Abstract
Background. Lateropulsion, a postural control disorder, delays recovery following hemispheric stroke. The number of stroke impairments may lead to differential recovery rates, depending on the intact systems available for recovery from lateropulsion. Objective. To study the impact of key postural control deficits on lateropulsion rate of recovery following stroke. Methods. Through retrospective analysis: 169 patients with hemispheric stroke in an in-patient rehabilitation facility were divided into 3 groups: (1) motor deficits only; (2) motor and hemianopic or visual–spatial deficits or motor and proprioceptive deficits; and (3) motor, proprioceptive, and hemianopic or visual–spatial deficits. Kaplan–Meier survival analysis determined if time to recovery from lateropulsion (achieving a score of 0 or 1 on the Burke Lateropulsion Scale) differed by group. Results. Log rank tests showed that time to recovery from lateropulsion differed based on the number of deficits (group, P = .012). Post hoc analyses by lesion side showed that group differences only occurred in right brain lesion (P < .05) as compared with left brain lesions (P = .34). Patients recovered from lateropulsion during in-patient rehabilitation if they had only motor deficits; those with all 3 postural control deficits showed the most protracted recovery. Conclusions. Rate of recovery from lateropulsion after stroke is dependent on the side of lesion, and number of key motor, proprioceptive, and/or hemianopic or visual–spatial deficits. The more postural control systems affected, the slower the recovery. Our data identify patients likely to need protracted rehabilitation targeting key postural control deficits.
Introduction
Lateropulsion following hemispheric stroke, defined as a postural bias toward the weak side1,2 accompanied by resistance to postural correction back to a vertical upright position, is also referred to as “pusher syndrome.” 1 Some view the resistance to postural correction as a pusher behavior, which is distinct from postural bias, 3 but for the purposes of this article we will use the terms “lateropulsion” and “pusher syndrome” interchangeably. Lateropulsion can compromise sitting and standing, and in more severe cases affect rolling, transferring, and walking. As a consequence, the behavior presents a significant barrier to rehabilitation because it delays training the functional activities needed for return home.4,5
The balance mechanism of humans involves a fine interplay of motor, vestibular, cerebellar, proprioceptive, and visual systems. 2 Pérennou et al 3 attributed lateropulsion and pusher behavior to impaired perception of an internal model of postural verticality,3,6 especially in cases of right hemispheric stroke. 3 They have observed that perception of haptic vertical and visual vertical are also impaired, yielding a transmodal model for the perception of verticality.3,6,7 Aside from their disrupted perception of verticality, patients with lateropulsion from hemispheric stroke may have primary visual or visual perceptual problems, impaired proprioception, and motor impairments, which leave them less equipped to relearn posture and balance.
This study examined whether recovery from lateropulsion was influenced by the patients’ positions along the impairment spectrum. We elected to examine the impairment count, as opposed to lesion size or severity because impairments directly relate to treatment planning and goal setting during rehabilitation. Such analyses may address mechanisms of recovery and guide selection of rehabilitation interventions. The purpose of this study was to clarify if recovery from lateropulsion differed for patients with 1, 2, or 3 major deficits associated with hemispheric stroke. A prior study by Abe et al 8 used a survival analysis strategy to compare patients’ recovery from lateropulsion based on lesion side. We incorporated this strategy to estimate courses of recovery of groups of patients with lateropulsion according to the number of impairments. Recovery from lateropulsion can occur with an extended rehabilitation hospitalization, which tends to exceed that of patients with hemispheric stroke without lateropulsion.4,5 We used the Burke Lateropulsion Scale (BLS) to define the endpoint for the survival analysis. The BLS provides a numerical rating of the degree of lateropulsion in supine rolling, sitting, transferring, standing, and walking. 9 A BLS score of 2 or greater indicates that a person has lateropulsion. The BLS has strong clinimetric properties in comparison with other tools that help describe the degree of lateropulsion.9-11 We operationally defined time to recovery from lateropulsion as the number of days needed to achieve a BLS score of 0 or 1 prior to discharge from in-patient rehabilitation. The null hypothesis was that groups would not differ in time to recovery from lateropulsion. The alternative hypothesis was that the greater number of deficits would result in fewer subjects achieving target BLS scores prior to discharge. We based our hypothesis of group-wise differences in recovery on the contention that more impairments leave fewer reserve systems for relearning posture and balance that are needed for the recovery from lateropulsion. Lesion location may influence findings 8 because of hemispheric specialization of function; therefore, separate post hoc analyses were conducted for patients with left and right brain lesions. Knowledge about the link between recovery from lateropulsion as an impairment of vertical perception and the number of other stroke impairments would facilitate intervention selection and discharge planning.
Methods
Subjects
This retrospective chart analysis included patients admitted to in-patient rehabilitation between May 2005 and December 2009 for hemispheric stroke who had BLS scores of 2 or greater at admission or at the first weekly assessment by physical therapists. Time since onset of stroke was less than or equal to 36 days. Patients with bilateral or brainstem or cerebellar stroke and subsequent admissions for stroke were excluded. The Burke Rehabilitation Hospital and Hunter College Institutional Review Boards approved this project.
Data Analysis
Data were extracted from the electronic medical record system into an Excel spreadsheet, checked for accuracy, and transferred to an SPSS (version 19, IBM) database. Using physician and therapist admission reports, patients with lateropulsion were stratified into 3 groups: (1) only motor impairments; (2) motor plus proprioceptive or motor plus hemianopic or visual–spatial impairments; and (3) motor, proprioceptive, and hemianopic or visual-spatial impairments. Motor impairments were determined by Fugl-Meyer Assessment for Stroke motor scores of less than 34 for the lower extremity or less than 66 for the upper extremity as reported by physical and occupational therapists, respectively. Proprioceptive impairments were determined using a limb placement test error of 6 inches or greater as recorded at the bedside by the admitting physician. The limb placement test was assessed by the examiner holding the affected index finger in all 4 spatial quadrants away from the patient’s body but within reach of the other hand. Allowing for multiple attempts, the largest consistent error touching the affected index finger with the unaffected index finger was estimated in inches with a maximum error recorded as ≥12 inches.12,13 The limb placement test was selected because it represents a functional measure of proprioception that can often be understood by globally aphasic patients with gestural cueing or hand-over-hand demonstration by the examiner. The physician evaluated visual field impairments by confrontation testing, scoring results as normal, or indicating a left or right hemianopic visual deficit. Hemispatial visual neglect, quadrantanopsia and hemianopsia were scored as one deficit as they are often difficult to differentiate during the early recovery phase following stroke, and each would be expected to affect function.
Patients’ discharge BLS scores of 0 or 1 defined recovery and constituted the exit point for time to recovery in the Kaplan–Meier survival analysis. Log rank (Mantel–Cox) tests examined the equality of the time to recovery distributions for the groups. This test compares observed cases for each group with the expected cases for the combined groups. Post hoc tests examined group differences based on side of lesion. Alpha levels were set at .05.
Results
Of the 200 patients with lateropulsion, 169 were eligible for the analysis with complete data to classify them into groups. Some patients failed to achieve the outcome by the time of discharge, therefore, their cases were considered “censored” within the time to recovery analysis (+ on the Figures). Table 1 presents demographic data (n = 169) by group and lesion side. One-way analysis of variance confirmed that admission BLS scores for those excluded from the survival analysis (n = 31) due to incomplete data were not statistically significantly different from that of people classified into groups, F(3, 32) = 1.542, P = .212.
Descriptive Statistics for Groups by Side of Lesion.
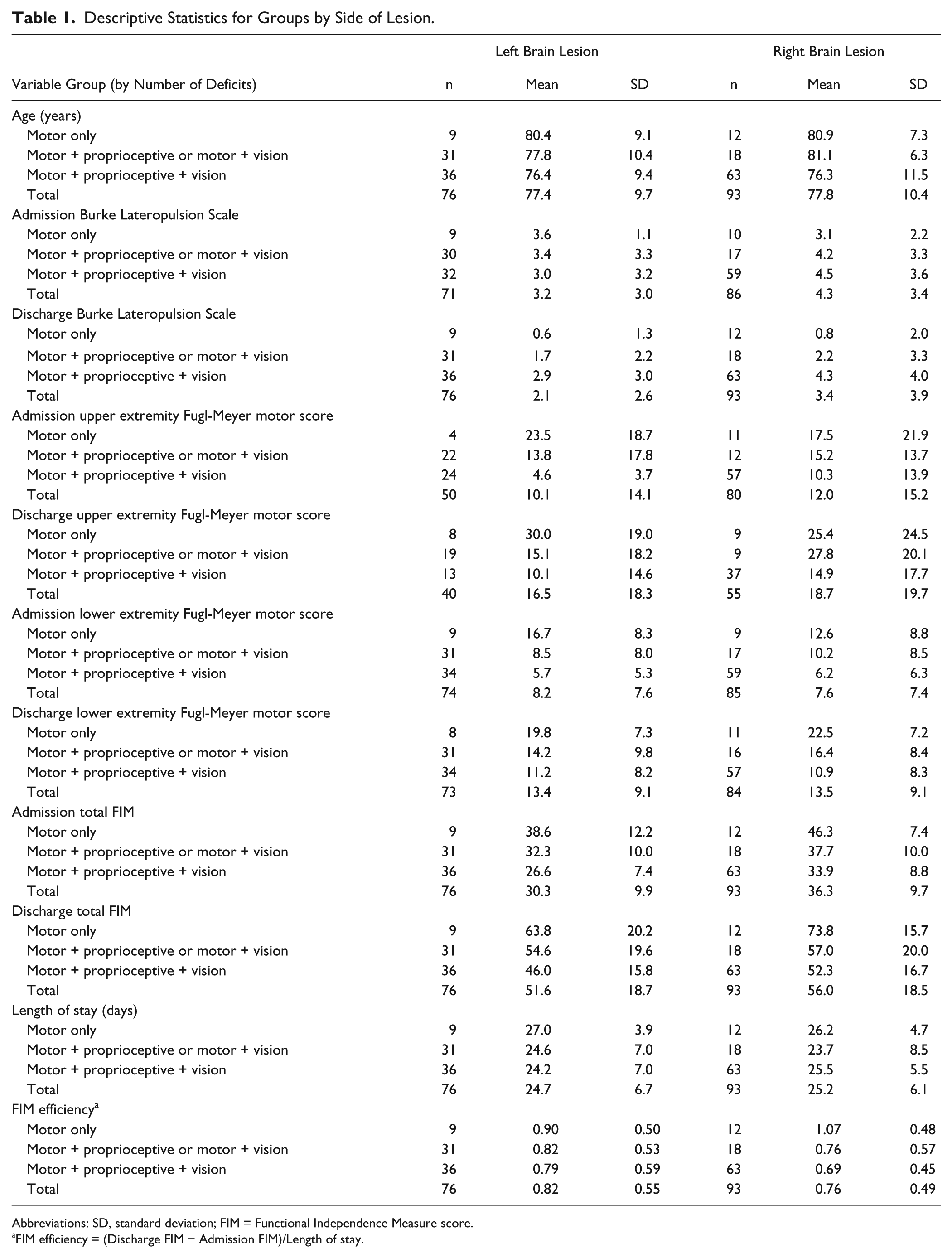
Abbreviations: SD, standard deviation; FIM = Functional Independence Measure score.
FIM efficiency = (Discharge FIM − Admission FIM)/Length of stay.
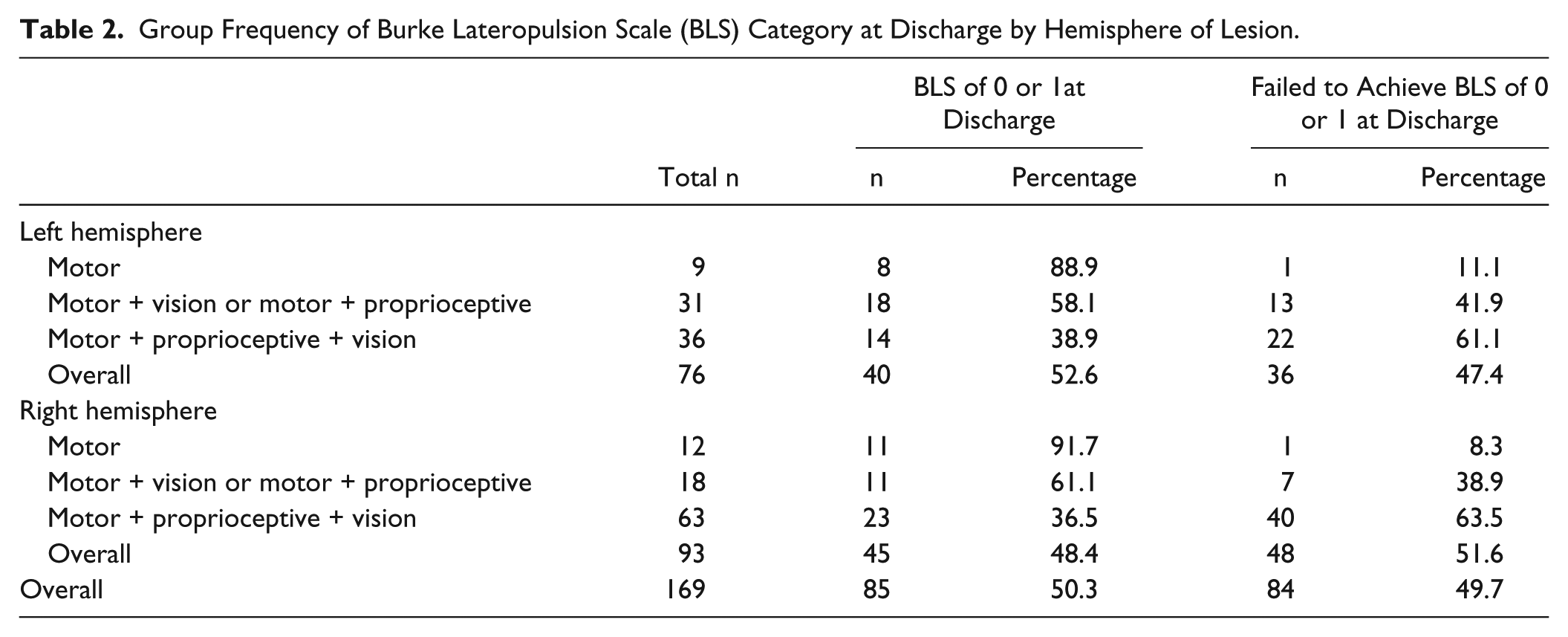
Log rank tests showed significant group differences in time to recovery from lateropulsion (χ2 = 8.783, P = .012). Figure 1 shows the percentage of patients with lateropulsion who have not achieved the target of a discharge BLS score of 0 or 1 as a function of length of stay for the respective groups. Post hoc analysis showed that groups had similar time to recovery curves with left brain lesion (Figure 2A) but differed in their recovery curves with right brain lesion (Figure 2B). Log rank tests confirmed that a significant group difference in time to recovery from lateropulsion occurred only for patients with right brain lesions (χ2 = 6.084, P = .048) as compared with left brain lesions (χ2 = 2.155, P = .340). Figure 3 shows how subjects within each group distributed relative to their achievement of the BLS score of 0 or 1 goal. Table 2 shows that approximately 47% of patients with left brain lesions and 52% of patients with right brain lesions failed to achieve a BLS score of 0 or 1 prior to discharge from in-patient rehabilitation. Patients with motor impairments alone had the best chance of recovery from lateropulsion during in-patient rehabilitation; patients with motor, proprioceptive, and visual impairments had the lowest probability of achieving the BLS scores of 0 or 1 prior to discharge.
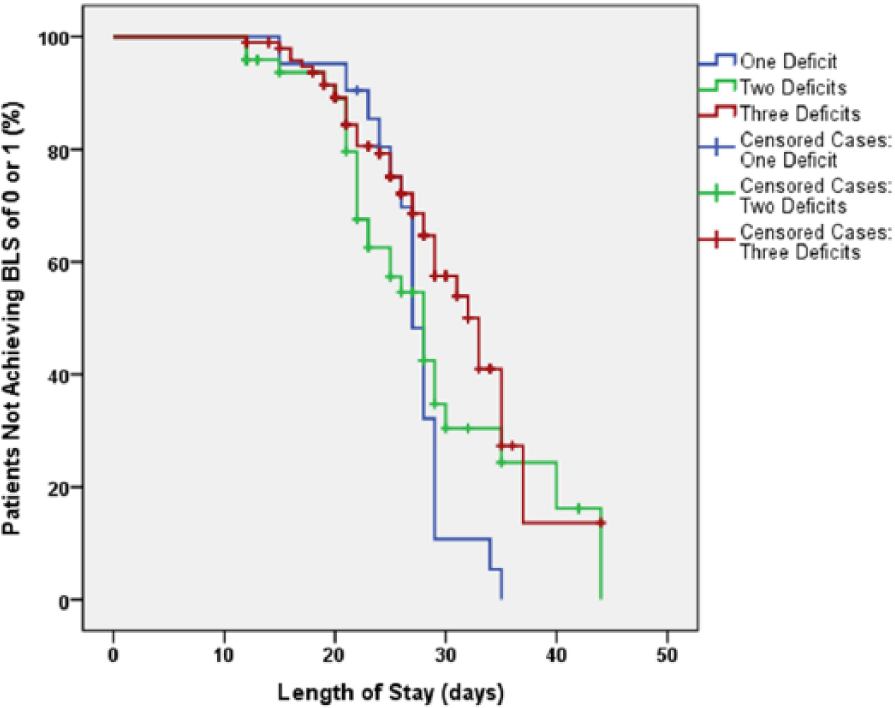
Time to achieve Burke Lateropulsion Scale (BLS) score of 0 or 1 based on number of stroke deficits. Censored cases (+) failed to achieve score of 0 or 1 by the time of discharge from in-patient rehabilitation.
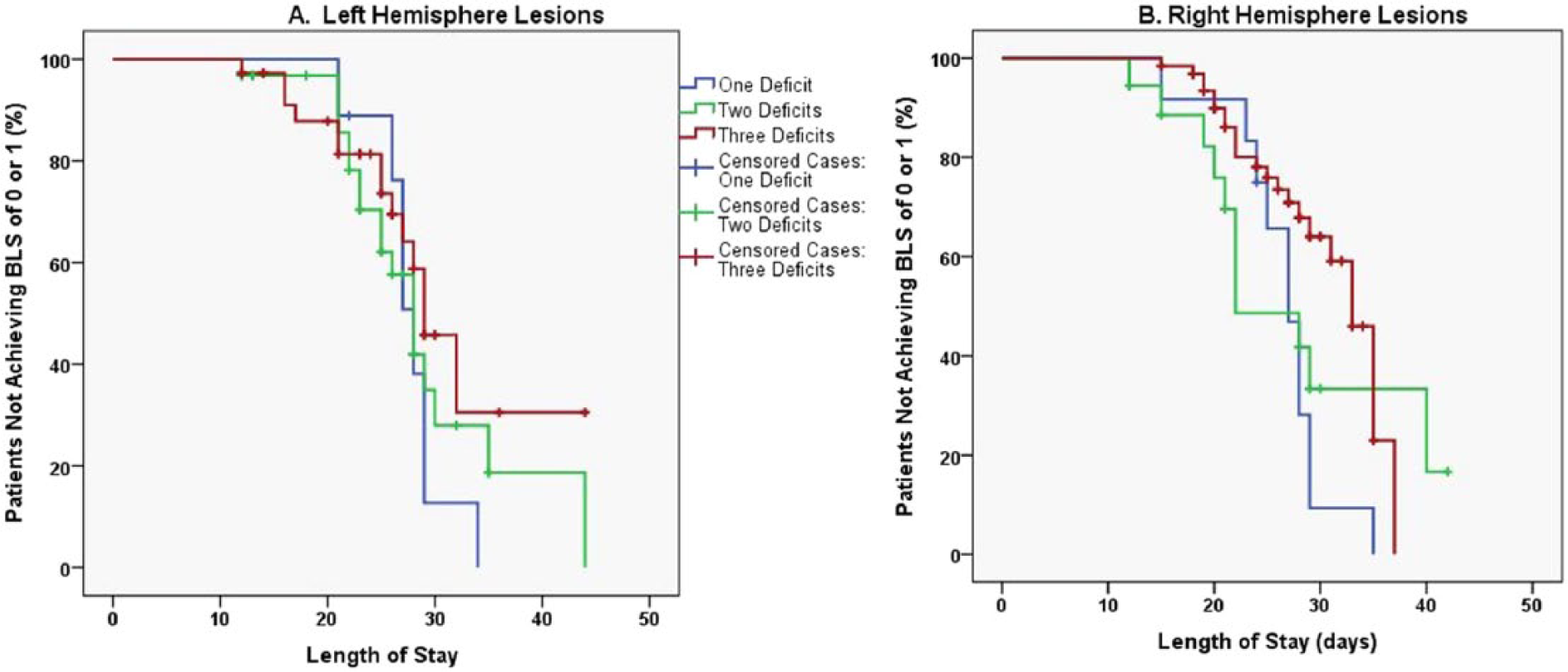
Time to achieve Burke Lateropulsion Scale (BLS) score of 0 or 1: (A) left hemisphere lesion and (B) right hemisphere lesion. Censored cases (+) failed to achieve score of 0 or 1 by the time of discharge from in-patient rehabilitation.
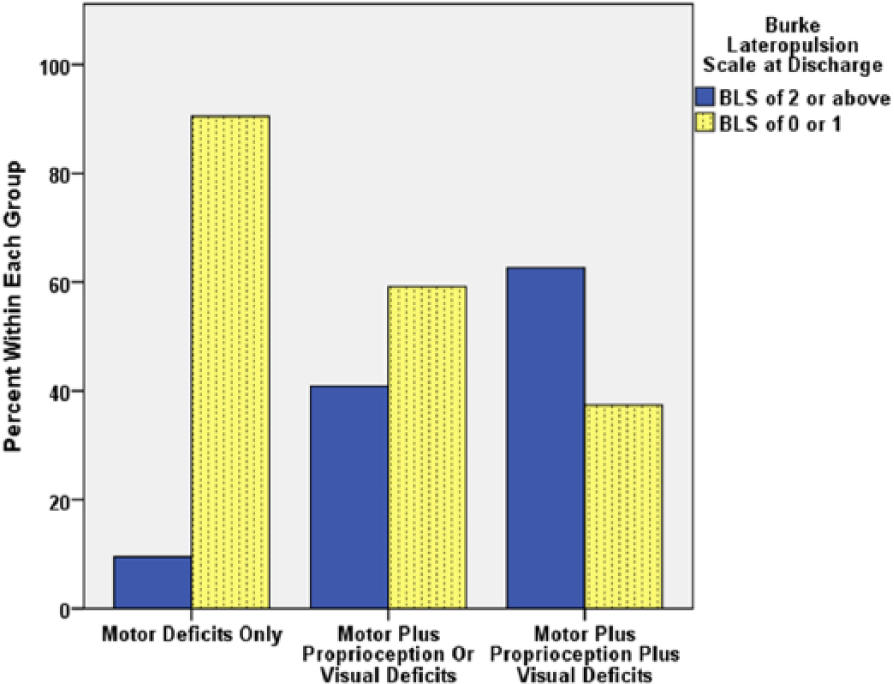
Frequency distribution (%) of subjects based on number of deficits and Burke Lateropulsion Scale (BLS) scores at discharge.
Group Frequency of Burke Lateropulsion Scale (BLS) Category at Discharge by Hemisphere of Lesion.
Discussion
As hypothesized, time to recovery from lateropulsion differed for patients according to the number of stroke impairments with which they presented at admission. Side of lesion made a difference in the time to recovery from lateropulsion but only patients with right brain damage showed a statistically significant difference in time to recovery among groups.
The findings of lesion side differences in time course of recovery from lateropulsion noted by Abe et al 8 appeared to hold true even as grouping by number of deficits was added to the paradigm. Other studies showed that right brain lesion often drives differences in recovery among patients with lateropulsion 14 and in comparisons to patients with stroke who do not have lateropulsion. 15
Even though patients with right brain damage had the clearest delineation of time course to recovery from lateropulsion based on number of deficits, Table 2 shows that like groups had similar percentages of patients who attained the target BLS score during their in-patient rehabilitation visit, regardless of lesion side. Most patients with lateropulsion who only had motor impairments (90.5%) achieved the target BLS score of 0 or 1 prior to discharge. On the other extreme, patients with motor, proprioceptive, and hemianopic or visual–spatial impairments achieved target BLS scores in roughly 37% of the cases. Patients with 2 deficits achieved the target in about 59% of cases.
Table 2 also shows that the most common combination of impairments in patients with lateropulsion is motor, proprioceptive, and visual deficits (59%). This higher percentage may necessitate caution in interpreting the time to recovery analyses. These individuals had statistically significantly lower motor Fugl-Meyer scores than the groups with 1 or 2 deficits for post hoc nonparametric tests. Of note, their initial BLS scores were not statistically significantly different among groups while their discharge BLS scores were. Our study was limited to the sample available over more than a 4-year period.
Other authors showed similar trends in the severity of stroke impairments for patients with lateropulsion relative to time to recovery.4,5 Pedersen et al 4 tracked patients with lateropulsion and found that patients with lateropulsion required 3.6 weeks longer than other patients with stroke to achieve the same degree of function. Danells et al 5 followed 65 patients with moderate to severe stroke from acute hospitalization through in-patient rehabilitation up to 3-months after stroke. Fifty-six percent of patients with lateropulsion had proprioceptive deficits and 29% had visual deficits at admission. Although they grouped patients with lateropulsion according to the time frame in which Scale of Contraversive Pushing Scores returned to 0/6, only motor scores were examined longitudinally for the groups; therefore, comparison with our results was not possible. Similarly, Abe et al 8 noted marked motor deficits for patients with lateropulsion and between 80% (left brain damage) and 91.3% (right brain damage) of patients with sensory deficits. Visual deficit frequency was not delineated in their study. 8 Perhaps their trend for patients with left brain lesions to experience more rapid recovery from lateropulsion when compared with those with right brain lesions was related to the relative proportions of sensory deficits but their study did not specifically address this point. Other studies relating to the time course of recovery had smaller samples.16,17 The present study gives a new perspective into the need to address patients at the impairment level, including the lateropulsion itself, in order to facilitate functional recovery, especially in cases of right brain lesion.
Prior studies indicate that motor recovery rates and the rate of recovery from lateropulsion are not codependent.5,15 Likewise, primary sensory and vestibular issues do not appear to create lateropulsion.18,19 Therefore, we cannot infer a cause–effect relationship between specific stroke impairments and lateropulsion. These results may point to the need for intact or less impaired motor, sensory, and/or visual systems for the most efficient learning processes needed in regaining postural control.
Our data support the value of specialized inpatient stroke rehabilitation even for patients with lateropulsion due to multimodal postural control deficits. Significant improvements were made by these patients in lateropulsion, Fugl-Meyer, and Functional Independence Measure scores during their in-patient rehabilitation stay (Table 1). We consider lateropulsion to be an impairment in vertical perception. Our results may help practitioners predict the course of recovery from lateropulsion based on lesion side and the number of additional stroke impairments to more effectively focus intervention strategies and to estimate length of stay and discharge destination. For example, patients with lateropulsion who have only motor symptoms can be expected to recover from lateropulsion prior to discharge. Relearning proper postural alignment appears to be influenced to some degree by available proprioceptive and visual systems. Whereas those with motor, proprioceptive, and visual impairments would need more protracted in-patient rehabilitation before they relearn how to maintain vertical postural positions. These patients appear to have the least neurological resources available to aid relearning of the vertical upright position. Patients with left brain lesions appear to have a lesser impact of the number of impairments during recovery from lateropulsion. The presence of right brain lesions augments the differential recovery rates from lateropulsion based on number of impairments. On a positive note, lateropulsion resolves with prolonged experience in the vertical upright position. The question arises about how to stimulate right brain centers for postural control especially when 2 or more motor, sensory or visual impairments coexist with lateropulsion. Pilot work suggested that supplementing the sensory 20 or locomotor systems 21 may affect lateropulsion. Controlled studies are needed to discern if impairment-based interventions may augment traditional interventions that focus on posture and balance with visual feedback.16,17,22 The impact of the severity of motor, proprioceptive, and visual impairments on recovery should also be examined.
Conclusion
Time to recovery from lateropulsion after stroke is related to the number of key stroke deficits noted at admission to in-patient rehabilitation. Patients with only motor deficits can recover from lateropulsion with an inpatient rehabilitation stay averaging 27 days. Those with 2 or 3 additional key postural control deficits and right brain lesions will need more protracted rehabilitation.
Footnotes
Acknowledgements
We are grateful for the assistance of Cathy Dwyer and Janet Herbold for database assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
