Abstract
Introduction
Motor impairments, including upper limb hemiparesis, are the most common deficits affecting ~80% of all stroke patients.1,2 Such impairments result from damage to specific cortical regions (eg, primary motor cortex), subcortical structures (eg, basal ganglia), and/or the corticospinal tract. 3 Indeed, evidence from multiple large-scale prospective clinical studies indicate that strokes primarily affect subcortical structures such as the basal ganglia, white matter, and/or the thalamus.4-6 In these studies, pure cortical strokes accounted for less than ~15% of the total number of strokes whereas subcortical-only and combined cortico-subcortical strokes occurred at a much greater frequency.
Despite the high prevalence of subcortical strokes, preclinical stroke research focuses almost exclusively on cortical strokes. A recent study compared lesion profiles between preclinical studies and a clinical trial (ICARE) and found that current preclinical models result in either too large or superficial strokes. Importantly, it is noted that these preclinical models of stroke are likely not representative of the segment of the clinical population with relatively small strokes. 7 Mismatch in lesion characteristics between preclinical and clinical research could negatively affect translational efforts. 7
Although several studies have examined the association between location of stroke injury and motor outcome, very few have directly compared recovery following cortical versus subcortical strokes. Some studies show that location of lesion matters, and that patients with a cortical injury show better motor recovery than those with a purely subcortical stroke (eg, basal ganglia, internal capsule).8,9 In contrast, others have reported that the location of lesion (cortical/subcortical) does not affect functional outcome.10,11 Clearly, further research is needed to clarify the effect of lesion location on motor recovery following stroke.
Both humans and animals demonstrate some degree of spontaneous biological recovery following stroke.12-14 However, the heterogeneity of stroke location and impairment severity in humans, coupled with the fact that the vast majority of stroke survivors receive rehabilitation, presents a challenge to investigating spontaneous recovery in humans. Preclinical models of stroke provide a better means to study spontaneous recovery since the poststroke environment can be easily controlled and manipulated.
The aim of the present study was to capture the heterogeneity of ischemic human stroke by investigating how lesion location and size influences spontaneous motor recovery. Focal strokes were directed at the forelimb motor cortex (FMC; homologous to the human primary motor cortex) and/or the dorsolateral striatum (DLS; homologous to the human caudate and putamen). 15 Using a battery of behavioral tests, we characterized the spontaneous recovery profile of the animals over an 8-week period.
Methods
Subjects
A total of 30 male, 3-month-old, Sprague-Dawley rats (Charles River Laboratories, Montreal, Quebec, Canada) weighing ~200 to 250 g on arrival were used. Animals were pair-housed in standard plexiglass cages on a reverse 12-hour light/dark cycle, and had ad libitum access to food and water except during behavioral training and testing periods. On arrival, animals were acclimatized to the housing facilities for one week and then handled daily for 5 days. All experimental procedures were approved by the University of Ottawa Animal Care Committee and complied with Canadian Council on Animal Care (CCAC) guidelines.
Stroke Induction
Cortical, striatal, or combined (cortical + striatal) strokes were produced by intracerebral injections of the vasoconstricting peptide endothelin-1 (ET-1) as previously described.16,17 Rats were anesthetized with isoflurane (4% induction, 1.5%-2.0% maintenance in oxygen) and secured in a stereotaxic frame. A small incision was made in the scalp and 1 (striatal-only), 2 (cortical-only), or 3 (combined) small, ~1.0-mm diameter burr holes were drilled in the skull. The stereotaxic injection coordinates targeted the FMC and/or DLS (Supplementary Table 1). All strokes were produced in the hemisphere opposite the dominant forelimb as determined by baseline performance on the staircase test.
At each injection site, a Hamilton syringe (80366; Hamilton, Reno, NV, USA), was slowly lowered into the brain and after a 1-minute delay, 1 μL of ET-1 (400 pmol/μL in sterile water; E7764; Sigma-Aldrich, St Louis, MO, USA) was infused at a rate of 250 nL/min. Following injection, the needle was left in place for an additional 2 minutes and then slowly withdrawn to minimize backflow of ET-1. The incision was sutured and a topical anesthetic (2% bupivacaine cream, Chiron, Guelph, Ontario, Canada) was applied. Animals were then placed in an incubation chamber and after awakening administered a subcutaneous injection of buprenorphine (0.05 mg/kg; Chiron, Guelph, Ontario, Canada) before being returned to their home cages.
In a number of previous studies we performed sham injection surgeries and found that they had minimal damage 18 and do not result in any behavioral impairments.19,20 In keeping with CCAC guidelines to reduce the number of animals used in research, we have not repeated this control condition.
Infarct Confirmation by Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) was performed 72 hours following surgery to verify successful infarcts. Scans were obtained using a 7T General Electric/Agilent MR901 small-animal scanner. Animals were anesthetized with isoflurane (4% induction; 2% maintenance) during the 15-minute procedure, and respiratory and cardiovascular function was monitored throughout with a SA Instruments Inc (SAII, Stony Brook, NY, USA) physiological monitoring system. A localizer scan was first performed, during which routine adjustments were made. T2-weighted structural images were obtained using the following parameters: 15 coronal slices; slice thickness = 800 µm; in-plane resolution = 78 µm; echo train length = 8; echo time = 27 ms; scan time = 5 minutes. MRI stacks were viewed using ImageJ (National Institute of Health, Bethesda, MD, USA) to verify and confirm that the infarcts occurred in the targeted location (FMC, DLS, or FMC + DLS). In each slice, any observable striatal and/or cortical damage was manually traced and the infarct area for each region was measured. Infarct volumes were calculated by multiplying the sum of infarcted area for all measured slices by the MRI slice thickness (0.80 mm).
Behavior Training and Testing
Prior to stroke, animals were trained on the Montoya staircase, beam, and adhesive strip removal task (Supplementary Section) and their baseline performance assessed. Baseline performance was also assessed on the cylinder test, which did not require pretraining. Subsequent behavioral testing was conducted at poststroke weeks 1, 2, 4, 6, and 8 (Figure 1). Testing took place during the animals’ dark cycle over 3 days at each time point. Experimenters were blinded to experimental condition during behavior testing and analysis.
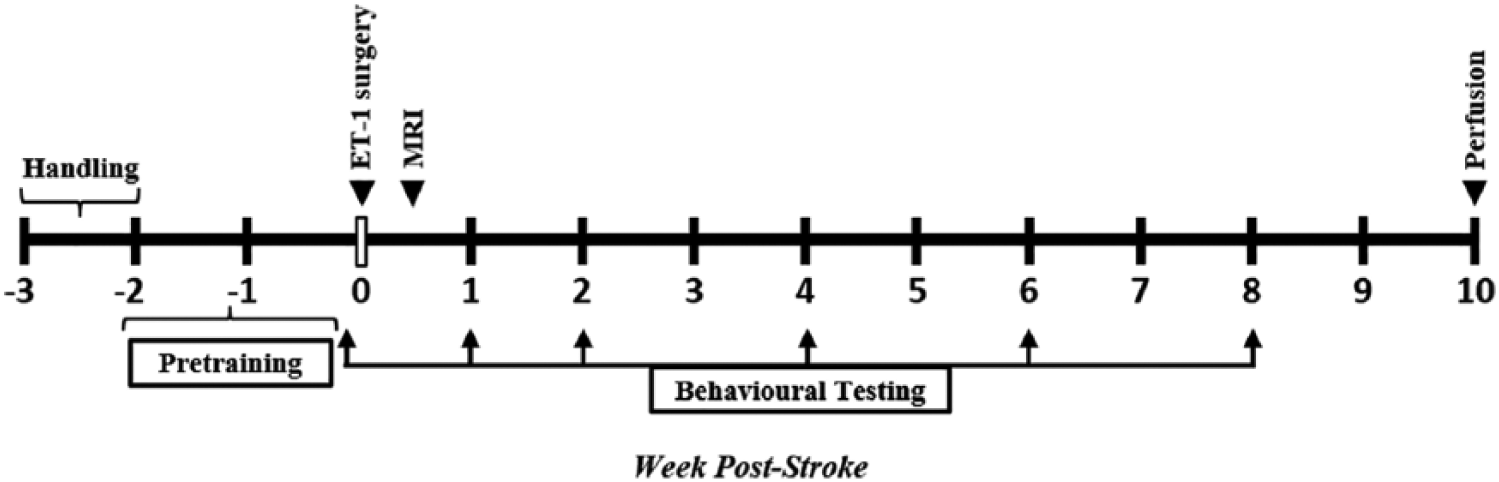
Experimental timeline. Following handling, animals were trained on behavioral tests over a period of two weeks and baseline performance was assessed. Following endothelin-1 (ET-1) stroke, behavior testing was conducted at poststroke weeks 1, 2, 4, 6, and 8. Magnetic resonance imaging (MRI) was performed at 72 hours poststroke to verify infarcts. Animals were perfused at 10 weeks.
Montoya Staircase Test
The staircase test requires animals to use fine motor skills to grasp and retrieve small food pellets with their forepaw. 21 The staircase chamber has 7-tiered descending steps on each side of a central platform. Each step has a food well containing 3 food pellets (45 mg; TestDiet, Richmond, IN, USA). The design of the staircase apparatus allows animals to retrieve pellets placed on steps only on the same side as their paw. Forepaw dexterity was measured by counting the total number of pellets retrieved (PR) versus those that were left or dropped.
Food restricted (90% of body weight) animals were trained on this task twice daily for 2 weeks (2 trials/day; 15 min/trial; 3 hours apart) to reach a minimum criterion of at least 14 ± 2 pellets. Prestroke performance was calculated as the average of all trials over the last 2 days of training. All poststroke time points were conducted over 3 day periods and performance was based on the last 2 days (2 trials/day; 15 min/trial; 3 hours apart). Percent improvement at 8 weeks was calculated as follow: % improvement = ([PRWeek 8/PRPrestroke] − [PRWeek 1/PRPrestroke]).
Cylinder Test
The cylinder test was used to assess asymmetrical limb use for postural support. 22 Rats were placed inside a tall plexiglass cylinder (20 cm diameter, 30 cm tall) on a glass tabletop and video-recorded from below. The number of independent (ipsilateral and contralateral) and bilateral wall forelimb contacts was determined from recorded videos, and the percentage of impaired forelimb use was calculated as follows 22 : % use of impaired paw = ([contralateral contacts +1/2 bilateral contacts]/total contacts) × 100%. A trial continued until a rat performed a minimum of 20 independent rears where they touched the walls of the cylinder using their forelimbs (~5 min/rat). One trial was conducted during each testing period.
Tapered Beam Test
The beam test was used to assess both forelimb and hindlimb placement.23,24 Rats were trained to cross a 160 cm long elevated/tapered, 2-level beam (6 cm at the wide end; 1.5 cm at the narrow end) to gain access to a dark goal box containing food pellets. Training consisted of gradually placing the rats at increasing distances from the goal box until the animals crossed the beam without any pauses. Baseline performance was assessed on the day following training. Following stroke, animals step or slip off the top level onto the wider lower beam level with increased frequency. 24 Trials were video recorded and the number of slips with both the left and right fore- and hindlimbs were assessed from videos. During each test-point, animals were required to cross the beam four times and data from all trials was averaged. The percentage of successful steps for each paw was calculated as 16 :
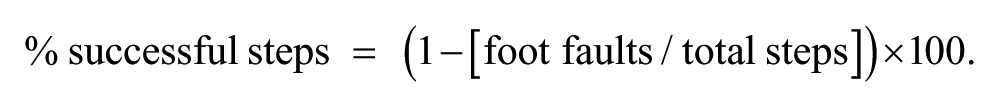
Histology and Infarct Measurement
Following completion of behavioral testing at 10 weeks poststroke, rats were deeply anesthetized with euthanyl (intraperitoneally 149.5 mg/kg; Bimeda-MTC Health Inc, Cambridge, Ontario, Canada) and transcardially perfused with cold heparinized saline followed by cold 4% paraformaldehyde (PFA) in phosphate buffered saline. Brains were extracted, postfixed in 4% PFA overnight at 4°C and then transferred to a solution of 20% sucrose until saturated. Brains were frozen in isopentane and cryo-sectioned at 20 μm (1:24 intervals). Coronal sections were stained with cresyl violet.
For each animal, a minimum of 8 evenly spaced sections, spanning the lesion were used for estimating infarct volume as previously described.18,25 Cresyl violet stained sections were imaged at 5× using a Leica DMRXE microscope (Leica Microsystems, Concord, Ontario, Canada). In each section, the area of intact tissue in the striatum and cortex of both hemispheres (ipsilesional and contralesional) was manually traced and measured using ImageJ. The total volume of injury (in mm3) was calculated for the cortex and striatum separately as follows 18 :

Statistical Analysis
All analyses were performed using SPSS (IBM Corp, Armonk, NY, USA). Behavioral data were analyzed using 1-way and repeated-measures analysis of variance (ANOVA) as appropriate. The length of time that animals remained significantly impaired was analyzed using simple contrast comparison at each poststroke test point using prestroke performance as control. Recovery on the tasks for each group was calculated as the difference in performance between the first (week 1) and the last poststroke time points (week 8). Infarct volumes were analyzed using a 1-way ANOVA with Ryan-Einot-Gabriel-Welsch (REGW-F) post hoc. Significance was set at
Results
Infarct Volumes
One rat in the combined stroke group died during surgery. A total of 3 animals were excluded; 2 animals in the striatal group had large nontargeted cortical injuries (~25 mm3 and 47 mm3) and were identified as being extreme outliers (cortical infarct volumes that were 3× the interquartile range). In addition, 1 animal from the cortical group was excluded due to having a small infarct (~3 mm3) and failing to show any behavioral impairments following stroke surgery. The remaining animals constituted n = 9 cortical, n = 8 striatal, and n = 9 combined.
Figure 2A depicts the typical injury profile seen in each of the 3 groups. There was a significant difference in total hemispheric damage between the groups (
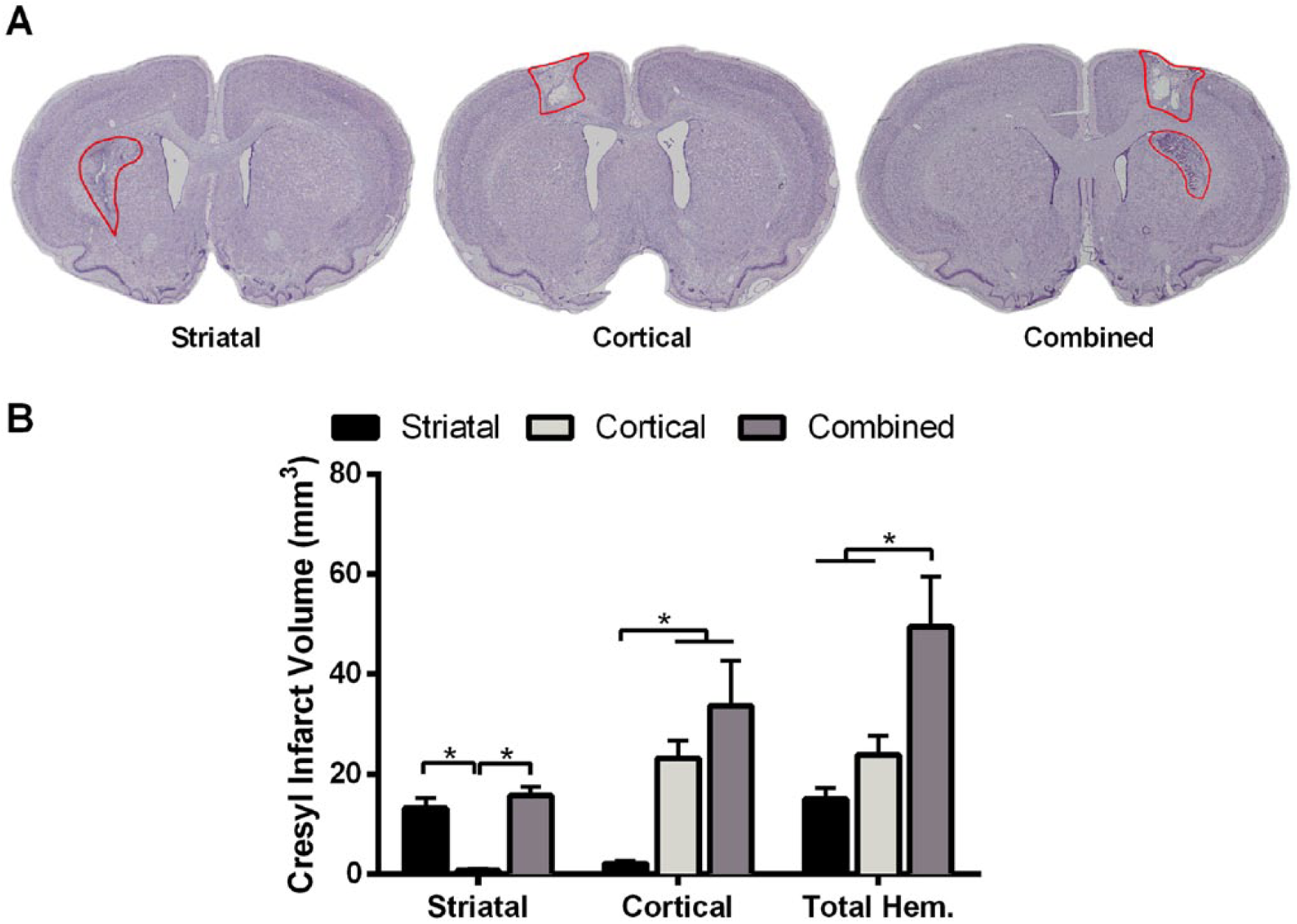
Infarct volumes. (A) Representative cresyl violet stained sections showing typical stroke lesions in the three groups. (B) Quantification of infarct volumes from cresyl violet–stained sections (10 weeks poststroke). The combined group had the largest total hemispheric damage (
Infarct volumes calculated from T2-weighted MRI images obtained 72 hours poststroke were significantly larger than those measured from cresyl violet–stained sections at 10 weeks poststroke (
Staircase Test
All groups reached a similar number of pellets (17.1 ± 0.4 out of 21 pellets) with their dominant forelimb prior to stroke (Figure 3A). Following ET-1 stroke, all three groups demonstrated a significant reduction in the average number of PR with the contralateral forelimb (PR: 14.2 ± 1.0 for striatal, 12.4 ± 0.9 for cortical, and 8.8 ± 1.0 for the combined group;
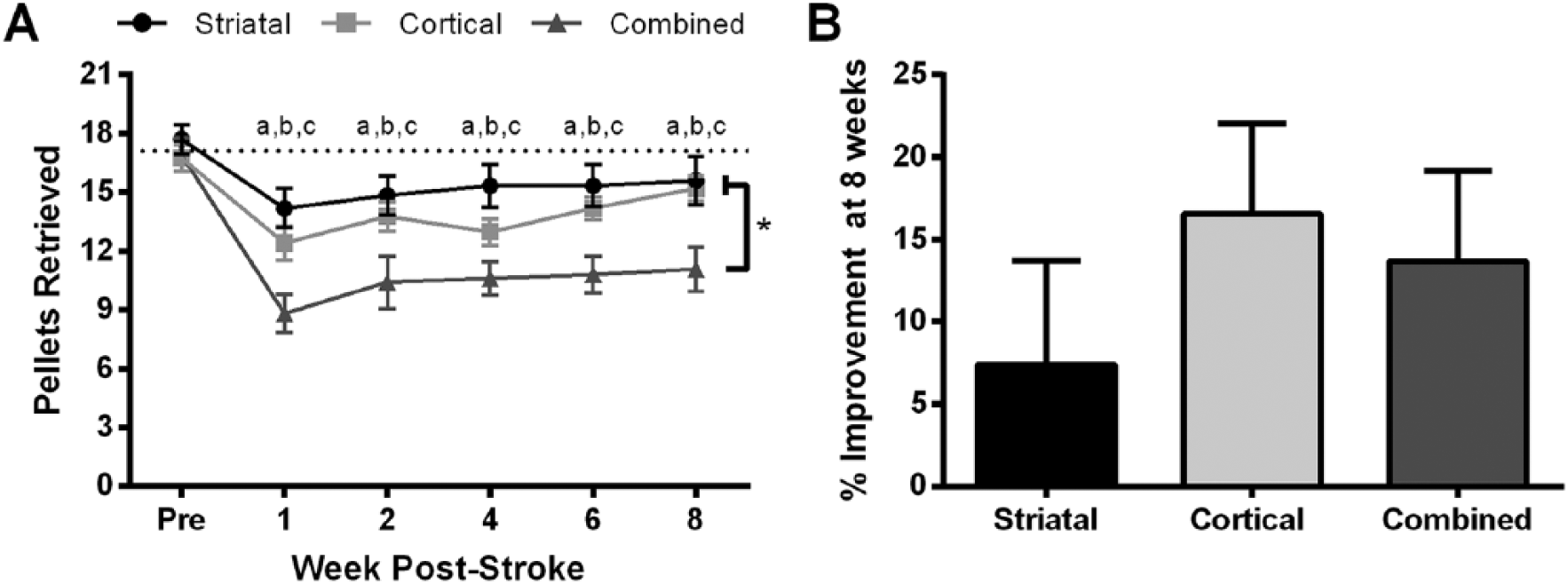
Staircase test. (A) Animals in all 3 groups retrieved fewer pellets with their contralateral forelimb and remained significantly impaired for the entire 8-week period (
Cylinder Test
Prior to stroke, animals tended to use both forelimbs equally for postural support on the cylinder task, and no differences existed between groups (Figure 4A). Following surgery, rats in all 3 groups significantly reduced their use of the contralateral forelimb from ~50% to 28.4% ± 3.8% for the striatal, 21.3% ± 3.9% for the cortical, and 21.7% ± 3.1% for the combined groups (
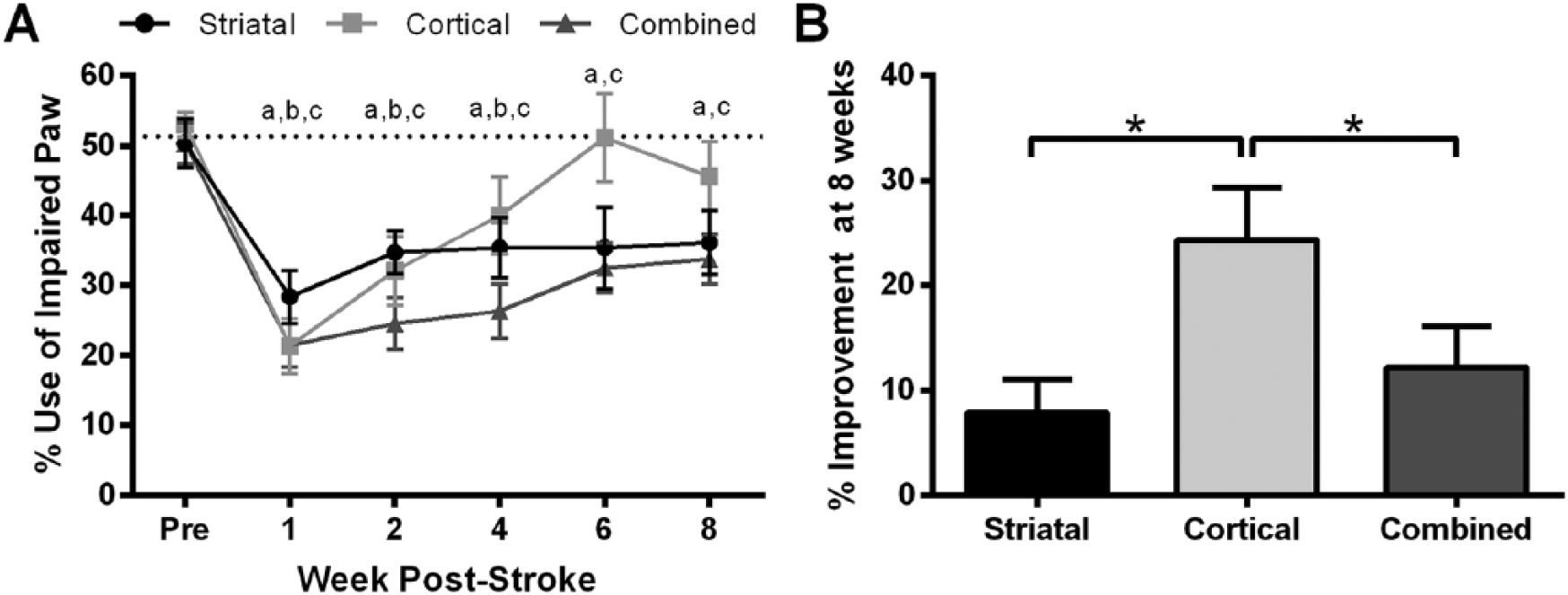
Cylinder test. (A) All groups displayed reduced reliance on their impaired forepaw after stroke. Both the striatal-only and combined groups were significantly impaired relative to prestroke for the entire 8-week testing period (
Beam Test
During baseline assessment, animals in all groups performed equally well and made few forelimb and hindlimb errors (~94% and ~96% successful steps, respectively; Figure 5A and C).
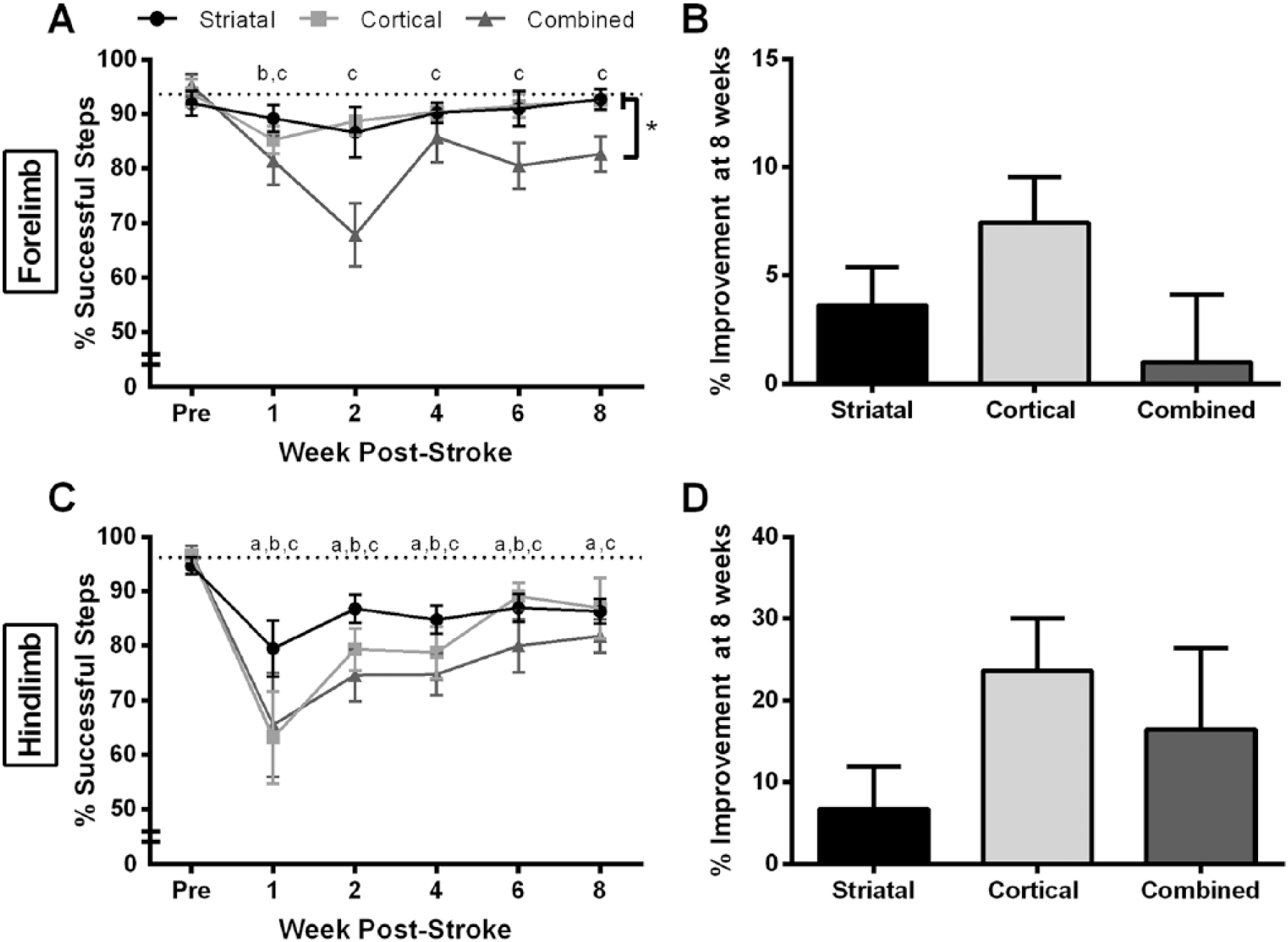
Beam-traversal test. (A) The cortical group had significant contralateral forelimb impairments at week 1 poststroke while the combined group remained significantly impaired at 8 weeks poststroke (
Forelimb Performance
Following stroke, the cortical and combined group made significantly more contralateral forelimb foot faults (
Hindlimb Performance
All 3 groups made significantly more hindlimb foot faults following stroke (
Overall, the combined stroke group showed the greatest motor deficits. Their impairments (staircase, cylinder, and beam) persisted for the entire 8-week testing period (Table 1, Figures 3-5) while the adhesive tape test showed only transient impairment (Table 1, Supplementary Figure 1). The striatal group had less severe deficits (Figures 3-5) than the combined group however the deficits remained evident at 8 weeks on the staircase, beam (hindlimb) and cylinder tests while there were no detectable deficits on the tape or beam (forelimb) tests (Table 1). The cortical group showed deficits of comparable magnitude to the striatal group (Figures 3-5) but the spontaneous recovery was much more variable depending on the test used (Table 1).
Duration of Motor Deficits. a
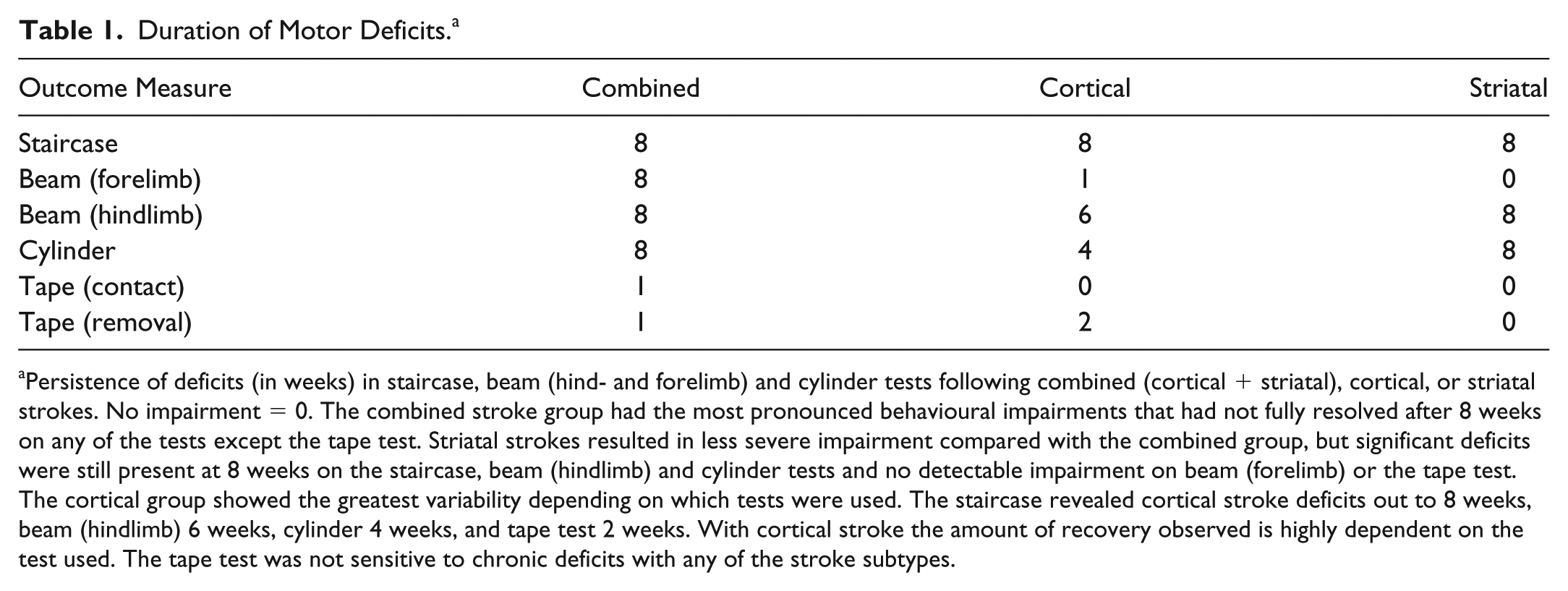
Persistence of deficits (in weeks) in staircase, beam (hind- and forelimb) and cylinder tests following combined (cortical + striatal), cortical, or striatal strokes. No impairment = 0. The combined stroke group had the most pronounced behavioural impairments that had not fully resolved after 8 weeks on any of the tests except the tape test. Striatal strokes resulted in less severe impairment compared with the combined group, but significant deficits were still present at 8 weeks on the staircase, beam (hindlimb) and cylinder tests and no detectable impairment on beam (forelimb) or the tape test. The cortical group showed the greatest variability depending on which tests were used. The staircase revealed cortical stroke deficits out to 8 weeks, beam (hindlimb) 6 weeks, cylinder 4 weeks, and tape test 2 weeks. With cortical stroke the amount of recovery observed is highly dependent on the test used. The tape test was not sensitive to chronic deficits with any of the stroke subtypes.
Discussion
The present study examined how the location of injury affects spontaneous motor recovery up to 2 months after stroke in the rat. The results show that lesion location differentially affects functional outcome following stroke. ET-1-induced stroke of the FMC and/or DLS resulted in significant impairments on the staircase, beam (hindlimb only for striatal group), and cylinder tests over the entire 8 week poststroke recovery period. Cortical strokes resulted in more variable deficits, with persistent impairments observed on the staircase test (up to 8 weeks poststroke), and less persistent deficits on the beam task (6 weeks post-stroke in the hindlimb) and the cylinder test (4 weeks poststroke). Forelimb impairments on the beam test resolved very quickly (1 week poststroke) after either cortical or striatal stroke.
Interestingly, our data also demonstrate that recovery is not simply dependent on the lesion volume. The combined group had the largest infarct volumes and most severe behavioral impairments; however, both the cortical and striatal groups with much smaller infarcts and deficits showed equally persistent impairments as the combined group. As reported by Carmichael et al, 27 “human strokes are mostly small in size” with lesions ranging from 4.5% to 14% of the ipsilesional hemisphere. However, the majority of preclinical stroke studies have used models that result in the destruction of a much greater proportion of the ipsilesional hemisphere than is typically observed in the clinical population. The infarct volumes in our study correspond to ipsilesional hemispheric damage of 1.9% in the striatal group and range up to 6.1% in the combined group, which is congruent with most human stroke volumes. Interestingly, even in the case of our smallest strokes, the animals showed chronic motor impairments. Similarly, persistent impairments after relatively small strokes have previously been reported by Jones et al. 28
The dorsolateral region of the striatum is critically involved in skilled motor control, 29 which may explain the chronic behavioral deficits observed in the groups with striatal damage. These results suggest that striatal damage is an important predictor of poststroke motor recovery. Similarly, in the clinical setting patients with subcortical strokes show reduced upper limb motor recovery compared with those with injuries confined to the cortex.8,9 The better outcome in patients and animals with cortical strokes may be due to the redundancy of the cortical motor system that allow spared cortical networks to take over functions of the damaged site.8,30-33 Others have hypothesized that poor functional outcome following strokes isolated to the striatum may be due to dysfunctional cortical-striatal interactions that impede recovery. 9
Regardless of stroke location, animals in all groups made significantly more contralateral hindlimb foot faults following surgery. Although strokes targeted forelimb areas, the hindlimb representation area in both the striatum and the cortex lies in close proximity to the forelimb area.15,34 As a result, animals had strokes that extended into the hindlimb regions. As with the staircase and cylinder test, the striatal and combined group displayed limited recovery on the task and had significant impairments throughout the testing period. In comparison, the cortical group remained impaired only until week 6. This finding lends further support to the view that subcortical strokes result in reduced functional outcome compared to cortical strokes.8,9 Further research is needed to understand the mechanisms contributing to difference in spontaneous recovery between cortical and subcortical strokes.
In contrast to the other tests, we were unable to detect chronic deficits using the adhesive removal task (Supplementary Figure 1). Other studies have reported long-term impairments (6 months) following stroke using this test.35,36 This may be due to the much larger infarcts (2-3 times compared with our combined group) produced by the intraluminal suture, middle cerebral artery occlusion model used in these studies.
Damage to white matter tracts is known to contribute to functional impairment and influence the degree of recovery. Approximately one-third of animals (9/26) had incidental damage to the corpus callosum that was equally distributed across the 3 stroke groups. Interestingly, there was no significant relationship between white matter injury and impairment on any of the behavioral tests (Supplementary Table 2). This suggests that it is the location of the gray matter lesion (ie, cortex/striatum) and not white matter damage was the defining characteristic of impairments in the present study.
Our results highlight the importance of using multiple behavioral tests to assess different functions following stroke in both cortical and subcortical brain regions as commonly occurs in human stroke.4-7 Most preclinical studies target the forelimb motor cortex and use only 1 or 2 tests to evaluate functional outcome.37,38 Here, we demonstrate that the degree of recovery shown on one test may not be similar to that of another test. For example, if we had only used the adhesive tape test or the cylinder test we would not have detected chronic reaching impairments that the cortical group exhibited in the staircase test.
Overall, the pattern of recovery observed on the different behavioral tasks in this study was similar to the nonlinear, logarithmic curve described in humans, albeit within a shorter time frame. 39 Most of the spontaneous recovery appeared to take place between poststroke weeks 1 and 2 whereas in humans the rapid changes are within the first month poststroke.40,41 The greatest amount of recovery on all behavioral tests occurred in the cortical group. Resolution of edema and diaschisis may be a likely mechanism that contributes to recovery during this early phase. After approximately 4 to 6 weeks following stroke, recovery on most of the behavioral tests reached a plateau. There is compelling preclinical evidence for the existence of a critical period of increased neuroplasticity in the days to weeks following stroke.12,19 The plateau observed in the behavioral tests in our study may reflect the closure of this critical period.
Stroke is a heterogeneous condition; therefore a “one-size-fits-all” approach to stroke rehabilitation might not be the most effective. Recently, there has been growing interest in identifying biomarkers to predict stroke recovery and optimize current rehabilitation approaches. Several different biomarkers have been investigated, including the use of neuroanatomical (eg, lesion size), physiological (eg, motor evoked potential), and behavioral measures (eg, initial level of impairment on Fugl-Meyer assessment) to predict recovery. 42 Our findings lend further support to the use of infarct location as a biomarker of functional outcome following stroke. Ongoing research in our lab is seeking to identify biomarkers of spontaneous and rehabilitation-induced recovery using different stroke models. 43
One limitation of the present study is that we cannot differentiate true spontaneous recovery from improvement due to behavioral compensation. It is possible that some of the behavioral improvement we observed may have resulted from animals using new adaptive strategies to perform a task in a different manner than they did prior to stroke. In a recent study, we used kinematic analysis in rats with injury similar to the combined group (ie, cortical + striatal) in this study and found evidence for poststroke compensatory movement patterns similar to those exhibited by stroke patients. 44 Compensation may also exist in other tasks such as the beam and cylinder tests where animals may be making subtle changes in posture and forelimb movements. It remains to be seen how compensatory patterns vary with lesion location or in response to recovery-promoting interventions. In addition, previous studies using aged stroke models have demonstrated that age is another critical factor that influences the degree of functional recovery.45,46 Thus it is possible there would be less spontaneous recovery in an older cohort of rats compared to the young rats used herein.
Conclusion
Our findings demonstrate that functional outcome is dependent not only on lesion volume, but also on lesion location and the behavioral test employed. A major goal in advancing stroke recovery research is to better align preclinical and clinical approaches.37,47 The present results illustrate the importance of incorporating the heterogeneity of human stroke injury location into preclinical animal models. Based on the spontaneous recovery patterns observed in this study, interventions targeting stroke recovery may appear quite effective after cortical stroke but less so with subcortical or combined subcortical and cortical stroke. Our results highlight key differences between cortical and subcortical strokes, suggesting that different types of stroke likely require individualized strategies to achieve more complete recovery.
Supplemental Material
Supplementary_Materials – Supplemental material for Characterizing Spontaneous Motor Recovery Following Cortical and Subcortical Stroke in the Rat
Supplemental material, Supplementary_Materials for Characterizing Spontaneous Motor Recovery Following Cortical and Subcortical Stroke in the Rat by Sudhir Karthikeyan, Matthew Strider Jeffers, Anthony Carter and Dale Corbett in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The University of Ottawa Preclinical Imaging Core and Dr Greg Cron were instrumental for conducting MRI imaging. The authors thank Dr Matthew McDonald for his helpful comments on an earlier version of this article. The data that support the findings of this study are available from the corresponding author on request.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant to DC from the Heart and Stroke Foundation and the Canadian Partnership for Stroke Recovery. SK received fellowships from Canadian Institutes of Health Research (CIHR) and the Ontario Graduate Scholarship (OGS) program.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
