Abstract
One hour of 20-Hz continuous electrical stimulation (ES) applied at the time of injury promotes the regeneration of axons in cut peripheral nerves. A more robust enhancement of peripheral axon regeneration is achieved by 2 weeks of daily treadmill exercise. We investigated whether repeated applications of brief ES (mES) would be more effective in promoting regeneration than a single application. Sciatic nerves of C57B6 mice were cut and repaired by end-to-end anastomosis. At that time and every third day for 2 weeks, the repaired nerve was stimulated for 1 hour at 20 Hz. In controls, injured mice were either untreated or treated with ES only once. Direct muscle responses recorded from reinnervated muscles in awake animals were observed earlier both in mice treated with ES and mES than untreated controls. Their amplitudes increased progressively over the post transection study period, but the rate of this progression was increased significantly only in animals treated once with ES. Monosynaptic H reflexes recovered to pretransection levels in both untreated and singly treated mice but in the animals treated repeatedly, they were maintained at more than twice that of the same reflexes recorded prior to injury. In anatomical analyses, both excitatory and inhibitory synaptic contacts with the cell bodies of injured motoneurons, including those expressing the vesicular glutamate transporter 1 (VGLUT1), were sustained in mice treated repeatedly but not in singly treated or untreated mice. Repeated ES does not enhance the rate of restoration of functional muscle reinnervation and results in the retention of exaggerated reflexes.
Introduction
Injuries to peripheral nerves are common,1,2 but full functional recovery from traumatic peripheral nerve injuries is rare. 3 We 4 and others5,6 have advocated for experimental treatments for peripheral nerve injury that promote axon regeneration by increasing neuronal activity, either by application of electrical stimulation (ES) 7 or moderate daily exercise.5,8,9 The most commonly used ES treatment is a single application of 1 hour of continuous stimulation at 20 Hz at the time of surgical repair of cut nerves. This stimulation activates the injured neurons and promotes axon elongation across the surgical repair site.4,10 However, it is less effective at inducing subsequent elongation of regenerating axons to reach their targets than 2 weeks of exercise.4,11 A prolonged and continuous application of ES did not enhance the regeneration of sensory 12 or motor 6 axons after peripheral nerve injury. Repeated daily applications of 1 hour of ES 13 produced a greater enhancement of motor axon regeneration than observed with a single application of ES at the time of nerve repair, especially if the ES was delivered for 6 consecutive days. These observations motivated us to hypothesize that repeated treatments of 1 hour of 20-Hz ES applied less frequently but over the same time course as treadmill exercise might be more effective in promoting axon regeneration/elongation than either a single or continuous application. Thus, we investigated the effectiveness of application of ES every third day (mES) on axon regeneration after peripheral nerve injury.
An additional impediment to functional recovery following peripheral nerve injury is a reorganization of spinal circuitry. Following nerve transection injuries, synapses on the somata and proximal most dendrites of motoneurons are withdrawn.14-16 Many of the withdrawn synaptic inputs are restored, especially if regenerating motor axons are successful in reinnervating muscle. 15 However, synaptic inputs onto motoneurons expressing the vesicular glutamate transporter 1 (VGLUT1) are not restored 15 and the terminal branches of these inputs are withdrawn progressively from the ventral horn. 14 Because VGLUT1+ inputs in the ventral horn are overwhelmingly from group Ia afferent neurons innervating muscle spindles, 17 their permanent loss could explain the permanent loss of the stretch reflex15,18,19 and reduction of the electrically evoked H reflex 9 observed in reinnervated muscles. Following sciatic nerve transection and repair, this withdrawal of synaptic inputs and the reduction in efficacy of the H reflex is abrogated with exercise20,21 in a brain-derived neurotrophic factor (BDNF)–dependent manner. Selective BDNF knockout in motoneurons blocks this effect of exercise. 22 Whether a similar effect occurs with ES is not known. A second goal of this study was to evaluate whether single or repeated applications of ES would affect synaptic inputs onto axotomized motoneurons.
Methods
All surgical procedures conducted using animals were approved by the Institutional Animal Care and Use Committee of Emory University (Protocol No. 2003261) and conformed to the Guidelines for the Use of Animals in Research of the Society for Neuroscience. All surgical procedures were performed under aseptic conditions. Numbers of mice in each experimental group were chosen based on an a priori power sample size estimate, using a power of 0.8, α = .05, and the interanimal variability and effect sizes observed in our previous studies.23,24
Surgical Procedures and Treatments
Transection and surgical repair of the sciatic nerve (Figure 1A), a well-established peripheral nerve injury model, was used in mice (C57B6) of both sexes. To investigate axon regeneration and functional recovery after this injury, mice were surgically implanted with stimulating and recording hardware. Under general anesthesia (ketamine [80 mg/kg] and xylazine [10 mg/kg]), a cuff electrode was placed around the right sciatic nerve in the mid-thigh to be used for stimulation (Figure 1A). These cuffs were constructed from small lengths of Silastic tubing (Dow Corning, Part No. 508-006), into which lengths of Teflon-coated stranded stainless-steel wires (AWG size 40, Cooner Wire, Chatsworth, CA; part number AS631) were sewn and then insulated with SILASTIC Medical Adhesive Silicone, Type A (Dow Corning). Four pairs of the same fine wire were used as electromyography (EMG) electrodes and were implanted into the gastrocnemius (GAST) and tibialis anterior (TA) muscles on each side of the animal (Figure 1A). All the wires from these implants were led subcutaneously to a connector plug (Omnetics, Minneapolis MN, Part A79110-001) mounted on the animal’s head with 2 stainless steel bone screws and dental acrylic (Figure 1B).
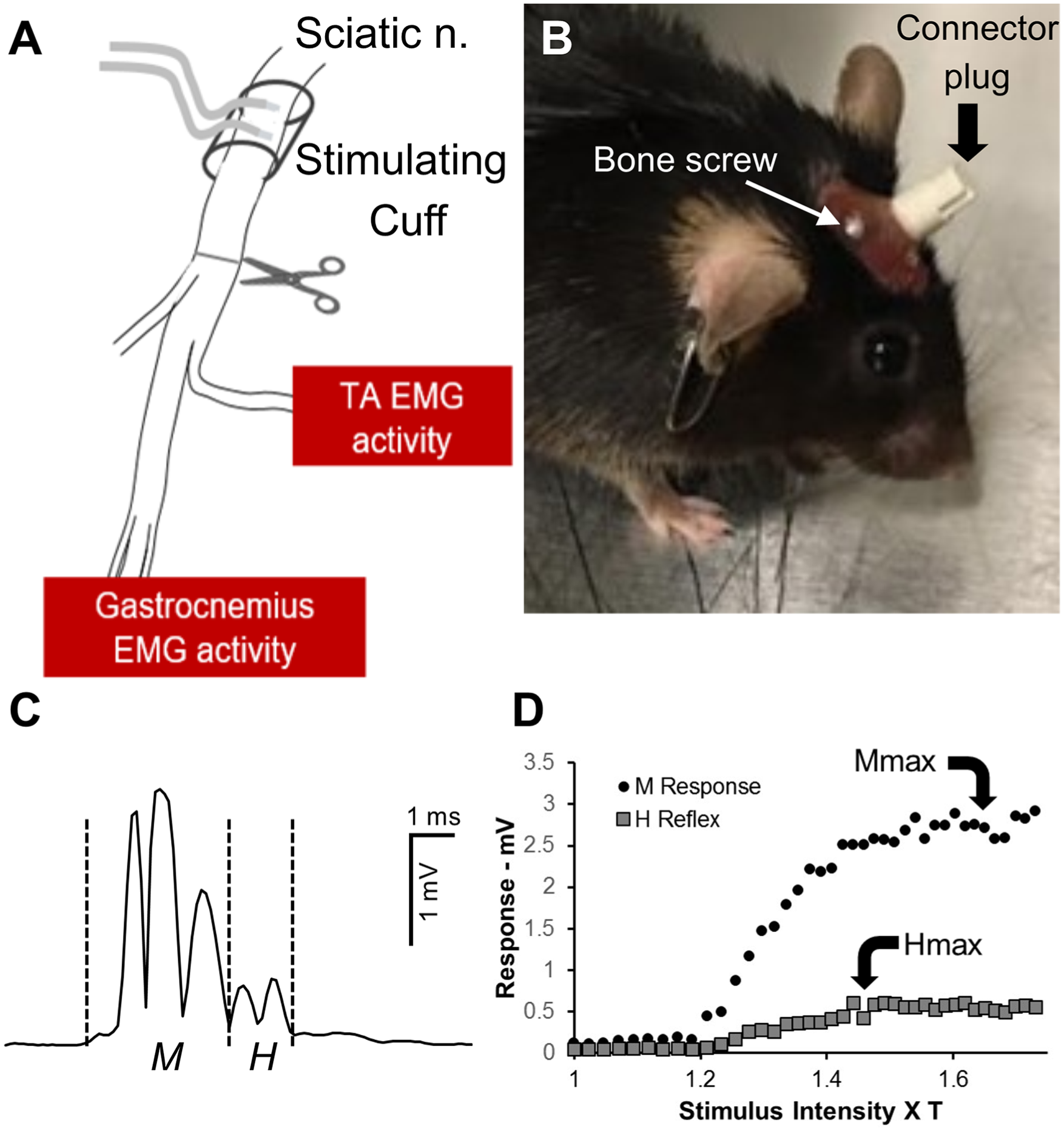
(A) Diagram of experimental setup. Animals were chronically implanted with electromyography (EMG) electrodes placed bilaterally in the tibialis anterior (TA) and gastrocnemius muscles and a bipolar cuff electrode around the proximal sciatic nerve. Electrode wires were led subcutaneously to a connector plug mounted on the animal’s head using small stainless-steel bone screws and epoxy (B). Following collection of baseline data, the sciatic nerve was cut and repaired at the site shown by the scissors in panel A. (C) A representative trace of stimulus evoked rectified EMG activity recorded from the gastrocnemius muscle of an intact mouse. Vertical dashed lines mark the windows for the direct muscle (M) response and monosynaptic H reflex recorded. (D) Recruitment curves for the responses noted in CA. The amplitude of the M responses (circles) increases with increasing stimulus intensity, expressed as multiples of M response threshold. The amplitude of the H reflex (squares) peaks at earlier stimulus intensities.
One week after implant surgery, pretransection EMG data were collected. In each such recording session, implanted mice were connected to recording amplifiers and an electrical stimulator through the head-mounted connector. In awake animals, ongoing EMG activity in the right GAST muscle was sampled at 10 kHz using a laboratory computer system and custom Labview software. When the rectified amplitude of this background activity fell within a user-defined voltage range, typically 50 to 100 µV, for 20 ms, the computer delivered a single, 0.3-ms duration, constant voltage stimulus pulse to the cuff electrode on the sciatic nerve. The EMG activity evoked from the implanted muscles by that stimulation was recorded from 20 ms prior to the stimulus pulse to 100 ms after the stimulus, and stored on disc. An example of an evoked direct muscle (M) response and a monosynaptic H reflex, recorded from the GAST muscles of an intact mouse is shown in Figure 1C. In each experiment, stimulus intensity was increased linearly in approximately 1% increments from subthreshold to generate a complete recruitment curve for the muscles on the right sides of the animals (Figure 1D). To avoid fatigue, stimuli were delivered no more frequently than once every three seconds. 25 Approximately 100 stimulus presentations were required to generate the recruitment curves. Once these data had been collected, the mice were reanesthetized and the sciatic nerve was cut ca. 1 mm below the cuff electrode and repaired by simple end-to-end anastomosis using fibrin glue. 26 In one group of mice (n = 4), electrical stimulation (20 Hz for 1 hour, 0.3-ms duration pulses), delivered at twice the intensity needed to evoke a muscle response, was applied to the anesthetized mice, through the implanted cuff electrode, immediately following nerve repair. In another group of 4 mice, this stimulation protocol was applied and then repeated in isoflurane anesthetized animals every third day for 2 weeks from the transection date. Others have shown that one hour of applied ES results in a rapid, but transient increase in expression of neuronal BDNF, 7 and that this increase was required for the enhancing effect of ES on axon regeneration. 27 Treating with ES every third day for 2 weeks was an effort to maintain elevated expression of BDNF in sensory and motor neurons during the same treatment period as used in treadmill exercise. In a third group of 4 implanted mice, the sciatic nerve was cut and repaired and no treatment was applied.
Electrophysiological Data Analysis
Stimulus evoked EMG data were collected from awake animals, as described above, at different times after injury. Data from untreated control mice were obtained at 3, 4, 6, 8, 10, and 12 weeks after injury. No evoked response could be recorded from these mice at earlier survival times. Recordings from mice in the mES group were made at 2, 3, 4, 5, 6, 8, 10, and 12 weeks after injury. In the ES group, recordings were made at 2, 3, 4, 5, 6, 8, and 10 weeks after injury. Recordings at longer (12 weeks) survival times in these mice were not possible because of multiple implant failures. These recordings were used to closely monitor the recovery of direct muscle EMG responses of the GAST and TA muscles and of reflex activity evoked by sciatic nerve stimulation. Short latency M responses and slightly longer latency H reflexes could be recorded. The latency and duration of the different responses changes during the period of reinnervation, as we have described previously. 25 Thus the poststimulus time windows for measurement of these responses had to be adjusted empirically. The shortest latency triphasic action potential recorded was assumed to be an M response and a slightly longer latency response the H reflex. Because complete recruitment curves were generated at each testing (see above), the responses at different stimulus intensities were also considered in determining the time windows for measurement of responses. The amplitude of M responses increases with increasing stimulus intensity until a plateau is reached (Figure 1D: Mmax). The amplitude of the H reflex reaches this plateau at lower stimulus intensities (Figure 1D: Hmax) and sometimes declines, as antidromically activated motoneurons become refractory to recruitment into the reflex. 25 The average rectified voltage within time windows for these responses was measured as the amplitude of these responses. In all the reinnervated muscles, this amplitude was scaled to the amplitude of the corresponding response recorded from that muscle prior to nerve injury.
Scaled maximal M response amplitudes recorded from mice treated with a single application of 20-Hz ES, from mice treated repeatedly (mES), and from untreated (UT) controls all increased linearly over the study period, as we have described previously for rats. 23 Significance of differences in the time course of recovery of these responses was evaluated in a pairwise manner using multiple linear regression analysis. Significant differences (P < .05) between groups in the slopes and intercepts of the regression lines fit to the data were considered evidence for an effect of the particular treatment (ES or mES).
To investigate the effect of repeated application of ES on the recovery of the H reflex, the ratio of the amplitude of the largest evoked H reflex to that of the largest evoked M response was determined at each time measured. This HMax/MMax ratio is thought to be a rough estimate of the proportion of the available motoneuron pool that is recruited into the H reflex. 28 To compare this H reflex efficacy at different times following sciatic nerve transection and repair, HMax/MMax ratios were scaled to the same ratio determined prior to transection. Significance of differences in scaled H reflex efficacy between the 3 treatment groups was evaluated at 10 weeks after nerve injury, using analysis of variance (ANOVA) and post hoc paired testing (Tukey-Kramer minimum significant difference).
Neuroanatomical Evaluations of Synaptic Inputs to Motoneurons
To study the effects of ES treatments on synaptic inputs to the axotomized motoneurons, animals were implanted with stimulating and recording electrodes as described above. At the time of this implantation surgery, motoneurons were labeled by injecting the tibialis anterior muscle with one microliter of a 1% solution of cholera toxin-B conjugated to Alexafluor 555. One week later, the mice were anesthetized and the common fibular nerve was cut, using sharp scissors, just distal to its branching from the sciatic nerve. The 2 cut segments of the nerve were then aligned and secured in place using fibrin glue, as described above. At this time, a single application of 1 hour of 20 Hz ES was applied to the sciatic nerve of one group of 4 mice. In a second group of 4 mice, the 20-Hz ES was applied to anesthetized mice every third day for 2 weeks, as described above. For controls, the same labeling and injury procedure was applied to 4 mice, but they remained untreated throughout the 2-week survival period. Comparisons were also made to data from a group of 6 intact mice whose common fibular motoneurons had been labeled by tracer injection.
At the end of the 2-week survival period, the mice were euthanized with an overdose of pentobarbital and perfused transcardially with saline, followed by periodate-lysate-paraformaldehyde solution. 29 The lumbar spinal cords were removed, and after cryopreservation in 20% sucrose solution, they were serially sectioned at 20 μm on a cryostat in the transverse plane. All sections were mounted onto charged slides and then reacted for the demonstration of 2 different synapse-associated antigens, according to protocols described in more detail in our previous publications.4,20,22,30 Antibodies to VGLUT1 (Synaptic Systems GmbH number TO30326,Gӧttingen, Germany, diluted 1:2000) and GAD67 (Millipore number MAB5406, Billerica, MA, diluted 1:200) were used to study contacts made by muscle spindle group Ia afferent axons and axons of CNS inhibitory neurons, respectively, onto the somata and proximal-most dendrites of retrogradely labeled motoneurons (Figure 5A).
Twenty motoneurons in each spinal cord were selected for confocal imaging if the retrograde label filled the soma and proximal-most dendrites of the neuron. Optical sections (10 µm thick) were obtained that contained the portion of the somata of the selected cells that contained a nucleus. A 5-µm wide region of interest (ROI) around the perimeter of the each of the cells studied in these images was generated using the thresholding feature of ImageJ software. The intensity of immunofluorescence for VGLUT1 and GAD67 beneath this ROI was then measured as a profile plot (Figure 5B). For each profile plot, a threshold was set as the mean intensity plus one standard deviation (horizontal dashed lines in Figure 5B). Immunofluorescence intensities above this threshold were assumed to be due to contacts within 5 µm of the labeled motoneurons by structures containing these synapse-associated antigens. For each motoneuron studied, the proportion of the total cell perimeter above this threshold was determined as percent synaptic coverage. We 30 and others31,32 have validated such measurement of the extent of synaptic inputs onto motoneurons. Differences in mean coverage by VGLUT1+ and GAD67+ inputs between groups was evaluated using 1-way ANOVA, with post hoc paired (Tukey-Kramer minimum significant difference) testing where appropriate.
Results
Effects of ES on Axon Regeneration and Muscle Reinnervation
Successful motor axon regeneration and muscle reinnervation were studied electrophysiologically by measuring the amplitudes of the direct muscle or M responses to sciatic nerve stimulation over an extended postinjury period. In untreated control mice, an evoked EMG potential that was significantly greater than background was first noted at three weeks after sciatic nerve transection and repair. In both ES and mES groups, a detectable M response could be recorded one week earlier. In intact mice, clear poststimulus time windows for M responses (1.0-3.2 ms for GAST and 1.2-3.5 ms for TA) and H reflexes (4.7-6.6 ms for GAST and 4.8-6.8 ms for TA) were observed. These values are in the same range as has been described by others in mice using a similar recording approach. 33 In reinnervated muscles, the latency between stimulus and the onset of these responses was initially much longer, as we 23 and others 28 have described for rats (Figure 2). This M response latency decreased markedly during the postinjury recovery period but remained significantly longer than the preinjury latency even as long as 12 weeks after injury (Figure 2). For both TA and GAST, there was no significant difference in this pattern of maturation of the M response latency of reinnervated muscles in the different treatment groups (Figure 2).
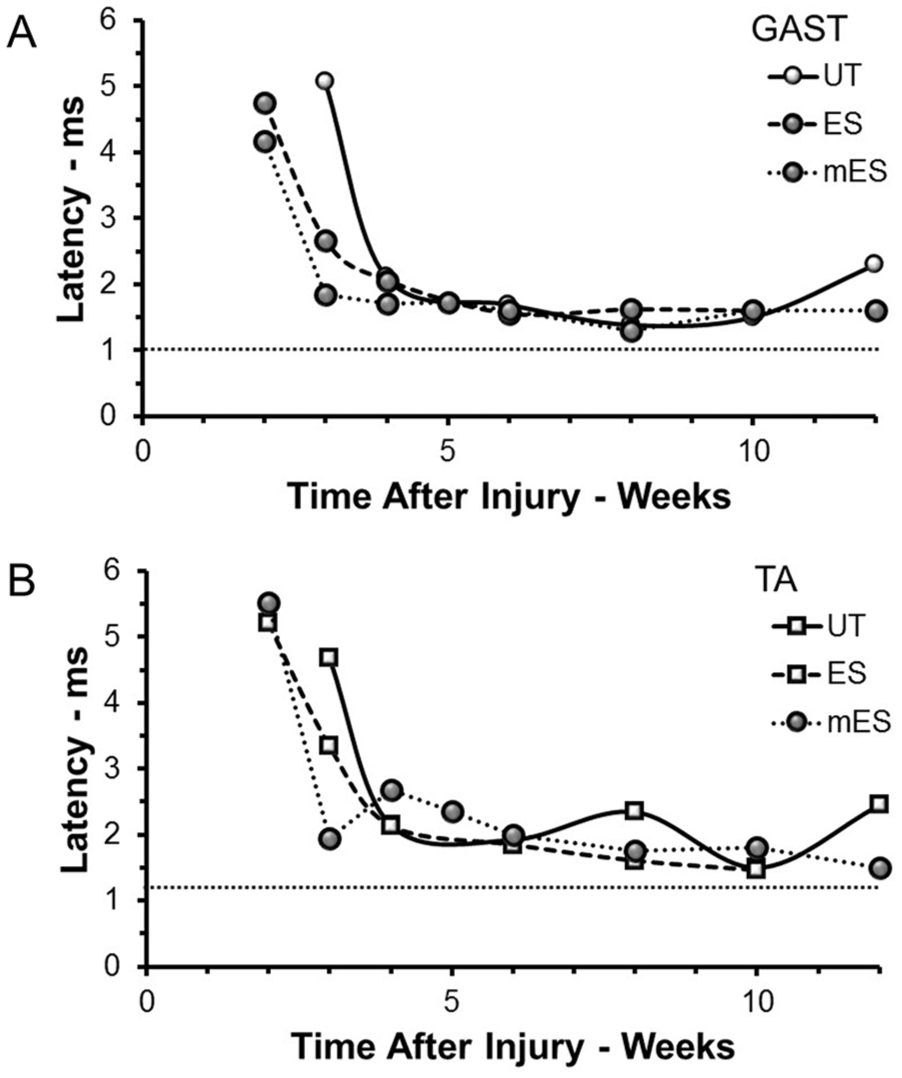
Changes in M response latency (time between stimulus and onset of M response) as a function of postinjury times are shown for recordings from GAST (A) and TA (B) muscles from mice of the three treatment groups studied: untreated (UT); treated once with ES (ES), or treated repeated times with ES (mES). Each symbol represents the average latencies measured in at least three mice at each time in each group. The first M response that could be evoked from reinnervated muscles in the UT group occurred later than in the other groups. The horizontal dashed line in each plot is the mean latency found in a group of 5 intact mice. GAST, gastrocnemius; TA, tibialis anterior; ES, electrical stimulation.
Using chronically implanted stimulating and recording electrodes, M responses were evoked in the GAST and TA muscles at different postinjury times in awake mice that had been treated once (ES) or repeatedly (mES) with 20 Hz electrical stimulation, as well as in untreated controls (UT). Because some implants failed over time in each of these groups, consistent recordings at times longer than 10 weeks postinjury could be obtained from only three of the implanted mice in the mES and UT groups. In the mice treated only once with ES, no recordings were available at survival times longer than 10 weeks.
For both GAST (Figure 3A) and TA (Figure 3B), the amplitude of largest M response recorded (MMax), when scaled to the amplitude of MMax recorded prior to injury, increased linearly over the study period in all 3 treatment groups. Using multiple linear regression analysis, the slopes of all the lines fitted to the data were significant and the correlation coefficients were very high. In both muscles, the slope of the regression line for mice treated once with ES was much larger than that of the other 2 groups of mice. This difference was significant, relative both to the UT mice and mice in the mES group for both GAST (P < .02, P < .01) and TA (P < .01, P < .01). Slopes of the lines for mice treated repeatedly with ES were not significantly different from those of untreated controls for both GAST or TA. Single treatments with ES produced a significant increase in the rate at which the M response was restored, relative to untreated controls, but repeated ES treatments did not. For both GAST and TA, the values of the average scaled M responses at the 10 week recovery time were significantly greater for the mice treated with ES one time than for untreated mice or mice treated repeatedly with ES (ANOVA, F2, 8 = 8.81, P < .01 for GAST, F2, 8 = 20.23, P < .01 for TA). No significant difference was found at this recovery time between the untreated mice and the mice that had been treated repeated times with ES.
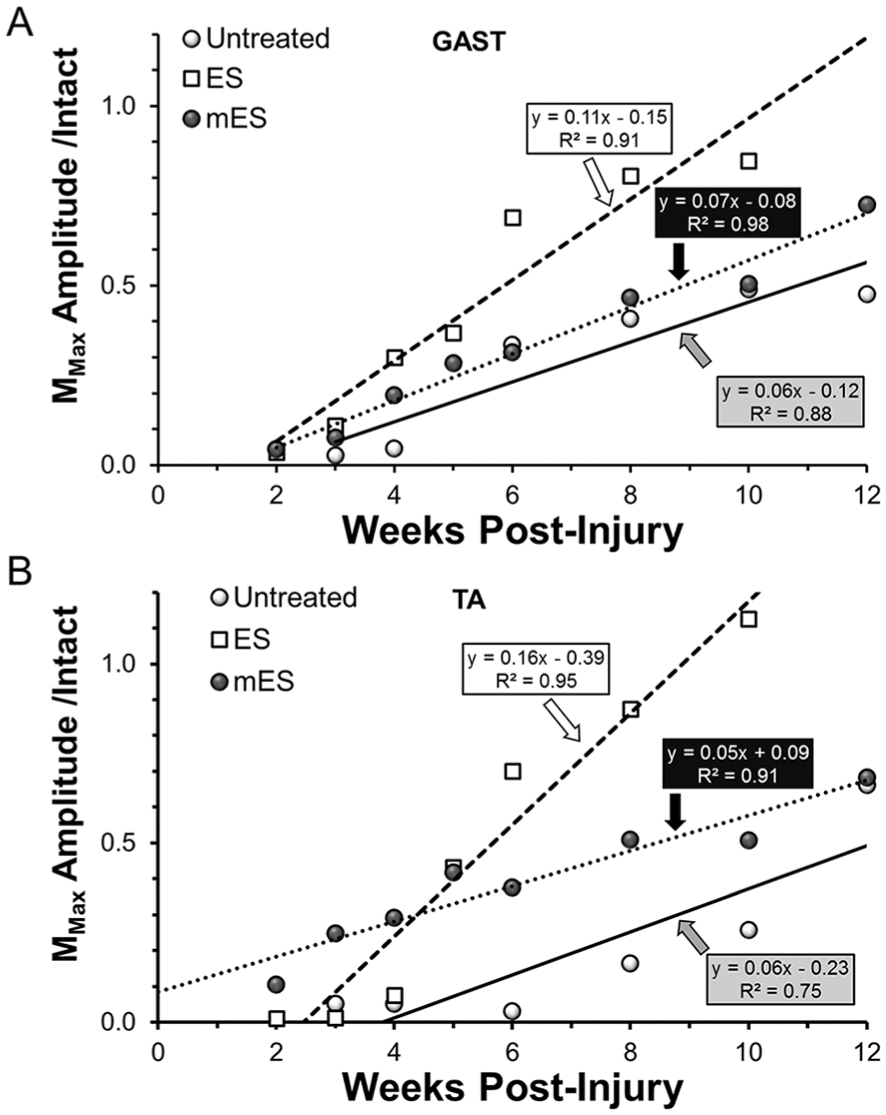
Maximally evoked direct muscle responses (MMax) to stimulation of the sciatic nerve proximal to a transection injury were studied in the same mice at several times following the injury. Changes in the amplitudes of these responses at different postinjury times, scaled to the pretransection amplitudes, are shown for untreated (UT) mice and mice treated with an hour of electrical stimulation either once (ES) or every third day for 2 weeks (mES), for (A) gastrocnemius (GAST) and (B) tibialis anterior (TA) muscles. Each symbol represents the average of values measured from at least 3 mice. Regression lines were fit to the data points using a least squares method. The formulae for the lines and correlation coefficients are shown next to each.
Effects of ES on the Efficacy of the H Reflex in Reinnervated Muscles
Monosynaptic H reflexes were elicited in awake mice in response to sciatic nerve stimulation at the same times as the M responses described above. The time course of scaled Hmax/Mmax ratios following peripheral nerve injury are shown for GAST and TA muscles in the 2 panels in Figure 4A. Horizontal dashed lines at unity in each panel mark the Hmax/Mmax ratio found in intact mice. For both muscles and in all 3 groups studied, the scaled Hmax/Mmax ratio was strikingly enlarged at early postinjury times but declined thereafter. Enlarged H reflexes during the period of early muscle reinnervation have been reported previously in rats.23,28 In untreated mice, the Hmax/Mmax ratio returned to pretransection levels by about 6 weeks after injury and remained not significantly different from that amount. In mice treated with a single application of 1 hour of 20-Hz ES, a more gradual decline in scaled Hmax/Mmax ratio was noted, reaching pretransection levels by 10 weeks after injury. In mice treated with repeated applications of ES, the scaled Hmax/Mmax ratio also declined but remained larger than found in intact mice. At ten weeks after nerve injury, the latest time available for all groups, Hmax/Mmax ratios in mice treated repeatedly with ES were significantly (F2, 7 = 20.174, P < .01, and F2, 7 = 34.05, P < .01, for GAST and TA, respectively) larger than those measured in both untreated mice and mice treated once with ES (Figure 4B). Thus, repeated treatments with ES resulted in a retention of exaggerated H reflexes in reinnervated muscles.
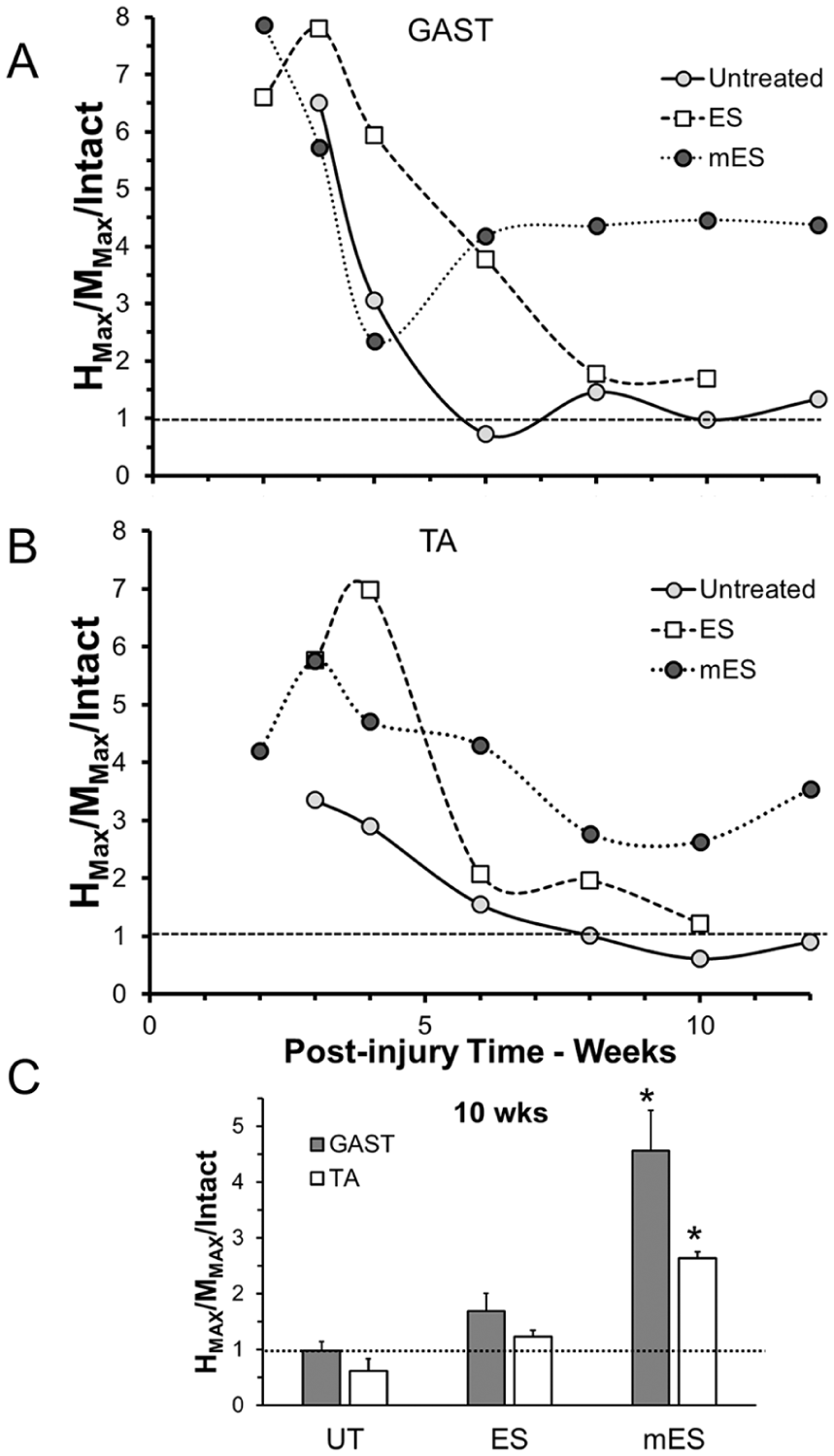
Monosynaptic H reflexes were recorded in response to stimulation of the sciatic nerve proximal to a transection injury at multiple post-injury times in untreated (UT) mice and mice treated with an hour of electrical stimulation either once (ES) or every third day for 2 weeks (mES). The amplitudes of the largest responses at each time were expressed relative to the amplitude of the maximal M response recorded during that experiment. This Hmax/Mmax ratio was then scaled to the same ratio recorded prior to injury. Changes in this scaled Hmax/Mmax ratio over post-injury time are shown in A for gastrocnemius (GAST, upper graph) and tibialis anterior (TA, lower graph) muscles. Horizontal dotted lines in each graph at unity mark the values observed in intact mice. In panel B, the mean (±SEM, n = 3) scaled Hmax/Mmax ratios at 10 weeks postinjury are shown for the 2 muscles in the 3 groups. *P < .05 versus UT, ES.
Effects of ES on Synaptic Inputs Onto Axotomized Motoneurons
We evaluated the extent of contacts made by structures immunoreactive to either of 2 synapse-associated molecules onto the somata and proximal-most dendrites of axotomized motoneurons (Figure 5A and B). We assumed that the percentage of the perimeter of the motoneurons contacted by these VGLUT1- and GAD67-immunoreactive structures was a measure of the extent of inputs to these parts of the motoneurons by these excitatory and inhibitory inputs. In Figure 5C, the mean (±SEM) synaptic coverage by these 2 types of inputs is shown for intact mice and for mice in the different treatment groups studied 2 weeks after sciatic nerve transection and repair. The 2-week survival period was chosen because synaptic withdrawal after peripheral nerve injury is well established at that time and study at this time enabled direct comparison to previously published studies of the effects of exercise in sustaining synaptic inputs onto axotomized motoneurons20,22,30 and their retention as long as 10 weeks later. 34
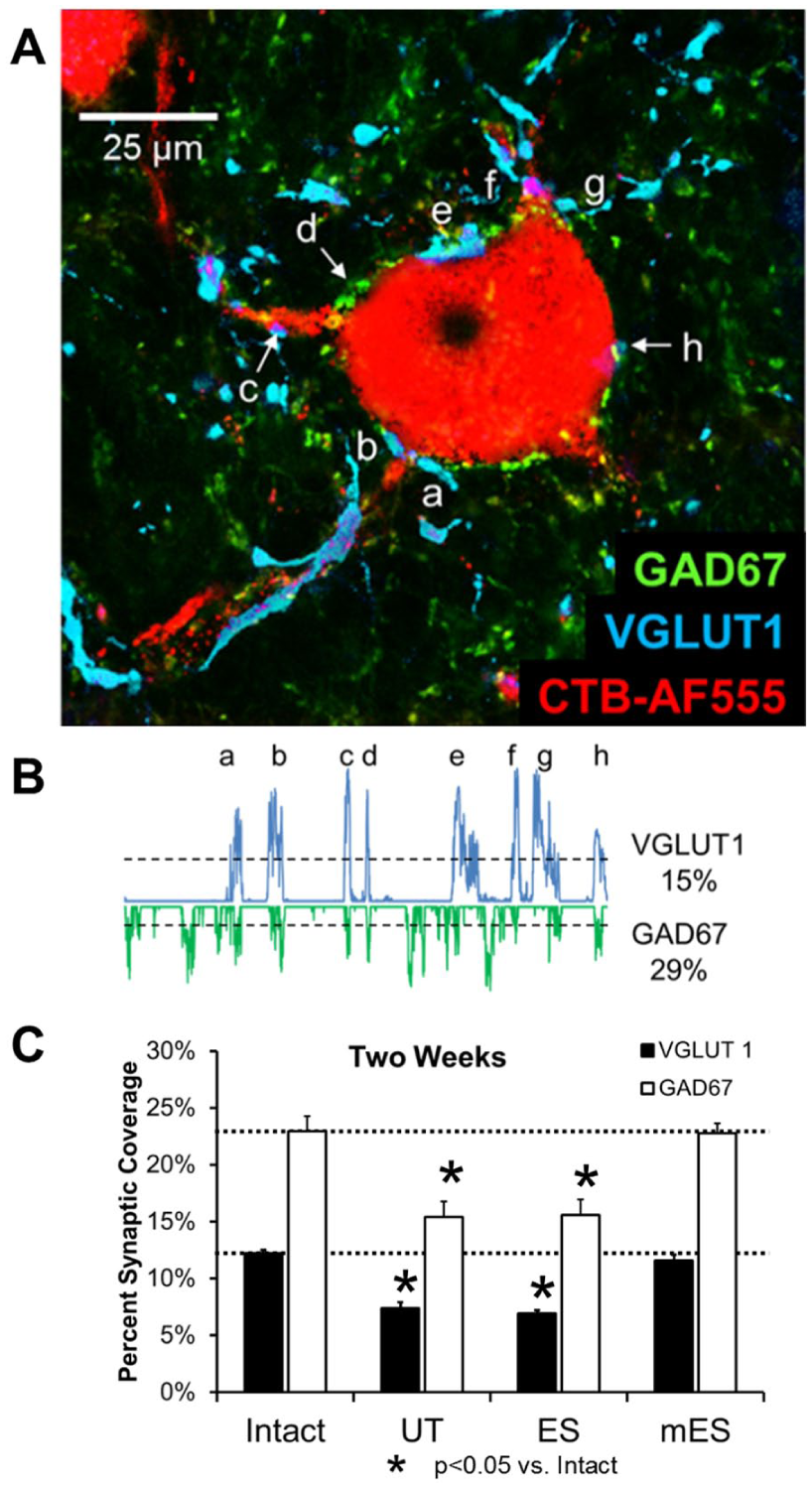
(A and B). Scheme for measurement of synaptic coverage of motoneurons. (A) One motoneuron from an intact mouse that was retrogradely labeled with cholera toxin-B conjugated to Alexafluor 555 (CTB-AF555) is shown. Structures immunoreactive to excitatory (VGLUT1) and inhibitory (GAD67) synapses on this neuron appear as cyan and green. The image is from a single 10-µm thick optical section through the cell. (B) A 5-µm wide region of interest was created around the perimeter of the cell and the intensity of VGLUT1 and GAD67 immunoreactivity was measured beneath it as profile plots. The lowercase letters a-f in the profile plots correspond to the positions marked in panel A. The mean plus 1 standard deviation of the entire plots was used as a threshold (dashed lines) and portions of the plot greater than this threshold were considered contacts between the motoneuron and the respective synapse-associated structures. The percentage of the perimeter of the cell in this image that is in contact with each type of synaptic structure is given. (C) The mean (± SEM) synaptic coverage by structures immunoreactive to VGLUT1 (black bars) and GAD67 (white bars) are shown. Data are from intact mice and from animals two weeks after sciatic nerve transection and repair. Horizontal dashed lines indicate the mean percent synaptic coverage in 6 intact mice. UT = untreated (n = 6); ES = single treatment of 20-Hz electrical stimulation (n = 4) of the proximal segment of the cut nerve at the time of injury; mES = treatments applied to the proximal segment of the injured nerve every third day for 2 weeks (n = 3).
In untreated mice (Figure 5C, UT), there was a marked reduction in coverage by both VGLUT1+ (F3, 12 = 51.86,P < .01) and GAD67+ inputs (F3, 12 = 9.80, P < .01), relative to intact mice, as we20,22,30 and others14-16 have also observed. Treatment of mice with a single application of ES (Figure 5C, ES) had no significant effect on this decrease in coverage. However, in mice treated repeated times with ES (Figure 5C, mES), no significant reduction in synaptic coverage by either type of input, relative to intact mice, was observed.
Discussion
Two different experimental activity-dependent therapies have been developed to address the poor functional recovery found after peripheral nerve injuries. One hour of 20-Hz ES, usually applied once at the time of repair of cut nerves, 7 and 2 weeks of moderate daily exercise in the form of treadmill walking5,8,9 have been shown to enhance axon regeneration. Both treatments enable regenerating axons to cross the injury site and enter a regeneration pathway in the distal nerve segment. The effects of a single application of ES at the time of injury on the elongation of axons once in the regeneration pathway are not as robust as those of exercise. 4 Results of application of ES more than once are mixed. Continuous application of 20-Hz ES resulted in inhibition of the growth of regenerating sensory 12 and motor 6 axons and have formed part of the rationale for limiting ES treatments to 1 hour. Very recently it has been reported that daily application of 1 hour of ES via implanted electrodes during the first week after injury resulted in greater enhancement of motor axon regeneration than a single application. 13 We chose to evaluate the effectiveness of repeated treatments with 1 hour of ES, applied every third day over the same time course as the application of treadmill exercise, for its effect on motor axon regeneration and functional recovery following peripheral nerve injury. An important finding of this study was that our repeated applications of ES did not increase the rate of M response recovery after peripheral nerve transection and repair; it did not enhance motor axon regeneration relative to untreated controls.
At least a part of our rationale for choosing to treat mice every third day for 2 weeks with 1 hour of ES was based on the observation that it produces a transient increase in the expression of BDNF and its trkB receptor in the stimulated neurons. 7 Both ES and exercise rely on increased BDNF-trkB signaling in the regenerating axons for their effectiveness in enhancing axon regeneration.27,35 By stimulating cut nerves every third day, we hoped to extend the duration of the increased expression of these molecules over the same 2-week period used with treadmill exercise. However, the anticipated prolonged elevation of BDNF-trkB expression also might underlie the lack of an effect on motor axon regeneration we observed. Application of recombinant BDNF to cut nerves can enhance regeneration but the amount applied may be critical. At higher concentrations, the efficacy of such treatments is reduced, via a mechanism thought to involve anti-growth signaling by BDNF through the common neurotrophin receptor, p75NTR 36 . It is possible that a more prolonged elevation of neuronal BDNF resulting from repeated application of ES could engage p75NTR and inhibit any trkB-mediated enhancement of regeneration. We believe that this hypothesis could be tested by manipulating p75NTR expression/signaling. Unfortunately, this theory does not explain the recently reported finding 13 that daily treatment with 1 hour of ES during the first week after sciatic nerve transection and repair produced an acceleration of the restoration of the M response in rats that was nearly as great as we have observed after treadmill exercise for 2 weeks. 23 An analysis of BDNF-trkB expression in motoneurons in this protocol will be important.
We also found that repeated, but not single, application of ES resulted in an extravagant ability of stimulated peripheral afferent axons to recruit motoneurons into activity. In all the mice studied, H reflex efficacy was greatly exaggerated during the early stages of muscle reinnervation, as we9,23 and others 28 have reported in rats. In untreated mice and mice treated once with ES, this enlarged reflex response declines over time but in mice treated repeated times with ES no such decline in H reflex efficacy was noted. It is possible that the retention of enlarged monosynaptic reflexes could be due to an effect of the mES on synaptic inputs onto motoneurons. Beginning approximately 1 week after peripheral nerve transection and repair, synaptic inputs are withdrawn from the somata and proximal-most dendrites of the axotomized motoneurons.15,16 Over time, some of these inputs are restored, especially if motor axons regenerate and reinnervate targets, but those expressing VGLUT1 are not, as the central processes of axons of these group Ia afferent neurons withdraw from the ventral horn.14,15 The permanent loss of these connections is an explanation for the permanent loss of the stretch reflex found in reinnervated muscles.18,19
We found that after a single application of ES, a reduction in motoneuron synaptic coverage like that found in untreated mice was encountered. Because a single application of ES did not sustain them for at least 2 weeks after injury, we presume that VGLUT1+ synaptic inputs will continue to be withdrawn and lead to permanent withdrawal of these proprioceptive axons from the ventral horn, as in untreated mice. 15 In our mice exposed to repeated application of ES, coverage of motoneuron somata by VGLUT1+, as well as inhibitory GAD67+ inputs, was similar to that found in intact mice. We believe that the sustaining of VGLUT1+ inputs onto motoneurons found 2 weeks after sciatic nerve transection and repair and repeated applications of ES may be related to BDNF expression. Two weeks after peripheral nerve injury followed by moderate daily treadmill exercise, the coverage on the axotomized motoneurons by VGLUT1+ terminals is not reduced, relative to intact animals20,22,30 and this sustaining of inputs is maintained for at least 10 weeks after the end of the exercise period. 34 This effect of exercise is dependent on BDNF expression in the postsynaptic motoneurons. 22 Thus, we speculate that the repeated applications of ES following peripheral nerve transection and repair employed here results in a prolonged expression of BDNF in the injured motoneurons and a sustaining of synaptic inputs onto them, especially those immunoreactive for VGLUT1, which is not found after a single treatment with ES. Whether the sustained inputs in the mES mice are due to a lack of withdrawal or because the repeated treatments with ES promoted the reformation of withdrawn inputs (or both) is not known. Similarly, it is not known whether the sustained VGLUT1+ inputs are retained, as they are after exercise, or whether they were withdrawn once the treatments with ES were stopped.
Withdrawal of synaptic inputs from axotomized motoneurons has been known for some time, but its functional significance has remained uncertain. The coincident lack of enhancement of motor axon regeneration reported here with mES and the sustaining of VGLUT1+ synaptic inputs onto the axotomized motoneurons might be interpreted to suggest that withdrawal of these monosynaptic inputs is necessary for the effectiveness of ES in promoting motor axon regeneration. However, this seems unlikely, as daily treadmill exercise both enhances motor axon regeneration21,23 and sustaining of these synaptic inputs.20,22 It would be interesting to know whether the daily application of ES during the first week after nerve injury that promotes robust enhancement of motor axon regeneration also results in sustained synaptic inputs onto the injured motoneurons.
The nature of the sustained inputs in the mES mice and in mice treated with daily treadmill exercise is not known. It is possible that the synapses in these mice are either not withdrawn or are withdrawn but then simply reestablish connections in the same manner as prior to injury. On one level, such a sustaining of VGLUT1+ inputs onto motoneurons might be considered advantageous to functional recovery, since it could enable greater proprioceptive feedback onto the motoneurons than is found otherwise. However, the extent that the repeated treatments with ES shown here also result in the retention of segmental reflexes that are abnormally large would seem to outweigh this advantage.
It also is possible that the origins of the sustained inputs are different from those present prior to injury. Following sciatic nerve crush injury, VGLUT1+ synaptic density onto motoneurons is restored, but monosynaptic EPSPs in those motoneurons are significantly larger than found in intact animals. 37 One explanation for this finding is that the small GABA-ergic terminals (P-boutons) on VGLUT1+ synapses, ones that likely mediate presynaptic inhibition, are not restored after crush injury. 38 If synaptic rearrangement is also present in the reestablishment of other inputs to motoneurons, then the process of synaptic withdrawal following peripheral nerve injury might be cast in an entirely different light. Post-injury withdrawal of synaptic inputs might represent an opportunity for a level of restoration of spinal circuitry that is congruent with the extent and specificity of reinnervation of muscle targets. If these speculative hypotheses were proved true, then the effects of potential therapies for treatments of peripheral nerve injuries, such as ES, on the restoration of spinal circuitry need to be considered as much as their effect on axon regeneration.
Footnotes
Acknowledgements
Many thanks to Amanda Mulligan and Olivia Mistretta for their superb technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants NS057190 and HD032571 from the USPHS.
