Abstract
Studies indicate that motor functions in patients with traumatic brain injury (TBI) can be improved with action observation. It has been hypothesized that this clinical practice relies on modulation of motor cortical excitability elicited by passive action observation in patients with TBI, a phenomenon shown thus far only in normal controls. The purpose of this work was to test this hypothesis and characterize the modulation of motor cortex excitability during passive action observation in patients with subacute moderate to severe TBI. We measured motor evoked potentials induced by single-pulse transcranial magnetic stimulation to the left primary motor cortex and recorded from the contralateral first dorsal interosseus while 20 participants observed videos of static and moving right index finger. Results were compared with those of 20 age-and gender-matched healthy controls. As expected, greater excitability was elicited during moving than static stimuli in healthy subjects. However, this was not observed in patients with TBI. Modulation of motor excitability during action observation is impaired in patients with TBI depending on motor dysfunction, lesion site, and number of days postinjury. These preliminary results suggest a strategy to identify patients in whom action observation might be a valuable neurorehabilitative strategy.
Keywords
Introduction
Cortical excitability is increased not only when performing an action but also by mere observation of a motor action.1-5 The activation of the internal representation of movement created by passive action observation may be beneficial for patients with brain damage and limited motor functions.6-10 Some studies have reported beneficial effects of action observation in patients with stroke, especially when used as an adjunct to physical therapy. 11 For instance, Celnik and colleagues found enhanced motor memory formation when motor training was combined with action observation in 8 patients with chronic stroke. 12 Franceschini and colleagues reported improved upper limb function in 28 chronic stroke patients who underwent a 4-week treatment course of action observation combined with imitation of hand action with the affected limbs. 13 These results support the use of action observation to augment physical therapy to improve motor functions in stroke. Similar arguments have been made regarding the clinical relevance of passive action observation in patients with spinal cord injury and traumatic brain injury (TBI). Action observation may promote relearning of lost motor capacities by activating the action brain network. This implies that modulation of motor cortical outputs is possible during action observation in patients after TBI. Here, we tested this notion. However, we hypothesized that patients with TBI affecting motor cortical connectivity may show smaller modulation of motor excitability during action observation than healthy controls. If so, evaluation of modulation of motor cortical outputs might predict which patients with TBI may benefit from action observation and which would not or less so.
An important hypothesis is that modulation of motor responses during action observation reflects mirror-neuron mediated mechanisms of interpersonal motor resonance. 14 The brain network of action observation has been extensively studied in healthy subjects, especially with single-pulse transcranial magnetic stimulation (TMS)-induced motor evoked potentials (MEPs) applied over the primary motor cortex (M1). For instance, observation of a moving index finger modulates excitability of the corticospinal pathway, which induces MEPs that are greater than those elicited during observation of a static index finger. Moreover, this modulation of MEPs is congruent and muscle-specific. Observation of a moving index finger induces greater MEP size in the first dorsal interosseus (FDI) than from the abductor digiti minimi (ADM) or abductor pollicis brevis (APB).1,2
In this preliminary study, we assessed modulation of motor responses during action observation in patients with TBI and healthy controls. We applied single-pulse TMS over the left M1 to assess motor excitability during presentation of static and moving right index finger videos and recorded TMS-induced MEPs from the right finger index (FDI) using electromyography (EMG). We further assessed 4 factors that may affect TMS-induced MEP size during action observation in TBI patients: motor capacities, presence or absence of a lesion in the frontal and parietal cortex, number of days since the injury, and severity of the head injury (described below).
Materials and Methods
Participants
Right-handed individuals with subacute TBI (20 adults, average age = 30.3 years old, range = 17-49 years) and healthy adults (20 adults, average age = 30.5 years old, range = 20-50 years) took part of the study. They were all recruited at the Institut Universitari de Neurorehabilitació Guttmann-UAB. Inclusion criteria for patients with TBI included having received a diagnosis of TBI based on clinical history and supported by neurological and neuroimaging findings. Exclusion criteria included pregnancy or medications that could modify seizure threshold. 15 Individuals with TBI were all inpatients at the Institut Universitari de Neurorehabilitació Guttmann-UAB. They were tested within 147 days postinjury (average = 94.7 days, range = 46-147). Severity of the TBI was assessed at admission using the Galveston Orientation and Amnesia Test and the Glasgow Coma Scale (GCS). The TBI group was composed of moderate (N = 6; GCS 9-12) or severe TBI (N = 14; GCS ≤8). Average score of the motor subscale of the Functional Independence Measure (FIM)/Functional Assessment Measure (FAM) was 45 (range = 16-107). Thirteen patients displayed paresis (left hemiparesis, n = 3; right hemiparesis, n = 2; left tetraparesis, n = 6; right tetraparesis, n = 1; symmetric tetraparesis, n = 1). Eleven patients had a history of substance use disorders and one had a history of depression prior to the TBI. Inclusion criteria for the healthy group included no history of any neurological or psychiatric disorders and eligible to undergo TMS following international guidelines. 15 They were age-and gender-matched with the TBI group (Table 1). Participants or their legal guardian gave written informed consent prior to entering the study, which was approved by the local ethics committee.
Characteristics of the Subjects.
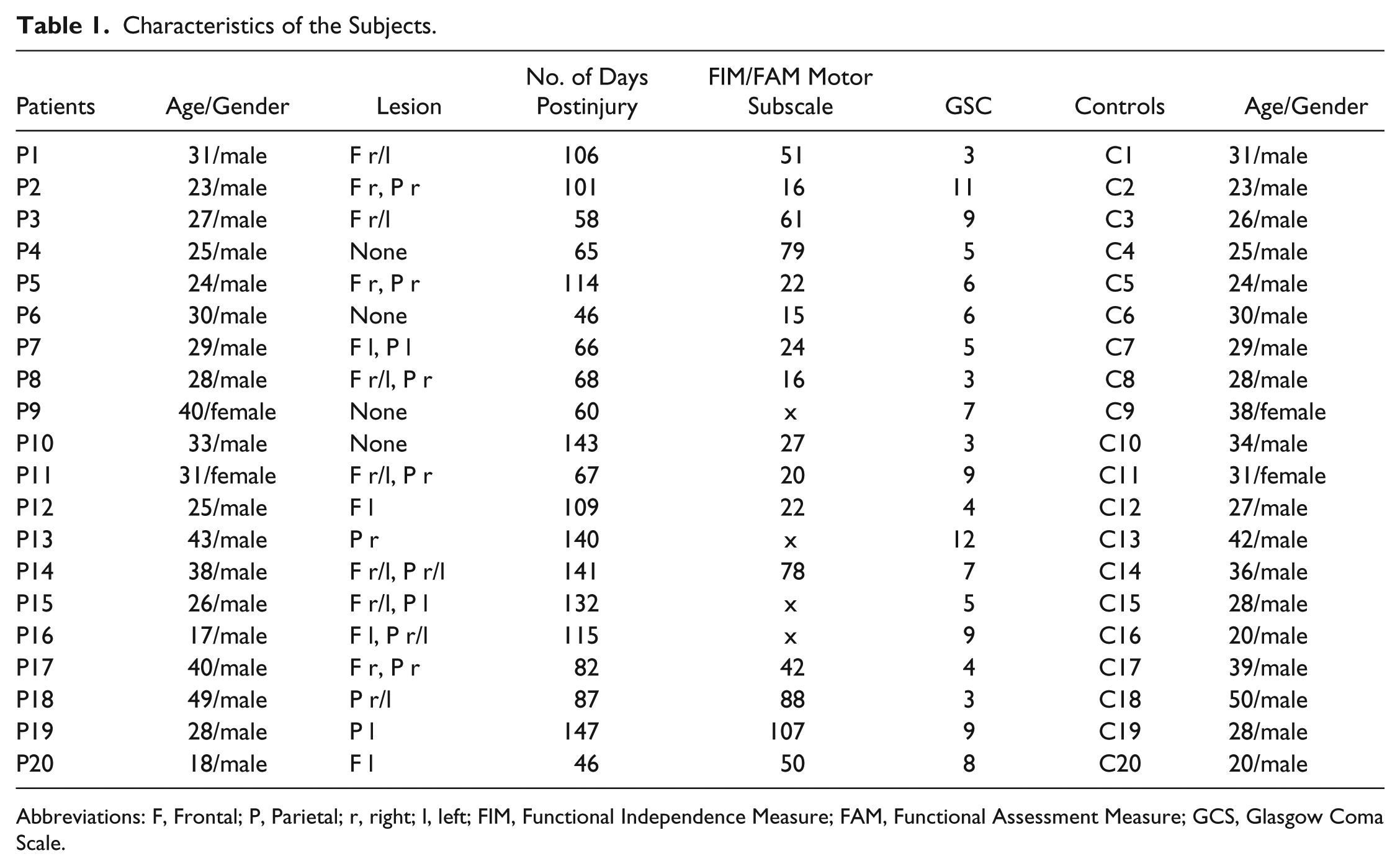
Abbreviations: F, Frontal; P, Parietal; r, right; l, left; FIM, Functional Independence Measure; FAM, Functional Assessment Measure; GCS, Glasgow Coma Scale.
Experimental Procedure
The experiment was performed in a quiet testing room at the research facilities of the Institut Universitari de Neurorehabilitació Guttmann-UAB. TMS-induced MEPs were recorded from the FDI and the ADM muscles while participants passively viewed movie clips presented in a random order on a computer screen of movement of the right index toward a dot (movement condition) and still hand (static condition; Figure 1). These stimuli were similar to those used in a previous study. 16 Duration of each video was 8 seconds. For the movement condition, the index moved 8 times at a pace of approximately 1 Hz. The entire session was composed of 48 stimuli (24 for the movement condition; 24 for the static condition). A single suprathreshold pulse was delivered over the left M1 during each movie clip presentation at 120% of resting motor threshold, which was measured in each subject following recommendations endorsed by the International Federation of Clinical Neurophysiology, 17 with its standard definition of reaching a 50 µV peak-to-peak amplitude FDI-MEP in at least 5 times out of 10 with the lowest stimulator intensity. Patients were wearing a swimming cap to mark the hot spot. Subjects were sitting at a distance of 1 m from the computer screen. TMS was performed with a commercially available Magpro figure-of-eight coil and a Magpro X100 magnetic stimulator (Dantec-Medtronic, Fridley, MN). The average motor threshold stimulation intensity was 52 ± 4% for the healthy group and 65 ± 4% for the TBI group. Relaxation of both muscles was documented by EMG recording for at least 20 ms before each TMS pulse. MEPs were recorded using pairs of Ag/AgCl surface electrodes placed over both the FDI and ADM of the right hand in a belly-tendon montage with a circular ground electrode on the wrist. The EMG signal was recorded using a Medelec System (Oxford Instruments Medical Inc, Oxford, UK).
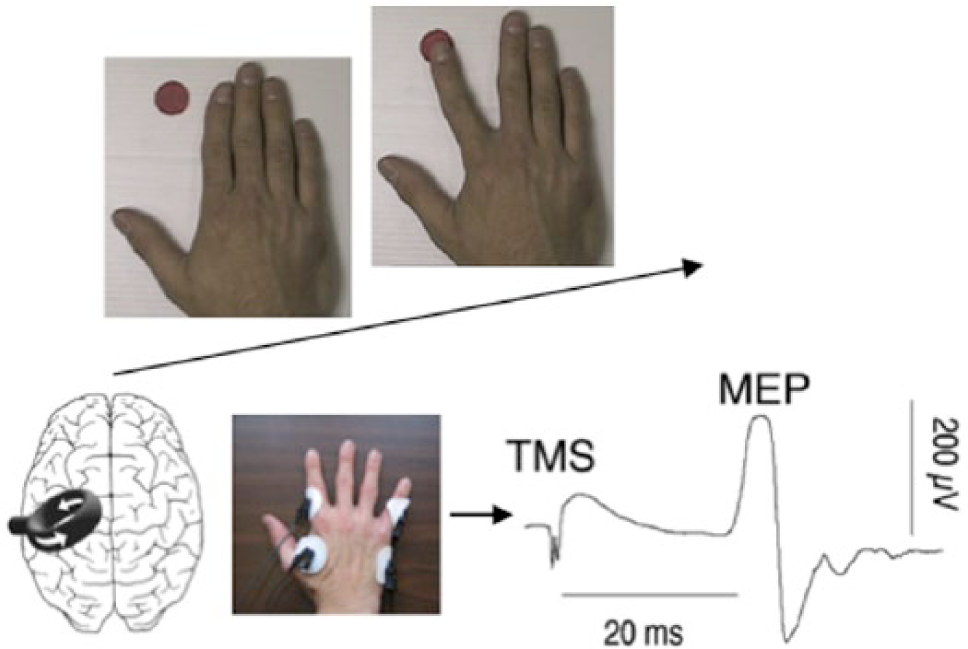
Study design.
In order to further characterize the modulation of motor excitability in TBI patients, we tested whether the modulation of TMS-induced MEP size during action observation was related to any one of the following 4 patients’ characteristics.
Motor Capacities
Most studies assessing motor excitability of action observation have been performed in healthy subjects, and some reported that excitability of action observation can be differently modulated by motor expertise or experience.18,19 However, it is not known whether motor capacities per se have an impact on excitability during passive observation of action. We thus explored whether motor capacities have an impact on modulation of excitability during action observation, as indexed by the modulation of TMS-induced MEP size. We used the FIM + FAM motor subscale administered at admission as a measure of motor function with demonstrated good interrater reliability and validity.20-22 We choose this subscale, although it is not the most precise, because it was part of patients’ clinical assessment. As a preliminary study, we aimed here at using routine clinical assessments to explore whether some of them might be relevant for eliciting modulation of motor excitability during passive action observation. We predicted that weaker motor capacities would be associated with smaller modulation of MEP size compared with better motor capacities.
Frontal and Parietal Lobe Status
Studies in healthy subjects have repeatedly reported the involvement of the frontal and parietal cortex in action observation, although there is no clear consensus with regard to the hemispheric contribution and no studies establishing a causal role of these brain regions in modulation of motor excitability. Some studies have found bilateral activations during action observation,2,23,24 while others argue for a left dominance.25,26 We thus explored the impact of the status of the patients’ left and right frontal and parietal cortices on the modulation of motor excitability during action observation. Information was based on the medical records from patients’ brain computed tomography (CT) or magnetic resonance imaging (MRI) scans. We postulated that patients with lesions in these regions would display weaker modulation of MEP size than patients with no lesion in these regions.
Number of Days Postinjury
Previous work has indicated that brain network reorganization is especially important during the subacute phase after the injury. 27 We thus tested whether the number of days between patients’ injury and participation in this study had an impact on the findings. We assumed that lesser number of days would be associated with weaker modulation of MEP size.
Severity of the Head Injury
Severity of the head injury may affect cortical excitability at rest as measured by TMS. 28 We thus investigated whether severity of the head injury had an impact on the modulation of motor excitability during action observation. Severity of the TBI was indexed using the GCS, one of the most common methods used to characterize severity of head injuries. 29 We proposed that more severe head injury would be associated with weaker modulation of motor excitability.
Data Analysis
Peak-to-peak amplitude of the MEPs was measured using Matlab software (The MathWorks, Natick, MA). For each participant, MEPs were averaged per stimulus condition (static, movement). First, we performed paired t test to compare TMS-induced MEP amplitude during action and static stimuli for each groups. Then, given the expectation that MEP size and its modulation would show substantial intervariability in the TBI group according to aforementioned patients’ characteristics, we calculated for each patient a percent change in MEP amplitude between the moving and static finger conditions, as described previously.16,30 To test whether motor capacities, number of days since the injury, and severity of the head injury influenced TMS-induced amplitude of action observation, we performed Pearson correlations for each of these factors. Finally, to test whether the presence or absence of a lesion in the frontal and parietal cortex had an impact on the TMS-induced amplitude of action observation, we performed an ANOVA. Statistical analyses were performed using SPSS software (version 22.0, SPSS Inc, Chicago, IL). Results with a P value ≤.05 were considered significant.
Results
None of the participants experienced or reported significant adverse effects during or after TMS even when explicitly asked.
TMS-Induced MEP During Action Observation
We first analyzed FDI-MEPs when healthy subjects passively observed videos of static and moving index to confirm that stimuli elicit differential neurophysiologic responses. There was an effect of stimulus conditions (static, action; paired t test; t = −5.62; P = .0001; Figure 2). There was no significant difference between static and movement conditions for the ADM-MEPs (paired t test; t = −1.87; P = .078).
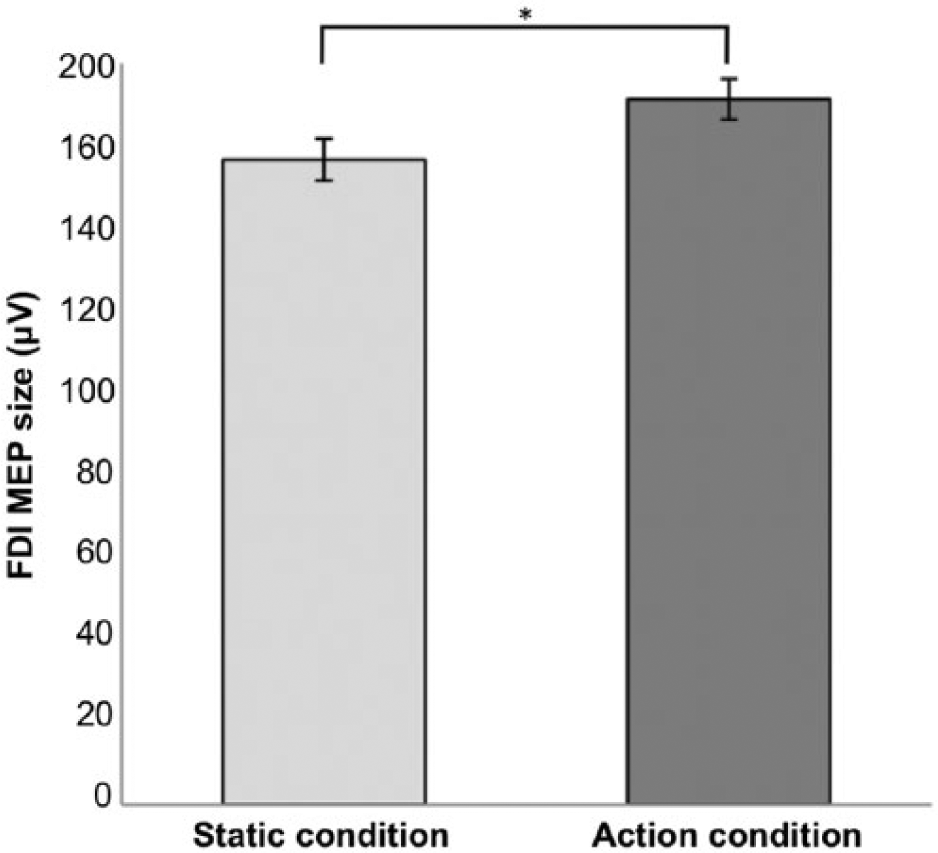
FDI-MEP size of the static and action condition healthy subjects. Error bars indicate SEM.
We then investigated FDI-MEPs in participants with TBI and found no significant difference between stimulus conditions (static, action; paired t test; t = −1.57; P = .132; Figure 3). Furthermore, there was no significant difference between stimulus conditions for the ADM-MEPs (paired t test; t = 1.79; P = .089).
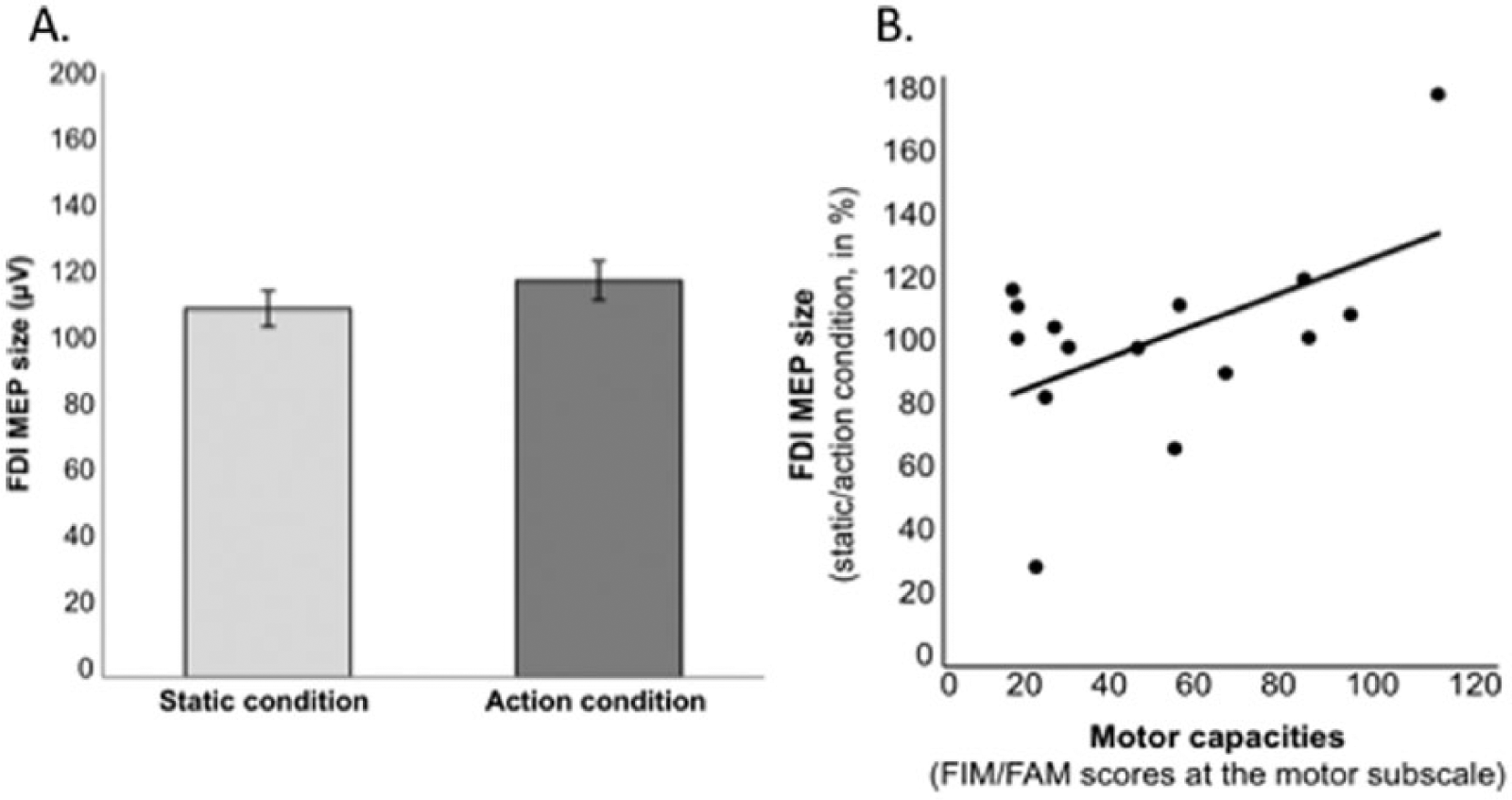
(A) FDI-MEP size of the static and action condition patients with TBI. Error bars indicate SEM. (B) FDI-MEP size of action observation in patients with TBI according to their motor capacities. Of note, FIM/FAM data were missing in 4 subjects.
TMS-Induced MEP in the TBI Group According to Motor Capacities
We examined whether motor capacities had an effect on the modulation of motor excitability during action observation. There was a correlation between FIM + FAM scores of the motor subscale and FDI-MEP size (percentage of change from moving to static stimuli; Pearson correlation; r = .536; 1-tailed value: P = .016; Figure 4). There was no significant correlation between motor capacities and ADM-MEP size (Pearson correlation: r = −.394; 1-tailed value: P = .066).
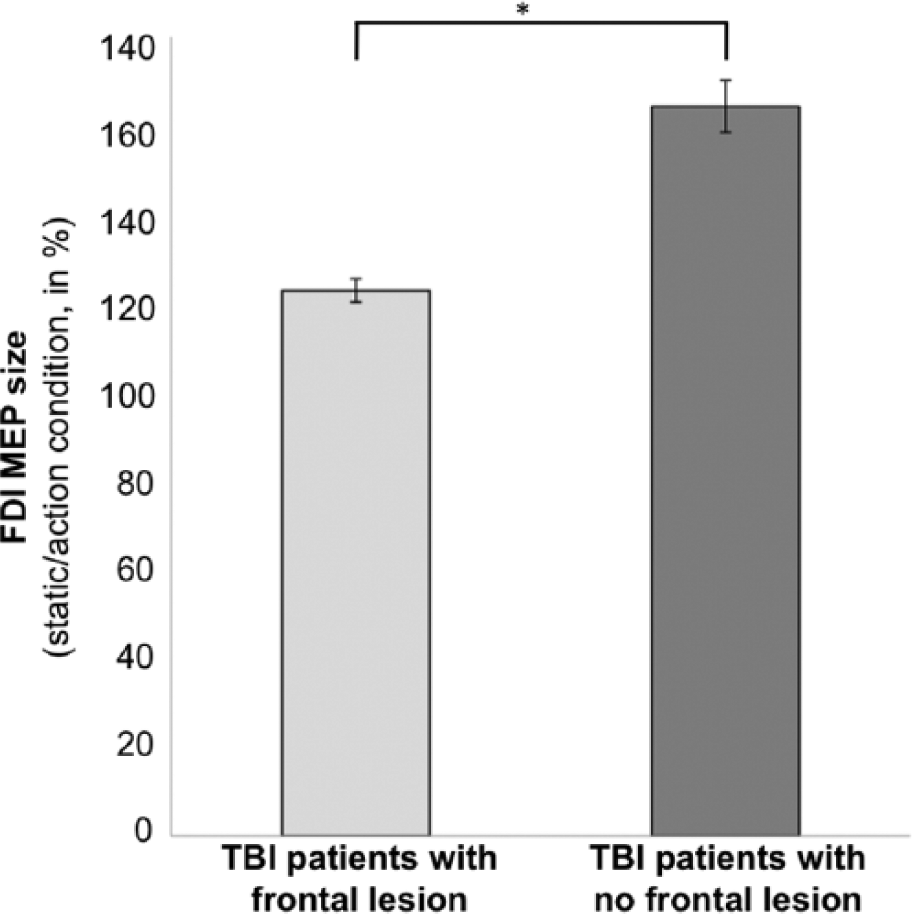
FDI-MEP size of action observation in patients with TBI according to the frontal lobe status. Error bars indicate SEM.
TMS-Induced MEP in the TBI Group According to the Frontal Cortex Status
We investigated whether the frontal cortex status had an impact on the modulation of FDI-MEP size. Twelve patients had a lesion in the frontal cortex, and 8 patients had no lesion in the frontal cortex. We found a difference between groups based on their frontal cortex status (absence, presence of a lesion; ANOVA; F1,19 = 5.66; P = .029; Figure 5). These 2 groups did not differ in modulation of ADM-MEP size (ANOVA; F1,19 = 0.38; P = .546).
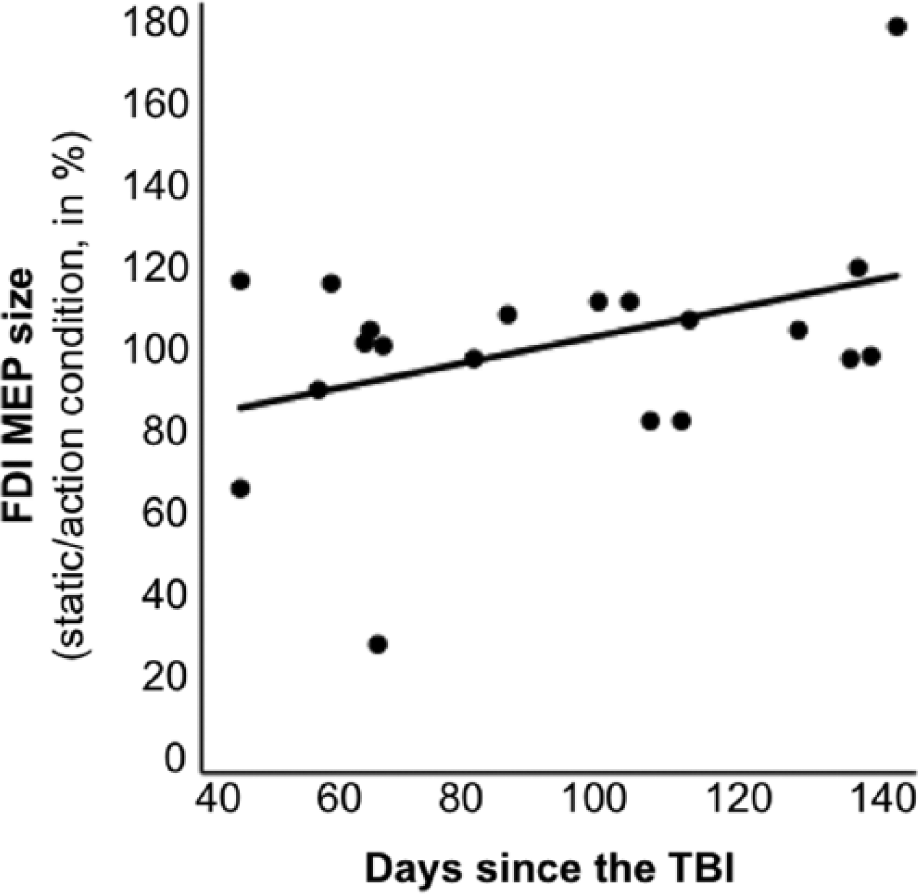
FDI-MEP size of action observation in patients with TBI according to the number of days postinjury.
We then explored whether a lesion in the left frontal cortex played a role in motor excitability elicited during action observation. Ten patients presented a lesion in the left frontal cortex, and 10 patients had no lesion in the left frontal cortex. There was an effect of left frontal cortex status (absence, presence of a lesion; ANOVA; F1,19 = 4.46; P = .049). Patients did not differ in modulation of ADM-MEP size (absence, presence of a lesion in the left frontal cortex; ANOVA; F1,19 = 0.49; P = .827).
We also tested whether a lesion in the right frontal cortex played a role in motor excitability of action observation. Nine patients had lesion in the right frontal cortex, and 11 patients had no lesion in the right frontal cortex. There was no difference between these 2 groups on the FDI-MEP size (ANOVA; F1,19 = 1.08; P = .313), nor on the ADM-MEP size (F1,19 = 0.99; P = .333).
TMS-Induced MEP in the TBI Group According to the Parietal Cortex Status
We examined whether the parietal cortex status had an impact on neurophysiologic responses of action observation. Twelve patients displayed a lesion in the parietal cortex, and 8 patients had no lesion in the parietal cortex. There was no difference between these 2 groups on FDI-MEP size (ANOVA; F1,19 = 0.02; P = .902), nor on ADM-MEP size (ANOVA; F1,19 = 1.67; P = .212).
TMS-Induced MEP in the TBI Group According to the Number of Days Postinjury
We investigated whether the number of days between the injury and the experiment had an effect on the modulation of neurophysiologic responses elicited by action observation. There was a correlation between the number of days and modulation of FDI-MEPs (Pearson correlation; r = .397; 1-tailed value: P = .042). There was no significant correlation between the number of days and ADM-MEPs (Pearson correlation: r = −.082; 1-tailed value: P = .366).
TMS-Induced MEP in the TBI Group According to Severity of the Head Injury
Finally, we examined whether severity of the head injury had an effect on the modulation of motor excitability during action observation. There was no significant correlation between GCS scores and modulation of FDI-MEPs (Pearson correlation; r = −.011; 1-tailed value: P = .481) and between the GCS scores and ADM-MEPs (Pearson correlation: r = −.139; 1-tailed value: P = .279).
Discussion
The goal of this preliminary study was to investigate the modulation of neurophysiologic responses from M1 elicited during passive observation of static and moving index finger in subacute patients with moderate to severe TBI. Observation of static and moving finger index elicited differential responses in healthy controls: Confirming prior findings, FDI-MEPs were greater during observation of action than static index finger. This is in line with an extensive literature reporting that action observation enhances motor excitability in healthy subjects.31,32 This was however not the case in patients with TBI: FDI-MEP was not significantly different between observation of action and static finger index. We further investigated motor excitability responses of action observation in patients with TBI according to factors that, based on the literature, could affect these responses: motor capacities, lesion in the frontal and parietal cortex, the number of days postinjury, and severity of the head injury.
Effects of Motor Capacities on TMS-Induced MEP During Action Observation in TBI
There was a positive correlation between motor capacities, as measured by the FIM + FAM motor scores, and modulation of FDI-MEP size during action observation. This reveals that better motor function is associated with greater modulation of motor excitability when passively observing movements. None of the patients who failed to show the expected modulation of FDI-MEP size during observation of index finger movements were able to execute themselves the isolated ab-adduction movement of the index finger. It thus appears that being able to execute a movement might be essential to preserve the resonance of the motor system to an observed action. In future work, it would be interesting to measure in a longitudinal design brain activations of action observation in patients who improve their motor capacities with time or therapy. Furthermore, a more specific motor functioning scale to the hands, such as the Wolf Motor Function test, should be included in future work. The FIM + FAM motor subscale may not be the optimal scale as it reflects upper and lower extremities as well as head and fails to provide a more fine-grained evaluation of specific motor deficits or limitation. It is however a clinically validated and broadly used measure of overall motor discapacity. As mentioned earlier, we decided to use this subscale because it was part of the routine clinical assessment.
Effects of Frontal Cortex Status on TMS-Induced MEP During Action Observation in TBI
Motor excitability of action observation was different between TBI patients with no lesion in the frontal cortex and those with lesions in the frontal cortex. This is in line with the literature on activations in the frontal cortex associated with action observation in healthy subjects. 33 For instance, Caspers and colleagues performed a meta-analysis on brain activations associated with action observation as assessed by functional magnetic resonance imaging (fMRI) and positron emission tomography (PET). They found that observation of right hand actions elicited activations in both inferior frontal gyri (pars opercularis), among other regions such as the dorsal premotor cortex and the inferior and superior parietal lobule. 23 Our results extend such findings and provide causal information on the role of the frontal role in modulation of motor cortical excitability during action observation. Of note, motor resonance likely requires perceptual awareness, which involves the frontal cortex. 34 For instance, Pobric and Hamilton found that TMS of the pars opercularis impaired perceptual judgments about other people’s actions. 35 Also, Chong and colleagues found that attention can suppress activity in the left inferior frontal gyrus during passive action observation. 36 Therefore, it is possible that perceptual awareness was impaired in TBI patients with lesion in the frontal cortex, which could in turn interfere with motor resonance.
We examined a potential difference between patients with lesions in the left or right frontal cortex. There was a weaker modulation of action observation in patients with lesions in the left, but not the right, frontal cortex. This finding supports the notion of a lateralization of fontal lobe role in motor resonance. However, MEPs were only recorded from the right hand, elicited by TMS delivered over the left hemisphere, and visual stimuli represented the right hand. The finding that left frontal lesions were associated with impaired MEP modulation may thus also be due to this unilateral testing procedure. Future work should also include MEP recording from the left hand with TMS delivered over the right hemisphere while presenting left hand stimuli.
Effects of Parietal Cortex Status on TMS-Induced MEP During Action Observation in TBI
Status of the parietal cortex had no significant impact on modulation of cortical excitability by action observation: Patients with a lesion in the parietal cortex displayed similar modulation than patients with no lesion in the parietal cortex. Some studies have reported activation in the inferior parietal areas during action observation,23,37 and some data indicate that the parietal cortex is associated with observation of goal-oriented movement more than observation of intransitive movement. For instance, Lorey and colleagues tested presentation of different conditions of action observation using fMRI. They found that the posterior parietal cortex was activated during observation for squeezing movement and observation of aiming movement, but not during observation of extension-flexion movement. 38 Similarly, Suchan and colleagues found that action observation with the instruction to subsequently imitate the viewed movement, as compared to judge the velocity of the movement, activated the left parietal cortex as measured by PET. 39 It is therefore possible that the lack of difference between our patients with lesion and no lesion in the parietal cortex might reflect that our stimuli did not sufficiently imply a goal (Figure 1). It is also possible, as mentioned above, that due to neural reorganization following a TBI the role of the normal parietal cortex in motor resonance might have been altered in our patients.
Effects of Number of Days Postinjury and Severity of the Head Injury on TMS-Induced MEP During Action Observation in TBI
Modulation of motor cortical excitability during action observation was correlated with the number of days postinjury. Greater modulation was observed with greater number of days. This supports the notion that modulation of motor excitability changes with time, indicating a natural improvement of the modulation of cortical excitability. Severity of the head injury (moderate, severe) as indexed by the GCS scores did not affect motor excitability of action observation. It is possible that severity of the head injury does not affect differently motor excitability of action observation. It is also possible that, in the subacute phase, both moderate and severe TBI are associated to weaker modulation of motor excitability, but with time they would show different neurophysiologic responses. Also, it is possible that the GCS scores are not sensitive enough for this given experiment focusing on moderate and severe TBI. Future longitudinal work with greater sample size including patients with mild, moderate, and severe TBI is required to further understand whether change in the modulation of motor excitability during action observation may occur over time.
Study Limitations and Perspectives
This study has important limitations to be considered. First, the sample size was modest (6 patients with moderate and 14 patients with severe TBI) in their subacute phase, tested between 46 and 147 days postinjury). Thus, the lack of significance in some analyses, such as severity of the head injury, might be attributed to underpowered analyses. Studies with larger sample size are needed to conduct multiple regression analyses to shed light on how patients’ characteristics interact and to evaluate the unique contribution of each one of these factors. Second, we did not directly analyze the CT and MRI scans (eg, lesion size; interrater reliability tests) because we only had access to the medical records. Also, future work should explore whether the types of lesions (eg, diffuse axonal injury, hemorrhage, contusions) and total lesion volume play a role on motor excitability. Longitudinal studies with greater sample size and repeated assessments of motor functions are warranted to explore potential clinical value of action observation in TBI patients. Such design would identify whether some patients show spontaneous remission and those who do not. In addition, studies including multiple methods of neuroimaging, especially functional connectivity MRI and diffusion tensor imaging, are warranted because MEPs are likely to reflect intact and damage connections between various brain regions, whether a given region is intact or not according to an anatomical MRI or CT brain scan as used here. Another limitation is the lack of an eye tracking system to ensure that patients were observing the stimuli. Furthermore, the cognitive status and cooperation of the patients during testing is a possible confounding factor in the results, as it may affect processing of the visual stimuli.
Despite these caveats, findings from this preliminary work support the notion that motor excitability of action observation is impaired in moderate and severe TBI in the subacute phase, especially in patients with limited motor capacities and/or lesion to the left frontal cortex. Passive action observation is believed to contribute to action understanding 40 and motor learning, 41 which appear to be impaired in TBI.42,43 From a practical perspective, assessment of the integrity of motor excitability of action observation may hold clinical value and ultimately guide rehabilitation strategies.
In healthy subjects, emotionally loaded stimuli elicited greater TMS-induced motor excitability than neutral stimuli. 44 Here patients displayed weak motor excitability when passively viewing simple finger movement. It would be interesting in future work to present larger set of stimuli, including movements that are individually impaired and intact in TBI patients in order to explore whether more meaningful stimuli would elicit greater modulation of excitability. Moreover, we previously showed that behavioral experience, such as visual priming, can refine these neurophysiologic responses. 30 This might also be relevant in neurorehabilitation in TBI, such as priming the brain network in order to elicit greater modulation of motor excitability. Finally, future protocols should also include additional conditions, for instance, observation with the intent to imitate the observed movement such as Ertelt and colleagues, who found improved motor functions in stroke patients who were asked to observe movements and execute them. 8
Footnotes
Acknowledgements
We thank all participants and their families for participating in this research project.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Pascual-Leone serves on the scientific advisory boards for Nexstim, Neuronix, Starlab Neuroscience, Neuroelectrics, Axilum Robotics, Magstim Inc., and Neosync; and is listed as an inventor on several issued and pending patents on the real-time integration of transcranial magnetic stimulation (TMS) with electroencephalography (EEG) and magnetic resonance imaging (MRI).
The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Catalyst, Harvard University and its affiliated academic health care centers, the National Institutes of Health or the Sidney R. Baer Jr. Foundation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Canada Research Chair in Cognitive Neuroplasticity to SF, from the Sidney R. Baer Jr. Foundation, the National Institutes of Health (R01HD069776, R01NS073601, R21 MH099196, R21 NS082870, R21 NS085491, R21 HD07616), Harvard Catalyst | The Harvard Clinical and Translational Science Center (NCRR and the NCATS NIH, UL1 RR025758). to APL, and by the Foundation La Marató TV3 (071931) and Grant PI082004 from the Instituto de Salud Carlos III to JMT.
