Abstract
Background. Erythropoietin (EPO) upregulates the mitogen activated protein kinase (MAPK) cascade, a central signaling pathway in cellular plastic mechanisms, and is critical for normal brain development. Objective. We hypothesized that EPO could modulate the plasticity mechanisms supporting spatial memory recovery in fimbria-fornix-transected animals. Methods. Fimbria-fornix was transected in 3 groups of rats. Seven days later, EPO was injected daily for 4 consecutive days within 10 minutes after training on a water maze task. Results. Our results show that EPO injections 10 minutes after training produced a substantial spatial memory recovery in fimbria-fornix-lesioned animals. In contrast, an EPO injection shortly after fimbria-fornix lesion surgery does not promote spatial-memory recovery. Neither does daily EPO injection 5 hours after the water maze performance. EPO, on the other hand, induced the expression of plasticity-related genes like arc and bdnf, but this effect was independent of training or lesion. Conclusions. This finding supports our working hypothesis that EPO can modulate transient neuroplastic mechanisms triggered by training in lesioned animals. Consequently, we propose that EPO administration can be a useful trophic factor to promote neural restoration when given in combination with training.
Introduction
Trophic factors are important molecules that regulate neurogenesis, neuronal differentiation, and survival, as well as neural plasticity mechanisms. Erythropoietin (EPO) has received particular attention in recent years. EPO has shown neuroprotective effects in animal models focused on ischemia,1,2 traumatic brain injury,3-5 ischemic preconditioning,4,6 and transgenic expression of a constitutively active EPO receptor. 7 More recently, EPO effects have been expanded to neurodegenerative diseases,8-11 multiple sclerosis, 12 and mood disorders.13,14 Moreover, it has been demonstrated that EPO is critical for normal brain development. 15 This diversity in role seems to be supported by the fact that EPO upregulates intracellular molecular signaling pathways that include the anti-apoptotic and mitogen-activated protein kinase (MAPK) cascades among others. 16 These results support the suggestion that EPO can promote neural plasticity mechanisms. 17
However, most of the experiments in injury models have been designed to demonstrate neuroprotection. Consequently, EPO was administrated before, or shortly after, brain injury, and behavioral assessments were performed several days later. To our knowledge, the effects of EPO administered in chronic stages after brain lesions have not been examined. In addition, the timing constraints of EPO administration with respect to the behavioral task are not known. Even when searching for EPO effects on memory and LTP in healthy mice, the protein was injected at least a week before the behavioral task. 18 Neural plasticity mechanisms, like those involved in learning and memory, are time-dependent processes. 19 Studies focused on possible direct modulating effects of EPO on neural plasticity mechanisms are needed to improve the efficacy of EPO administration. In particular, it is important to know the timing of EPO administration with regard to other significant events, such as the time of the lesion and rehabilitation interventions.
Here we address the question of whether EPO can (a) enhance functional recovery after the lesion has been established and (b) modulate neuroplastic mechanisms involved in recovery. We have examined these questions using a well-established brain injury model: the fimbria-fornix (FF) transection. This lesion produces a severe, irreversible impairment of spatial memory.20-22
Methods
Animals
A total of 108 two-month-old (250-300 g) male Wistar rats were used. The animals were obtained from a professional breeder (CENPALAB, Bejucal, Cuba) and housed in translucent plastic cages (5 animals per cage) under controlled environmental conditions (23°C, 50% relative humidity, 12-hour light–dark cycle) with free access to water and food throughout the experiment. All efforts were made to minimize the number of animals used, as well as pain and distress. The experimental protocols for this study followed the National Guidelines for the Care and Use of Laboratory Animals (CENPALAB, Cuba) and were approved by the institutional Bioethics Committee of the International Center for Neurological Restoration (CIREN) Cuba.
Fimbria-Fornix Lesion
Surgery was performed under chloral hydrate narcosis (420 mg/kg, i.p.) according to the procedure described elsewhere. 23 The animals were mounted on a stereotactic frame (David Kopf Inst, Saint Louis, MO), the skin was incised, and the skull exposed. A bilateral window was opened in the skull at the following coordinates: AP, 1.4 mm; ML, 0.5 to 5.2 mm. A reduced #11 surgical blade was lowered at 15° (with respect to the vertical direction) and a bilateral knife transection was carried out at the following coordinates: AP, 1.4 mm; ML, 0.8 to 3.1 mm; and DV, 5.0 mm. 24 Lesions were performed bilaterally to prevent recovery by sprouting of contralateral fibers. Sham-operated animals received the same treatment, but no knife was inserted and no transection was performed. Control animals did not undergo surgical procedures. To confirm the lesion procedure, after completion of the behavioral test, we randomly selected 3 animals from each lesion group for acethylcholinesterase immunostaining as described elsewhere.23,25
Experimental Design
To examine the effects of intraperitoneal EPO administration (using saline vehicle as control) on behavioral recovery, rats were randomly distributed into 11 experimental groups:
FF (N = 13): bilateral fimbria-fornix lesion; no other treatment
FF-EPO (N = 16): bilateral fimbria-fornix lesion; EPO injection immediately after lesion (to test EPO neuroprotective effects)
FF + 10 min-EPO (N = 16): bilateral fimbria-fornix lesion; EPO injection 10 minutes after daily water maze training on post-lesion days (PLD) 7 to 10 (to test EPO neurorestorative effects)
FF-EPO + EPO (N = 11): bilateral fimbria-fornix lesion; EPO injection immediately after lesion; EPO injection 10 minutes after daily water maze training on PLD 7 to 10 (to test a possible synergism between neuroprotective and neurorestorative effects)
FF + 5 h-EPO (N = 8): bilateral fimbria-fornix lesion; EPO injection 5 hours after daily water maze training on PLD 7 to 10 (to test if EPO effects on spatial memory recovery are time-dependent)
FF + 10 min-NaCl (N = 10): bilateral fimbria-fornix lesion; NaCl (0.9%) solution injected 10 minutes after daily water maze training on PLD 7 to 10 (control to group 3)
FL (N = 7): sham lesion; no other treatment (control to group 1)
FL + 10 min-EPO (N = 6): sham lesion; EPO injection 10 minutes after daily water maze training on PLD 7 to 10 (control to group 3)
Healthy controls (N = 24): no lesion or treatment.
No blinding was used, but possible observer biases were minimized by the fact that all behavioral data were obtained and processed by an automatic video processing system (see below). Additionally, the technicians performing FF lesion or sham-lesion do not participate in the group distribution of the animals, which was performed randomly. Moreover, the behavioral training was performed by 2 different technicians who handle the animals and recorded the track of each trail.
To provide data for possible genes involved in spatial memory recovery after FF lesions, bdnf and arc gene expression was studied in the hippocampus after the end of the experiments (PLD 11, 2 hours after training) in a sample of 5 animals from each group. In addition to that, bdnf and arc gene expression was also studied in the frontal cortex of naïve, nonlesioned, nontrained animals 30 minutes after EPO (N = 5) or NaCl injection (N = 5). Figure 1A summarizes the experimental design.
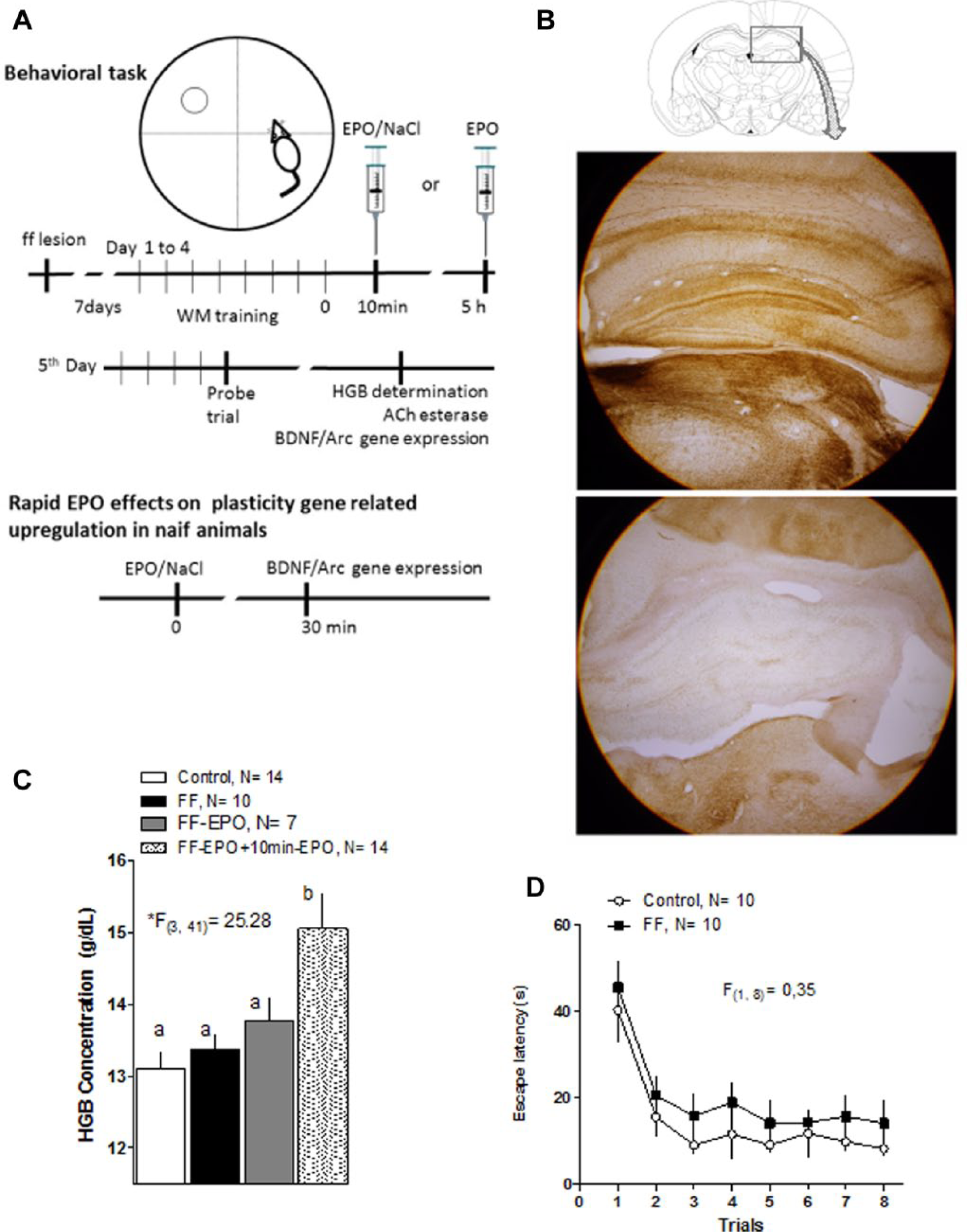
Experimental design and control experiments.
Hemoglobin Study
To verify the EPO biological activity, blood samples were taken from randomly selected animals immediately after the end of water maze training on PLD 15. The hemoglobin concentration was determined using standard procedures in a clinical laboratory.
Behavioral Training
One week after surgery, the animals were trained for 5 successive days in a Morris Water Maze (place learning). During the first 4 days (PLD 7-10), animals performed 8 consecutive trials, during which they could search (60 second maximum) for a hidden platform to escape from the water. On the fifth day (PLD 11) they performed 4 standard trials and then 1 probe-trial (ie, without platform). We have also performed a visible-platform training session (day 6) with the control and FF-lesioned animals to explore whether visual or motor impairments are involved in the deficits that might be shown by FF-lesioned animals. The training maze consisted of a circular swimming pool (diameter = 1.50 m, filled with water at 22°C) marked by 4 virtual equally spaced points marked N, S, E, and W, respectively, on the external tank wall. On each trial, animals were released into the water from one of these positions following a previously selected, random sequence for each day. The behavior of the animals was captured by a television camera connected to a personal computer. The data were collected and processed using the SMART program (Panlab, v 2.0). Several behavioral variables were measured, escape latency (in seconds) and distance (in cm) were selected as the primary outcomes, as reliable measures of learning and memory. We also report thigmotaxis (measured as the time that the animals spend swimming along the walls) as well as time in the target quadrant and the number of crossings over the place where the platform used to be located in the probe trial (last trial in last training day, escape platform removed). The escape latencies and the distance travelled of 4 consecutive trials were averaged and presented in the graphic as blocks. Thus, Block 2.1 (B2.1) is the average of the 4 first trials of the second day of training (see Figure 2A and B).
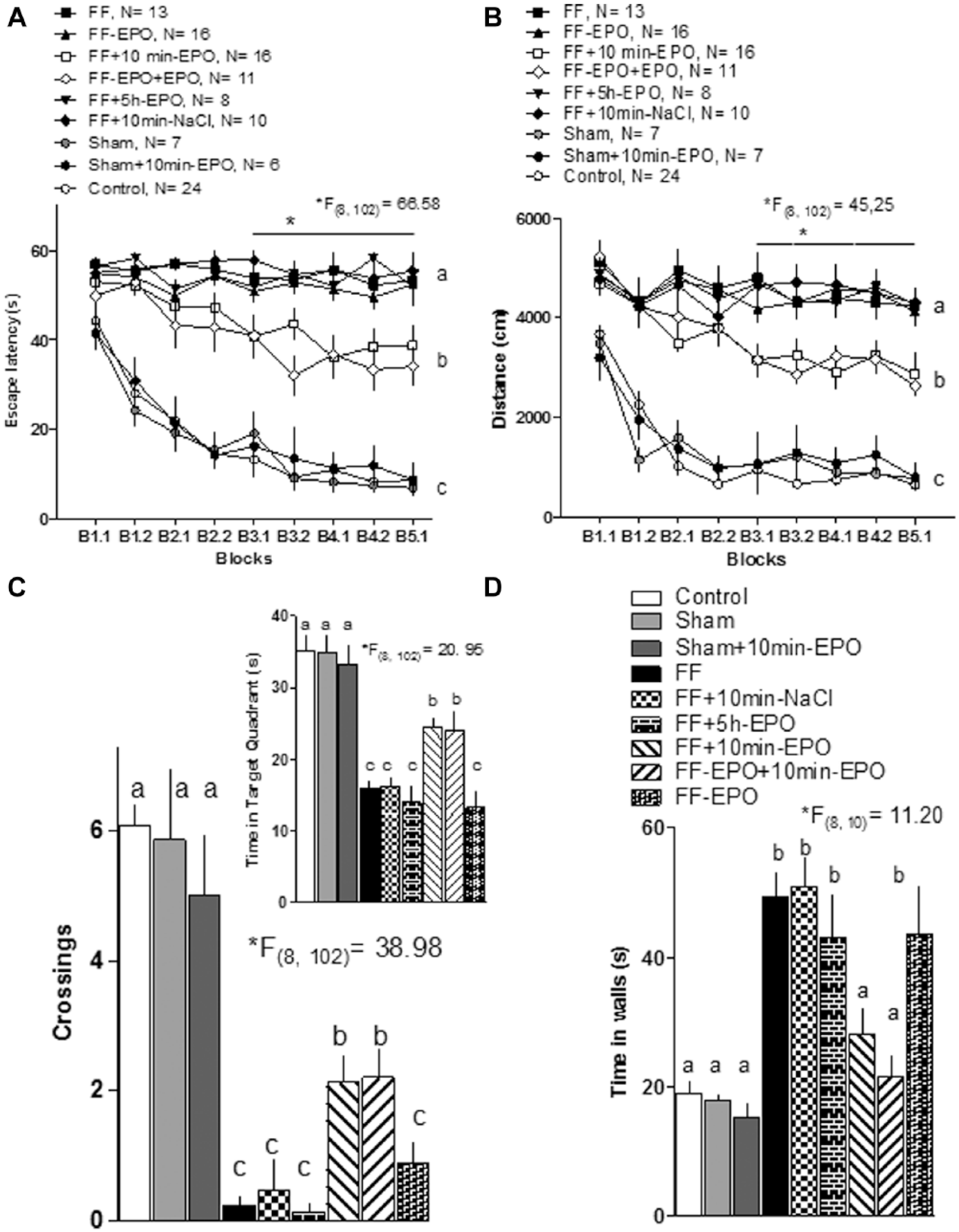
Erythropoietin (EPO) administration improves spatial memory in fimbria-fornix (FF)-lesioned animals.
EPO Administration
Human recombinant erythropoietin (ior EPOCIM, Center for Molecular Immunology, Havana, Cuba) was used. The animals were injected intraperitoneally immediately after the FF lesion (Figure 1A, 9000 U/kg), or on PLD 8 to 11, 10 minutes after completing each daily training session (5000 U/kg) or 5 hours after the each training session (5000 U/kg). As a control, we injected the same volume by weight of NaCl, 0.9% (see Figure 1A).
Arc and bdnf Expression 30 Minutes After Intraperitoneal EPO Injection
To evaluate EPO involvement in plasticity mechanisms, arc and bdnf gene expression were measured in 2 structures: in the hippocampus of trained animals (5 from each group) lesioned or not, and treated or untreated with EPO. In the prefrontal cortex of nonlesioned, nontrained animals were euthanized 30 minutes after injection to measure how fast and how far EPO can modify gene expression.
The gene expression was measured using β-actin as a control and following a conventional protocol described previously. 26 These genes have been shown to be involved in learning and memory and synaptic plasticity.27-31
In brief, the sequence of each primer, the annealing temperatures, and the length of the amplified products were as follows: BDNF, forward and reverse primer sequences (5′ to 3′): forward: TTGGCCTACCCAGCTG TGCGGAC/reverse: CTCTTCGATCACGTGCTCAAAAGTG (annealing temperature 60°C, product length 130 bp); Arc, TGC TCC AGG GTC TTG GGG AGT/AGG AGA GCT GCC TGA GCA GG (60°C, 472 bp); β-actin, ATTTGGCACCACACTTTCTACA/TCACGCACGATTTCCCTCTCAG (51°C, 379 bp). β-Actin was used as an endogenous control. DNA products were electrophoresed on 1.2% agarose gels at 100 mV and visualized with ethidium bromide. Polymerase chain reaction products were separated and documented by digital imaging. 29 We performed the electrophoresis 2 times, which were used in the semiquantification analysis of the second one. For the semiquantitative analysis, we used the free online program ImageJ (Version 1.44; Wayne Rasband, National Institute of Health; http://imagej.nih.gov/ij). For this analysis we first subtracted the background activity to the target band and then normalized using β-actin as reference.
Statistical Analysis
Repeated-measures ANOVA was used to compare the escape latencies and distance among groups, and one-way ANOVA to compare thigmotaxis, time in the target quadrant and the number of crossings. As a post hoc test we used the Tukey HSD test. Significant differences were considered when P < .05.
Results
Controlling Lesion Efficacy and EPO Activity
The FF lesion reliably produced a virtually complete, bilateral cholinergic denervation in the hippocampal formation. Representative photos are shown in Figure 1B. Notice the intense AChE staining above and below the granule cell layer at the dentate gyrus and pyramidal cells in the cornus ammonis in control animals (upper photo), compared with an almost complete absence of staining for the same enzyme in FF-lesioned animals (below).
The EPO used in these experiments showed its classic erythropoietic activity as can be seen in the results of measuring hemoglobin (HGB) concentration in blood samples taken from animals in different experimental groups (Figure 1C). A one-way ANOVA demonstrated significant differences (*F[3, 41] = 25.28) among groups and the post hoc Tukey test showed that HGB concentration was higher in the group receiving daily after training EPO, but not in the one that received a single EPO injection immediately after lesion.
Water Maze Training, FF Lesion, and EPO Treatment
Figure 1D shows results of a visible target training session performed with the control and FF-lesioned groups at the end of training in the Morris Water Maze. A repeated-measures ANOVA (F[1, 8] = 0.35) showed no differences between groups in the latency to reach the platform.
In analyzing the data presented in the following figures, it should be remembered that every BLOCK is the result of averaging the results of 4 consecutive trails, where some learning effects may already be present, particularly in the non- or sham-lesioned groups. A one-way ANOVA of the latencies in the first trail showed no significant differences (F[8, 102] = 0.25, NS data not shown) among groups. However, a similar analysis for the distance travelled showed significant initial differences (F[8, 102] = 2.47, data not shown). A post hoc analysis (Tukey HSD) showed that the lesioned groups receiving one single EPO injection immediately after lesion, those receiving EPO after lesion and after training, and those receiving EPO after training (but not those receiving EPO 5 hours after training or other lesioned groups) were slightly, but significantly, worse than the control group in the first trial. The comparison of the results in the water maze over the 5 training days showed interesting results that support the assumption that EPO can promote neural plasticity favoring learning and memory.
Statistical comparison of escape latency for all the experimental groups (Figure 2A) showed a significant difference (repeated-measures ANOVA, factor GROUP, F[8, 102] = 66.58). The post hoc Tukey HSD test showed that the nonlesioned groups (labeled c) differed significantly from all the lesioned groups. Among the lesioned groups, the 2 receiving EPO 10 minutes after each daily training session (labeled b) showed a significant improvement in learning compared to those groups (labeled a) treated with saline, but also from those receiving EPO in a single dose immediately after the lesion and those receiving daily posttraining injection 5 hours later. A post hoc analysis (Tukey HSD) of the interaction GROUP/BLOCK showed that both groups receiving EPO 10 minutes after training significantly differed from the lesion (FF) group from block 3.1 on.
The distance travelled to target showed quite similar results (Figure 2B). The statistical analysis (repeated-measures ANOVA, factor GROUP, F[8, 102] = 45.25) demonstrated differences among groups showing the same segregation pattern: control and sham operated groups (c), the 2 groups receiving EPO injections daily after training (b), and the rest of the lesioned groups (a) (Tukey HSD). The post hoc (Tukey) of the GROUP/BLOCK interaction confirmed significant differences among lesioned groups (FF, FF-EPO, FF + 5 h EPO and FF + 10 min NaCl) and those receiving EPO 10 minutes after each training session (FF-EPO + EPO and FF + 10 min EPO) from block 3.1 on.
A similar pattern of group segregation was obtained in the probe trial. The number of crossings (Figure 2C, ANOVA, F[8, 102] = 39.89), the time spent in the target quadrant (Figure 2C inset, ANOVA, F[8, 102] = 20.95), and thigmotaxis (Figure 2D, ANOVA, F[8, 102] = 11.20) showed significant differences among groups. The post hoc Tukey HSD test showed again that nonlesioned or sham-lesioned animals (a) showed more crossings over target, spent more time on target quadrant, and showed less thigmotaxis than lesioned animals (c). The 2 groups receiving EPO 10 minutes after daily training showed an intermediate behavior (b) regarding the crossings and time in target quadrant, but were statistically undistinguishable from the control in regard to the reduced thigmotaxis (a).
Summarizing, the behavioral results showed a significant effect of EPO ameliorating the severe spatial memory impairment produced by the FF lesion, but only when EPO was administered shortly after the daily training session. EPO administration immediately after the lesion did not show any effect. Also, this early injection did not increase the efficacy of EPO posttraining administration when both treatments were combined. The importance of timing for the EPO effect on memory is reinforced by the absence of effect when the hormone was given long after the end of the training session (5 hours). It should also be noticed that injecting EPO to nonlesioned animals neither improved nor impaired performance in the maze.
EPO Administration Triggered Plasticity-Related Early Gene Expression
In Figure 3A, we show the results of a parallel molecular study designed to examine the short-term effect of EPO administration and the penetration of the effect. As can be seen, both arc and bdnf increased their expression in about 80% in prefrontal cortex samples taken 30 minutes after injection. This result demonstrates that EPO effects on gene expression can occur at relative short time even in distant structures. Notice as well that the intraperitoneal injection of NaCl has no effect on the expression of both genes.
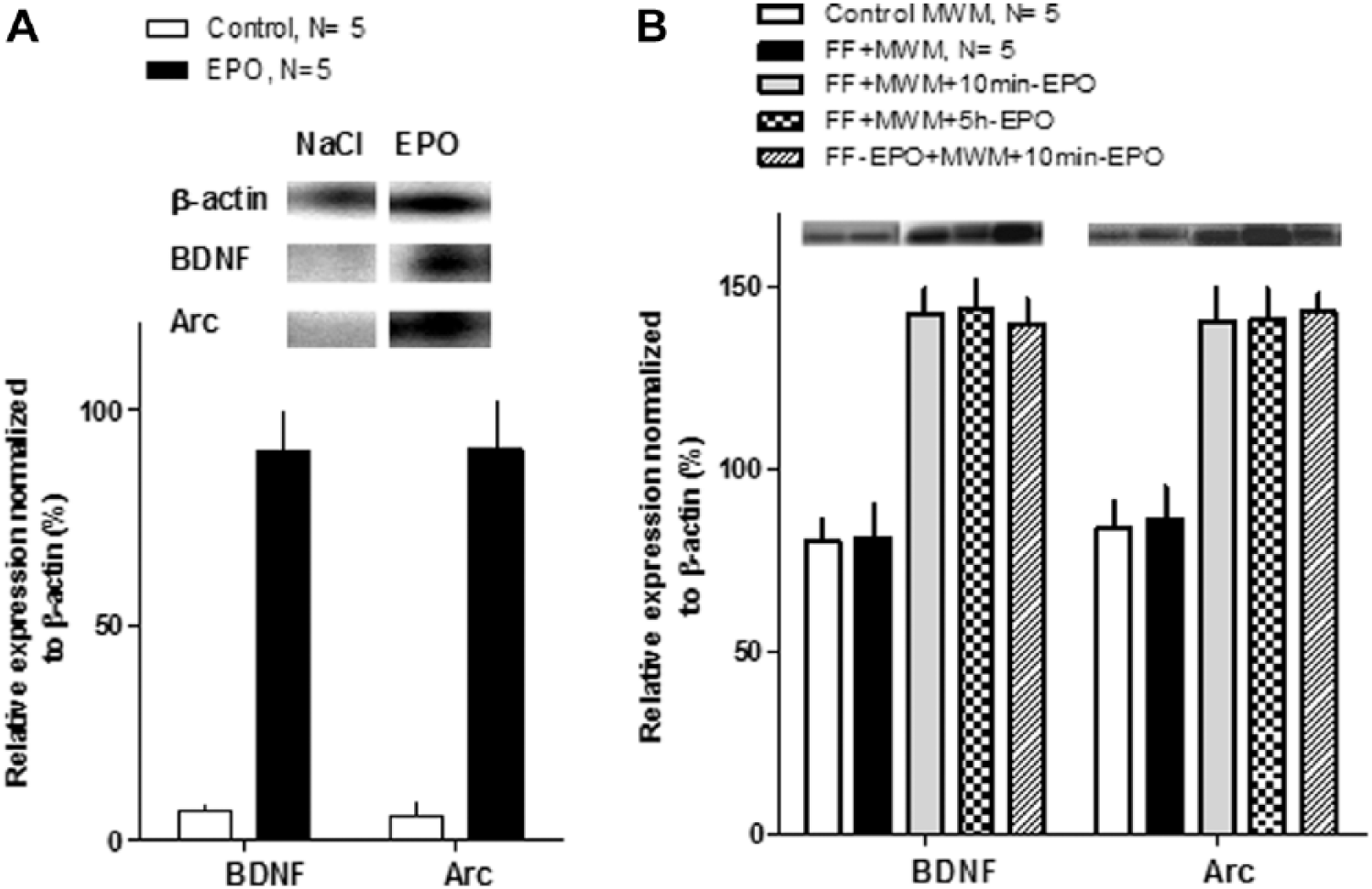
(A) Early gene expressions in naïve animals by semiquantitative RT-PCR expressed as percentage of β-actin. Erythropoietin (EPO) increased the expression of bdnf and arc in the prefrontal cortex 30 minutes after the intraperitoneal injection. (B) Early gene expressions by measured by semiquantitative RT-PCR expressed as percentage of β-actin in the different groups after the end of the training sessions. The expression of both arc and bdnf genes was increased in all groups receiving repeated EPO injection, irrespective of the time of administration.
Figure 3B shows the results of measuring arc and bdnf gene expression after FF lesion, water maze training, and EPO. As can be seen in the bar graph, EPO induced an increase in the expression of both plasticity-related genes, and this effect takes place with independence of the timing of injection with regard to training. On the other hand, the FF lesion did not modify the expression of the genes with respect to control, nonlesioned animals.
Discussion
The present results provide evidence that EPO administered during the chronic phase after a FF lesion results in improvement of spatial memory. The behavioral improvement is time dependent, because it is only observed when EPO is delivered soon after behavioral training. No evidence was found for presumed neuroprotective effects at the behavioral level.
Gene expression results suggest that activation of plasticity-related genes like arc and bdnf can contribute to the efficacy of EPO in this model, but also indicate that additional, complementary mechanisms initiated by the combination of lesion, training, and EPO given shortly after training must be involved.
The FF lesion is a classical biological model of a severe and irreversible spatial-memory acquisition deficit,20-22 which is commonly used to assay treatments aimed to improve memory impairments.20,32-34 FF lesions also affect long-term potentiation (LTP) 35 as well as LTP reinforcement by motivational stimuli. 23
The memory deficit induced by the FF lesion is severe, but it can be ameliorated. Using a similar experimental strategy we have shown that basolateral amygdala stimulation (BLA) 15 minutes after the water maze training25,36 induces a significant improvement in spatial learning abilities. Here we report that daily EPO administration, 10 minutes after the behavioral training, improved spatial memory acquisition in FF-lesioned rats. Interestingly, those FF-lesioned animals that received daily EPO 5 hours after the end of the learning task did not show any behavioral sign of improvement. These data suggest that the temporal relationship between training and EPO administration is critical for spatial memory recovery.
The critical timing of EPO administration with relation to the behavioral task could be explained by assuming that an increased availability of EPO during a transient, early phase supports specific cellular mechanisms involved in memory acquisition and maintenance, which take place in a precise time sequence.37-40 The bdnf and arc gene upregulation induced by EPO administration supports this interpretation. Our data showing that acute administration of EPO induces a rapid, increased expression of the plasticity-related genes bdnf and arc supports this interpretation. Especially because, in the chronic study all animals that received daily EPO, irrespective of the time of administration (10 minutes or 5 hours after training), showed increases in gene expression. However, those animals that received EPO administration 5 hours after the water maze training do not show spatial memory recovery.
We have previously shown that novelty exploration 10 minutes after water maze training preserves memory traces altered by an intense posttraining footshock and that this effect is also accompanied by an increased expression of bdnf and arc. 26 Additionally, posttraining BLA stimulation, a procedure that ameliorates the memory deficits in FF-lesioned animals, induces an increase of BDNF concentration in the hippocampus. 36 A recent report from an independent laboratory confirmed the ability of EPO to enhance the expression of BDNF and Arc among others. 17
The existence of a time window for the EPO effect on recovery of spatial memory suggests that increased erythropoiesis is not a likely causal factor, since animals receiving a 5-hour delayed injection with the same EPO dosage did not show an improvement in behavior. The time dependence of memory enhancing procedures or substances is well established 19 and can be explained at the cellular level by mechanisms of synaptic tagging.41,42 According to this hypothesis, the activation of synaptic inputs sets a tag that can become reinforced from the capture and insertion of plasticity-related proteins activated by another mechanism within a restricted time window. Equivalent mechanisms, known as behavioral tagging, are thought to operate at a behavioral level.26,43 Accordingly, it is possible that EPO promotes the synthesis of plasticity-related proteins necessary for memory stabilization, which are captured by the tags set during the learning task, but only when they become available within a limited time window.
One argument against a direct action of EPO on the brain is the low permeability of this protein across the blood–brain barrier. 44 In the case of our results this difficulty is reduced simply by the fact that the lesion procedure disrupts the integrity of the barrier, allowing the access of peripherally injected EPO. However, there have been claims that even the low amounts of EPO able to cross the intact blood–brain barrier can induce neuroprotective effects.44,45
As mentioned earlier, neuroprotective effects of EPO have been reported in different animal models.1-5,8-11 The lack of presumptive neuroprotective effects of EPO based on behavioral recovery in our results was surprising, particularly considering previous reports showing that EPO injected shortly after FF lesions improved behavioral performance in the water maze. 46 However, a preliminary explanation could emerge from the simple fact that our FF-lesioned animals showed no sign of training-induced recovery, while this is clearly the case in the Mogensen’s study. More important, we provide histological data showing that the lesion produced virtually complete, bilateral cholinergic denervation in the hippocampal formation, even in those lesioned animals that received EPO immediately after the lesion procedure.
Regarding the brain regions involved in the beneficial effects of EPO on memory, the hippocampus appears as a likely candidate. Despite the cholinergic and aminergic denervation induced by the lesion, 20 FF-lesioned rats can improve in hippocampus-dependent tasks like the Morris Water Maze after appropriate treatment.32,33,36,47-50 The prefrontal cortex participates in spatial information processing in parallel with the hippocampus51,52 and in final memory stabilization and subsequent reconsolidation.53-57 Consistent with this assumption, our data show that systemic EPO administration induces the expression of the plasticity-related genes bdnf and arc58-61 one hour later in the prefrontal cortex.
To summarize the present results, we show that EPO can promote the recovery of spatial memory after FF injury, when administered during a chronic stage of the lesion. The effects of EPO on recovery are time dependent with respect to behavioral training, suggesting that this molecule modulates neural plasticity mechanisms responsible for the improvement. Consequently, our data support the idea that it may be possible to use EPO to promote neural plasticity mechanisms during rehabilitation procedures.
Footnotes
Acknowledgements
We thank the Molecular Biology Department of the Cuban Neuroscience Center, especially Viana Manrique, MSc, for her generous support. We also thank Dr Alain Garcia for excellent animal care.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
