Abstract
Introduction. Fatigue is a common and debilitating symptom in patients with multiple sclerosis (MS). Self-reported levels of perceived fatigue are associated with both patient characteristics and clinical measures. Pilot analysis indicated that muscle fatigability combined with depression scores was highly associated with perceived fatigue in patients with MS. Studies that combine physiological and psychological constructs to explain MS-related fatigue are scarce. Therefore, the present study aimed to evaluate the robustness of the association between perceived fatigue, muscle fatigability, and depression scores in MS. Methods. Eighty-six patients with relapsing-remitting MS completed 2 fatigue questionnaires (Fatigue Severity Scale [FSS] and Modified Fatigue Impact Scale [MFIS]) and a depression questionnaire (Hospital Anxiety and Depression Scale [HADS]). Maximal index finger abduction force (maximum voluntary contraction [MVC]) was measured, as well as muscle fatigability during a 2-minutes sustained maximal contraction. Multivariable regression analyses were used to analyze the association between perceived fatigue, and muscle fatigability and depression scores. Results. Perceived fatigue was associated with depression, muscle fatigability, and, depending on the questionnaire, to sex or to MVC. The model explained 40% and 48% of the variation in perception of fatigue as indexed with FSS questionnaire (rpartial: HADS 0.45, muscle fatigability 0.45, MVC −0.14, sex 0.32), and MFIS physical questionnaire (rpartial: HADS 0.59, muscle fatigability 0.49, MVC −0.38), respectively. Conclusions. The found association accentuates the importance of including both physiological fatigability-related and psychological mood–related constructs in models to explain perceived fatigue in patients with MS. The model also directs future research toward applying effortful conditions and emphasizes the importance of assessing different constructs when evaluating rehabilitation strategies to reduce MS-related fatigue.
Keywords
Introduction
A large percentage of patients with MS report increased levels of perceived fatigue, having a strong impact on the quality of life.1,2 MS-related fatigue is described by the Multiple Sclerosis Council as “a subjective lack of physical and/or mental energy that is perceived by the individual or caregiver to interfere with usual and desired activities.” 3 Factors underlying perception of fatigue are multidimensional and not yet fully understood. Associations with clinical features have been examined, but only depression showed a consistent association with perceived fatigue.4-6
Several studies have investigated the association between perceived fatigue and muscle fatigability. Most studies did not find an association (see supplementary material). Conversely, we found a clear association between perceived (self-reported) fatigue and muscle fatigability but only after correction for maximal force. Furthermore, pilot analyses showed that the combination of depression scores and muscle fatigability predicted perceived fatigue even better. 7 Recent literature8,9 has underlined the importance of combining physiological and psychological constructs to explain perception of fatigue. Studies that combine both dimensions in a single model are, however, scarce. Since our sample size was relatively small (n = 20) and, because of the intensive protocol, included highly motivated patients, 7 we felt that we had to test the robustness of the association between perceived fatigue, muscle fatigability, and depression scores (1) in a larger and less well-controlled group of patients with MS and (2) by including an additional fatigue questionnaire that distinguishes between perception of cognitive and physical fatigue. We also sought to investigate effects of medication, and contribution of other patient characteristics, such as sex, age, and disease duration. We hypothesized that models incorporating muscle fatigability result in significantly better predictions of perceived fatigue than a model that only includes depression scores.
Parts of the data were previously presented as an abstract. 10
Methods
Subjects
Of 100 patients with MS, 86 relapsing-remitting (RRMS) patients were recruited (patients were excluded for the following: non-RRMS, other neurological disorders, other conditions affecting fatigue, or not being able to walk at least 100 m). None suffered from a relapse at the time of the measurements or had taken steroids in the preceding 6 weeks.
All participants reported to be right handed. The study was approved by the University Medical Center Groningen Medical Ethical Committee and all patients signed an informed consent before participation.
Experimental Setup
Before the experiment, patients completed 3 questionnaires: the Hospital Anxiety and Depression Scale (HADS), 11 the Fatigue Severity Scale (FSS), 12 and the Modified Fatigue Impact Scale (MFIS). 3 The FSS consists of 9 questions with scores ranging from 1 to 7. The MFIS consists of 21 questions and distinguishes between effects of fatigue on physical (range 0-36), cognitive (range 0-40), and social functioning. The HADS consists of 14 questions, of which 7 questions determine the depression subscale (range 0-21). Higher scores indicate stronger feelings of fatigue or depression.
During force measurements, patients sat in a comfortable chair with arm support and held a force transducer in their right hand (Figure 1A). 13 The thumb and fingers 3 to 5 were taped to the transducer to prevent changes in hand position during the test. The proximal interphalangeal joint of the index finger was taped to a wedge connected to the transducer. Force was exerted by abducting the index finger. Force data were amplified and sampled at a rate of 200 Hz (analog-to-digital conversion: 1401Plus, CED, Cambridge, UK; accompanying software Spike 2, version 5.03).
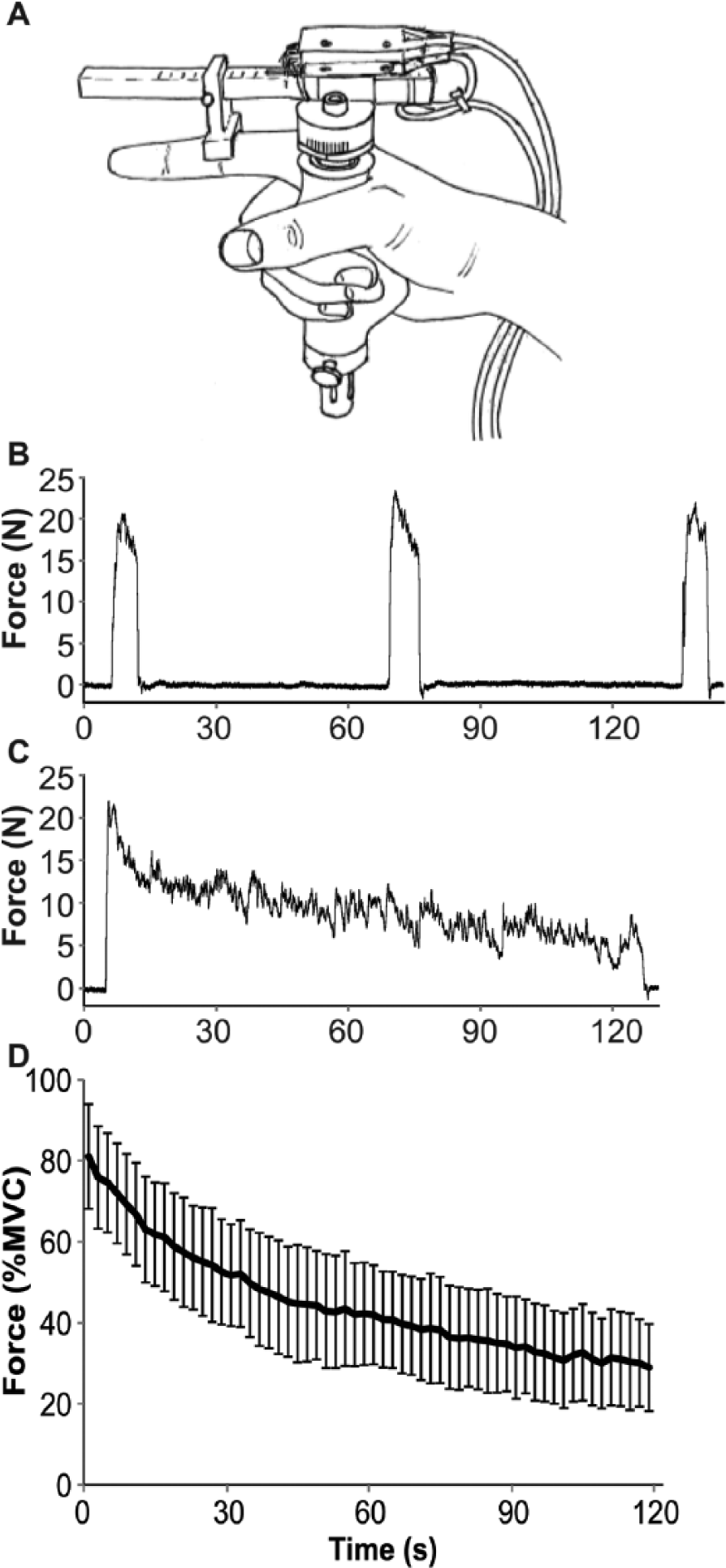
Force transducer and force data. (A) Force transducer. (B) Maximal voluntary contractions (MVCs) of a single patient. (C) Force decline during a 2-minute sustained maximal contraction of a single patient. (D) Mean force decline of all patients (as percentage of MVC) during the fatiguing 2-minute task. Data are averaged over 2-second periods for graphic purposes. Error bars represent standard deviations.
Tasks
Three 5-second maximal voluntary contractions (MVCs) were performed, with 1 minute in between attempts. If force increased across the 3 attempts, an extra MVC was performed until the force reached a stable level. Subsequently, patients performed a fatiguing, sustained maximal index finger abduction for 2 minutes (Figure 1B and C). During the tasks, participants received online feedback of their force and were encouraged by the investigator.
Outcome Measures
Perceived fatigue was quantified by the FSS and MFIS physical subscale; the first reflecting the impact of fatigue on daily life, the latter reflecting the impact on physical functioning.3,12,14 Maximal force obtained during the MVCs (n = 57) or during the fatiguing task (n = 26) was considered the MVC. Muscle fatigability was defined as the decline in force during the 2-minute sustained contraction expressed as a percentage of MVC, that is, 1 − (force last 6 seconds/MVC) × 100%.
Statistical Analyses
All analyses were performed with SPSS version 20. The FSS, HADS depression, and disease duration were not normally distributed according to the Kolmogorov-Smirnov analysis. To normalize the data, FSS scores were cubed (FSS 3 ), and disease duration data were log-transformed. We did not succeed in normalizing the HADS depression scores.
Correlations were investigated using Pearson’s correlation coefficients or Spearman’s ρ (HADS data). Differences in questionnaire scores between groups were tested using independent t tests.
We performed multivariable regression analyses for both the FSS and MFIS questionnaires. The residuals of the models were normally distributed after including original FSS data and no collinearity was found between any of the parameters. Therefore, all analyses were performed with the original data (without transformation).
Several patients used medication, and since medication may alter (perception of) fatigue, this was further investigated by analyzing a subgroup of patients without medication.
Results
Patient Characteristics
Three patients did not perform the sustained contraction correctly, and were excluded from analysis (2 patients repeatedly stopped during the contraction, 1 patient strongly increased force after 1 minute). In total, 83 patients were included in the analyses (Table 1). Three patients scored 12 or more on the HADS depression questionnaire, indicating possible presence of depressive symptoms. However, inclusion of these subjects did not affect the direction of our analysis. Fifty-six patients (67%) were employed or weekly volunteers. Six patients were ambidextrous rather than right-handed according to the Edinburgh Handedness Questionnaire (scores −23 to +41). 15
Patient Characteristics and Medication Information. a
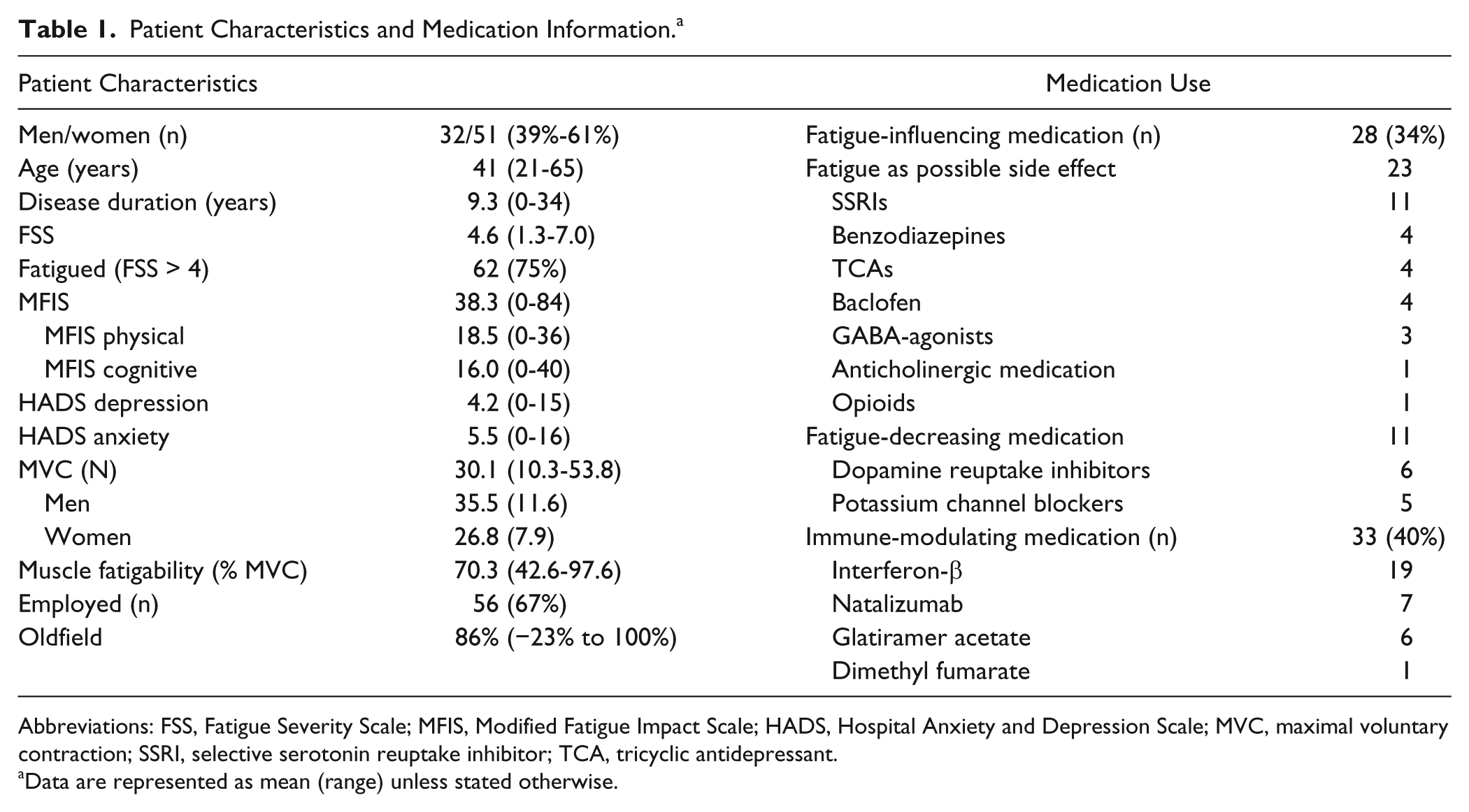
Abbreviations: FSS, Fatigue Severity Scale; MFIS, Modified Fatigue Impact Scale; HADS, Hospital Anxiety and Depression Scale; MVC, maximal voluntary contraction; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant.
Data are represented as mean (range) unless stated otherwise.
Medication
Thirty-three patients used immune-modulating medication, and 28 used medication that affects (perception of) fatigue. Nine patients used both types of medication (Table 1). Some medication is known to have perceived fatigue as a side effect (n = 22), but patients also used potassium channel blockers and dopamine reuptake inhibitors, that have been prescribed to reduce feelings of fatigue (n = 11). In general, women used more fatigue-modulating medication than men did (43% vs 19%, P = .016).
Perceived Fatigue
Patients reported an average FSS score of 4.6 (SD = 1.4). In total, 62 patients (75%) scored higher than 4.4,16 Mean scores were higher for females (4.9 vs 4.1, P = .016), patients using fatigue-modulating medication (ie, all medication listed in Table 1; 5.2 vs 4.3, P = .003), and unemployed patients (5.4 vs 4.3, P < .001). The FSS score was not associated with disease duration (P = .34).
The MFIS physical subscale ranged from 0 to 36. Higher scores were obtained by patients using fatigue-modulating medication (21.9 vs 16.7, P = .005) and unemployed patients (22.8 vs 16.4, P < .001). Neither sex (P = .29) nor disease duration (P = .081) was associated with the MFIS physical subscale.
The MFIS cognitive subscale ranged from 0 to 40 and was only associated with the HADS depression score (ρ = 0.59, P < .001).
The FSS 3 and MFIS physical were strongly associated (r = 0.76, P < .001), as well as the FSS 3 and the MFIS cognitive (r = 0.64, P < .001), and the MFIS physical and MFIS cognitive (r = 0.68, P < .001).
Patients with and without immune-modulating medication did not differ in their scores on fatigue questionnaires.
Force Tasks
Figure 1B and C shows the force recording for a representative patient. The maximal index finger abduction force equaled 30.1 ± 10.3 N. Men (35.5 ± 11.6 N) were significantly stronger than women (26.8 ± 7.9 N, P = .001). With increasing age, patients became weaker (r = −0.37, P = .001). This effect was still significant after correction for sex (rpartial = −0.32, P = .003). The fatigue-modulating medication did not affect force (P = .444).
The mean force (averaged over 6 seconds) during the 2-minute sustained contraction declined from 78.0% MVC (SD = 9.6) to 29.7% MVC (SD = 10.2; Figure 1D), which resulted in a fatigability of 70.3% (range 42.6-97.6).
Larger MVC (r = 0.28, P = .009) and lower age (r = −0.35, P = .001) resulted in more muscle fatigue. However, when age and MVC were simultaneously included in a regression model, only age significantly predicted fatigability (age rpartial = −0.28, P = .011, MVC rpartial = −0.18; P = .114). Muscle fatigability was associated with perceived fatigue, measured with either the FSS 3 (r = 0.29, P = .009) or MFIS physical (r = 0.31, P = .005). Muscle fatigability was neither associated with sex (r = −0.62, P = .578) nor with disease duration (r = −0.15, P = .170).
Hospital Anxiety and Depression Scale
Patients scored an average of 4.2 (range 0-15) on the HADS depression questionnaire. FSS 3 was associated with HADS depression score (ρ = 0.34, P = .002), as were the subscales MFIS physical and MFIS cognitive (ρ = 0.52, P < .001 and ρ = 0.59, P < .001, respectively). No association was found between HADS and muscle fatigability (r = −0.01, P = .931).
Associations Between Perceived Fatigue, Muscle Fatigability, and Depression
The final step was to investigate associations between perceived fatigue and muscle fatigability, taking the aforementioned factors into account. The regression analysis included HADS depression scores on the first level, and muscle fatigability and MVC (see Steens et al 7 ) on the second level. Since sex and fatigue-modulating medication were associated with perceived fatigue, we added them stepwise on the third level.
One subject was excluded from the regression analyses because his data deviated more than 3 standard deviations from the model.
Fatigue Severity Scale scores were best explained by a model consisting of HADS depression scores, muscle fatigability and MVC, and sex (R2 = 0.40, P = .005; Table 2). For patients without medication (n = 55), the model including HADS depression scores, muscle fatigability, and MVC explained FFS scores best (R2 = 0.33 P = .013). HADS depression scores, together with muscle fatigability and MVC explained the MFIS physical scores best (R2 = 0.48, P < .001, Table 2), both in the whole group as well as in patients without medication (n = 55; R2 = 0.43, P = .003).
Associations With Fatigue Questionnaires. a
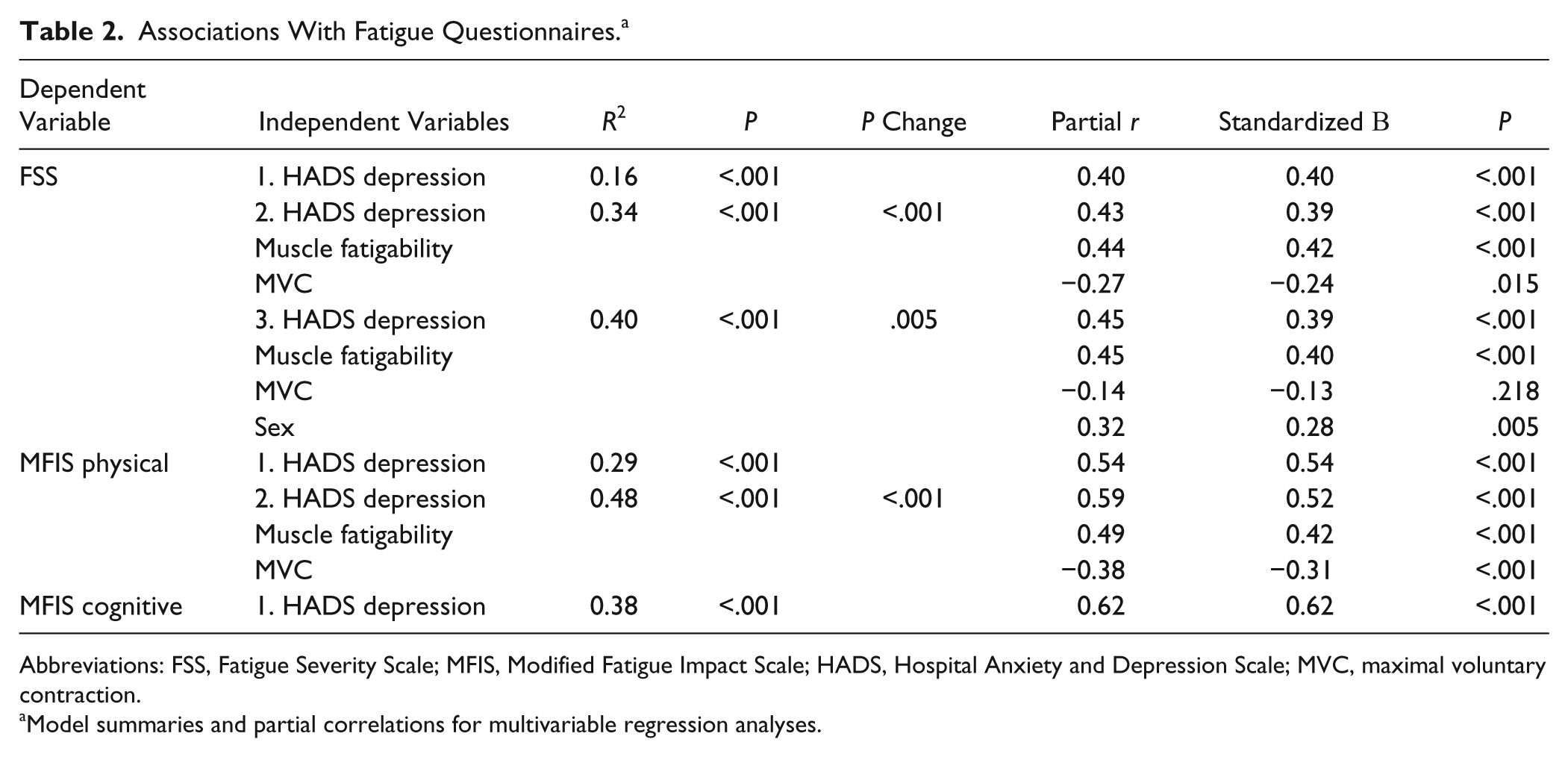
Abbreviations: FSS, Fatigue Severity Scale; MFIS, Modified Fatigue Impact Scale; HADS, Hospital Anxiety and Depression Scale; MVC, maximal voluntary contraction.
Model summaries and partial correlations for multivariable regression analyses.
Modified Fatigue Impact Scale cognitive was best explained by HADS depression scores (R2 = 0.38, P < .001; Table 2). Also in patients without fatigue-modulating medication, HADS depression was the only factor to predict the MFIS cognitive subscale (R2 = 0.47, F = 46.05, P < .001).
Discussion
The present data confirmed the association between fatigue as perceived by patients with MS, and the combination of muscle fatigability and depression in a large group of patients. The combination of muscle fatigability and HADS depression scores explained the variation in FSS scores better than a model consisting of either one of the parameters. We extended our previous finding by showing that this was true for different questionnaires of perceived fatigue. We further found that fatigue-modulating medication did not change these results significantly; nor did patient characteristics such as disease duration and age.
Perceived Fatigue Is Associated With Muscle Fatigability and Depression
In accordance with previous studies, we found an association of perceived fatigue and depression scores1,4-6 Most studies, however, showed no association between muscle fatigability and perceived fatigue (see supplementary material). Although we are not certain why studies failed to find associations between perceived fatigue and muscle fatigability, several possible factors are presented in Table 1 of the supplementary material.
Additionally, it is known that fatigability is affected positively and negatively by several parameters (such as MVC and age); these parameters also interact with each other and will therefore interfere with the association between perceived fatigue and fatigability. One study 5 with a large range of perceived fatigue scores and a large sample size also failed to find an association. It is possible that the inclusion of MVC values or sex would have resulted in a significant association (see also Steens et al 7 ).
During a sustained contraction fatigue-related changes in the muscle fibers 17 and the motoneurons 18 require increased cortical drive19-21 to maintain force production. In patients with MS, this increase in activation is reduced, resulting in a decline in voluntary drive.7,20,22,23 The association between muscle fatigability and perceived fatigue found presently could therefore point to difficulties in maintaining a strong voluntary drive. This observation is particularly interesting in relation with the increased activation of the anterior cingulate cortex in patients with higher levels of perceived fatigue.6,24 This area is thought to be involved in effort-related cost-benefit analysis. 25
Muscle Fatigability and Maximal Force
Muscle fatigability is task and muscle dependent. 26 The association with perceived fatigue may therefore change depending on the muscle (group) that is fatigued or the performed task. However, a previous study using repeated index finger abductions of 40 seconds at 30% MVC followed by 5-second MVC and 5-second rest, demonstrated similar findings. 27 However, when leg muscles are targeted, it is possible that deconditioning will confound muscle fatigability.
In control subjects, stronger subjects generally show a larger force decline. 28 In patients with MS, a positive relationship between maximal force and muscle fatigability was found in some studies (present data7,29) but not in others. 30 This relation will also affect the association between perceived fatigue, and fatigability and depression scores. It is therefore, important to include MVC values in models using fatigability to predict perceived fatigue.
Effects of Medication on Fatigue
The immune-modulating medication (ie, interferons, copaxone) did not affect perceived fatigue in the patients with MS, even though studies specify perception of fatigue as a possible side effect. 31 Patients using fatigue-modulating medications—mostly medication to combat symptoms of MS such as spasticity or pain—scored higher on the FSS questionnaire. However, our model for perceived fatigue was not affected by inclusion of medication use.
Fatigue Questionnaires
Both FSS and MFIS physical did associate with fatigability and depression. FSS was, however, also associated with sex whereas MFIS physical was associated with MVC. This result most likely reflects the interaction between fatigability, MVC and sex. The multidimensional nature of fatigue, and the focus of the questionnaires on different aspects of perceived fatigue probably also contribute to variation in the amount of perceived fatigue that is explained by fatigability and depression scores.4,14 The observation that muscle fatigability is associated with MFIS physical, but not with MFIS cognitive, supports the notion that muscle fatigability is associated with the perception of physical fatigue. This is in accordance with a previous study involving a cognitive-motor dual task, where we found an association of the MFIS cognitive with a decrease in cognitive performance, and of FSS with fatigability measures. 16
Conclusions and Future Recommendations
The main finding of the present study is the observation that a combination of physiological (muscle fatigability) and psychological (HADS depression scores) measures were better in explaining variation in MS-related fatigue than each single parameter. The fact that both depression and fatigability are associated with perceived fatigue, and complement each other, confirms the multidimensional nature of fatigue and suggests that a combination of constructs is necessary to explain MS-related fatigue. We further hypothesize that the association between muscle fatigability and perceived fatigue might reflect activity changes in areas involved in cost-benefit trade-offs, suggesting a role for more effortful tasks in future research. Although this study has a cross-sectional nature and no causal relation can be established from our data, it supports the need for well-controlled exercise training programs as possible treatment for MS-related fatigue. 32 Furthermore, it is likely that a combination of treatments would be more successful to combat feelings of fatigue in patients with MS.
Footnotes
Acknowledgements
We would like to thank Tjalling Nijboer for his help with the experimental setup, Cobi Bolwijn for her help with patient inclusion, and Astrid de Vries for her assistance with the measurements.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
