Abstract
Introduction
Parkinson’s disease (PD) is one of the most common neurological diseases. 1 Although pharmacological treatment has changed its evolution, PD still leads to major disability that impairs the quality of life of patients. 2 The characteristics of the disease, the long life span after diagnosis, the short duration of the effectiveness of drugs, and the frequent adverse effects related to drugs prompt the need for additional therapeutic interventions to improve motor performance. The acute positive effects of different rehabilitation treatments are established, but whether the effects of the rehabilitation persist over time and what the optimal content of an intervention is (type of exercises and intensity and duration of treatment) remain open questions.3-6
Treadmill 7 and visual and auditory cues improved gait in PD patients. 8 A recent study showed that balance training improves postural stability and the level of confidence perceived while performing daily activities that require balance and reduces the frequency of falls. 9 Several studies suggest that exercise duration and intensity might play a role in the effectiveness of rehabilitation.10-15 We thus developed a multidisciplinary intensive rehabilitation treatment (IRT) in which standard physical therapy techniques were combined with treadmill with auditory and visual cues (treadmill plus) and a stabilometric platform.16,17
The present study was devised (
Methods
Study Population
This was a random-assignment, parallel-group, pilot study with a 1:1 allocation ratio. Randomization was based on a computer-generated list. Eligibility criteria for patients were diagnosis of “clinically probable” idiopathic PD according to Gelb et al, 18 Hoehn–Yahr stage 3, 19 ability to walk without physical assistance, no cognitive impairment (mini-mental state examination score ≥ 26 20 ), no comorbidity, no vestibular/visual dysfunction limiting locomotion or balance, and anti-Parkinsonian medications stable for >4 weeks.
PD patients were screened by a neurologist specialized in movement disorders and eligible patients were invited, according to the randomization list, either to be admitted to the Rehabilitation Institute of Montescano for a 4-week treatment to be repeated 1 year later or to follow usual care. The study was approved by the local ethics committee, and all participants gave their informed written consent.
Assessment of Outcome
The primary outcome measure was the Unified Parkinson’s Disease Rating Scale. 21 Sections II and III (UPDRS II and III) and total score (UPDRS tot) were used. UPDRS II is an ADL scale (0-52), whereas UPDRS III is a scale of motor performance (0-56); lower scores correspond to a better clinical condition. UPDRS tot summed up the scores of all sections: I, II, III, and IV. The dosage of levodopa equivalent was considered as the secondary outcome measure.
Patients in group 1 were evaluated at the beginning and end of the first and second cycle of IRT. Those in group 2 were assessed at the beginning of the study and at 12 months. All patients were examined by a neurologist, who was blinded to treatment allocation and to the study design, in the morning, 1 hour after the first dose of levodopa. The dosage of levodopa equivalent was recorded at the beginning of the study, after 12 months, and from the patients’ clinical reports, 1 year before enrollment.
Sample Size Computation
From published studies, we found that the standard error of measurement is 1.4, 4, and 4.7 for UPDRS II, UPDRS III, and UPDRS tot, respectively. We expected an effect size of around 2, 5.2, and 9.1 (moderate clinically important difference according to Shulman and colleagues)
22
for UPDRS II, UPDRS III, and UPDRS tot, respectively. Hence to detect a change with a 2-tailed type I error of 0.05 and a power of 80%, the estimated sample size (the largest between the 3 estimates) was 20 patients for each treatment group (Student’s
Intervention
Patients allocated to group 1 were admitted to the Rehabilitation Institute of Montescano (Pavia), Italy, where they underwent IRT, a 4-week cycle of physiotherapy that entailed 3 daily sessions (2 in the morning and 1 in the afternoon, each for 1 hour), 5 days a week. The first session comprised cardiovascular warm-up activities; relaxation exercises; muscle stretching (scapular, hip flexor, hamstring, and gastrocnemius muscles); exercises to improve the range of motion of spinal, pelvic, and scapular joints; and exercises to improve the functionality of abdominal muscles and postural changes in the supine position. 23 The second session included exercises to improve balance and gait using a stabilometric platform with a visual cue and treadmill training associated with auditory and visual cues (treadmill plus). Briefly, using the stabilometric platform, the patients were asked to follow a circular pathway on the screen by means of a cursor sensitive to the movements made by their feet on the platform. For treadmill training, the visual cue was a target, displayed on a screen, that the patient had to reach within a stride, whereas the auditory cue consisted of musical beats, synchronized with the visual cues, with a frequency of 0.5 c/s. 16 The last session was occupational therapy to improve autonomy in daily living activities: transfers from sitting to standing, rolling from supine to sitting and from sitting to supine, dressing, use of tools, and exercises to improve hand functionality and skills (eg, using screws and bolts). At discharge, the patients were instructed to continue a set of learned exercises in order to maintain functionality of the spine and scapular and pelvic joints, along with the instruction to walk at least 30 minutes a day.
Patients allocated to group 2 underwent only usual care (pharmacological treatment) and were instructed by a neurologist during the first visit to practice generic physical exercises at home and to walk.
Statistical Analysis
Descriptive statistics are given as mean ± standard deviation. The Shapiro–Wilk statistic was used to test the normality of the distribution of all variables.
To investigate the primary end point, we tested for a difference in the time course of UPDRS scores in treated patients compared with control patients over a year. A 2-factor analysis of variance was carried out: the first factor was treatment (rehabilitation vs control group) and the second factor was time (first admission and second admission 1 year later for treated patients, and first visit and second visit 1 year later for controls), with repeated measurements in the time factor.
To compare the effect sizes of the first and second interventions in treated patients (secondary end point), within-group comparisons of the changes in UPDRS scores (T1-T0 vs T3-T2) were carried out by paired
Finally, to assess the time course of levodopa dosage in treated and control patients, a 2-factor analysis of variance was carried out: the first factor was treatment (rehabilitation vs control group), and the second factor was time (first admission and second admission 1 year later for treated patients, and first visit and second visit 1 year later for controls), with repeated measurements in the time factor.
A
Results
Figure 1 describes the progress through the phases of the study. Of 68 eligible patients approached, 18 refused to participate in the study. There were no dropouts, leading to a final population of 25 + 25 patients. Figure 2 shows the relevant observation times of the study. Demographic, clinical, and functional characteristics of studied patients at observation times T0, T1, T2, and T3 are reported in Table 1. The time between T2 and T0 was 12.1 ± 1.7 months.
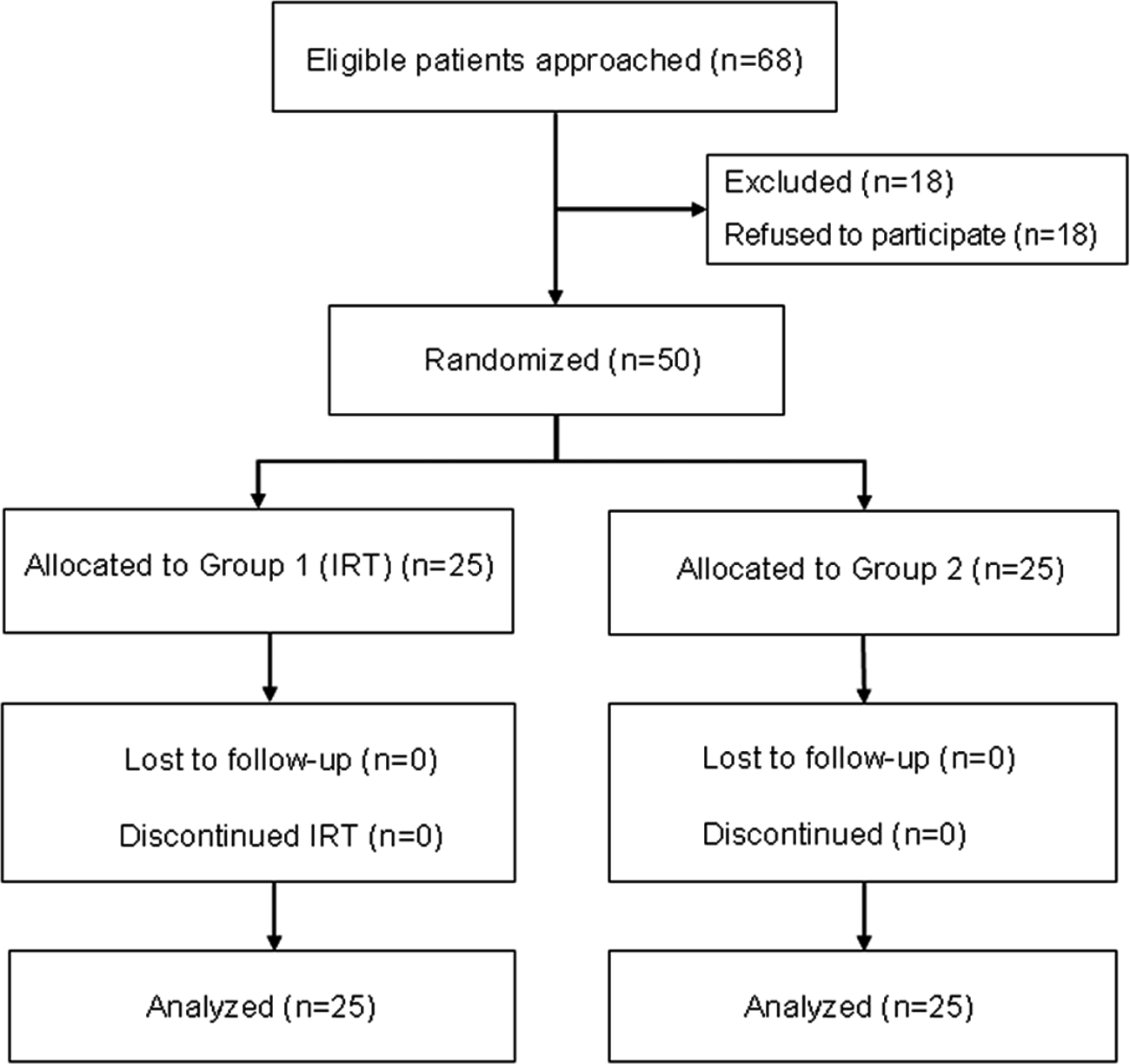
Flow diagram of the progress through the phases of the study: of 68 eligible patients approached, 18 refused to participate in the study. There were no dropouts, leading to a final population of 25 + 25 patients
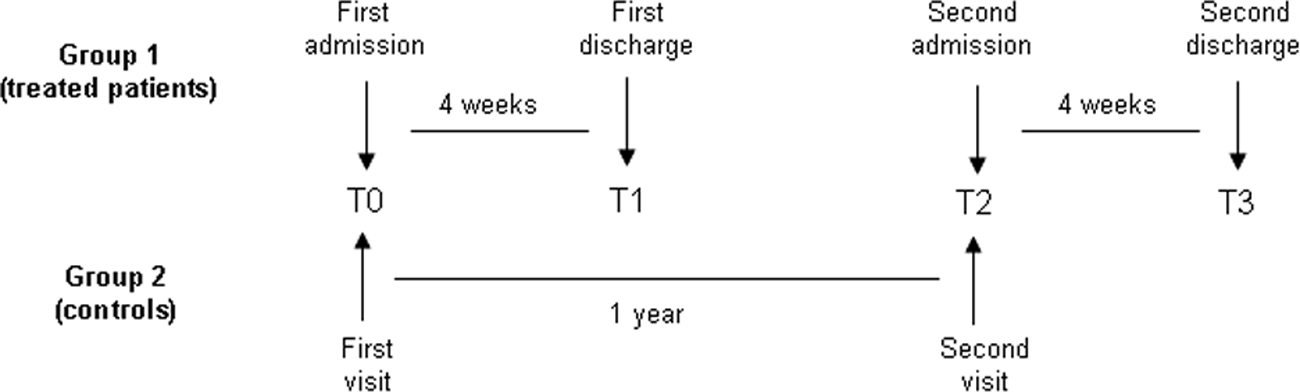
Relevant observation times: T0, time of first visit/admission; T1, time of first discharge for treated patients (4 weeks after T0); T2, time of second visit/admission (1 year after T0); T3, time of second discharge for treated patients (4 weeks after T2)
Demographic, Clinical, and Functional Characteristics of the Patients a
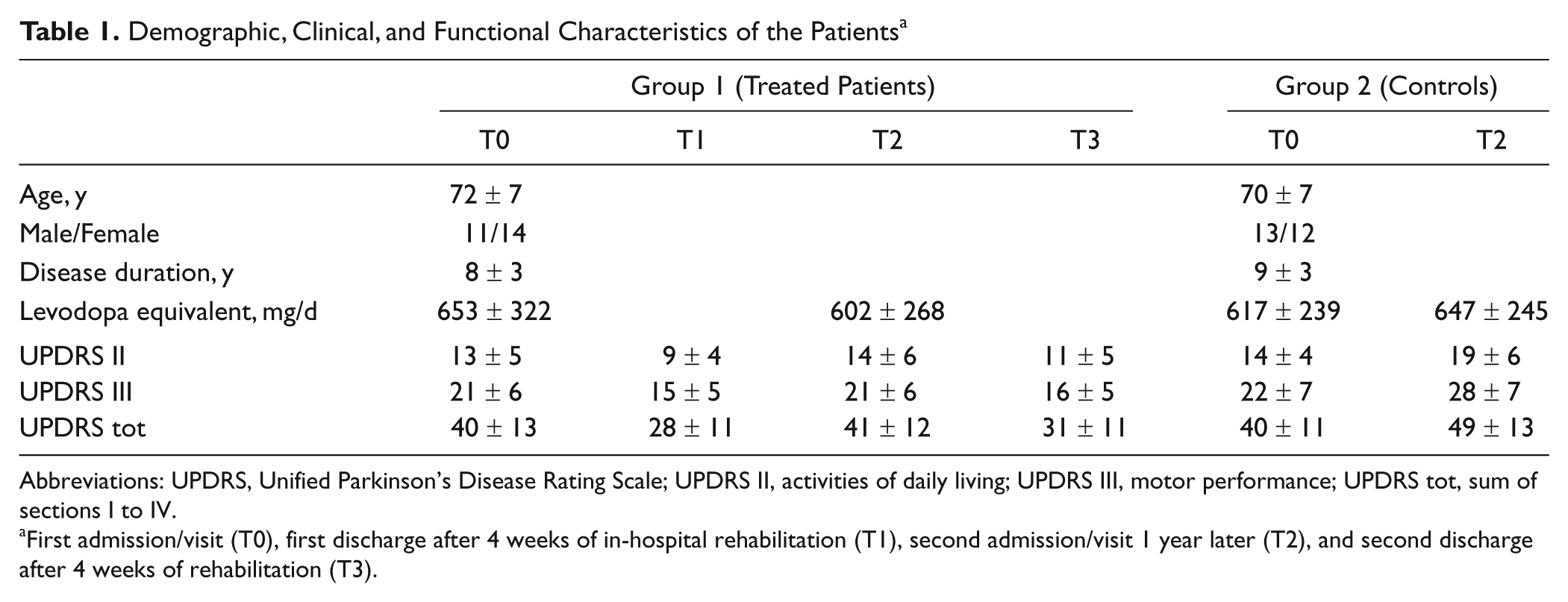
Abbreviations: UPDRS, Unified Parkinson’s Disease Rating Scale; UPDRS II, activities of daily living; UPDRS III, motor performance; UPDRS tot, sum of sections I to IV.
First admission/visit (T0), first discharge after 4 weeks of in-hospital rehabilitation (T1), second admission/visit 1 year later (T2), and second discharge after 4 weeks of rehabilitation (T3).
We found a highly significant time–treatment interaction in the analysis of variance for all UPDRS scores (Time × Group:
Even though no variable markedly violated the normality assumption, all results were checked using a nonparametric Wilcoxon’s matched pairs test, and superimposable results were obtained.
Considering the effects of the first and second intervention (T1 to T0 and T3 to T2) in treated patients (secondary end point), both cycles of IRT improved UPDRS scores. In particular, UPDRS II was reduced by 4.2 (
In regard to drug intake, again we found a significant time–treatment interaction in the analysis of variance (Time × Group:
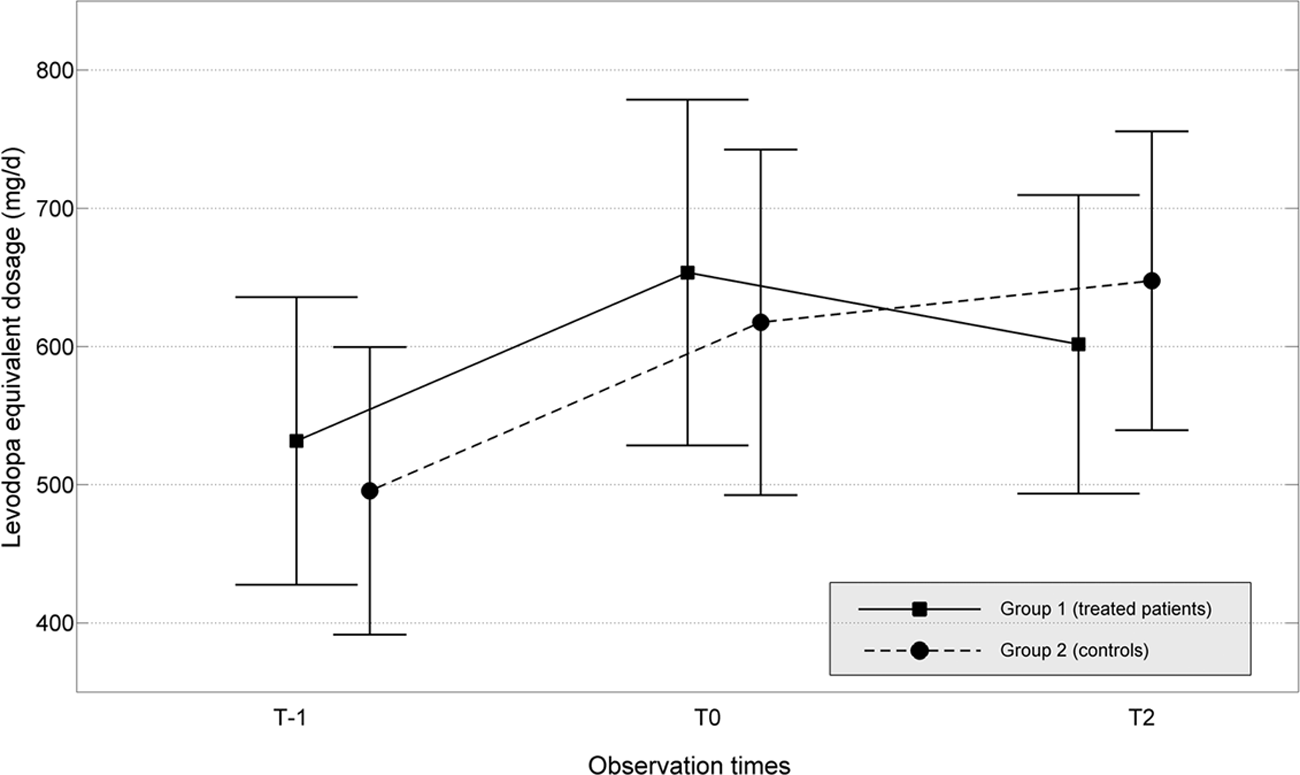
Dosage of levodopa equivalent in treated (solid line) and control patients (dashed line) at the time of first visit/admission (T0), at the time of second visit/admission (T2), and from the patients’ clinical reports, 1 year before enrollment (T-1). Data are plotted as mean ± 95% confidence interval
Discussion
We found a significant difference between treated patients and control patients in the changes over time of UPDRS scores. In patients who underwent IRT, UPDRS scores at the end of the 1-year follow-up period were very similar to the values at first admission, whereas in control patients, the values were significantly increased by a clinically important amount, 22 indicating that the effects of IRT do persist over time, in contrast to expectations of the natural worsening of symptoms associated with the degenerative nature of PD.
We also found that a second rehabilitation cycle administered after 1 year was as effective as the first treatment and that both cycles determined a clinically important improvement in UPDRS scores. Finally, we found that daily medication dosage was reduced in treated patients, whereas it was significantly increased in control patients.
Published studies support the time dependence of the effects of rehabilitation for PD to improve motor performance, but the follow-up has been brief and benefits have declined over time after discharge.24-28 On the basis of previous studies, 29 Herman and colleagues 30 developed an intensive treadmill program that improved gait parameters in PD patients, and other studies demonstrated the efficacy of intensive treadmill training and sensory cues.7,16,31 Ellis and colleagues 14 showed the efficacy of an inpatient rehabilitation program, even though there was a confounding effect caused by drug therapy optimization during hospitalization. Morris and colleagues 32 reported that an intensive 2-week rehabilitation treatment reduced disability, but at 3-month follow-up, some regression in the performance of patients had occurred. The authors hypothesized that longer treatment might produce better results. The improvement in UPDRS motor score observed in our study was similar to that reported by Herman et al 30 and higher than that reported by Morris et al. 32 However, because of the very low level of disability of the patients in the study by Morris et al, no direct comparison can be made.
Taken together, these findings suggest that intensity, duration, type of exercises, and use of cues are key factors for short- and long-term effectiveness of a rehabilitation treatment. Differences in these factors might explain the varied results. Our study is the first to show the benefits of inpatient rehabilitation and a second course of therapy 1 year later.
Another novel finding was that the daily dosage of levodopa at follow-up was reduced in treated patients and increased in control patients. This result was strengthened by the observation that during the year preceding enrollment in the study, no difference in changes in drug dosage was observed for the 2 groups. To explain this finding, we hypothesize that repetitive exercise improves synthesis and release of endogenous dopamine in the basal ganglia.33-35 A recent study showed that despite optimal treatment, the UPDRS III score worsened after 1 year in 25.6% of PD patients, and the levodopa dose had to be increased in 51.6%. 36
Feasibility
The rationale to develop our IRT derived from studies demonstrating the importance of a properly designed protocol in terms of intensity, duration, and type of exercises. Our protocol of 15 hours per week for 4 weeks was feasible in our institution because usual clinical practice and the compliance of patients was very high. In published studies on PD patients, the hours of treatment per week vary from 2 to 21.14,15,30,32 Our protocol should be feasible in other health care systems. However, because we did not make a cost-effectiveness analysis, further studies must address this issue.
Study Limitations
Patients could not be blinded to the treatment, allowing for a possible placebo effect.37-39 The blinded evaluator and persistence of beneficial effects at 1-year follow-up support a treatment effect. Another possible limitation was the lack of data on the adherence of patients to the instructions to practice physical activity during the year spent at home. In addition, we considered only parts of the UPDRS score as measurements of outcome. Even though this might constitute a limitation, the UPDRS is judged as an important measurement of outcome that reflects the severity of PD.
Conclusions
Our findings suggest that the natural worsening of symptoms associated with PD and the parallel increase in drug dosage can be countered by a properly designed rehabilitative treatment. Adding periodic cycles of intensive rehabilitation to the pharmacological treatment should be considered to improve motor performance and autonomy in daily activity as well as to delay the increase in drug dosage to avoid possible side effects. Further studies could assess whether rehabilitation administered at early stages of PD can reduce the progression of symptoms.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
