Abstract
Background. Pharyngeal electrical stimulation (PES) appears to promote cortical plasticity and swallowing recovery poststroke. Objective. We aimed to assess clinical effectiveness with longer follow-up. Methods. Dysphagic patients (n = 36; median = 71 years; 61% male) recruited from 3 trial centers within 6 weeks of stroke, received active or sham PES in a single-blinded randomized design via an intraluminal pharyngeal catheter (10 minutes, for 3days). The primary outcome measure was the Dysphagia Severity Rating (DSR) scale (<4, no-mild; ≥4, moderate-severe). Secondary outcomes included unsafe swallows on the Penetration-Aspiration Scale (PAS ≥ 3), times to hospital discharge, and nasogastric tube (NGT) removal. Data were analyzed using logistic regression. Odds/hazard ratios (ORs/HRs) >1 for DSR <4, hospital discharge, and NGT removal and OR <1 for PAS ≥3, indicated favorable outcomes for active PES. Results. Two weeks post–active PES, 11/18 (61%) had DSR <4: OR (95% CI) = 2.5 (0.52, 14). Effects of active versus sham for secondary outcomes included the following: PAS ≥3 at 2 weeks, OR (95% CI) = 0.61 (0.27, 1.4); times to hospital discharge, 39 days versus 52 days, HR (95% CI) = 1.2 (0.55, 2.5); NGT removal 8 versus 14 days, HR (95% CI) = 2.0 (0.51, 7.9); and DSR <4 at 3 months, OR (95% CI) = 0.97 (0.13, 7.0). PES was well tolerated, without adverse effects or associations with serious complications (chest infections/death). Conclusions. Although the direction of observed differences were consistent with PES accelerating swallowing recovery over the first 2 weeks postintervention, suboptimal recruitment prevents definitive conclusions. Our study design experience and outcome data are essential to inform a definitive, multicenter randomized trial.
Introduction
Oropharyngeal dysphagia is a major complication of stroke (detected in up to 78% of patients 1 ) and is considered a major adverse prognostic factor with increased risks of pulmonary aspiration, 1 malnutrition, increased lengths of hospital stay, and death. 2 Current rehabilitative strategies, including behavioral therapies have limited evidence base. 3 However, with recent developments in our understanding of the role of neuroplasticity and cortical reorganization in swallowing recovery,4-6 there is now increasing interest that neurostimulation techniques can drive this natural recovery process. 7 Intraluminal pharyngeal electrical stimulation (PES) is one such neurostimulation technique that has been shown to promote this type of plasticity in healthy individuals and achieve measurable improvements in swallowing function in dysphagic stroke patients in studies spanning more than a decade.8-12 In contrast to behavioral swallowing treatments, PES appears to promote functional benefit through a central nonbiomechanical mechanism, such that dysphagia is improved regardless of physiological deficit. Furthermore, the optimal stimulation parameters (5-Hz frequency, 10 minutes duration, and 75% of maximum tolerated intensity) 8 have been shown to reverse the neurophysiological and behavioral effects of experimental brain lesions,9,13 and a dose-response study provided an optimal regimen in dysphagic stroke patients—once-daily stimulation for 3 days. 9 Moreover, in randomized clinical studies, PES improved swallowing within an hour poststimulation in both acute 8 and chronic 11 dysphagic stroke patients, as measured with videofluoroscopy (VFS). Finally, data from a small phase II randomized controlled trial in acute stroke patients (n = 28) demonstrated positive effects of 3 days of active PES, which at 2 weeks postintervention improved feeding status as measured by the Dysphagia Severity Rating (DSR) Scale, 9 which correlated with the Penetration-Aspiration Scale (PAS) 14 and was linked to reduced length of hospital stay. 9
The aim of this phase II, single-blind randomized controlled trial was to assess whether PES improves swallowing in poststroke dysphagia, with clinical effects in longer-term follow-up.
Methods
The study was approved by a regional NHS research ethics committee (Greater Manchester South 09/H1003/74) and complied with CONSORT guidlines. 15
Sample Size
Data from the pilot trial of PES 9 revealed a mean improvement (SD) of 1.8 (1.77) in the number of swallows scoring >3 on PAS 14 after active compared with a deterioration of 0.6 (1.56) swallows following sham. 9 Based on these data we calculated that 50 patients per group would provide 80% power at the 5% significance level to detect a difference between groups of 1 swallow, based on a common SD of 1.75. Unfortunately, during the trial, one of the recruiting sites unexpectedly became unable to accomodate requests for VFS examinations. Because of these unforseen circumstances, the DSR at 2 weeks 9 was upgraded to be the primary outcome measure prior to unblinding of any data.
Screening and Recruitment
Stroke patients who fulfilled inclusion criteria were identified and approached on the stroke units in 3 Greater Manchester hospitals (Salford Royal, University Hospital of South Manchester, and Trafford General). All recruited participants either provided informed written consent, or if they lacked capacity as a result of stroke, they were recruited if a named consultee declared that in their opinion, the patient would not have objected to participation in the trial.
Patients included (no age limit) were those with new-onset dysphagia following anterior or posterior cerebral circulation, within 6 weeks of ictus. Patients with both ischemic stroke and hemorrhagic stroke were included. All recruited patients were medically stable at inclusion. None of the included patients had been intubated/trachiotomized during hospitalization prior to recruitment.
Exclusion criteria included advanced dementia, other neurological conditions that may explain dysphagia, previous history of dysphagia, presence of cardiac pacemaker or implanted cardiac defibrillator, a diagnosis other than stroke (eg, brain tumor), any severe concomitant chronic medical condition that compromises cardiac or respiratory status (severe emphysema or heart failure that may render catheter intubation unsafe), and significant structural abnormalities of the mouth or throat. Patients requiring continuous oxygen treatment were also excluded.
Randomization
Following consent and baseline assessment, patients were randomized through a concealed program created by our information technology department. Allocation was blocked in randomly permuted sizes and stratified by center and feeding status (presence/absence of artificial feeding) to optimize balance. Patients were enrolled by the neurogastroenterology clinical research fellows, and participants were assigned to their groups by the chief investigator.
Procedures
Bedside Screening Swallowing Test
All recruited patients had a standardized bedside swallowing test using the validated Toronto Bedside Swallowing Screening Test (TOR-BSST) 16 conducted by a trained researcher at study entry. Those failing TOR-BSST proceeded to a baseline instrumental swallowing examination, which consisted of either VFS by the research team (where available) or a fiberoptic endoscopic examination of swallowing (FEES) carried out by the supervising clinical team. When neither instrumental test was possible, those failing TOR-BSST were directly randomized into the trial. In contrast, patients who passed TOR-BSST were classed as nondysphagic, and their participation was discontinued.
Videofluoroscopy
Participants who were fit enough at baseline had a standardized VFS assessment of their swallow (where available). This examination comprised up to 6 swallows of 5-mL boluses of liquid barium (60% w/v, EZ-HD, E-Z-EM Limited, UK), with lateral view images (Siemens Fluorospot H SIRESKOP SX Unit, Germany) carried out at the radiology department of the recruiting sites. As a safety precaution, when significant signs of swallow compromise were demonstrated during VFS (eg, PAS of 7 or 8 for 3 consecutive 5-mL volume swallows), the procedure was stopped (regardless of the number of swallows performed up to that point).
Digitally captured images were reviewed offline frame by frame for evidence of ingress of material into the airway 14 independently by 2 blinded speech and language therapists (SaLTs) with special interest in dysphagia, blinded to study allocation. These examinations were repeated at 2 weeks and 3 months postrandomization.
Patients with normal swallowing at baseline VFS (where all recorded swallows had PAS <3) were excluded from receiving their randomized interventions, whereas those with evidence of dysphagia (at least 1 swallow of PAS ≥3) on VFS or FEES received randomized intervention (active or sham).
Fiber-optic Evaluation of Swallowing
In some patients, where VFS was not possible (because of patients’ fitness or unavailability of VFS slots), FEES was performed by the clinical supervizing team using standard methods to assess laryngopharyngeal structures and secretions to liquid boli. FEES was applied via the right or left nostril using a Pentax scope connected to a Digital Swallowing Workstation (Model 7200, Kay Elemetrics Corp, Lincoln Park, NJ). PAS scores derived from FEES were used to confirm dysphagia in these patients and were used in the data analyses for secondary outcome measures.
Dysphagia Severity Rating Scale
Based on detailed clinical bedside assessments performed by independent (blinded) trained SaLTs based at the sites, feeding recommendations were incorporated to calculate the patients DSR 9 at baseline, 2 weeks, and 3 months postinterventions. This scale, originally adapted from the Dysphagia Outcome and Severity Scale, 17 has previously been validated against VFS 9 and reflects the feeding status achieved by the patient across 3 domains: fluids, diet, and level of supervision required for feeding. The score for each domain ranges from 0 to 4 and is summated to give a total composite score. These scores are calculated based on clinical recommendations from independent SaLTs with special interest in dysphagia.
Pharyngeal Electrical Stimulation
Interventions were delivered at the patient’s bedside. Based on previous pharyngeal electrode placement experience in clinically dysphagic patients, 11 the intraluminal pharyngeal “stimulation” catheter (Gaeltec, Dunvegan, Isle of Skye, UK) was inserted either orally or nasally (depending on patient preference) such that its bipolar electrodes were secured at the midpharyngeal level (17 cm from the nasal flare or 15 cm aboral). The catheter was connected to a stimulator (Model DS7; Digitimer, Welwyn-Garden City, Herts, UK) via a trigger generator (Neurolog System, Digitimer), and stimuli were delivered (0.2 ms pulses, maximum 280 V) at the previously defined optimal parameters (5 Hz frequency and an intensity [current] 75% of the maximum tolerated). 8 The maximum tolerated intensity was determined from each patient’s perception and pain thresholds; these values were calculated from an average of 3 consecutive measurements on each of the 3 days. Group 1 received 3 sessions of PES for 10 minutes on 3 consecutive days. Group 2 received sham stimulation (catheter in situ with stimulator turned off) for the same period. Both groups continued to receive standard swallowing treatments as decided by SaLTs of the respective hospitals. Interventions were delivered by a trained researcher independently of the clinical team. Patients and SaLTs who independently assessed the outcomes (DSR/instrumental swallowing examinations) were blinded to group allocation. The researcher who delivered the interventions was not blinded to the group allocation.
Data Analyses and Statistical Methods
Logistic regression analyses were performed by independent medical statisticians at the University of Manchester using the R statistical package. 18 The primary analysis was conducted under the intention-to-treat framework: data were analyzed according to whether patients were allocated to active or sham treatment, not according to the treatment they actually received. If a patient withdrew from the study, we endeavored to obtain their hospital records for the study period and use any appropriate and relevant outcome data.
Primary Outcome: DSR at 2 Weeks Postintervention
Examination of an independent patient group with characteristics similar to those in this study showed the distribution of DSR scores to be highly nonnormal, bimodally distributed at the extremes, and with a median DSR of 3.5. 9 Thus, DSR scores at 2 weeks were dichotomized from the 13-point severity scale (0-12) into a binary outcome, indicating mild or no dysphagia (scores from 0 to 3) or moderate to severe dysphagia (scores from 4 to 12). Thus, the binary DSR score at 2 weeks was analyzed as the response in a generalized linear model, with treatment allocation as the factor of interest and including age (<75 or ≥75 years) and the stratified randomization factors—namely, feeding method and treatment center—as covariates. Compared with the sham group, an odds ratio (OR) >1 would indicate a favorable outcome for the active group.
Secondary Outcomes
DSR at 3 months was analyzed similarly to DSR scores at 2 weeks. The time from randomization until medically fit for hospital discharge (days) was analyzed using a Cox proportional hazards model, including feeding method, age, and treatment center as covariates. For those patients with either a nasogastric tube (NGT) or a percutaneous endoscopic gastrostomy (PEG) feeding tube inserted, the time from randomization until tube removal censored at study end was similarly analyzed.
For models of time to discharge and time to tube removal, a hazard ratio (HR) >1 for the treatment factor would indicate shorter times to discharge and a positive treatment effect. The proportional hazards assumption of these models was assessed by visual inspection of plots of the scaled Schoenfeld residuals over time. 19
Instrumental swallowing examination (VFS/FEES) outcomes were analyzed for patients who had these examinations. VFS/FEES swallows with a PAS ≥3 were defined as unsafe. The counts of these unsafe swallows, as a proportion of the number of trials (up to a maximum of 6), were analyzed in a binomial regression model using data from all centers involved in the study at both 2 weeks and 3 months. An estimated OR <1 would indicate a favorable outcome for the active treatment group compared with the sham treatment group.
The number of adverse events—chest infections and death—before study end were also evaluated and compared between groups.
Results
Patients were assessed for eligibility and recruited at acute stroke units of the participating sites between February 2009 and January 31, 2012. The recruitment target of 100 patients proved to be unachievable during the time frame because the majority of eligible patients (137/207, 66%) were either transferred from the hyperacute stroke center for ongoing rehabilitative care at a nonrecruiting site or were deemed to be clinically unsuitable (severe pneumonia, concurrent acute illnesses, inability to comply with swallowing assessments and interventions because of consciousness levels, or requiring supplemental oxygenation). A total of 36 patients were randomized, 18 in each interventional arm (Figure 1). One patient withdrew from the trial for reasons independent of health, and no further data could be obtained for this patient. Two patients died, and their missing outcomes were imputed as the worst possible values for all analyses. Baseline characteristics of patients are summarized in Table 1. Of the 14 patients who actually received active PES, only 1 patient received suboptimal stimulation (2 doses), whereas the rest received all 3 doses.
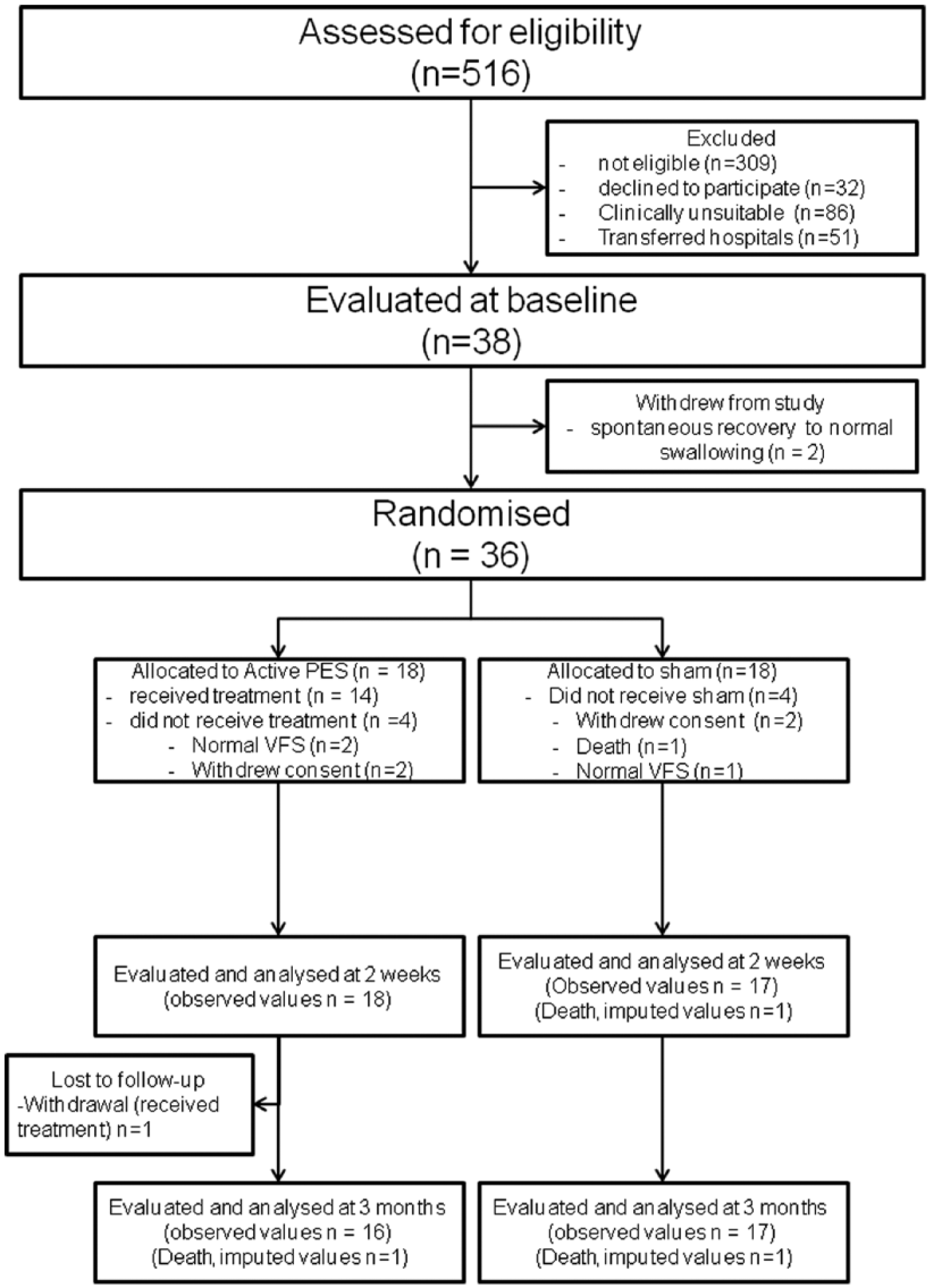
Data flowchart: numbers, and reasons for dropping out.
Summary of Patient Characteristics. a
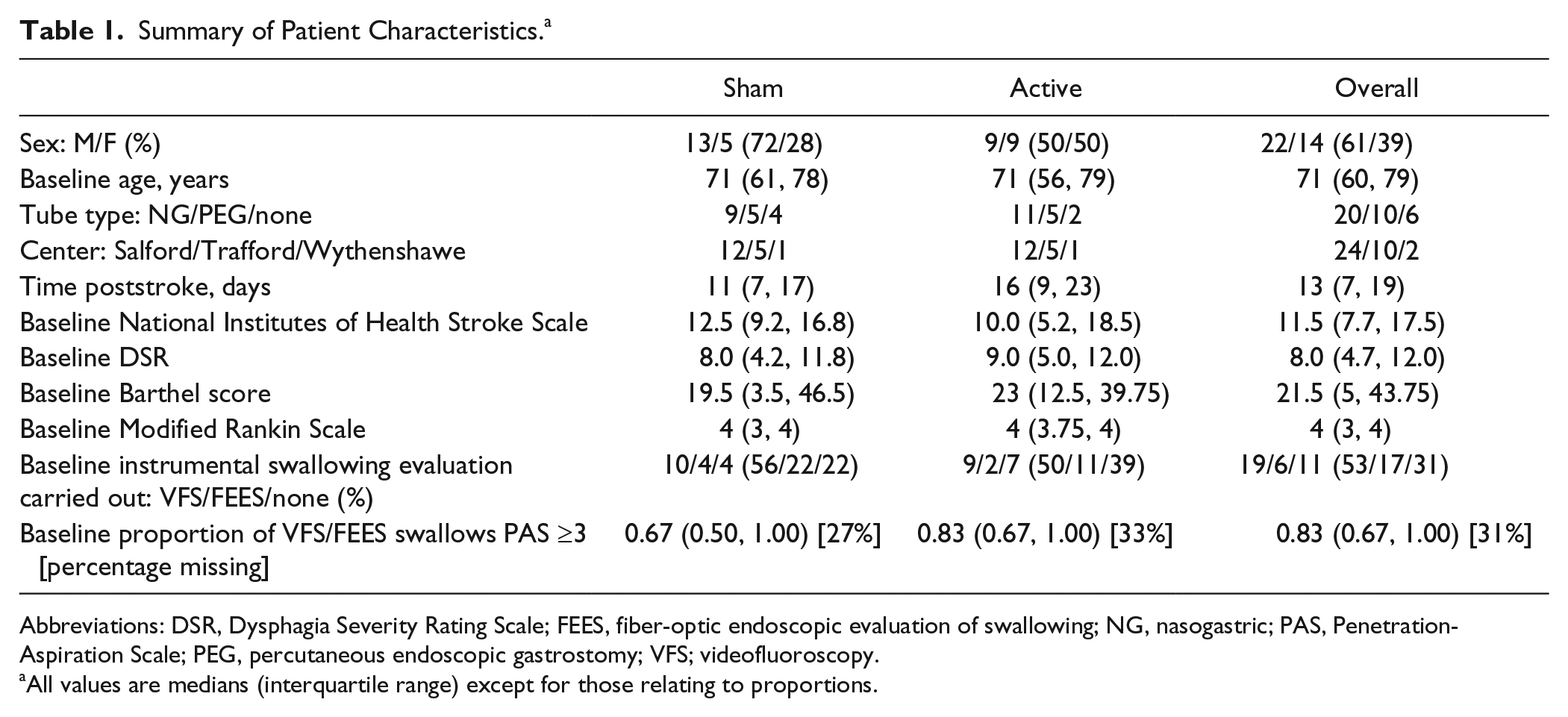
Abbreviations: DSR, Dysphagia Severity Rating Scale; FEES, fiber-optic endoscopic evaluation of swallowing; NG, nasogastric; PAS, Penetration-Aspiration Scale; PEG, percutaneous endoscopic gastrostomy; VFS; videofluoroscopy.
All values are medians (interquartile range) except for those relating to proportions.
Stimulation Intensity
Median stimulation intensities (in milliamperes) for active PES (interquartile range), derived from daily measurements of sensory and pain thresholds (see Methods), appeared to decrease between day 1 and day 3 of treatment: day 1, 19.9 (12.3, 25.9); day 2, 18.1 (10.3, 21.3); day 3, 12.5 (10.7, 23.9). This numerical change was not as apparent in sham PES where the catheter was in situ but the current was switched off: day 1, 17.2 (12.9, 28.1); day 2, 17.7 (12.2, 27.4); day 3, 16.3 (12.2, 18.6).
Primary Outcome
A total of 36 patients were included in the analysis for the primary outcome at 2 weeks: 18 in the active treatment arm and 18 in the sham arm (1 patient had died). At the 2-week follow-up, 11 patients (61%) had no/mild dysphagia in the active treatment group, and 9 patients (50%) had no/mild dysphagia in the sham group. Only 1 patient in the sham group had a DSR that was worse at the 2-week follow-up than at baseline assessment (from a score of 9 to 10), and no patient in the active treatment group had worse DSR than baseline at 2-week follow-up. The treatment effect of the relative presence of no/mild dysphagia (by DSR) at 2 weeks was estimated by an OR (95% CI) of 2.5 (0.52, 14), not significant by the likelihood ratio test (P = .26). Figure 2 illustrates the observed DSR scores in both arms over the study duration.
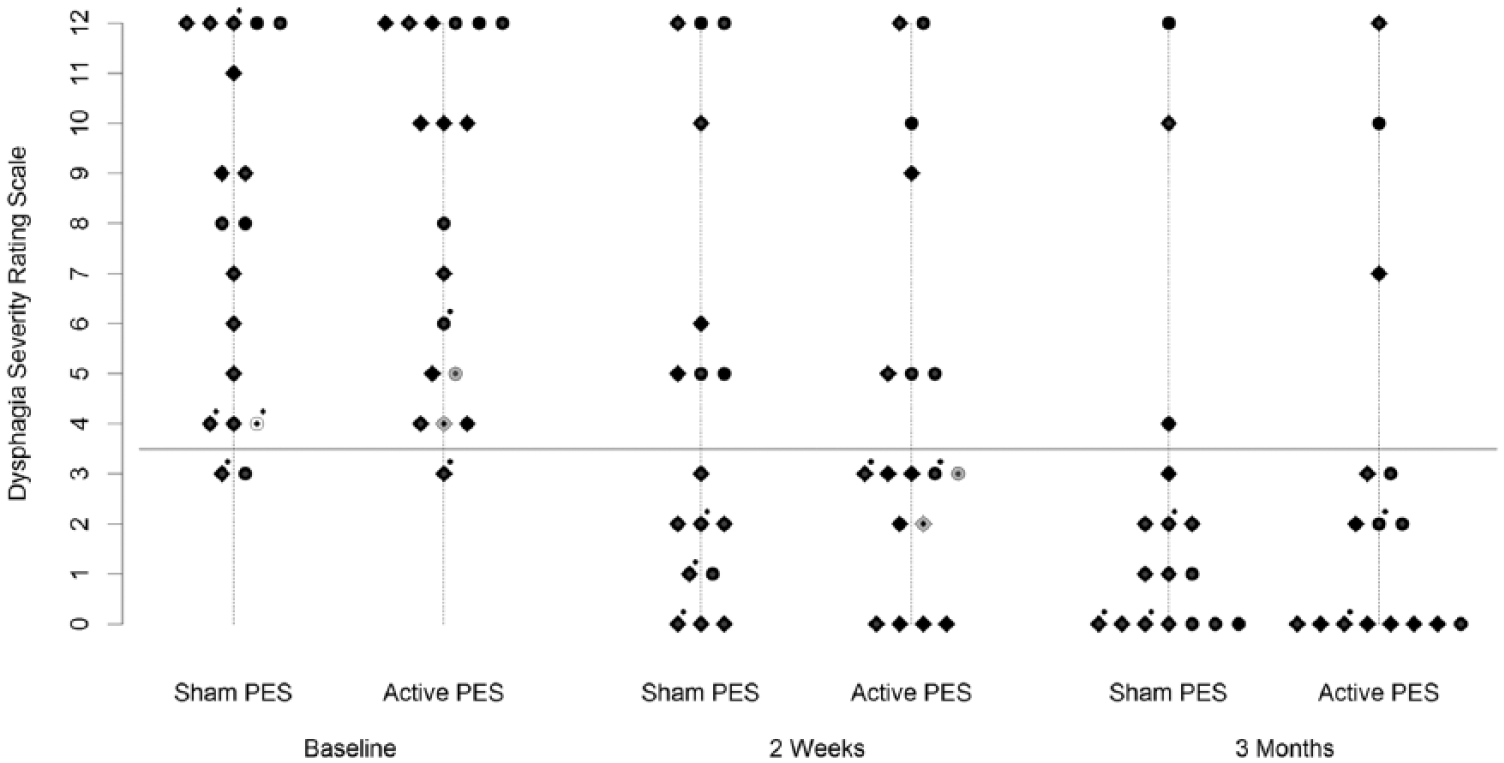
Observed Dysphagia Severity Rating Scale measurements in each trial arm and at each time point. White and gray-filled points indicate patients withdrawing from the trial before the 2-week and 3-month evaluations, respectively. Diamonds indicate patients treated at the primary center (Salford) and circles, those treated outside (Trafford/Wythenshawe). Gray outlines indicate male patients, and black outlines indicate female patients. Dots over points indicate patients feeding naturally; all others were feeding by either nasogastric or percutaneous endoscopic gastrostomy feeding tubes.
Secondary Outcomes
There were 35 patients included in the analysis for secondary outcomes at 3 months—18 in the control arm and 17 in the active treatment arm (1 patient in each group had died)—with 1 patient in the active arm having been lost to follow-up. No patient in either arm had an observed DSR that increased between the 2-week and 3-month follow-up assessments. At the 3-month follow-up, 14 patients (78%) had no/mild dysphagia in the control group, and 13 patients (76%) had no/mild dysphagia in the active treatment group. The treatment effect of the relative presence of mild/no dysphagia (by DSR) at 3 months was estimated by an OR (95% CI) of 0.97 (0.13, 7.0), not significant by the likelihood ratio test (P = .97).
The median times from randomization until hospital discharge were 39 days and 52 days in the active and sham groups, respectively (Table 2). A Cox proportional hazards analysis of time from the date of randomization until hospital discharge estimated a HR (95% CI) of 1.2 (0.55, 2.5). No significant difference between arms was observed by the stratified log-rank test (P = .62). Figure 3A shows the observed survival probabilities (probability of not being discharged) for patients in each study arm.
Number and Median Survival Times of Hospital Discharges and Feeding Tube Removal From Randomization Until 90 Days.
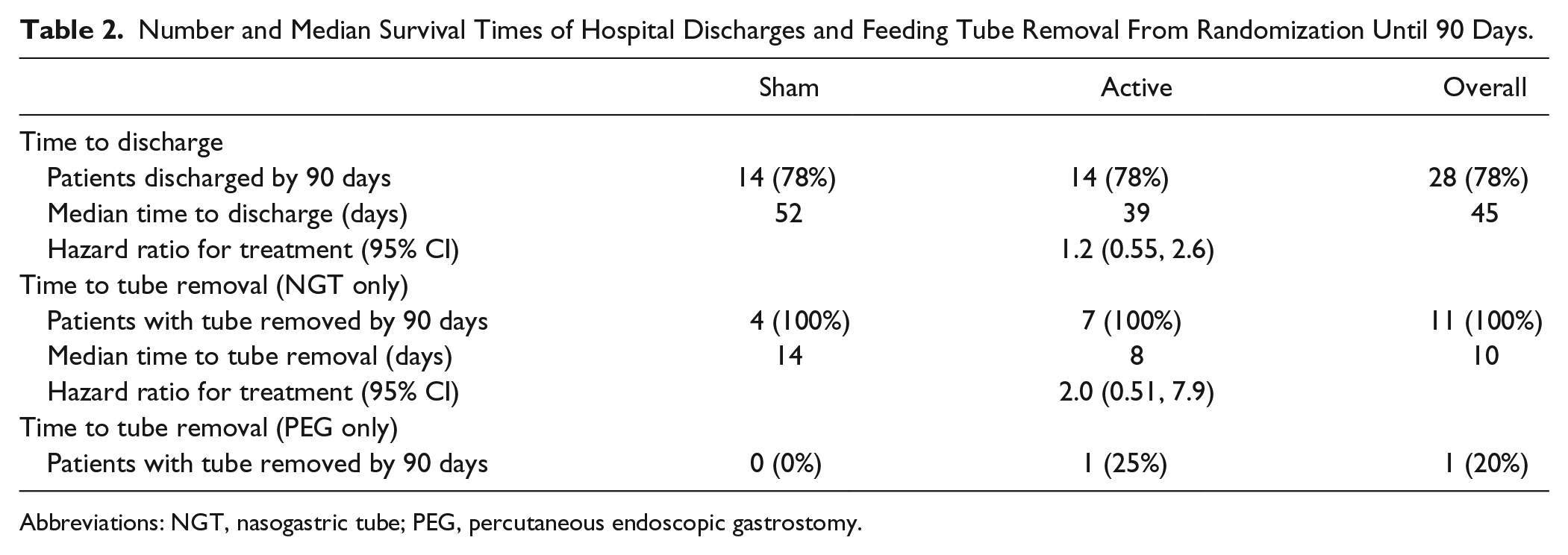
Abbreviations: NGT, nasogastric tube; PEG, percutaneous endoscopic gastrostomy.
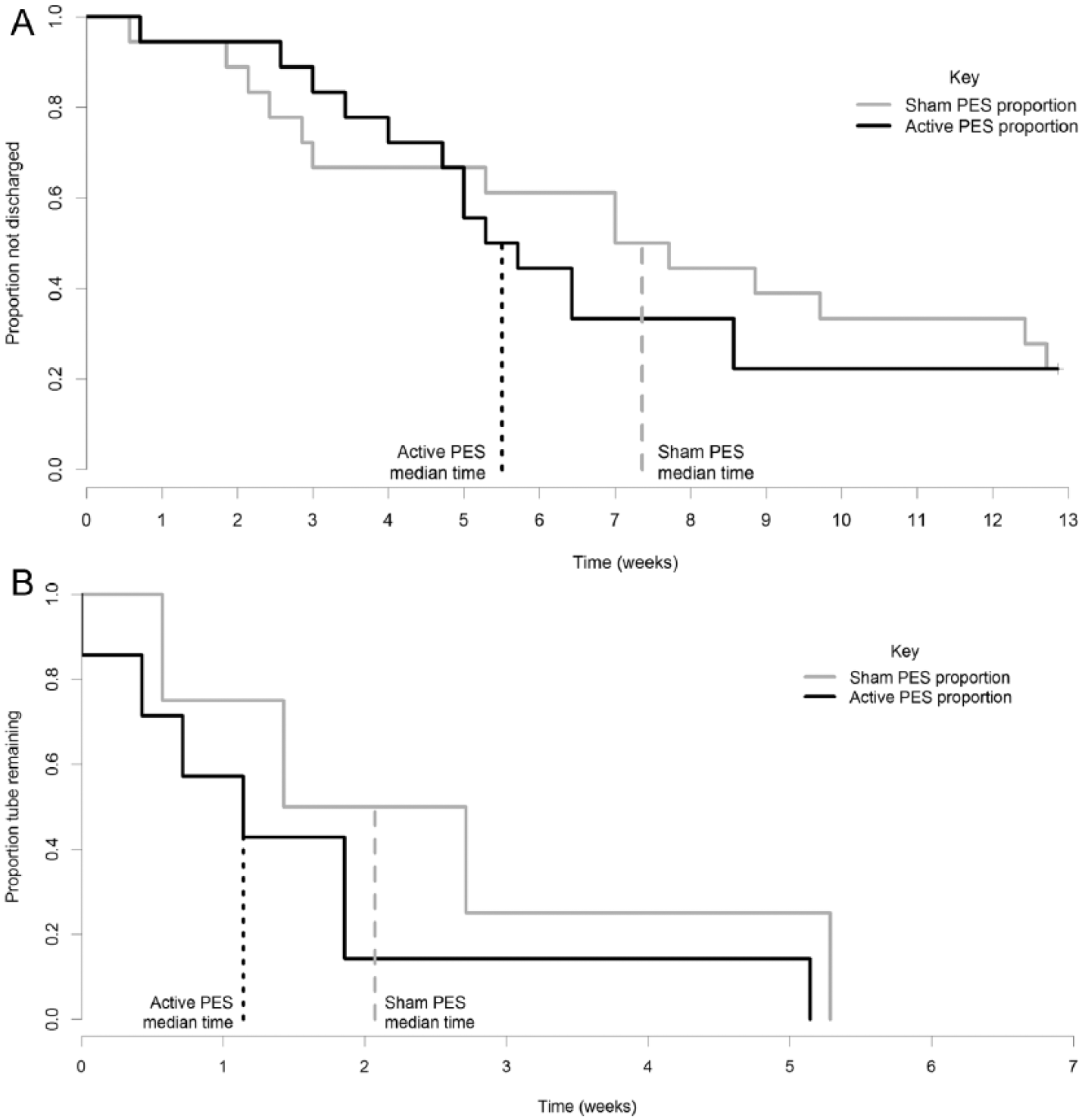
Kaplan-Meier estimate of survival curves for time postrandomization to (A) hospital discharge (B) removal of nasogastric feeding tubes.
A total of 21 patients had a feeding tube inserted at the time of randomization: 11 NGTs and 10 PEG tubes. At study completion, all 11 NGTs had been removed, whereas only 2 PEG tubes (20%) had been removed. The median time to removal of NGTs from the date of randomization was 8 days for active compared with 14 days for the sham treatment group (Table 2 and Figure 3B). A Cox proportional hazards analysis estimated a HR (95% CI) of 2.0 (0.51, 7.9), not significant by the stratified log-rank test (P = .33).
At baseline, the majority of patients had instrumental swallowing evaluation (VFS or FEES; Table 1) confirming dysphagia (ie, PAS ≥ 3). At 2 weeks, only 18/36 (50%) had instrumental swallowing examinations (16 VFS and 2 FEES with at least 1 swallow recorded) and were included in the analysis, with 9 (50%) of these in each intervention group. In the sham group, 7/9 patients had 6 VFS swallows recorded, whereas 2 patients had 1 swallow recorded at FEES. In the active treatment group, 8/9 patients had 6 VFS swallows recorded, and 1 patient met the stopping criteria after 5 swallows. For the 50% of patients in whom instrumental swallowing examinations were carried out at 2 weeks, the median (IQR) proportion of trials with a PAS ≥3 was 0.67 (0.67, 1.00) for the sham treatment group and 0.5 (0.16, 0.83) for the active treatment group. At 2 weeks, the estimated OR (95% CI) for PAS ≥3 was 0.61 (0.27 to 1.4); P = .23. At 3 months, 13 participants (36%) had VFS and 0 patients had FEES. Of the VFS patients, 5 were from the sham treatment group and 8 from the active treatment group—all participants had at least 5 VFS swallows. For the 36% of patients on whom VFS was carried out at 3 months, the median (IQR) proportion of trials with a PAS ≥3 was 0.50 (0.00, 1.00) for the sham treatment group and 0.08 (0.00, 0.25) for the active treatment group. The estimated OR (95% CI) for PAS ≥3at 3 months was 0.38 (0.13 to 1.08); P = .07.
One patient in the active group suffered a chest infection approximately 2 months after randomization, which responded to a 5-day course of antibiotics. One patient from each arm died within the duration of the study; the patient in the sham group was withdrawn from the study before application of sham PES and died 2 weeks postrandomization (from aspiration pneumonia). The patient in the active treatment group had self-discharged from hospital against medical advice (42 days after randomization) and died 11 days later from an unknown cause. These patients had baseline DSR scores of 4 and 5, respectively.
Discussion
From this study, given the smaller-than-planned recruitment target, the effects of PES on dysphagia recovery in stroke patients remain inconclusive. Despite this, there are some indications of a potentially favorable outcome to this treatment, as suggested by the consistency of the direction of the estimated effects relating to dysphagia severity at 2 weeks, the time to NGT removal, the time to discharge (ORs and HRs are all >1), and the observed improvement in the number of unsafe swallows (PAS ≥ 3) in the limited number of patients who had instrumental examinations by 3 months. PES was well tolerated, with no adverse effects related to the therapy. These findings taken together would be consistent with the hypothesis and add to the evidence base, including the previously demonstrated effects of PES in expediting the recovery of poststroke dysphagia.9,11
Differences in the baseline characteristics between the first randomized control trial 9 and those in the present study may partially explain the estimated lesser effect of PES on DSR than in the previous study. Baseline data in the present study (Table 1) reveal higher National Institutes of Health Stroke Scale and DSR scores (both overall mean values were approximately 2 points higher than in the study by Jayasekeran et al 9 at baseline) compared with the first trial. This could produce differences in the rehabilitation needs and resultant length of stay (which would be influenced by factors independent of swallowing status). Higher baseline stroke severity also has an impact on the incompleteness of VFS data from the centers where this was available, with many patients being physically unable to sit in a chair and unable to follow commands for VFS examination either because of impaired cognitive function and/or disabilities secondary to stroke severity. Second, in the present study, there was more improvement in the sham group (presumably related to natural swallowing recovery) during the first 2 weeks than predicted from the power calculation. This level of natural recovery was not so evident following sham PES in the study by Jayasekeran et al, 9 where there was an observed deterioration in VFS PAS and only minimal improvement in DSR at 2 weeks. The present cohort of patients may represent a more realistic population than the former study, bearing in mind that the lower baseline DSR (5.6 in the sham group 9 ) may have offered these patients less room for improvement over time compared with those in the present study. At 2 weeks, 7/18 (39%) patients were nonresponders to active PES based on DSR. Similar to our findings, Jayasekeran et al 9 described a distinct group of nonresponders to PES. There is now evidence that genetic variance is an important factor in the neuroplastic response to neurostimulation interventions, 20 and in particular, polymorphisms in the brain-derived neurotrophic factor gene may be predictive of nonresponse to PES but favorable response to other cortical neurostimulation modalities. 21 Future studies may explore the role of genetic variance in subselecting patients for specific neurostimulation interventions in dysphagia rehabilitation.
By the end of the 3-month follow-up in the present study, only 17% of patients had moderate/severe dysphagia, with no difference in the prevalence between the 2 interventional groups. This suggests that the main effects of PES may be in early expedition of the natural swallowing recovery process, rather than in influencing the long-term swallowing outcome. Such expedition of swallowing recovery could be seen as clinically useful, in line with other clinically relevant outcomes: we estimated earlier NGT removal and shorter lengths of inpatient stay (although neither effect was statistically significant). In terms of serious complications in the present study, there was 1 respiratory infection and 1 death in each interventional group; such end points usually require many hundreds if not thousands of patients to demonstrate an effect, so it is not surprising that PES interventions could not be shown to influence these particular secondary outcome measures. There are limitations to this study. First, it was powered to detect a VFS-related difference in swallowing outcome. However, for reasons unrelated to the study population, VFS measurement was not possible in all patients. As such, given that the study was powered using a VFS outcome, the statistical power to detect a clinically important treatment effect on the DSR was going to be difficult. The protocol specified 50 patients per arm, but in the final study trial, recruitment only reached 36% of its targeted number of patients. Our recruitment experience from this trial highlights the difficulties in obtaining larger sample sizes in interventional studies in subacute poststroke dysphagia. Given the high incidence of clinical instability in a large proportion of eligible patients, a larger number of participating sites (including nonhyperacute stroke centers) would be required to achieve the recruitment targets for a much-needed definitive study. It is possible that the current pragmatic outcome measure may require even more patients; our estimate is that it is closer to 100 patients per arm rather than 50 patients. This lower recruited number also diminishes the ability of randomization to achieve balance on important prognostic factors. In addition to higher baseline DSR, patients in the active group were recruited later in relation to the date of stroke (Table 1) compared with the sham group, and these factors may also have important adverse prognostic significance for these recruited participants in terms of their dysphagia neurorehabilitation potential at baseline. DSR, which is a more pragmatic outcome measure, may be less sensitive than VFS because it scores largely on observations from bedside clinical assessments of swallowing by trained SaLTs rather than instrumental examinations (VFS/FEES). It is recognized that bedside testing would not detect silent aspiration, and there is evidence that without direct visualization of the swallowing mechanism, subtle abnormalities can be missed in a bedside examination. 22 It is also possible that the lack of a consistent test to determine dysphagia at baseline may have contributed to nonsignificant findings, given that bedside screening tools such as the TOR-BSST generally have lower sensitivity, with a higher false-positive rate. Thus, using the TOR-BSST when VFS/FEES was not available to determine dysphagia may have resulted in patients with normal swallowing being identified as dysphagic in either of the 2 groups. However, this only represented a very small proportion of patients recruited. The rationale for including this group with clinical dysphagia in the analyses was based on the primary end point (DSR) also being a clinical measure of dysphagia severity, given that DSR, in the preliminary trial, 9 was shown to change in accordance with VFS measures. A further limitation is that many patients did not receive their first intervention on the day of randomization because of the scheduling of instrumental examinations, which resulted in a mean delay of 2.5 days to treatment. Nonetheless, under the intention-to-treat analyses, DSR values used are aligned to 2 weeks postrandomization rather than 2 weeks postintervention. This discrepancy may miss some additional improvements of DSR (with or without PES) between 2 weeks postrandomization and 2 weeks posttreatment. Finally, it is possible that behavioral interventions performed as part of routine clinical practice by SaLTs may have varied between treatments centers and therapists. However, in contrast to behavioral therapies, given that PES appears to promote swallowing recovery regardless of the physiological deficits via a central, nonbiomechanical mechanism,8,11 it is doubtful whether this would have influenced outcomes. Moreover, it is expected that the groups would be matched for standard therapies, given the similar baseline characteristics (Table 1). The effects of PES combined with specific behavioral exercises by SaLTs directed against specific swallowing deficits were not examined here and could be explored in future studies.
Conclusions
Because of the listed limitations, this study could not confirm that PES accelerates swallowing recovery in poststroke dysphagia. Despite not being a definitive study, the observed direction of the differences in outcome measures was consistently in favor of active treatment. The important lessons learned will help inform an independent, definitive multicenter international trial of this intervention, which is now ongoing. 23
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of Professor Pippa Tyrrell; Dr Ed Gamble from University Hospital of South Manchester; Dr Sajjad Anwar from Trafford General Hospital; Dr Vanoo Jayasekeran; Dana Sandars (an independent SaLT), who was the second blinded reader of VFS examinations; research practitioners from Greater Manchester Stroke Research Network; and senior radiographers Lisa Renaut, Jackie Johnson, Rachel Rae, and Danielle Burgess.
Authors’ Note
Clinical Trial Registration: ISRCTN 83103698.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SH provides scientific advice via a secondment agreement with the University of Manchester to a medical device company focusing on dysphagia (Phagenesis Ltd), which manufactures the Phagenyx device. He also sits on the Phagenesis Ltd board of directors as a founder and owns shares in the company.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article presents independent research funded by the National Institute for Health Research (NIHR) under its Research for Patient Benefit (RfPB) Programme (Grant Reference Number PB-PG-0107-12076). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
