Abstract
Background. The trunk plays a critical role in attenuating movement-related forces that threaten to challenge the body’s postural control system. For people with Parkinson’s disease (PD), disease progression often leads to dopamine-resistant axial symptoms, which impair trunk control and increase falls risk. Objective. This prospective study aimed to evaluate the relationship between impaired trunk muscle function, segmental coordination, and future falls in people with PD. Methods. Seventy-nine PD patients and 82 age-matched controls completed clinical assessments and questionnaires to establish their medical history, symptom severity, balance confidence, and falls history. Gait characteristics and trunk muscle activity were assessed using 3-dimensional motion analysis and surface electromyography. The incidence, cause, and consequence of any falls experienced over the next 12 months were recorded and indicated that 48 PD and 29 control participants fell at least once during this time. Results. PD fallers had greater peak and baseline lumbar multifidus (LMF) and thoracic erector spinae (TES) activations than control fallers and nonfallers. Analysis of covariance indicated that the higher LMF activity was attributable to the stooped posture adopted by PD fallers, but TES activity was independent of medication use, symptom severity, and trunk orientation. Furthermore, greater LMF and TES baseline activity contributed to increasing lateral head, trunk, and pelvis movements in PD fallers but not nonfallers or controls. Conclusions. The results provide evidence of neuromuscular deficits for PD fallers that are independent of medications, symptom severity, and posture and contribute to impaired head, trunk, and pelvis control associated with falls in this population.
Introduction
Parkinson’s disease (PD) is a heterogeneous neurodegenerative condition characterized by early symptoms of resting tremor, 1 slowness of movement, 2 and muscle rigidity. 2 However, with disease progression, patients may develop symptoms of postural instability 2 and difficulties with initiating and maintaining rhythmic walking patterns (eg, freezing of gait) that are often only partially responsive to pharmacological intervention.3,4 Declines in physical functioning expose people with PD to a 9 times greater risk of recurrent falls,5,6 and prospective research shows up to 65% of people with PD will fall at least once in a 12-month period.7-10 Unfortunately, this increased risk of falling is compounded by the presence of impaired postural responses,11,12 which significantly increase the risk of injury and future hospitalization. Furthermore, the higher incidence of falls and fall-related injuries in individuals with PD leads to an increased fear of falling, activity limitation, and overall declines in independence and quality of life.
Prospective research shows more than 50% of falls experienced by people with PD occur during walking or transferring tasks, 13 implicating postural instability and gait difficulties as likely contributors to a large percentage of the falls experienced by this population. By adulthood, human locomotion has become a relatively autonomous skill that relies on the powerful and precisely timed contractions of skeletal muscles to effectively coordinate the body’s segments. To better understand the effects of PD on neuromuscular function during gait, research has traditionally focused on the lower limb muscles, while the neuromuscular control of larger segments is commonly overlooked. From a mechanical perspective, the trunk represents almost 50% of the body’s mass14,15; hence, effective control of this segment is essential to maintaining equilibrium during dynamic tasks. During both locomotor and nonlocomotor tasks, it is the role of the postural control system to coordinate the body’s segments to ensure that head movements are kept controlled, rhythmic, and predictable (ie, the head remains stable).16,17 The head is one of the body’s most important natural frames of reference, and given the significance of vision and vestibular function to postural control and orientation, any unpredictable or excessive movement of this segment has the potential to increase one’s risk of balance loss and/or falls during dynamic tasks.18-20 In bipedal gait, the structures of the trunk are believed to contribute to head stability by attenuating the forces associated with locomotion.16,21 This task requires a fine balance between trunk stiffness and mobility, 22 a balance that is likely affected in people with PD who develop axial motor symptoms that increase segmental rigidity and postural abnormalities. 23 The reduced capacity for the trunk to attenuate movement-related forces may help explain why PD fallers have an impaired capacity to stabilize their head during gait 7 and a reduced ability to adapt to more challenging walking surfaces. 24
Despite the importance of trunk control in maintaining equilibrium during locomotion, there is a relative lack of research aimed at better understanding the relationship between deficits in trunk muscle function and the higher incidence of falls in people with PD. As such, this prospective study aimed to assess trunk muscle activation during walking in people with PD and age-matched controls to examine whether differences in trunk muscle function influence head, trunk, and pelvis motion in these populations. Furthermore, to provide insight into the possible relationship between neuromuscular deficits, segmental motion, and falls in these populations, these measures were also contrasted for participants who prospectively reported falling and those who did not. Given research has shown that movements of the head and pelvis are larger7,24 and less rhythmic 25 for people with PD who fall, it was hypothesized that patients with PD would have increased trunk muscle activity compared with controls and that these differences would be most prominent in patients who prospectively report falls. Additionally, it was hypothesized that increased muscle activity would correspond with increased segmental motion, due to the established relationship between axial rigidity and postural instability for people with PD. 23
Methods
Study Population
Seventy-nine participants clinically diagnosed with idiopathic PD based on the United Kingdom Brain Bank Criteria 26 were recruited from neurology clinics and community support groups in Southeast Queensland between August 2011 and June 2013 (Figure 1). During the same period, 82 healthy age- and gender-matched controls were randomly recruited from the Brisbane metropolitan area via the Queensland state electoral roll. Prospective participants were sent an invitation via post, which outlined the aims, expected risk/benefits, and time commitment associated with the study and were encouraged to contact the research team to discuss their eligibility if they were keen to be involved. Participants were excluded if they had (a) recently undergone surgery; (b) a recurrent history of musculoskeletal injury; (c) an inability to walk without assistance; (d) significant visual (Bailey-Lovie high contrast visual acuity >0.30 logMAR) or cognitive (Addenbrooke’s Cognitive Examination score <82 out of 100 27 ) impairment; or (e) received deep brain stimulation. In accordance with the Declaration of Helsinki, all participants provided written informed consent, and the experimental protocol was approved by the Human Research Ethics Committee at the Australian Catholic University (Approval #Q2011 04). Given the lack of literature concerning differences in muscle activation in people with PD who fall, a sample size estimate was derived using lower limb muscle activations presented previously for PD patients and age-matched controls. 28 On the basis of this calculation, it was determined that a minimum of 23 participants per group would be required to detect differences in muscle activation (effect size = 0.82, power = 0.8, P = .05). Given the falls rates reported for people with PD7,8 and community-dwelling older adults,29,30 it was anticipated that for a sample comprising 80 participants per group, approximately 52 PD patients (66%) and 26 controls (33%) would report at least one fall during the 12-month follow-up. As such, a target of 80 participants per group was deemed appropriate to not only achieve the required sample size per group but also to accommodate an attrition rate of up to 15%.
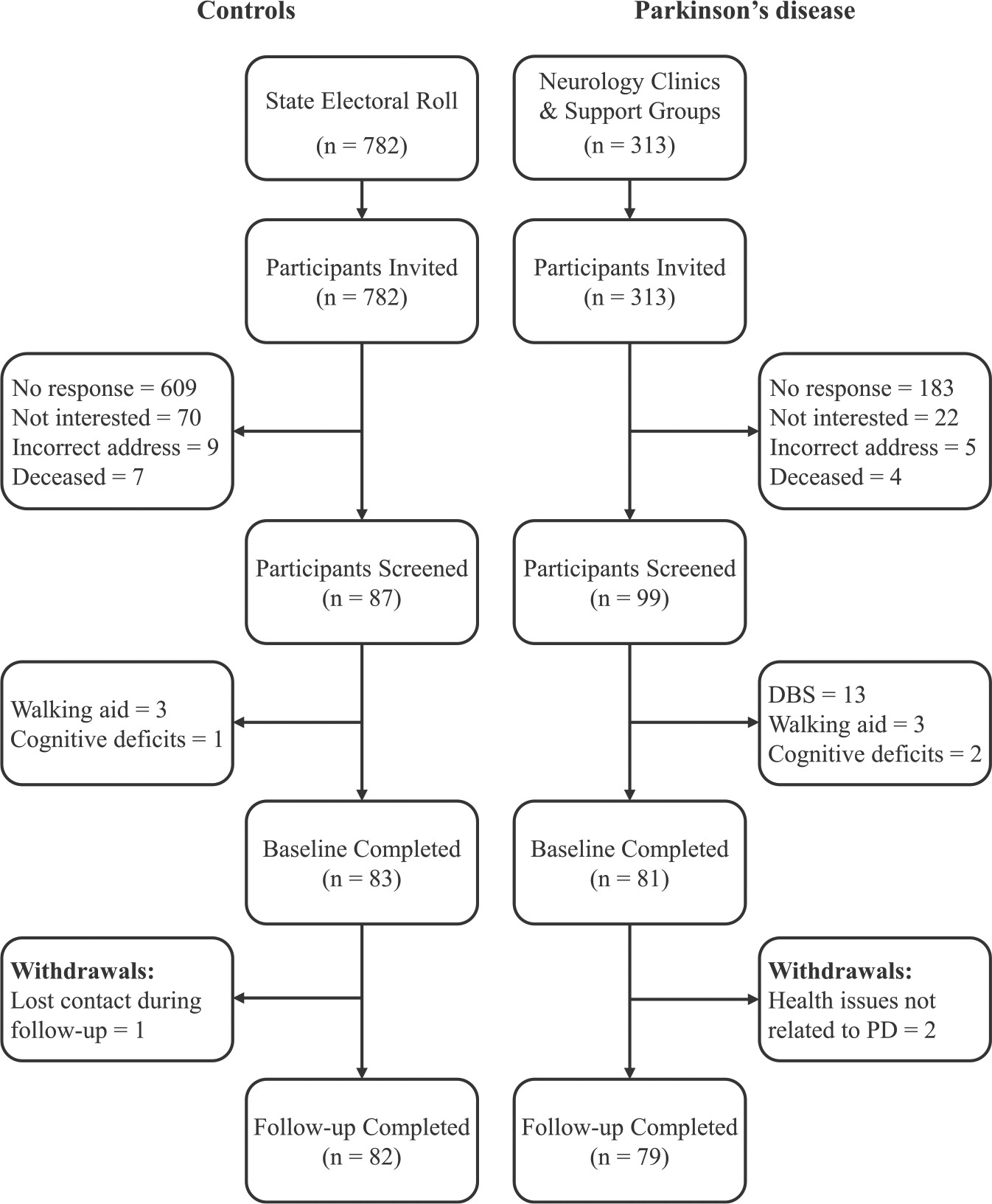
Flow diagram summarizing the recruitment, patient screening, data collection, and follow-up procedures.
Clinical Assessment
Participants completed a series of questionnaires and clinical assessments with an experienced movement disorders researcher to establish their medical history, symptom severity, and balance confidence. Specifically, symptom severity was assessed using the Unified Parkinson’s Disease Rating Scale (UPDRS), 31 the Hoehn and Yahr stage score, 32 and the Schwab and England Activities of Daily Living scale. 33 Additionally, freezing of gait and balance confidence were assessed using the Freezing of Gait questionnaire 34 and the Activities-specific Balance Confidence scale,35,36 respectively. Postural instability and gait disability (PIGD) were assessed by summing the scores for Items 13 to 15 and 27 to 30 from the UPDRS, 37 while PD subtypes 38 and a global rigidity score 39 were determined from the UPDRS using previously described methods. All assessments were completed within 1 to 2 hours of patients receiving their usual anti-parkinsonian treatment to ensure that they were optimally medicated. Of the 79 PD patients, 74 (93.7%) were being treated with levodopa or dopamine agonists.
Gait Assessment
The 3-dimensional walking patterns and bilateral activity of the superficial trunk muscles were evaluated as participants completed 4 self-paced and barefoot walking trials along a 9-meter-long firm walkway. To facilitate the assessment of stride characteristics and segmental control, 34 reflective markers were positioned on the participants in accordance with the full-body Vicon Plug-In Gait model (Vicon, Oxford, UK). Specifically, markers were attached to both legs (second metatarsal heads, lateral malleoli, calcanei, mid-shanks, lateral femoral epicondyles, mid-thighs), the pelvis (anterior-superior iliac spines, sacrum), the trunk (T10 and C7 spinous processes, xiphoid process, suprasternal notch, spine of right scapula), both arms (acromioclavicular joints, lateral humeral condyles, radial and ulnar styloids), and the head (bilaterally over the lateral brow and lambdoid sutures). The 3-dimensional positions of these markers were tracked (150 Hz) for 2 complete gait cycles (1 right; 1 left) within the central 6-meter section of the walkway by a previously calibrated 8-camera motion analysis system (Nexus 1.7; Vicon).
Data were reconstructed to facilitate the calculation of spatiotemporal characteristics, including walking velocity, stride length, stride frequency (cadence), and step width. Furthermore, the lateral and vertical displacements of the head, trunk, and pelvis were calculated and normalized to walking velocity, providing insight into the control and stability of these segments. Given that the stooped (flexed) trunk posture adopted by PD patients 40 has the potential to influence trunk extensor activity, differences in mean trunk flexion were assessed in the sagittal plane by calculating the angle formed between the longitudinal axes of the pelvis (defined by the anterior-superior iliac spines and sacrum markers) and trunk segments (defined by the T10, C7, xiphoid, suprasternal notch, and scapula markers). Finally, arm swing was calculated as the average linear displacement of both wrists in the sagittal plane.7,24 Differences in spatiotemporal characteristics have been previously reported for people with PD,41-43 and differences in head control has been observed in PD fallers while walking on firm 7 and compliant surfaces. 24
Trunk Muscle Function
Bilateral activation of the thoracic erector spinae (TES), lumbar multifidus (LMF), and external obliques (EO) were assessed using a 16-channel wireless surface electromyography (EMG) system (TeleMyo DTS, Noraxon USA Inc, Scottsdale, AZ). Pairs of Blue Sensor M-00-S silver-silver chloride surface electrodes with a pre-gelled area of 154 mm2 and an overall diameter of 34 mm (Ambu Inc, Glen Burnie, MD) were positioned bilaterally over the muscles of interest with a center-to-center distance of 34 mm. Specifically, the TES was monitored by electrodes positioned 5 cm lateral to the 10th thoracic spinous process, while electrodes placed 2 cm lateral to the 3rd lumbar spinous process recorded LMF activity. 44 Electrodes placed midway between the anterior superior iliac spine and the lowest point of the 10th rib recorded the bilateral activity of the EO muscles. 45 Prior to positioning the electrodes, each site was prepared with an abrasive paste (Nuprep Skin Prep, Weaver and Company, Aurora, CO) and cleaned thoroughly with isopropyl alcohol to reduce impedance. For individuals with excessive hair, the skin was shaved prior to abrasion to improve adherence of the equipment to the skin.
Myoelectric activity was sampled at 1500 Hz and at a resolution of 16-bit using wireless EMG transmitters with integrated pre-amplifiers (gain × 400, input impedance >100 MΩ, common mode rejection ratio >100 dB, noise = <1 µV). To assist with combining the myoelectric and motion caption data, these systems were synchronized via a 16-bit analog-to-digital converter.
Following data collection, the raw EMGs were detrended and cropped to include a complete gait cycle for each leg using the initial foot contact data derived from the motion analysis system. Data were then processed using a wavelet-based adaptive filter 46 to attenuate the influence of the electrocardiogram signal on trunk EMGs. Following full-wave rectification, data were low-pass filtered using a fourth-order dual-pass Butterworth filter (20 Hz cutoff) and processed using the root mean square (RMS) method with a 100-ms window length (150 samples) and an overlap of 149 samples. To minimize the likelihood of inadvertently filtering out important data, the cutoff frequency for the low-pass filter was informed by a residual analysis of the data and is commensurate with parameters used in previous research. 47
To facilitate group comparisons, EMGs were expressed as a percentage of the peak RMS amplitude of the EMGs collected during a series of 3 maximum voluntary isometric contraction (MVIC) performed for each of the muscles. As early research48,49 demonstrates that the rate of force development and relaxation is reduced in people with PD compared with age-matched controls, there is a risk that a patient’s MVIC may be underestimated if appropriate measures are not taken during data collection. To ameliorate this risk, participants performed a minimum of 2 submaximal practice trials to confirm that (a) they understood the movement that they would be required to perform and (b) to warm-up the muscles before the maximal efforts. Furthermore, as maximal force responses are more variable in people with PD, 49 each participant performed 3 maximal efforts separated by a minimum of 30-second rest. Finally, due to the slower rate of force production evident in people with PD, 48 raw EMGs were visually inspected in real-time during data collection to ensure that activations plateaued and/or decreased prior to the participant relaxing. The peak RMS values recorded for each muscle during the MVIC trials were used to normalize the data, and the peak normalized amplitudes for each muscle were averaged within each group to facilitate further analyses. Trunk muscles were examined as they have been shown to have regular activation patterns during unconstrained walking 50 and contribute extensively to upper body orientation and segmental control during locomotion. 51
Twelve-Month Prospective Follow-up
Following the assessments, participants were asked to prospectively record any falls experienced over the next 12-month period on a series of falls diaries, which they completed daily and returned at the end of each month via reply-paid post. When participants recorded a fall, they were asked to report the timing, location, and circumstances surrounding that fall. A fall was defined as an unintentional coming to the ground or some lower level not as a result of a major intrinsic event or overwhelming hazard. 29 Using the prospective falls data, participants were divided into 4 groups that included people in the PD and control groups who reported no falls during the follow-up period (PD nonfallers; Control nonfallers) and those who reported one or more falls (PD fallers; Control fallers).
Statistical Analysis
A one-way analysis of variance (ANOVA) was used to assess differences in demographics, movement characteristics, and muscle activity patterns between the PD patients and controls or the different faller subgroups. When a significant main effect was identified for the faller subgroups, the Tukey’s honestly significant difference post hoc test was used to determine where the statistically significant differences existed. The Tukey’s honestly significant difference procedure controls for the overall significance level when performing all pairwise comparisons in the ANOVA and therefore reduces the likelihood of a Type 1 error. In circumstances where the assumptions of the ANOVA were violated, the nonparametric Kruskal-Wallis test was used. The degree of association between the categorical variables was assessed with the χ2 test.
The EMG outcomes were also analyzed using an analysis of covariance (ANCOVA) to determine whether differences in medications (antidepressants, benzodiazepines 52 ), symptom severity (UPDRS total score, Hoehn and Yahr stage score), and/or trunk posture potentially influenced the amplitude of trunk EMGs. Furthermore, linear regression was used to determine whether differences in trunk muscle function were related to differences in head, trunk, and pelvis control during walking. In addition to reporting the level of significance (P values) for the selected outcomes, the minimal detectable change (MDC) was calculated using the formula 1.96 × √2 × standard error of measurement and presented to provide insight into the clinical meaningfulness of the reported outcomes. 53 Statistical procedures were conducted using SPSS 22, with the level of significance set at P < .05.
Results
Falls
Forty-eight people with PD (61%) and 29 control participants (35%) reported at least one fall during the 12-month period, while 28 (35%) participants with PD and 13 (16%) controls reported falling twice or more. On the basis of the prospective falls data, participants were divided into 4 groups: PD Fallers (n = 48), PD Nonfallers (n = 31), Control Fallers (n = 29), and Control Nonfallers (n = 53).
Demographics and Clinical Assessments
The PD and control groups and the 4 faller subgroups did not differ significantly for age, gender, height, mass, or body mass index. However, PD participants had more previous falls and less balance confidence than controls, while PD fallers reported more previous falls and less balance confidence than PD nonfallers and the 2 control groups. PD nonfallers reported less balance confidence than the control fallers and nonfallers, while PD nonfallers and control fallers both recorded more previous falls than control nonfallers.
The PD-specific clinical assessments indicated that PD fallers had more advanced symptoms, took larger daily doses of levodopa, and had greater difficulties performing activities of daily living (UPDRS II) and simple motor tasks (UPDRS III). Despite this, subclassification of patients in the 2 PD groups highlighted that each group comprised a similar proportion of patients with tremor-dominant, PIGD-dominant, and mixed symptoms. With respect to the specific medications being used by the study cohort, the results indicated that a greater proportion of PD patients were taking antidepressants and benzodiazepines than the control participants. Furthermore, a significantly greater percentage of PD fallers were taking antidepressants and benzodiazepines compared with the control nonfaller group. With respect to the anti-parkinsonian medications being taken by the patient cohort, a similar proportion of PD fallers and nonfallers reported taking dopamine agonists, catechol-O-methyl transferase inhibitors, and/or monoamine oxidase inhibitors (Table 1). There were no significant differences between the 2 patient subgroups with respect to the proportion of each cohort that was taking 1, 2, 3, or 4 anti-parkinsonian medications at the time of their involvement in this study (χ2 = 3.49; P = .48).
Demographic Data and Disease-Specific Scores for the Participants With Parkinson’s Disease, the Age-Matched Controls Participants, and the Faller and Nonfaller Subgroups*.
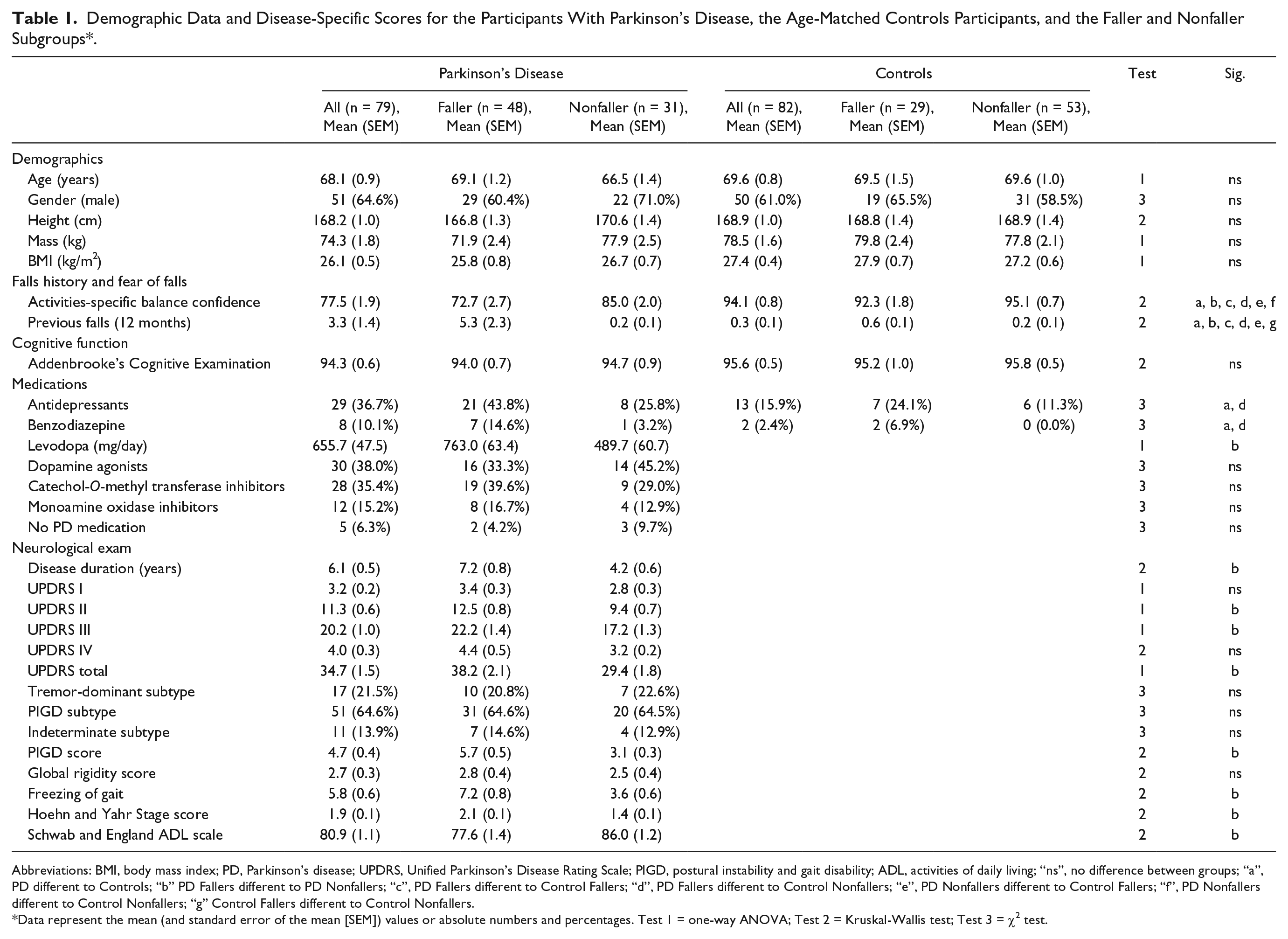
Abbreviations: BMI, body mass index; PD, Parkinson’s disease; UPDRS, Unified Parkinson’s Disease Rating Scale; PIGD, postural instability and gait disability; ADL, activities of daily living; “ns”, no difference between groups; “a”, PD different to Controls; “b” PD Fallers different to PD Nonfallers; “c”, PD Fallers different to Control Fallers; “d”, PD Fallers different to Control Nonfallers; “e”, PD Nonfallers different to Control Fallers; “f”, PD Nonfallers different to Control Nonfallers; “g” Control Fallers different to Control Nonfallers.
Data represent the mean (and standard error of the mean [SEM]) values or absolute numbers and percentages. Test 1 = one-way ANOVA; Test 2 = Kruskal-Wallis test; Test 3 = χ2 test.
Spatiotemporal Characteristics
Participants with PD took shorter strides (P = .001) and walked slower (P = .009) than the age-matched controls, but stride frequency (cadence) and step width did not differ between the 2 groups. Comparison of the 4 faller subgroups indicated that the PD fallers, PD nonfallers, and control fallers all took significantly shorter strides and walked slower than control nonfallers. However, the MDC values reported for these outcomes indicated that only the differences observed between the PD fallers and control nonfallers and the PD nonfallers and control nonfallers were clinically meaningful. The 4 faller groups did not differ for cadence or step width (Table 2).
Mean (and Standard Error of the Mean [SEM]) Segmental Movement Patterns, Sagittal Trunk Kinematics, and Trunk Muscle Activation for the People With Parkinson’s Disease, Age-Matched Controls, and the Faller and Nonfaller Subgroups*.
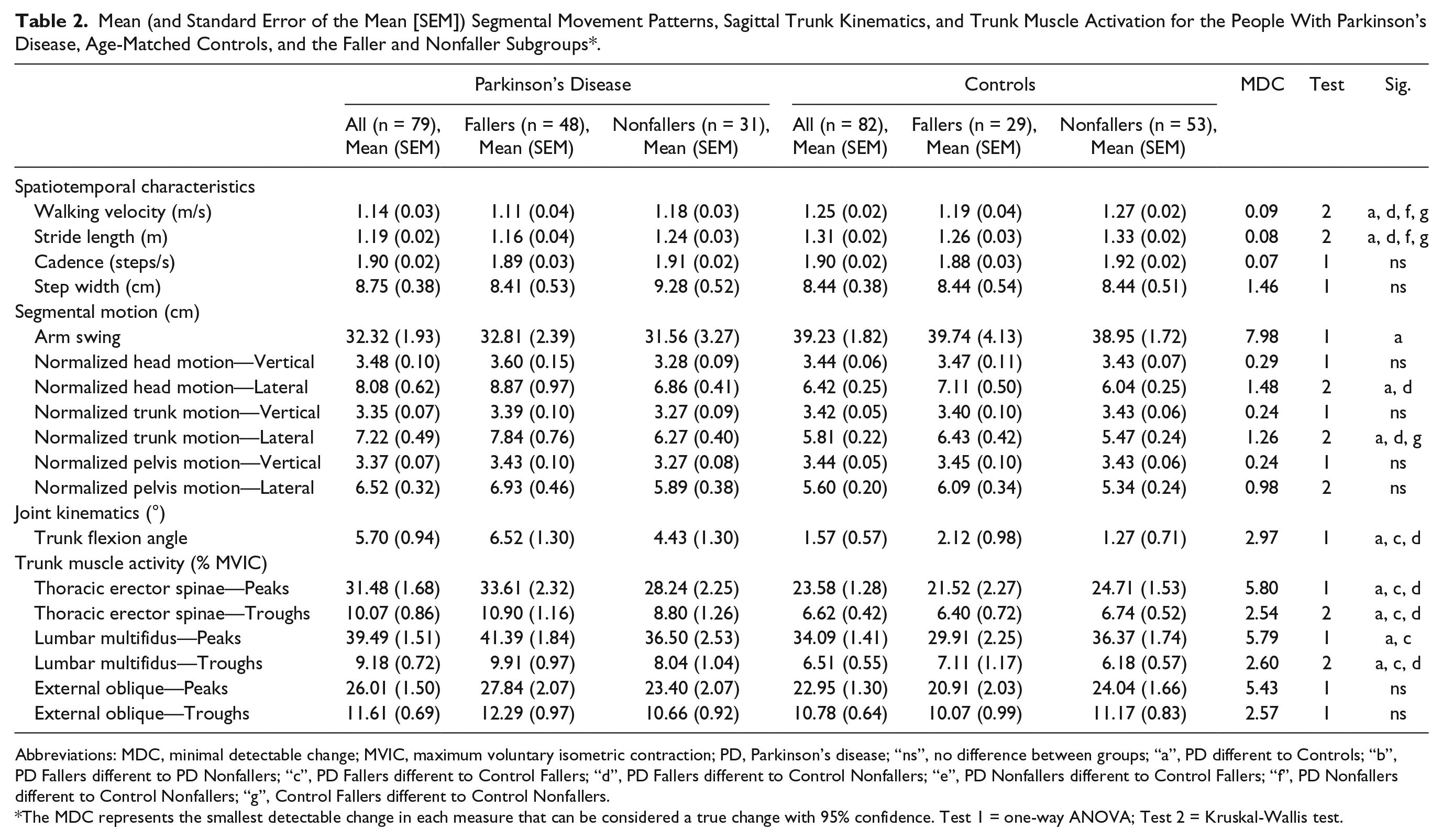
Abbreviations: MDC, minimal detectable change; MVIC, maximum voluntary isometric contraction; PD, Parkinson’s disease; “ns”, no difference between groups; “a”, PD different to Controls; “b”, PD Fallers different to PD Nonfallers; “c”, PD Fallers different to Control Fallers; “d”, PD Fallers different to Control Nonfallers; “e”, PD Nonfallers different to Control Fallers; “f”, PD Nonfallers different to Control Nonfallers; “g”, Control Fallers different to Control Nonfallers.
The MDC represents the smallest detectable change in each measure that can be considered a true change with 95% confidence. Test 1 = one-way ANOVA; Test 2 = Kruskal-Wallis test.
Segmental Motion and Joint Kinematics
Compared with controls, PD participants adopted a more stooped posture (greater trunk flexion) and had greater lateral head and trunk movement following adjustment for walking speed. Furthermore, the statistical analyses suggested that the patient group had less arm swing than the control group, but the MDC for this variable indicated that this difference was not clinically meaningful. The groups did not differ with respect to lateral pelvic movement or vertical head, trunk and pelvis displacement.
PD fallers had more trunk flexion than control fallers (P = .038) and nonfallers (P = .001) and recorded more lateral head (P = .009) and trunk (P = .008) motion than control nonfallers. While control fallers also had greater lateral trunk motion than their nonfalling counterparts (P = .025), the MDC value suggested that this difference was not clinically important. Participants in the PD nonfaller group did not differ significantly from the other faller subgroups for any of the measures of segmental motion or joint kinematics (Table 2).
Trunk Muscle Activation
The normalized trunk EMGs indicated that while the participants with PD demonstrated similar trunk muscle activation, they achieved significantly greater peak values for both the TES and LMF. Furthermore, the PD group had significantly greater TES and LMF activation between each of the dominant bursts of these muscles (ie, greater baseline activity). Given that benzodiazepine and antidepressant use was more common in PD fallers and that these patients had more advanced motor symptoms than their nonfaller counterparts, the TES, LMF, and EO data were further analyzed using an ANCOVA to determine whether these factors may have influenced the outcomes. The results of these analyses indicated that the reported activations of the anterior and posterior trunk muscles were not significantly influenced by benzodiazepine use, antidepressant use, UPDRS total score, or Hoehn and Yahr stage score. Furthermore, given PD patients walked with a more stooped posture than controls, the ANCOVA procedure was also used to account for any potential influence of trunk orientation on the TES and LMF data. Even following adjustment for differences in trunk posture, the PD group had significantly greater peak (F[1, 158] = 9.125, P = .003) and baseline (F[1, 158] = 6.295, P = .013) TES activation. In contrast, bilateral activation of the EO did not differ between the PD and control groups.
Figure 2 provides representative bilateral TES activation patterns for the PD fallers, PD nonfallers, control fallers, and control nonfallers. The subgroup analyses indicated that the PD fallers typically demonstrated greater peak TES (Figure 3) and LMF muscle activity than both control groups, although the difference between PD fallers and control nonfallers for peak LMF activity did not reach statistical significance (Table 2). Similarly, compared with both control groups, PD fallers had greater baseline activity between successive TES and LMF bursts, while PD nonfallers did not differ from the other groups for TES or LMF activation patterns. The ANCOVA results indicated that the higher peak (F[3, 156] = 4.445, P = .005) and baseline (F[3, 156] = 2.766, P = .044) TES activities for the PD fallers were completely independent of the trunk flexion evident in these patients. The faller groups did not differ for peak or baseline levels of EO activity.
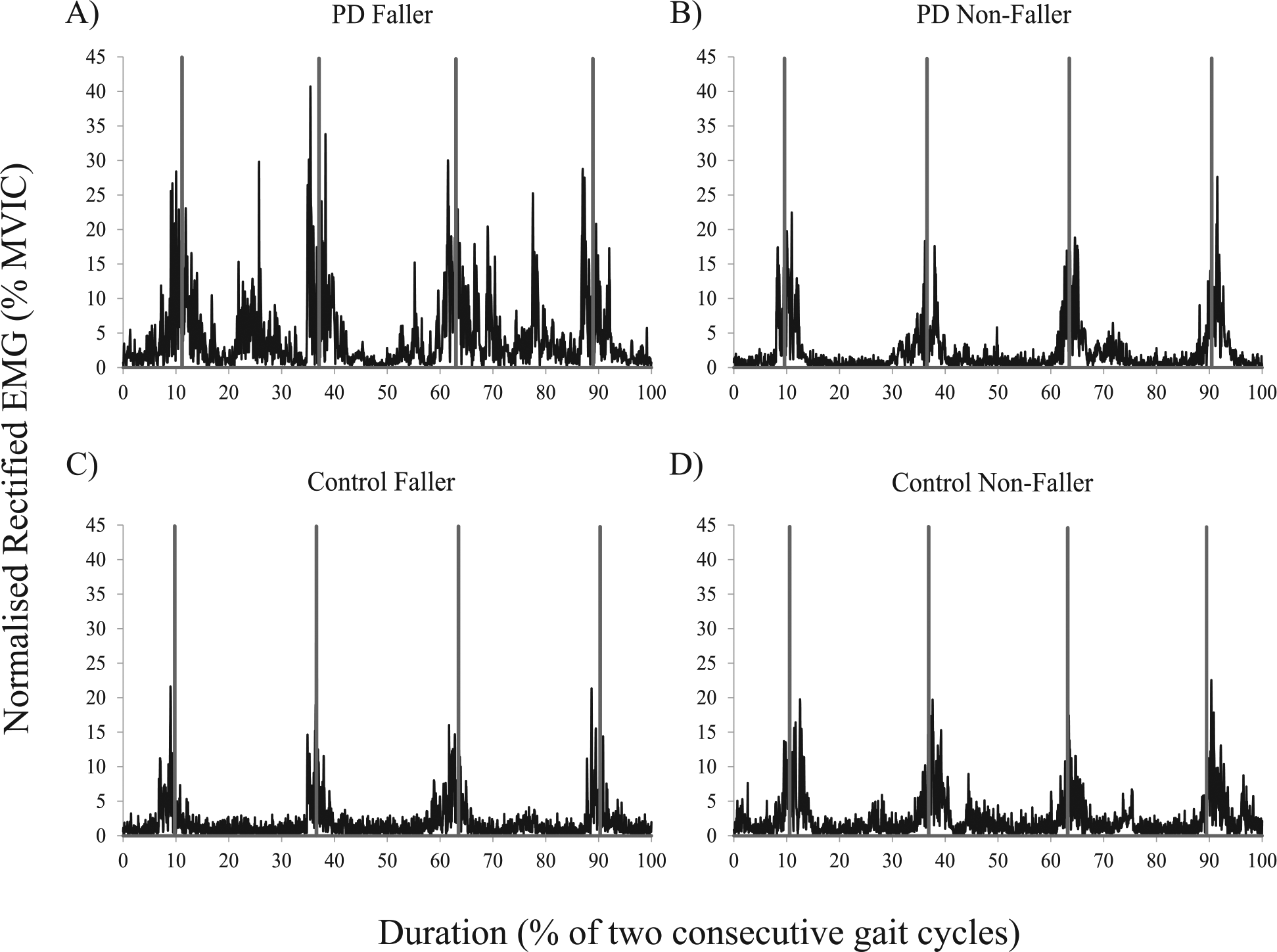
Representative bilateral thoracic erector spinae muscle activity for one participant from the (A) Parkinson’s disease Faller; (B) Parkinson’s disease Nonfaller; (C) Control Faller; and (D) Control Nonfaller groups.
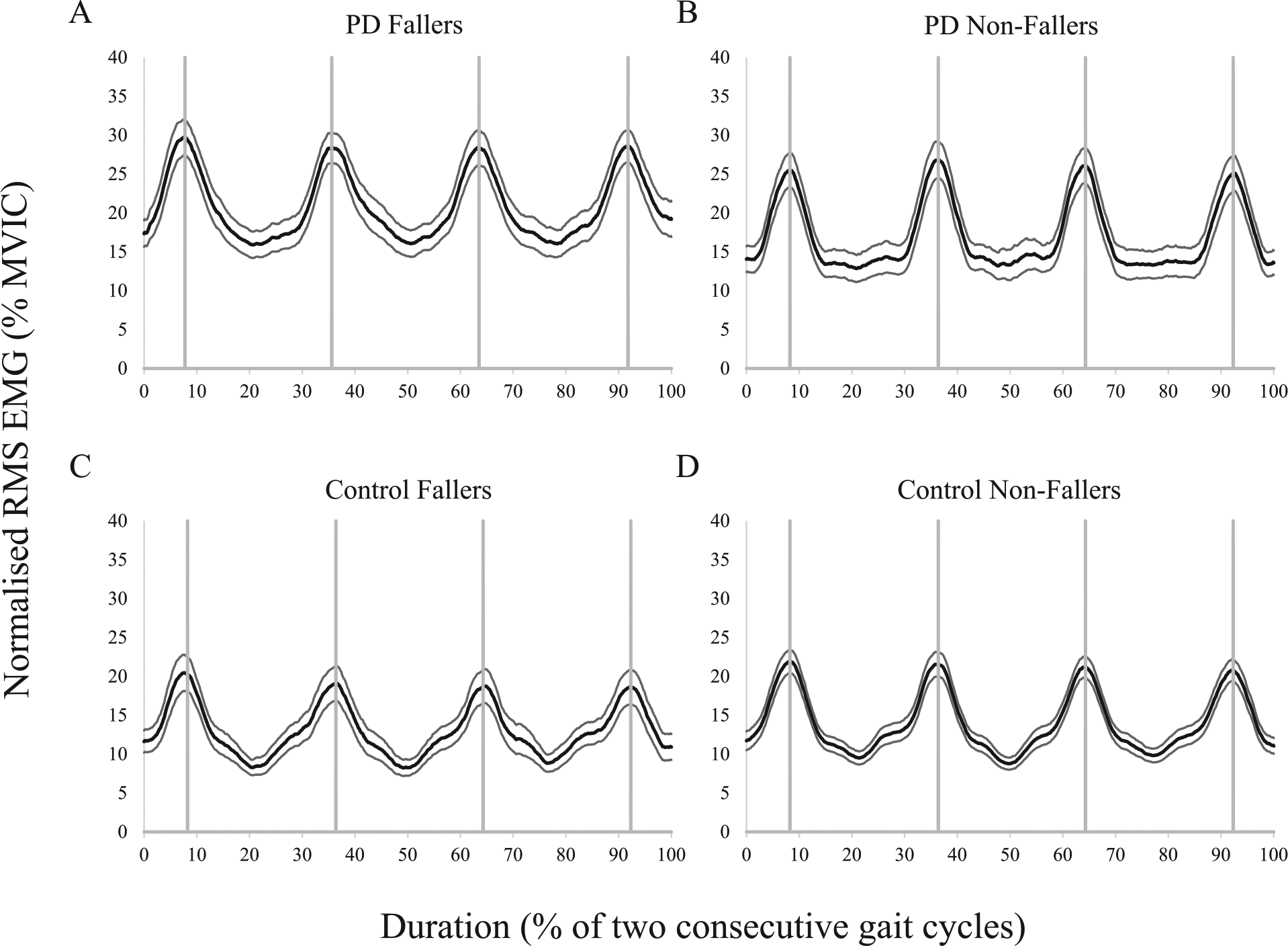
Mean (±1 SEM) bilateral thoracic erector spinae muscle activity for the (A) Parkinson’s disease Fallers; (B) Parkinson’s disease Nonfallers; (C) Control Fallers; and (D) Control Nonfallers.
Trunk Muscle Activation and Segmental Motion
Simple linear regressions evaluated the relationship between the trunk muscle activity (peaks and baseline) and the lateral displacement of the head, trunk, and pelvis. For the PD fallers only, significant regression equations indicated that increased baseline TES activity was predictive of greater normalized lateral head (F[1, 47] = 13.985, P = .001, B = 4.180), trunk (F[1, 47] = 17.105, P < .001, B = 3.523), and pelvis (F[1, 47] = 15.564, P < .001, B = 2.011) movements. Similarly, significant regression equations showed that greater baseline LMF activity was predictive of larger normalized lateral head (F[1, 47] = 8.218, P = .006, B = 4.027), trunk (F[1, 47] = 9.213, P = .004, B = 3.307) and pelvis (F[1, 47] = 12.936, P = .001, B = 2.257] movements for PD fallers. The regression equations returned for the other 3 subgroups identified no significant association between trunk muscle activation and segmental motion.
Discussion
This study investigated deficits in the activation of the anterior and posterior trunk muscles in people with PD who prospectively reported falling and their relationship to difficulties with coordinating and stabilizing the head and trunk during locomotion. The results highlighted significant differences in trunk muscle function during walking for people with PD who prospectively fall. Commensurate with previous research,7,24 PD fallers were also less able to control gait-related head and trunk movements when compared with the participants in the age-matched control groups. Interestingly, the apparent difficulties that these patients had with regulating segmental control were directly influenced by their altered trunk muscle activation patterns, which suggested that such deficits in neuromuscular function may impair dynamic postural control in these individuals.
In healthy adults, the activation patterns of the erector spinae and multifidus are characterized by 2 main bursts of activity during the gait cycle; each separated by a period of rest or relatively little activation. 50 Each of these bursts coincides with (or occurs shortly after) initial contact between the foot and the walking surface.22,50,54 This bilateral activation of the erector spinae and multifidus muscles is believed to assist with keeping the trunk upright and over the pelvis by counteracting the trunk flexor moment that occurs during this phase.54,55 The TES and LMF data collected in this study indicated that this basic pattern of activation was largely unaffected by the ageing process or the presence of PD. However, participants with PD and, in particular, those patients who prospectively reported falling typically had greater peak TES and LMF activations than both control groups. Furthermore, these participants also presented with higher levels of baseline activity for these muscles, which was suggestive of a greater level of coactivity during walking.
Arguably, the higher levels of TES and LMF activity demonstrated by the PD fallers may be related to their more stooped walking posture and an increased reliance on these muscles to suspend the trunk’s mass against gravity. Interestingly, our results suggest that while the larger LMF peak and baseline activities recorded for the PD fallers were largely attributable to differences in trunk orientation, the observed TES activations were independent of trunk posture. Furthermore, differences in symptom severity and benzodiazepine or antidepressant use were also shown to have little influence on TES muscle activation in this cohort. These findings are somewhat in contrast with previous research that has highlighted that the number of falls experienced by a person with PD in the previous month was significantly positively associated with antidepressant use. 52 Nevertheless, our results seem to suggest that the higher TES activity observed in PD fallers was unlikely to represent a compensatory strategy to account for increased symptom severity or a neuromuscular adaptation in response to altered trunk posture, but rather represented a genuine abnormality in neuromuscular control.
While the greater trunk muscle activity evident for the PD fallers could be viewed as a form of active stabilization adopted by these patients, this interpretation incorrectly assumes that a stiffer trunk is representative of a more stable trunk. During activities of daily life, the human body is rarely truly static and, hence, requires the head and trunk to move in an independent and coordinated manner to maintain equilibrium.17,20 The linear regression analyses indicated that, for the PD faller group only, TES and LMF baseline activities were predictive of lateral head, trunk, and pelvis movement during the walking task. Moreover, for every 1% increase in normalized baseline activity of the TES and LMF, lateral head movements were predicted to increase by approximately 4 cm, while predicted increases in lateral trunk and pelvic motion were 3.5 and 2 cm, respectively. These findings suggested that the increased levels of baseline activity evident for the TES and LMF muscles served to increase the overall stiffness (rigidity) of the kinetic chain, resulting in the pelvis, trunk, and head tending to move more as a single rigid unit rather than a collection of independent and well-coordinated segments. Previous research provides support for this notion, showing that increased axial stiffness causes people with PD to rotate the thorax and pelvis in unison (ie, in-phase),56,57 which significantly reduces their stability and increases their risk of overbalancing. 58 Furthermore, given differences in trunk muscle activation patterns and lateral head and trunk movements were most prominent in those PD patients who prospective reported falling, our findings seem to strengthen the argument that such characteristics may be linked to the higher rate of falls experienced by this population.7,24 Nevertheless, it should be noted that although control fallers had significantly greater lateral trunk motion than control nonfallers, neither peak nor baseline TES or LMF activation were significant related to this observation. These results seem to suggest that the mechanism(s) of falling in otherwise healthy older adults may be quite different to those experienced by people with PD. This notion may be supported by epidemiological research, which demonstrates that 69% of the falls from standing that are reported by PD patients are attributed to a loss of balance, 13 while only 9% of the falls experienced by community-dwelling older adults are attributed to this cause. 59 Given the apparent uniqueness of the physiological deficits exhibited by people with PD, it seems reasonable to suggest that falls prevention strategies be developed to specifically assist these individuals, rather than simply adopting programs developed for other populations.
The identified relationship between increased axial rigidity and falls is well established.23,60 Indeed, for over 2 decades, clinicians and scientists have reported the existence of at least 2 major subtypes of PD that are discernible based on their dominant motor symptom(s).38,61-63 Despite the availability of several protocols for determining a patient’s disease subtype, all tend to identify a group of patients whose primary motor symptom is resting tremor (tremor-dominant type), a group whose primary symptoms are bradykinesia and rigidity (akinetic-rigid or PIGD type) and a group who presents with a combination of these 2 types (mixed or indeterminate type). Given these classifications, one may intuitively assume that patients who present primarily with symptoms of bradykinesia and rigidity (akinetic-rigid type) or postural instability and gait disability (PIGD type) would be more likely to present with impaired axial muscle function. However, given there was a similar proportion of tremor-dominant and PIGD-dominant patients in the PD faller and PD nonfallers groups, it seems that impaired trunk muscle function is not unique to patients who present with bradykinetic and/or rigid symptoms. Furthermore, linear regression revealed no significant relationship between the global rigidity score derived from the UPDRS and normalized ML head (P = .701), trunk (P = .697), or pelvis (P = .726) movement in the PD population. These findings suggest that the assessment of trunk muscle function during gait may offer far greater benefits to existing clinic tests with respect to identifying patients who experience stability issues during dynamic tasks. As such, there appears to be a need to improve existing clinical assessments to ensure that “at risk” patients can be effectively identified within the constraints of a standard clinical consultation.
The relatively low rate of acceptance to participate in this study (25% for PD patients) may raise questions about the potential transferability of our findings to the wider PD community. With the information available for the nonresponders, it was not possible to compare the disease characteristics, medication types, and/or symptom severity of those included in the study with those who declined or did not respond. However, statistical comparison of the groups for age (P = .765), gender distribution (P = .586), and proximity to the testing location (P = .512) showed no significant differences between those who were included and those who were not. While these results suggesting that the sample was somewhat representative of the wider PD community in this geographical region, the applicability of the reported outcomes to other PD populations should be considered. Furthermore, while our results provide evidence of impaired neuromuscular control of the superficial paraspinal muscles in PD fallers, it should be noted that it very difficult to accurately assess the activation of the deeper trunk muscles (eg, thoracic multifidus), which are typically considered the primary stabilizers of the spine. 22 As such, it is plausible to suggest that the higher TES activity demonstrated by the PD fallers was representative of muscle substitution, rather than a specific deficit in this muscle’s function. The potential limitations associated with surface electromyography should therefore be considered when reviewing our findings. Finally, while the incidence of falls was established prospectively using daily falls diaries, participants were not required to return these until the end of each month. As such, it may be reasonable to argue that some participants may have completed multiple daily entries on a single day (ie, retrospectively), which would have inadvertently increased the risk of recall bias. Nevertheless, it should be noted that the methods used were commensurate with previous research reporting the incidence of prospective falls64-68; hence, the potential risk of bias would not be expected to be any greater than observed in these earlier studies.
Collectively, the results of this study assist with better understanding the mechanisms of postural instability in people with PD and suggest that those patients who fall demonstrate greater trunk muscle activity that is independent of medication use, symptom severity, and trunk flexion and contributes to the impaired head and trunk control evident in this subpopulation. Given that head and trunk control are critical to maintaining dynamic stability during locomotion, it is essential that the underlying mechanism(s) of axial PD symptoms are identified. Furthermore, as current pharmacological and surgical interventions are not always effective for managing these dopamine-resistant axial symptoms of PD, 23 future research should seek to identify alternate approaches to assist with the improvement of these symptoms. Exercise-based interventions seeking to improve the coordination, endurance, and mobility of the trunk 69 may prove beneficial for people with PD, as systematic evidence indicates that exercise is effective for improving some motor symptoms and patient scores on a number of balance and mobility scales.70-74
Footnotes
Acknowledgements
The authors would like to thank those people with Parkinson’s disease and the age-matched controls who gave so generously of their time to help make this research possible.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by an Australian National Health and Medical Research Council (NHMRC) Early Career Researcher Fellowship (Project #GNT1016481) and research funding provided by the Australian Catholic University (Project #2013000570). The funding bodies did not contribute to data collection or data analysis and played no part in the decision to prepare and publish this manuscript.
