Abstract
Introduction
Stroke is a leading cause of death and serious long-term disability.1,2 Unilateral upper-limb impairment is one of the most frequent and persistent disabilities reported in stroke survivors.3-6 Stroke affects a diverse age group, with about 34% of the patients hospitalized for stroke in 2009 being younger than 65 years. 7 A number of rehabilitative strategies are used with stroke survivors, but they are ineffective in attaining full recovery. Most patients achieve a behavioral outcome that represents about 70% of their prestroke ability after much less intensive therapy than believed to be necessary to optimize function. 8 To improve the efficacy of rehabilitative strategies, a better understanding of the impact of various behavioral interventions and the mechanisms that support them is necessary.
Rats are commonly used as behavioral models in stroke rehabilitation because of their ability to learn complex tasks that are similar to human activities, such as reaching and grasping objects. Furthermore, rats exhibit deficits in forelimb function following stroke that resemble the upper-limb impairments observed in humans.9-16 Though mice have been a less-popular choice as model systems for behavioral research, they are useful for modeling human behavioral and anatomical deficits because of their motor map homologies with humans and their dexterous forelimb use.17-20
It has been repeatedly demonstrated that skilled reaching training improves motor performance following stroke in rodent models,21-23 but these effects can vary with the amount of task practice. 24 Whether the efficacy of skilled reach training is altered by the intensity as well as the quantity of training has not been determined. High-intensity (HI) motor skill training increases skill acquisition rates in intact mice. 25 The present study was conducted to determine the impact of HI rehabilitative training on reach performance following ischemic sensorimotor cortical (SMC) damage in mice. SMC is involved in motor skill acquisition and performance,26,27 and sufficient SMC damage results in enduring forelimb impairments in rodents that bear resemblance to clinical poststroke upper-extremity impairments.11,14,16,28,29 Our first experiment tested the influence of training intensity over a restricted time span. The second experiment matched the total number of training sessions and investigated the maintenance of performance improvements following cessation of rehabilitative training. Results suggest that higher training intensity affects the efficiency of rehabilitative training, as indicated by faster rates of functional improvements per time and training session.
Materials and Methods
Participants
A total of 52 male C57BL/6 mice from Jackson Laboratories (Bar Harbor, ME) were obtained at 2 months of age and maintained on a 12:12 light-dark cycle. Mice were group housed (4/cage) and received standard enrichment (ie, 8 hours of classical music during the dark cycle, nesting material, PVC pipe, small wooden objects for chewing, and a piece of a cardboard roll).30,31 Food restriction was implemented 5 days prior to preoperative training and maintained throughout the experiment. Mice were fed 2.5 to 3 g of standard rodent chow daily. Weights were monitored and food intake was adjusted to maintain a body mass ≥85% of free-feeding body weight.
In experiment 1, 28 mice received preoperative training on the pasta matrix reaching task (PMRT). One animal did not meet reaching criteria (described below in Behavioral Training). A total of 27 mice received ischemic insult via intracortical infusion of endothelin-1 (ET-1; see Surgical Procedures). Three mice did not recover from perioperative anesthesia and were excluded from the study. The remaining 24 mice were randomly assigned postoperatively to a high intensity (HI; n = 8), low-intensity (LI; n = 8), or no-training group (n = 8). In experiment 2, 24 mice received preoperative training and intracortical infusion of ET-1. Two mice were excluded from the study because of postoperative (PO) mortality. The remaining 22 mice were randomly assigned to a HI (n = 11) or LI (n = 11) group. All animal use was in accordance with a protocol approved by the Animal Care and Use Committee of the University of Texas.
Surgical Procedures: Intracortical Infusion of ET-1
Following preoperative behavioral training, mice were given unilateral intracortical infusion of ET-1 as described previously. 20 Mice were anesthetized with ketamine (100 mg/kg, intraperitoneal [ip]) and xylazine (10 mg/kg, ip) and placed into a stereotaxic frame (Stoelting, Wood Dale, IL). Lidocaine (2 mg/kg, subcutaneous [sc]) was injected into the scalp, and a midline incision was made. A small burr hole was drilled over the center of the forelimb region of SMC (0.3 mm anterior to Bregma; 1.5 mm from the midline). A total of 3 µL of ET-1 (American Peptide; 320 pmol, 0.2 µg/µL in sterile saline) was injected into layer V of the motor cortex 800 µm below the surface of the cortex over the course of 10 minutes. The burr hole was then filled with gelfoam and covered with UV curing dental cement (Wave A2; Southern Dental Industries, Victoria, Australia). The wound was sutured and covered with antibiotic ointment before the animal was injected with 3 mL of prewarmed, 37°C electrolyte (Ringer’s solution, sc). All mice awoke in a heated chamber before receiving buprenorphine (3 mg/kg at 0.015 mg/mL in sterile saline, sc) and returning to the home cage.
Behavioral Training
The mouse PMRT chamber was adapted from a rat chamber, 32 as described previously. 20 Mice were trained to reach through a small slit located in the center wall of a Plexiglas chamber to break pieces (3.2 cm in height, 1 mm in diameter) of vertically oriented, uncooked capellini pasta (De Cecco brand). Pasta pieces were located 2 mm apart in a 10 × 10 grid in a heavy-duty plastic block located outside of the reaching chamber. For a successful reach, mice were required to reach across their body, through the reaching aperture, to grab, break, and retrieve a piece of pasta protruding from the matrix block.
Prior to training, all mice underwent shaping procedures in which limb preference was determined. Animals were placed individually in reaching chambers once daily for 3 to 5 days. During this time, the matrix stage was completely filled, allowing mice to reach for pasta with both limbs. Each daily trial lasted for 10 minutes or until the mouse reached a minimum of 10 times. A reach was defined as extending a single limb through the reaching aperture such that the entire forepaw was outside the chamber walls. Limb preference was determined when a minimum 70% of daily reaches were made with a single forelimb.
Preoperative Training
Following shaping procedures, mice were trained on the PMRT with their preferred limb. Preoperative training is necessary to establish a motor skill under a controlled environment, mimicking general motor skills observed in humans. During training, one half of the matrix was filled with pasta (contralateral to the preferred limb), forcing the mice to use their preferred limb to reach for pasta pieces. Each daily session consisted of 15 minutes or 100 reaches, whichever occurred first. Mice were trained 6 days a week for a total of 16 days. Preoperative performance level was determined by averaging the total number of pasta pieces broken the final 3 days of training. All but 1 mouse reached criterion performance, which was established as successful retrieval of 9 pasta pieces (half of the theoretical maximum 20 ) and received ET-1 lesions (PO day 0).
Postoperative Training
On PO day 4, mice were probed with a single PMRT session targeting their affected limb to assess deficits following ischemic insult. On PO day 5, the mice began rehabilitative training based on their experimental group requirements. The HI group was trained with their affected limb twice daily with 90 minutes between each session. The LI group was trained with their affected limb once daily. Each training session was identical to preoperative training. The control group was placed in reaching chambers and given bits of capellini too small to handle to simulate the time training and consumption of pasta in the absence of dexterous forelimb use.
In experiment 1, mice were trained for 9 consecutive days (PO days 5-13) before receiving a final probe (single PMRT session) on PO day 14. They were then sacrificed and perfused intracardially, and brain tissue was harvested. In experiment 2, mice in the HI group were trained for 9 consecutive days (PO days 5-13) before beginning a regimen of probe sessions every 10 days. The LI group was trained for 19 consecutive days (PO days 5-23) before beginning a regimen of probe sessions every 10 days. This procedure ensured that the HI and LI groups received the same number of training trials during rehabilitation training, permitting the assessment of differences between intensity and trial quantity. The probe trials following the rehabilitation allowed for the assessment of potential differences in retaining the improved performance based on varying intensity. Following the last probe (PO 84), all mice were sacrificed and perfused intracardially.
Tissue Processing and Lesion Analysis
After the final probe on the PMRT, mice were euthanized with an overdose of sodium pentobarbital (euthasol, 175 mg/kg, ip) and perfused intracardially with approximately 25 mL of 0.1 M phosphate buffer and approximately 50 mL of 4% paraformaldehyde delivered at 10 mL/min. The brains were harvested and stored in 4% paraformaldehyde at 4°C for at least 1 week before the tissue was sectioned into 50-µm thick coronal sections using a vibratome (Leica Biosystems, Wetzler, Germany). Every sixth section was mounted onto gelatin-coated slides and Nissl stained with toluidine blue.
Neurolucida software was used to estimate lesion volume. Coronal sections were viewed at a total magnification of 17. The cortical areas of 8 coronal sections from approximately 2 mm anterior to 1.5 mm posterior to bregma, each 300 µm apart, were measured by tracing cortical boundaries of both the contralesional and ipsilesional cortex. The SMC fell within the area of tissue measured, and no lesion extended beyond these boundaries. Cortical volume was estimated with the Cavalieri method, by multiplying the sum of section areas by the distance between sections.33,34 Lesion volume was calculated as the difference between the contralesional and ipsilesional cortex.
Lesion reconstruction was performed to visualize the placement and consistency of the lesions within and between experimental groups. Nissl-stained sections were visualized under a microscope and copied to a coronal section template paper. The illustrations were then scanned, digitalized using Adobe Photoshop, and overlaid onto a master template. The master template is darker in areas where more animals have overlapping lesions.
Statistical Analysis
The statistical significance of the behavior was determined by using SPSS software. Univariate analyses of variance (ANOVA) were run for all preoperative comparisons. Planned comparisons with a Bonferroni correction were run for all probe trials in experiment 1. Repeated-measures ANOVA were run for PO data in experiments 1 and 2 using day as a within-subjects variable and intensity of training group as a between-subjects variable. The level of significance for all tests in this study was an α level of .05. Data are presented as mean ± standard error of the mean (SEM).
Results
Lesion Volume and Reconstruction
ET-1 infusion resulted in damage in all mice. In experiment 1, ET-1 infusion produced damage to the forelimb representation area of the motor cortex, extending in an approximately 1 mm radius from the infusion site (Figure 1). The average interhemispheric volume difference was 2.00 ± 0.43 mm3 (Figure 1A). In experiment 2, ET-1 infusion produced similar damage, with an average lesion volume of 2.78 ± 0.32 mm3 (Figure 1C). No damage was observed in the underlying white matter or striatum in either experiment (Figures 1B and 1D).
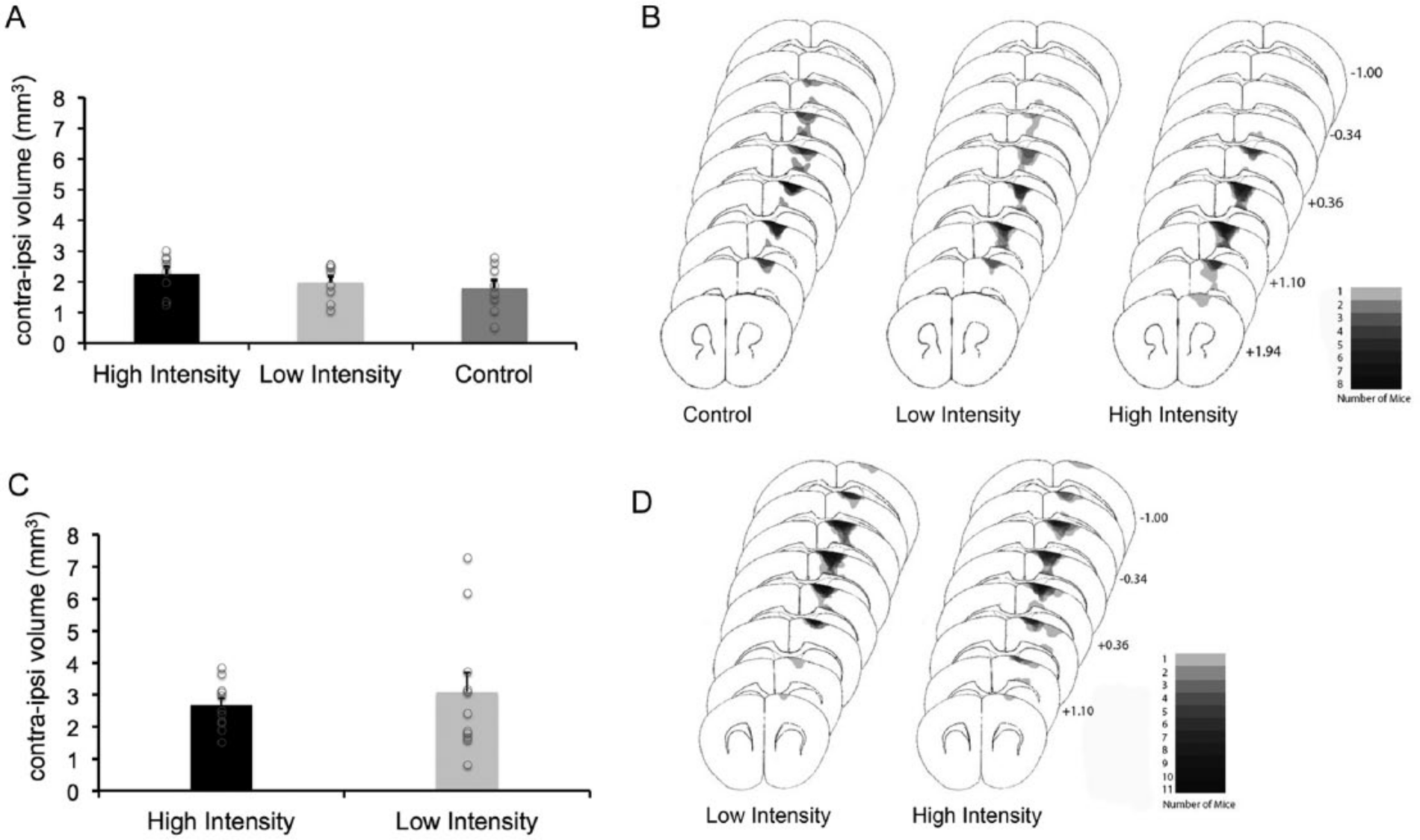
Lesion verification and analysis for experiment 1 (A and B) and 2 (C and D). There were no significant differences between groups in lesion size for experiment 1 (A) or 2 (B). Individual lesions are represented by open circles. Lesions were also consistent in placement, centered around the sensorimotor cortex (experiment 1B; experiment 2C). Error bars represent standard error of the mean.
As seen in Figures 1A and 1B, lesion volumes were generally similar among groups in both experiments. In experiment 1, there was an overlapping range of, and no significant difference in, lesion volumes between HI, LI, and control mice (
The lesions in experiment 1 were consistently placed in the motor cortex anterior of bregma, with high consistency between +1.10 and +0.36, as demonstrated in Figure 1B. Lesion reconstruction for experiment 2 revealed a similar pattern of damage, though lesions were consistently placed slightly more posterior in the motor cortex (Figure 1D).
Experiment 1: HI Training Speeds the Temporal Rate of Functional Improvement
Results from experiment 1 are depicted graphically in Figure 2. Data from the HI group represent an average of the 2 daily training sessions. All mice in experiment 1 demonstrated similar preoperative performance levels on the PMRT, with no statistical difference between groups (
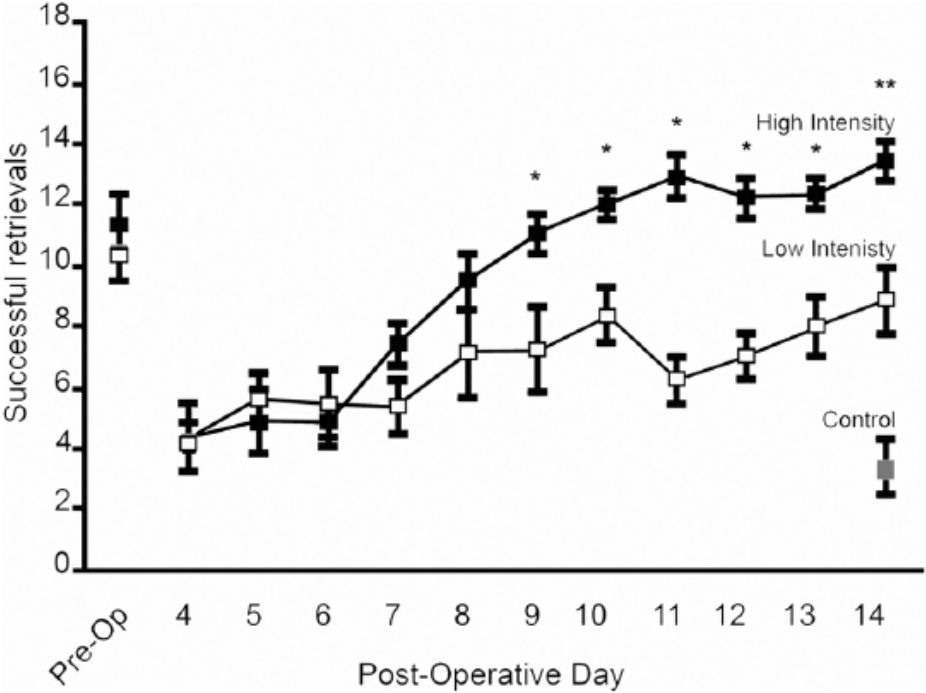
Experiment 1: high-intensity training results in improved behavioral outcome compared with low-intensity training. High-intensity training resulted in significantly better performance beginning on postoperative day 9 through the termination of motor skill training.a
HI training resulted in significantly better performance than did LI training, as confirmed by a repeated-measures ANOVA (
Experiment 2: HI Training Speeds the Per-Session (or Per-Trial) Rate of Functional Improvement
Results from experiment 2 are depicted graphically in Figure 3. There were no statistical differences between groups on the final preoperative training day (
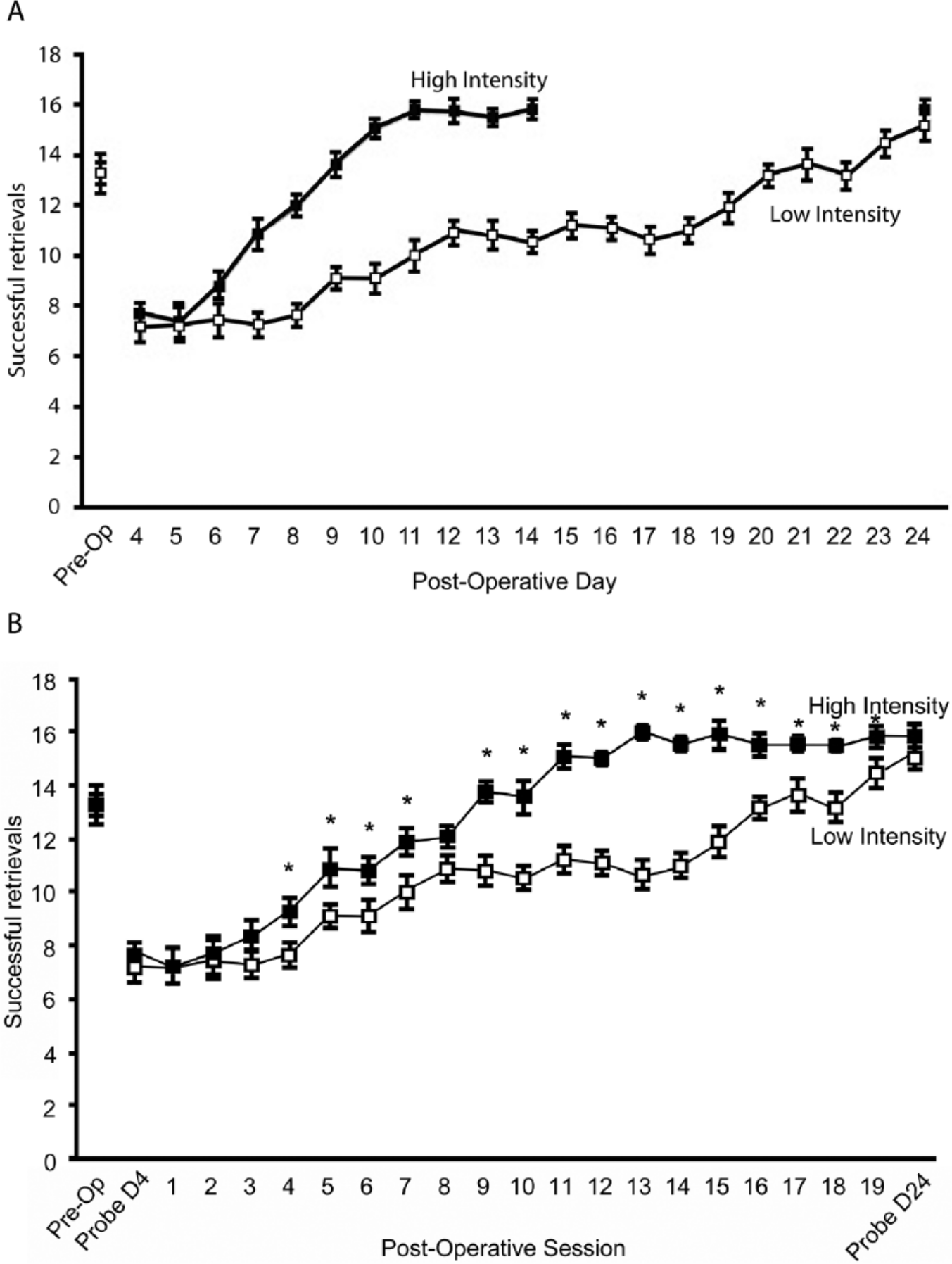
Experiment 2: high-intensity training results in faster functional improvement but not better skilled motor performance. When the number of training sessions were accounted for, training regimens resulted in similar behavioral outcome (B). However, a session-by-session analysis revealed that high-intensity training resulted in quicker gains of function, with low-intensity trained mice requiring the full motor skill training regimen to reach a similar performance level (B).a
Performance per training session was also analyzed. A repeated-measures ANOVA confirmed a significant group effect in session performance (
Experiment 2: Training Intensity Did Not Consistently Affect Maintenance of Performance Improvements
Following the cessation of rehabilitative training, there was a gradual decrease in successful retrievals in both groups as time progressed (Figure 4). However, all mice continued to exhibit performance levels that were above their initial postoperative performance. The pattern of behavioral performance was consistent between groups following the cessation of rehabilitation. A repeated-measures ANOVA confirmed that there was no statistical difference between groups in postoperative probe performance (
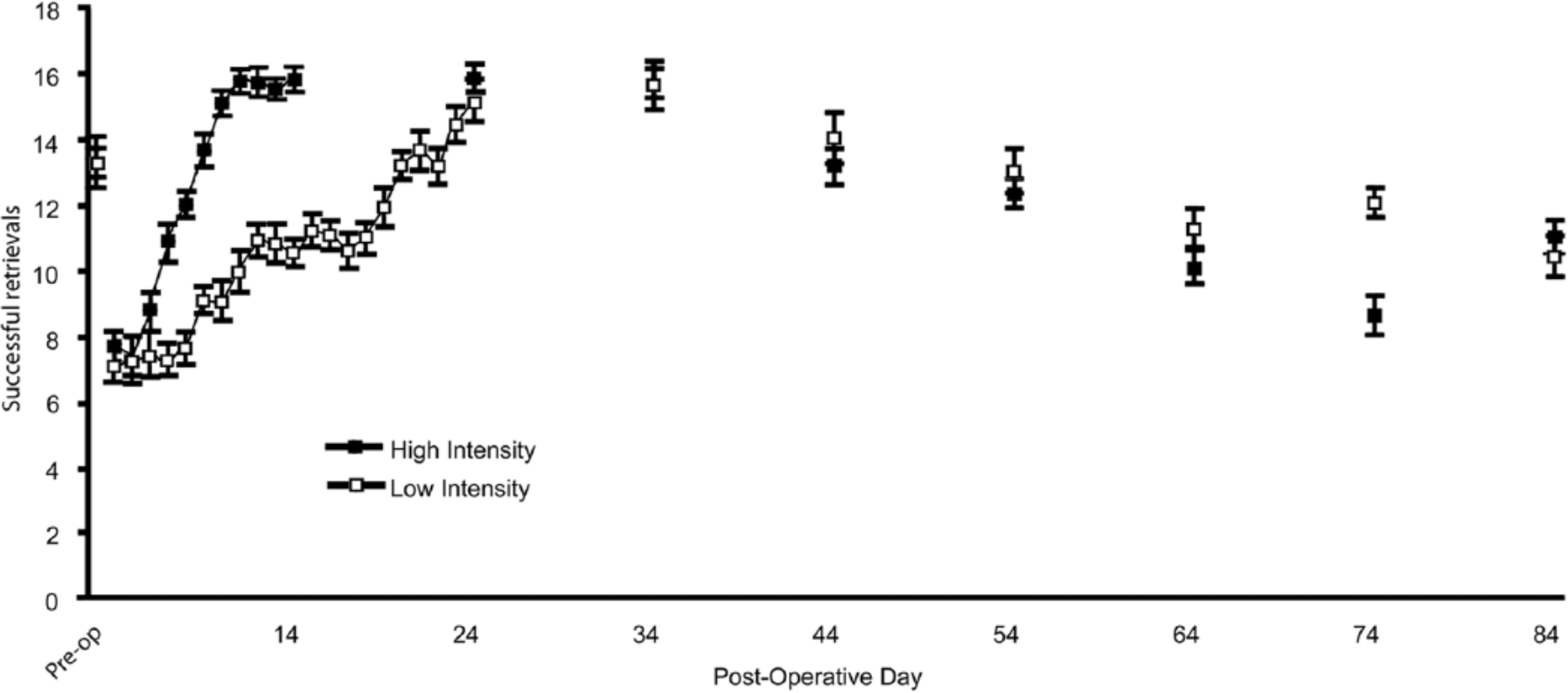
Experiment 2: performance maintenance over time. Both high-and low-intensity trained mice exhibited a decrease in performance levels following the cessation of rehabilitative motor skill training. There were no differences between groups in the maintenance of performance improvements over time. Error bars represent standard error of the mean.
Discussion
C57BL/6 mice have been previously established as a reliable model for behavioral deficits following focal ischemic insult of the SMC,20,35 with impairments resembling those produced by similar lesions in rats29,36,37 and chronic impairments observed in human stroke survivors. 38 It has also been established that focused skilled training following SMC damage improves skilled motor performance in rats and mice.35,39 In this study, we report that HI rehabilitative training leads to better behavioral outcome following ischemic insult. Furthermore, increasing the period of time over which training occurs, at either LI or HI, may increase the persistence of rehabilitated performance of a skilled reaching task.
Previous research reports that motor skill learning in intact mice progresses more quickly with increased training intensity. 25 Mice trained on the PMRT with 2 daily training sessions exhibit faster acquisition of the task and more persistent performance. Our results suggest that this “intensity effect” also plays a role in motor skill rehabilitation, with higher levels of intensity favoring a quicker return to preinjury performance levels (see Figure 2). These findings are in concert with similar results reported in a rat model of stroke, whereby a greater number of training trials resulted in better recovery from injury, although rats never reach preoperative performance levels. 24 There are several important differences between the current study and that of MacLellan et al. 24 Although both rodent models use ET-1-induced stroke, the damage in the rat model is much more extensive and may, therefore, result in behavioral deficits that are more extensive and difficult to rehabilitate. Differences in task assessment (staircase task vs PMRT) and/or species differences may also explain differences in recovery potential. Although the motor cortex of both rats and mice share extensive homologies with the human motor cortex, the mouse motor cortex expresses greater redundancy, with a greater proportion of digit representation, 40 which may result in more behavioral flexibility and improved recovery potential. The extent of digit representation observed in the mouse also suggests greater homologies between mouse and human motor cortices compared with that of the rat.
Unlike previous studies, 24 the current study establishes an effect of training intensity that is independent of training quantity (see experiment 2). By examining the number of training trials, it appears as though HI training merely accelerates functional improvement because it involves additional training trials (see Figure 3). Although this appears to be the case, it is interesting to note that a session-by-session analysis of reach performance reveals that although LI training successfully improves behavioral outcome, it does so at a slower pace than HI training regardless of the number of training sessions. HI training results in reaching performance that is similar to preoperative levels in fewer training sessions than does LI training (see Figure 3B). In fact, it is not until the final training session that the LI group reaches performance levels similar to that of the HI group.
Although HI training increases the rate of functional improvements during the training regimen, it does not benefit the long-term retention of these improvements (see Figure 4). It was hypothesized that a quicker, more-intense rehabilitation would limit retention of the reacquired skills compared with the slower, less-intense rehabilitation because the neural pathways would not have as much time to become permanent, and this would negatively affect skill persistence. 41 When comparing the impact of HI training and LI training on long-term maintenance of performance improvements following the cessation of rehabilitation, the 2 groups did not exhibit statistical differences until 74 days after injury, when the LI group appeared to show an increase in rehabilitative persistence. However, it is difficult to conclude that this indicates better overall rehabilitation in the LI group because there were no differences between groups on the final probe day (84 days after injury).
Correlational analyses revealed no significant relationships between lesion volume and performance level, indicating that behavioral results cannot be explained by lesion size. However, the present study was not designed to explore the influence of lesion severity. It remains possible that lesion size and training intensity interact—for example, training intensity may moderate the influence of lesion severity. Future studies should consider the impact of differing levels of intensity training after lesions of varying severities.
There was a general decline in successful reach performance with the cessation of rehabilitative training in all groups. Similar results have been commonly reported in rats after 2 months following the cessation of rehabilitative motor skill training10,42; performance returned to optimal levels with a few rehabilitation sessions. 42 Similar declines have also been detected in intact rats following periods of inactivity after motor skill learning (O’Bryant, Adkins, Sitko, Combs, Nordquist, & Jones, unpublished data, manuscript in revision). It is believed that the decline in reach success is the result of a lack of practice following acquisition of a new motor skill. That is, without continued practice of the newly acquired motor skill, the quality of the skill decreases. These results suggest that rehabilitative training following injury results in neuronal changes that resemble new motor skill acquisition and point to the importance of continued motor skill practice following injury in order to maintain optimal behavioral performance. It is possible that such changes reflect a new way of performing the skill (such as with compensatory limb movements) or a rewiring of the original motor engram that was damaged by the ischemic event (ie, true recovery).
It is well established that performance improvements resulting from rehabilitative training reflect, at least in part, the development of compensatory movement patterns rather than the recovery of the movements used prior to the lesion.43-45 The increased time course in the LI group may suggest a learning mechanism whereby mice have acquired a new, compensatory strategy to complete the PMRT. Such compensatory strategies have been observed in similar studies of monkeys.46,47 Although the present study did not include a kinematic analysis of individual reaches, we did assess the pattern of successful reaches across sessions (supplementary Figure 2). This analysis revealed that HI training results in a greater number of successful distal reaches early after injury. After approximately 1 month of training, reach patterns between the 2 groups are more similar. Further studies involving kinematic analysis of individual reaches as well as the anatomical changes occurring with both intact motor learning and poststroke rehabilitative training would be necessary to determine the mechanism of recovery in the current paradigm.
Whereas human stroke exhibits greater heterogeneity than our mouse model, the present study suggests that skilled motor behavior, compromised following stroke, benefits from HI, focused training of the impaired limb. Typical clinical rehabilitation is not nearly as frequent as that modeled in animal studies, and in many cases, the rehabilitation in the clinical setting does not start as early as basic research studies have suggested that it should.8,48 Our findings indicate that an ideal poststroke therapy would include a HI rehabilitation regimen that continues for long enough to maximize performance improvements and, as other studies suggest, ideally begins early after a patient has suffered a stroke. 49 However, increasing the intensity of rehabilitative therapies to at least match that practiced in basic research studies seems likely to be beneficial in promoting better long-term recovery of function in stroke patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH RO1 NS056839.
