Abstract
Background. Dysfunction of corticospinal pathways has been implicated in motor impairments in people with bilateral spastic cerebral palsy (CP). While structural damage to corticospinal pathways in people with CP is known, its impact on the activation of these pathways is not. Objective. To provide the first, complete activation profile of corticospinal pathways in adults with CP using a full range of transcranial magnetic stimulation (TMS) intensities and voluntary contractions. Methods. TMS targeted the soleus muscle of 16 adults with bilateral spastic CP and 15 neurologically intact (NI) control participants. Activation profiles were generated using motor-evoked potentials (MEPs) produced by varying both stimulation intensity and degree of voluntary muscle activity. Anatomical integrity of corticospinal pathways was also measured with diffusion tractography. Results. Participants with CP had smaller MEPs produced by TMS at 1.2× active motor threshold during submaximal (20%) muscle activity and smaller maximal MEPs produced under any combination of stimulation intensity and voluntary muscle activity. At a fixed stimulation intensity, increasing voluntary muscle activity facilitated MEP amplitudes to a lesser degree in the participants with CP. Consistent differences in diffusion tractography suggested structural abnormalities in the corticospinal pathways of participants with CP that correlated with maximal MEPs. Conclusion. People with bilateral spastic CP have impaired activation of low and high-threshold corticospinal pathways to soleus motoneurons by TMS and reduced facilitation by voluntary activity that may be associated with structural damage to these pathways. These impairments likely contribute to impaired voluntary movement.
Keywords
Introduction
Difficulty with voluntary muscle activation contributes to impaired movement in people with spastic cerebral palsy (CP). However, the strength of descending motor pathways in CP is yet to be comprehensively characterized. We do know that following an injury to the developing brain resulting in a disorder of movement and posture, 1 people with spastic CP have muscular weakness2,3 mediated, in part, by reduced activation of spinal motoneurons.4-6 While it has long been postulated that damage to descending pathways from the brain that project to spinal motoneurons may contribute to motor impairments in spastic CP, 7 the evidence of this is incomplete. In this study, we used transcranial magnetic stimulation (TMS) to characterize the activation of corticospinal pathways in adults with bilateral spastic CP. Using diffusion tensor imaging (DTI), we also evaluated anatomical microstructure abnormalities that may contribute to dysfunction of these pathways.
Activation of corticospinal pathways from TMS, which include the corticospinal tracts and tracts that connect the cortex and spinal cord via synapses in the brainstem, 8 has only been explored to a limited extent previously in people with spastic CP. Studies have focused on outcomes that can be determined with relatively low intensities of stimulation, such as hotspot locations9,10 and motor thresholds.11-14 Although it is expected that corticospinal pathways that are contralateral to affected muscles are weaker, surprisingly motor-evoked potentials (MEPs) evoked from the contralateral motor cortex that are smaller compared to neurologically intact controls has not been demonstrated.14,15 In the majority of studies in people with CP, MEPs were measured with the participants at rest, and thus, did not examine the stimulation of corticospinal pathways during voluntary motor activity. For instance, MEPs recorded in the soleus muscle at rest were actually larger compared to controls 15 and leaves open the question of how voluntary activity influences corticospinal tract recruitment in CP. Moreover, while MEPs increase in size with increasing levels of voluntary activity in neurologically intact populations,16-19 the influence of this has also not been previously examined in people with CP.
Most anatomic evaluations of the corticospinal pathways using DTI also characterize both the corticospinal and cortico-brainstem tracts.20,21 Studies consistently demonstrate a loss of white matter integrity in corticospinal pathways in children with spastic CP. 22 These pathways are known to develop into adulthood. 23 However, most of the previous studies have focused on children and adolescents and thus cannot distinguish between differences in developmental trajectories and differences in white matter integrity that will persist in adults.
Here we examined the activation and anatomic integrity of the corticospinal pathway using TMS and diffusion tractography in adults with bilateral spastic CP. We recorded TMS responses in the soleus muscle because plantarflexor muscles in people with CP exhibit greater weakness and spasticity contributing to gait impairments than the more commonly studied dorsiflexors.24,25 TMS responses to a large range of stimulation intensities evoked during voluntary muscle activity were compared to neurologically intact (NI) participants of similar age and sex distribution. We also examined how increasing voluntary activity impacted the activation of corticospinal pathways. In a subgroup of these CP and NI participants that were age and sex matched, we used diffusion tractography to determine if the anatomical integrity of the corticospinal pathway projecting to all muscles was also reduced in adults with CP.
Methods
Participants
This study was approved by the Health Research Ethics Board at the University of Alberta (Pro00023530), in accordance with the Declaration of Helsinki. All participants provided written informed consent. Sixteen adults with spastic CP (9 female, 7 male, age 19-57 years, Table 1) and 15 neurologically intact (NI) controls (9 female, 6 male, age 19-59 years) participated in this study. No participant had absolute contraindications to TMS, 26 botulinum toxin injections in the lower leg within the past 2 years, or dorsal rhizotomy. Participants with CP underwent clinical assessment of the Gross Motor Function Classification System (GMFCS) 27 and plantarflexor strength (Medical Research Council grading, MRC) using a manual muscle break test.28,29 The maximum MRC score of 5 may have been overestimated as testing was performed in a non-weightbearing position. Plantarflexor spasticity was assessed using the modified Ashworth scale 30 and the Penn Spasm Frequency scale 31 by a physician (author EGC) with a score of 1 or more in either scale indicating spasticity.
Setup
Each participant took part in 3 TMS protocols during a single experimental session: location of the soleus MEP hotspot, stimulus-recruitment curve, and voluntary modulation of MEPs. In the CP group, the leg that interfered more with function (referred to as the target leg) was tested. This was defined by the participant or on clinical examination of strength, spasticity, and voluntary movement in a few cases where the participant was uncertain. The right leg was targeted in the NI group except in 3 participants with previous right ankle injuries. Throughout the experiment, participants were seated comfortably with their knee at 80° to 90° of flexion and their ankle stabilized at 90°. A lap belt and leg straps were used to minimize movements caused by co-activation of other muscles particularly given the known impairments in selective motor control in people with CP. 32 Surface electromyography (EMG) was recorded from the soleus muscle in the target leg using pairs of conductive adhesive hydrogel electrodes (3.81 × 2.24 cm, Covidien Ltd, Dublin, Ireland). EMG signals were amplified (1k gain), filtered (10-1000 Hz) (AMT-8, Bortec Biomedical Ltd, Calgary, Alberta, Canada), digitized and sampled at 5 kHz using Axon hardware (Digidata 1440A) and software (Axoscope 10.3, Molecular Devices LLC, Sunnyvale, CA). Visual feedback of soleus activity was provided on an oscilloscope. Analysis of EMG data was performed offline using Matlab (R2011b, The Mathworks Inc, Natick, MA).
The maximal voluntary activity (MVA) of the soleus muscle over 1 second was determined for each participant following recommendations for optimizing maximal voluntary efforts. 33 The maximal M-wave (Mmax) was measured to determine the maximal evoked response possible in the soleus muscle. TMS was delivered using a custom batwing coil (P/N 15857: 90-mm wing diameter), designed to produce focal activation of the leg motor cortex and oriented to induce posterior-anterior current in the brain. TMS was delivered using a MagStim 200 stimulator or its BiStim module (The Magstim Company Ltd, Carmarthenshire, UK). See the Supplementary Material (available in the online version of the article) for further details on MVA, Mmax, and the stimulators.
TMS Protocols
To ensure we obtained the largest MEP responses possible, we determined the location of the soleus hotspot, which was defined as the position resulting in the largest MEP (average of 4 responses) produced by submaximal stimulation with a background contraction of 20% MVA (see “Hotspot Localization” in the Supplementary Material for further details). If consistent responses could not be elicited from the cortex contralateral to the target leg, as occurred in 4 of the participants with CP, TMS was applied to the ipsilateral cortex to verify that responses could be elicited in that muscle. The Euclidian distance between the vertex and hotspot was calculated for all participants with contralateral responses.
Stimulus-recruitment curves were generated while participants performed submaximal contractions (20% MVA) to evaluate the relationship between the stimulation intensity and MEP amplitude. The active motor threshold (aMT), defined as the lowest stimulation intensity that produced a reproducible waveform at the MEP latency in 3/5 individual responses (as per Palmer et al 34 and Roy et al 35 ), was first determined. Stimulation intensities above and below aMT were then applied in steps no greater than 0.2 × aMT with finer adjustments at high intensities to ensure we obtained the largest MEP amplitude possible. Five stimulations were applied at each TMS intensity, and the peak-to-peak amplitude of each MEP was averaged across the 5 trials.
To evaluate the ability to voluntarily modulate descending pathways, the relationship between MEP amplitude and voluntary activity was explored. Participants varied their background contractions from rest to maximal activation while TMS was delivered at a fixed intensity (median 34 trials). This was performed at 2 TMS intensities: 1.2 × aMT and the intensity that produced the largest MEP amplitude in the stimulus-recruitment curve.
MEP amplitudes are expressed in raw peak-to-peak voltage and separately normalized by Mmax. Normalization can introduce additional sources of error but does reduce the impact of differences in factors that impact surface EMG such as lower muscle mass in people with CP. Three measures were used to compare the activation of corticospinal pathways by TMS between the CP and NI groups. (1)
Magnetic Resonance Imaging and Diffusion Tractography
On a separate day from the TMS experiments all participants with CP, except one with a contraindication to magnetic resonance imaging (MRI) (15/16), were scanned using a 1.5T Siemens Sonata. Eleven of the 15 NI participants were scanned; three were not included in the MRI protocol, and one declined to be imaged. Sequences included T1-weighted, T2-weighted, fluid-attenuated inversion recovery (FLAIR), and diffusion tensor imaging (DTI) for a total scan time of ~20 minutes. DTI was acquired using a dual spin-echo, single shot echo-planar imaging sequence with 40 axial-oblique slices, 3-mm slice thickness with no interslice gap; repetition time (TR) = 6900 ms; echo time (TE) = 100 ms; 30 non-collinear gradient directions with b = 1000 s/mm2, 1 b = 0 s/mm2, 2 averages, field of view (FOV) 220 × 220 mm2, matrix of 128 × 128 with 75% phase partial Fourier zero-filled to 256 × 256, acquisition time 8:11 minutes. A radiology resident and a neuroradiologist (authors DTJ and DJE) characterized any anatomical abnormalities as per Towsley (2011) 36 and provided a qualitative ranking of severity.
While MEP responses are stable in the age range of participants in this study,
37
DTI metrics are known to undergo an age-dependent evolution.
38
Therefore to evaluate the differences in the anatomic integrity of the corticospinal pathways between the CP and NI groups, diffusion tractography was performed in a subset of participants who were individually sex- and age-matched (11 in each group, see boldfaced IDs in Table 1). Partial tracts
39
contralateral to the target leg between the posterior limb of the internal capsule and basis points were identified using deterministic methods in ExploreDTI v4.8.3.
40
Fractional anisotropy (FA), mean diffusivity (MD), parallel diffusivity (
Statistical Analysis
Statistical analysis was performed in SPSS (version 21, IBM, Armonk, NY). Because most parameters were not normally distributed, as determined from the Shapiro-Wilk test, groups were described by their median and range values and compared using nonparametric statistics. Differences between groups were evaluated using the non-parametric Mann-Whitney U test. To determine if differences noted in the activation of corticospinal pathways were related to structural differences, associations between MEP parameters and DTI measures in the group with CP were explored with the non-parametric Spearman rank correlation. The alpha level for significance was set at .05.
Results
Participant Characteristics
As shown in Table 1, participants in the CP group represented a range of functional abilities (GMFCS I-IV) and motor impairments (MRC 1-5, modified Ashworth Scale 0-3, Penn 1-3). Almost all (14/15 participants who underwent MRI) had evidence of bilateral brain lesions (see Supplementary Figure S1 for representative slices). The diagnosis of bilateral CP for the remaining participant with CP who had no visible brain lesions (CP-14) and the participant without an MRI (CP-4) was confirmed with review of their clinical records.
Demographics and Clinical Measures of Participants With Cerebral Palsy. a
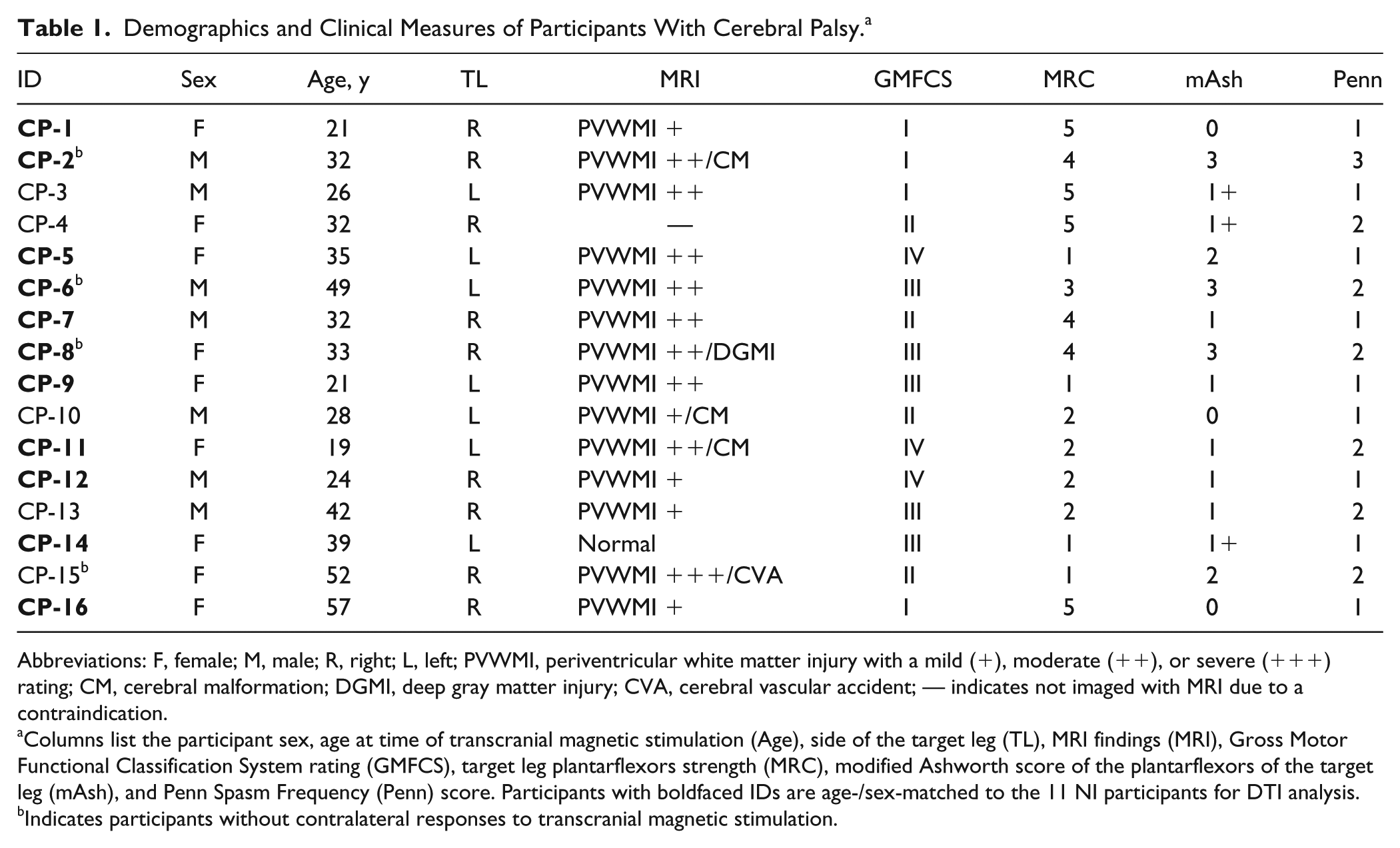
Abbreviations: F, female; M, male; R, right; L, left; PVWMI, periventricular white matter injury with a mild (+), moderate (++), or severe (+++) rating; CM, cerebral malformation; DGMI, deep gray matter injury; CVA, cerebral vascular accident; — indicates not imaged with MRI due to a contraindication.
Columns list the participant sex, age at time of transcranial magnetic stimulation (Age), side of the target leg (TL), MRI findings (MRI), Gross Motor Functional Classification System rating (GMFCS), target leg plantarflexors strength (MRC), modified Ashworth score of the plantarflexors of the target leg (mAsh), and Penn Spasm Frequency (Penn) score. Participants with boldfaced IDs are age-/sex-matched to the 11 NI participants for DTI analysis.
Indicates participants without contralateral responses to transcranial magnetic stimulation.
No differences were found between the participant ages (P = .61) or sex distributions (P = .83) between the 2 groups. As expected, the group with CP displayed motor weakness in the plantarflexors as reflected in the MRC grades and in lower maximal voluntary muscle activity (MVA). See “MVA” in the Supplemental Text for further details.
Location of Soleus MEP Hotspot
MEP responses in the soleus muscle were evoked from the contralateral cortex in all but 4 participants with CP (see footnote b in Table 1) and in all NI participants. The hotspots for soleus MEPs in the NI group were located 2 to 3 cm lateral from vertex in a tight cluster (open circles, Figure 1A). In contrast, the hotspot locations in the CP group were farther from the vertex (P < .001, Figure 1B, Table 2), more dispersed and very lateral in some individuals (solid circles, Figure 1A).
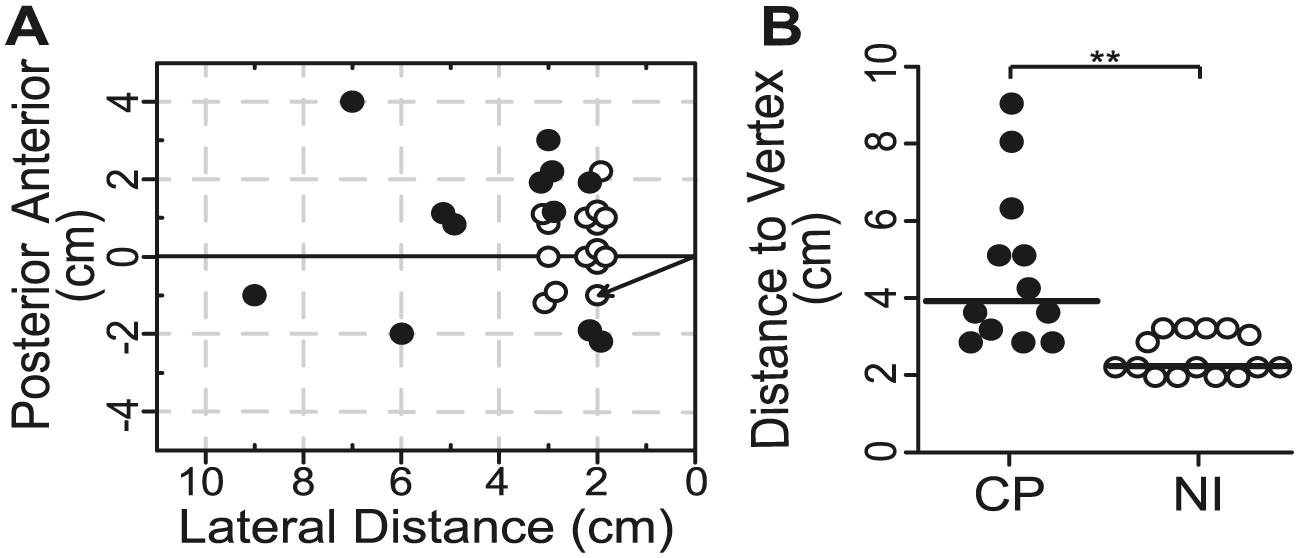
Location of soleus motor-evoked potential (MEP) hotspot. (A) Cartesian coordinates of the soleus MEP hotspot in the contralateral cortex for each cerebral palsy (CP; solid circles, n = 12) and neurologically intact (NI; open circles, n = 15) participant relative to vertex (0, 0) where contralateral MEPs could be obtained. The Euclidian distance from vertex is marked by the arrow for one NI participant. (B) Values of the Euclidian distance from vertex for each of the CP and NI participants shown in A. Horizontal lines represent the group medians. **P < .001.
Comparison of TMS and DTI Measures Between Groups. a
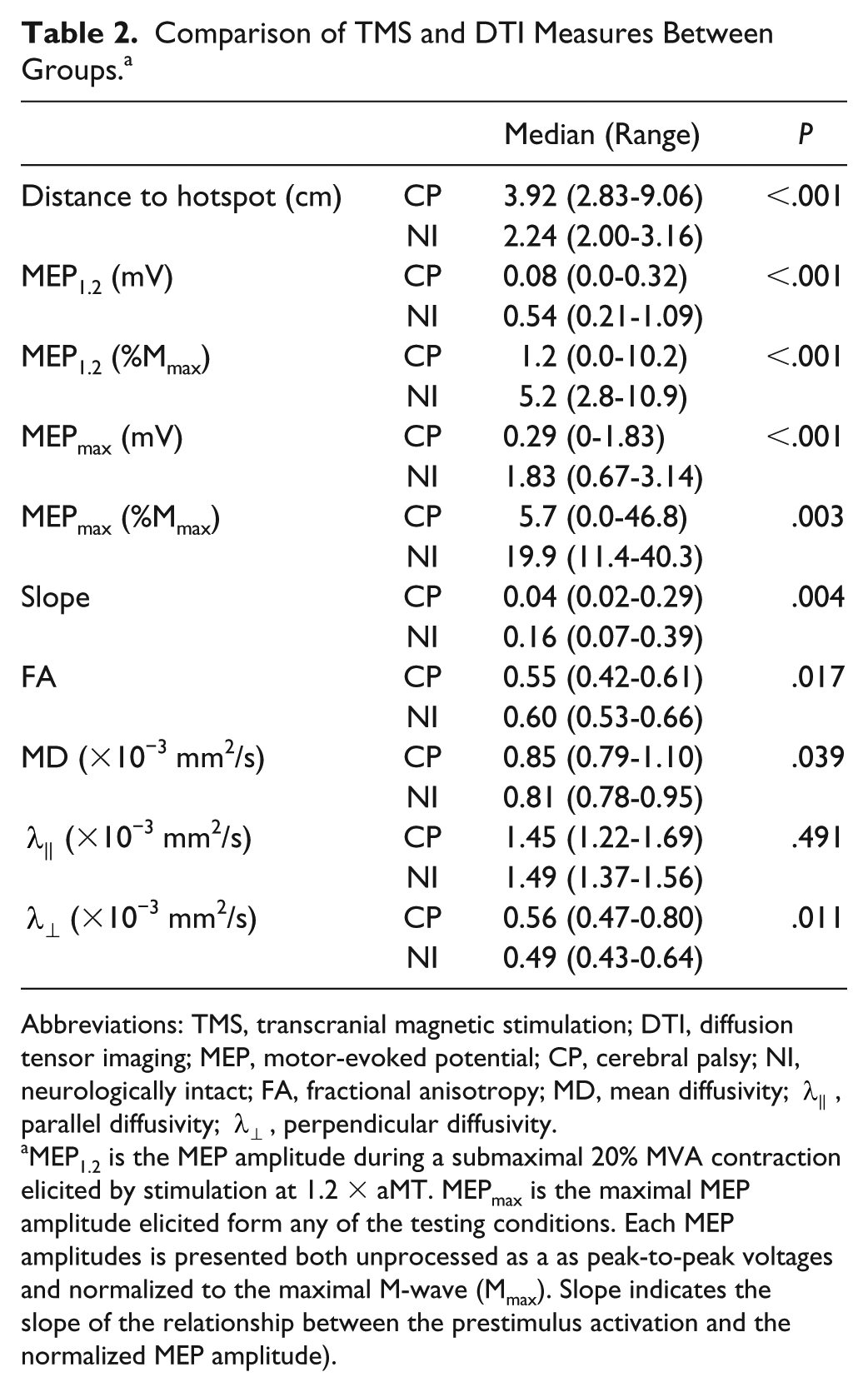
Abbreviations: TMS, transcranial magnetic stimulation; DTI, diffusion tensor imaging; MEP, motor-evoked potential; CP, cerebral palsy; NI, neurologically intact; FA, fractional anisotropy; MD, mean diffusivity;
MEP1.2 is the MEP amplitude during a submaximal 20% MVA contraction elicited by stimulation at 1.2 × aMT. MEPmax is the maximal MEP amplitude elicited form any of the testing conditions. Each MEP amplitudes is presented both unprocessed as a as peak-to-peak voltages and normalized to the maximal M-wave (Mmax). Slope indicates the slope of the relationship between the prestimulus activation and the normalized MEP amplitude).
Stimulus-Recruitment Curve
MEPs evoked in the contralateral soleus muscle of participants with CP were generally smaller and their shape was less consistent than the MEPs measured in NI control participants (Figure 2A, note the different vertical scales). While MEP amplitudes increased for stimulation intensities above the active motor threshold (aMT) until reaching a plateau or peak in all participants in the NI group (eg, NI-7 in Figure 2B), full stimulus-recruitment curves during submaximal contractions (20% MVA) could not be generated in most participants with CP (14/16 participants with CP) prior to reaching the maximum stimulator output (eg, CP-4 in Figure 2B). In 2 participants with good motor function (CP-3 and CP-16), MEP amplitudes plateaued prior to reaching the limits of the stimulator, creating stimulus-recruitment curves with a profile similar to the NI participants. Interestingly, expressing the TMS intensity relative to aMT revealed that a similar range of intensities were explored in both groups. Specifically, the intensity that produced the largest MEP for each participant was 1.56 (1.15-2.00) × aMT in the CP group and 1.56 (1.40-2.11) × aMT in the NI group (P = .56).
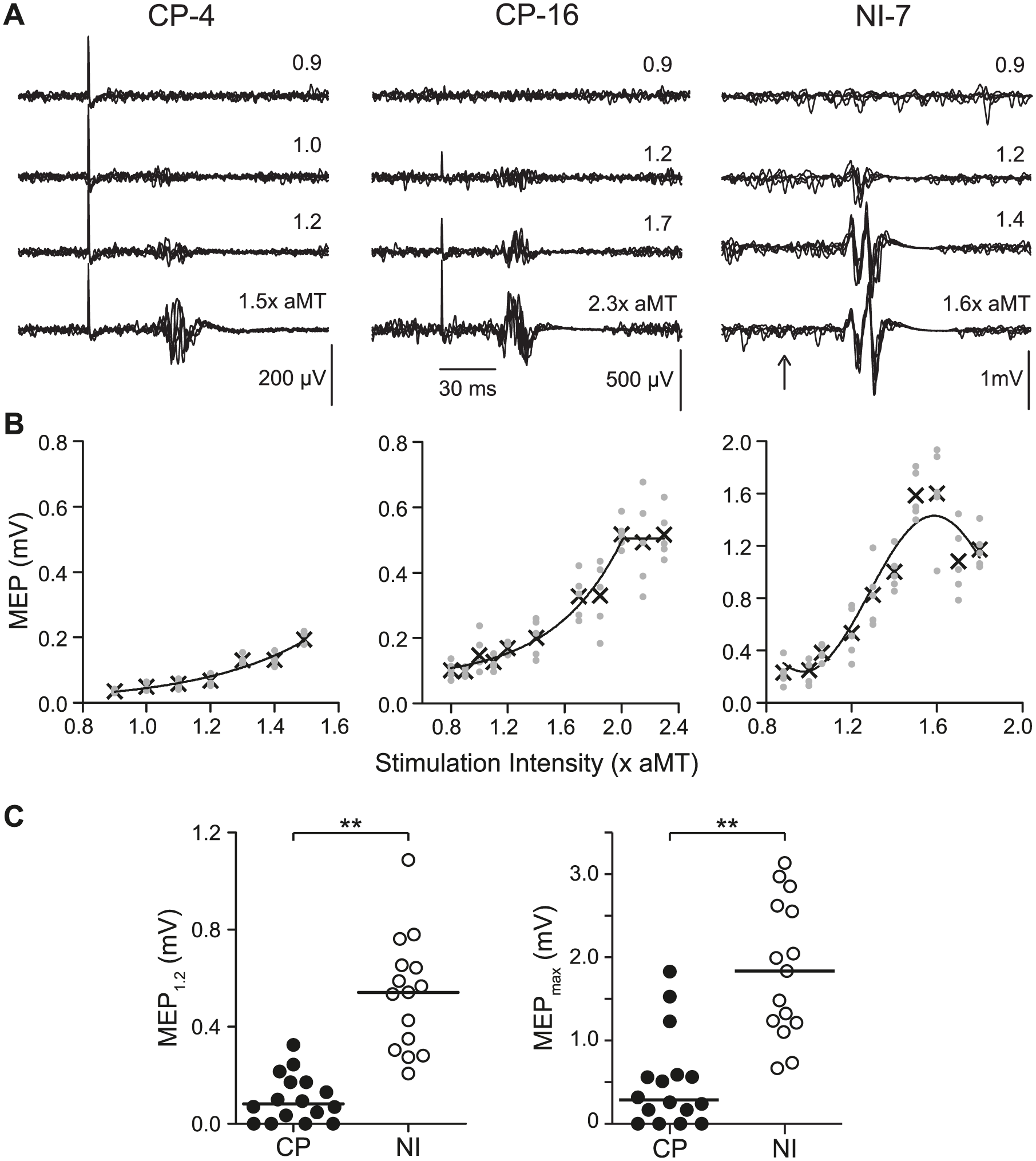
Motor-evoked potentials (MEPs). Example soleus MEPs (A) and stimulus-recruitment curves (B) are shown for 3 representative participants. (A) Five traces of unrectified electromyogram (EMG) obtained during submaximal activation are superimposed at incrementing intensities of stimulation expressed as a multiple of the active motor threshold (× aMT). The time of stimulation is marked by the stimulus artifact in CP-4 and CP-16 and by the vertical arrow for NI-7. (B) Stimulus-recruitment curves demonstrate the relationship between stimulation intensity and MEP amplitude. Each gray dot represents a single MEP and Xs indicate the mean MEP amplitude at each intensity. (C) Dot-plot comparison of MEP1.2 and MEPmax amplitudes between the CP (solid circles) and NI (open circles) groups. Horizontal lines represent the group medians. Note values of 0.0 mV were used for the 4 participants with CP who did not have contralateral MEP responses. **P < .001. CP, cerebral palsy; NI, neurologically intact.
MEP Amplitude
MEP amplitudes evoked from the contralateral motor cortex were smaller in the CP group compared with NI control participants (Figure 2C). Low threshold MEPs (MEP1.2), that is, MEPs evoked during submaximal (20% MVA) background contractions at a fixed low stimulation intensity (1.2 × aMT), were smaller in the CP group than the NI group (P < .001, Table 2). Evaluating higher threshold pathways with MEPmax also revealed significantly smaller responses in the CP group (P < .001, Table 2). These MEPs in the CP group were smaller irrespective of whether amplitudes were compared in raw magnitude (mV) or normalized to Mmax (Ps <.005, Table 2).
In the 4 participants with CP having MEPs evoked from the ipsilateral cortex only, MEPmax from these participants (CP-2, CP-6, CP-8, and CP-15, footnote b in Table 1) were 0.65, 0.62, 0.94, and 0.15 mV respectively, compared with the median contralateral MEPmax of 0.29 mV (Table 2). Description of the data from the ipsilateral cortex in these participants and statistical analysis are not described further because of the small number of participants.
Voluntary Modulation of MEPs
Voluntary modulation of corticospinal pathways was explored by evoking MEPs at different levels of background activation from rest to maximum. The slope of the relationship between MEP amplitude, expressed as a percentage of a participants maximal evoked response (Mmax), and background activation, expressed as a percentage of their maximum activation (MVA), was then calculated (as illustrated in Figure 3A for a CP and NI participant). Increasing voluntary activity increased MEP amplitudes to a lesser extent in participants with CP compared with NI participants (Figure 3B). In fact, the median slope of the relationship between MEP amplitude and background voluntary activity was 4 times lower in participants with CP than in NI participants (P = .004, Table 2).
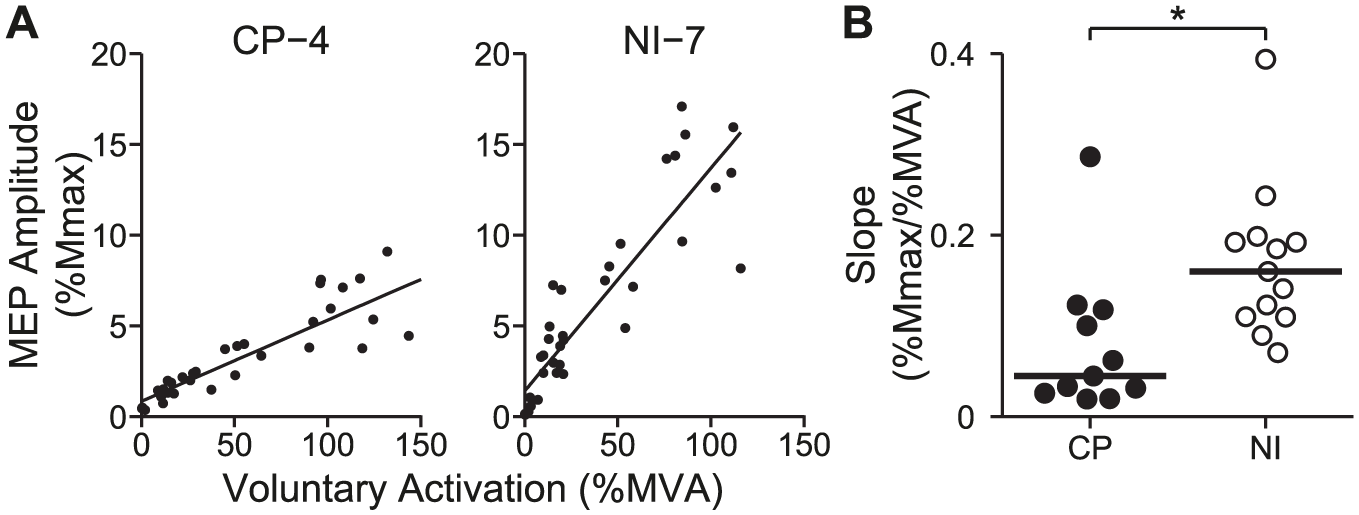
Voluntary modulation of motor-evoked potentials (MEPs). (A) The relationship between voluntary activity (% MVA) and MEP amplitude (expressed as a percentage of Mmax) plotted for a representative neurologically intact (NI) (left) and a cerebral palsy (CP) (right) participant. Transcranial magnetic stimulation (TMS) was applied at 1.2 × aMT. It is worth noting here that levels of voluntary activity could exceed 100% MVA given that the voluntary activity measured 100 ms before the TMS pulse could be greater than 100% MVA, which is the voluntary activity averaged over 1000 ms used to quantify the MVA at the beginning of the experiment. (B) Slope of the relationship between voluntary activity and MEP amplitude for each CP (solid circles) and NI (open circles) participant. Horizontal lines represent the group medians. Data from 2 NI and 1 CP participant is omitted due to problems with Mmax. *P < .05.
Diffusion Tractography
Tractography successfully generated streamlines attributed to the corticospinal pathway that was contralateral to the target leg in all 11 age- and sex-matched pairs of CP and NI participants (examples in Figure 4A). The tendency for water molecules to diffuse in a preferred direction, that is, fractional anisotropy (FA), was lower in the CP group (P = .017, Table 2, Figure 4B). Overall there was less restriction to diffusion (higher mean diffusivity, MD) in the CP group (P = .039), which appeared to be driven by an increase in the perpendicular diffusivity,
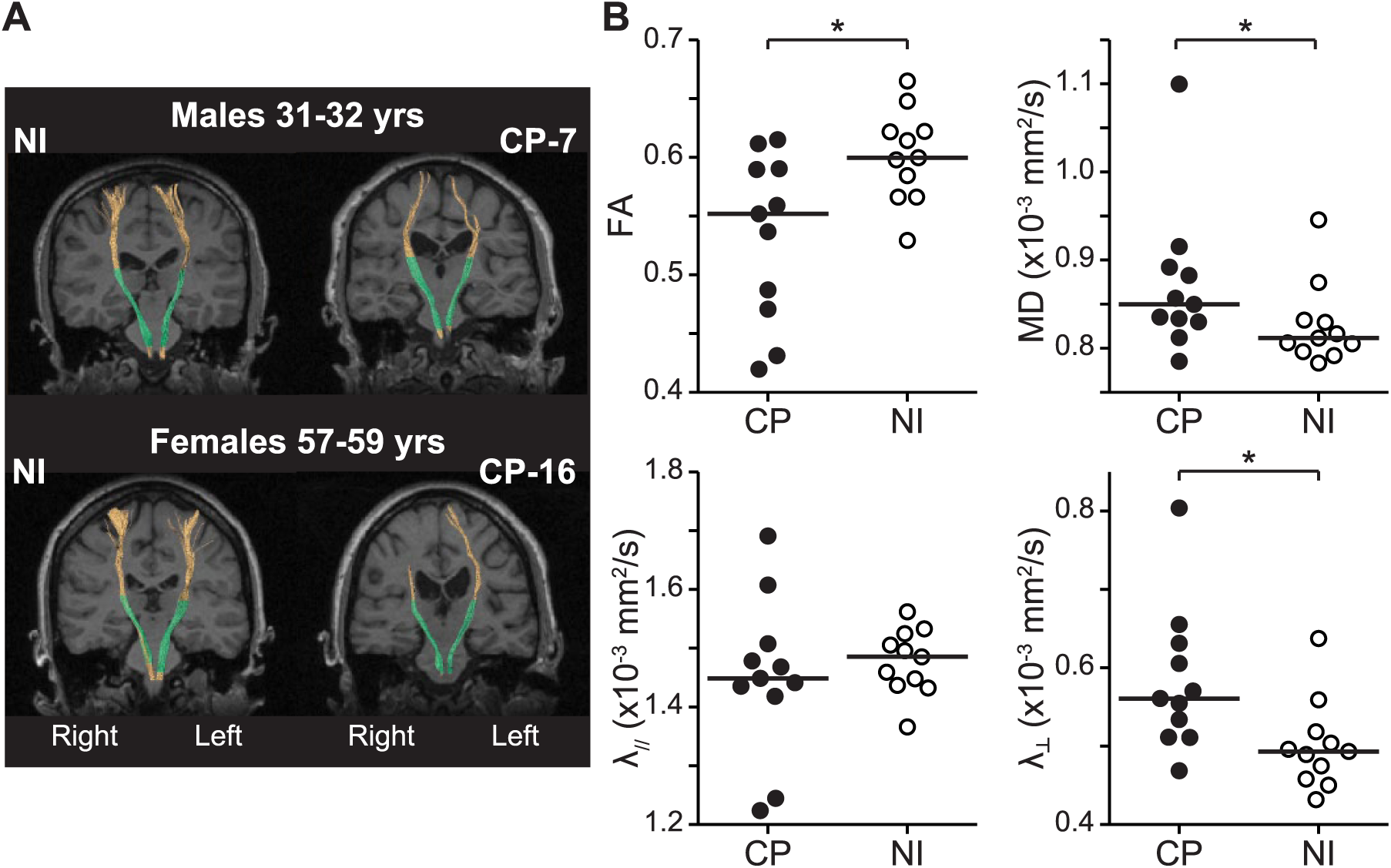
Tractography of corticospinal pathway. (A) Tractography of the corticospinal pathway superimposed on a coronal T1-weighted image for representative sex- and age-matched pairs of neurologically intact (NI) (left) and cerebral palsy (CP) (right) participants. The sex and age range are indicated above each pair. Full tracts are shown in orange and the analyzed partial tracts between the “seed” and “target” region of interest (ROI) are displayed in green. (B) Fractional anisotropy (FA, top left), mean diffusivity (MD, top right), parallel diffusivity (
Because some participants with CP had DTI values similar to controls (Figure 4B), we examined if there was an association between corticospinal tract integrity, as assessed with DTI, and activation of corticospinal pathways with TMS, as assessed with MEP measures. There were significant associations between MEPmax (both raw voltage and normalized to Mmax) and FA at r = 0.65 (P = .030) for MEPmax (mV) and r = 0.66 (P = .039) for MEPmax (%Mmax). No associations were found between FA and MEP1.2 or slope, nor were any associations found between MD or parallel diffusivity (
Discussion
This is the first study comparing participants with spastic CP and NI control participants to examine full activation profiles of corticospinal pathways using a large range of TMS intensities and voluntary muscle activity. The results demonstrate that in people with spastic CP, the activation of corticospinal pathways by TMS produces smaller MEPs both when activated at a relatively low intensity (1.2 × aMT) and when under conditions that produce the largest responses for that individual (MEPmax). Furthermore, as increasing the level of voluntary muscle activity provided less facilitation of MEP responses, the ability to modulate excitability in their motor pathways is impaired in people with CP. All the observed differences existed in both raw and normalized MEP values. These novel findings provide direct evidence that impairments in the function of corticospinal pathways occur and likely contribute to impaired muscle activation in people with bilateral spastic CP.
Impaired Activation of MEPs in People With CP
There have been 2 previous studies comparing the magnitude of activation of contralateral corticospinal pathways by TMS between people with CP and NI peers.14,15 In one study performed in children and adolescents with bilateral CP, the amplitude of MEPs measured in the tibialis anterior was not significantly smaller compared with the control group despite the mean MEP amplitude being half the magnitude in the CP group. 14 The lack of statistical difference likely resulted from high variability in both groups. MEPs were produced at a low TMS intensity while participants performed a maximal effort, which likely contributed to this variability. In the second study performed in adolescents and adults with mainly spastic diplegia, 15 soleus MEPs were the same amplitude as in the control group when measured at rest, in contrast to our findings. The smaller amplitudes of the soleus CP MEPs in the current study may have resulted, in part, from a reduced facilitation of MEPs from voluntary activity, as demonstrated by the shallower slope of the MEP/MVA relation (see Figure 3). Moreover, even though participants with CP could produce graded activation of the soleus muscle, there was likely activation of other agonist and antagonist muscles.32,41 Thus, a less focused and isolated activation of circuits within the soleus motor cortex may have also contributed to the reduced soleus MEPs in the CP group. Evidence that a different circuit organization was present in some of the participants with CP group is suggested by the marked lateral location of the soleus hotspot as shown here and by others,9,10 with some shifts between 4 and 7 cm, much larger than would be expected by differences in head size and shape alone.
Smaller MEPs were found both through lower threshold pathways (1.2 × MT) during submaximal voluntary activity and through higher threshold pathways (MEPmax). Because of the technical limit of the stimulator (ie, maximum stimulator output), a clear maximal MEP could not be found during a 20% MVA contraction (ie, during the stimulus-recruitment curve testing) despite testing a similar range of stimulation intensities relative to each participant’s active motor threshold. It seems likely that the stimulus-recruitment curve may be more prolonged in people with CP, requiring higher intensities to reach a maximum. However, using higher levels of voluntary muscle activity, we were able to compare maximal MEPs for each individual and demonstrate responses evoked through higher threshold pathways are also smaller in people with CP.
The reduced amplitude of soleus MEPs in the present study could have also resulted from microstructural damage to corticospinal and/or cortico-brainstem47 tracts as assessed by the DTI parameters FA and mean, parallel, and perpendicular diffusivity. The magnitude of anisotropic diffusion, or FA, is thought to be mainly related to axonal count and density, as well as modulation by the degree of myelination and fiber organization.42,43 It has also been shown that parallel and perpendicular diffusivity can sometimes be related more specifically to axonal and myelin integrity, respectively, in cases of known timing-dependent Wallerian degeneration.44,45 The pattern of reduced FA and increased perpendicular diffusivity (as the cause of the reduced anisotropy) suggests reduced or damaged myelination of corticospinal pathways and/or axonal loss within them.44-46 This is in agreement with the finding that FA and perpendicular diffusivity have greater correlation to white matter damage compared with mean and parallel diffusivity.46,48 The positive association between FA and MEPmax and the negative trend for perpendicular diffusivity supports the idea that reduced corticospinal axon and myelin integrity contributed to the reduced maximum activation of soleus motoneurons by TMS. It is interesting that these associations and trends occurred given that the DTI parameters represent all the corticospinal tracts and not just those projecting to soleus motoneurons. The lack of association to MEP1.2 and slope may be due to these TMS values reflecting factors more closely related to the excitability of the corticospinal pathway rather than total tract connectivity. However, larger numbers of comparisons are needed to resolve these ideas more clearly.
Reduced Voluntary Modulation of Soleus MEPs
With increasing voluntary muscle activity, normalized MEP amplitudes increased to a lesser degree in the participants with CP (Figure 3). This is consistent with previous indirect evidence of impaired descending modulation of spinal motoneurons. People with CP were unable to fully activate all their motor units and submaximal contractions required higher levels of voluntary effort to reach similar degrees of increased motor unit recruitment and firing rates. 4 Taken together with our results, this suggests that in people with CP, voluntary activity must be increased further before the proportion of the spinal motor neuron pool available for activation by TMS reaches a comparable magnitude to that of people without neurologic injuries.
Increasing voluntary activation of muscles increases both cortical and spinal excitability facilitating the amplitude of MEPs.16,19 Given that spinal circuits are either more excitable or unchanged in people with spastic CP and not more inhibited,49,50 the reduced voluntary facilitation of MEPs in people with CP may be predominantly mediated by cortical mechanisms. Evidence for cortical mechanisms comes from recording descending volleys (I-waves) in the epidural space of the spinal cord that are evoked by TMS. 51 Increasing voluntary contractions increase the amplitude and number of I-waves, providing larger activation of spinal motorneurons. 16 Thus, the reduced efficacy of corticospinal pathways to facilitate spinal motoneurons likely causes the inefficient voluntary activation of muscle in CP.5,52
Implications
People with bilateral spastic CP demonstrate impaired activation of low- and high-threshold corticospinal pathways to soleus motoneurons by TMS and impaired modulation of these pathways by voluntary activity. This was found despite heterogeneity within the CP group (see “Heterogeneity in the Participants With CP” in the Supplementary Material), making the result more generalizable to the broad population of people with bilateral spastic CP. These impairments may be indicative of reduced function in corticospinal pathways that mediate difficulties with voluntary movement including weakness, particularly as it contributes to reduced push-off in gait and during graded contractions required for skilled walking. 25 In line with this, our data corroborate recent findings in adults with CP that suggest reduced descending drive to the soleus muscle cannot compensate for its reduced sensory activation during push-off. 53 Future studies examining TMS responses during different phases of movement may shed light on how early brain injury affects the functional organization and recruitment of cortical circuits and corticospinal pathways in CP and how these pathways respond to interventions.
Supplemental Material
Supplemental_Materials – Supplemental material for Full Activation Profiles and Integrity of Corticospinal Pathways in Adults With Bilateral Spastic Cerebral Palsy
Supplemental material, Supplemental_Materials for Full Activation Profiles and Integrity of Corticospinal Pathways in Adults With Bilateral Spastic Cerebral Palsy by Elizabeth G. Condliffe, Dean T. Jeffery, Derek J. Emery, Sarah Treit, Christian Beaulieu and Monica A. Gorassini in Neurorehabilitation and Neural Repair
Footnotes
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Authors’ Note
All data from this article can be provided by contacting the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding for this work was provided by CIHR MOP106549. E.G.C. was supported through the Clinical Investigator Program and Alberta Innovates: Health Solutions 201100202.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
