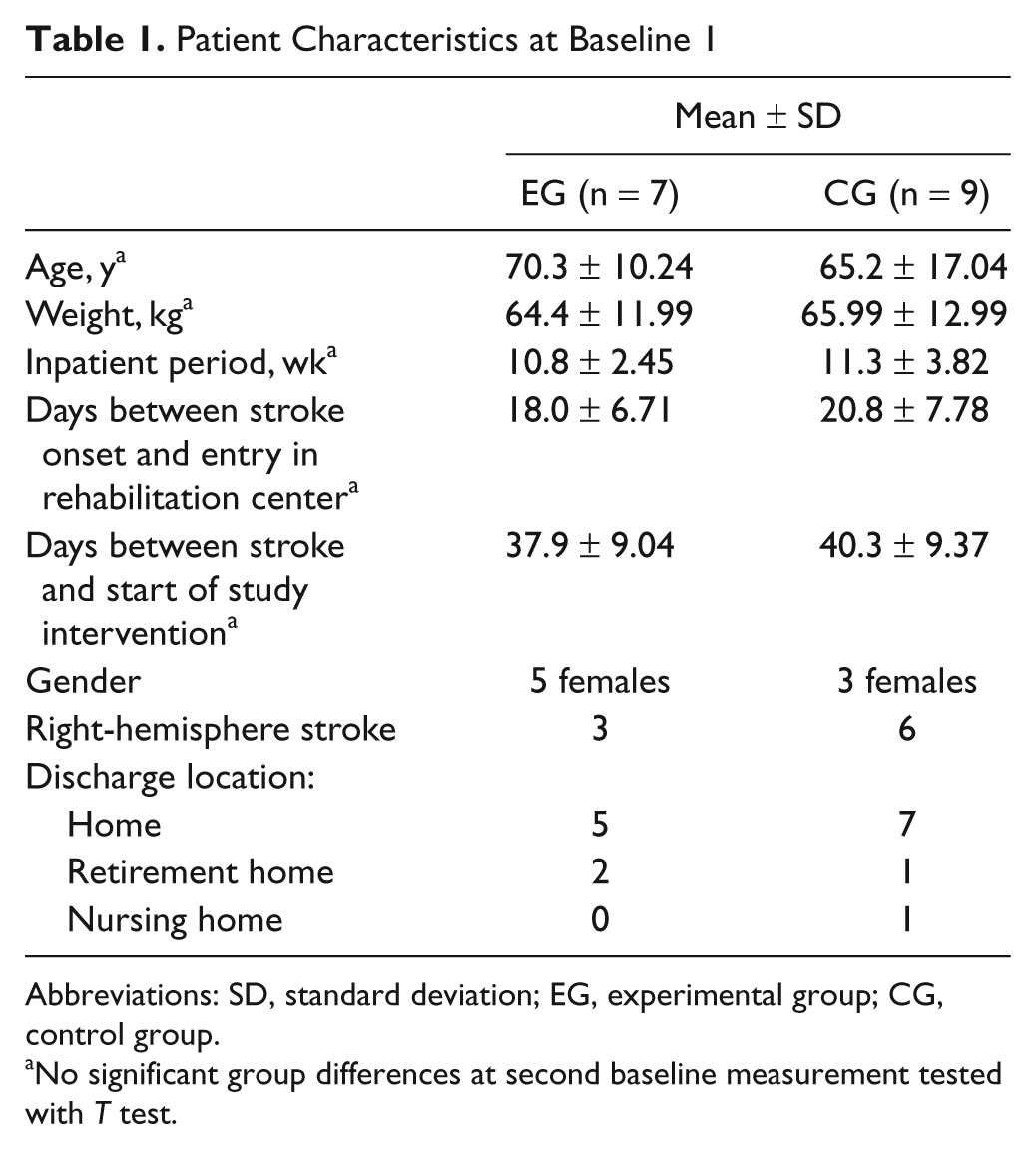
Abbreviations: SD, standard deviation; EG, experimental group; CG, control group.
No significant group differences at second baseline measurement tested with
Results
Baseline
A total of 16 patients were recruited (Table 1). All participants completed the study, whereby one patient had to be transferred to acute care during the intervention period because of health problems not related to the study. She received a part of the intervention during the study period and was again assessed for the 12-month follow-up. The missing values method was applied 3 times in EG in 3 different cases. For one patient in EG, who left the study after S2, data were completed with the group mean difference from one measurement point to the next.
Group comparisons at baseline showed no statistically significant differences at B1 and B2 for any of the CMSA subscales. A nonsignificant age difference of 5 years was detected in favor of the CG. A statistically significant improvement in ADL was found for both groups from B1 to B2.
Outcomes
Two-factor ANOVAs to test for group-specific differences revealed a significant effect for EG for CMSA subscale ADL and arm from B2 to F1. This confirms a significantly better recovery in ADL and arm function of EG compared with CG from B2 to F1. Furthermore, both groups showed a significant change from B2 to F1, B2 to F2, and B2 to F3 in all subscales. Other subscales did not differ (Table 2).
ANOVA Results for Group Interaction Over Time a
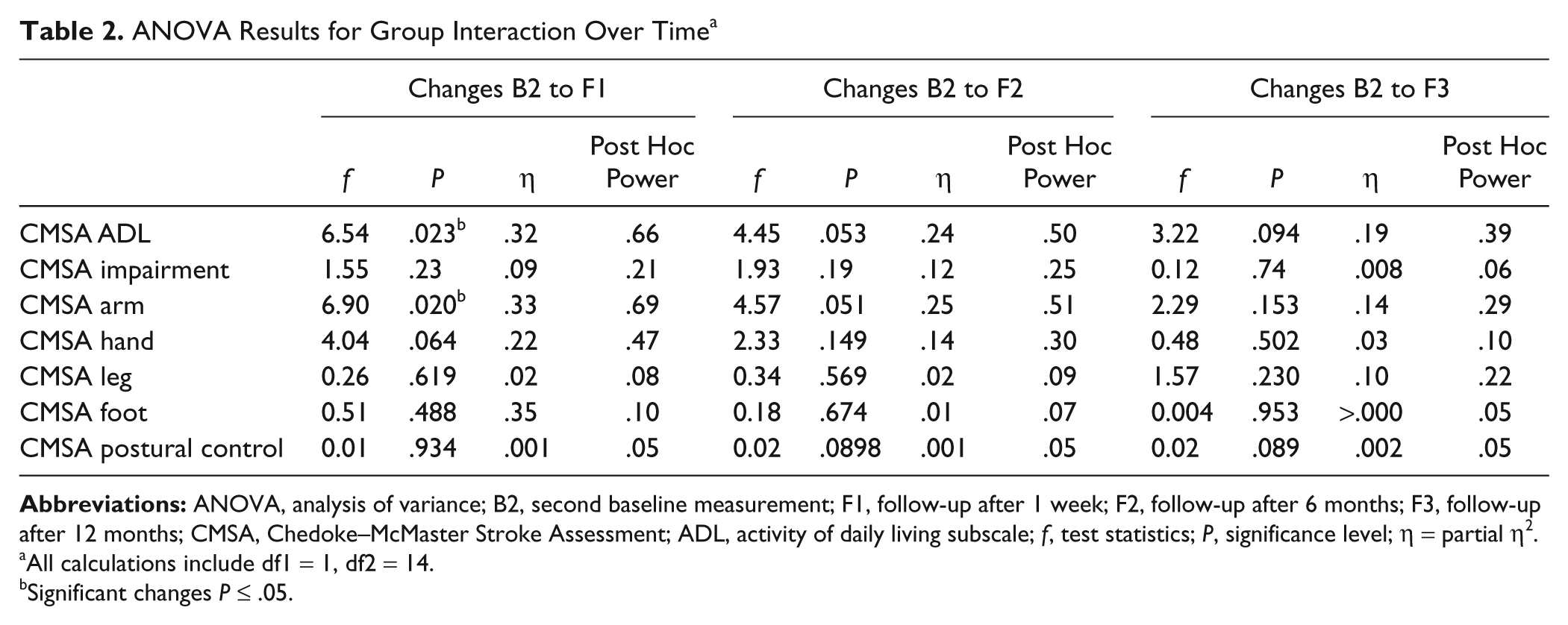
All calculations include df1 = 1, df2 = 14.
Significant changes
Figures 2 and 3 show mean values for the CMSA ADL and arm subscales for all measurement events of both groups. The CMSA ADL subscale showed a +45% improvement in EG from B2 to finalization of the study (F1), compared with +27% in CG. The improvement to F3 was 33 points (+65%) for EG and 20 points (+36%) for CG.
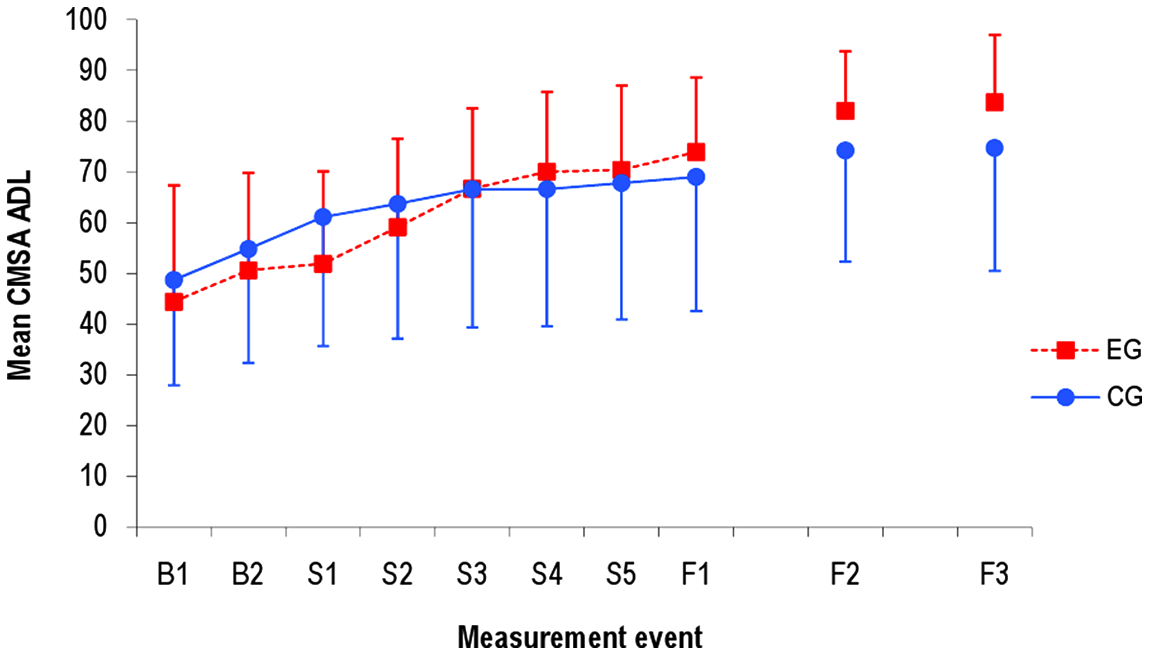
Mean score for CMSA ADL subscale in EG and CG at all measurement events. Abbreviations: CMSA, Chedoke–McMaster Stroke Assessment; ADL, activities of daily living; EG, experimental group; CG, control group; B1, baseline 1 (11 days before intervention start); B2, baseline 2 (4 days before intervention start); S1 to S5, intervention period (once every week during 5-week intervention); F1, follow-up 1 week after intervention stop; F2, follow-up 6 months after intervention stop; F3, follow-up 12 months after intervention stop.
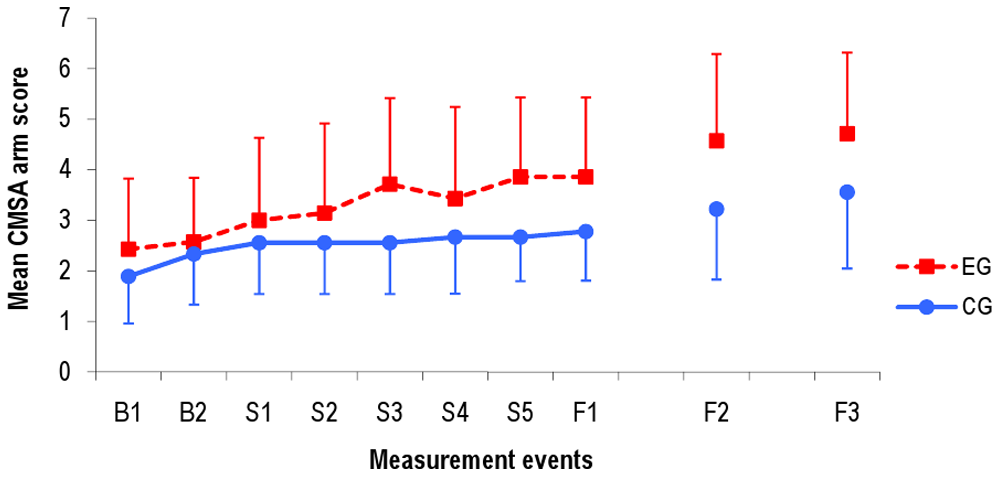
Mean score for CMSA arm subscale in EG and CG at all measurement events. Abbreviations: CMSA, Chedoke -McMaster Stroke Assessment; EG, experimental group; CG, control group; B1, baseline 1 (11 days before intervention start); B2, baseline 2 (4 days before intervention start); S1 to S5, intervention period (once every week during 5-week intervention); F1, follow-up 1 week after intervention stop; F2, follow-up 6 months after intervention stop; F3, follow-up 12 months after intervention stop.
Patients in EG started to show better results in the ADL assessment after the third intervention week, and after 6 oral amphetamine dosages. Improvement in arm motor function on the CMSA was shown by an increase from 2.6 up to 4.7 (+80%) in EG, compared with 2.3 up to 3.6 (+56%) in CG.
Recovery in Both groups (Effect of Time)
Table 3 provides an overview for both groups on CMSA mean values for all subscales and the total impairment score at B2, F1, F2, and F3.
Motor Function at Preintervention (B2), at 1-Week (F1), 6-Month (F2), and 12-Month (F3) Follow-Up a
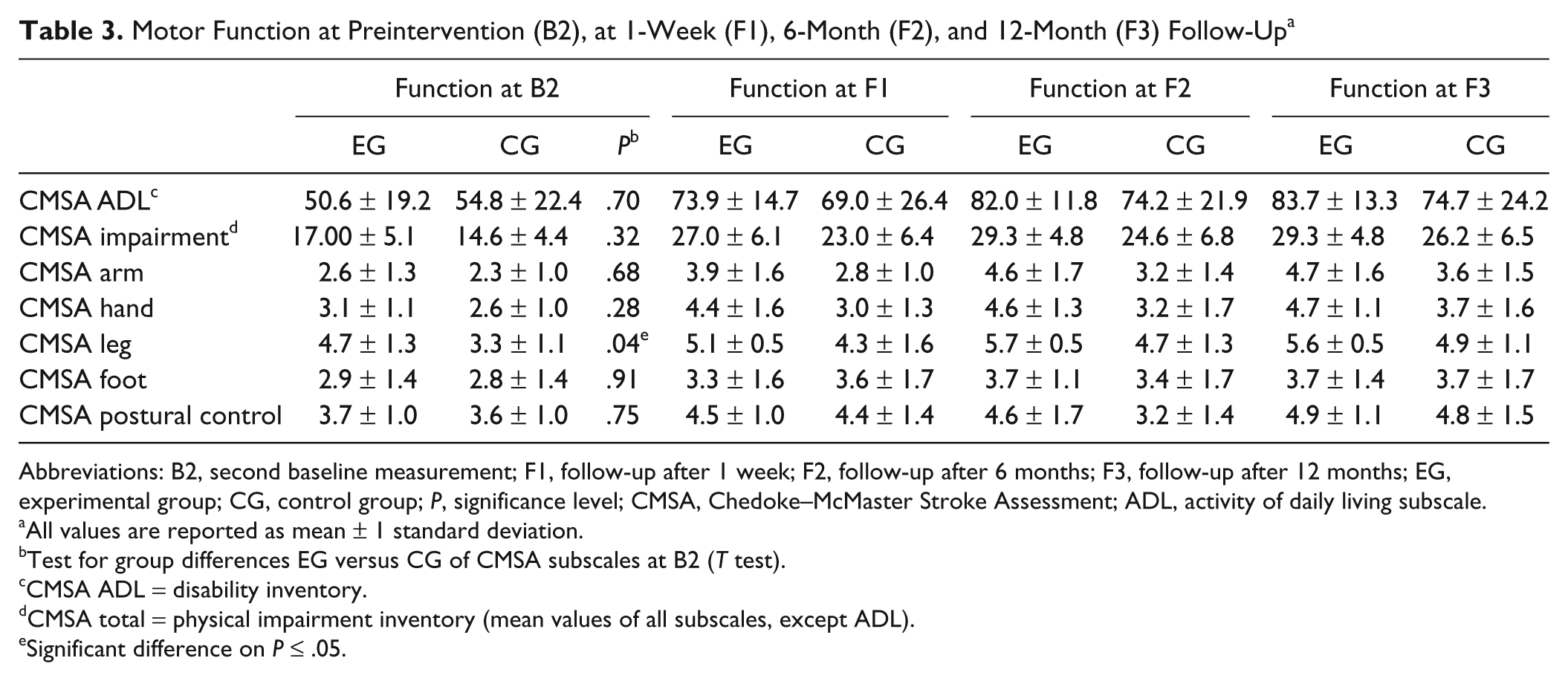
Abbreviations: B2, second baseline measurement; F1, follow-up after 1 week; F2, follow-up after 6 months; F3, follow-up after 12 months; EG, experimental group; CG, control group;
All values are reported as mean ± 1 standard deviation.
Test for group differences EG versus CG of CMSA subscales at B2 (
CMSA ADL = disability inventory.
CMSA total = physical impairment inventory (mean values of all subscales, except ADL).
Significant difference on
Adverse Events
No adverse events (nausea, diarrhea, increased blood pressure, obstipation) or mood changes were observed. In EG, one death occurred between the 6- and 12-month follow-up, which was not related to the intervention.
Discussion
Of the published randomized controlled trials for stroke, only 4 have demonstrated a beneficial effect of dexamphetamine.4,5,17,18 Our trial revealed significant improvement of ADL and arm motor function in favor of the combination of dexamphetamine with physiotherapy. The calculated power of .66 and .69 for ADL and arm CMSA subscales, respectively, is promising, considering the small sample size of 16 participants. The additional improvement moved the EG participants from needing assistance toward just supervision in transfer tasks, walking, and changing body positions. Both groups started with assisted movements in most cases. Patients in EG were able to lift the arm to eye level, whereas CG patients gained the ability to touch the chin or opposite knee only at F2, which was maintained until F3. Patients in EG were able to move their arm in all joints and degrees of freedom against gravity. Patients in CG were only able to move the arm without gravity or not in the whole range of motion.
The inpatient multidisciplinary rehabilitation program produced nonsignificant improvements for the entire cohort in all subscales of the CMSA. Sample sizes would have to be from 25 to 50 patients to show possibly significant improvements for subscales leg and foot respectively (post hoc power analysis with software Gpower version 3.08). Therefore, our results have to be interpreted cautiously. Additionally, we would like to raise attention to an intensive assessor training. Similar to Gowland et al, 8 our interrater reliability showed the least correlation for the subscale leg, which could have led to under or over scoring.
The improvements for ADL and arm motor function are in line with the results reported by Walker-Batson et al,5,18 who published 2 articles on studies with five and twelve patients in EG, respectively. Gladstone et al, 19 who investigated 31 patients in EG, did not find an overall group difference between EG and CG, which could be because of lower mean ADL values in the CMSA at baseline. However, in a subgroup analysis for patients with a moderate hemiparesis, they found a significant time and group interaction for upper limb motor function in favor of EG. 19 Our positive results stand in contrast to the study by Treig et al, 20 including 12 patients in EG. Their study population was 10 years younger, almost 20 kg heavier, and included only 2 women of 24 participants. Amphetamine was given on average 3 weeks earlier and only combined with 45 minutes of physical therapy. These parameters led to our hypothesis for future studies that an amphetamine dosage of 10× 10 mg has to be adapted in relation to the patient’s weight. Our study was designed according to 2 successful studies suggesting an intervention duration of 5 weeks, an oral dosage of dexamphetamine of 10 mg administered every third to fourth day, and an intervention begun 14 to 60 days after stroke onset5,18 and administered in combination with practice. 21 The timing of drug administration after stroke may be a crucial factor. In our trial, dexamphetamine intake started at a mean of 37.9 (EG) and 40.3 (CG) days after stroke. Other studies with positive results started amphetamine intake 16 to 30 days.5,18 In studies with no significant results, the first dose was administered from less than 72 hours to 10 days poststroke,19,22-24 which could also be because of the variability in the patients’ state <30 days after stroke onset.
Our results suggest that the CMSA is sensitive to changes in aspects of functional recovery for an amphetamine intervention. We assumed that the CMSA would be more applicable for amphetamine interventions because of its higher responsiveness than the Fugl-Meyer Motor Scale 8 that was used in most previous studies.
Trials to date suggest the possibility that starting medication >3 weeks after stroke in older patients (>60 years) and earlier <3 weeks in younger patients (<60 years) should be considered in future designs of trials with larger numbers of subjects.
Other factors that may influence the effectiveness of dexamphetamine are the time between drug intake and physiotherapy, the frequency of drug administration, and the amount and specific type of physiotherapy that patients receive. Our single center design allowed a highly consistent methodology for all study participants and control of these factors, but we only tested one regimen.
Previous studies reported that only a small proportion of screened patients were included because of the strict selection criteria for the administration of amphetamine. Recruiting periods in past trials ranged from 24 to 42 months with recruitment versus inclusion ratios of 9 to 40.5,20,23 In the present study, 57 times the number of included patients were screened for study eligibility over 60 months. Since Walker-Batson et al 5 in 1995 and Martinsson et al 17 in 2003 showed the safety of the 10-mg dosage of dexamphetamine, by relaxing the inclusion criteria (all levels of paresis severity, treatment with anticonvulsant, anxiolytic, neuroleptic medication) it may become feasible to treat more patients with dexamphetamine in the future.
Conclusion
Amphetamine enhanced recovery of ADL and arm motor control at the completion of the 5-week inpatient intervention and did not alter other outcomes of the CMSA. This suggests that the dose and timing of dexamphetamine employed can augment physiotherapy. Effect size calculations based on our results recommend the inclusion of at least 25 patients per group in future studies.
Footnotes
Acknowledgements
The authors thank all patients for their participation. We are grateful to all assessors who are not coauthors of this article: Katja Blaettel and Anja von Doellen and thank all physicians who helped with the patient recruitment and selection: Hans Dieter Huellstrung, MD, and Oliver Findling, MD. We thank Dr Markus Stoecklin for advice on the statistical analysis and Dr Oliver Amft for comments on an earlier version of the manuscript.
The author(s) declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
The study was supported by a research grant from the Hugo and Elsa Isler-Foundation. Medication was provided by G. Streuli & Co AG at no charge.
