Abstract
Keywords
Introduction
Even after severe cervical or thoracic spinal cord injury (SCI), the human lumbosacral neural circuitry can process sensory information and generate rhythmic motor output to paralyzed leg muscles in response to assisted stepping movements 1 or epidural spinal cord stimulation (eSCS). 2
Peripheral, mechanical stimulation of the receptors in lower-limb muscles, tendons, joints, and skin during therapist-assisted treadmill stepping with body-weight support (BWS) provides step-phase synchronized activation to the lumbosacral spinal circuitry. 3 In motor-complete SCI individuals, the circuitry can react to these sequences of feedback by generating motor output to the legs timed to specific phases of the gait cycle.4,5 The produced electromyographic (EMG) signals include rhythmic activity, as well as rhythmically triggered clonus and stretch reflexes.6-8 Robotic-driven gait orthoses (DGOs) that provide assistance during BWS treadmill stepping produce similar EMG patterns. 9 Afferent information related to lower-extremity load 1 and step frequency 6 can be processed by the human lumbosacral spinal cord 10 to modulate the generated rhythmic output. Another source of sensory input important in the control of stepping is the hip extension angle.5,9 Exaggerated hip extension at the end of stance may trigger a flexor synergy and initiate swing of the paralyzed limb in SCI individuals.5,11 Different explanations for the rhythmic EMG activities produced during assisted treadmill stepping have been proposed. Some observations have suggested that they are stretch reflexes, cyclically entrained by the externally imposed movements.7,12,13 Other findings have implied that the lumbosacral circuitry acts like a processing interface that integrates the multimodal step-induced sensory information and reacts by generating step-phase-dependent motor outputs.1,6 The appropriate combination of sensory cues may activate flexion–extension synergies. 11 The direct involvement of spinal pattern generators for locomotion has also been put forth.14-16
Afferents of similar modalities as those activated peripherally during assisted treadmill stepping, that is, large- and medium-diameter myelinated fibers, are also electrically depolarized by eSCS of the lumbar cord, proximally within the posterior roots.17,18 Yet eSCS generates tonic inputs of constant frequency that are highly synchronized across spinal cord segments and sides. eSCS of the lumbar cord at frequencies of 25 to 50 Hz can produce rhythmic EMG patterns in motor-complete SCI individuals in the absence of step-specific feedback information, revealing spinal rhythm generating circuitry in humans.2,19,20
Case studies showed that the lumbar spinal cord can integrate inputs provided by stepping and eSCS to generate rhythmic motor patterns that are not produced in the presence of either input alone.18,21-23 Delivering multisegmental driving inputs to the human lumbar spinal cord has been considerably simplified by transcutaneous spinal cord stimulation (tSCS).24-26 Preliminary data suggested that 20 to 50 Hz tSCS can modify rhythmic EMG activity generated during assisted treadmill stepping.22,27-29
The aim of the present exploratory study was to investigate rhythm generation by the lumbar spinal neural circuitry, largely deprived of supraspinal influence, in response to phasic step-related sensory feedback and tonic tSCS when provided alone or in combination. We applied DGO-guided treadmill stepping with different speeds and hip-extension conditions without and with concomitant 30-Hz tSCS in subjects with clinically classified complete SCI. We presumed that EMG activity in individual muscles directly related to specific mechanical events 7 of DGO-stepping—or to their variations—would suggest the activation of reflex circuits. Changes in the EMG patterns across muscles, especially when applying exaggerated hip-extension,5,11 would hint at more complex mechanisms of spinal rhythm generation. We further examined whether tSCS would provide additional excitatory drive as well as activate spinal rhythm generating networks, 30 reflected in an immediate increase of the number of rhythmically activated muscles during DGO-stepping.21,23 Synergistic effects of sensory feedback cues and tonic drive in spinal rhythm generation may give a perspective for new therapeutic approaches to promote recovery of function even after severe SCI.
Methods
Subjects
We studied 4 subjects (Table 1) with traumatic, chronic motor- and sensory-complete SCI, classified as American Spinal Injury Association Impairment Scale (AIS) “A” using the International Standards for Neurological Classification of SCI. 31 The participants were otherwise healthy adults with preserved stretch and cutaneomuscular lower limb reflexes; preserved lower limb range of motion allowing passive stepping motions, and stability for weight bearing; no implants at the site of stimulation. None of the subjects had participated in treadmill training prior to this study. Medications were not changed for the study (subject 1, Baclofen; subjects 2 and 3, Gabapentin). Informed consent was obtained as approved by the institutional human research committee at Shepherd Center, Atlanta, GA, USA.
Demographic Data and Neurological Classification of Spinal Cord Injury a .
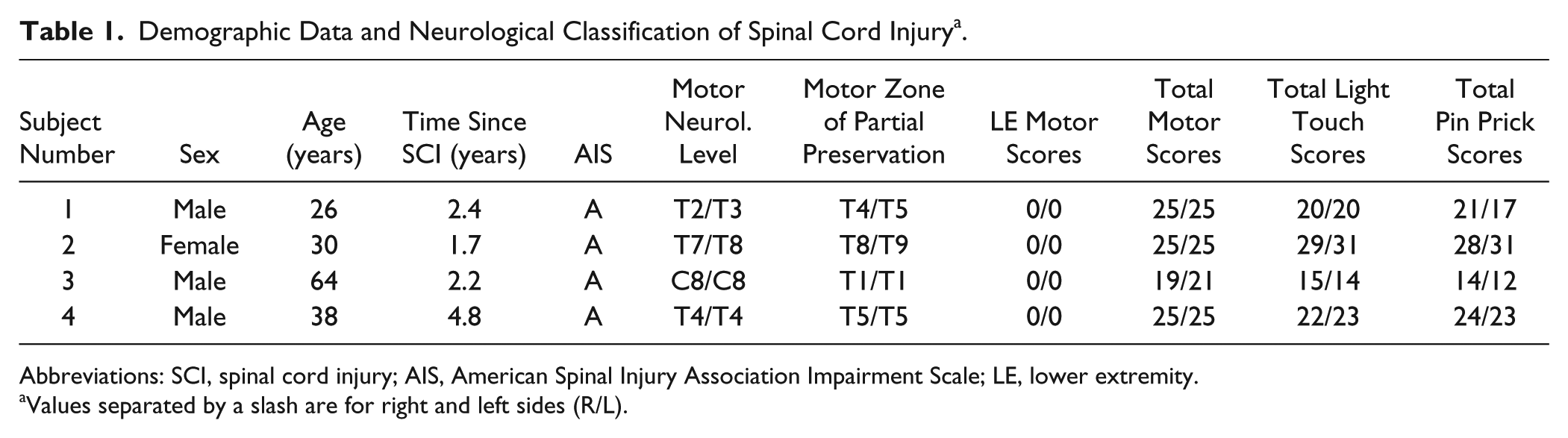
Abbreviations: SCI, spinal cord injury; AIS, American Spinal Injury Association Impairment Scale; LE, lower extremity.
Values separated by a slash are for right and left sides (R/L).
Data Acquisition
Surface-EMG activity was recorded bilaterally from quadriceps (Q), hamstrings (Ham), tibialis anterior (TA), and triceps surae (TS) in all, and additionally from tensor fasciae latae (TFL) in subjects 1 to 3 with pairs of silver–silver chloride surface electrodes (Intec Medizintechnik GmbH, Klagenfurt, Austria) with an interelectrode distance of 3 cm. 32 A pair of recording electrodes was placed on the right side of the trunk to monitor stimulus artifacts. Abrasive paste (Nuprep, Weaver and Company, Aurora, CO) was used for skin preparation to reduce contact resistance. EMG signals were amplified using a poly-EMG system (Phoenix, EMS-Handels GmbH, Korneuburg, Austria) set to a gain of 502 with a bandwidth of 20 to 500 Hz and digitized at 2048 samples per second and channel.
Robotic-Driven Gait Orthosis
The LokomatPro (Hocoma AG, Volketswil, Switzerland) DGO was used to assist treadmill stepping.33,34 Subjects were fitted with a harness connected to the BWS system, set to unload them by 60%. A “slow” and a “fast” step frequency of 0.86 and 1.12 steps per second, respectively, was tested. Treadmill-belt speed was adjusted according to the subjects’ leg lengths and varied between 1.7 and 1.8 km/h for the slow step frequency and between 2.3 and 2.5 km/h for the fast step frequency. At both step frequencies, stepping with neutral hip range and with exaggerated hip extension at the end of stance (hip extension increased by 5°), without changing the total hip and knee ranges of motion, was tested.
Stimulation
tSCS was applied through a coupled pair of round (Ø = 5 cm) self-adhesive electrodes (Schwa-medico GmbH, Ehringshausen, Germany) placed over the T11 and T12 spinous processes, 1 cm apart on each side of the spine24,35 (Figure 1A) and connected to the negative output of the stimulator. The positive output was connected to a pair of rectangular skin electrodes (each 8 × 13 cm, Schwa-medico GmbH) covering the abdomen. The constant current stimulator (DS7AH, Digitimer Ltd, Hertfordshire, England) produced rectangular, monophasic pulses of 1 ms in duration, and was triggered by an external pulse generator (DG2A, Digitimer Ltd) to deliver single and paired pulses, or tonic 30-Hz stimulation. Since the pulses generated were not charge-balanced we limited the stimulation duration and monitored the skin condition.
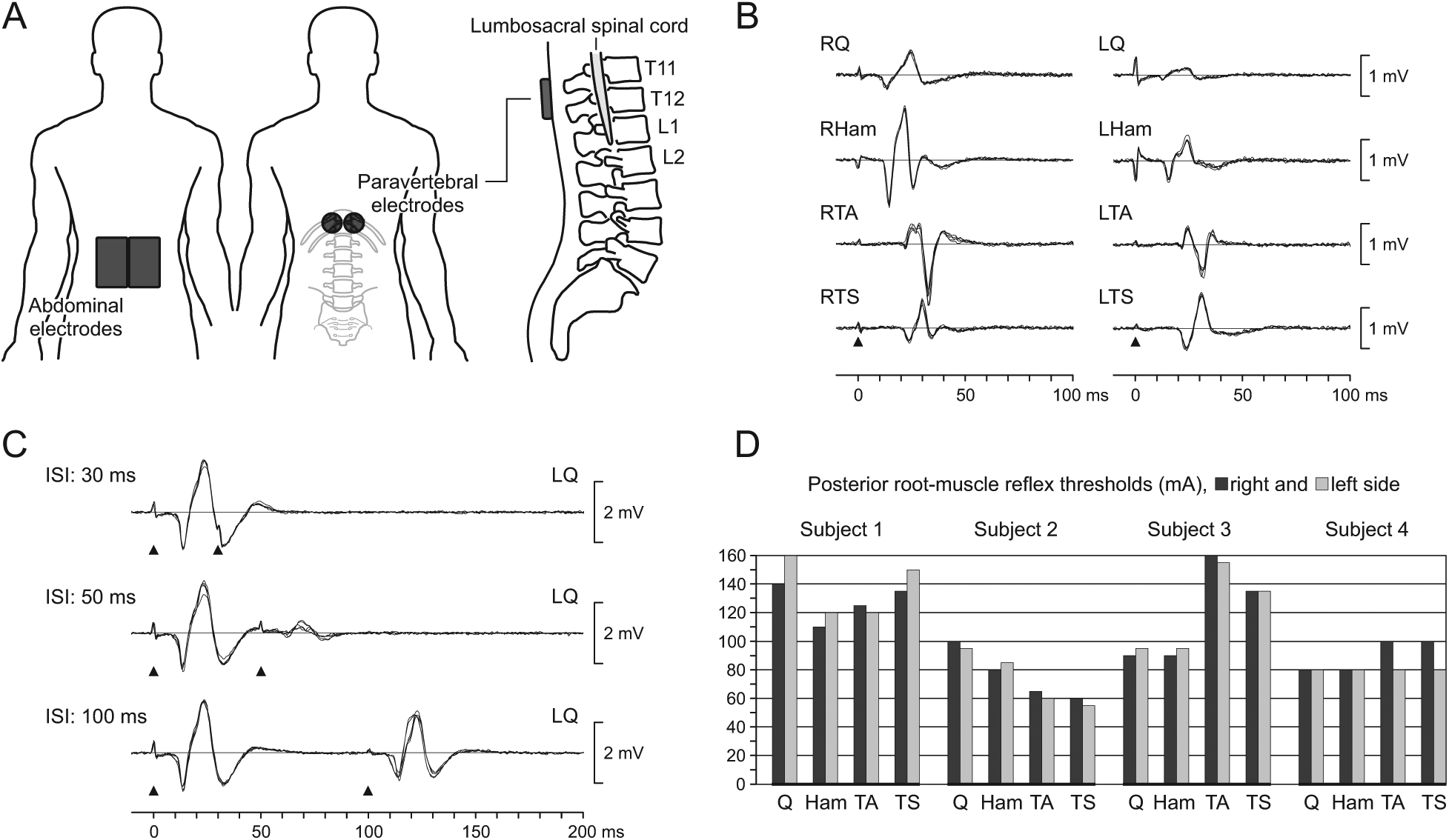
Transcutaneous posterior root stimulation. (A) Paravertebral and abdominal skin electrode placement. (B) Posterior root-muscle reflexes evoked in right and left quadriceps (Q), hamstrings (Ham), tibialis anterior (TA), and triceps surae (TS) verify the effective paravertebral electrode position; responses to 5 stimuli are shown superimposed; subject 1. (C) Postactivation depression of the evoked responses revealed by paired stimulation (ISI, interstimulus interval) confirms stimulation of afferent structures, exemplarily shown for left Q; subject 3. Black arrowheads mark the timing of stimulus application. (D) Posterior root-muscle reflex thresholds in the standing position with 60% body weight support.
Study Protocol
The paravertebral electrode positioning was first tested in the supine position by the elicitation of posterior root-muscle (PRM) reflexes 24 in Q, Ham, TA, and TS as a measure for the stimulation of L2–S2 posterior-root afferents (Figure 1B).18,36 Subsequently, subjects were suspended in the DGO and posterior-root stimulation was reverified while standing with 60% BWS and manual assistance to hold limb-extension. Afferent stimulation was confirmed by the presence of postactivation depression using a paired-pulse paradigm with interstimulus intervals of 30 ms, 50 ms, and 100 ms (Figure 1C).25,37,38 The PRM reflex threshold of each lower limb muscle studied was determined by increasing stimulation intensity in 5-mA increments (Figure 1D).
The stepping protocol included, with the stimulation off, the recording of 20 steps each at the slow and fast step frequencies with neutral hip range and then with the exaggerated hip extension. Subsequently, in the standing position with 60% BWS, 30-Hz stimulation was applied with slowly increasing intensity, individually targeting the level of the highest PRM-reflex threshold across muscles (see Figure 1D). In subject 1, tonic stimulation at such level produced a feeling of tightness caused by the neuromuscular activation of the paraspinal and abdominal muscles and was hence reduced. In subjects 3 and 4, stimulation was increased beyond the initially targeted level, because 30-Hz stimulation at the intensities effective to evoke PRM reflexes when single stimuli were applied (see Figure 1D) failed to generate output in some muscles, probably due to interindividual differences in postactivation depression introduced by the repetitive stimulation.24,25 The final, individually set intensities amounted to, subject 1, 140 mA; subject 2, 100 mA; subject 3, 170 mA; and subject 4, 125 mA; and produced EMG activity in at least one lower limb muscle group per side in all subjects. The stepping protocol was then repeated in the presence of tonic 30-Hz stimulation with unchanged intensity.
Signal Processing
Data were analyzed offline using Matlab R2012a (The MathWorks, Inc, Natick, MA). Stimulus artifacts superimposed during 30-Hz stimulation on the recorded physiological EMG activities were removed, as follows, prior to further analysis. The recordings from the trunk-located electrode pair were used to trigger a blanking interval of 2 to 5 ms, beginning with the leading edge of the stimulus. The length was adjusted to cover any die-away effects following the falling stimulus edge as visible in the different EMG channels. The derived sequence of blanking intervals was applied to the EMG recordings by setting the traces to zero. This had the effect of removing 6% to 15% of the EMG data.
Data Analysis and Statistics
For each of the 4 stepping conditions, 10 consecutive gait cycles per leg were analyzed. Stance and swing phases were identified using the heel-strike and toe-off information provided by the DGO. Activity of each of the 8 studied muscles was calculated as root mean square (RMS) value of the EMG for total gait cycles. All values obtained were baseline-corrected by subtracting the RMS calculated from 5-second periods obtained from the end of each recording, when subjects were brought into an upright position without ground contact and stimulation was turned off.
Effects of step frequency and hip-movement condition were described per muscle by comparing the RMS values of the respective 10 consecutive gait cycles and analyzed using 2-way factorial analyses of variance, independently for the stimulation-on and -off conditions. Group effects of stimulation and muscle group on the mean EMG output of the 10 gait cycles were evaluated with a mixed generalized linear model with each leg as a single case and gait parameters as repeated measures. Pair-wise post hoc tests were performed and Bonferroni corrected. Model assumptions were checked and an α-error of
Results
Motor Output During Supported Standing
No EMG activity was produced in subjects 1 and 3 during standing with 60% BWS and manually assisted knee-extension. In subject 2, there was an irregular occurrence of clonic motor unit firing in the RTA. In subject 4, repetitive motor unit firing with occasional bursting was detected in the RTA.
Motor Output During Robotic-Driven Stepping
In subject 1, DGO-stepping induced low-amplitude EMG activity in RHam, LHam, and LTS. Ham activation occurred during the hip-flexion and concomitant knee-extension segments of the swing phases and was increased by the faster step rate and decreased when stepping with exaggerated hip extension (all
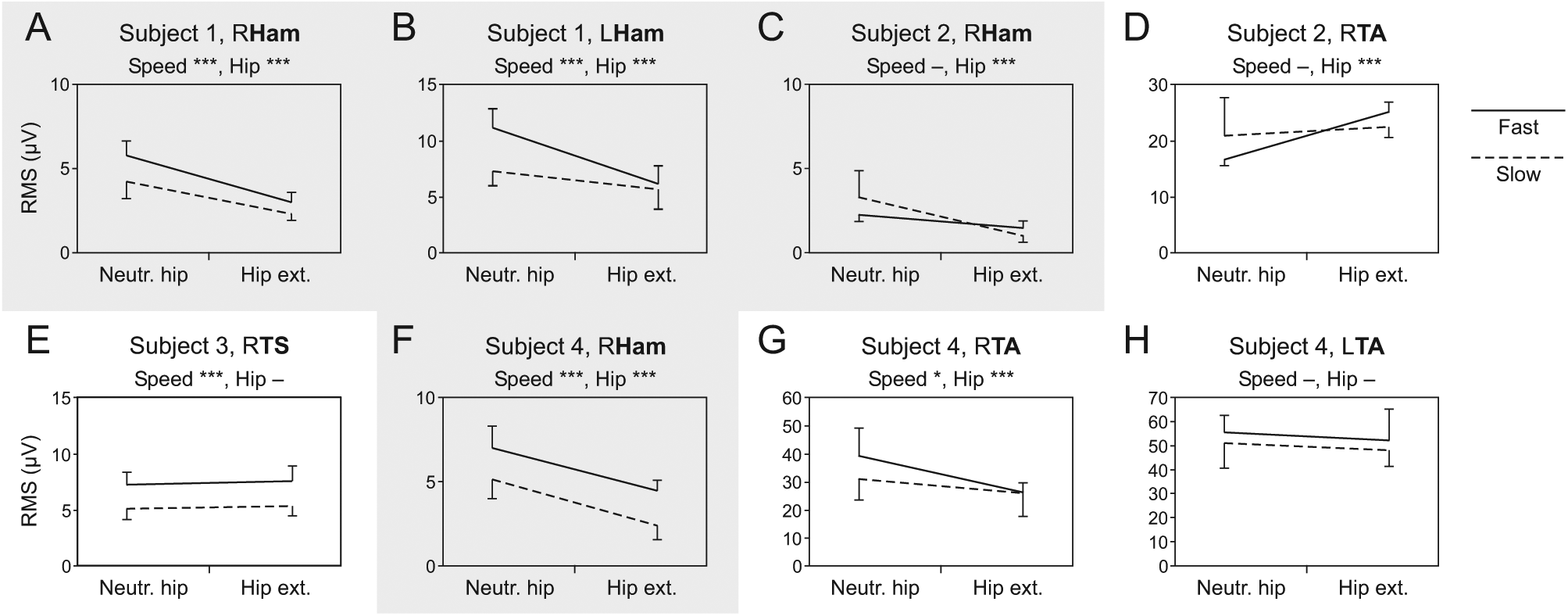
Leg muscle activity produced by sensory feedback during robotic-driven treadmill stepping. (A-H) Average EMG RMS values per gait cycle with SD produced during stepping with neutral hip movement range (Neutr. hip) and hip extension increased by 5° (Hip ext.) at a slow (0.86/s) and fast (1.12/s) step frequencies. Nonparallel dashed and solid lines indicate interaction effects between hip-movement condition and step frequency. Ham, hamstrings; TA, tibialis anterior; TS, triceps surae. Gray background results derived from Ham. Asterisks indicate significant main effects (*,
In subject 2, low-amplitude EMG activity was recorded from RHam during swing phases. RTA activation appeared as clonus during the stance and early swing phases. EMG activity in RHam decreased with exaggerated hip extension (
Subject 3 produced the lowest level of EMG activity of all subjects. Some stance-phase synchronized activity was observed in RTS that increased with faster stepping (
Subject 4 developed low-amplitude activity in RHam during swing and higher-amplitude clonic EMG activity in both TA during the respective late stance phases and stance-to-swing transitions. RHam activity increased with the higher step frequency (
For the group, 33 of 128 muscles (4 stepping conditions × 4 subjects × 8 muscles) or 26% responded to DGO-stepping. Among the responses, 12 were stance-phase initiated clonic activity in the TA muscles of 2 subjects. No activity was observed in Q or TFL. The variation of step frequency and hip-movement condition did not change the distribution of activity or recruit additional muscles, except for LTS in subject 1.
Thirty-Hertz tSCS Effects on Motor Output During Supported Standing
Tonic 30-Hz tSCS produced rhythmic EMG activity in lower limb muscles without step-related sensory feedback in 2 of the 4 subjects. These EMG patterns were series of stimulus-time locked PRM reflexes with amplitude modulation appearing as burst-like envelopes. In subject 1, stimulation produced rhythmic modulation in RHam and simultaneously, but with smaller amplitude, in LHam with an average cycle frequency of 0.41 Hz (Figure 3). In subject 3, 30-Hz tSCS generated synchronous rhythmic modulation in LQ and LTA, and to some extent in the same muscle groups of the right side. The rhythmicity was more erratic and inconsistent than in subject 1 and had an average cycle frequency of 0.36 Hz. In other muscles (13 of 32), the stimulation produced series of PRM reflexes in the form of a tonic motor output with unsystematic modulation. In the remaining 13 cases, no activity was produced.
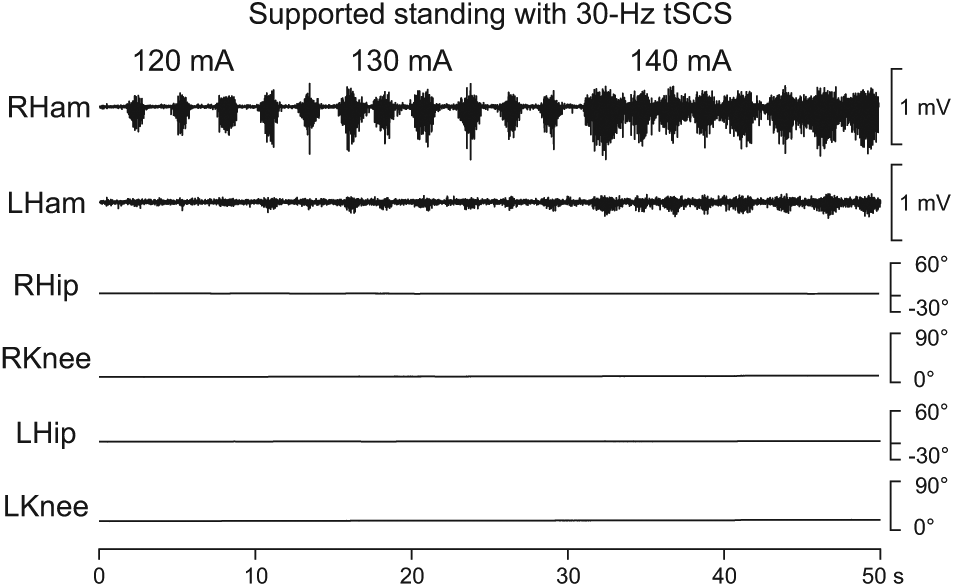
Rhythmic EMG activity produced by 30-Hz tSCS in the standing position with manually supported knee extension and 60% body weight support; right and left hamstrings (Ham), subject 1. Hip and knee angle data confirm static conditions; stimulation intensities are shown above the EMG traces.
Thirty-Hertz tSCS Effects on Motor Output During Robotic-Driven Stepping
EMG activity produced by DGO-imposed stepping was either augmented or suppressed when tonic tSCS was added, and rhythmic activity could be generated in muscles that had not responded to stepping alone (Figure 4A). These effects could be found concomitantly in a single leg. All motor outputs evoked by tSCS were amplitude-modulated PRM reflexes. Figure 4A(i) shows an example of swing-phase synchronized burst-like modulation produced in Ham that was not activated during treadmill stepping alone. Figure 4A(ii) illustrates rhythmic activity generated in Ham by 30-Hz tSCS that required several steps to synchronize to the externally imposed step frequency. Figure 4A(iii) displays tSCS-induced, inconsistently modulated EMG activity in Ham. Figure 4A(iv) shows an example of rhythmic activity, modulating with a frequency of 0.37 to 0.38 Hz, that did not synchronize to the DGO-imposed rhythm cycle frequency. Such autonomously modulating burst-like activity produced by tSCS was found in a single muscle (LQ, subject 3, all stepping conditions). Overall, 30-Hz tSCS had an enhancing or inducing effect on the motor output (Figures 4A(i)-(iv)) in 58 of the 128 cases (45.3%). Attenuation of motor activity during tSCS was observed in 13 cases (10.2%), including suppression of clonus in RTA of subject 2 in all stepping conditions (Figure 4A(v)). In another 4 cases (3.1%; RTA of subject 4), stimulation did not alter the amount of generated motor output, but modified its pattern (Figure 4A(vi)). There, the clonic EMG activity during late stance produced by DGO-stepping alone was replaced by PRM reflexes that were more frequently evoked during swing. Finally, in 53 cases (41.4%), stimulation did not generate motor outputs (Figure 4A(vii)). It is noteworthy that tSCS failed to elicit activity only in those muscles that did not respond to DGO-stepping alone either. Furthermore, in these nonresponding muscles, the applied stimulus intensity was on average 1.19 ± 0.26 times their respective PRM reflex thresholds, while it was 1.52 ± 0.27 times the thresholds in the 58 cases with an enhancing or inducing effect of tSCS.
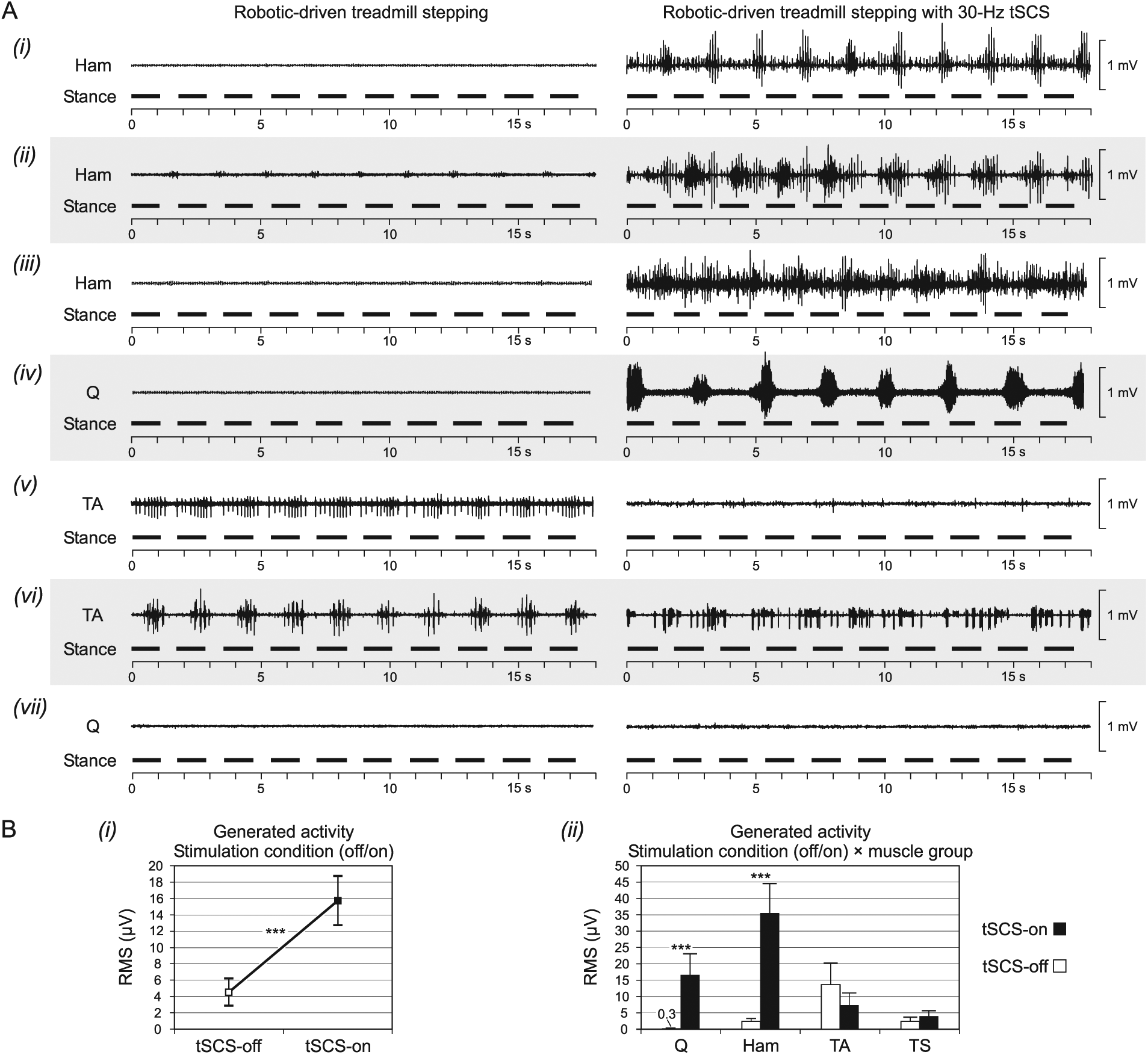
Modification induced by 30-Hz tSCS during robotic-driven treadmill stepping. (A) Exemplary EMG patterns and their changes by stimulation. Stepping parameters in all: 60% body weight support, 1.12/s step frequency, hip extension increased by 5°. EMG derived from, (i) and (vi), subject 4; (ii), subject 1; (iii) and (iv), subject 3; (v) and (vii), subject 2. (B) Group effects of (i) stimulation and (ii) muscle group on the EMG output. Q, quadriceps; Ham, hamstrings; TA, tibialis anterior; TS, triceps surae. ***denote highly significant effects,
Across subjects, muscles and stepping conditions, 30-Hz tSCS increased the EMG RMS values per gait cycle by a factor of 3.5 (
Varying step frequency and hip-movement condition under tSCS modified the amount of EMG activity to different degrees across muscle groups as well as in a given muscle group across subjects (Figure 5). These complex relationships are well reflected in the results derived from Ham. Across step-frequencies, stepping with exaggerated hip extension significantly decreased the activity in 4 (eg, Figure 5A-C) but did not significantly alter it in the remaining 3 responding Hams (eg, Figure 5F) and thus did not have the consistent effect observed in the stimulation-off condition. Across hip-movement conditions, increasing the step frequency significantly attenuated the activity in 2 Hams (eg, Figure 5A), did not significantly alter its amount in 3 Hams (eg, Figure 5B and C), and significantly increased the output in 2 Hams (eg, Figure 5F).
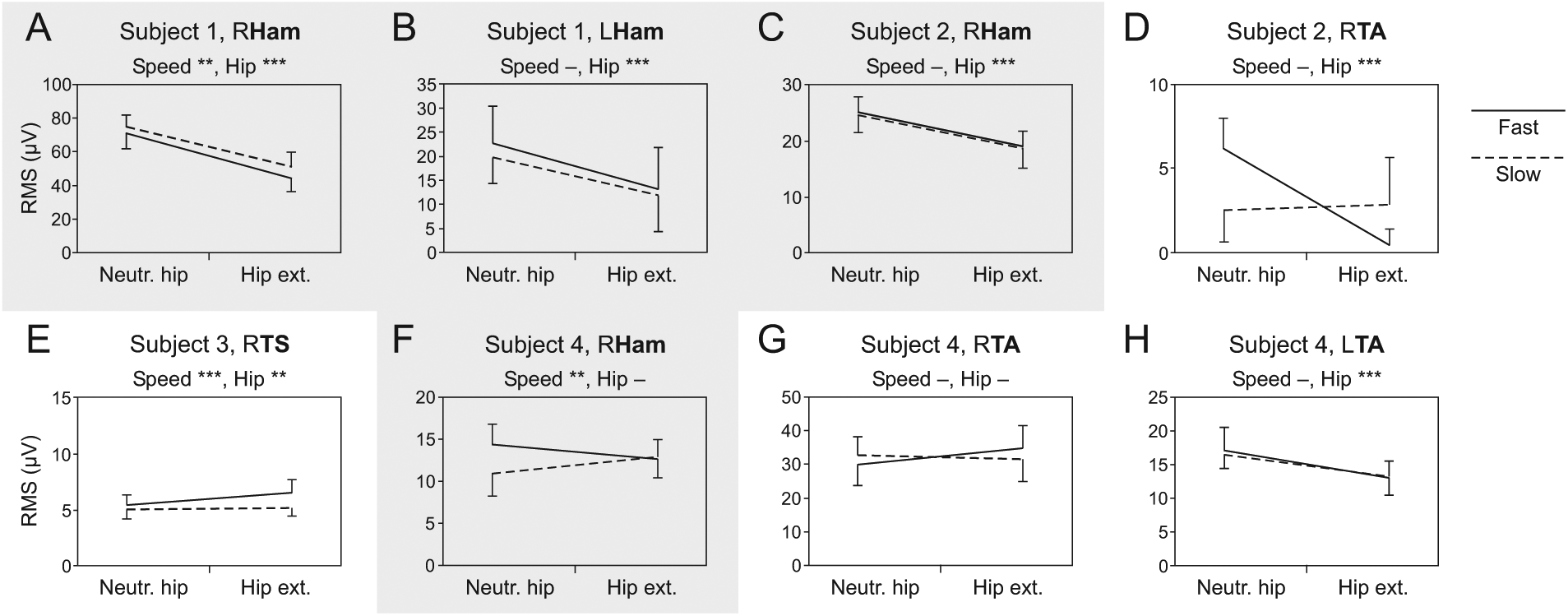
Leg-muscle activity produced by 30-Hz tSCS and robotic-driven treadmill stepping. (A-H) Average EMG RMS values per gait cycle with SD produced during stepping with neutral hip movement range (Neutr. hip) and hip extension increased by 5° (Hip ext.) at a slow (0.86/s) and fast (1.12/s) step frequencies. Nonparallel dashed and solid lines indicate interaction effects between hip-movement condition and step frequency. Examples are all muscles that had responded to robotic-driven stepping alone (see Figure 2). Ham, hamstrings; TA, tibialis anterior; TS, triceps surae. Gray background results derived from Ham. Asterisks indicate significant main effects (**,
In summary, sensory information related to the changes of stepping conditions could interact with tSCS, but there was no direct relationship between changes in a single stepping parameter and the recorded motor output. Furthermore, with tSCS on, the effect of changing step frequency or hip-movement conditions on the generated motor output was often different from the stimulation-off condition (see Figure 2).
Discussion
Rhythmic motor outputs were produced in some lower limb muscles by step-related sensory feedback as well as by tonic 30-Hz tSCS in 4 subjects with chronic, clinically complete SCI. When applied in combination, the number of rhythmically activated muscles was considerably increased. Changing the stepping conditions could change the amount of the generated motor output. The addition of tSCS also modified the multimuscle activation patterns. We suspect that the cyclical step-related feedback and the tonic drive provided by tSCS engaged different spinal mechanisms of rhythm generation. Their respective contribution and interaction, along with the responsiveness of the spinal circuitry altered after SCI, in the generation of rhythmic motor output will be elaborated in the following.
Central State of Excitability
Some degree of spastic muscle tone, that is, an altered state of excitability of the lumbosacral circuitry caudal to the SCI, is critical for the generation of rhythmic EMG activity during assisted treadmill stepping.4,11,30 The responsiveness of this circuitry may also be influenced by some residual descending long-tract fibers or propriospinal connections39,40 that can survive an SCI clinically classified as motor-complete. 41 The resulting interindividual differences in the relative excitability of various spinal pathways may then cause the varied responses observed during assisted treadmill stepping, ranging from minimal EMG activity limited to few muscles to moderate activity throughout the muscles of both legs. 8 Similarly, different rhythmic EMG patterns across motor-complete SCI subjects are generated by eSCS, both with 21 and without 20 step-related sensory feedback.
Here, DGO-stepping alone generated clonus in TA in subjects 2 and 4 in whom clonus and spasm-like activity also occurred in the passive standing position. Furthermore, the lower PRM-reflex thresholds in the same subjects (see Figure 1D) may suggest a higher degree of central excitability in the stretch-sensitive reflex arcs. Conversely, 30-Hz tSCS alone produced rhythmic muscle activity only in the other 2 subjects (1 and 3). Together, these observations imply that interindividual differences in the central state of excitability may have contributed to the varied effects across subjects in this study.
Although some differences may stem from the SCI levels, all lesions were well rostral to the lumbosacral circuitry examined in this study. Injury-level related differences in trunk afferent and propriospinal inputs were essentially minimized by the DGO and the BWS system that stabilized pelvic and trunk movement. Some of the interindividual variations in the EMG output are likely attributable to the specific peripheral feedback produced by DGO-stepping and the afferent population activated by tSCS.
Mechanisms of Rhythm Generation Induced by Step-Related Feedback
DGO-stepping elicited gait-phase synchronized EMG activity mainly in Ham and TA. A lack of Q activation by treadmill stepping in motor-complete SCI individuals was also noted in other studies.4,7 The timing of the Ham activity coincided with the stretch applied to the muscle when the lower leg was drawn forward during swing, as was previously observed for DGO 9 and manually assisted treadmill stepping.4,7 Accordingly, when maximum Ham stretch was reduced with increased hip extension, 42 its EMG activity decreased. In TA, the clonic EMG activity was generated during both shortening and stretching of the muscle and was not consistently influenced by stepping conditions. Inappropriate activation of TA during the stance phases of passive stepping4,7 with clonus-like EMG patterning1,8 is common in SCI individuals. The TA activity was hence likely dependent on spinal mechanisms of clonus, 43 their individual excitability, and afferent activation during the stance phases.
We propose that the motor patterns observed during DGO-stepping alone were immediate responses of spinal reflex pathways (with increased excitability after SCI) that were triggered by the sequential afferent inputs related to stretch and other modalities. The operation of rhythm and pattern generating networks is not necessary to explain these patterns of EMG responses. 44 The exaggerated hip extension at the end of stance did not produce new multimuscle activation patterns in our study set-up. Sensory activation of flexor and extensor synergies or of some elements of the spinal pattern generator may require specific sensory cues or SCI profiles. 11
Mechanisms of Rhythm Generation Induced by 30-Hz tSCS
The neuronal target structures of tSCS are afferent fibers within the most proximal portions of posterior roots/rootlets.24-26,45 Afferents with diameters corresponding to group I24,46 and group II27,37 of multiple lumbar and upper sacral segments, bilaterally, are recruited. As in epidural stimulation, 19 their repetitive stimulation presumably generates motor output transsynaptically through direct projections, evoking PRM reflexes, and recruits spinal interneuronal circuits as well that modulate the concomitantly elicited PRM reflex output. The rhythmic EMG activity in the form of modulating PRM reflexes with burst-like envelopes induced by tSCS without (see Figure 3) or independent of (see Figure 4A(iv)) step-specific peripheral feedback described here is consistent with observations obtained during eSCS.2,18,19 Therefore, tSCS, similarly to the epidural method, is capable of organizing spinal interneuronal networks to produce rhythmic motor outputs from a constant-frequency input. Functionally, these networks can be regarded as elements of a spinal pattern generator.20,47
Combined Effects of Robotic-Driven Stepping and 30-Hz tSCS
Adding tSCS during DGO-stepping drove the spinal circuitry into a physiological state not accessed by phasic feedback alone, as reflected by the generation of new multimuscle activation patterns, the observation of a motor output with autonomous rhythm frequency, the putative modulation of central mechanisms of clonus, 48 as well as the partially altered processing of sensory input related to the variation of stepping conditions.
tSCS and step-induced mechanical stimulation of peripheral receptors produced neural inputs carried by fibers with similar diameters and projections within the lumbosacral spinal cord. However, the patterns of the neural signals generated were different. The increased prevalence of rhythm generation when the 2 inputs were applied in combination may be explained by “coupling” effects of the respective spinal processes induced. Tonic tSCS likely activated some elements of the spinal rhythm generating circuitry and provided a base excitatory support. The rhythmic feedback inflow to this circuitry would then have facilitated its activation. Also, the step-related feedback presumably modulated the motoneuronal excitability in a gait phase–dependent manner and thus affected the PRM-reflex output. 49 Considering its augmentative effects on the motor output, tSCS could provide a countermeasure against the fading of leg muscle EMG activity that can occur within the first 10 minutes of assisted treadmill stepping in individuals with chronic motor-complete SCI.50,51
Methodological Considerations
In this exploratory study, we applied tSCS at 30 Hz across all subjects, because eSCS at this frequency was shown to generate rhythmic lower-limb EMG activities in motor-complete SCI individuals without2,20 and with step-induced feedback.21,23 Case-by-case adjustment may identify individually more appropriate tSCS frequencies. 52 Furthermore, the relative tSCS intensities were higher for the activated muscles than for the nonresponding muscles during stepping. Increased stimulation intensities may increase the number of recruited muscles.20,21 Absolute intensities of tSCS are however limited by the co-activation of trunk muscles. Individual stimulation-site adjustment as well as new electrode designs may facilitate the control over the relative intensities across muscles. 53
We used the DGO to keep stepping parameters constant and produce comparable sensory feedback during the stimulation-off and -on conditions. The level of unloading used for DGO-stepping50,51 may have posed some limitation in maximizing sensory input, although even with less than 60% BWS, the relationship between increased limb load and the generated motor outputs would not necessarily be monotonic. 1
With our focus on spinal burst generation, we described the combined effects of DGO-stepping and tSCS in terms of increased amount and prevalence of rhythmic EMG activities. To further substantiate a functional implication would also require assessing the degree of DGO assistance necessary for stepping. Changes in the torques exerted by the DGO to maintain the predefined movement trajectories (adapted from gait in healthy subjects) could be analyzed. However, it is not known whether induced muscle activation that would lead to rudimentary stepping movements 11 not in accordance with the reference trajectories would increase or decrease the DGO torques. Until the independence of induced stepping becomes quantifiable, EMG may remain the best measure for function. It reflects relative changes in the number of activated motor units as well as the muscle force produced54,55 and basic stepping function could be assessed by the EMG activity of knee extensors during stance and of hip flexors at the stance-to-swing transition. Appropriate amplifiers and EMG electrode orientations (with respect to the generated electric field) will be needed to study proximal muscles close to the tSCS site, such as the TFL in our study. EMG recordings from the main hip flexor, the deeply located iliopsoas, would require fine-wire electrodes.6,52
Conclusions
Regaining functional lower-limb movement with task-specific training is limited in individuals with severe SCI.11,50 Combining conventional training with other modalities that increase the excitability of the lumbosacral circuitry is thought to shift these limits.30,56 Studies employing eSCS have fueled ambitious expectations on the level of functional recovery that may occur after clinically complete SCI.2,23,52,57 The present exploratory study demonstrated that tonic tSCS can enhance rhythmic motor output over purely assisted stepping movements. It also showed that the individuals with clinically classified complete SCI responded with a spectrum of motor patterns to the step-related and tonic inputs. To identify the relationship between different lesion profiles within the group of clinically complete SCI and the obtainable motor outputs will require larger-scale studies with nuanced clinical and neurophysiological assessment of the SCI and standardized externally generated inputs across subjects. Such studies would also need to consider the impact of antispasticity medication that cannot be discontinued in all patients. Rigorous examination in a select group of subjects can study various stimulation parameters in each individual to test whether some independent stepping movements can be induced with proper lower-limb positioning and shifting the center of gravity11,58 and hence substantiate clinical relevance of tSCS.
Footnotes
Acknowledgements
We wish to express our gratitude to Milan R. Dimitrijevic, Department of Physical Medicine and Rehabilitation, Baylor College of Medicine, Houston, TX, USA, for mentorship and for initiating and encouraging the collaboration between the Vienna and Atlanta groups. Special thanks are due to Leslie VanHiel, Lauren McCollough, and Jason Tidwell, all affiliated with the Hulse Spinal Cord Injury Lab, Crawford Research Institute, Shepherd Center, Atlanta, GA, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wings for Life Spinal Cord Research Foundation (WfL), Grant Number WFL-AT-007/11; the Vienna Science and Technology Fund (WWTF), Grant Number LS11-057; and the Hulse Spinal Cord Injury Laboratory Grant.
