Abstract
Introduction
Traumatic brain injury (TBI) is an important cause of cognitive deficits as studied in humans1-3 and animals,4,5 and there are no therapies that are effective in mitigating the consequences of TBI. In particular, concussive brain injury is a major cause of neuropsychological disability in spite of no obvious neuronal death, 4 which suggests that subtle disturbances in neuronal function can be underlying causes of such disability. TBI elicits oxidative damage to plasma membrane phospholipids, which can result in long-term harm in interneuronal communication and cognition.5,6 Accordingly, strategies directed to preserve phospholipids in the plasma membrane such as the use of dietary docosahexaenoic acid (C22:6n-3; DHA) 5 can have beneficial effects for post-TBI recovery. DHA is the most abundant polyunsaturated fatty acid (PUFA) in the brain, where the DHA-containing phospholipids contribute to plasma membrane biogenesis and receptor signaling. The fact that TBI-induced oxidative damage to phospholipids can also damage DHA has raised interest to optimize its healing capacity after TBI. The phenolic yellow curry pigment curcumin has potent anti-inflammatory and antioxidant activities that can function to reduce oxidative damage and cognitive deficits associated with neurological disorders.7-9 Curcumin provided in the diet before TBI can reduce oxidative damage and counteract TBI-related cognitive dysfunction. 10 This study was performed to assess whether curcumin provided after TBI can complement the action of DHA in the TBI pathology and foster functional recovery.
The metabolism of DHA from its precursor α-linolenic acid shares pathways with the metabolism of n-6 PUFAs from its precursor linoleic acid11,12 and opens the possibility that the production of DHA can be enhanced at the expense of reducing n-6 PUFAs. This is actually a desirable objective because n-6 PUFAs often antagonize the action of DHA, with detrimental consequences for brain function. The metabolism of these fatty acids is regulated by several enzymes, of which the Δ6-desaturase (FADS2) and 17β-HSD4 are very important. The function of DHA in the plasma membrane is to maintain the fluidity of the membrane, and 4-hydroxy-2-nonenal (4-HNE) and 4-hydroxy-2-hexenal (4-HHE) are markers of membrane lipid peroxidation affecting n-6 fatty acids and n-3 fatty acids, respectively. Membrane fluidity affects the signaling through transmembrane embedded receptors such as the brain-derived neurotrophic factor (BDNF) receptor TrkB. The function of BDNF is important for synaptic plasticity underlying cognitive processing in models of TBI,13,14 such that loss of BDNF function can have consequences for post-TBI recovery events. Accordingly, the current study was conducted to explore how the combinatory action of DHA and curcumin could help preserve plasma membrane stability, with potential effects on synaptic plasticity and cognition.
Materials and Methods
Experimental Design and Tissue Preparation
Male Sprague-Dawley rats (Charles River Laboratories, Inc, Wilmington, MA) weighing between 200 and 240 g were housed in cages (1 rat per cage) and maintained in environmentally controlled rooms (22°C-24°C), with a 12-hour light/dark cycle. After acclimatization for 1 week on standard rat chow, the rats (n = 5-6 within each group) were exposed to moderate fluid percussion injury (FPI) or sham injury and then maintained on control diet, curcumin diet (Cur; 500 ppm), DHA diet (1.2% DHA, Nordic Naturals, Inc), or DHA+Cur diet (DHA, 1.2%; Cur, 500 ppm) for 2 weeks. The rats were divided into 5 groups: CTL (sham-control diet), FPI, FPI-Cur, FPI-DHA, and FPI-DHA-Cur. The components of control and DHA diets are shown in Table 1. The diets were provided in powder form ad libitum (TestDiet Inc, Richmond, IN) in large bowls. After 2 weeks of feeding postinjury, the tissues were collected for biochemical analysis, including analyses for lipid and protein markers. Our previous studies indicated that learning ability significantly declined after 1 week of FPI, together with molecules involved in plasticity and cogniton. 13 Therefore, we chose 2 weeks of FPI as a suitable postlesion time to assess the effects of DHA and curcumin. The first 2 weeks of TBI are the most critical for the course of the chronic phase of the injury recovery process because the brain is metabolically dysfunctional during this period. 15 All experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals by the National Institutes of Health and were approved by the UCLA Chancellor’s Animal Research Committee. The suffering experienced by the animals and the number of animals used were minimized.
Composition of Experimental Diets.
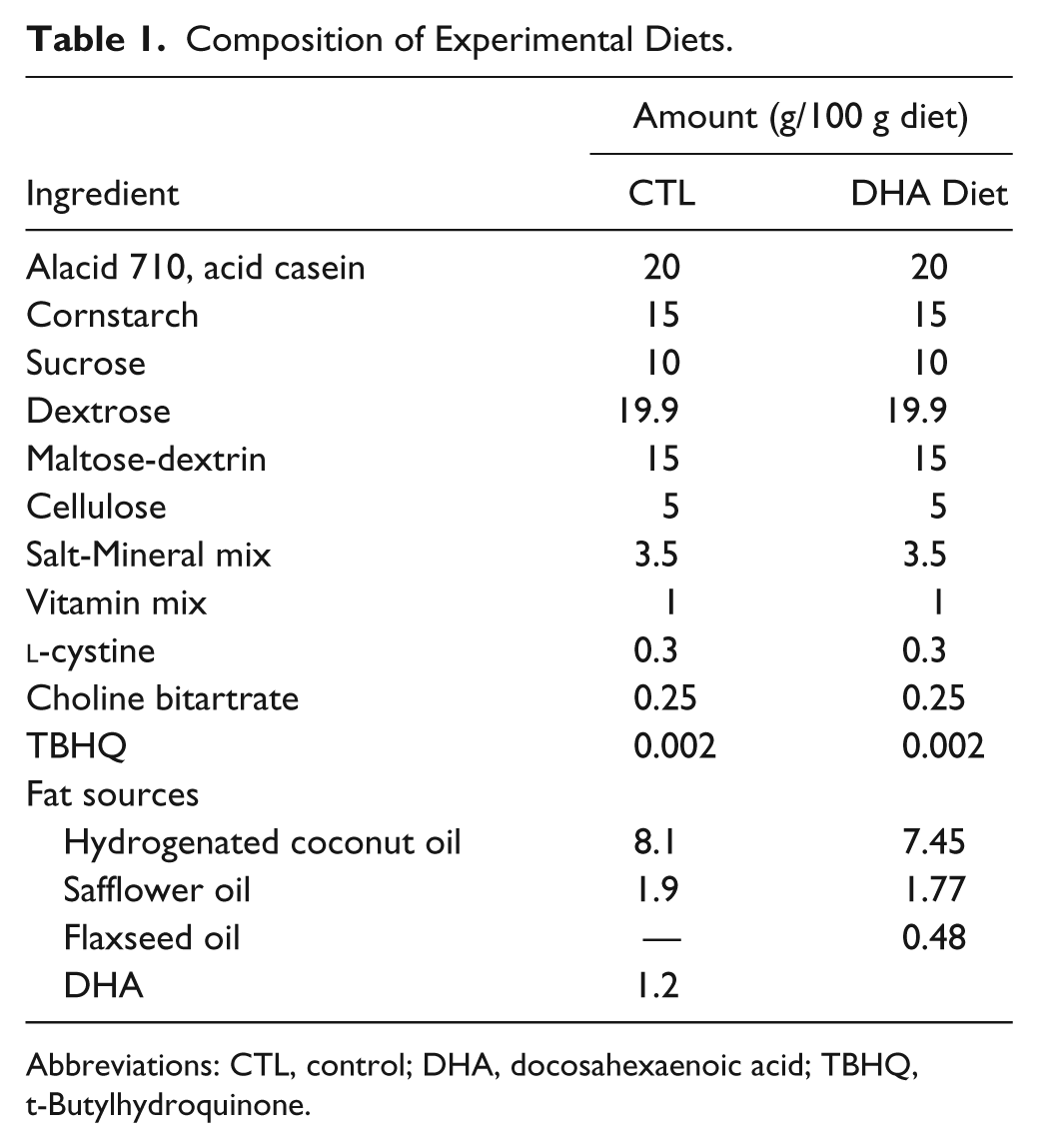
Abbreviations: CTL, control; DHA, docosahexaenoic acid; TBHQ, t-Butylhydroquinone.
Fluid Percussion Injury
The injury was performed as previously described (Wu et al 13 ) with minor modifications. In brief, with the aid of a microscope (Wild, Heerburg, Switzerland), a 3.0-mm diameter craniotomy was made 3.0 mm posterior to the bregma and 6.0 mm lateral (left) to the midline with a high-speed drill (Dremel, Racine, WI). A plastic injury cap was placed over the craniotomy with silicone adhesive and dental cement. When the dental cement hardened, the cap was filled with 0.9% saline solution. Anesthesia was discontinued and the injury cap was attached to the fluid percussion device. At the first sign of hind-limb withdrawal to a paw pinch, a moderate fluid percussion pulse (2.0 atm) was administered. Sham animals underwent an identical preparation with the exception of the fluid percussion. Immediately on responding to a paw pinch, anesthesia was restored, and the skull was sutured. Neomycin was applied on the suture and the rats were placed in a heated recovery chamber for approximately an hour before being returned to their cages.
Cognitive Testing
The cognitive testing was performed in a Barnes maze 1 week postinjury. Briefly, the rats were trained in the Barnes maze with 2 consecutive trials per day for 5 days. Visual cues were prominent and constant. The maze was cleaned between trials to control for odor detection. The rats were placed in the center of the platform with 14 holes. One of the holes had a black box attached, so that the rat could escape into this hole. Each trial lasted until the rat found the hole or for a maximum of 5 minutes. If the rat failed to find the platform in the allocated time, it was gently placed into the box. At the end of each trial, the animals were allowed to rest inside the box for 30 s. The escape latencies to find the box were recorded.
Serum Cholesterol
Serum cholesterol was assayed enzymatically by the ACE cholesterol reagent using the ACE Alera chemistry analyzer (Alfa Wassermann, NJ, USA).
Western Blot
The total proteins from hippocampal tissue were extracted. Protein samples were separated by electrophoresis on an 8% polyacrylamide gel and electrotransferred to a nitrocellulose membrane. Nonspecific binding sites were blocked in Tris-buffered saline, overnight at 4°C, with 2% bovine serum albumin and 0.1% Tween-20. Membranes were rinsed for 10 minutes in buffer (0.1% Tween-20 in Tris-buffered saline) and then incubated with anti-actin, anti-BDNF, anti-p-trkB, anti-4-HNE, anti-FADS2, anti-17β-HSD4 (1:1000, Santa Cruz Biotechnology, Santa Cruz, CA), and anti-4-HHE (1:1000, a gift from Dr Picklo 16 ), followed by antigoat or antirabbit IgG horseradish peroxidase conjugate. After rinsing with buffer, the immunocomplexes were visualized by chemiluminescence using an ECL kit (Amersham Pharmacia Biotech Inc, Piscataway, NJ). The film signals were digitally scanned and then quantified using NIH Image software. Actin was used as an internal control.
Lipid Analysis
Total lipids were extracted from cerebral cortical tissues with a lysis solution of chloroform-methanol (2:1, vol:vol) containing 0.005% butylated hydroxytoluene. The ratio of sample to lysis solution was 1:20 (ie, 100 mg tissue + 2 mL lysis solution). After centrifugation at 9000 rpm for 10 minutes, the chloroform layer was transferred to a 15-mL tube and then mixed with 0.9% saline. After centrifugation again, the chloroform layer was transferred to another 15-mL tube and dried under nitrogen. The dried sample of total lipids was dissolved in hexane and methylated by methanol in the presence of BF3 at 90°C for lipid analysis. Fatty acid profiles were determined by using gas chromatography (GC). The system consisted of model Clarus 500 gas chromatograph (PerkinElmer) with a built-in autosampler. An Elite-WAX column (60 m, 0.32-mm internal diameter, PerkinElmer) was used, with hydrogen as the carrier gas. The GC oven temperature was initially held at 140°C for 2 minutes and raised with a gradient of 5°C/minute to 250°C and held for 10 minutes. The total run time was 34 minutes. The injector and detector were maintained at 250°C and 300°C, respectively. A 1-µL sample of fatty acid methyl esters (FAME) was injected in a split injection mode, with a 100:1 split ratio. Peaks of resolved FAME were identified and quantified by comparison with standards (Supelco 37-component FAME Mix).
Statistical Analysis
For DHA, docosapentaenoic acid (DPA; n-3), and protein levels, the values were converted to percentage of sham control group as presented in bar figures and represented the mean ± standard error of the mean. The Barnes Maze test data were analyzed by repeated-measures ANOVA. The biochemical analysis and learning slope data were analyzed by ANOVA followed by Fisher’s protected least-significance post hoc test. The correlation between DHA and cholesterol was statistically analyzed by linear regression. Statistical differences were considered significant at
Results
Curcumin Potentiates the Action of DHA on Reducing Cognitive Impairment After TBI
Our previous study indicated that n-3 fatty acids supplemented in the diet counteracted learning disability after TBI,
5
and the current studies aim to determine whether curcumin can complement these effects of DHA after TBI. The separate groups of rats were maintained on control diet, Cur, DHA diet, or DHA+Cur diet for 2 weeks, after TBI or sham injury. The learning ability was tested after day 7 postinjury, as described in the Methods section. The latencies to safe box were recorded and analyzed by repeated-measures ANOVA. There was an overall significant difference in learning performance among groups (
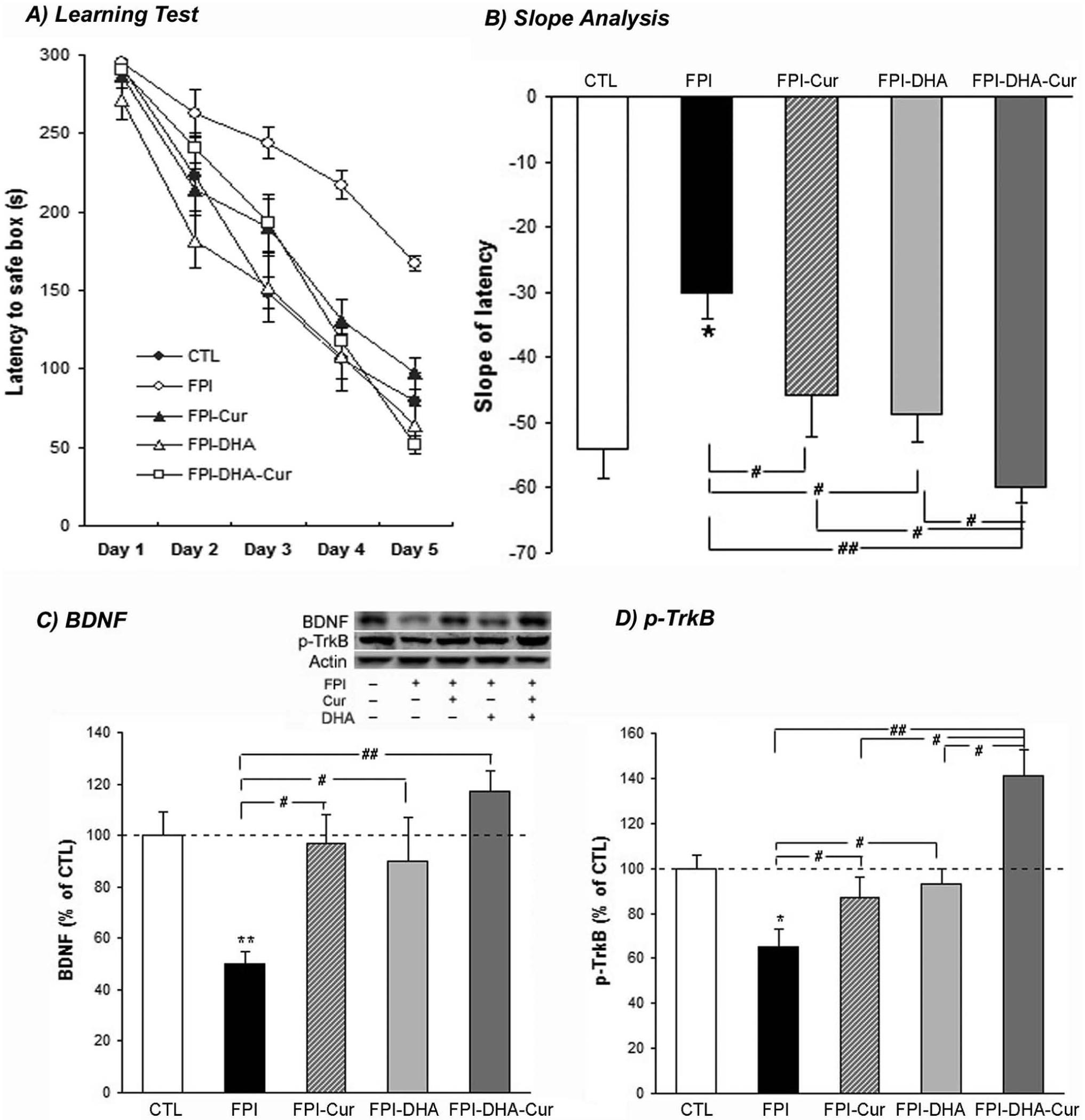
DHA and curcumin supplementation provide protection against learning deficit and synaptic impairment after FPI. A. The latencies to safe box during the Barnes Maze test were significantly higher in the FPI group than in other groups. B. The learning speed (slope of escape latency to find the platform during the Barnes Maze test) was reduced in FPI rats fed on a control diet, which was counteracted after DHA or curcumin treatment. A greater effect in learning speed was observed when DHA and curcumin were combined. C. FPI reduced BDNF levels, which were counteracted by DHA, curcumin, or a combination of both. D. FPI reduced p-TrkB levels, which were counteracted by DHA or curcumin. A greater effect was observed when DHA was combined with curcumin.a
Curcumin and DHA Affect the BDNF System and Cognition
There was a significant group effect on BDNF (
There was a significant group effect on p-TrkB (
Curcumin Preserves DHA in the Plasma Membrane After TBI
The action of TBI and dietary interventions to selectively affect lipid peroxidation in n-6 fatty acids and n-3 fatty acids was assessed by using lipid peroxidation markers 4-HNE and 4-HHE, respectively. It is known that 4-HNE is formed when the n-6 PUFAs, such as arachidonic acid or linoleic acid, are oxidized. 4-HNE has been linked to the pathology of many diseases such as Alzheimer’s disease, atherosclerosis, and cancer. The results showed that there was a significant group effect on 4-HNE (
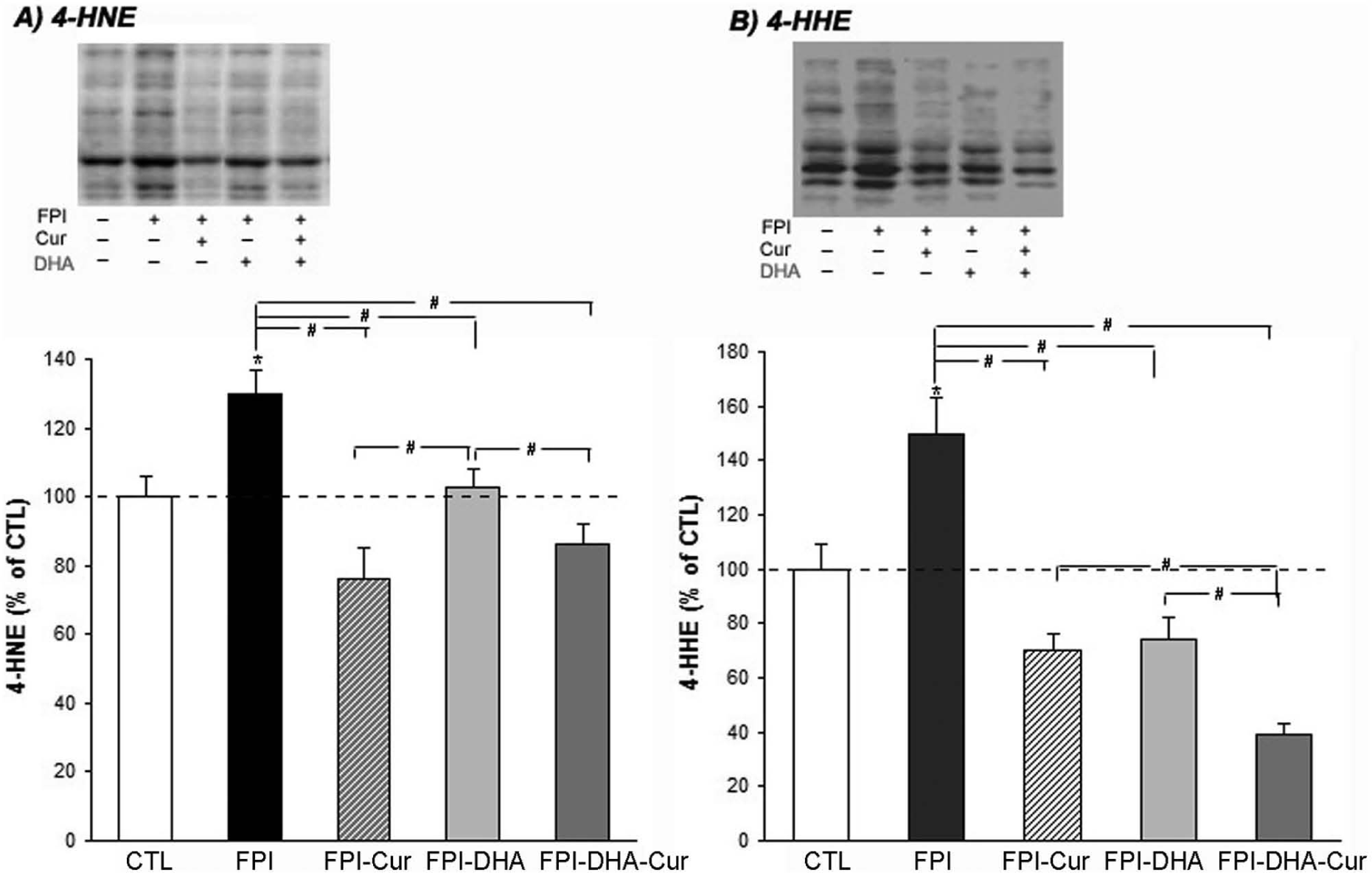
DHA and curcumin supplementation reduce 4-HNE (A) and 4-HHE (B) after FPI. Curcumin, DHA, or DHA and curcumin reduce 4-HNE and 4-HHE levels in FPI rats compared with untreated animals with FPI. A greater reduction was observed when DHA was combined with curcumin compared with separate DHA or curcumin treatment.a
Another lipid peroxidation marker is 4-HHE, which is derived from oxidative damage to n-3 PUFAs. The results showed that FPI also elevated levels of 4-HHE (150% of CTL,
Action of Curcumin Overlaps DHA Pathways and Enhances DHA Stability
FADS2 and 17β-HSD4 are important enzymes involved in the metabolism of DHA from the essential fatty acid α-linolenic acid. The results indicated a significant group effect on FADS2 (F4,25 = 9.481,
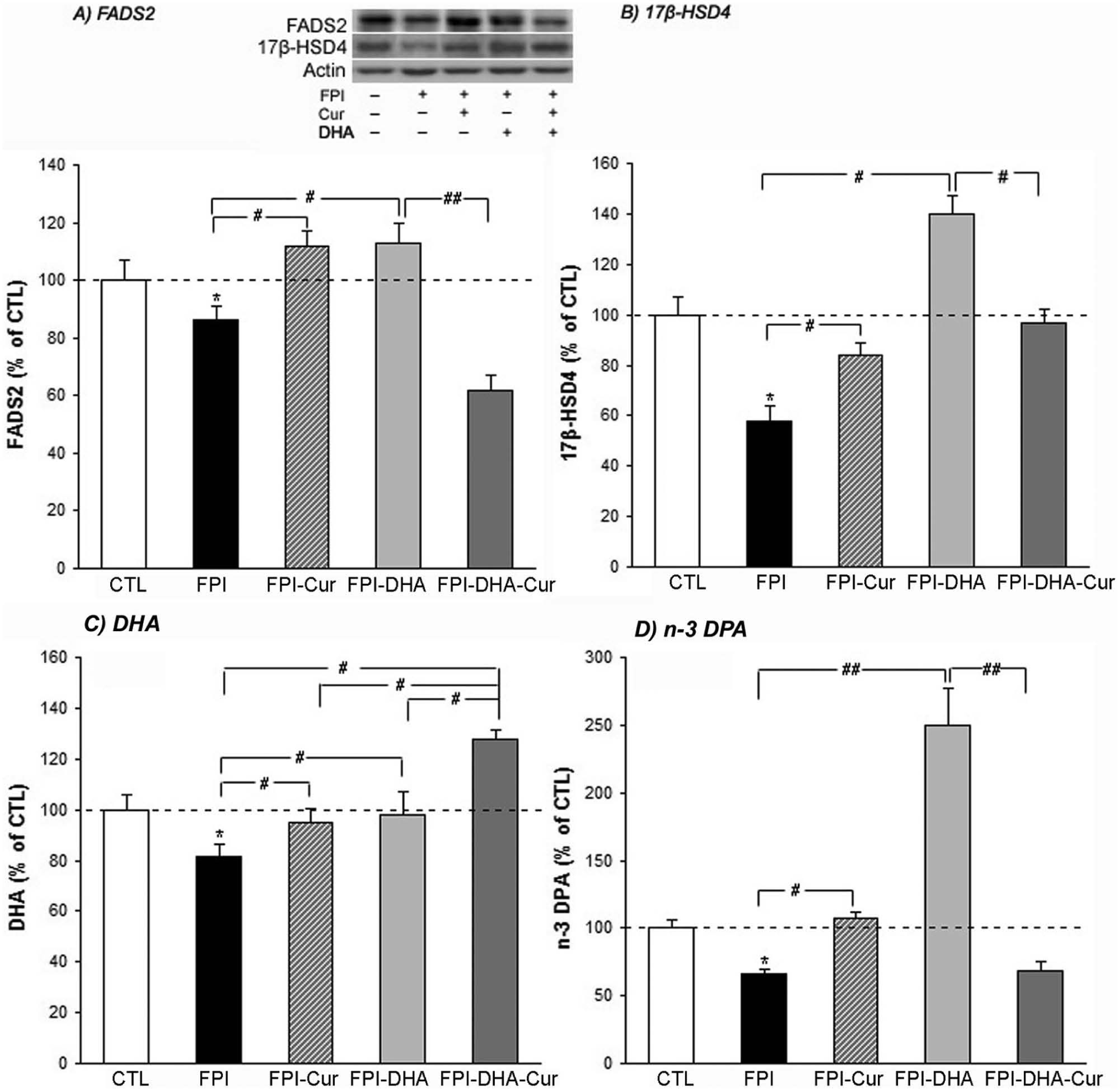
DHA and curcumin regulate enzymes of FADS2 and 17β-HSD4 and the contents of DHA and n-3 DPA after FPI. A. FPI resulted in reduction of FADS2, which was counteracted by DHA or curcumin. However, a reduction of FADS2 was observed when curcumin and DHA were combined. B. FPI resulted in significant reduction of 17β-HSD4, which was significantly reversed by curcumin or DHA. C. FPI resulted in reduction of DHA, which was reversed by a diet of both curcumin and DHA. A greater increase in DHA was observed when DHA and curcumin were combined in the diet. D. FPI also reduced n-3 DPA, which was reversed by both curcumin and DHA. However, a significant reduction was shown when n-3 and curcumin were combined compared with either of them alone.a
The results showed that there was a significant group effect on 17β-HSD4 (
Curcumin Regulates the Content of DHA and n-3 DPA in the Brain
GC was used to determine possible effects of FPI and curcumin on levels of DHA or n-3 DPA in the brain. The results of DHA and n-3 DPA were converted to percentage of controls and are presented as a bar plot. There was a significant group effect on DHA (
Because n-3 DPA (C22:5n-3) is another important n-3 fatty acid that can be a source for making DHA inside the body, we analyzed the n-3 DPA content in the brain with or without FPI. There was a significant group effect on n-3 DPA (
Curcumin Reduces Serum Cholesterol, Which May Reflect Brain DHA Degradation
Among its many functions, cholesterol is considered a degradation product of lipid damage in the membrane.
19
The blood cholesterol levels in FPI groups with or without curcumin, DHA, or a combination of both were measured. Our results showed that FPI increased levels of blood cholesterol (116% of CTL,
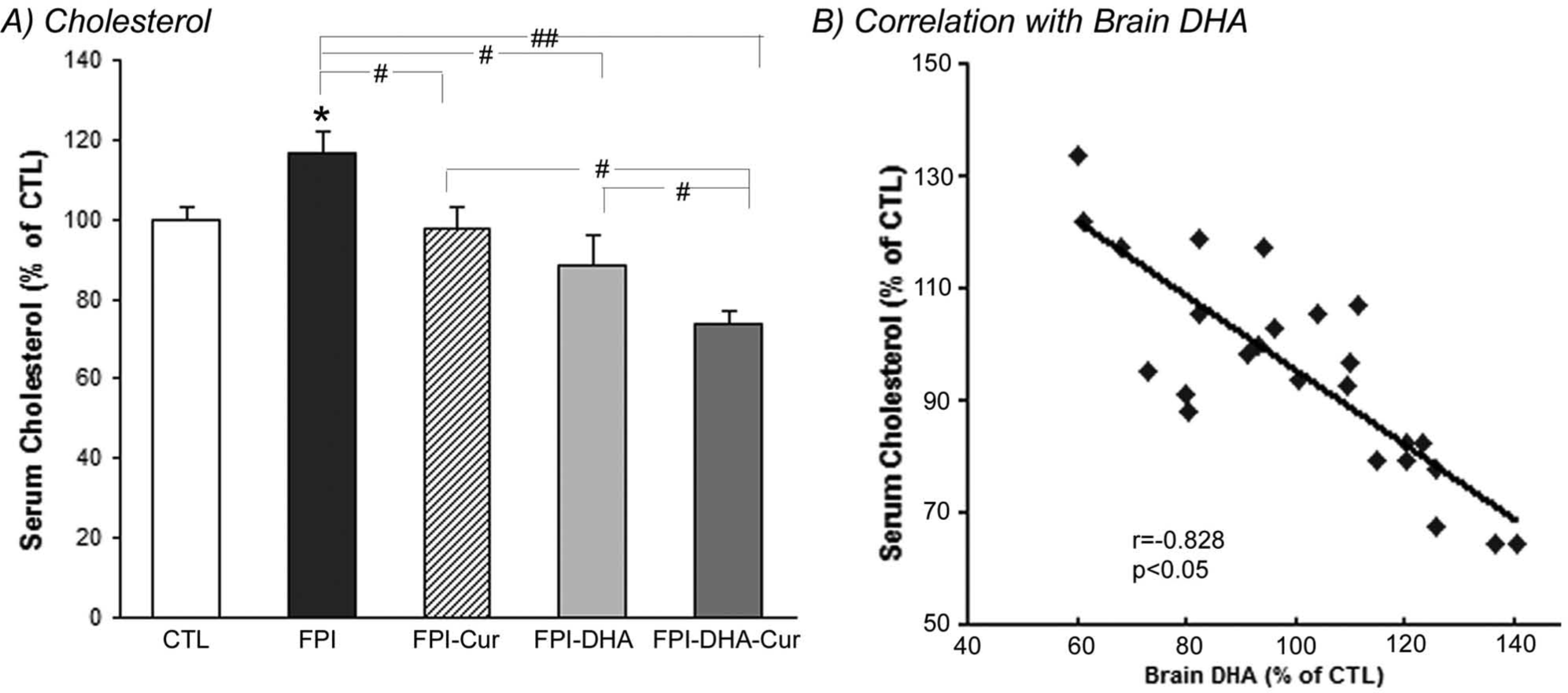
Serum cholesterol (A) and its relationship with brain DHA (B) after TBI. A. FPI resulted in increased cholesterol, which was counteracted by DHA, and a greater reduction of cholesterol was observed when DHA was combined with curcumin. B. Brain DHA level was negatively correlated with serum cholesterol.a
Discussion
This study was performed to explore the possibility that curcumin could complement the action of DHA and contribute to support plasma membrane homeostasis after TBI. The results indicated that curcumin contributed to enhance the action of DHA, protecting against cognitive impairment, and these effects were associated with elevations in the BDNF receptor signaling. This study presents original evidence that curcumin preserves membrane homeostasis by acting on enzymes involved in the metabolism of DHA, which were compromised after TBI (Figure 5). Our findings provide new, important information that can be used as a framework for the design of potential therapies for the treatment of TBI. The overall results are consistent with the notion that dietary manipulations can be used as a strategy to promote neuronal plasticity and function.
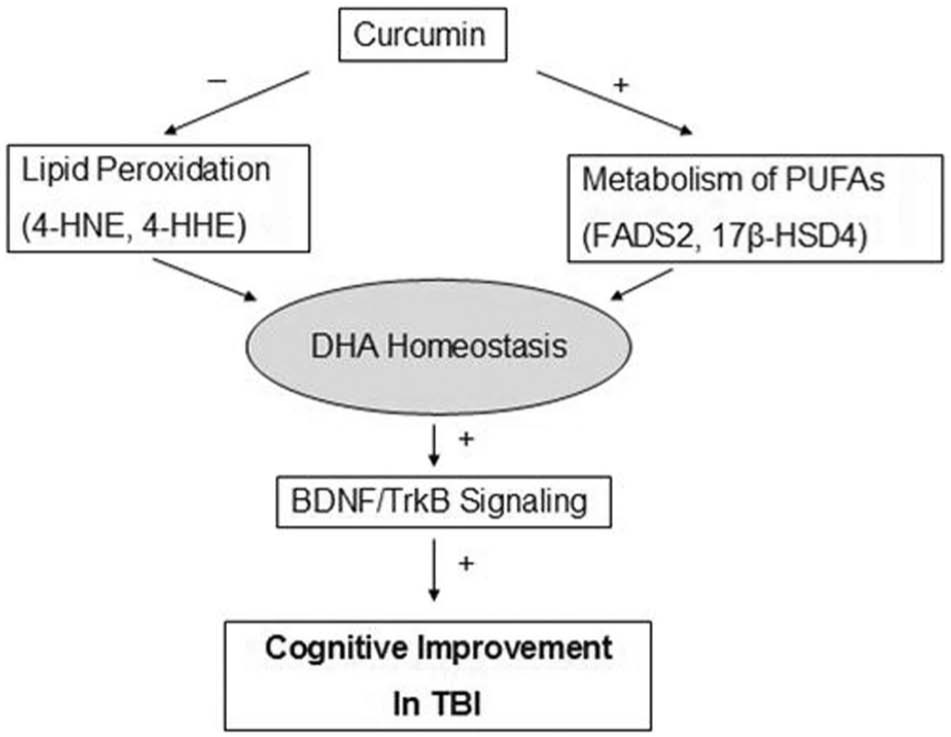
Possible mechanisms underlying the beneficial effects of curcumin on DHA homeostasis and cognition after TBI. Curcumin may preserve brain DHA by protecting against oxidative stress and regulating enzymes involved in the metabolism of DHA. This may contribute to maintain DHA in the plasma membrane after TBI, thereby supporting interneuronal communication through BDNF-TrkB or other signaling systems. These events are important for improving cognitive ability after trauma.
Curcumin Enhances the Effects of DHA on Cognition
Our current results show that curcumin contributes to enhance the effects of DHA on TBI by promoting phosphorylation of the BDNF receptor TrkB in the hippocampus. In particular, we found that the action of curcumin on DHA was also instrumental in counteracting a reduction in p-TrkB after TBI. It has been reported that BDNF-TrkB signaling is positively associated with the cognitive recovery observed in animal models of TBI. In addition, previous evidence indicates that curcumin 10 and DHA 5 counteract TBI-related learning disability by involving BDNF. The combined action of curcumin and DHA results in greater learning performance than that seen with curcumin or DHA alone. These results prompted us to investigate the mechanisms involved in the collaborative action of curcumin and DHA. These studies were focused on the actions of curcumin and DHA on the plasma membrane because this is pivotal for proper neuronal signaling, with the capacity to affect cognition and fundamental mechanisms of functional recovery.
Curcumin Contributes to Preserving DHA in the Membrane After TBI
The brain is highly vulnerable to oxidative damage because of the high content of membrane phospholipids such as DHA and relatively low antioxidant defenses. TBI produces peroxides that give rise to α,β-unsaturated aldehydes such as 4-HNE and 4-HHE, leading to subsequent degradation of membrane phospholipids.6,20 These products bind to proteins, particularly cysteine, histidine, or lysine residues, and can cause deleterious effects in cells, including inhibition of DNA synthesis, disturbance in calcium homeostasis, and inhibition of mitochondrial respiration. The results showed that TBI increased levels of 4-HNE and 4-HHE, which have been associated with the peroxidation of n-6 and n-3 PUFAs, respectively. In particular, results showing that curcumin reduced the levels of 4-HHE, an indicator of oxidized n-3 fatty acids, provide evidence for a protective action of curcumin on DHA oxidation. The fact that the combination of DHA and curcumin elevated levels of DHA beyond those observed with the sham control diet suggests that dietary curcumin has some capacity to influence DHA metabolism and homeostasis.
The metabolism of DHA is regulated by several enzymes, of which FADS2 and 17β-HSD4 play an important role. TBI reduced levels of DHA in conjunction with FADS2 and 17β-HSD4. The fact that treatment with curcumin alone counteracted the reduction of both enzymes suggests potential mechanisms by which curcumin maintains homeostatic levels of DHA after TBI. The combination of curcumin and DHA did not yield higher levels of the enzymes, and even FADS2 was reduced, which suggests that DHA may produce negative feedback (see Figure 3).
It is known that n-3 DPA has important biological effects—that is, it can reduce age-related decline in cognition. 11 Furthermore, n-3 DPA is an intermediary product between eicosapentaenoic acid (EPA) and DHA and can be retroconverted to EPA but has limited conversion to DHA. Both enzymes, FADS2 and 17β-HSD4, are involved in these processes. Our findings indicate that curcumin counteracted the TBI-related reduction in n-3 DPA. The fact that n-3 DPA was reduced when curcumin is combined with DHA (see Figure 3) suggests that curcumin may promote the conversion of n-3 DPA to DHA by regulating enzymes FADS2 and 17β-HSD4, even though the body has limited conversion of n-3 DPA to DHA. 11
It is important to consider that alternative mechanisms could be responsible for benefit to the injured brain from the combination of DHA and curcumin other than those discussed above. For example, the combination of both nutrients has been reported to produce anti-inflammatory action 21 and support insulin/trophic factor signaling. 22 These actions can be significant, based on information that the TBI pathobiology is associated with acute immune inflammatory events.23 -25 It is also significant that TBI elevated levels of cholesterol in blood, whereas curcumin potentiated the effects of DHA on reducing levels of peripheral cholesterol (see Figure 4A). Our results also showed that blood cholesterol is negatively associated with brain DHA levels (see Figure 4B). The current view is that peripheral cholesterol may be a subproduct of membrane phospholipid degradation. 19 According to this view, the enhanced actions of curcumin and DHA in reducing cholesterol levels could be interpreted as preservation of levels of phospholipids in the plasma membrane by this dietary treatment. An alternative explanation is that curcumin and DHA may contribute to reduce inflammation associated with the action of cholesterol in the pathology of TBI.
Curcumin Contributes to the Action of DHA to Foster Plasticity After TBI
This study examined the possibility that cognitive dysfunction related to TBI is the result of membrane dysfunction, which disrupts neuronal signaling through membrane-embedded receptors. Results indicate that curcumin collaborates with DHA to stabilize pathways important for maintaining membrane homeostasis and synaptic plasticity, underlying cognitive abilities. In particular, curcumin makes the action of DHA more efficient by affecting enzymes involved in the metabolism of omega-3 fatty acids such as FADS2 and 17β-HSD4 (Figure 5). The overall evidence seems to suggest the existence of intrinsic adaptive mechanisms in the maintenance of proper levels of omega-3 fatty acids in the membrane, which are crucial for supporting learning ability after TBI (see Figure 1). The effects of the DHA diet and curcumin on cognitive enhancement were consistent with enhanced elevations in BDNF receptor signaling (see Figure 1). These results are important for the design of therapeutic strategies using diet to counteract the effects of TBI and other neurological disorders. In particular, our results provide a mechanistic framework to explain how different components of the diet can collaborate at the molecular level to influence brain plasticity and mechanisms of functional recovery. We assessed the effects of DHA and curcumin up to 2 weeks after TBI because this is the most critical period for the course of injury recovery because the brain is metabolically dysfunctional during this time. 15 Accordingly, we expect that this treatment can have important consequences for the chronic phase of recovery. However, further studies are required to document this assumption. We expect that our 2-week paradigm after TBI can serve as a baseline for the planning of dietary interventions to boost functional recovery. A crucial advantage is that these interventions are noninvasive and devoid of side effects, so that they can be easily prolonged and accommodated according to individual needs. Another advantageous aspect of our results is that this dietary treatment can be used to complement the action of other interventions, such as rehabilitation for optimal functional recovery.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by NIH award NS50465.
