Abstract
Introduction
Falls are a significant problem for people with Parkinson’s disease (PD), with fall rates double 1 that of community-dwelling older people without neurological disease.2,3 Many falls experienced by people with PD result in injury4,5 and require costly health care. It is well known that previous falls1,6-8 are a strong predictor of future falls in people with PD. Several studies have identified a range of factors that are associated with an increased risk of falls in people with PD, such as impaired balance,5,6,9 impaired mobility,5,9 freezing of gait,5,9 decreased muscle strength,5,9 impaired cognition,4-6 and disease severity,4-7,9 but the relative contribution of these risk factors to falls is unclear. Other risk factors such as multiple drug use, depression, orthostatic hypotension, and limitations with daily activities have been proposed but do not show a consistent association with increased fall risk in people with PD.1,4,5,9-11
Understanding which risk factors are important in determining overall fall risk has the benefit of allowing clinicians to target and prioritize interventions. An understanding as to whether the underlying cause of the impairment is remediable is also important as again it will determine whether an intervention is put in place to address the risk factor (eg, cataract removal to correct the associated visual impairment) or minimize the impact of the identified deficit (eg, provision of mobility aids for people with severe balance problems). This is an important contrast to other variables such as “past falls” that have been used in previous prediction models, that is, although predictive, such marker variables provide little insight into why future falls occur and do not guide intervention strategies.
To gain a greater understanding of the causes of falls, some studies in the general older population have investigated possible physical and cognitive risk factors that may explain mechanisms underlying falls.12,13 To date, only 2 prospective studies of falls undertaken in people with PD have included both physical and cognitive factors.5,14 One study found carer-rated cognitive impairment and freezing of gait to be important explanatory fall risk factors but this study contained only a small sample size (n = 52). 14 The other study 5 included a larger sample (113 participants) and identified freezing of gait, poor balance, reduced muscle strength, impaired executive functioning, and abnormal posture as independent risk factors for falls in a multivariate explanatory model.
As some of the impairments identified above are amenable to intervention, these studies provide valuable information for informing fall prevention initiatives in people with PD. Given that only 2 studies have investigated potentially remediable physical and cognitive fall risk factors (without including prior falls in the models),5,14 further work is required to confirm such findings in large external samples and identify possible additional important explanatory fall risk factors. Therefore, this study aimed to identify the relative contribution of a comprehensive range of physical and cognitive factors to future falls in a sample of over 200 people with PD. We hypothesized that both physical and cognitive factors would make independent contributions to overall fall risk in people with PD and that these important fall risk factors would be potentially amenable to intervention.
Methods
Participants
A total of 205 participants, recruited from Parkinson’s support groups and private neurology clinics, participated in this study: 133 were drawn from the control groups of 2 randomized controlled trials assessing exercise interventions15,16 and the remainder comprised participants who did not meet the inclusion criteria for the randomized controlled trials or chose not to participate in them. The eligibility criteria for this study were as follows: having idiopathic PD as confirmed by a medical practitioner, older than 40 years, and able to walk independently with or without an aid. Participants were excluded if they had significant cognitive impairment (Mini Mental State Examination [MMSE] 17 score <24) or suffered from any unstable cardiovascular, orthopedic, or neurological conditions that would interfere with the safety of assessment and/or interpretation of results.
Each participant was assessed at home during a single assessment session of approximately 2.5 hours duration by trained research staff. The order of testing was standardized for all participants and the assessment was conducted when each person’s PD medications were working optimally, usually 1 hour after taking the medication. For participants who reported going “off” during the assessment, testing was suspended until they had taken their medication and reported being “on” again. Similarly, for participants who fatigued during the assessment, rest breaks were given and testing recommenced only when the participant reported being able to continue.
Following baseline assessment, the number of falls sustained by each person was monitored prospectively over 6 months by the use of monthly falls diaries with routine monthly telephone follow-up. 16 Participants were subsequently categorized into fallers (≥1 fall during follow-up) and nonfallers according to the following definition of a fall: unintentionally coming to rest on the ground or other lower surface without overwhelming external force or a major internal event. 18 This study was approved by the relevant human ethics committee and all participants gave written informed consent prior to assessment.
Measurement of Potential Risk Factors
Background information including age, falls history in the previous 12 months, physical activity levels, and time since PD diagnosis were recorded. PD severity was ascertained from the motor section of the Unified Parkinson’s Disease Rating Scale (UPDRS). 19
Tasks Requiring Anticipatory Control Without a Change in Base of Support
Functional Reach 25 was performed 3 times and measured to the nearest 0.5 cm. Near tandem stand with eyes shut 26 was performed twice and timed with a handheld stopwatch for up to 10 seconds each. Postural sway path length was measured in mm using a portable swaymeter while participants stood barefoot on the floor and on a 7 mm medium-density foam rubber mat with eyes open for 30 seconds. 27 Maximum balance range in the anterior–posterior direction 28 required each participant to lean forward and backward from the ankles as far as possible 3 times without losing balance and was measured in mm. The coordinated stability task measured participants’ ability to adjust balance in a steady and coordinated way while placing them near or at the limits of their base of support. The swaymeter was attached to the participant’s waist with the rod extending anteriorly. Participants were asked to adjust balance in a steady manner so that the pen on the end of the rod followed and remained within a convoluted track that was marked on a piece of paper attached to the top of a height-adjustable table. To complete the test without errors, participants had to remain within the 14 mm wide track and be capable of adjusting the position of the pen 29 cm laterally and 18 cm in the anterior–posterior plane. A total error score was calculated by summing the number of occasions the pen on the swaymeter failed to stay within the path. Where participants failed to negotiate an outside comer (because they could not lean sufficiently), 5 error points were accrued. Participants attempted the test twice, with the better trial taken as the test score. 28 Standing balance with a narrow base of support was calculated as the sum of standing without arm support and with eyes open for up to 10 seconds in 3 positions: standing with feet together, in semi-tandem stance, and in tandem stance. 29
Tasks Requiring Anticipatory Control With a Change in Base of Support
The alternate step test 26 required each participant to alternately place each foot up onto an 18 cm block for 8 repetitions as quickly as possible; this test was performed twice and timed to the nearest millisecond. The 5-repetition sit-to-stand 26 was performed once as fast as possible with arms folded across the chest and timed to the nearest millisecond. Comfortable and fast walking pace (timed with a stopwatch) was measured over 4 m with an extra meter at either end; participants completed 2 trials at each pace. Walking pace was calculated in meters per second (m/s).
Tasks Requiring Reactive Adjustments to External Perturbations
The pull test was rated using item 30 of the UPDRS and scored from 0 to 4. This score was dichotomized at the most discriminating cut-point. Scores of 0 to 1 indicated less impairment in reactive adjustments, while scores of 2 to 4 indicated more impaired reactive adjustments.
The majority of these balance and mobility tests have been shown to be valid30,31 and to have fair to excellent reliability 32 in people with PD.
Statistical Analyses
Data were analyzed using Stata version 12 (Stata Corp, College Station, TX). Some participants were physically unable to perform one or more of the mobility measures. These participants were assigned a score of 0 (for measures where high scores indicate better performance) or a value of the sample mean plus 3 standard deviations (for measures where low scores indicate better performance; <6% for any one measure). Other missing data for continuous variables were imputed using the “fill in missing values” command in Stata using data from similar tests (<1% of data; <6% of data for any variable).
Associations between each performance measure and falling during the follow-up period were assessed using univariate modified Poisson regression with robust error variance. 36 Standardized risk ratios (RRs) were also calculated to allow comparison of the relative association of each risk factor with future falls.
To identify significant and independent fall risk factors across multiple domains, the variable with the strongest association with falls from each of the domains or their subsets (ie, PD signs and symptoms, anticipatory balance without a change in base of support, anticipatory balance with a change in base of support, reactive balance, proprioception, muscle strength, orientation, and executive function) was entered into the multivariate model. This method decreased the risk of spurious findings from entering too many predictor variables in multivariate models in relation to the sample size. Backward stepwise regression, with
Results
Characteristics of the 205 participants are described in Table 1. Falls data for the complete 6-month follow-up period were available for all participants; 120 participants (59%) fell at least once during follow-up. The univariate associations and standardized RRs between individual performance measures and falls during the follow-up period are shown in Table 2.
Participant Characteristics for the Entire Sample, the Nonfallers and the Fallers. a
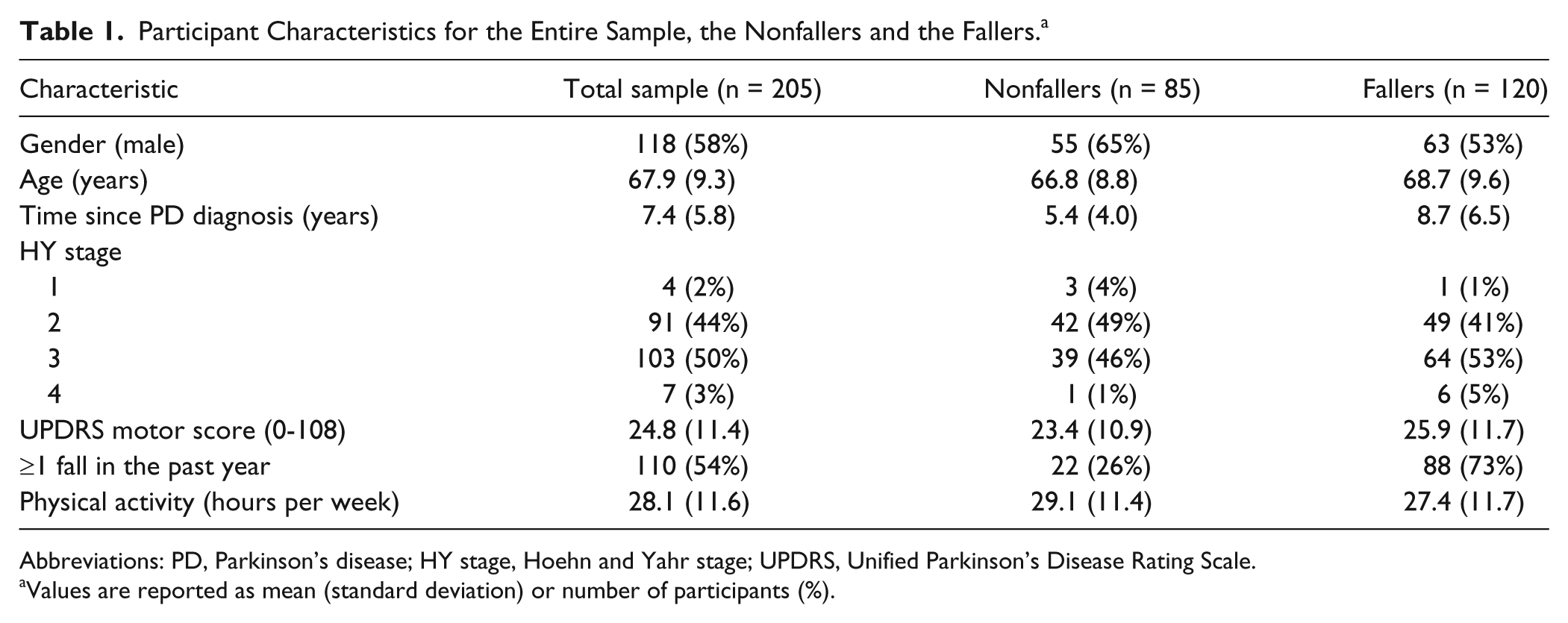
Abbreviations: PD, Parkinson’s disease; HY stage, Hoehn and Yahr stage; UPDRS, Unified Parkinson’s Disease Rating Scale.
Values are reported as mean (standard deviation) or number of participants (%).
Univariate Associations Between Risk Factors and Future Falls.
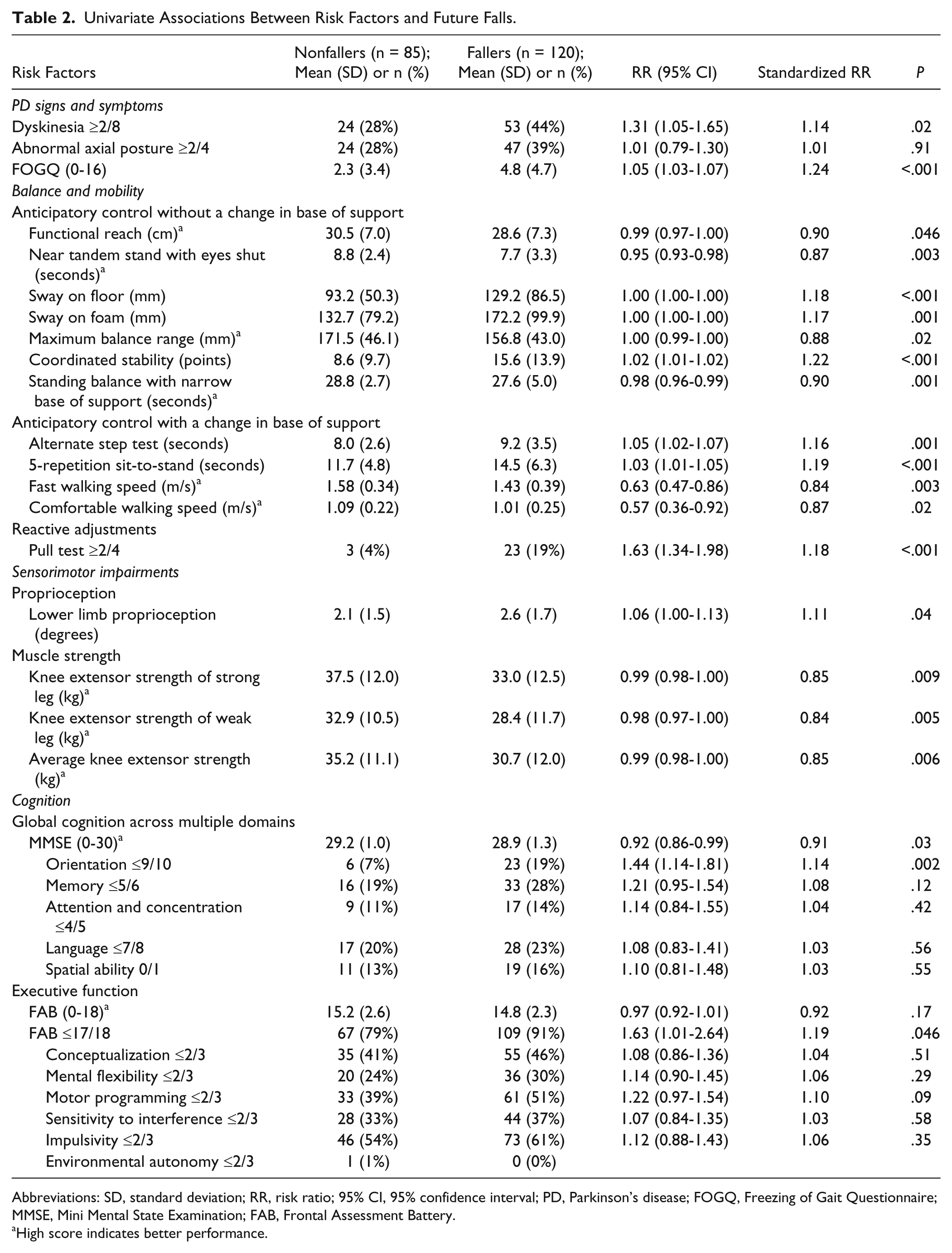
Abbreviations: SD, standard deviation; RR, risk ratio; 95% CI, 95% confidence interval; PD, Parkinson’s disease; FOGQ, Freezing of Gait Questionnaire; MMSE, Mini Mental State Examination; FAB, Frontal Assessment Battery.
High score indicates better performance.
Certain measures within each physical and cognitive domain were significantly associated with future falls in univariate analysis. Of the PD signs and symptoms, freezing of gait (standardized RR = 1.24) and dyskinesia (standardized RR = 1.14) were significantly associated with falling. In the balance domain, all measures of anticipatory and reactive balance were associated with falls, although coordinated stability (standardized RR = 1.22), repeated sit-to-stand (standardized RR = 1.19), fast walking speed (standardized RR = 0.84), postural sway on a firm surface, and the pull test (standardized RR = 1.18) showed the strongest associations. Lower limb proprioception (standardized RR = 1.11) and knee extensor strength (standardized RR = 0.84-0.85) were sensorimotor factors significantly associated with falling. The cognitive measures which showed significant associations with falls were the total MMSE score (standardized RR = 0.91), impaired orientation on the MMSE (standardized RR = 1.14), and Frontal Assessment Battery scores ≤17 (standardized RR = 1.19).
The multivariate model revealed freezing severity, impaired balance (impaired coordinated stability and poor reactive adjustments), and impaired cognition to be the most important factors contributing to future falls (Table 3). Lower limb proprioception, knee extensor strength, and repeated sit-to-stand were not associated with falls once the other factors were adjusted for. The model was moderately accurate in predicting future falls (AUC = 0.73, 95% confidence interval = 0.66-0.80 with variables retained following backward stepwise regression). Variance inflation factors ranged from 1.1 to 1.6 for factors entered into the multivariate model, indicating a low risk of collinearity between risk factors.
Multivariate Model (AUC = 0.73) Showing the Relative Association of Physical and Cognitive Factors With Future Falls.
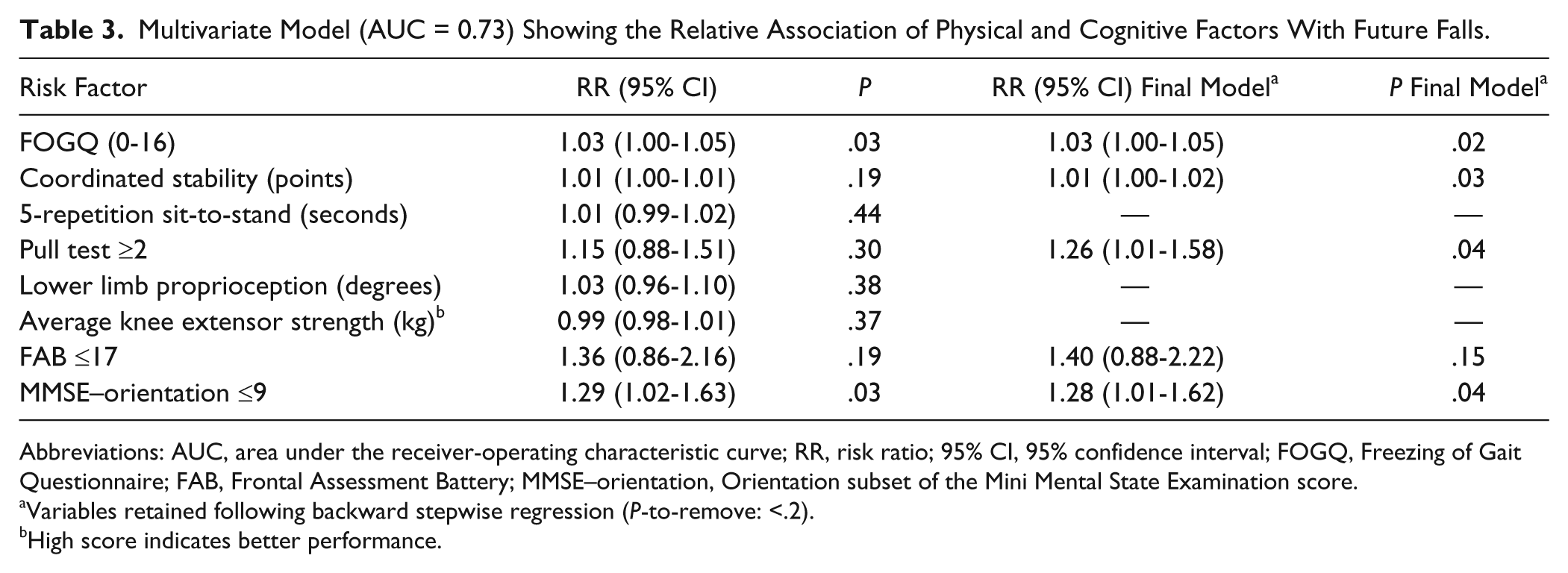
Abbreviations: AUC, area under the receiver-operating characteristic curve; RR, risk ratio; 95% CI, 95% confidence interval; FOGQ, Freezing of Gait Questionnaire; FAB, Frontal Assessment Battery; MMSE–orientation, Orientation subset of the Mini Mental State Examination score.
Variables retained following backward stepwise regression (
High score indicates better performance.
Discussion
This large prospective study aimed to identify cognitive, sensorimotor, balance, and gait factors associated with falls in community-dwelling people with PD. A range of measures were significantly associated with falls, with more severe freezing of gait, impaired coordinated stability, impaired reactive balance, and impaired orientation being significantly associated with falls in a final multivariate model that provided good discrimination between fallers and nonfallers (AUC = 0.73).
With regard to balance and mobility task performance, tasks requiring both anticipatory and reactive postural adjustments were found to be strongly associated with falls. These findings are consistent with observations that most falls reported by people with PD occur due to loss of balance11,37 and with prior findings showing that falls are strongly associated with poor coordinated stability, 5 a test involving control of body position while moving near the limits of stability without stepping. Additionally, our finding that impaired reactive balance was associated with falls is consistent with previously reported trends in studies with smaller sample sizes1,11 and clinical observation.
The finding that participant-reported freezing of gait severity was a significant and independent risk factor for falls is in line with previous studies9,10 and consistent with prior work reporting that many people with PD fall due to freezing.11,37 Mild impairments of executive function and orientation were also associated with falls and these impairments were retained in the multivariate explanatory model, but only impaired orientation was found to make a significant independent contribution (ie,
Finally, although impaired sit-to-stand, reduced leg muscle strength, and impaired proprioception did not make significant contributions to falls in multivariate analysis (probably due in part to their associations with the other included balance measures), the significant univariate associations suggest that these deficits40-43 and dyskinesia1,9 cannot be discounted as contributors to falls in people with PD.
As expected, the accuracy of the explanatory multivariate model developed in this study was less than predictive models that include a history of falling.7-9 While predictive models including past falls are useful in identifying an individual’s absolute risk of falling, 8 they do not necessarily direct assessment and intervention to reduce falls. A positive falls history probably represents a composite measure of individual risk factors from multiple domains that contribute to future falls. Therefore, the inclusion of this composite measure in predictive models likely masks the contribution of potentially remediable risk factors. The strength of multivariate explanatory models that do not include falls history, such as that developed in this study, is their ability to focus on potentially remediable fall risk factors. We have shown that when falls history, freezing of gait, and reduced gait speed are accounted for, 8 impaired balance and cognition do not remain strongly predictive of falls. However, the multivariate explanatory model developed in this study demonstrates that, in addition to freezing of gait, impaired balance (both anticipatory and reactive adjustments) and cognition are important risk factors to consider when determining potential intervention strategies.
A potential limitation of this study was that it was conducted in conjunction with 2 randomized controlled trials investigating the efficacy of exercise on risk factors for falls 15 and fall rates, 16 so it may be that the risk factor profile or the proportion of fallers in our study was not truly representative of community-dwelling people with PD. Nevertheless, the proportion of fallers identified in this study (59%) is in the same range (45% to 68%) as reported in other moderately large prospective studies (n = 101-164) investigating falls in community-dwelling people with PD.1,4,5,9,10,44 However, there were insufficient numbers of fallers in this study to subdivide them according to fall frequency. Further research is required to explore the risk profile of fallers based on their frequency of falls. Although there are limitations in self-reporting falls, this method remains best practice 45 and all participants in this study were required to report their falls according to the standardized definition. 45 The results of this study can be best generalized to people with moderate PD without significant cognitive impairment, defined as MMSE scores ≥24.
This study has a number of implications for clinical practice. The finding that impaired balance involving both anticipatory and reactive adjustments was strongly associated with falls suggests that interventions targeting balance may have a beneficial impact on falls prevention. There is substantial high-quality evidence demonstrating that intervention targeting anticipatory balance control is effective in improving balance in people with PD.46-49 A recent study has also found that falls were reduced in people with PD following a program of Tai Chi, 49 which involves stepping and controlled movements near the limits of stability. Given the strong association between reactive adjustments and falls, interventions involving stepping in response to external perturbations may also improve balance and potentially reduce falls.50-52 In addition, there is evidence showing that severity of freezing can be reduced with strategies such as auditory and visual cues53-55 and medications.56,57
The strong association between impaired orientation and future falls, even when physical deficits were considered, highlights the need for clinicians to be aware of cognitive deficits in their clients and to modify their approach to intervention based on the individual’s cognitive abilities. There are also encouraging findings that multiple domains of cognition may be improved following cognitive training in individuals without significant cognitive impairment. 58
Multifaceted fall prevention strategies have been found to be effective in the general older community
59
but are yet to be evaluated in people with PD. It is recommended that future studies investigating intervention strategies targeting falls consider (
Footnotes
Acknowledgements
The authors wish to thank Natalie Allen, Susan Murray, Sandra O’Rourke, Wendy Robinson, Lauren Wade, and Geraldine Wallbank for their assistance with recruitment and data collection. We would also like to thank Parkinson’s NSW for their assistance with recruitment and the people with Parkinson’s disease who participated in this research.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VSCF is on advisory boards and/or has received travel grants from Abbott, Allergan, Boehringer-Ingelheim, Hospira, Lundbeck, and Novartis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a National Health and Medical Research Council of Australia (NHMRC) Project Grant (ID: 512326), a grant from Perpetual Philanthropic Services and a Parkinson’s NSW research grant. SSP received financial assistance from a NHMRC postgraduate scholarship. CS and SRL receive salary funding from the NHMRC.
