Abstract
Background. The brain may reorganize to optimize stroke recovery. Yet relatively little is known about neural correlates of training-facilitated recovery, particularly after loss of body sensations. Objective. Our aim was to characterize changes in brain activation following clinically effective touch discrimination training in stroke patients with somatosensory loss after lesions of primary/secondary somatosensory cortices or thalamic/capsular somatosensory regions using functional magnetic resonance imaging (fMRI). Methods. Eleven stroke patients with somatosensory loss, 7 with lesions involving primary (S1) and/or secondary (S2) somatosensory cortex (4 male, 58.7 ± 13.3 years) and 4 with lesions primarily involving somatosensory thalamus and/or capsular/white matter regions (2 male, 58 ± 8.6 years) were studied. Clinical and MRI testing occurred at 6 months poststroke (preintervention), and following 15 sessions of clinically effective touch discrimination training (postintervention). Results. Improved touch discrimination of a magnitude similar to previous clinical studies and approaching normal range was found. Patients with thalamic/capsular somatosensory lesions activated preintervention in left ipsilesional supramarginal gyrus, and postintervention in ipsilesional insula and supramarginal gyrus. In contrast, those with S1/S2 lesions did not show common activation preintervention, only deactivation in contralesional superior parietal lobe, including S1, and cingulate cortex postintervention. The S1/S2 group did, however, show significant change over time involving ipsilesional precuneus. This change was greater than for the thalamic/capsular group (P = .012; d = −2.43; CI = −0.67 to −3.76). Conclusion. Different patterns of change in activation are evident following touch discrimination training with thalamic/capsular lesions compared with S1/S2 cortical somatosensory lesions, despite common training and similar improvement.
Keywords
Introduction
The brain may reorganize to optimize stroke recovery, 1 but how does rehabilitation affect neural plasticity? One of the most potent modulators of cortical structure and function is behavioral experience, 2 consistent with learning-dependent neural plasticity. Yet relatively little is known about neural correlates of training-facilitated recovery, particularly after loss of body sensations. 3 Knowledge of how the brain adapts in response to a clinically effective therapy in patients with interruption to different parts of function-specific brain networks may provide important insights into individual targeting of interventions. 4
Evidence that specific interventions facilitate neural plastic changes and improved motor recovery after stroke is growing.5-8 Reviews of neuroanatomical outcomes associated with specific motor interventions, revealed changes in the ipsilesional primary sensorimotor cortex, 7 and variable changes in associated motor areas such as premotor cortex, supplementary motor area, and cerebellum in both hemispheres.5-7
Brain adaptation may be influenced by time poststroke,9-12 lesion location,13,14 and interruption to remote regions and functional brain networks.12,15,16 In motor recovery, different activation patterns are reported following cortical or subcortical lesions. 13 Recovery following cortical motor lesions is more distributed, involving secondary motor regions and contralesional hemisphere, while subcortical lesions showed return of activation to cortical motor areas in chronic stroke. 13
In the somatosensory domain, impairment was correlated with distributed activation in the chronic phase. 17 In patients with thalamic lesions, return of S1 activation is reported with good recovery,18-20 or sparing with mild impairment. 20 Furthermore, touch impairment was differentially correlated with brain activity after thalamic/capsular compared with primary (S1) and/or secondary (S2) somatosensory cortex lesions 1-month poststroke. 14 These findings suggest lesion-specific mechanisms of brain adaptation and different rehabilitative strategies to target specific brain regions.
One in 2 people experience loss of body sensations after stroke, with major and ongoing impact on recognizing objects via touch, manipulating objects, use of the limb in daily activities, and functional outcome.21-23 Despite the high prevalence and negative impact, factors that affect brain recovery and adaptation following somatosensory loss remain virtually unknown.3,14
We have systematically developed an approach to somatosensory rehabilitation that applies robust neuroscience evidence to help stroke survivors regain a sense of touch.3,24-26 The approach is operationalized and evaluated using 2 different training protocols: stimulus-specific training 24 and transfer-enhanced training.25,26 Effectiveness has been established using meta-analysis 27 and a randomized controlled trial. 26
Our aim was to characterize and compare changes in brain activation associated with clinically-effective touch discrimination training (stimulus-specific training) in stroke patients with touch discrimination impairment following lesions interrupting different parts of the somatosensory network, that is, primary and secondary somatosensory cortices (S1/S2 group) or subcortical structures that relay somatosensory information such as thalamus, especially the ventral posterior nucleus, and internal capsule (thalamic/capsular group). We asked the question: Do changes in brain activation following clinically effective touch discrimination training differ in patients with touch impairment following lesion to S1/S2 regions compared to thalamic/capsular lesions?
We hypothesized that activation involving primary and secondary somatosensory regions would occur with training-facilitated recovery, based on prior literature of return of activation to “typical” cortical regions with good recovery.7,9,11,13,14 We also predicted involvement of regions involved in attention, vision, and tactile learning, as these were specifically targeted in discrimination training.3,24 Given prior evidence of different activation with motor 13 and somatosensory 14 recovery following cortical and subcortical lesions, we predicted differences between patients with lesions primarily of S1/S2 cortical processing regions compared with thalamic/capsular subcortical relay regions. Specifically, we expected that those with thalamic and/or capsular somatosensory lesions would activate primarily in ipsilesional cortical somatosensory regions following training, while those with lesions of S1 and/or S2 would activate distributed regions, including nonprimary somatosensory regions and contralesional hemisphere.
Methods
Participants
Stroke patients with impaired touch discrimination of the upper limb following first-episode infarct were recruited. Patients had to be medically stable, able to give informed consent, comprehend simple instructions, and be right-hand dominant. Patients with brain-stem infarct, hemorrhagic stroke, previous neurological dysfunction, neglect based on standard neuropsychological tests, history of impaired hand function, or history precluding magnetic resonance imaging (MRI) were excluded. The study was approved by hospital and university human ethics committees and all participants gave written informed consent.
Study Design
At 6-months poststroke, 11 patients with somatosensory impairment received stimulus-specific training of touch discrimination. 24 Clinical and MRI testing occurred at 6 months poststroke, then again following a 6-week intervention of the clinically effective touch discrimination training. All measures were repeated at each time. Most (n = 10) were also investigated at 1-month poststroke, and are a subset of the sample reported by Carey et al. 14 Patients with lesions of S1 and/or S2 were grouped, given the cortical nature of these lesions and their role in processing of somatosensation for perception and discrimination, while those with lesions primarily of thalamus and/or internal capsule/corona radiata were grouped given their role in relay of information and evidence that regions such as ventral posterolateral nucleus of the thalamus and lenticulocapsular regions are indicated in the differential diagnosis of pure sensory stroke.28,29
Touch Discrimination Intervention
Touch discrimination training involved stimulus-specific training of texture grids, based on perceptual learning, as previously described. 24 During training, the individual is required to attentively explore and discriminate the “odd texture” from a set of 3 texture grids (sets vary in stimulus difficulty). Feedback is given on accuracy and manual exploration. The individual is then guided to calibrate their impaired touch sensation internally, by reference to a more normal touch sensation experienced through the other, “unaffected,” hand exposed to the same stimulus set, and via vision. Anticipation trials, in which the patient is cued for a particular stimulus difference, are used to “prime” the sensory system to discriminate particular sensory attributes, and achieve repetition.3,27 The approach facilitates intra- and cross-modal plasticity and learning-dependent neural plasticity.3,30 Intervention involved 15 sessions of 45 to 60 minutes each, 3 times a week over 6 weeks. Patients were not involved in any other rehabilitation at the time of the study.
Demographic and Clinical Profile
Demographic and clinical background included: age; gender; premorbid hand dominance 31 ; stroke type; lesion location; severity of neurological impairment using the National Institutes of Health Stroke Scale; hand function using the Action Research Arm Test 32 ; touch detection using WEST (Weinstein Enhanced Sensory Test) hand monofilaments 33 ; limb position sense using the Wrist Position Sense Test 34 ; tactile object recognition using the functional Tactile Object Recognition Test 27 ; and temperature discrimination using the Roylan hot-and-cold discrimination kit.
Quantification of Touch Discrimination Impairment and Response to Somatosensory Rehabilitation
The Tactile Discrimination Test (TDT) 35 was the primary outcome measure for touch discrimination impairment and response to stimulus-specific texture discrimination training. The TDT has high retest reliability, age-appropriate normative standards, and good discriminative properties. 35 Touch detection, using the WEST hand monofilaments, and a visual analogue scale of ability to feel the touch stimulus during the functional MRI scan were also monitored pre- and postsensory rehabilitation.
Functional MRI: Touch Stimulation Paradigm
Whole-brain functional MRI studies were conducted using a controlled dynamic touch discrimination stimulus to the fingertips, as previously described. 14 Touch stimulation and rest conditions were alternated in a block design comprising 6 × 30-second epochs of each condition for each hand. During the task condition a plastic texture grating was repeatedly moved from side-to-side under the second, third, and fourth digits at a controlled pressure and speed, with the hand held stationary in the device. In the control condition, the surface was moved at the same speed but did not contact the fingertips. Participants were instructed to “feel the stimulus and keep feeling it as it moves under your fingertips.” Separate runs were conducted for each hand, the affected hand first. Immediately after each session, participants reported on their degree of concentration during the task; ability to feel the stimulus and perception of stimulus intensity, using visual analogue scales.
A 3-T GE Horizon LX Signa MRI scanner with quadrature head coil (GE Medical Systems, Waukesha, WI, USA) acquired the functional images in 25 axial slices spanning the cerebellum to the apex of the cerebrum using a gradient-echo, echoplanar imaging (EPI) sequence (repetition Time [TR] = 3000 ms; slice thickness 4 mm; interslice gap = 1mm; in-plane voxel size = 1.95 mm × 1.95 mm; bandwidth = 100). 14 Sixty volumes were acquired per condition, per hand. A high-resolution 3-dimensional (3D) anatomical image, 2D T1-weighted, and axial 2D T2-weighted images in the same plane as the EPI were also acquired. 14
Data Analysis
Behavioral Data
Behavioral data were analyzed using the Statistical Package for Social Sciences (version 20). Descriptive statistics were calculated for all measures. Improvement in the TDT score was tested using the Wilcoxon signed-rank statistic.
Imaging Analysis
Imaging analysis used SPM8 (www.fil.ion.ucl.ac.uk/spm/software/spm8/), MILXView (http://research.ict.csiro.au/software/milxview), and the iBrain Analysis Toolbox for SPM (www.brain.org.au/software). Preprocessing was conducted as previously described. 14 A lesion mask 36 was created by the study neurologist for each participant, co-registered and masked as part of the normalization process at each time point. A local EPI brain template was derived from study participants through a process of iterative spatial normalization to the group mean. Images of patients with right hemisphere lesions were flipped such that all infarcts were in the left hemisphere.
First-Level Imaging Analysis
A first-level analysis was conducted for each individual using the general linear model as implemented in SPM8. Motion correction parameters were included as covariates of no interest. Additional gross-motion rejection regressors were included when scan-to-scan motion exceeded 1mm, to completely account for the affected volume and the following 3 volumes. 37 For each individual, task-related changes in BOLD (blood oxygen–level dependent) signal intensity were mapped for each session, and also contrasted between sessions to determine change over time.
Second-Level Imaging Analysis
Individual contrast images from the first-level were taken to a second-level random-effects analysis. Two second-level analyses were conducted: one using a 2 × 2 design of time (pre/post) and group (S1/S2–thalamic/capsular), and the other using the change over time contrasts by group (S1/S2–thalamic/capsular), assessed using Student t tests. Analyses were conducted separately for the affected and “unaffected” hands.
Significant results are reported at cluster-corrected familywise error of P < .05 (2-tailed; 1-tailed threshold of P ≤ .025 when testing for increased/positive and decreased/negative activation). The cluster-defining primary threshold was selected a priori at the voxel-height threshold of P < .001 uncorrected, consistent with recent recommendations. 38 Location of activation was objectively quantified using probabilistic cytoarchitectonic maps. 39 Post hoc analyses examined the signal intensity of the peak voxel coordinate within individuals over time in regions found to be significant at the group level. Change in signal intensity over time was tested using Wilcoxon signed-rank test.
Results
Background Demographic, Lesion, and Clinical Status
Eleven stroke patients (6 males, mean = 58.45 years, SD = 11.33 years) with impaired touch discrimination were studied. Seven had lesions involving S1 and/or S2 (4 males, 58.7 ± 13.3 years) and 4 had lesions involving thalamus and/or capsular somatosensory regions (2 males, 58 ± 8.6 years). Demographic and lesion location details are presented in Table 1. Background neurological and clinical data prior to intervention are outlined in Table 2.
Demographic and Lesion Location Data for Each Stroke Individual, With Summary Lesion Overlay Maps for Subgroups. a
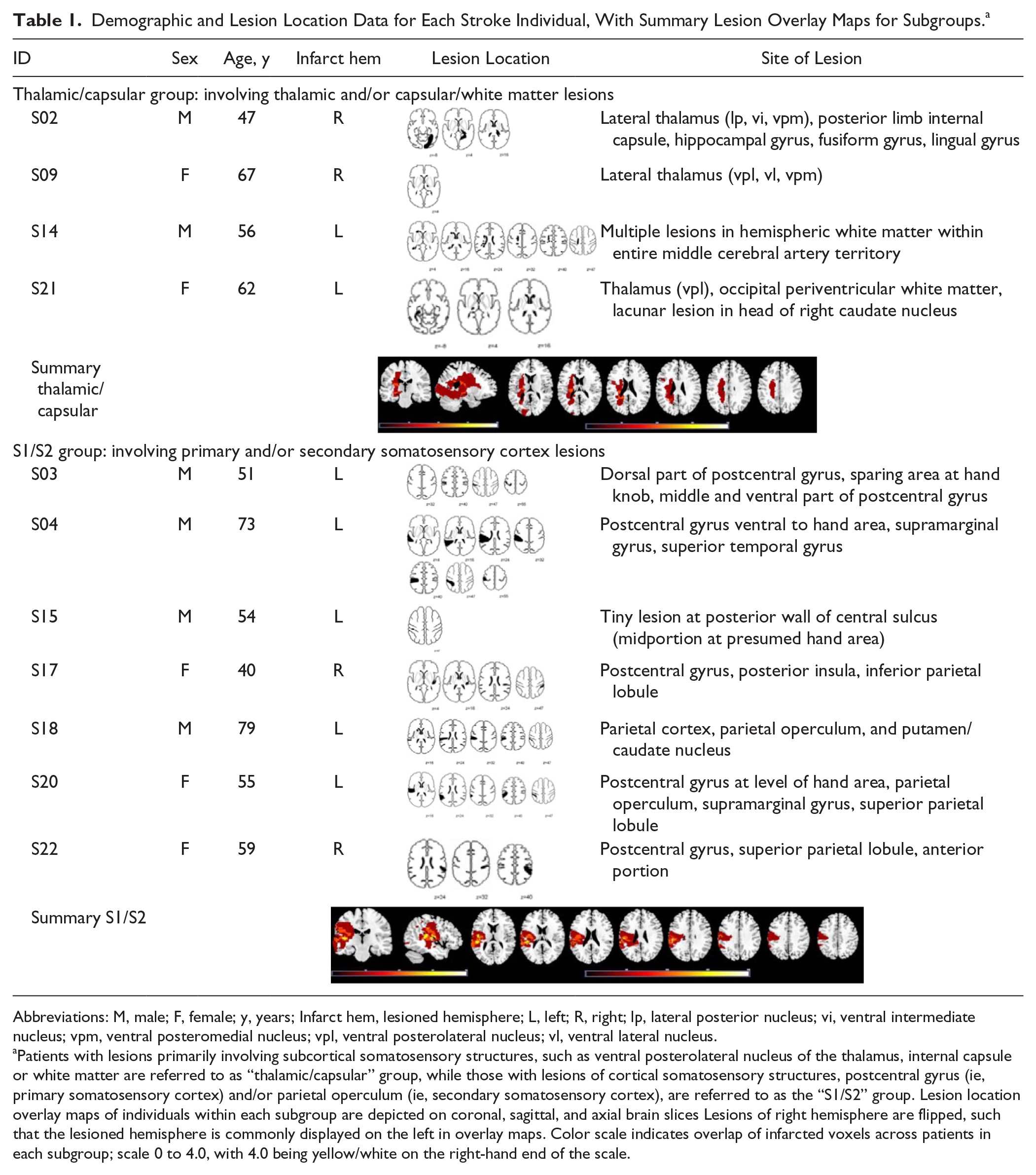
Abbreviations: M, male; F, female; y, years; Infarct hem, lesioned hemisphere; L, left; R, right; lp, lateral posterior nucleus; vi, ventral intermediate nucleus; vpm, ventral posteromedial nucleus; vpl, ventral posterolateral nucleus; vl, ventral lateral nucleus.
Patients with lesions primarily involving subcortical somatosensory structures, such as ventral posterolateral nucleus of the thalamus, internal capsule or white matter are referred to as “thalamic/capsular” group, while those with lesions of cortical somatosensory structures, postcentral gyrus (ie, primary somatosensory cortex) and/or parietal operculum (ie, secondary somatosensory cortex), are referred to as the “S1/S2” group. Lesion location overlay maps of individuals within each subgroup are depicted on coronal, sagittal, and axial brain slices Lesions of right hemisphere are flipped, such that the lesioned hemisphere is commonly displayed on the left in overlay maps. Color scale indicates overlap of infarcted voxels across patients in each subgroup; scale 0 to 4.0, with 4.0 being yellow/white on the right-hand end of the scale.
Background Clinical Scores Preintervention: Neurological Impairment, Motor Function, Related Somatosensory Function.
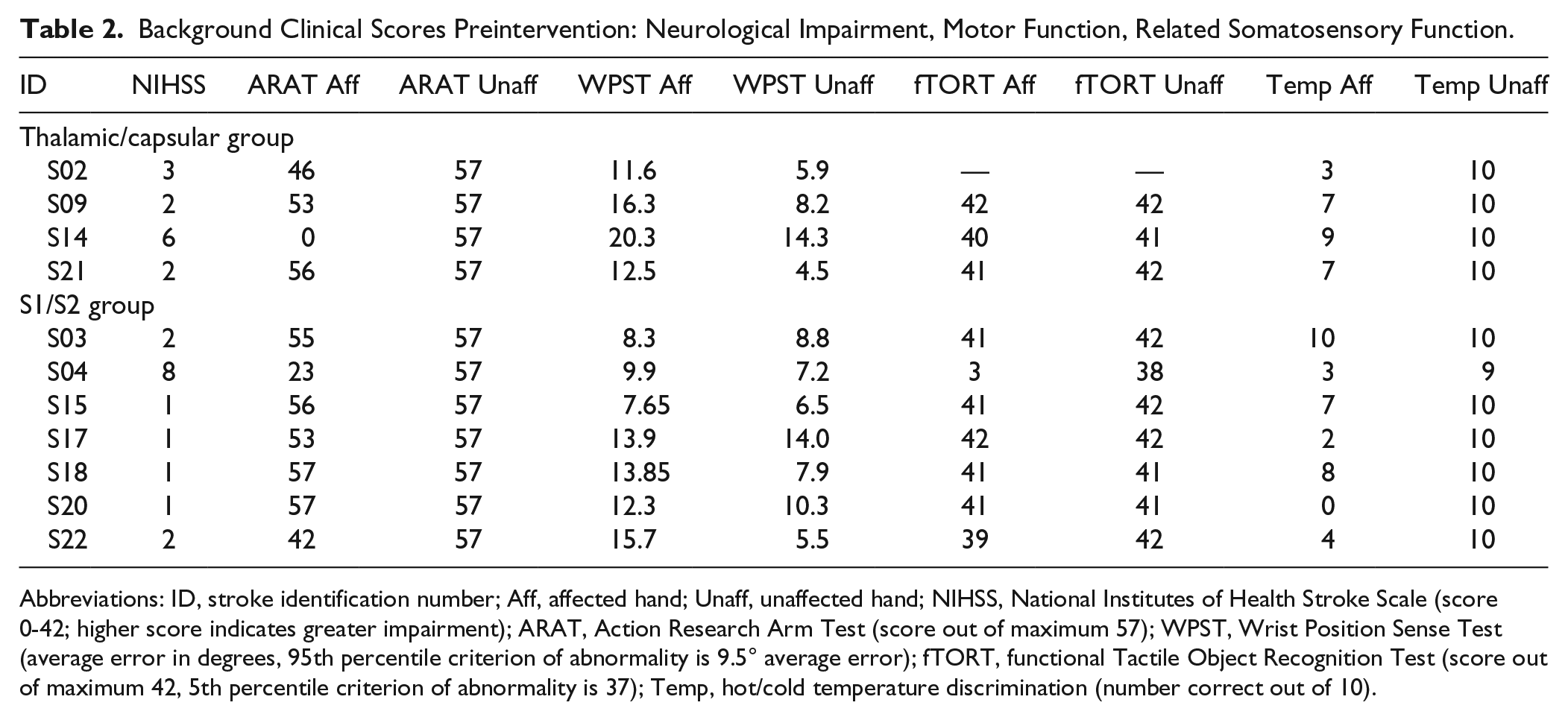
Abbreviations: ID, stroke identification number; Aff, affected hand; Unaff, unaffected hand; NIHSS, National Institutes of Health Stroke Scale (score 0-42; higher score indicates greater impairment); ARAT, Action Research Arm Test (score out of maximum 57); WPST, Wrist Position Sense Test (average error in degrees, 95th percentile criterion of abnormality is 9.5° average error); fTORT, functional Tactile Object Recognition Test (score out of maximum 42, 5th percentile criterion of abnormality is 37); Temp, hot/cold temperature discrimination (number correct out of 10).
Change in Touch Discrimination Over the Intervention Period
Touch detection and discrimination scores and self-reported ability to feel the touch stimulus during the functional MRI scan are presented for each individual pre- and postintervention in Table 3. The mean self-reported visual analogue scale score for concentration during the functional MRI task was: preintervention 8.26 (out of 10; SD = 1.26) for the affected hand and 8.78 (SD = 1.3) for the unaffected hand; postintervention 8.64 (SD = 1.6) for the affected hand, and 8.9 (SD = 1.26) for the unaffected hand.
Touch Discrimination Ability Pre– and Post–Somatosensory Intervention for Patients With Lesions of Thalamic/Capsular Compared With S1/S2 Somatosensory Regions.
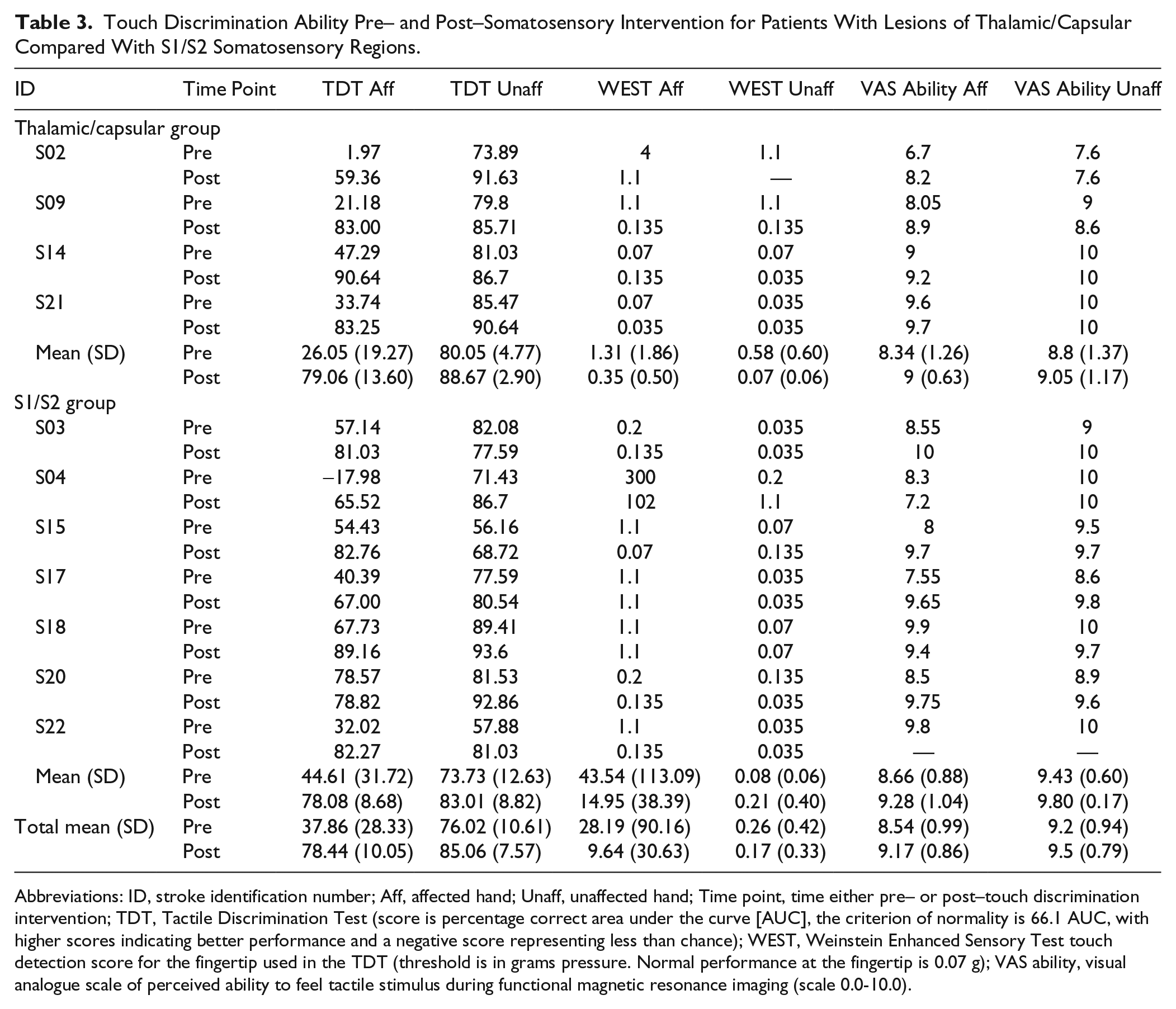
Abbreviations: ID, stroke identification number; Aff, affected hand; Unaff, unaffected hand; Time point, time either pre– or post–touch discrimination intervention; TDT, Tactile Discrimination Test (score is percentage correct area under the curve [AUC], the criterion of normality is 66.1 AUC, with higher scores indicating better performance and a negative score representing less than chance); WEST, Weinstein Enhanced Sensory Test touch detection score for the fingertip used in the TDT (threshold is in grams pressure. Normal performance at the fingertip is 0.07 g); VAS ability, visual analogue scale of perceived ability to feel tactile stimulus during functional magnetic resonance imaging (scale 0.0-10.0).
Patients demonstrated significant improvement in the primary outcome touch discrimination (TDT score) of the affected-hand. Improvement was of a similar magnitude and approaching discrimination within the normal range, as found in our previous clinical trials.24,26 Wilcoxon signed-rank test indicated a significant difference (improvement) in the affected hand, post > pre, (Z = −2.93; P = .003). The mean change in TDT scores for the affected hand of the thalamic/capsular group was 53.02 area under the curve (AUC; SD = 8.21) and for the S1/S2 group was 33.47 AUC (SD = 26.45).
A significant difference (improvement) was also found for the unaffected hand, post > pre (Z = −2.67; P = .008). Mean change was 8.62 AUC (SD = 6.09) for the thalamic/capsular group and 9.29 AUC (SD = 9.10) for the S1/S2 group.
Task-Related Activation Pre– and Post–Touch Intervention
Affected Hand
Preintervention, patients with thalamic/capsular lesions showed an activation cluster involving left ipsilesional supramarginal gyrus, and including the inferior parietal cortex (IPC) and parietal operculum, OP1. Postintervention, significant activation was observed in left ipsilesional insula, extending to the temporal pole, and supramarginal gyrus, including the IPC and OP1 (Figure 1; Table 4). Those with S1/S2 lesions did not show significant common activation preintervention. There was however significant decreased activation (relative to rest) in contralesional superior parietal lobe (SPL), that included contralesional S1 (Brodmann areas [BA] 2, BA3b, and BA3a), and in ipsilesional anterior cingulate cortex postintervention. There were no significant between-group differences pre- or postintervention for the affected hand. However, a trend (P = .047) was shown for greater activation in the ipsilesional insula in the thalamic/capsular compared with the S1/S2 lesion group postintervention.
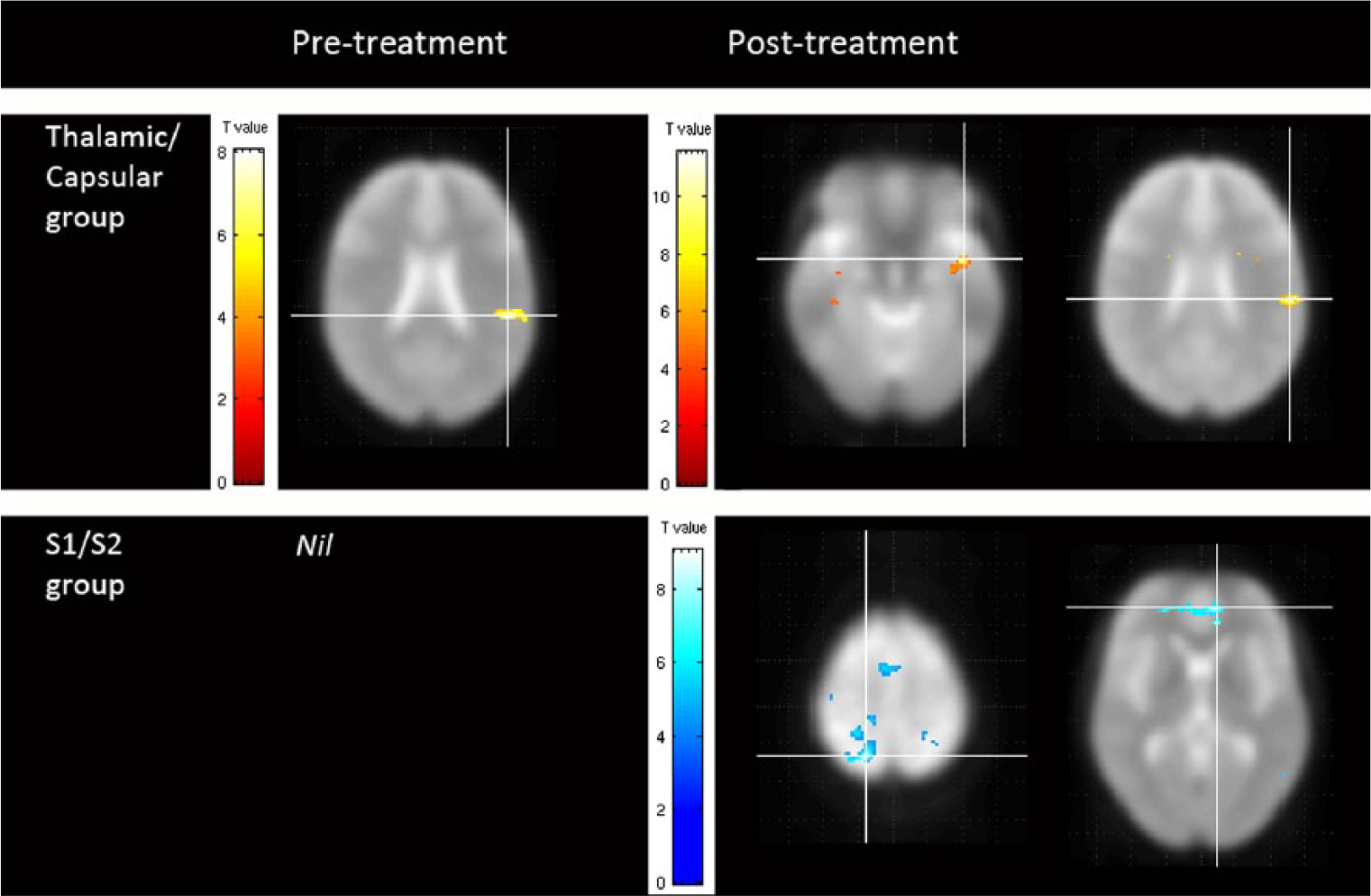
Brain activity associated with touch discrimination of the affected hand pre– and post–touch discrimination training in stroke survivors with interruption to thalamic/capsular compared with S1/S2 somatosensory regions. Axial images show significant clusters of brain activation (hot colors) and deactivation (cold colors). Significant clusters (FWE [familywise error] P < .05) are displayed with crosshairs positioned at the peak voxel within the cluster. The cluster-defining primary threshold for contrasts entered in the analysis is P < .001.
Task-Related Activation Pre- and Postintervention and Change in Task-Related Activation (Post- > Preintervention). a
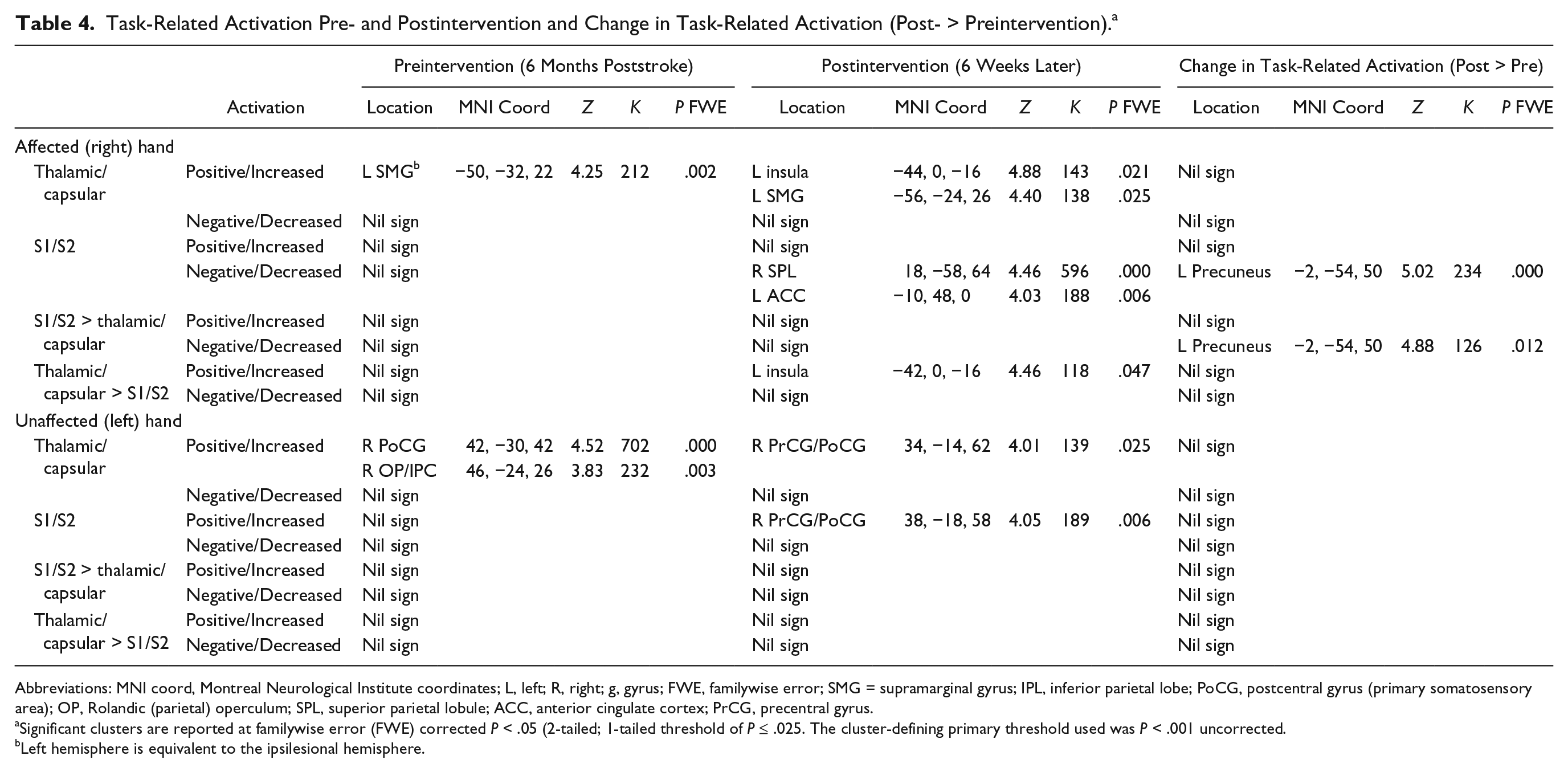
Abbreviations: MNI coord, Montreal Neurological Institute coordinates; L, left; R, right; g, gyrus; FWE, familywise error; SMG = supramarginal gyrus; IPL, inferior parietal lobe; PoCG, postcentral gyrus (primary somatosensory area); OP, Rolandic (parietal) operculum; SPL, superior parietal lobule; ACC, anterior cingulate cortex; PrCG, precentral gyrus.
Significant clusters are reported at familywise error (FWE) corrected P < .05 (2-tailed; 1-tailed threshold of P ≤ .025. The cluster-defining primary threshold used was P < .001 uncorrected.
Left hemisphere is equivalent to the ipsilesional hemisphere.
“Unaffected” Hand
The thalamic/capsular group consistently showed expected activation of the right contralesional primary sensorimotor cortex (ie, the noninfarcted cortex contralateral to the hand stimulated), including BA6, BA2, BA3b, and BA4a, and the right secondary somatosensory cortex, including OP1, OP3, and IPC preintervention. The S1/S2 group also activated in right sensorimotor cortex, including BA2, BA1, BA3b, BA6 pre- and BA4a, postintervention. They did not show significant activation at the corrected level pre-intervention. There were no significant differences between groups pre- or postintervention for the “unaffected” hand. It is noted that the hand commonly referred to as the “unaffected hand” may show some impairment in addition to the impairment traditionally expected for the hand linked with the site of infarct. 22
Change in Task-Related Activation Over the Intervention Period
Affected Hand
The S1/S2 group demonstrated significant change over the intervention period involving the ipsilesional precuneus; such that deactivation of precuneus was greater postintervention compared to pre-intervention. In comparison, the thalamic/capsular group did not show significant common change (Figure 1, Table 4). Change in activation over time was greater for those with S1/S2 lesions, compared to thalamic/capsular lesions, in the left ipsilesional precuneus (Figure 2; Table 4). The magnitude of the difference was large: d = −2.43, with a confidence interval of −0.67 to −3.76. The difference was a relatively greater decreased activation in the precuneus.
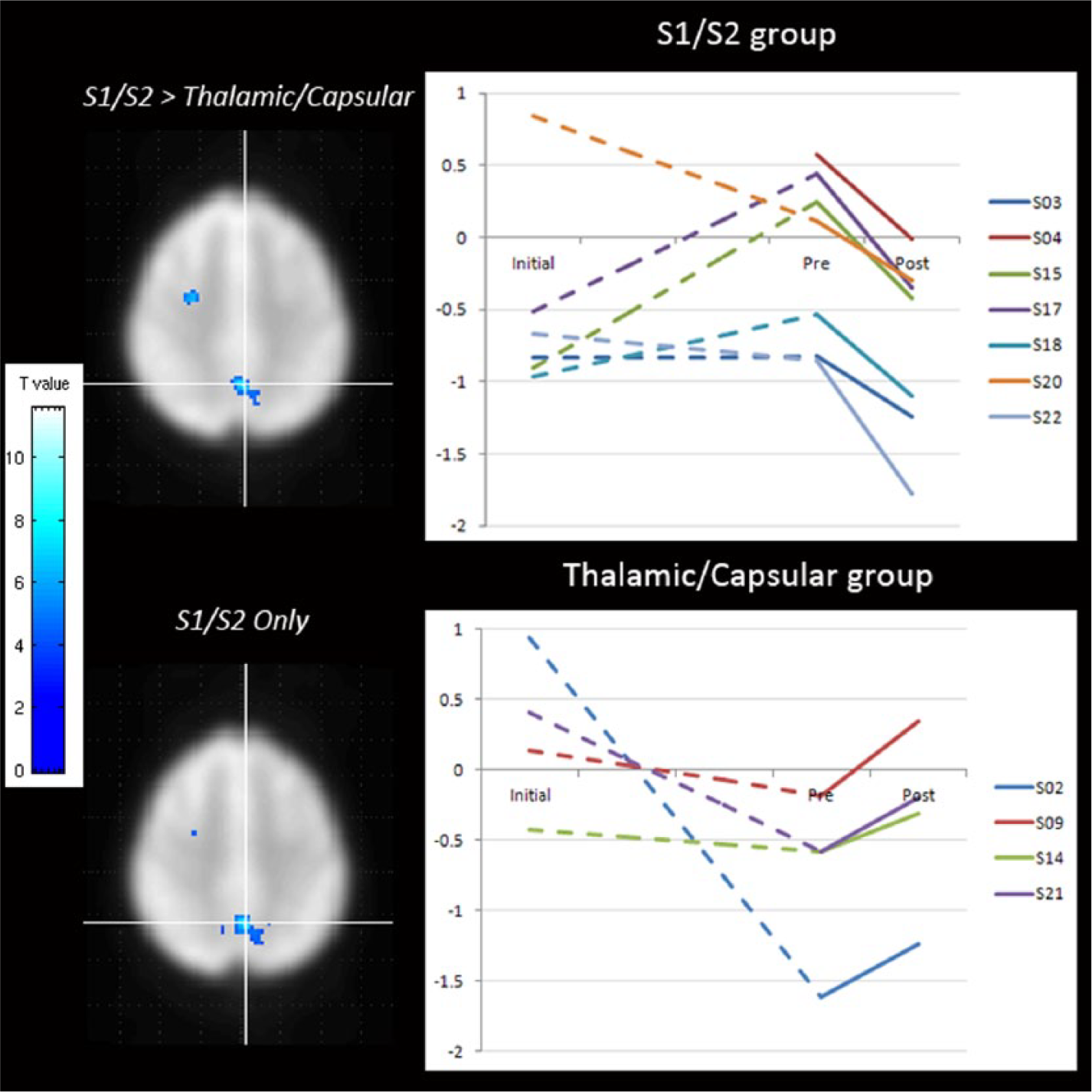
Change in brain activity associated with touch discrimination of the affected-hand over time. Brain maps depict significantly greater change in left, ipsilesional precuneus in those with S1/S2 lesions compared with thalamic/capsular somatosensory lesions (S1/S2 > thalamic/capsular; left panel, top image). The change in deactivation (cold colors) observed indicates greater deactivation postintervention compared with preintervention in those with S1/S2 lesions (S1/S2; left panel, bottom). Significant clusters (FWE [familywise error] P < .05) are depicted on axial brain images at the cluster-defining primary threshold of P < .001 uncorrected. The graphs depict BOLD (blood oxygen–level dependent) signal intensity in the left precuneus site pre- and postintervention and at the initial scan 1-month poststroke. Individuals with S1/S2 lesions show more negative slopes compared with those with thalamic/capsular lesions during the intervention period, and in comparison with the period prior to intervention, from 1 to 6 months poststroke (within-subject inspection).
Unaffected Hand
There was no significant change over time for the unaffected hand in relation to any of the above comparisons.
Post Hoc Analysis of Signal Change
Post hoc analysis examined whether the significant change in BOLD signal in ipsilesional precuneus (S1/S2 group > thalamic/capsular) during the touch-intervention period was a continuation of a preexisting change in signal prior to intervention or represented a change in signal direction/slope with intervention. Signal intensity at this site was graphed for inspection in individuals pre- and postintervention and at 1-month poststroke (mean = 30.8 days; SD = 7.1 days). As illustrated in Figure 2, those with S1/S2 lesions showed a reduction in signal intensity (negative slope) in precuneus over the intervention period. In comparison, slopes were variable and often positive prior to intervention. Change in signal over the extended preintervention (5 months) compared to intervention (6 weeks) intervals was significantly different (Z = −2.37; P = .018). Individuals with thalamic/capsular lesions showed relatively positive slopes during the training interval, but variable and negative slopes over the preintervention interval. There was no significant difference over the 2 intervals for the thalamic/capsular group (Z = −1.21; P > .05).
Discussion
We report the novel finding of involvement of different brain regions and different longitudinal change in activation with touch discrimination of the affected hand following the same touch discrimination training, in stroke patients with S1/S2 cortical lesions compared with subcortical thalamic/capsular somatosensory lesions. While the sample size is small, this study provides novel insight into changes in brain activation within individuals and between groups, relative to site of lesion and experimental manipulation of touch sensation ability via a somatosensory discrimination intervention shown to be effective in controlled clinical trials.24,26
Activation Pre- and Postintervention Following Lesions of Thalamic/Capsular Compared With S1/S2 Somatosensory Regions
Patients with lesions of sensory thalamus and/or internal capsule demonstrated activation in distributed somatosensory cortical regions prior to training followed by activation in left ipsilesional insula, in addition to supramarginal gyrus, postintervention. Our finding supports involvement of ipsilesional cortical somatosensory regions, but not S1 as hypothesized. The activation cluster in supramarginal gyrus, part of the somatosensory association cortex, also included an S2 region, OP1. Activation of ipsilesional insula is of interest given its close proximity to and reciprocal connection with S2, and input from thalamic nuclei.29,40 It is also consistent with activation of the hemisphere contralateral to the stimulated hand in healthy controls and with the role of insula in tactile representation of the body, discrimination and learning.29,41 Previous studies in patients with thalamic lesions, report return of activation in ipsilesional S1 with good recovery.18-20 Activation of S1 was not evident in our patients with thalamic and/or capsular lesions. This may in part be due to low power with the small sample (S1 activation was observed at .001 uncorrected), and that we report activation following targeted sensory rehabilitation, rather than with “good recovery” under spontaneous recovery conditions. While different regions of significant activation were observed at each time point, we did not find a significant common change over time with thalamic/capsular lesions.
Patients with somatosensory lesions of S1/S2 did not show common activation preintervention. This may in part be explained by the extensive and variable location of lesions directly affecting cortical somatosensory regions. However, a relative decreased activation in contralesional S1, within the SPL cluster, was observed postintervention. This may reflect a functionally effective response, given evidence in healthy individuals of negative BOLD changes in a spatially extended S1 region in the same hemisphere as the hand stimulated 42 (ie, equivalent to contralesional S1 in stroke) that is correlated with a simultaneous increase in perception thresholds of the unstimulated finger. These findings highlight the importance of relative activation and deactivation across hemispheres, and their role in touch perception and recovery.
In contrast to changes observed for the affected hand, activation of the “unaffected” hand was consistent with that reported for age-matched healthy controls. There were no between-group differences or significant changes over time.
Change in Activation Over Time and Role of Precuneus
Stroke patients with S1/S2 lesions showed a significant and common change in activation in ipsilesional precuneus, involving greater deactivation over the intervention period. Involvement of precuneus has significance from a number of perspectives. Baseline activity in precuneus is associated with conscious perception, such that baseline activity fluctuations predict somatosensory perception in humans. 43 Precuneus has a role in mental representations of self during rest, 44 shifting attention, 45 and processing of spatial location of sensory information of touch 46 and independent of modality. 47 It also has a role in memory 48 and a wide range of cognitive processes. 49 The precuneus displays one of the highest resting metabolic rates in the brain and is transiently decreased with goal-directed actions. 44 It has widespread connectivity with cortical and subcortical structures 50 and is suggested to be the “core node” of the default mode network. 51 Precuneus has been associated with mental imagery strategies, 44 with poststroke motor recovery, 52 and with somatosensory discrimination in stroke patients with S1/S2 or thalamic/capsular lesions and variable chronicity. 17
Change in ipsilesional precuneus following intervention was greater in those with interruption to S1/S2 compared with thalamic/capsular somatosensory regions. This finding highlights significantly different change over time between groups in response to touch discrimination training, despite a common training protocol. The change in precuneus signal in the S1/S2 group (negative slope) during intervention was not a continuation of a preexisting slope over the 5-month interval of recovery preceding the intervention (Figure 2). The competing hypothesis of a preexisting trajectory associated with natural recovery is therefore unlikely. Moreover, the direction/slope of signal change observed over the intervention period differs between S1/S2 and thalamic/capsular groups, consistent with our main effect.
Structural and functional connections of the precuneus may position it as a viable and accessible candidate for change following S1/S2 lesions. The precuneus is situated between the somatosensory cortex (lesioned) and the cuneus. 44 It comprises 3 regions with axonal links to somatosensory processing areas (eg, links to BA2, parietal operculum, and insula from the sensorimotor anterior region), working memory (angular gyrus and prefrontal areas from the cognitive associate central region), and vision (eg, cuneus from the ventral posterior region). 53 Our coordinates lie in the ventral posterior portion, potentially linked with calibration of touch via vision in training. Precuneus also has connections with subcortical structures such as the thalamus. 44 These subcortical connections may be important for training-facilitated recovery in those with cortical S1/S2 lesions.
Can Rehabilitation Shape Plasticity?
We trained stroke survivors using stimulus-specific touch discrimination training 24 and found improvement in touch discrimination capacity. In healthy individuals stimulus-specific perceptual learning involves focal changes in primary sensory cortex 54 and entrains unique patterns of spontaneous brain activity in the same neural networks specifically recruited by the task. 55 In patients with somatosensory impairment following thalamic/capsular lesions we found activation of ipsilesional insula postintervention, a region known to have a role, together with S1 and S2, in tactile learning and memory.29,41 While activation of S1 was not significant at a group level postintervention, ipsilesional insula is within the distributed neural network required for the task of tactile learning and discrimination.
In comparison, following S1/S2 lesions, cortical somatosensory areas were directly infarcted and thus access to more distributed areas including precuneus and contralesional somatosensory regions appears important, at least in the chronic phase of recovery and following touch discrimination training. Our touch discrimination intervention specifically involves attentive processing of spatially defined textures, use of goal-directed anticipation trials and mental-imagery of what the sensation is supposed to feel like during recalibration via vision and touch.3,24 Each training element involves putative functions of the precuneus. Interestingly, whilst the precuneus changed over the intervention period with S1/S2 lesions, it did not significantly change for those with interruption to thalamic/capsular regions. Perhaps access to this additional region was not needed given that cortical somatosensory regions typically accessed for this task were still intact.
Participants in our study demonstrated differences in brain reorganization (brain regions activated and change in activation over time) following intervention, despite the fact that they received the same training, and showed similar clinical improvement. It appears that the training (behavioral experience) provided a challenge and the brain reorganized in a manner influenced by interruption to specific somatosensory structures and access to brain networks related to somatosensory processing and training procedures. This begs the question: Should training be modified according to lesion location to specifically target the viable networks typically recruited for this task and/or related to training? Alternatively, in providing the sensory stimulus and training conditions that require somatosensory and related networks, does the individual respond accordingly with brain regions and networks that are accessible to them? This question has major significance in relation to personalized medicine and individual targeting of therapy. A recent study revealed that different approaches to motor rehabilitation are associated with different brain adaptation. 56 In our study, different types of brain adaptation were observed with the same approach to sensory rehabilitation in patients with interruption to different parts of the somatosensory network. Systematic investigation using different interventions and patient characteristics, including individual differences in interruption to different parts of the somatosensory system, will help to further this important field of inquiry.
A number of limitations are linked with this field of research and our study. First, the sample size is small and we did not have a separate control group. Grouping participants by lesion location exacerbated this limitation. However, this distinction could not be overlooked given our research question and evidence of the impact of lesion site on brain activation and reorganisation.13,14 While we compared patients with S1/S2 versus thalamic/capsular somatosensory lesions, clinically patients may present with mixed lesions, involving relay and cortical structures within the somatosensory system. This may further affect regions available to be accessed during sensory rehabilitation. Finally, BOLD signal may be influenced by medication and compromised blood flow in stroke. 57 While patients were not excluded on the basis of medication, readers need to be mindful of these factors when interpreting results.
In conclusion, we provide novel evidence that change in activation following touch discrimination training differs in stroke survivors with disruption to different parts of the somatosensory network (ie, thalamic/capsular compared with S1/S2 cortical somatosensory lesions), despite the same rehabilitation intervention and similar improvement. This has theoretical and clinical implication for the role of rehabilitation in driving neural plastic changes in the brain as well as the individual’s response to therapy based on brain regions that may be accessed during therapy.
Footnotes
Acknowledgements
The authors wish to acknowledge and thank the participants involved in the study and the Neurorehabilitation and Recovery Research Team at the Florey Institute of Neuroscience and Mental Health. Matthew Harvey and Pierrick Bourgeat assisted with analysis of imaging data, with additional scientific and technical assistance from the National Imaging Facility at the Florey Institute.
Authors’ Note
The funding sources had no role in conduct of the study or writing of the report.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge financial support for the research from the National Health and Medical Research Council (NHMRC) of Australia (Grants 307902 and 1022694); a NHMRC Career Development Award to LMC (307905); an Australian Research Council Future Fellowship awarded to LMC (FT0992299); a James S. McDonnell Foundation 21st Century Science Initiative in Cognitive Rehabilitation–Collaborative Award (220020413) to LMC; fellowship funding from the National Imaging Facility, Australia to DFA; the Dunlop foundation; and the Victorian Government’s Operational Infrastructure Support Program.
