Abstract
Background. Exercise may decrease the risk of Parkinson’s disease (PD) in humans and reduce PD symptoms in animal models. The beneficial effects have been linked to increased levels of neurotrophic factors. Objective. We examined whether intensive rehabilitation treatment reduces motor disability in patients in the early stages of PD and increases brain-derived neurotrophic factor (BDNF) serum levels. Methods. Thirty participants in the early stages of PD treated with rasagiline were randomly assigned to 3 hours of rehabilitation treatment that included aerobic exercise for 28 days (Group 1) or to not therapy (control; Group 2). BDNF serum levels were assessed at time T0 (baseline, before treatment), T1 (10 days), T2 (20 days), and T3 (28 days). At T0 and T3, we assessed the Unified Parkinson’s Disease Rating Scale (UPDRS) III in both groups, as well as the UPDRS II and total, Berg Balance Scale, and 6-minute walking test only in Group 1. Results. BDNF levels significantly increased at T1 in Group 1, an increase that was maintained throughout the treatment period. At T3 compared to T0, UPDRS III scores significantly improved in Group 1 along with scores for UPDRS II, total, Berg Balance Scale, and 6-minute walking test. Conclusions. Intensive rehabilitation treatment increases the BDNF levels and improves PD signs in patients in the early stages of the disease. These results are in line with studies on animal models of PD and healthy subjects.
Introduction
Parkinson’s disease (PD) is a neurodegenerative disorder that is imposing progressively heavier burdens on Western countries, as its incidence is 160 new cases per 100 000 person/year with a prevalence of 950 patients per 100 000 in the population over 65 years. 1 The majority of drugs used to treat PD-related disabilities acts on the dopaminergic nigrostriatal system. However, the impairment of balance and gait, one of the most prominent disabilities of PD, may not respond to dopaminergic treatment, as it probably stems from dysfunction of other neurotransmitter systems.2,3 These symptoms likely represent the onset of a vicious circle that leads to a sedentary lifestyle and a decrease of physical fitness, which further worsens balance and gait as well as the patients’ autonomy. 4
The onset of balance and gait problems might be prevented by maintaining physical activity. Indeed, in the last decade, a considerable number of reports have shown that exercise is effective in improving gait, balance, and general motor performance in PD.5,6 In addition, studies have highlighted that (a) moderate to vigorous activities decrease the risk of PD and (b) intensive exercise leads to optimal results in PD rehabilitation.7-10 Finally, intensive exercise in animal models of PD has been shown to ameliorate performance possibly by promoting cell proliferation and neuronal differentiation.11,12 The results of animal studies also suggest that these neuroplastic effects are probably related to increased expression of a variety of neurotrophic factors, in particular, brain-derived neurotrophic factor (BDNF).13,14 Interestingly, in normal human subjects, serum BDNF levels increase after physical exercise, and this increase is proportional to exercise intensity.15,16
So far, only 2 studies have measured BDNF serum levels in PD patients. In one of them, 17 average BDNF serum levels were lower in a group of PD patients (47 patients, Hoehn & Yahr stage from 1 to 4) as compared to a control group of 23 age-matched healthy subjects. In the other study, 18 the BDNF serum levels in 21 PD patients (Hoehn & Yahr stage from 1.5 to 4) were not compared to a control group but were positively correlated to striatal dopamine transporter availability, suggesting that BDNF levels might decrease along with loss in striatal DAT binding.
The aim of this study was to assess whether intensive rehabilitation treatment reduces motor disability and increases BDNF serum levels in patients in the early stages of PD.
Methods
In this randomized study, inclusion criteria were the following: a diagnosis of “clinically probable” idiopathic PD according to Gelb et al, 19 Hoehn & Yahr stage 1-1.5, no other neurological condition, rasagiline monotherapy for at least 8 weeks, Mini-Mental State Examination score greater than 26, visual and hearing function sufficient to perceive cues, and ability to walk without any physical assistance. Exclusion criteria were the following: postural hypotension, cardiovascular disorders, musculoskeletal disorders, vestibular dysfunctions limiting locomotion or balance, and depression (Hamilton DRS >8). We chose to evaluate patients who were all treated with the same drug (rasagiline) in order to avoid possible influences of different pharmacological classes on both motor performance and BDNF levels.
Twenty-five eligible patients, admitted to the movement disorder ambulatories of the Rehabilitation Institute of Montescano (Italy) from January to December 2012, agreed to participate in the study and were randomized to 2 groups: the treatment group (Group 1, N = 15), which underwent IRT, or the control group (Group 2, N = 10), which did not receive rehabilitation. Patients of both groups continued on rasagiline. For the allocation of the participants, a list of random numbers was generated using Matlab random numbers generator (MATLAB R2012a; MathWorks Inc, Natick, MA). The sequence was concealed until assignment, and the people enrolling participants did not know in advance which treatment the next patient was assigned.
The primary outcome measures were serum BDNF concentration and the motor score of Unified Parkinson’s Disease Rating Scale (UPDRS, section III).
20
BDNF serum levels were assessed in both groups, at baseline (T0) and at 10 (T1), 20 (T2), and 28 (T3) days after study onset. All blood samples were drawn at 7
Neurological examinations were performed at baseline (T0) and after 28 days of treatment (T3) at 10
Intervention
We defined our intervention as “intense” on the basis of the total time spent doing it. In fact, IRT consisted of a 4-week inpatients cycle of physiotherapy that entailed 3 daily sessions (2 sessions, not consecutive in the morning, and 1 session in the afternoon), for 5 days a week. The total duration of each session, including recovery periods, was about 1 hour, for a total of 3 hours per day. The first session comprised cardiovascular warm-up activities; relaxation exercises; muscle-stretching exercises (scapular muscle group, hip flexor, hamstring, and gastrocnemius muscles); exercises to improve the range of motion of spinal, pelvic, and scapular joints; exercises to improve the functionality of the abdominal muscles; and postural changes in the supine position. The second session included exercises to improve balance and gait using a stabilometric platform with a visual cue (patients were asked to follow a circular pathway on the screen by using a cursor sensitive to their feet movements on the platform) and treadmill plus (treadmill training with both a visual and an auditory cue). 22 All treadmill therapy was aerobic with a heart rate reserve ≤60% and a maximum speed of treadmill scrolling of 3.5 km/h. Subjects received training for 30 minutes every day for 4 weeks (20 sessions in all). The patients were observed during treadmill training by a physiatrist. Maximum tolerated walking speed was determined before the training session. This speed was reduced (−40%) and used for a 2-day warm-up period. After that, the belt speed was increased every 3 days by 0.05 stride cycles/second. The third was a session of occupational therapy aimed at improving autonomy in daily living activities: transferring from sitting to standing position, rolling from supine to sitting position and from sitting to supine, dressing, use of tools, and exercises to improve hand functionality and skills (eg, using screws and bolts).
Statistical Analysis
Since there are no studies estimating the standard error of measurement (SEM) for BDNF serum levels in patients with PD, we could not compute sample size according to this primary outcome. Hence, we based the study on the scores of UPDRS III. The minimum difference we wished to detect between treated and control groups was 6.7 (ie, moderate clinically important difference). 23 As previous studies have shown that the SEM 24 of UPDRS III is 4, for detecting the predefined difference with a 2-tailed type I error of 0.05 and a power of 80%, the sample size was equal to 11 patients per group.
We determined the effect of intensive rehabilitation treatment on serum concentration of BDNF and UPDRS III with a 2-factor ANOVA, where the first factor was treatment (2 groups, treatment vs control groups) and the second factor was time (4 measurements, T0, T1, T2, and T3, for BDNF, and 2 measurements, T0 and T3, for UPDRS III), with repeated measurements in the time factor. To quantify the effectiveness of the treatment, the effect size (standardized mean difference between the 2 groups) of changes in BDNF and UPDRS III was estimated. The effects of treatment on UPDRS, BBS, and 6MWT scores were assessed with paired t test or, in case of violation of the normality assumption (tested by the Shapiro–Wilk statistics), with Wilcoxon’s matched pairs test. We also assessed correlations between changes in BDNF (difference T3 − T0) and changes in behavioral scores with the Spearman R coefficient. A P value <.05 was considered statistically significant. The results of descriptive statistics are reported as mean ± standard deviation. All analyses were carried out using the SAS/STAT statistical package, release 9.2 (SAS Institute Inc, Cary, NC).
Results
Figure 1 reports the flow chart of the study. One patient in Group 1 discontinued treatment because of urinary tract infection, while all 10 patients in Group 2 completed the study. Thus, our analyses were performed on a total of 14 treated patients (Group 1) and 10 control patients (Group 2). Patients of these 2 groups had similar age (67 ± 5 vs 65 ± 4 years, P = .15), disease duration (8 ± 5 vs 8 ± 2 months, P = .34), and UPDRS III scores (16.4 ± 3.5 vs 15.6 ± 1.5, P = .53).
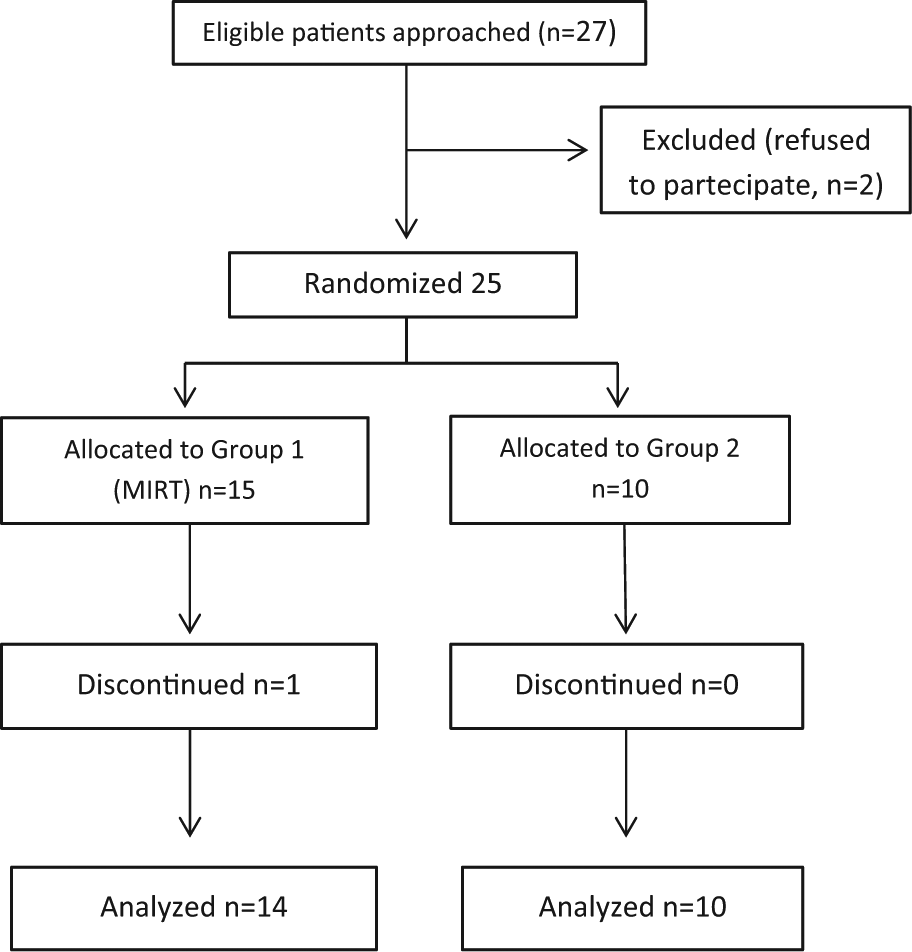
Flow diagram of the progress through the study.
Values of BDNF at observation times for both groups of patients are reported in Table 1, together with the motor score of UPDRS scale. The main finding was the different time course of BDNF serum levels in treated patients with respect to controls. While BDNF serum levels were stable over time in the control group (Figure 2, Group 2: white), they increased after 10 days in the treated group (Figure 2, Group 1: black). This between-group difference was confirmed by a significant time by treatment interaction in the repeated measurements analysis of variance, F(3, 66) = 5.63, P = .0017. Post hoc analysis showed that BDNF serum levels were significantly increased in Group 1 patients after 10 days (P < .001), and subsequently remained higher, whereas they were unchanged in Group 2 patients throughout the 4-week period (P > .5). The effect size of the changes in BDNF was 1.1. Subject-by-subject analysis of the changes in BDNF between baseline (T0) and final (T3) levels revealed that, while BDNF levels remained unchanged (±5% variation) only in 2 out of 14 patients, with 7 showing an increase between 5% and 15% and 5 an increase greater than 15%, all patients in Group 2 maintained BDNF serum levels substantially unchanged (within ±4% change).
BDNF Serum Concentration and UPDRS III at Considered Observation Times in Treated Patients (Group 1) and in Controls (Group2).
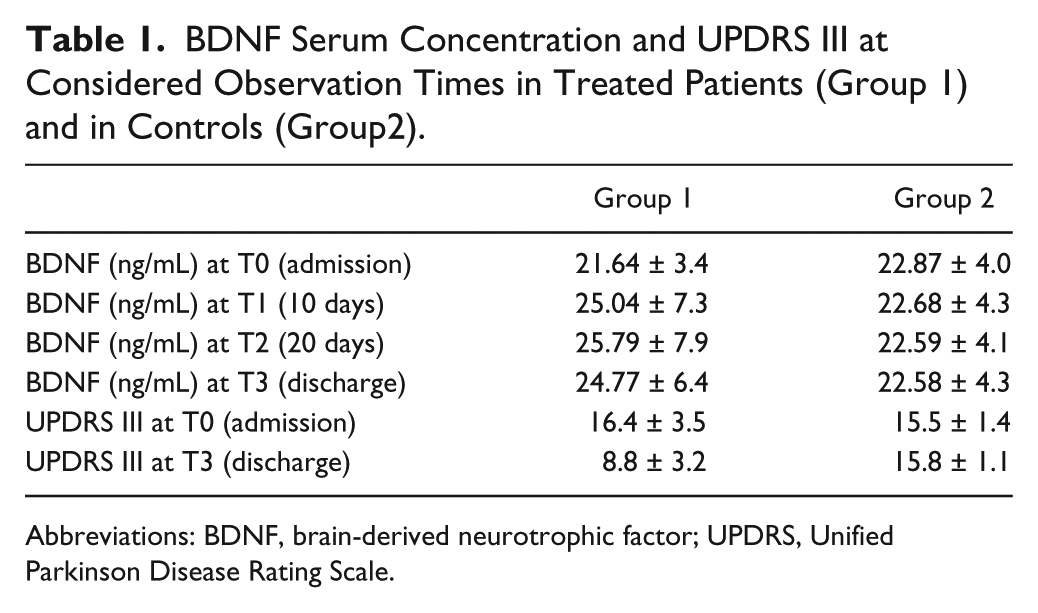
Abbreviations: BDNF, brain-derived neurotrophic factor; UPDRS, Unified Parkinson Disease Rating Scale.
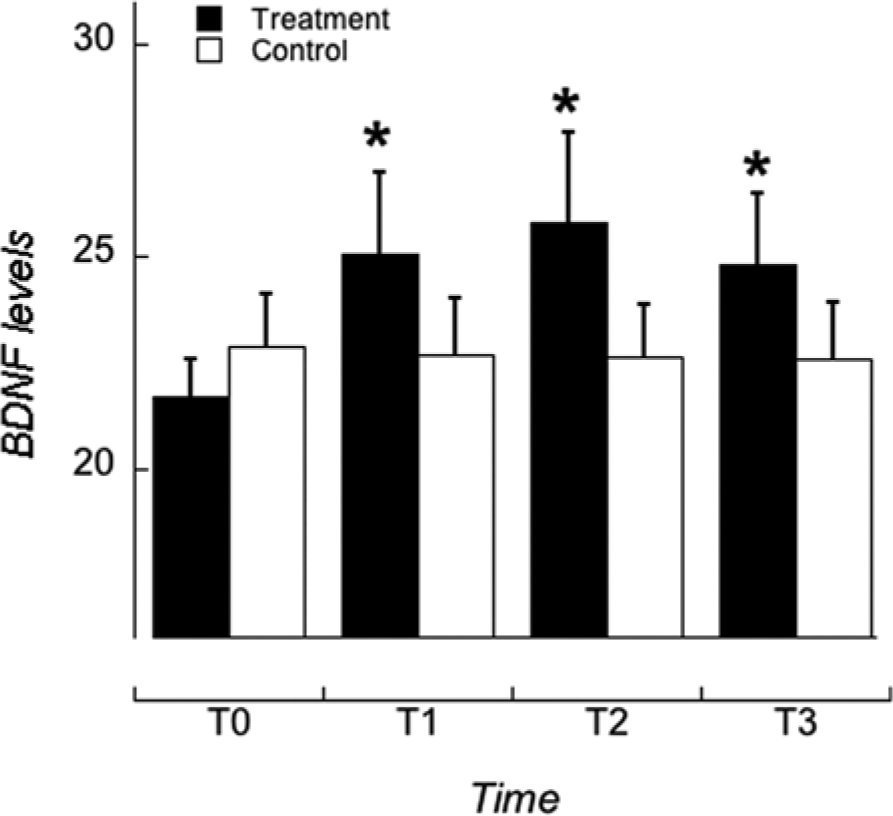
Time course of brain-derived neurotrophic factor (BDNF) serum levels.
For motor responses, a between-groups difference was observed in the time course of UPDRS III score, as assessed by a significant time by treatment interaction, F(3, 66) = 66.5, P < .0001. While the motor performance of patients in Group 1 improved at the end of treatment (P < .0001), it was unchanged in the control group (P = .1934). The effect size of the changes in UPDRS III was −3.3. All functional variables assessed only in Group 1 (UPDRS II score, BBS, and 6MWT) significantly improved at T3 as compared to T0 (Table 2). Finally, no significant correlation between changes (T3 − T0) in BDNF levels and changes in UPDRS III, UPDRS II, UPDRS total, BBS, and 6MWT was observed in Group 1 patients (r = −0.13, P = .65, r = −0.25, P = .38, r = −0.18, P = .52, r = −0.11, P = .69, r = 0.05, P = .88, respectively).
Functional Variables at Enrolment and After Rehabilitation Treatment in Treated (Group 1) Patients.
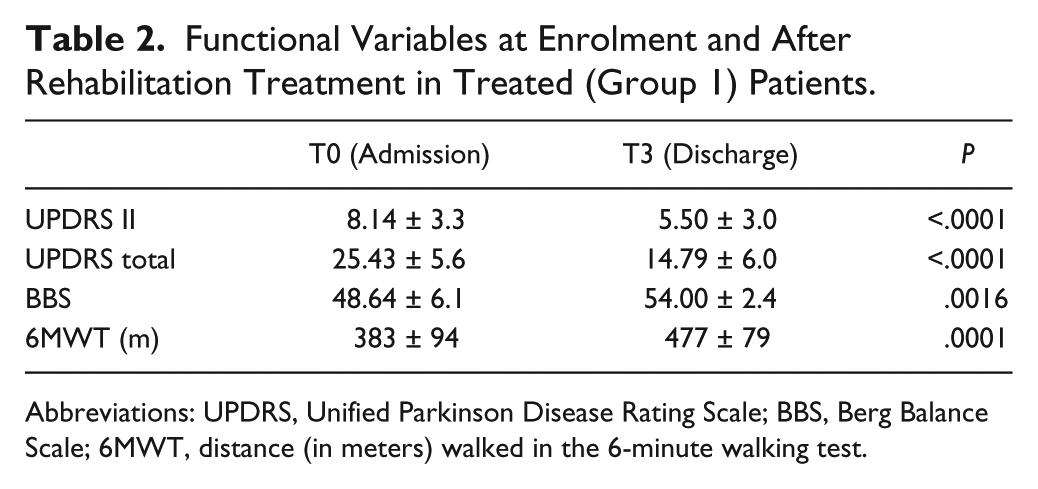
Abbreviations: UPDRS, Unified Parkinson Disease Rating Scale; BBS, Berg Balance Scale; 6MWT, distance (in meters) walked in the 6-minute walking test.
Discussion
This study shows that following IRT, patients in the early stages of PD may improve in several aspects of motor performance including balance and gait. This improvement was accompanied by significant increases in BDNF serum levels, which persisted throughout the treatment period. The average increase in BDNF serum levels in our patients was of 14%, a result similar to the one obtained in older adults (11%) after long-term aerobic exercise training. 25 Conversely, a control group of patients with similar degree of disability receiving only rasagiline treatment did not show any significant change in either the BDNF levels or UPDRS motor scores over the same 28-day period.
As far as baseline values of BDNF are concerned, the only studies reporting values of BDNF serum levels in PD patients are those by Scalzo et al 17 and Ziebell et al. 18 The mean values reported were 40.6 ng/mL and 32.9 ng/mL computed on 47 and 21 patients, respectively. These values are somewhat higher than those observed in our population. However, since Scalzo and coauthors report a very strong correlation between BDNF values and the disease duration (r = 0.526, P < .0001), we argue that this difference might be due to the very large difference of disease duration in the populations studied (7.6 years in the study from Scalzo, 29 months in the study from Ziebell, and 8 months in our study).
In summary, the results of this pilot study indicate that intense exercise might promote motor improvement and increase in BDNF serum levels. There are, however, some caveats. The groups tested were relatively small, and the durability of these findings is not yet known. The changes in BDNF serum levels and motor scores in Group 1 patients were not significantly correlated. This result might be simply due to the small sample size or to the reduced range of variability of the motor scores. However, there is also the possibility that the effects of exercise on BDNF and motor scores could have been independent from each other. Another issue that was not addressed in this study is the origin of the BDNF increases. It is unlikely that this increase resulted from the activation of muscles, as the increase of serum BDNF in normal humans following exercise seems to be independent from that produced by muscles. 26 Finally, before advocating such an intensive exercise program for the general PD population, its efficacy needs to be confirmed, its feasibility verified, and its costs defined.
Intensive exercise can improve motor performance in Parkinsonian patients5,27,28 and in animal models of PD. Although the research on PD animal models is not always directly applicable to clinical practice, 29 several studies have shown that in 6-hydroxy-dopamine (6-OHDA) or 1-methyl,4-phenyl,1,2,3,6-tetrahydropyridine (MPTP) animal models, an intensive use of a treadmill or a running wheel not only stopped the worsening of Parkinsonian signs but actually improved motor performance.29,30 These motor changes were associated with improvement of different aspects of dopaminergic and glutamatergic transmission.30-36 The net effect is a decrease of synaptic strength and relief from overactivation in basal ganglia circuits. 37
The neuroplastic effects of exercise have been related to increased expression of a variety of neurotrophic factors,13,14 in particular, BDNF and glial-derived neurotrophic factor (GDNF). Indeed, treadmill exercise induced striatal upregulation of BDNF and GDNF in rat models of PD 31 and increased the level of endogenous BDNF and GDNF in the substantia nigra and striatum. 38 BDNF, a key component in neurogenesis, synaptogenesis, and cell survival, 39 exerts protective effects against MPTP and 6-OHDA40,41 and increases striatal DA levels and tyrosine hydroxylase activity.31,42
Several studies tested the effect of training on BDNF in healthy people and showed that in the elderly, physical exercise, especially of moderate intensity, can increase the levels of peripheral BDNF.25,43 However, many aspects of the interactions between exercise and BDNF in humans are not clear. For instance, aerobic exercise and stretching may have similar effects on BDNF levels and there may be different responses between genders. Importantly, the reported BDNF increase in normal elderly was similar to our present findings in PD patients. 44 These results provide a further link between BDNF and the effectiveness of intensive rehabilitation treatment in the early stage of PD, and as suggested by our earlier studies with patients in Hoehn & Yahr Stage 3, also in the advanced stages of the disease. 45 Since BDNF can cross the blood–brain barrier in both directions, 46 the increase of circulating BDNF may represent a relevant reserve for brain cells and promote a neuroplastic effect in PD, at least in the early stages of the disease. Nevertheless, intensive exercise programs with the use of treadmill and other devices might represent an important and novel approach for the treatment of PD, especially for impairment of balance and gait, problems that do not respond well to conventional pharmacological treatments.
Finally, the results of this study raise questions about the specific mechanisms involved and their clinical relevance, in particular, whether BDNF levels and motor improvement are related phenomena; how long the BDNF increase and the beneficial effects on motor performance are going to last; how much, when, and which type of exercise is required to maintain high BDNF levels and improvement of motor performance. Further studies, both in humans and in animal models, are needed to address all these points.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
