Abstract
Background. Traumatic brain injury (TBI) often leads to mood and cognitive complications, affecting functional recovery. Understanding neurobiological alterations common in post-TBI depression (PTD) and cognition may identify novel biomarkers for TBI complications. Brain-derived neurotrophic factor (BDNF) is a likely target based on evidence of reduced BDNF signaling in experimental TBI and depression models and its role in learning and memory. Objective. To evaluate BDNF as a biomarker for PTD, cognitive impairment, and functional cognition in a prospective cohort with severe TBI. Methods. Participants with TBI (n = 113) were evaluated for PTD (Patient Health Questionnaire-9 [PHQ-9]), cognitive impairment (cognitive composite score), and functional cognition (Functional Independence Measure–Cognition, FIM-Cog). BDNF levels were measured in cerebrospinal fluid and serum at 0 to 6 days postinjury and in serum at 6 and 12 months postinjury. Results. Serum BDNF was reduced after TBI versus controls at all time points. Acute serum BDNF positively correlated with memory composites (6 months: r = 0.43, P = .019, n = 30; 12 months: r = 0.53, P = .005, n = 26) and FIM-Memory scores (6 months: r = 0.35, P = .019, n = 45; 12 months: r = 0.38, P = .018, n = 38). Acute serum BDNF negatively correlated with 12-month PHQ-9 scores (r = −0.38; P = .044; n = 29). At 12 months, chronic serum BDNF tended to be lower in participants with PTD (P = .07) and correlated with PHQ-9 scores (r = −0.41; P = .019; n = 32). Conclusions. Acute BDNF associations with memory recovery may implicate hippocampal damage/degeneration. Comparatively, BDNF associations with PTD status were not as strong as associations with PTD severity. Further investigation may delineate longitudinal BDNF patterns, and BDNF responsive treatments, reflecting mood and cognitive recovery following TBI.
Introduction
Traumatic brain injury (TBI) is increasingly recognized as a chronic medical condition, with accompanying mood and cognitive complications. Impaired cognition and negative mood can adversely affect quality of life and influence return to work or school following TBI.1-4 Identifying sensitive TBI biomarkers for these 2 major complications may be useful for evaluating therapeutic needs and measuring responses to pharmacological and/or behavioral interventions.
Post-TBI depression (PTD) is the most common neurobehavioral complication following TBI. Individuals with TBI are 10 times more likely than the general population to experience a depressive episode during their first year of recovery (53% 5 compared with 6% 6 per 12 months in the general population). The identification of an early biomarker for PTD development would aid in screening and early intervention by identifying those at greatest risk of depression and with the greatest need for high frequency tracking and follow-up. Additionally, a biomarker that is reflective of PTD symptoms may be useful in monitoring and improving treatment effectiveness by informing dose or timing of interventions.
In non–brain-injured populations, individuals with depression often have comorbid cognitive impairment, likely because of common underlying pathology. 7 Whereas individuals with depression report a varying number of cognitive difficulties, 8 memory impairment is consistently problematic. 9 Similar to individuals with depression, individuals with TBI commonly exhibit significant memory, executive function, and attentional difficulties after their injury.10-13 We recently reported that individuals with PTD have no additional cognitive deficits compared with nondepressed individuals with TBI (M.D. Failla, S.B. Juengst, K.M. Graham, P.M. Arenth, A.K. Wagner, unpublished data). Yet, in this same study, individuals with PTD had significantly greater functional cognitive limitations than those without PTD, suggesting that, for individuals with TBI, remittance of depressive symptoms may lead to improvement in functional cognitive limitations, 14 which may be a result of overlapping biological pathways or co-occurring symptomology. Depression and cognitive dysfunction have known effects on multidimensional outcomes15-18 relevant to the Rehabilomics framework, 19 strongly supporting an analysis of biomarker relationships to both conditions.
One potential biomarker that may be relevant to the neurobiological substrates involved with both mood and cognitive dysfunction post-TBI is brain-derived neurotrophic factor (BDNF). BDNF, a neurotrophin involved in neuronal survival and synaptic plasticity, has been implicated in depression, 20 memory and learning, 21 and TBI pathology.22-24 In the hippocampus, BDNF affects synaptogenesis and maintenance, particularly through long-term potentiation associated with activity-dependent secretion of BDNF. 25 BDNF is also reportedly an underlying substrate for persistent long-term memory storage.26,27
Reduced BDNF is known to be associated with depression, and serum BDNF levels are a consistent marker for depressive symptomology in neurologically intact populations. 28 Serum BDNF levels are decreased in untreated depression but increase with antidepressant treatment, indicating the viability for BDNF serum levels as a biomarker of depressive symptoms.28-30 In TBI, serum BDNF is acutely decreased, correlating with injury severity. 31 Hippocampal BDNF is chronically decreased in experimental TBI, 24 and hippocampal BDNF expression has been linked to spatial memory in experimental TBI studies. 22 It is important to note that therapies that increase brain BDNF expression, such as environmental enrichment 24 and exercise,22,32,33 show promise for mood and cognitive recovery post-TBI. This body of work suggests that BDNF may be a viable biomarker for long-term complications such as depression and memory impairments that affect TBI recovery.
In this study, we assessed BDNF as a viable biomarker for PTD, cognitive impairments, and functional cognitive limitations (with specific attention to memory) in the first year following TBI. BDNF serum levels have never been examined in clinical TBI beyond the first week or in relation to PTD or cognition. Thus, BDNF serum levels may be a novel biomarker reflecting these complications and may help elucidate convergent pathways to target for treatment and symptom monitoring in cognitive and depressive symptomology post-TBI.
Methods
Participants
Participants in this study, approved by the University of Pittsburgh’s Institutional Review Board, were recruited while receiving care at inpatient and/or outpatient clinics within the University of Pittsburgh Medical Center. All participants had sustained a nonpenetrating TBI, with evidence of intracranial injury on computed tomography. Exclusion criteria included the following: cardiac arrest or documented prolonged hypoxia or hypotension prior to admission or penetrating TBI. All participants survived for at least 1 year postinjury and were a subset of a larger study investigating biomarkers and genetic factors related to individual recovery following TBI.
Healthy adult controls were also recruited for comparison in biomarker analysis. Criteria for enrollment of controls included the following: (1) 18 to 70 years old and (2) no current or past history of brain injury, neurological disease, psychiatric disease, or bleeding disorder. Data on current depressive symptoms were not collected for this control group. All healthy control participants were Caucasian and ranged from 18 to 60 years old, with 40% being women. Women were excluded if they were pregnant, were taking oral contraceptives or hormone replacement therapy, or had any history of reproductive or endocrine disorder.
Injury severity was described using the best Glasgow Coma Scale score (GCS) obtained within the first 24 hours postinjury. Demographic information, including age, sex, and education, was collected by chart review as well as through participant or caregiver interviews. Similarly, antidepressant use at 6 and 12 months was extracted from both participant interview and chart review. An individual was considered taking an antidepressant if any of the following medications were prescribed within 1 month of the 6- or 12-month assessment: fluoxetine, citalopram, sertraline, escitalopram, paroxetine, trazodone, duloxetine, venlafaxine, bupropion, and mirtazapine. A preinjury history of mood disorders, including depression, bipolar disorder, and anxiety, was established by self-report and chart review.
Cognitive Assessment
Participants’ functional cognitive limitations were assessed with the Functional Independence Measure–Cognition (FIM-Cog) subscale34,35 conducted via interview 36 at both 6 and 12 months. FIM-Cog has 5 component scales: expression, comprehension, social interaction, problem solving, and memory. Each scale is rated from 1 to 7, with a 5 or lower indicative of need for caregiver assistance. The sum of these 5 components was considered the FIM-Cog Score.
Previous studies in TBI37-41 have illustrated the use of composites or global test statistics to evaluate general cognitive performance and to improve study design consistency through aggregation of multiple tests.42-44 Similar to previous studies, 40 cognitive impairment was measured at both 6 and 12 months postinjury using a cognitive composite score developed with a battery of 8 neuropsychological tests targeting 4 domains of cognition (attention, language fluency, memory, and executive function). Attention was measured using the Trails Making Test A 45 and the combined score of the forward and backward digit span tests from the Wechsler Adult Intelligence Scale-R. 46 Memory was evaluated using the Rey-Osterreith Complex Figure Test 47 and the Long Delay Free Recall Subsection of the California Verbal Learning Test. 48 Language Fluency domain scores were calculated using Controlled Oral Word Association 49 Animals Subsection and the Delis-Kaplan Executive Function Systems Verbal Fluency Letter Fluency subsection. Finally, executive function was measured using the Trails Making Test B 45 and the Stroop Task 50 Interference Subscore. These tests were selected as representative measures for their associated domains. Raw scores from each test were converted into T scores using appropriate metrics (ie, education, age, sex, race) based on norms indicated by the test manufacturer. T scores were averaged within each domain to create a domain subscore. To calculate an overall cognitive composite score, participants had to complete at least 1 test in each of the 4 domains. Mean values across domain subscores were calculated for the overall cognitive composite score. Additionally, because T scores have a mean of 50 and a standard deviation of 10, a t score cutoff of 40 was used to delineate impaired (greater than 1 SD below the mean) versus unimpaired performance in the TBI population. This cutoff is traditionally used in neuropsychological assessment to indicate the presence of mild cognitive impairment. 51
Depressive Symptom Assessment
At 6 and 12 months, depressive symptoms were evaluated using the Patient Health Questionnaire-9 (PHQ-9), a brief self-report symptom inventory based on the 9 Diagnostic and Statistical Manual of Mental Disorders (DSM), 4th edition, diagnostic criteria for major depressive disorder. The PHQ-9 asks participants to rate how often they have experienced symptoms of depression, on a scale between 0 (none) and 3 (nearly every day), over a 2-week period. Higher total scores (PHQ-9 Total) reflect greater number of and/or greater severity of depressive symptoms, with the maximum score being 27. Participants were grouped as “depressed” versus “nondepressed” using the PHQ-9 questions as they map to DSM diagnostic criteria (previously described). 52 For a categorization of depression (PTD), individuals responded positively to at least 5 symptom questions on the PHQ-9, with at least one pertaining to a cardinal symptom (anhedonia or depression). Compared to the Structured Clinical Interview for DSM Diagnosis, 52 this method has been validated in populations with TBI, showing a sensitivity of 93% and a specificity of 89%. It is important to note that the PHQ-9 is reliably able to discriminate between chronic TBI symptoms and depression symptoms. 53
BDNF Sample Cohort, Collection, and Processing
BDNF levels were measured in cerebrospinal fluid (CSF) and serum. When possible, CSF samples were collected via passive drainage up to twice daily for 6 days postinjury by an external ventricular drain placed for clinical care. Serum was collected, via venipuncture, daily for the first 6 days and chronically at 6 months and 12 months postinjury via venipuncture. Acute CSF and serum samples were binned by day, and an average was calculated for each day postinjury for each participant.
CSF and serum samples were stored at −80°C and were thawed immediately for measurement. BDNF was assayed by an enzyme-linked immunosorbent assay (ELISA) kit (RayBiotech, Norcross, GA). Briefly, standards (of varying BDNF concentrations) and samples were pipetted onto a 96-well plate precoated with human BDNF antibody. Following shaking for 2.5 hours at room temperature, the plate was washed and then incubated with biotinylated BDNF antibody for 1 hour. HRP-conjugated streptavidin was then added for an incubation of 45 minutes. The addition of a tetramethylbenzidine substrate allowed for a color reaction. Concentrations were calculated using mean absorbance of each sample at 460 nm because it correlates with the amount of BDNF present in samples, as plotted on a per-assay standard curve. Samples were diluted within the range of the ELISA kit (no dilution for CSF, 1:250 for serum samples), with a sensitivity of 80 pg/mL, a kit supplied intra-assay variation of <10%, and an interassay variation of <12%.
For biomarker analysis, participants (n = 113) were assayed for BDNF levels in CSF or serum samples. BDNF levels have been shown to be moderated by racial background. 54 Because the study population was 91.9% Caucasian, there was not enough power to detect racial differences between BDNF level associations and outcome in the current data set. As a result, we limited BDNF level associations to Caucasians only, with the plan to collect further data that include additional racial groups. There were 97 participants with CSF samples (n = 446) and 81 with acute serum samples (n = 204). One acute serum sample measurement and 4 CSF sample measurements were removed as outliers (based on ±1.5 times the interquartile range). Chronic serum samples were collected at 6 and 12 months (±1 month) postinjury and averaged for each time point for each participant. At 6 months, there were 54 participants with 112 samples. At 12 months, there were 36 participants with 36 samples. A subset of participants (n = 44) had both acute and chronic samples.
For biomarker comparisons, healthy controls were recruited as a reference group (n = 9). Control CSF was obtained via lumbar puncture for research purposes and was not a part of a clinical workup. Serum and CSF samples were obtained at ~7
Statistical Analysis
Analysis was conducted using Statistical Analysis Software (version 9.4; SAS Institute, Cary, NC). Descriptive analysis included mean and standard deviation and/or median for continuous and ordinal variables such as age, GCS, and education. Frequencies were calculated for categorical variables such as sex and antidepressant use. Demographic and relevant clinical information was assessed for relationships with BDNF levels using Student’s t tests or ANOVA to compare means. Nonparametric tests (Mann-Whitney and Kruskal-Wallis) were used when appropriate. Outliers were assessed using ±1.5 times the interquartile range. Pearson’s or Spearman’s ρ (r) correlations were used to assess relationships between 2 continuous variables.
Results
Specific cohort demographics are shown in Table 1. Overall, participants had a GCS (best in 24 hours) of 3 to 15 (mean GCS = 7.7 ± 2.8, median = 7). Participants were 16 to 72 (mean age = 33.1 ± 13.3) years old, and 17.7% of participants were women. In comparison, healthy controls were 18 to 60 (mean age 27.6 ± 13.3) years old, and 35.7% were women. At 6 months postinjury, 38.3% of participants with TBI had PTD, whereas 30.3% had PTD at 12 months. Participants with PTD tended to have a higher mean age compared with those without PTD (6 months, P = .094; 12 months, P = .098). At 6 months, women were more likely to be depressed than men (62.5% of women compared with 30.4% of men, P = .018). Participants with premorbid mood disorders had higher PTD rates at both 6 (22.6 vs 5.6%, P = .022) and 12 months (33.4 vs 8.3%, P = .007) compared with those without premorbid mood disorders. At 6 months, 51.6% of participants with PTD were taking an antidepressant, compared with 26.4% of those with no PTD (P = .021). There was no significant difference in antidepressant use between PTD groups at 12 months. Table 2 shows relationships between BDNF levels and demographic variables. Only sex was associated with BDNF levels at any time point. At 12 months, women had lower BDNF levels compared with men (P = .009). It is important to note that acute serum and CSF levels tended to be negatively correlated (r = −0.31; P = .069; n = 35).
Demographic Description of Study Population.
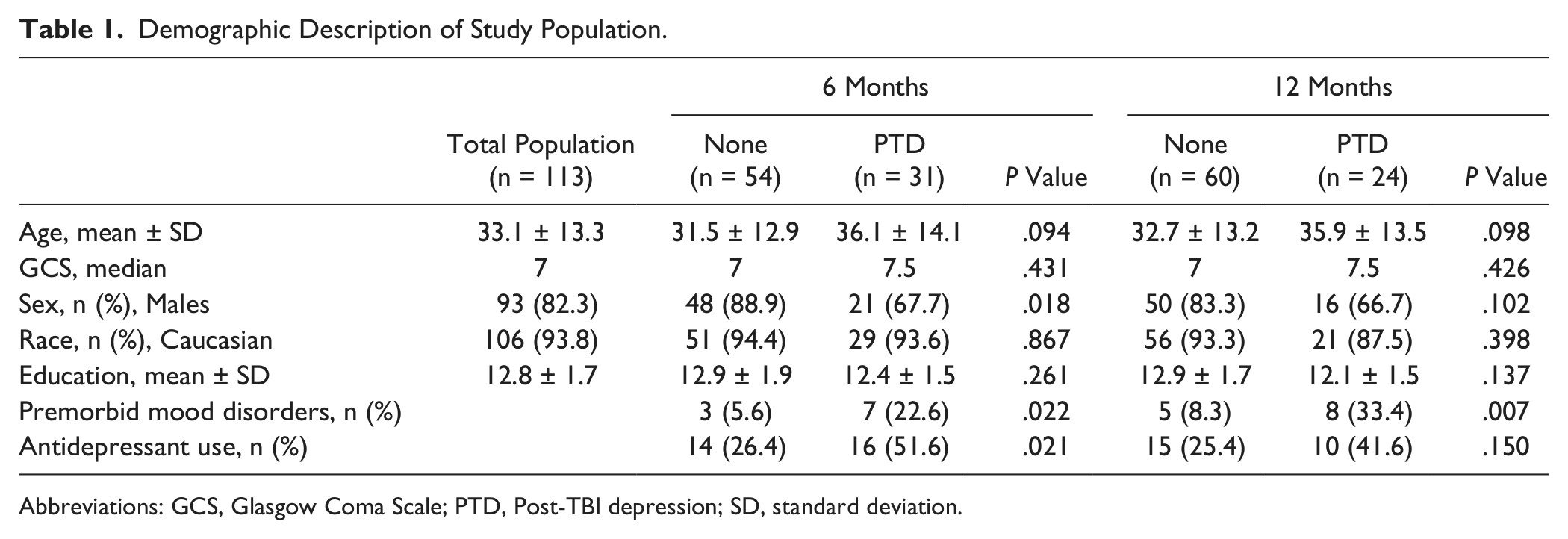
Abbreviations: GCS, Glasgow Coma Scale; PTD, Post-TBI depression; SD, standard deviation.
Demographic Associations With BDNF Levels.
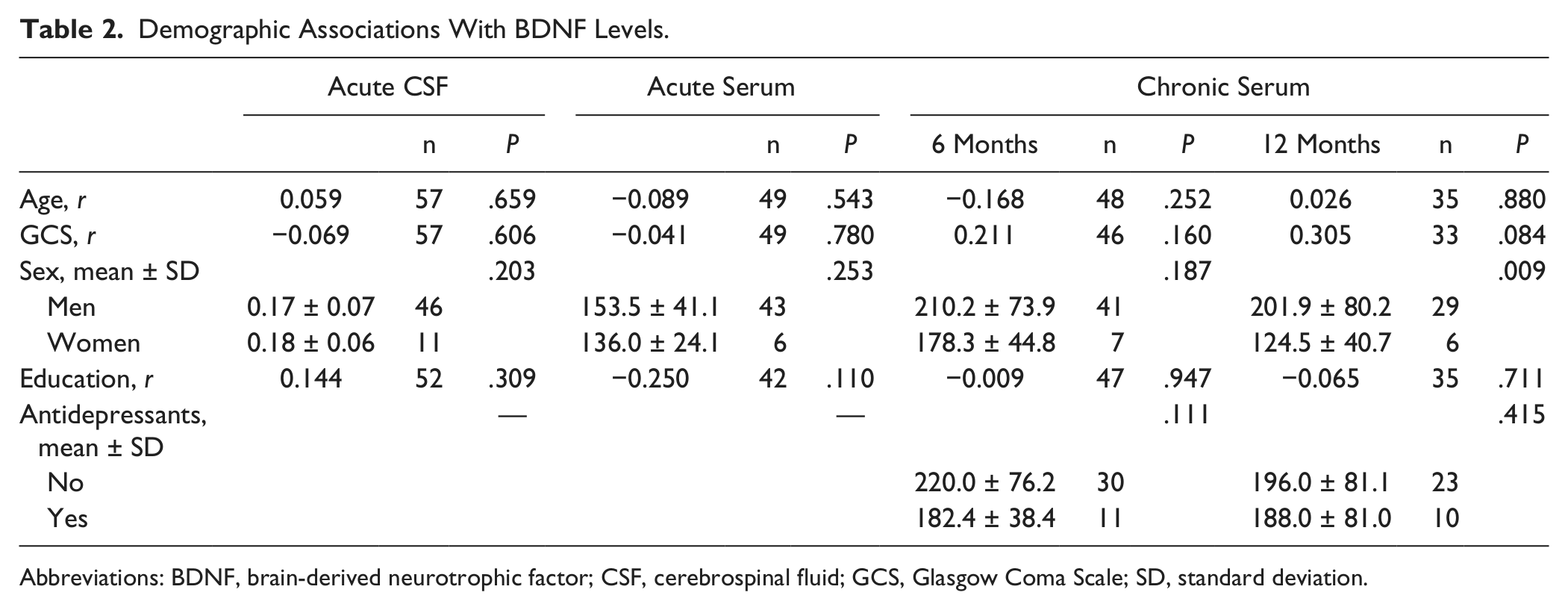
Abbreviations: BDNF, brain-derived neurotrophic factor; CSF, cerebrospinal fluid; GCS, Glasgow Coma Scale; SD, standard deviation.
BDNF Associations With PTD, Cognitive Impairment, and Cognitive Function
As shown in Figure 1A, daily mean CSF BDNF levels among TBI participants did not differ from that among healthy controls. Conversely, daily mean serum BDNF levels in participants with TBI were consistently reduced compared with healthy control levels beginning day 0 (206.27 ± 16.4 ng/mL vs 277.86 ± 28.1 ng/mL for healthy controls, P = .024) and remained below that of healthy controls for all days (all comparisons P < .01, n = 49, Figure 1B). Serum BDNF levels at 6 (205.57 ± 10.2 ng/mL, P = .011) and 12 (188.67 ± 13.5 ng/mL, P = .008) months were also below that of healthy control levels.
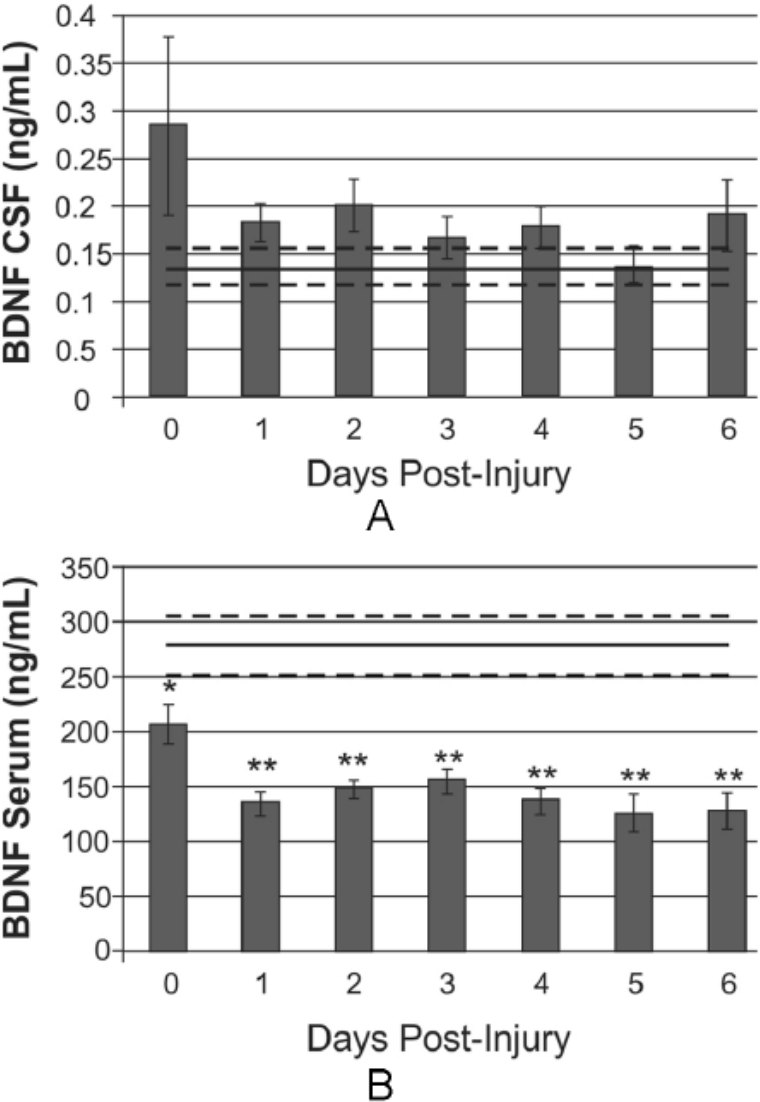
Daily BDNF levels over the first 7 days postinjury, compared with levels in healthy controls (mean in black line, standard error in light gray dashed lines). A. Daily mean CSF BDNF levels do not differ significantly from control levels. B. Daily mean serum BDNF levels fall below control levels at day 0 postinjury and remain reduced through day 6 (day 0, P < .05; days 1-6, P < .001).
BDNF levels were examined for associations to PTD status and symptom severity. CSF BDNF levels were higher in participants with PTD at 6 months, though this association was not significant (P = .089). Acute serum BDNF levels in participants with PTD tended to be reduced compared with those with no PTD at 6 months (P = .074). Acute serum BDNF did not predict PTD status at 12 months. Figure 2 shows chronic serum BDNF levels by PTD status. At 6 months, there were no significant differences in chronic serum BDNF levels by PTD status (P = .174). Yet those with PTD had significantly lower serum BDNF compared with controls (P = .012), whereas those without PTD did not differ significantly from controls (P = .070). At 12 months, participants with PTD tended to have lower serum BDNF levels than those without PTD (P = .066). Participants with and without PTD showed lower chronic serum BDNF levels compared with controls (P = .037 and .004, respectively). Table 3 shows associations between BDNF levels and PTD severity (PHQ-9 Total Score). Acute serum BDNF was negatively correlated with PHQ-9 scores at 12 months (r = −0.38; P = .044; n = 29), such that lower serum BDNF levels were associated with higher (worse) PHQ-9 scores. Similarly, chronic levels at 12 months were negatively correlated with PHQ-9 scores (r = −0.41; P = .019; n = 32).
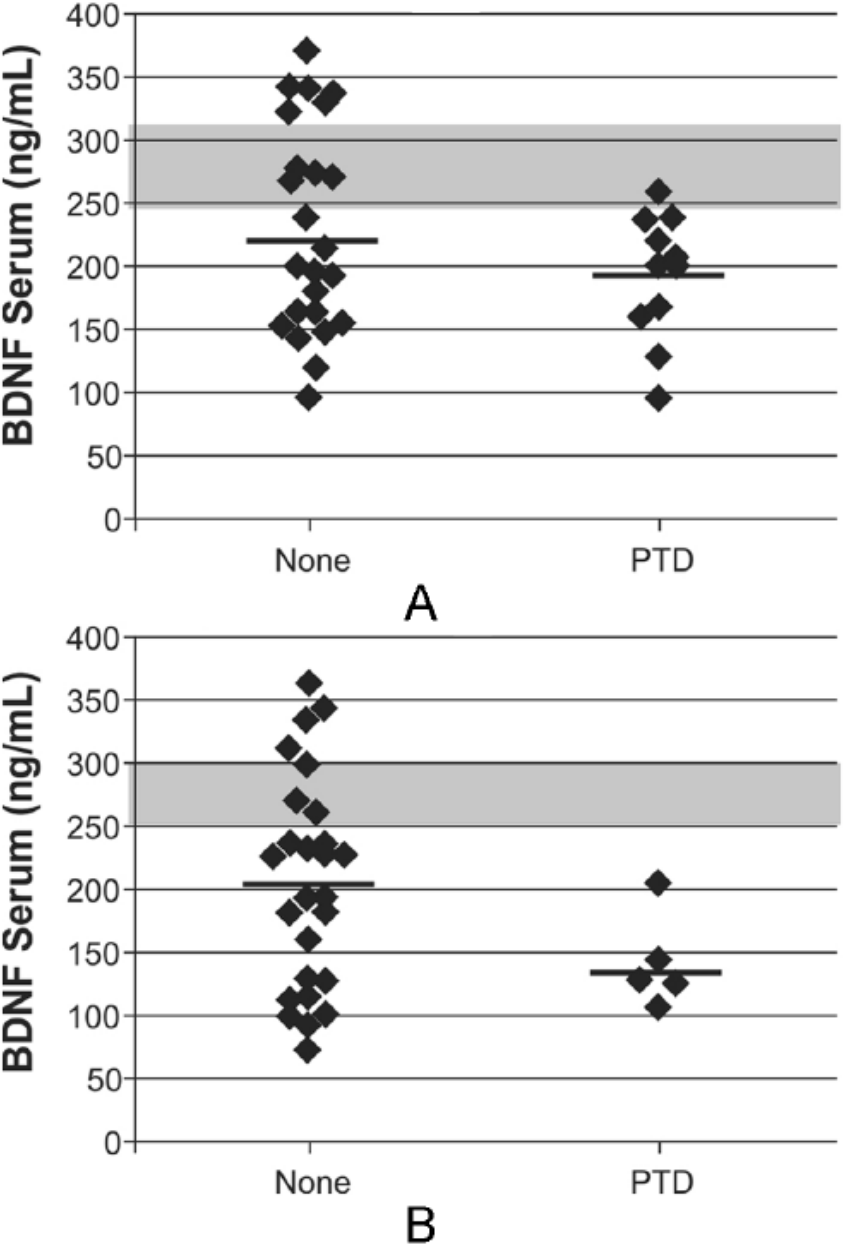
Serum brain-derived neurotrophic factor (BDNF) levels during chronic recovery at 6 and 12 months were examined in participants with PTD and no PTD. At 6 months (A), there were no significant differences in serum BDNF by PTD status (P = .174). At 12 months (B), participants with PTD had lower serum BDNF levels compared with participants with no PTD, though this was not significant (P = .066). At both 6 and 12 months, BDNF levels in participants with PTD were reduced compared with control levels; similarly, levels for participants with no PTD were reduced compared to controls (P < .05 for all comparisons).
Bivariate Correlations Between BDNF Levels and Mood and Cognitive Outcome.
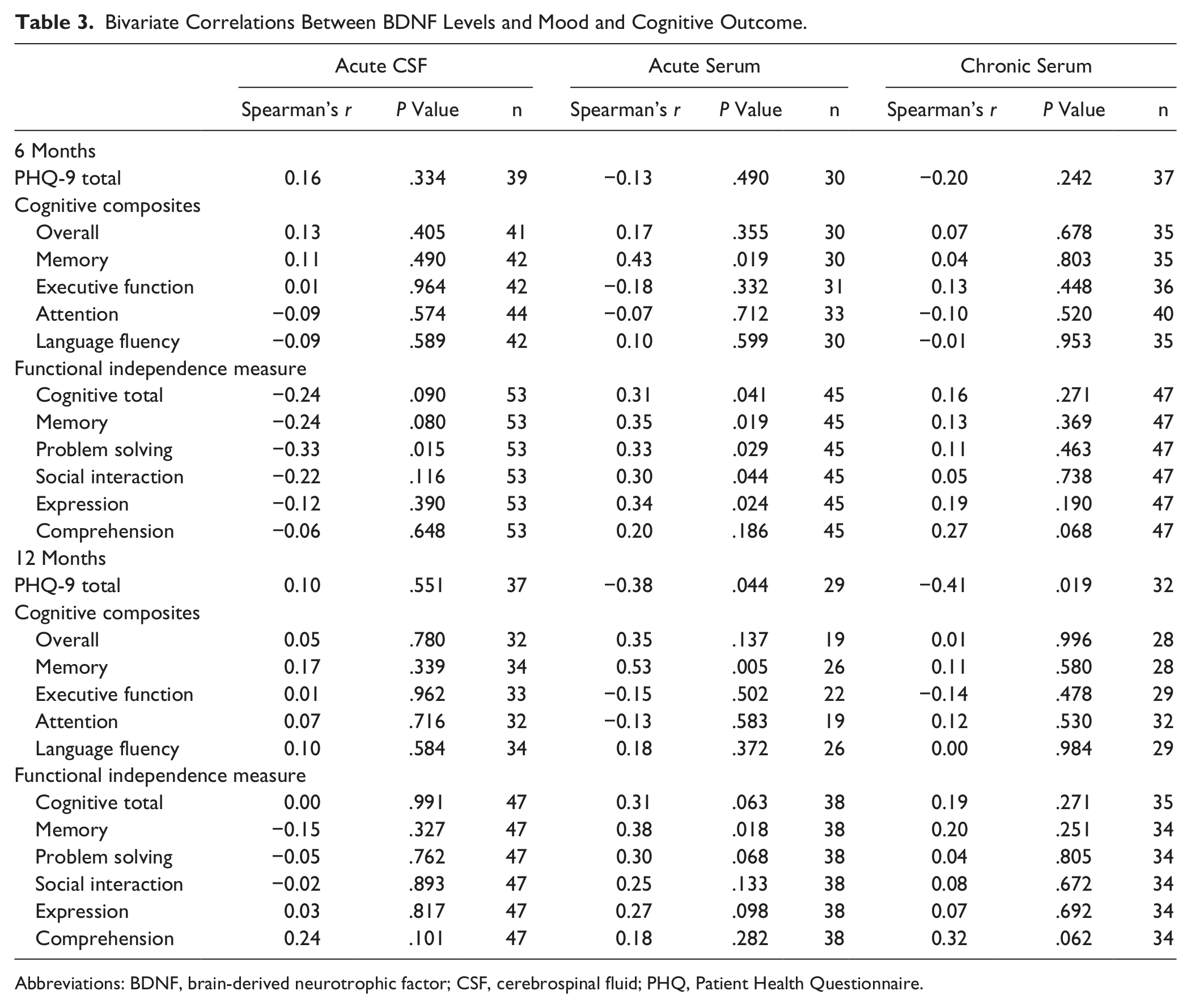
Abbreviations: BDNF, brain-derived neurotrophic factor; CSF, cerebrospinal fluid; PHQ, Patient Health Questionnaire.
BDNF levels were examined for associations to cognitive impairments and functional cognitive limitations (Table 3). Acute serum BDNF levels were positively correlated (Figure 3) with memory composite scores at 6 (r = 0.53; P = .005; n = 30) and 12 months (r = 0.38; P = .018; n = 26). Using a t score cutoff of 40 to designate impaired performance, acute BDNF levels were reduced in those individuals with a memory composite score <40 (impaired) compared with those with memory composites >40 at 6 months (146.9 ± 8.4 vs 168.2 ± 8.4, P = .024) and 12 months (144.9 ± 10.9 vs 167.2 ± 7.5, P = .027). CSF BDNF and chronic serum BDNF levels were not associated with any cognitive composite components. Acute serum BDNF levels were positively correlated with FIM-Cog at 6 months (r = 0.31, P = .041, n = 45). All FIM-Cog component scales were positively associated with higher serum BDNF at 6 months (Table 3). CSF BDNF levels tended to show a negative correlation with FIM-Cog at 6 months (r = −0.24; P = .090; n = 53), with a significant association with FIM-Problem Solving (r = −0.33; P = .015). At 12 months, acute serum BDNF levels were positively correlated to FIM-Memory (r = 0.38; P = .018); CSF BDNF did not show any significant correlations to 12-month FIM-Cog or FIM-Cog components. There were no significant relationships between chronic BDNF and functional cognition at either 6 or 12 months.
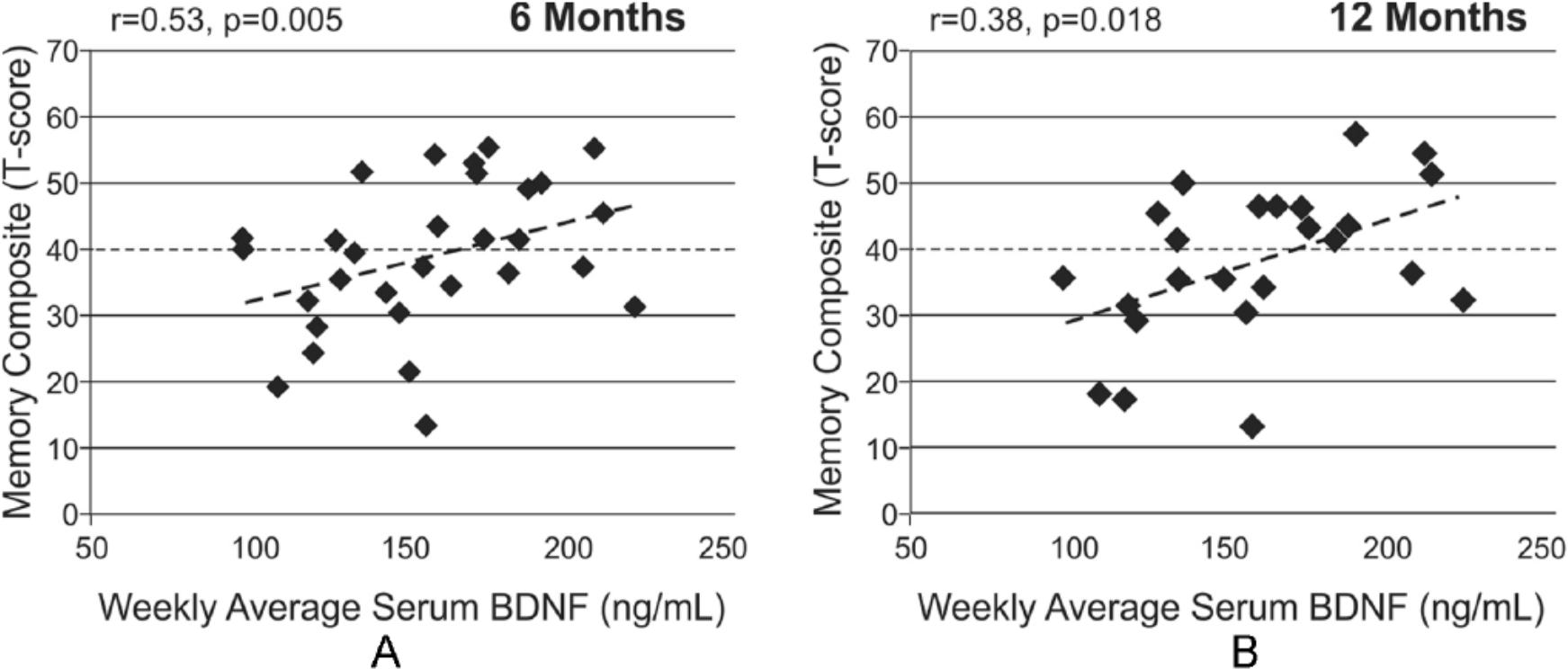
Serum brain-derived neurotrophic factor (BDNF) levels during the first week postinjury were investigated for predictive associations with memory impairment at 6 and 12 months postinjury. At 6 months (A) and 12 months (B), acute serum BDNF levels predicted performance on memory composite (r = 0.53, P = .005, and r = 0.38, P = .018, respectively). A dashed line denotes the T score cutoff for impairment on the memory composite.
Discussion
In this study, we investigated BDNF levels as a common biomarker for PTD pathology, cognitive dysfunction, and functional cognitive impairment post-TBI. Because PTD and cognitive difficulties often co-occur postinjury, we aimed to evaluate BDNF as a biomarker representing common pathology underlying these complications. In this study, acute serum BDNF levels were associated with chronic memory impairments, global functional cognitive limitations, and depressive symptom severity. Chronic serum BDNF levels, however, were not associated with any cognitive outcomes but tended to be lower in individuals with PTD. Yet, acute CSF levels tended to correlate with acute serum, but were not predictive of chronic outcome measures. These findings are summarized in Figure 4.
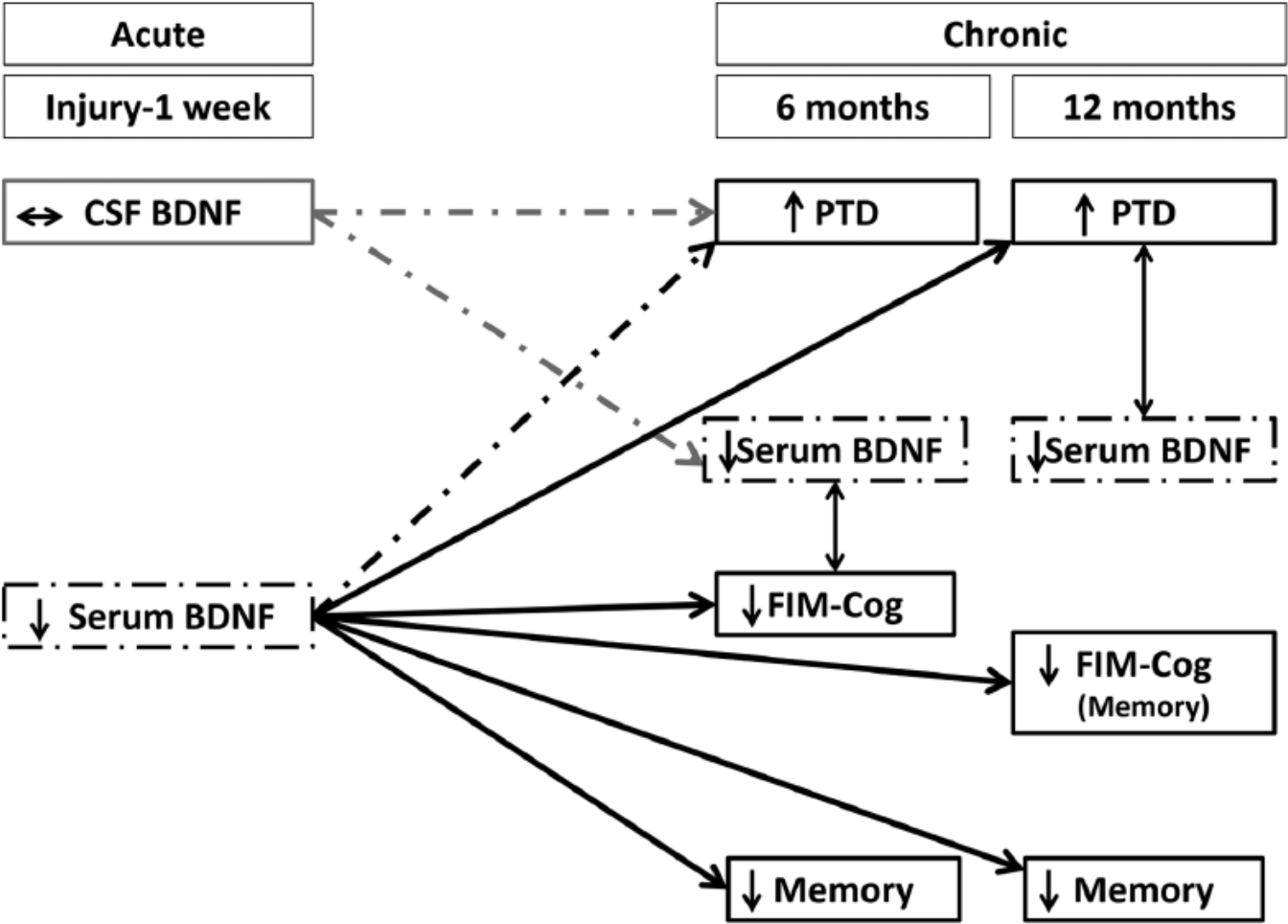
The following schematic summarizes the main findings regarding associations between brain-derived neurotrophic factor (BDNF) levels and depression or cognition following traumatic brain injury (TBI). Dashed lines and boxes depict trends in the data, whereas solid lines and boxes represent significant findings. These data are shown across a timeline from the date of injury through the first week (acute) and then 6 and 12 months postinjury (chronic). Memory refers to cognitive composites of neuropsychological tests examining memory.
This work has 2 potentially important implications: first, acute serum BDNF may be a viable predictive biomarker for both mood and cognitive complications within the first year post-TBI, indicating a possible treatment window in the acute phase that could have implications for long-term mood and cognitive recovery; second, chronic serum BDNF may be reflective of PTD severity, suggesting a potential biomarker for tracking treatment response and effectiveness in real-time. Although future studies will need to evaluate this work in larger cohorts, the findings presented here suggest that BDNF may be a viable treatment target, with dynamic utility across recovery.
Serum BDNF levels were low immediately after injury, consistent with previous studies.31,55 Also, our own previous work demonstrates that low serum BDNF is a mortality marker following TBI, suggesting that immediate reductions in serum BDNF levels are indicative of greater TBI-related pathology. Kalish and Phillips 31 demonstrated immediate reductions in serum BDNF, but this finding was associated with GCS. 31 In our current study, there is no relationship between GCS and acute serum BDNF. However, our population included participants with moderate to severe injury, whereas the Kalish and Phillips 31 study included those with mild TBI. We also report that serum BDNF levels remain reduced chronically, adding evidence to the emerging concept of TBI as a chronic condition with long-term pathophysiological alterations.
In uninjured populations, BDNF serum levels are consistently reduced among individuals experiencing depressive symptoms. 56 Under uninjured conditions, serum BDNF levels likely reflect CNS functioning because BDNF is primarily synthesized in the brain and secreted in an activity-dependent manner. 57 Several studies suggest that serum increases in BDNF are a result of brain-level changes.58,59 Serum BDNF is likely reflective of BDNF expression in the hippocampus, where tissue BDNF levels are decreased in correlation with stress and depression. 60 Hippocampal BDNF signaling is also implicated in mechanisms of antidepressant treatment. 61 In rat models, intracerebral BDNF infusions have antidepressant effects, whereas decreased BDNF signaling results in decreased hippocampal neurogenesis. 62
Reduced BDNF may be indicative of a depressive state following neurological insult. Following acute stroke, there were relative reductions in acute serum BDNF for those with depressive symptoms that did not occur in those without depression within the same time frame. 63 Similarly, chronic BDNF levels at 12 months in our study correlated with depressive symptom severity within the corresponding time frame. However, there was limited discriminatory ability of BDNF levels at chronic time points, possibly because of issues with sample size and variability in biomarker values. Additionally, there may also be a “floor effect” following TBI, where BDNF levels are already significantly lower than in controls, impairing our ability to discriminate PTD status.
The data from this study suggest that acute serum BDNF levels are highly associated with memory impairment post-TBI. Other studies have demonstrated similar relationships between serum BDNF levels and cognitive impairment. 64 Specifically, in patient populations (schizophrenia, bipolar disorder, mild cognitive impairment) and healthy controls, 65 there are relationships between BDNF levels and memory. 66 Animal studies using conditional BDNF knock-out mice show impaired hippocampal-dependent cognition and behavior. 67 Thus, injury-induced acute reductions in serum BDNF may be indicative of the degree of direct damage to the hippocampus. Similarly, levels may also be indicative of an acute injury state that is conducive to neuronal death and atrophy that would predict later memory performance. Low BDNF signaling in the hippocampus may diminish synaptic plasticity and neurogenesis, negatively affecting these chronic recovery end points. Future studies are needed to examine the relationship between acute BDNF levels and chronic hippocampal volume because this may aid interpretation of these findings. Because experimental TBI models show that hippocampal BDNF expression is correlated with cognitive recovery, 22 it will be critical to understand if/how early interventions targeting BDNF might improve chronic memory problems. Participants with lower BDNF levels acutely do tend to have lower levels chronically. One recent study demonstrated detrimental effects of long-term BDNF reductions on age-related decline in animal models. 68 Thus, understanding the trajectory of BDNF profiles and their relationship to memory performance could guide treatment and intervention strategies and improve long-term outcomes. Acute BDNF serum levels did not correlate with any other measures of cognitive performance but were significantly associated with multiple components of functional cognition. This finding may be a result of the importance of memory in other aspects of functional cognition. 69
BDNF CSF levels did not show consistent associations with mood or cognitive recovery. However, higher CSF levels tended to be associated with worse outcomes on the FIM-Cog subcomponents. This trend is consistent with our previous work 55 showing that high levels of CSF were associated with greater mortality post-TBI. However, it is important to understand interactions between CSF and serum BDNF levels. In fact, we show a trend for a negative correlation between CSF and serum BDNF (consistent with previous work 55 ). Whereas it has been suggested that BDNF can cross the blood-brain barrier in both directions under normal conditions,70,71 disruption of the blood-brain barrier following TBI could allow for increased BDNF transit into the brain, especially during the initial days after TBI.71,72 This increased transit from blood into CSF could reflect a possible protective process because platelets dump BDNF in response to vascular injury. 73 Therefore, lower BDNF levels in serum acutely may be suggestive of more extensive injury and subsequent expenditure of systemic stores of BDNF into the CNS, correlating with later development of chronic conditions.
In line with personalized medicine and Rehabilomics, 19 it is important to consider several demographic, medical, and genetic factors that may influence BDNF levels. Some studies suggest that BDNF levels vary by BDNF genetic variation. 74 Future studies in larger cohorts are needed to determine the effect of BDNF variation on chronic BDNF levels after TBI. It will also be important to evaluate serum BDNF post-TBI in relation to therapies that stimulate BDNF signaling (eg, exercise, SSRIs). In uninjured populations, serum BDNF levels are decreased in untreated depression but increase with antidepressant treatment.28-30 However, this study was not designed to evaluate the utility of BDNF as a biomarker of PTD remittance following antidepressant use. A limitation in this study is the inclusion of individuals on antidepressants; however, BDNF levels are thought to be more indicative of depressive symptomology and remittance than antidepressant use. 75 Additionally, many individuals who are on antidepressants are still depressed; the opposing effects of antidepressant use and depression may reduce associations with BDNF levels. Future studies in PTD will need to examine these relationships in carefully designed studies where relationships between BDNF levels, antidepressant type/dose, and depressive symptoms are examined.
It is also possible that this study was underpowered to detect some associations between BDNF and TBI-related outcomes, given possible effects of demographics such as age and sex on BDNF levels. 76 In this study, age and sex were not consistently associated with BDNF levels, though BDNF levels were significantly lower in women at 12 months postinjury. Future studies are needed to examine the impact of demographic variables on BDNF levels specifically following TBI and in concert with TBI outcomes.
There are other important limitations to consider concerning this study and BDNF evaluations. Similarly, it will be important to understand the relationship between BDNF levels in participants with PTD, with and without cognitive impairment. A general limitation is the assumption that serum BDNF levels represent brain levels because there is a substantial peripheral production of BDNF in vascular endothelial cells, 73 with storage in platelets. 77 The literature suggests that platelet release is not altered in depression, 56 but it is unclear if platelet release could be altered in PTD or TBI, or how peripheral stores of BDNF might influence our findings. As a reference group for biomarker levels, the sample size is small and cannot account for potential genetic or other sources of variability. Controls were not screened for depressive symptoms. It is possible that some healthy controls could have had depressive symptoms or other disorders that they did not report at the time of their interview. However, if that is the case in controls, then our results comparing individuals with TBI with controls would be biased toward the null.
The work presented here provides preliminary evidence of potential utility for BDNF as a predictive biomarker for cognitive recovery following TBI. Larger studies are needed to evaluate BDNF utility in discriminating PTD status; however, our data indicate that BDNF may be associated with depressive symptoms post-TBI. Examining BDNF levels in stratified cohorts with and without PTD and cognitive impairment may elucidate the specificity of BDNF as a biomarker post-TBI. It will be important to understand, mechanistically, utilizing animal models, what the resulting BDNF levels in this study indicate about underlying TBI pathology and possible treatment development. Future studies are likely needed to evaluate temporal BDNF trajectories across recovery, as PTD and cognitive impairments develop and/or resolve, for a better understanding of BDNF effects on TBI recovery.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by NIH R01 HD048162; NIDRR H133A120087; DODW81XWH-071-0701; CDC R49 CCR 323155, University of Pittsburgh Women’s Studies Faculty Research Fund.
