Abstract
Background. Nearly 15 million people suffer from stroke every year worldwide, with about 20% of the survivors retaining chronic aphasic symptoms. Spontaneous recovery is limited to 3 to 6 months. Cortical stimulation techniques have been proposed to enhance the recovery process. Objective. The goal of this study was to evaluate the benefit of epidural cortical stimulation for the treatment of poststroke aphasia, based on a systematic review of the literature. Methods. An extensive PubMed search was performed for English language articles published from 1990 to 2014 with the keywords (cortical OR epidural) AND stimulation AND stroke AND (aphasia OR language OR speech). The criteria analyzed included the type of study, epidemiology of patients, stroke, aphasia, stimulation protocol, concurrent rehabilitation therapies, language evaluations, results observed, and follow-up. Results. Seven cases were reported to date (3 case reports, 1 randomized controlled trial). All patients experienced nonfluent aphasia following an ischemic stroke. All four studies reported encouraging effects of the stimulation with improved lexical access and fluency for all patients. The effects were specific, independent of the motor recovery or of the pain reported by the patients, and they were linked to the stimulation parameters. Conclusions. Due to the small number of existing cases in the literature, the strength of the evidence is still low. Two main hypotheses of neurobiological mechanisms have been explored: either using continuous stimulation to modify cortical perilesional inhibition or using intermittent stimulation during the speech and language therapy sessions to explore synaptic plasticity and long-term potentiation or depression. To establish the role of epidural stimulation and the relevant stimulation protocols and parameters, large randomized controlled trials are mandatory. We suggest avenues of investigation.
Keywords
Introduction
Nearly 15 million people suffer from stroke every year worldwide 1 ; one fifth of them will die and one third will experience permanent disability. 2 Stroke is the leading cause of disability in modern society affecting the activities of daily living and the quality of life.1,3 About 35% of adult stroke survivors initially suffer from aphasia 4 and 20% will retain chronic aphasic symptoms.5,6 Spontaneous recovery that occurs in most patients after a stroke is usually considered to be limited to a period of 3 to 6 months.7,8 Speech and language therapy (SLT) performed during the first weeks can enhance recovery. However, the majority of patients with poststroke aphasia are left with some chronic deficit for which current rehabilitative treatments are limited.3,8
Cortical stimulation techniques have been proposed to enhance or extend functional recovery. Although numerous noninvasive trials have shown promising results for the treatment of chronic poststroke aphasia,1,9 -37 these techniques are still considered only for research protocols and are not employed routinely, mainly because of their presumably temporary effects, as previously demonstrated for the treatment of neuropathic pain38,39 and poststroke motor recovery. 40 From that perspective, a continuous stimulation using an implanted epidural device might afford higher therapeutic potential with a sustainable effect, as shown in the treatment of neuropathic pain. 41 Moreover, implanted epidural devices give the opportunity to provide high-frequency stimulation, which is deemed to carry excitatory effects 42 and an enhanced spatial selectivity. 43 Yet the exact role of epidural cortical stimulation (ECS) is still to be defined. We performed a systematic review of the literature with the aim of exploring the advantages and drawbacks of this technique in comparison to noninvasive stimulations. After presenting the results of this literature review, the present article summarizes the pathophysiology of poststroke recovery, then describes the possible mechanisms underlying the various cortical stimulation techniques, discusses the results of noninvasive1,3,8 -37,44,45 and ECS trials,46 -51 and finally explores the perspectives of ECS in the rehabilitation of poststroke aphasia.
Methods
An extensive PubMed search was performed for articles published from 1990 to 2014 with the keywords (cortical OR epidural) AND stimulation AND stroke AND (aphasia OR language OR speech). Out of the 2000+ entries screened, only 6 articles met our inclusion criteria, that is, English language articles reporting studies relevant to implanted epidural stimulation and to poststroke aphasia. The criteria analyzed in the results section were the number, gender, and age of the patients reported; the type of study (randomized and/or controlled trial, series, case report); the type and location of the stroke; the type of aphasia (fluent or nonfluent) and its severity; the delay between the stroke and the stimulation; the type, number, and position of the electrodes; the stimulation protocol (continuous or intermittent mode); the stimulation parameters (polarity, rate, pulse width, amplitude, hemisphere); the concurrent rehabilitation therapies; the language evaluations performed; the results observed; and the follow-up.
Results
A total of 7 cases (4 males and 3 females, age at stroke 33-58 years) were reported by 4 teams in different countries in the period 2006 to 2013.46 -51 Three cases were reported independently while the 4 other patients were included in a randomized controlled trial. All patients experienced an ischemic stroke of the left internal carotid territory. All patients suffered from nonfluent aphasia following the stroke. Considering all studies, the stimulation was initially started 13 to 195 months after the stroke, and then the follow-up lasted 4 to 75 months. The extracted individual results are collated into a tabular format (Table 1). Two protocols were used, continuous or intermittent stimulation, focusing on 2 pathophysiological hypotheses, a tonic or a remnant effect of stimulation, respectively.
Studies Reporting Poststroke Aphasia Rehabilitation Using Epidural Cortical Stimulation.
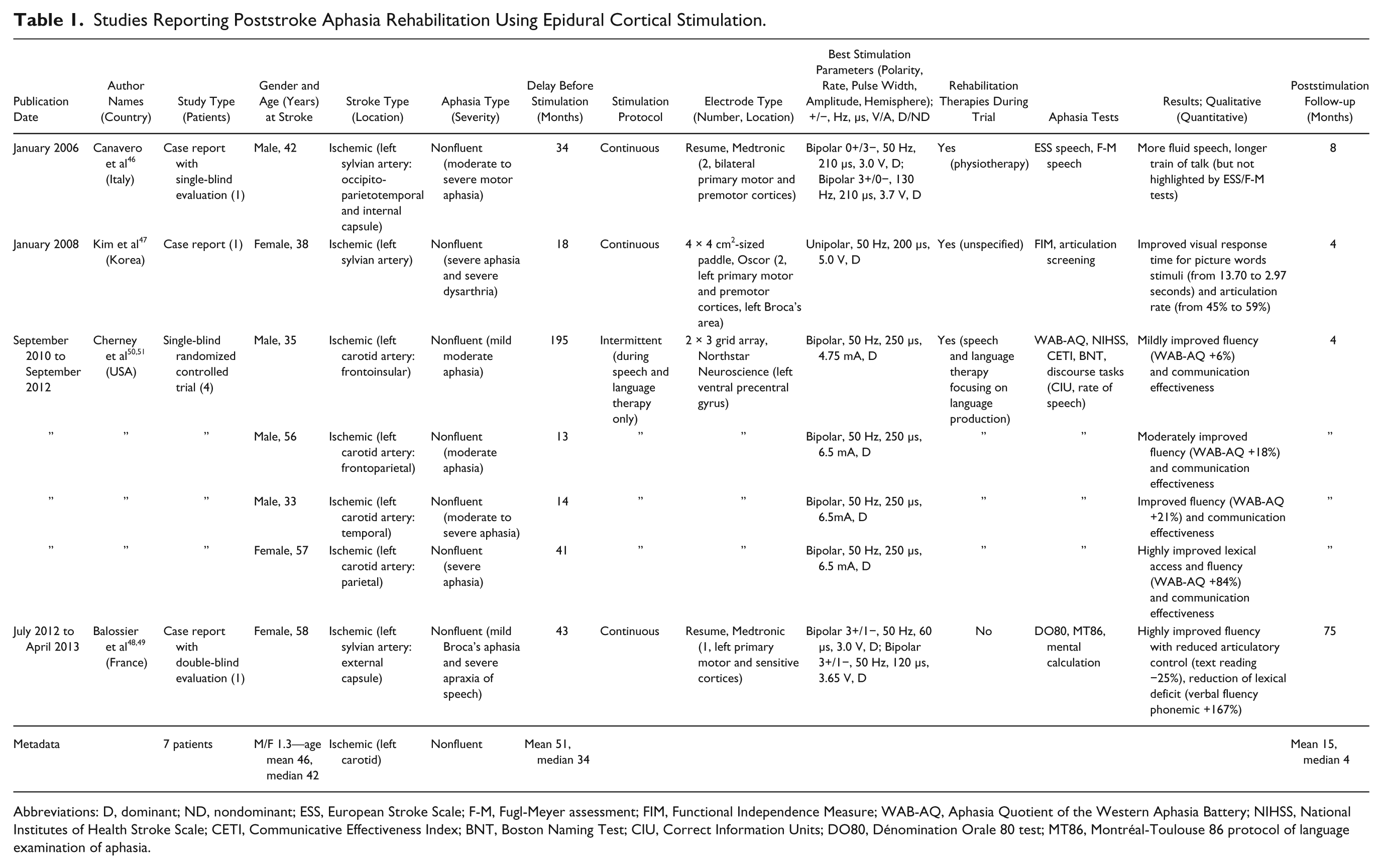
Abbreviations: D, dominant; ND, nondominant; ESS, European Stroke Scale; F-M, Fugl-Meyer assessment; FIM, Functional Independence Measure; WAB-AQ, Aphasia Quotient of the Western Aphasia Battery; NIHSS, National Institutes of Health Stroke Scale; CETI, Communicative Effectiveness Index; BNT, Boston Naming Test; CIU, Correct Information Units; DO80, Dénomination Orale 80 test; MT86, Montréal-Toulouse 86 protocol of language examination of aphasia.
Continuous Stimulation
The 3 case reports used continuous stimulation. Two of them investigated both motor and language recovery, while a third was designed primarily to evaluate the treatment of pain.
Canavero et al published in 2006 the first article describing recovery of poststroke aphasia subsequent to ECS. 46 The authors reported the case of a 42-year-old man suffering from right-hand monoplegia, severe right-arm paresis, and moderate-to-severe motor aphasia after a left middle cerebral artery infarct. The patient initially benefited from physiotherapy but subsequently experienced no further improvement. Thirty-four months after the stroke, one epidural stimulation paddle was implanted on each hemisphere with the electrode overlying the premotor and motor cortices. Then, the patient resumed physiotherapy with the stimulation turned on. Four combinations of stimulation parameters were evaluated on each hemisphere. Stimulation of the dominant hemisphere was reported to enhance language recovery with more fluid speech and longer speech duration. Yet aphasia remained a secondary focus of this study, after motor rehabilitation, and these improvements were not measured by the speech tests employed. The effects were linked to the stimulation parameters. Stimulations of the nondominant hemisphere involved measurable language impairments. After 8 months of stimulation with selected parameters, the same benefit was still seen. The authors concluded that the improvements observed were tonic and long-lasting, sustained by GABA modulation.
Kim et al described in 2008 two cases of poststroke recovery enhanced by ECS, one patient suffering from aphasia. 47 The authors reported the case of a 38-year-old woman experiencing marked limitation of speech (articulation error rate 45%), severe aphasia (not testable), and hemiparesis due to a left middle cerebral artery infarct. The patient benefited from the implantation of 2 epidural electrodes over the upper portion of the left motor and premotor cortices and Broca’s area 18 months after the stroke. At the same time, the patient underwent rehabilitation training. The language impairment was assessed at 4 months. Improved visual response times for picture words stimuli, reduction of articulation error rate, and improved recognition of common nouns were found. Yet the language impairment was not precisely characterized prior to ECS and the improvement was not measured by validated scales. Moreover the content of the active rehabilitation training is not described and the contribution of the SLT in the improvement is unknown.
We published in 2012 another case report describing poststroke recovery subsequent to ECS. 48 A 58-year-old woman experienced aphasia and facial pain after a left middle cerebral artery infarct. Four years after the stroke, a single epidural electrode was implanted over the left primary motor cortex for the treatment of neuropathic pain. As the patient reported an improvement in her speech after the stimulation, a speech evaluation was added to the initial protocol and 2 evaluations were performed by a speech and language therapist, blind to stimulation parameters, during 2 randomized periods of 1 month when the stimulation was turned on then off. An examination of the oral and written language as well as mental calculation was performed. During the off period, the aphasic syndrome was categorized as mild Broca’s aphasia associated with a severe apraxia of speech. The on period resulted in a drastic improvement in spontaneous language fluency with a reduction of control and jerks that were due to the stressing of every first syllable. After these randomized periods, the stimulation was turned on again and the patient was followed-up. At 5 years, while the parameters had been previously slightly adjusted for pain relief, the patient underwent the same speech evaluation showing a sustained effect of the stimulation. After 6 years, as the battery reached end of life, the patient experienced resurgence of her facial pain and speech deficit. The same speech evaluations were then performed before and after the replacement of the battery. The same exact language deficit as during the initial off period was observed just before the replacement. After the replacement of the battery the patient recovered again from her deficit. 49 This study showed that ECS could sustainably enhance poststroke recovery even in chronic deficit and that the effect of the stimulation was tonic and linked to the parameters.
Intermittent Stimulation
Cherney et al published in 2010 the sole randomized controlled trial evaluating the impact of ECS associated with behavioral SLT. 50 Four investigational and 4 control subjects participated. The included patients suffered from chronic nonfluent aphasia with slow agrammatic output and apraxia of speech due to a left carotid ischemic infarction at least 12 months before enrollment. Each patient implanted was matched to a control patient on the basis of their speech deficit. A single epidural electrode was implanted over the left ventral precentral gyrus, with its position previously determined by the functional magnetic resonance imaging (fMRI)-language task. Both investigational and control subjects underwent the same intensive SLT for 6 weeks, then the therapy was stopped and the device removed for the investigational group. Testing was always performed with the stimulation turned off, just after surgery, at 6 weeks postsurgery, and at 6 weeks and 12 weeks following the end of the treatment. The investigational subjects showed a better improvement than the controls at the end of the therapy. This difference was maintained at 6 weeks and was even greater 12 weeks after the end of the trial. 51 The authors reported that the best results were observed for the patients with the most severe aphasia compared to mild aphasia. The authors also showed that the improvement observed in the investigational group was correlated with the decrease in whole brain activation on the fMRI. They concluded that this decrease was linked with more efficient processing from better circuit reorganization corresponding to a mechanism of long-term potentiation (LTP) and reorganization of neural circuits.
Discussion
Pathophysiology of Recovery
Spatial Reorganization
Two main theories have been reported to be relevant for poststroke recovery3,8,52,53: recruitment of dominant-hemisphere perilesional regions for language-related tasks and acquisition or refinement of language processing ability in the nondominant hemisphere.
The language function or the regulation of this function can be transferred to undamaged areas surrounding the ischemic lesion. These perilesional undamaged areas either correspond to neurons situated in the ischemic penumbra or to neurons that are distant from the core of the lesion but are functionally connected to the infarcted tissue. 53 While the mechanisms underlying increased perilesional activation in language recovery have not been fully elucidated, one important contributor may be the absence of collateral inhibition from the lesioned cortex, leading to increased activity in nearby cortical areas. 3 Animal studies of cortical plasticity suggest that the persistent recruitment of the surrounding areas linked with this disinhibition could lead to structural modifications that allow reorganization of perilesional networks.3,53 -55 Nevertheless, although these mechanisms have been described for primary motor and somesthetic cortices, their existence in language areas is still to be proven.
Contralateral activations of the inferior frontal gyrus (IFG) and of the superior temporal gyrus are also seen in the early stages of recovery. These activations seem to appear early in the recovery process but disappear before the second month after the stroke. 56 One of the essential remaining questions is the role of the activation of the nondominant hemisphere and whether this activation is essential for recovery or not. 9 While in the subacute phase the nondominant hemisphere seems to gain activation, in the chronic phase the dominant hemisphere regains dominance.3,9,57 The recruitment of contralesional areas then seems to facilitate recovery in the subacute poststroke period, whereas the sustainable activation of these regions in fMRI could be the reflection of inefficient mechanisms of language processing and may limit the recovery process.3,9,10,53,58 It has been shown in functional imaging studies that patients achieving a better recovery had a greater activation of the dominant hemisphere compared to the nondominant one.8,9,53,56 One possible explanation is that increased nondominant-hemisphere activation, initially resulting from the reduced inhibition by the dominant hemisphere after the stroke, might in turn induce abnormally increased and deleterious transcallosal inhibition of the already damaged dominant hemisphere, leading to incomplete recovery. 3
Depending on whether they favored the hypothesis of recruitment of perilesional regions or the effect of the nondominant hemisphere, the investigators of cortical stimulation chose to stimulate the dominant hemisphere or to inhibit the nondominant hemisphere, respectively.
Temporal Reorganization
Additionally, behavioral and histological data indicate that considerable changes in the cortical representation of language networks can occur within the few weeks following the stroke, and that language recovery after stroke depends significantly on these dynamic neuroplastic changes.3,59
Considering the cellular aspect, 2 main functional mechanisms seem to participate in the recovery process: synaptic plasticity in the surrounding areas and restoration of inter- or intrahemispheric balance. The underlying mechanisms of synaptic plasticity involve the reduction in the efficacy of neuronal synapses known as long-term depression (LTD) and/or the potentiation of existing synapses and unmasking of latent synapses known as LTP that can lead to the creation of new connections known as synaptogenesis.60,61 The cellular processes that occur likely involve variations in gene expression and neurotransmitter levels. 62 The neurotransmitter systems involved in these effects include the inhibitory GABAergic system and the excitatory glutamatergic system with the activation of NMDA receptors. 59 Another possible explanation for recovery is the tonic modulation of synaptic efficacy without structural modifications. These mechanisms are not mutually exclusive, vary among subjects, and seem to be entangled in a dynamic framework depending mostly on the size and exact position of the ischemic lesion in the dominant hemisphere.8,11,56,63,64 Lesions that spare regions surrounding language areas may then favor perilesional neuroplastic changes, while larger lesions that engulf primary language areas may rely on nondominant-hemisphere homotopic language areas. 8
Depending on whether they favored the hypothesis of synaptic plasticity in the surrounding areas or the restoration of inter- or intrahemispheric balance, the investigators of cortical stimulation chose to apply an intermittent or a continuous stimulation protocol, respectively.
Mechanisms of Cortical Stimulation
Two main processes have been developed to modulate cortical and subcortical excitability either with noninvasive stimulation devices such as repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current (tDCS), or with invasive devices such as ECS. Since the early 1990s, stimulation of selected cortical regions has been proposed to treat various neurological disorders. 39 First, Tsubokawa et al showed the potential of continuous stimulation of the precentral cortex by means of implanted epidural electrodes to relieve drug-resistant neuropathic pain. 65 In parallel, methods of rTMS have been developed.
Noninvasive Methods: rTMS and tDCS
Using the principle of electromagnetic induction, magnetic stimulators were capable of delivering very safe repetitive magnetic pulses of sufficient power to induce efficacious depolarizing currents into the brain. 39 The goal of therapeutic neurostimulation or neuromodulation was then to change the excitability or activity of some cortical-subcortical networks involved in the pathophysiological mechanisms of a disease. Various parameters modulate the geometry of the induced electric field and thereby the neural structures that can be activated by rTMS pulses, the major one being the frequency of stimulation. It has been shown, either in healthy volunteers or some patients, that high-frequency stimulation (more than 1 Hz) applied over the primary motor cortex was able to excite the underlying cortex, while low-frequency stimulation (1 Hz or less) caused inhibition. 39 These findings are still debated for nonmotor cortices.66,67 The effects of rTMS are either thought to outlast the stimulation sessions or to vanish after the stimulation. 42 While physiological or clinical effects outlasting the period of stimulation for days suggest a LTP or LTD mechanism,12 -14,39,44 short-lasting effects suggest a tonic effect of the stimulation with temporarily maintained depolarization. 10
In tDCS, weak constant direct currents are delivered by an anode or a cathode placed on the scalp over a targeted cortical area with a reference electrode over the contralateral forehead or the chin. 39 Polarizing currents are able to cross the skull for inducing sustained changes in the membrane potential and in the excitability of cortical cells and fibers that outlast the stimulation. Cortical excitability is thought to be reduced by cathodal stimulation and increased by anodal stimulation, due to processes of neuronal hyperpolarization and depolarization. The effects observed tend to disappear after the sessions. 39
Invasive Method: ECS
The anatomic location and neurochemical mediation induced by ECS have not been clearly identified. Yet the effects are believed to rely on the same mechanisms as those putatively underlying noninvasive stimulation techniques. 68 It has been shown that not only the frequency but the amplitude of stimulation influenced the distribution of electric fields induced in the brain tissue, lower amplitude (<50% of the motor threshold) being more prone to stimulate the pyramidal cell fibers whereas higher stimulation amplitude (>75% of the motor threshold) would activate inhibitory networks.69,70 These findings have been corroborated by other studies and the stimulation parameters have a significant impact on the results.70 -73
Learning From Noninvasive Stimulation Trials
Generally, the treatment of aphasia is based on SLT for the improvement of communication. 15 Given the positive neuroplastic influence of combining noninvasive stimulation techniques with rehabilitative interventions on the motor system, a promising avenue for aphasia rehabilitation may lie in the combination of behavioral therapy and noninvasive brain stimulation methods to further engage brain regions that are important for language recovery.11,15 Two approaches have been primarily used in the application of noninvasive cortical stimulation to rehabilitation of poststroke aphasic patients, both based on the model of interhemispheric rivalry between the residual speech areas in the affected or unaffected hemisphere with either inhibitory or excitatory parameters.10,12 -37,44,45
Stimulation of the Nondominant Hemisphere
Most rTMS studies have evaluated the benefit of low-frequency inhibitory stimulation on the nondominant-hemisphere homotopic language areas, sometimes associated with SLT, with the goal being to focally diminish neuronal activity and suppress maladaptive cortical plasticity.10,12 -14,16 -26,44 The authors postulated that a reduction of the interhemispheric inhibition would favor perilesional recovering of the language abilities. Most of these studies demonstrated improvement either in picture naming accuracy, auditory comprehension, verbal fluency, or repetition after daily rTMS sessions. Not all patients with chronic aphasia respond to low-frequency stimulation. Martin et al reported 2 cases of patients treated by rTMS for chronic nonfluent aphasia. 13 The authors showed that only the patient who experienced aphasia after a left ischemic stroke of Broca’s area responded to the treatment with improved naming and phrase length. The other patient with a more extended lesion of the frontal lobe including the supplementary motor area showed no improvement. These data suggest that differences in lesion anatomy may strongly modulate the functional and behavioral effects of rTMS. Some authors have applied cathodal tDCS stimulation on the right Broca’s homotopic area with the same encouraging results as rTMS studies.15,27 -29
Stimulation of Perilesional Regions
Some authors have chosen to target the dominant hemisphere in order to enhance recovery. rTMS studies using excitatory stimulation over the perilesional areas associated with SLT have shown improvements in language function.30,31 Szaflarski et al showed that this improvement was associated with an increase in the frontal, temporal, and parietal activations of the dominant hemisphere and a nondominant-hemispheric decrease during language fMRI. 30 The authors reported that the effects observed were sustainable and linked with LTP mechanisms. Allendorfer et al showed that not only excitatory stimulation of the left perilesional areas could favor speech improvement, but also that it could trigger changes in white matter structural integrity in the language regions of aphasic patients. 31 Yet the authors reported no correlation between the speech improvement and the degree of white matter increase. The same clinical results were reported with anodal tDCS applied over the left language areas.28,32 -37,45 Still some authors have found that inhibitory tDCS applied over the perilesional areas of the dominant hemisphere could enhance recovery. 28 The authors postulated that the improvement observed was linked to a specific inhibitory effect on overactive interneurons in the damaged dominant hemisphere.
In order to determine the best target for each patient, Abo et al used an fMRI-language task prior to the rTMS sessions and chose either to stimulate the lesioned hemisphere when the highest activation was observed in the perilesional area or to inhibit the contralateral hemisphere when the highest activation was observed in the contralateral areas. 24
Limits of Noninvasive Stimulation Trials
While mounting evidence suggests that noninvasive brain stimulation techniques may be useful as adjunctive treatment for patients suffering from aphasia after stroke, both rTMS and tDCS studies have shortcomings. First, the number of patients included in each study is very small. Although most tDCS studies have shown encouraging results, a meta-analysis failed to prove the benefit of this technique on poststroke aphasia recovery. 1 On the contrary, Ren et al proved the efficacy of low-frequency rTMS in the short term. 25 Yet no meta-analysis has shown the efficacy of high-frequency stimulation. Last, both techniques are deemed to have temporary effects and are time-consuming, limiting their everyday clinical practice.62,74 In order to sustain the effects of noninvasive devices, the stimulation can be combined with SLT. Some authors postulated that tDCS applied as a conditioning treatment before SLT could increase its effects.15,28,29 In contrast, several studies have used tDCS delivered during a language task and during language learning in healthy volunteers with encouraging results. 74 Increased excitability within a language-engaged network and the associated neuronal facilitation are deemed to enhance neuronal plasticity within the damaged language system in a process of relearning language capabilities. 74
Effects of ECS on Aphasia Recovery
While few articles have been published on the use of ECS as a potential treatment for poststroke aphasia rehabilitation, the results are encouraging with improved lexical access and fluency.46 -51 Although most patients were implanted over the left primary motor cortex close to Broca’s area, the improvement observed was not linked to an improved articulation but to an increased verbal fluency and an improved lexical access. The effects were specific, independent of the motor recovery or of the pain reported by the patients and were linked to the stimulation parameters.46,48,49 Only one study 46 evaluated the benefit of stimulation of the nondominant hemisphere; the authors reported measurable language impairments. These results contrast with most of the rTMS studies and may be linked to the impossibility of inducing inhibitory stimulations with epidural electrodes.
The main advantage of ECS over noninvasive stimulation techniques is that it permits high-frequency stimulation of high spatial selectivity to targeted neuronal populations of particular regions of interest. 50 The stimulation can be continuously applied whereas daily repeated rTMS sessions would be time consuming. 42 Eventually, ECS enables brain modulation via a set of parameters (rate, pulse width, amplitude) whose devices currently available afford larger ranges compared to noninvasive stimulations, especially regarding high frequencies (up to 130 Hz for ECS vs 30 Hz only for rTMS, or 100 Hz at the penalty of a lower maximum amplitude for theta burst stimulation). Those parameters can then be adapted to each patient, depending on the deficit, the anatomical lesion, and the functional imaging. Considering the safety of ECS, although it requires surgery, very few adverse events are reported in the literature. Concerning the 7 published cases,46 -51 there were no occurrences of wound infection or postoperative pain reported, and the devices were all well tolerated. Only one patient complained of tingling around the implanted neurostimulator. 50 The treatment was suspended for 1 day and the symptoms spontaneously remitted. This low complication rate is consistent with rates reported in the treatment of neuropathic pain showing the safety of this technique with hundred cases published.39,75
Neurobiological Mechanisms
The exact neurobiological mechanism induced by the stimulation is still debated. While we have shown in our longitudinal study48,49 that the effects disappear a few days or weeks after the end of the stimulation, suggesting a tonic effect of cortical stimulation, Cherney et al have shown that the effects continued to increase after the end of the stimulation, corresponding to a physiological mechanism of LTP.50,51 Both phenomena have been similarly observed in animal models for motor poststroke recovery.70 -73 Some authors showed that the stimulated animals had a better outcome than the controls and that this recovery was linked to an inhibition of astroglial response in the perilesional cortex, and to an increase in dendritic structures and vascular density, suggesting a LTP effect.70 -72 The authors also postulated that these effects were linked to the stimulation parameters. Unlike these observations, we have shown in a nonhuman primate model that poststroke recovery could be improved by primary motor cortex stimulation and that the effects disappeared when the stimulation was turned off. 73 The motor improvement was linked to the stimulation parameters and more specifically to the amplitude of stimulation.
Limits of ECS Trials
As noninvasive stimulation trials, the ECS studies have major limitations. First, the number of patients is very small with mostly single cases being reported. Then, all patients were treated in the chronic phase. To date, no studies have evaluated the role of ECS on fluent aphasia. As for noninvasive techniques, in order to get higher proof of efficacy, further studies are needed. A randomized controlled trial including 10 patients (5 investigational patients and 5 controls) was launched in 2009. 76 The patients included suffered from Broca’s aphasia. Both investigational patients and controls were to benefit from intensive behavioral therapy with the stimulation turned on only during the sessions. The results are still awaited and may bring forward new arguments regarding the hypothesis of synaptic plasticity and LTP. Considering our experience, we believe that even higher expectations could be created from continuous stimulation. Yet, to our knowledge, there is no such trial ongoing.
Perspectives of ECS for Poststroke Aphasia Rehabilitation
Prior to considering this technique in everyday clinical practice, several questions need to be answered. Further studies are required to determine the appropriate subpopulation (fluent or nonfluent aphasia), the timing of the stimulation (poststroke acute or chronic phase), the target (ipsi- or contralateral hemisphere), the protocol (continuous or intermittent mode), and the parameters (polarity, rate, pulse width, amplitude).
Population
To date, no studies have evaluated the effects of ECS for the treatment of fluent aphasia, and the data concerning the effects of noninvasive stimulation are rare. Fridriksson et al showed in a double-blind sham-controlled study of 8 chronic poststroke fluent aphasic patients that the patients under anodal-tDCS associated with SLT had an improved naming reaction time compared to sham-tDCS. 37 This improvement was observed just after the session and was maintained over the 3-week follow-up. Yet the authors used an aspecific test for fluent aphasia, which limits the scope of their results. Abo et al evaluated the role of rTMS in fluent and nonfluent aphasic patients with a frontal target or a temporal target, respectively. 24 The type (inhibitory or excitatory) and target (dominant or nondominant hemisphere) of stimulation were determined based on the results of the fMRI. The authors showed that fluent aphasic patients had an improved spontaneous speech only and nonfluent aphasic patients showed increased auditory comprehension, reading comprehension, and repetition. Yet the improvement was greater for nonfluent aphasic patients.
Thompson-Schill et al explored the role of the IFG in 2 main studies in the selection among competing sources of information.77,78 They first showed in healthy volunteers that the degree of left IFG activity in fMRI could be modulated by the selection demands of the semantic task with a higher activation of the IFG for selective tasks. 77 In a second study, the authors showed that nonfluent aphasic patients with lesions that encompassed left IFG experienced impairments in generating semantically appropriate verbs for concrete nouns with high demands for selection, among competing responses for nouns with low selection demands. 78 The error rate correlated with the size of the lesion in the pars opercularis (Brodmann area 44 dedicated to phonological and syntactic processing). The authors suggested that the IFG subserves a mechanism that is similar to other operations performed by the prefrontal cortex, which can be described as the selection of a response among competing information rather than a specialized semantic retrieval mechanism. Then, although the IFG appeared to be necessary for verb generation, the authors believed that it was not due to the semantic retrieval demands of the task but rather to a semantic selection process that is engaged when there are many sources of competing information. These studies showed that nonfluent aphasia can either be altered by a direct lesion of areas specified for semantic generation or by a lesion of associated areas leading to dysregulation of language. These observations were confirmed by Hamilton et al 14 and Naeser et al, 26 who demonstrated improvement when inhibitory rTMS was applied over the right pars triangularis (Brodmann area 45 dedicated to semantic decision tasks and generation tasks) and not over the pars opercularis. Based on these studies, we suspect that the patients who would best respond to ECS would be those experiencing nonfluent aphasia due to a focal lesion preserving the pars triangularis. The effects induced by neuromodulation devices would then vary depending on the type of regions stimulated, semantic generation areas, or regulation areas.
Timing
All the studies of ECS for poststroke aphasia recovery have focused on chronic deficits, with the patients being implanted more than 12 months after the stroke. Still, would the recovering process be increased if the stimulation was applied during the first weeks after the stroke? The major problem encountered in early stimulation protocols is to distinguish the effects induced by the stimulation from the spontaneous recovery. One of the challenges would be to identify patients with an unfavorable spontaneous outcome which would benefit from the stimulation.
Target
Only one study has evaluated the role of contralateral stimulation with no benefit. 46 We believe that a selection of the stimulation target could be made using fMRI-guided rTMS as this technique has already showed its predictive value in the selection of good responders to ECS for the treatment of pain. 79 Abo et al used the same process to select the stimulation target for their study in aphasic patients. 24 The patients would then be implanted over the hemisphere for which the best improvement in language fluency and lexical access is observed.
Protocol
We have shown in our longitudinal study that the effects of ECS were temporary and disappeared a few weeks after turning off the stimulation.48,49 This observation was confirmed by an experimental study on poststroke motor deficit in a nonhuman primate model. 73 Those observations suggest that the effects of the stimulation are mostly due to a tonic modulation and not to an LTP/LTD mechanism. The stimulation should then be applied continuously and not only during the SLT sessions. To decide this hypothesis, a trial using randomized on and off double-blind periods should be designed, the off periods corresponding in this technique to a sham stimulation as ECS does not induce immediate perception by the patient.
Parameters
To date, no studies have evaluated the exact role of each parameter (polarity, rate, pulse width, amplitude) and the cortical modulation induced in poststroke aphasia. A few authors have already shown that the effects of ECS were linked with the stimulation parameters in preclinical trials on poststroke motor recovery.25,71 -73 Moreover, contrary to noninvasive trials, the effect of the stimulation seems mostly linked with the amplitude based on the motor threshold and not with the rate. 73
Conclusion
ECS studies on poststroke aphasia have shown encouraging effects of the stimulation with improved lexical access and fluency for all patients. The effects were specific, independent of the motor recovery or of the pain reported by the patients and were linked to the stimulation parameters. Yet to date, the strength of the evidence is still low, and the research on this promising rehabilitative technique is in its early stages. To establish the role of ECS in the therapeutic arsenal, large randomized controlled trials are mandatory. The major challenges will be to define the relevant population, timing, target, protocol, and parameters of the stimulation.
Footnotes
Acknowledgements
We thank Mr Graham P. Rhodes for his assistance in reviewing the final version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
