Abstract
The remarkable scientific and technological advances in the field of cell research have not been translated into viable restorative therapies for brain disorders. In this article, we examine the best available evidence for the clinical efficacy of reconstructive intracerebral transplantation in people with Parkinson’s disease (PD), with the aim of identifying methodological obstacles to the translation process. The major stumbling block is the fact that the potential contributions of people with neural grafts and the effects of the physical and social environment in which they recover have not been adequately investigated and applied to advancing the clinical stages of the research program. We suggest that the biopsychosocial model along with emerging evidence of targeted rehabilitation can provide a useful framework for conducting research and evaluation that will ensure the best possible outcomes following intracerebral transplantation for PD.
Keywords
Introduction
The reconstruction of the human brain is one of the great challenges facing neuroscientists and clinicians working to develop cellular therapies for people suffering from a variety of central nervous system (CNS) disorders.1-3 The intrastriatal transplantation of embryonic cells in animal models of basal ganglia impairments began in the late 1970s, initiating the contemporary research program targeting a variety of brain disorders, including Parkinson’s disease (PD), Huntington disease (HD), and stroke.4,5 The central idea driving this research program is that immature cells from various sources, including embryonic, stem, or xenogenic, can be used to replace neurons lost through disease or injury, restore impaired neural networks, and reinstate behavioral functioning.1,6,7 The newly grafted cells do not restore impaired brains to their original state, but rather a conceptual framework (Composite Brain Model) is applicable to identify the ways in which the grafted cells are integrated into functional neural circuits. Considering that there is substantial evidence that movement rehabilitation in people with PD is of therapeutic benefit,8,9 it is puzzling that combining rehabilitation with neural transplantation has received scant attention.
Research in the area of embryonic and stem cell research has generated major discoveries of neuronal and CNS functioning and has advanced the technology of repairing neuronal circuitry in mammalian brains.10,11 Despite numerous reports of clinical benefits, none of the transplanted cell lines have been identified as the best available treatment for any of the targeted brain disorders. Abundant reviews have been published, seeking to identify the obstacles to the translation of laboratory discoveries to clinical practice and to identify the best path to follow for ensuring the advancement of reconstructive cellular therapies.1,10,12-15
The flowchart shown in Figure 1, modified from Polgar and Morris, 16 describes the process followed by researchers to develop and evaluate reconstructive cellular therapy for PD. The process begins with the selection of a cell line that has been identified as having the potential for development into the best-practice treatment for a specific CNS disorder. 17 Figure 1 represents that most current research is conducted at the level of the basic biological sciences (stages 1 and 2). There is a bottleneck in the translation process 18 because only a small number of cell lines that are studied in the laboratory are subjected to further clinical research and evaluation (stages 3 and 4). Consequently, only a limited number of cell lines have been adequately evaluated to produce evidence for conducting systematic reviews and meta-analyses and to enable evidence-based policy decisions for implementing practices (stages 5 and 6).
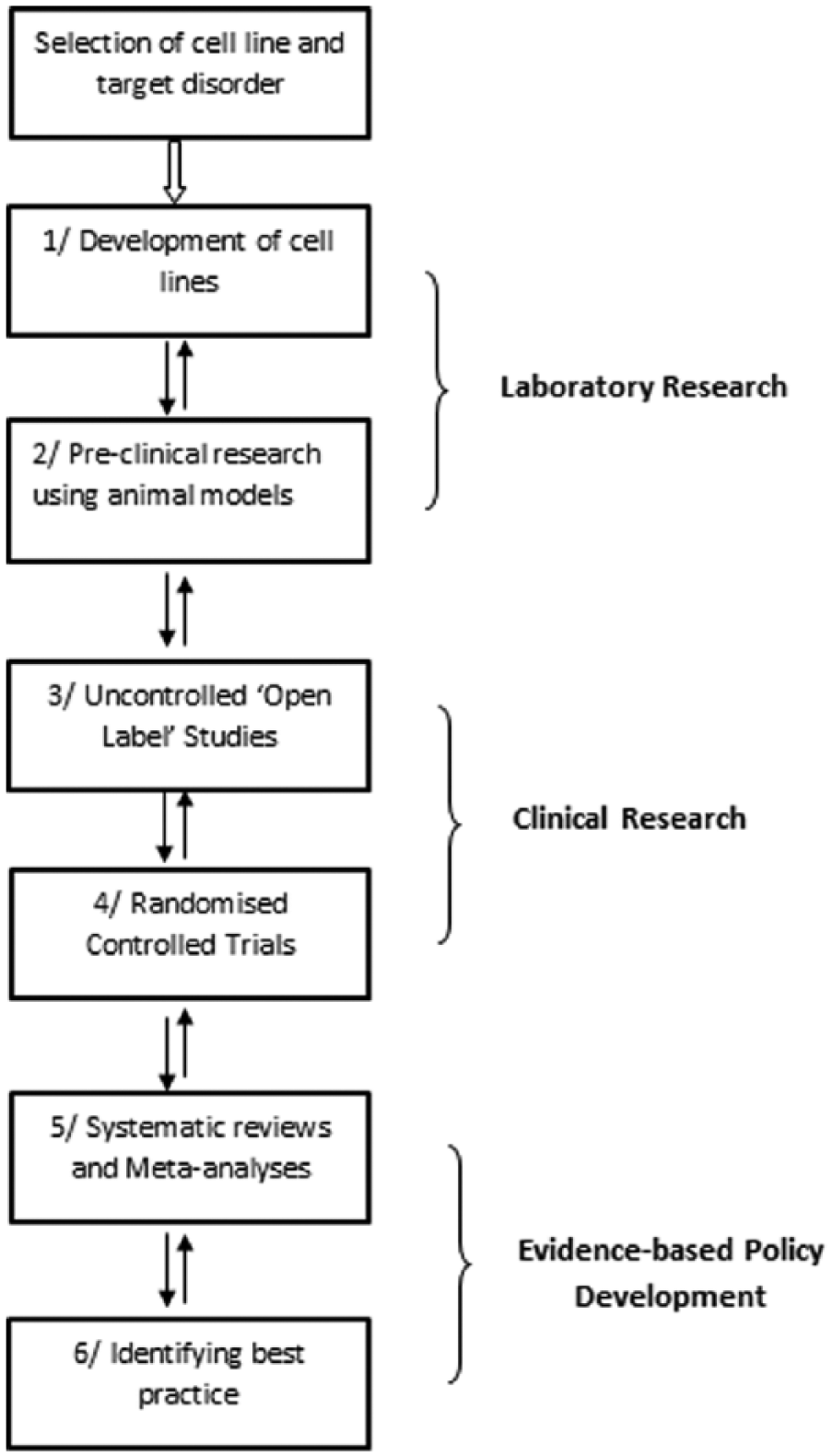
Structure of the research program.a
In the present article, we focus on a subset of the methodological problems that have been bedeviling the clinical stages of the translation process and outline a strategy for ensuring the advancement of the research program. The term method is used in the present context with 2 different but interrelated meanings; first, in reference to the procedures followed in devising and implementing the practice of reconstructive cellular therapies and, second, the way in which research and evaluation are conducted to achieve the scientific and clinical objectives. The specific aims of this position paper are to the following:
discuss the best available evidence for the efficacy of intracerebral grafts for the treatment of PD;
describe specific methodological challenges associated with the selection of the designs used to conduct research and evaluation; and
briefly outline an integrated methodological approach to optimize the translation process for creating the best-practice cellular therapies for people with PD.
The reason for selecting reconstructive cellular therapies for PD is that all the stages of laboratory and clinical research shown in Figure 1 have been completed, at least in a preliminary fashion, to reveal the key methodological obstacles in the translation process.
Preclinical Research
Preclinical studies (stages 1 and 2, Figure 1) provide the foundation evidence and conceptual frameworks for research in the area of cellular therapies. These studies use experimental designs and are usually laboratory based. They rely on animal models of brain disorders to investigate the causal mechanisms by which the transplantation of various cell lines result in structural and functional benefits. The evidence produced enables researchers to estimate the efficacy of the cell lines for reconstructing the brain and to identify some of the risks associated with the transplantation of a specific cell line.10,19
One of the problems confronting clinical researchers is that while investigating the potential benefits of grafting a cell line, the understanding of the neuropathology of the targeted brain disorder may change. For example, the neuropathology of PD was originally attributed to the degeneration of neurons in the pars compacta of the substantia nigra, resulting in reduced dopamine (DA) transmission in the neostriatum. Although the impairment of DA expression and the effect of this on synaptic transmission in the basal ganglia remains the cornerstone for understanding the etiology of PD as a movement disorder, there are also progressive impairments in other parts of the CNS. 20 Concomitantly, symptoms other than motor dysfunctions and movement limitations have been identified as being symptomatic of PD, including cognitive and emotional impairments. 21 It is now recognized that the replacement of DA-producing cells will not be a cure for PD, but rather that the research program may lead to the best available treatments for patients with advanced movement impairments.
The central theme developed in the present article is the importance of the reciprocal interaction between the grafted neurons, the host brain, and the actions and experiences of the graft recipients in the environments in which they recover.7,22,23 Decades of experimental research in the field of neural plasticity13,24 -26 has demonstrated that enriched environments and increased physical activity
facilitated neuronal and overall CNS development in neurologically intact individuals;
enhanced neural plasticity and behavioral recovery in animal models of HD, PD, AD, and stroke;
facilitated the growth and integration of grafted cells with the host brain; and
improved the functional efficacy of intracerebral transplantation in animal models.
There are multiple layers to the problem of using the above evidence for informing clinical research leading to the development of effective cellular therapies. The most obvious obstacle is the species-specific differences in brain anatomy, physiology, and behavior between animal models and people with brain disorders.13,27 In laboratory settings, we can operationalize and control for factors such as the nature and intensity of environmental stimulation and opportunities for increased activity, but questions arise regarding what constitutes a complex and stimulating physical and social environment for a person with an intracerebral graft. The meaning of events, the reasons for actions, and the experience of social and physical environments are culturally constructed and personally interpreted. These questions specific to human behavior are difficult to answer through data obtained in preclinical studies using animal models. 23 Regardless of these challenges, the failure to make use of the preclinical evidence for the potential benefits of activity and participation in clinical studies has been a major obstacle to creating viable cellular therapies. In contrast, evidence for biological processes, such as the optimal methods for the preparation and targeting of cells, has been successfully translated to clinical research.7,14
Open-Label Studies
The basic objective of stage 3 of the research program (Figure 1) is to transform the discoveries of the laboratory stages of cellular research into safe and effective clinical practices. The first exploratory phase of the clinical research program is aimed at determining the safety and feasibility of implanting the cells. In the second phase, uncontrolled time series, referred to as open-label, before-after designs are used to guide the preliminary stage of clinical research. 28 These designs involve a sequence of baseline assessment of symptoms and levels of functioning, followed by the surgical grafting of the cells and the subsequent evaluation of the participants’ progress according to explicit assessment protocols. 29 The results of these studies are essential for estimating the safety and efficacy of clinical interventions in comparison to the baseline. Moreover, they provide data on the optimal parameters that may influence treatment outcomes.14,15,28
Selecting relatively small groups of participants and conducting uncontrolled open-label investigations as a series over several years is justifiable on both methodological and ethical grounds. However, this method has created problems when using the results to make inferences regarding the general safety and efficacy of cellular therapies for PD.30-32 The inherent problem with inadequate sampling is that population estimates are based on data that are subject to large error margins, impeding power calculations and, therefore, the accuracy of identifying the sampling requirements for further studies.31,32
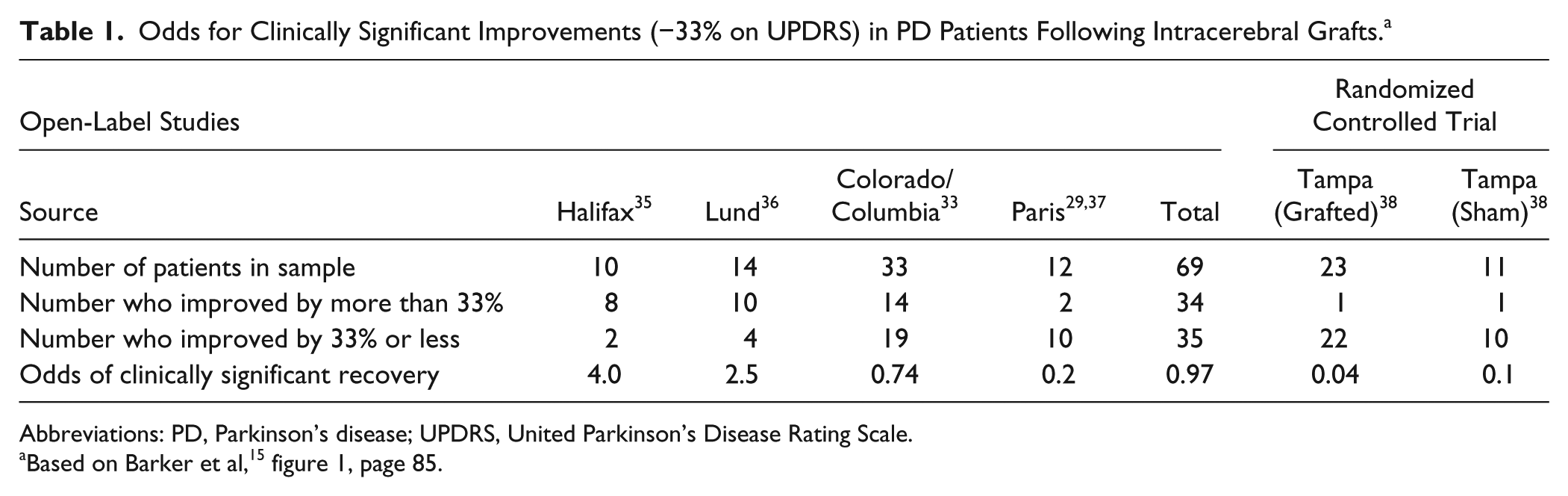
Owing to the inadequacy of the descriptive and inferential statistics reported in published research, previous attempts to synthesize evidence for the safety and efficacy of intracerebral grafts for PD created intractable problems for extracting the results and producing credible meta-analyses.30,31 Recently, Barker et al 15 collated and analyzed updated data from 5 research teams reporting on the efficacy of human embryonic cell transplantation for the treatment of PD. The data set consists of outcomes measured on the United Parkinson’s Disease Rating Scale (UPDRS), specifically the motor subscale under off-medication conditions, tabulated as the percentage change in patients’ scores before and after transplantation at the specified end points of each of the studies.
Overview of the database indicates that there are numerous differences between the studies regarding the treatment histories of the participants in each of the transplanted groups (figure 1 in Barker et al 15 ). For instance, 20 of the participants with neural grafts in the Colorado/Columbia study were initially enrolled in a double-blind RCT, 33 and following this, 14 of the participants in the sham operated control group also received neural grafts. Subsequently, 33 participants with neural grafts were reassessed approximately 2 years after completing the RCT under open, unblinded conditions. 34 Consequently, the percentage changes at the end points of the studies (as noted in the data set shown in Table 1) refer to data collected at different points in time following transplantation, which can influence the validity of comparisons between the transplanted groups.
Odds for Clinically Significant Improvements (−33% on UPDRS) in PD Patients Following Intracerebral Grafts. a
Abbreviations: PD, Parkinson’s disease; UPDRS, United Parkinson’s Disease Rating Scale.
Furthermore, the reported outcomes were influenced by differences between the groups on factors such as the age of the participants, the type and duration of the posttransplantation immunosuppression, and the quantity and location of the dopaminergic cells grafted within the basal ganglia. 15 The problem inherent in the available database is that there are too many interacting variables operating, making it difficult to apply methodologically acceptable statistical analyses to identify the effects of the multiple interacting factors that determine the therapeutic benefits of the intracerebral grafts. 15 At present, there are several research groups working collaboratively,10,11 with the aim of producing accurate evidence for correctly identifying optimal parameters for cellular therapies.
Even with the methodological limitations of the available database, we were able to estimate the overall clinical efficacy of intracerebral dopaminergic grafts for PD (Table 1). We used a percentage improvement of at least 33% or greater from baseline on the UPDRS motor subscale as a conservative indicator of a sufficiently large effect size for inferring clinically meaningful improvements. 29 The outcome of the secondary data analysis for estimating effect sizes is shown in Table 1, where we tabulated the number of patients in each sample, the percentage improved or not improved by 33%, and the odds of a clinically significant recovery.
In the 4 open-label studies, the total or combined odds of 0.97 indicates that 34 out of 69 participants with intracerebral grafts (49%) demonstrated meaningful improvements. Considering that PD is a progressive disorder, intractably leading to increasingly severe motor symptoms and disability, the total odds of 0.97 for clinically meaningful recovery from baseline indicates a very positive efficacy for grafting dopaminergic embryonic cells. This conclusion is consistent with the results of a previous meta-analysis (table 19 in Polgar et al30(p15)), which showed statistically and clinically significant effect sizes (Cohen’s d = 1.13; 95% CI = 1.04-1.22) on UPDRS (motor) outcomes. However, the above evidence does not imply that neural transplantation is a viable treatment option for PD, given that the highly variable results of efficacy provide insufficient evidence to establish reliable treatment guidelines required for conducting intracerebral transplantation (stage 6 in Figure 1). Furthermore, the elementary problem with relying on the results of open-label studies is that the improvements may not have been caused by the intervention but rather by bias and confounding factors resulting from the influence of uncontrolled extraneous variables such as placebo effects and observer bias. 39
Randomized Controlled Trials
In the context of evidence-based practice, randomized controlled trials (RCTs) are valued as gold standard designs for producing the most credible confirmatory evidence for the safety and efficacy of intervention (stage 4 in Figure 1). Depending on a number of considerations, such as the specific research question, the phenomenon being investigated, and the ethical necessity of protecting patients, researchers select the most appropriate control group(s). These may be as follows: no treatment, best available practice, or placebo control group(s). Double-blind placebo-controlled RCTs ensure the highest level of internal validity because neither the randomly allocated participants nor the assessors of the treatment outcomes are aware of the participants’ assignment to levels of the intervention. Sham surgery has been used to enable the implementation of double-blind surgical trials; this procedure is conducted in a way that imitates the actual cell transplantation.28,40,41
Initially, 2 double-blind RCTs33,38 for evaluating the safety and efficacy of reconstructive cellular therapies for PD using human embryonic cells were approved and funded in the United States by the National Institutes of Health. There were no meaningful improvements on the primary outcome measures at the designated end points, and an unacceptable proportion of the participants developed severe medication-independent dyskinesias approximately a year posttransplantation. 42 These negative findings clearly indicate (stage 6, Figure 1) that reconstructive cellular therapies are not ready to be introduced as a standard treatment for PD. 43 Moreover, concerns about the findings of the 2 RCTs resulted in a reduced number of studies being initiated while the causes of the weak efficacy and the harmful side effects were investigated.11,44,45
The question open to debate was whether the research program should continue in the light of the disappointing findings of the double-blind RCTs.1,11 We hold the view that the core concept driving the research program has not been falsified, but rather that a host of unresolved methodological difficulties and gaps in knowledge emerged during the translation process. The methodological problem relevant to the present discussion is illustrated by the evidence in Table 1, showing that whereas 34 of the total 69 (49%) participants with grafts experienced meaningful improvements in open-label studies, only 1 out of 23 (4%) recovered to this level in the Tampa/Columbia double-blind RCT. 38 The odds ratio is a commonly used statistic for estimating effect sizes for nominal scale data. For the combined open and the Tampa grafted groups, it is 21.37 (95% CI = 2.73-167.49; P = .0036) 46 ; that is, the combined outcome of the 4 grafted groups assessed under open conditions demonstrate approximately 21 times the odds for meaningful clinical recovery in comparison to the grafted group in the RCT. This result is statistically significant (P = .0036). Why is there such a large difference in the efficacy of the grafts under the 2 designs?
Alterman et al 32 compared the results of a series of published open-label and double-blind RCTs reporting on the efficacy of different types of cells used for the treatment of PD. They concluded that the primary explanation for the difference was the elimination of observer bias in assessing the outcomes for the participants of the RCTs. The argument for observer bias operating in uncontrolled open-label studies is justifiable. However, this is only one of the many arguable causes for the difference in treatment efficacy associated with the 2 designs, including numerous differences in the way in which the cells were prepared and the participants were selected. 47
It is relevant to note that there is evidence in the open-label studies (Table 1) for significant positive correlations between objectively determined graft development and the UPDRS motor outcomes, demonstrating association between the structural and functional benefits.15,30,48,49 Also, significant improvements have been reported posttransplantation on clinically relevant outcomes, such as improved medication benefits, as indicated by the reduced need for
Theory and Methods
The Composite Brain Model is based on the premise that the cells grafted to replace those lost through disease or injury do not restore impaired brains to their premorbid states, but rather create biologically and functionally novel neural systems. 23 Over time, the newly grafted cells gradually develop into mature neurons, interacting in multiple ways with the impaired neural networks, enhancing the neural plasticity of the host brain.7,10
Neural plasticity refers to the mechanisms, including the modification of synapses and the functional reorganization of neural networks, by which the brain responds to biological and environmental challenges.55,56 The intrastriatal transplantation of cells creates the potential for functional reorganization of the brain and the increased capacity of the graft recipients to interact with the physical and social environment. Accordingly, it has been suggested that the practice of neural reconstruction may be conceptualized as a 2-stage process7,13,16,22,23: first, the grafting of the immature cells into the host brain, and second, the provision of personal support and environmental stimulation for the graft recipients to ensure
the development and adaptive integration of the grafted cells with the host brain and
the optimal recovery of the patients in their social and physical environments by learning to make use of the graft (ie, rehabilitation).
The above model leads to an alternative explanation to that proposed by Alterman et al, 32 by taking into account the extent to which the open and double-blind research designs enable persons with intracerebral grafts to effectively engage with their physical and social environments. In contrast to open-label designs, the concealment of the intervention, inherent to double-blind designs, creates uncertainty in the participants regarding their allocation to sham or the active treatment. The consequence of masking the actual procedure is to limit the actions that may be undertaken by the graft recipients and their health care providers to ensure the most effective integration of the immature neurons with host neural networks. 54 We propose that one of the important causes for the diminished efficacy is the reduced opportunity for the participants in double-blind RCTs to learn how to make use of the newly grafted DA-rich cells for achieving optimal recovery.13,54
The results of decades of clinical research57,58 indicate that the best way to ensure optimal outcomes is to provide clear information about what to do during the recovery process and also to negotiate clear therapeutic goals. It is arguably advantageous, following the grafting of neurons in people with PD, to systematically enhance motivation, goal-directed activities, physical activity levels, and the reinforcement of improvements in functioning if and when they emerge. There is an obvious need to develop evidence-based movement rehabilitation programs targeted at integrating grafted cells with the circuitry of the host brain and systematically reinforcing the adaptive changes as they emerge over time.7,13,22,54
The methodology for research and evaluation for integrating rehabilitation with intracerebral transplantation is readily conceptualized in the framework of a biopsychosocial model. 23 This model, as formulated by Engel, 59 envisages health practice and research in the framework of hierarchically organized, dynamically interacting systems, where data are required for each of the biological, psychological, and social dimensions of illness and recovery. There is nothing novel about the suggestion of using biopsychosocial approaches in the area of cellular therapies for PD. For example, Swedish researchers 60 presented detailed narrative descriptions of the activities and social participation of the patients with intracerebral transplants. The biopsychosocial approach to practice and research is consistent with current views of disability 61 and the patient-centered position taken by health professionals in fields such as physiotherapy and neuropsychology.8,55,62
Further Research
The fundamental weakness of the above argument is that there is no credible, direct evidence for demonstrating that the actions of patients with neural grafts improve treatment efficacy. Furthermore, current initiatives for future research appear to focus on biological mechanisms such as cell sources 63 and optimal parameters for cell transplantation.11,64 Although these projects will produce crucial data for the advancement of cellular therapies for PD, 65 we suggest that additional evidence is needed to resolve the obstacles discussed in the present article.
The first obstacle to progress has been the failure to apply evidence for the benefits of activity and learning obtained in preclinical studies to the clinical stages of cell transplantation research. Important insights relevant to advancing cellular therapies may have been lost by not taking into account the relationship between the environment, activity, and the function integration of the transplanted cells. A useful method for making use of the preclinical evidence is to construct a general model for representing the mechanisms of posttransplantation neural plasticity in mammalian brains and use this model for generating clinically relevant hypotheses, which may lead to improved outcomes for cellular therapies.10,13,23
The second obstacle has been the lack of interest in research to understand the participant as a person with a disability. There are many questions that need to be investigated, such as the participants’ knowledge and expectations of the transplantation procedure, their optimism regarding the treatment outcomes, and the nature of the physical activities and social participation that they intend to undertake. 66 Understanding the psychological and social resources available to participants is relevant for devising ways of enhancing their capacity to make the best use of the transplanted cells. Therefore, exploratory, descriptive research is required, using mixed research designs that include the use of both quantitative and qualitative methods. 67 Mixed designs generate both subjective and objective data to provide evidence for a holistic overview of the experiences and actions of people recovering after intracerebral transplantation.
The third obstacle to the translation process is the absence of a systematic effort to devise, implement, and evaluate targeted rehabilitation programs for achieving optimal recovery. This lack of effort is surprising considering the depth and scope of the evidence regarding the importance of movement rehabilitation for people with basal ganglia disorders.8,9,62,68 The appropriate method to develop a targeted program is to modify existing evidence-based programs of movement rehabilitation for PD to articulate with emerging understanding of the process of neural reorganization following intracerebral transplantation.10,13,16,23 Preliminary research is required using multifactorial designs to identify the nature of the interactions between the transplanted cells and neurorehabilitation. Multifactorial designs are required to capture the interaction between time-dependent changes in neuronal activity following grafting and environmental changes, and the outcome variables as a measure of recovery.
Finally, the commitment to evaluating the efficacy of double-blind RCTs may be an obstacle to the advancement of cellular therapies. If rehabilitation is shown to be effective and emerges as an integral part of best-practice cellular therapies for PD, pragmatic rather than double-blind RCTs are required for evaluating clinical safety and efficacy. 54 Pragmatic trials aim to identify the comparative benefits of an intervention as administered under actual conditions, such that the implantation of cells cannot be concealed from participants or the therapists providing the targeted rehabilitation. Deep brain stimulation has emerged as best-practice symptomatic surgical treatment for suitable patients with advanced PD, 69 providing a clinically meaningful control group in future RCTs.
Summary
The problem addressed in the present article is that after more than 3 decades of research, the transplantation of DA expressing cells has not produced safe and effective interventions for PD. We proposed, on theoretical grounds, that in addition to transplanting the cells, the active involvement of the participants is required to ensure the functional integration of the cells into host neural networks.
The relevance of the participants’ actions and the social context in which they recover was illustrated by the difference in the recovery from baseline under open and double-blind conditions. Best available evidence indicated very promising levels of recovery under open conditions, in contrast to the poor benefits reported in double-blind RCTs. This difference was interpreted as demonstrating that masking the transplantation of the cells, under the double-blind protocol, minimized the potential contributions of the participants to their recovery.
However, there are a multitude of factors that interact to determine the outcomes of intracerebral transplantation. Therefore, the above evidence is merely consistent with, but in no way proof of the importance of, the participant’s role in the effective development of reconstructive cellular therapies for PD. Clearly, there is a need for direct evidence, acquired by methodologically rigorous means, to discover the extent to which the experiences and actions of the participants contribute to recovery. In particular, research was called for developing targeted rehabilitation for persons with neural grafts. This research will need to rely on methods that can integrate knowledge of the biology of the CNS following the transplantations with techniques used to enhance activity and participation of people with PD.
Footnotes
Acknowledgements
We would like to acknowledge the contribution of Michele Bernshaw and Fathimath (Sheeza) Mohamed for editing and proofreading the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
