Abstract
Background. Visual vertical (VV) measurement provides information about spatial cognition and is now part of postural disorders assessment. Guidelines for clinical VV measurement after stroke remain to be established, especially regarding the orientation settings for patients who do not sit upright. Objectives. We analyzed the need to control body orientation while patients estimate the VV. Methods. VV orientation and variability were assessed in 20 controls and 36 subacute patients undergoing rehabilitation after a first hemisphere stroke, in 3 settings: body not maintained (trunk and head free), partially maintained (trunk maintained, head free), or maintained (trunk and head). VV was analyzed as a function of trunk and head tilt, also quantified. Results. Trunk and head orientations were independent. The ability to sit independently was affected by a tilted trunk. The setting had a strong effect on VV orientation and variability in patients with contralesional trunk tilt (n = 11; trunk orientation −18.4 ± 11.7°). The contralesional VV bias was severe and consistent under partially maintained (−8.4 ± 5.2°) and maintained (−7.8 ± 3.5°) settings, whereas various individual behaviors reduced the mean bias under the nonmaintained setting (−3.6 ± 9.3°, P < .05). VV variability was lower under the maintained (1.5 ± 0.2°) than nonmaintained (3.7 ± 0.4°, P < .001) and partially maintained (3.6 ± 0.2°, P < .001) settings. In contrast, setting had no effect in patients with satisfactory postural control in sitting. Conclusion. Subject setting improves VV measurement in stroke patients with postural disorders. Maintaining the trunk upright enhances the validity of VV orientation, and maintaining the head upright enhances the validity of within-subject variability. Measuring VV without any body maintaining is valid in patients with satisfactory balance abilities.
Keywords
Introduction
The visual estimation of the vertical (visual vertical [VV]) is the most simple and widely used test to assess verticality perception in research and clinical practice. The recent evolution of knowledge of balance disorders after stroke 1 has progressively led to the use of VV as part of routine assessments.2-11 VV assessment is now included as a basic measurement of spatial cognition in addition to other tests12-15 and is used to better understand the specific part due to a biased internal model of verticality as a potential cause of balance disorders in brainstem2,16-18 or hemisphere stroke.6,19,20 Therefore, the measurement is useful to guide rehabilitation 1 and to follow recovery.5,20
However, this increasing use of VV for assessing spatial orientation after stroke is made without standardized protocol,2-11,19 and our knowledge of the clinimetric characteristics of this test remains limited in this context. Furthermore, major differences in methodological approach exist among studies. One of the most important differences in the assessment of stroke patients pertains to participant settings. In some studies, the positions of the trunk and head are free,3,5,8-10 whereas in other studies, the trunk and/or the head are maintained in a fixed upright position.2,4,6,7,11,20 This question is critical for stroke patients given (a) their frequent postural disorders, especially spontaneous lateral whole body tilts, which may compromise their ability to sit on their own; (b) the well-known impact of body tilt on verticality perception.21-24
In this study, we analyzed the interest of controlling body orientation while stroke patients estimate VV. We hypothesized that to achieve valid results, maintaining the trunk upright might be useful, especially in stroke patients showing postural disorders. We also aimed to determine whether additional head maintaining would be beneficial. We measured VV in controls and stroke patients with or without postural disabilities, in 3 different settings: nonmaintained body (trunk and head free), partially maintained body (trunk maintained and head free), and maintained body (trunk and head).
Methods
Participants
All participants gave their informed consent in accordance with the guidelines of the institutions’ ethics committees. Table 1 presents the characteristics of the 56 participants of the study: 20 healthy controls without disorders affecting the nervous system or the skeleton and 36 consecutive hemisphere stroke patients recruited from a neurorehabilitation unit. Clinical examination included collection of data on motor weakness, sensory loss, and spatial neglect. Motor weakness was assessed by a standardized examination of muscle strength adapted for patients with central neurological disorders. 25 As proposed by Pérennou et al, 26 the somatosensory threshold was assessed by an investigation of pressure sensitivity using a Semmes-Weinstein aesthesiometer. 27 Pressure sensitivity was tested both at the pulp of the big toe and in the area of the second metacarpophalangeal joint. Spatial neglect was assessed by using bell cancellation, 28 the ability to bisect two 200-mm length lines, 29 and the Catherine Bergego scale. 30 Patients were considered as having spatial neglect when performance was pathological in at least 2 of the 3 tests.
Characteristics of Controls and Patients With Independent Sitting Posture (ISP) and Without Independent Sitting Posture (Non-IS) a .
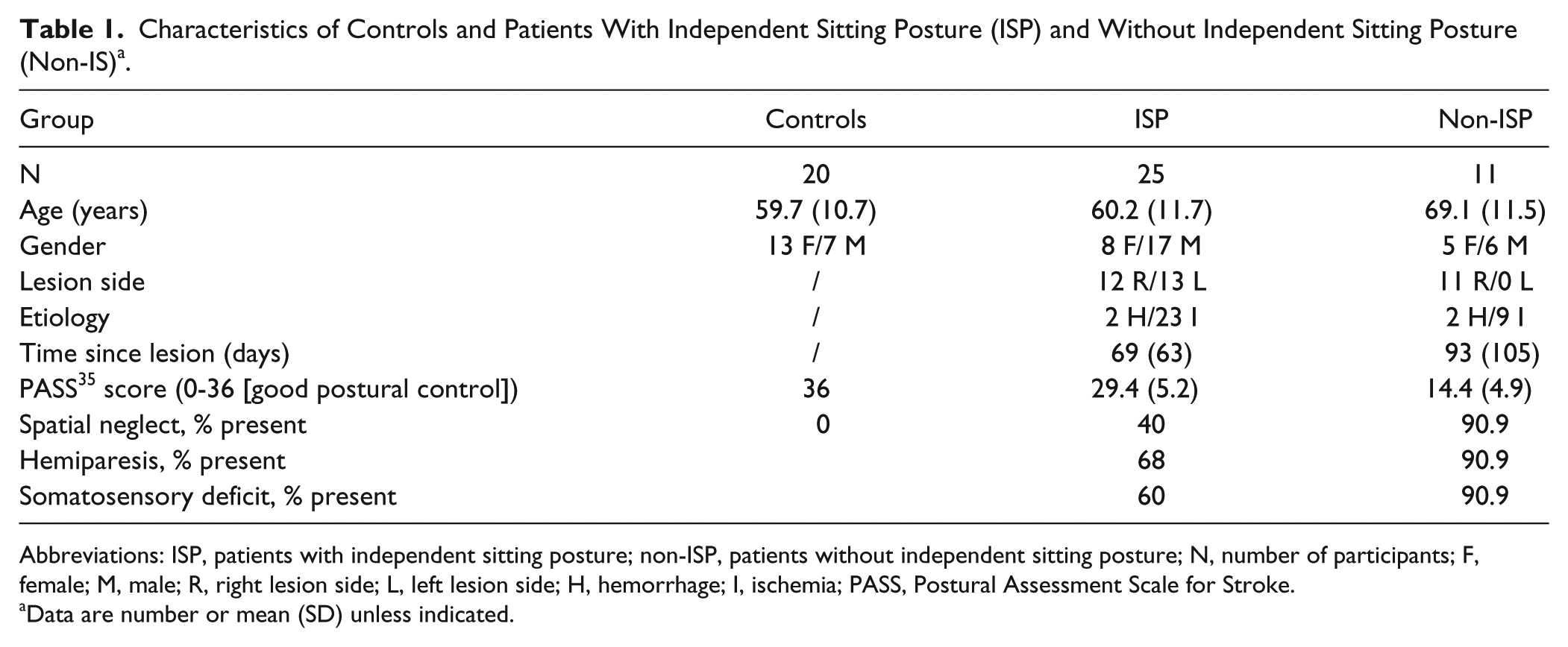
Abbreviations: ISP, patients with independent sitting posture; non-ISP, patients without independent sitting posture; N, number of participants; F, female; M, male; R, right lesion side; L, left lesion side; H, hemorrhage; I, ischemia; PASS, Postural Assessment Scale for Stroke.
Data are number or mean (SD) unless indicated.
We focused on patients with subacute stroke who had experienced a first and solitary recent (2.5 ± 2.6 months) hemorrhagic or ischemic hemisphere stroke (Figure 1). Patients with brainstem or cerebellar strokes involving vestibulo-ocular signs were not included because of a different mechanism linking abnormal VV perception and lateral postural disorders.1,6,16,17,32-34 Patients with neuropathy, psychiatric disorders, or major comprehension problems due to aphasia or dementia were not included, nor were those with unstable status.
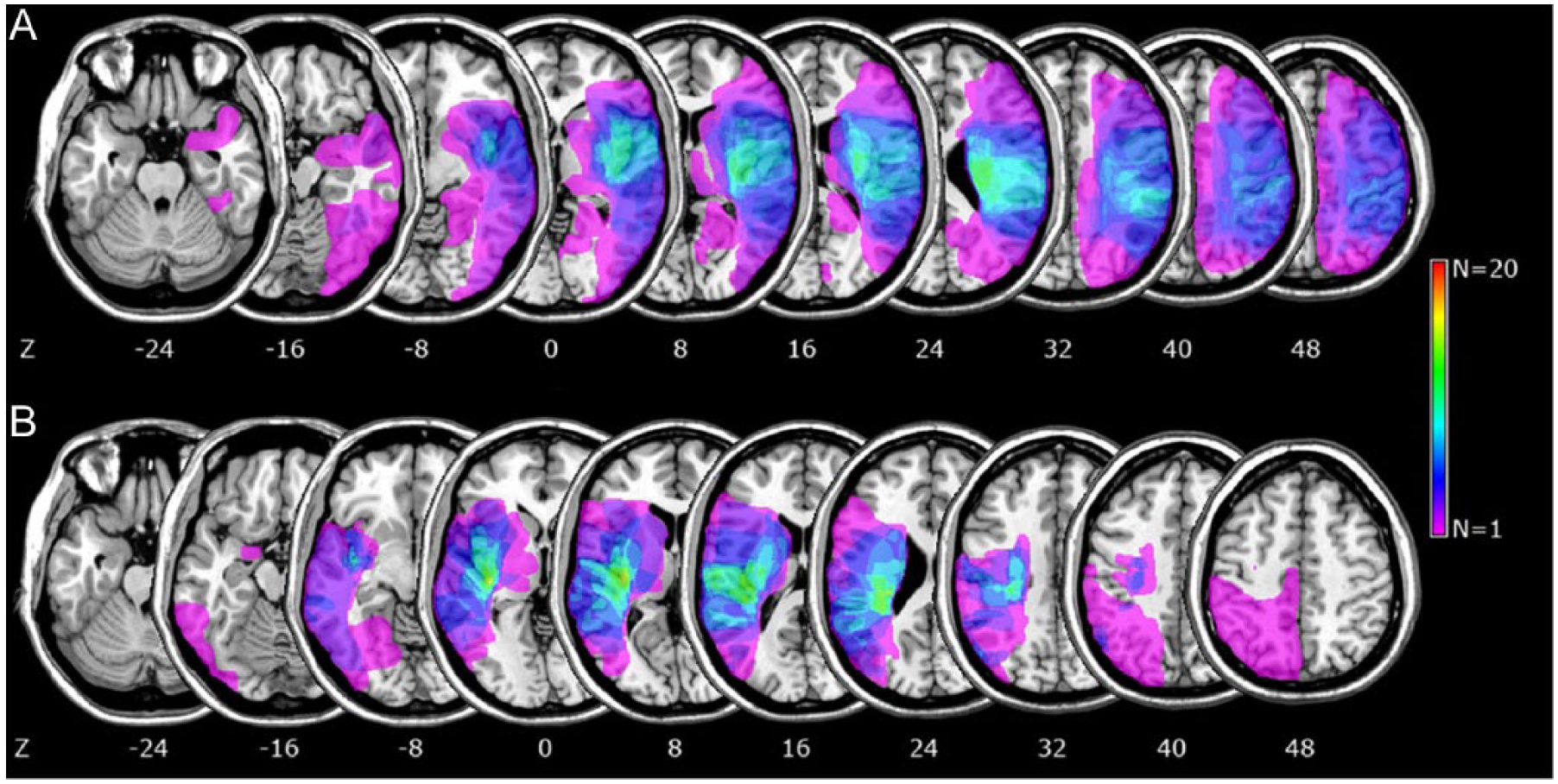
Overlapping lesion plots of the 23 right (A) and the 13 left (B) hemisphere strokes. The number of overlapping lesions is illustrated by different colors coding increasing frequencies from violet (n = 1) to red (n = 20). Talairach z-coordinates of each transverse slice are given. 31
Patients were classified into 2 groups according to their score for the “maintaining a sitting posture” item of the Postural Assessment Scale for Stroke 35 (PASS). A score of 3 defined an upright Independent Sitting Posture (ISP) classification and <3 a Non-Independent upright Sitting Posture (Non-ISP) classification. In total, 25 patients had a score of 3 for the PASS for an upright ISP classification, and 11 had a score <3 for a non-ISP classification. These latter patients were not able to maintain a sitting posture without support for 5 minutes (score = 2, n = 6) or at least 10 seconds (score = 1, n = 2) or required a support to sit (score = 0, n = 3). All these patients showed a net lateropulsion with an average Score for Contraversive Pushing (SCP) of 3.97 ± 1.4. 36
The control group and patient groups did not differ in age (P = .98 for ISP and P = .08 for non-ISP; Table 1). The 2 groups of patients did not differ in age (P = .09), time since stroke (P = .45), gender (χ2 = 0.60, P = .43), or stroke etiology (χ2 = 0.80, P = .37). They differed in lesion side (χ2 = 7.92, P = .004) and size, t(34) = 2.43, P < .05, with only right-sided and more extended lesions in non-ISP than ISP patients.
Verticality Perception
VV perception was assessed by subjects who were in complete darkness with 10 adjustments of the direction of a luminous line (15-cm long, 2-mm wide) presented on a computer screen, at eye level. The initial orientation of the luminous line was randomly determined to be 5° to 30° for each trial. The side of the initial tilt of the line was randomized for the 10 trials, but the order was kept constant across participants. Participants were asked to verbally adjust the line to the vertical. Participants closed their eyes between adjustments. There was no time limitation or feedback. For each setting (nonmaintained, partially maintained, and maintained body), measurements started after 2 minutes in darkness. Between 2 settings, participants’ eyes were open with the room lights on.
For each participant and each setting, VV orientation (mean) and VV variability (standard deviation) were calculated. By convention, after sign transformation depending on the lesion side, a negative value corresponded to a rotation relative to the true vertical toward the contralesional side for patients and toward the left for controls.
Procedure
VV was assessed in 3 settings. In one setting, participants were without constraints on a bench with the trunk and head free (nonmaintained body). Patients who were not able to actively maintain a sitting position (non-ISP) seated themselves in a comfortable posture. An armrest was available to prevent falling due to lateral postural disorders. In a second setting, the trunk was maintained upright by lateral devices,6,23 preventing lateral tilts or rotations while the head was free (partially maintained body). In a third setting, participants were seated upright with the trunk and head restrained and maintained upright (maintained body). The order of the 3 settings was pseudorandomized because the partially maintained and maintained settings were always successive to limit the manipulation of patients. Four orders resulted from this control: maintained, partially maintained, nonmaintained; partially maintained, maintained, nonmaintained; nonmaintained, maintained, partially maintained; and nonmaintained, partially maintained, maintained. Each participant was randomly assigned to 1 of the 4 possible orders. The experiment lasted about 75 minutes.
Spontaneous Trunk and Head Orientations
To precisely analyze the impact of body tilt on VV, the lateral body orientation of participants was measured in the nonmaintained and partially maintained settings (Figure 4). Trunk and head orientations were measured by use of an inclinometer (Pro 360 Digital Protractor) by a trained physiotherapist who was blinded to the study hypotheses and VV results. Trunk orientation in the roll plane was quantified by measuring the tilt of the axis colinear to the first thoracic vertebra and the fourth lumbar vertebra with respect to the vertical plane. Head orientation in the roll plane was quantified by measuring the tilt of the bipupillary line with respect to the horizontal plane. Measurements were performed before and after VV estimates and averaged to provide segmental body orientation in roll (spatial resolution 1°). By convention, a negative value corresponded to a leftward tilt in controls and a contralesional tilt in patients.
Statistical Analyses
The normality of data distribution was tested by the Kolmogorov–Smirnov test. Regarding trunk and head orientations, given that distributions were normal in each group (all P > .1), a 1-factor ANOVA was used for each setting (nonmaintained, partially maintained) with groups (controls, ISP, non-ISP) as between factors. Trunk and head orientations were analyzed by a 1-sample t test (against verticality [0°]). The predictive values of trunk tilt on the capacity to maintain an independent sitting posture were analyzed using a Fisher exact test. Because the distributions of these data were Gaussian, the range of normality of trunk orientation was defined as mean ± 2 SD in controls ± 1° of measurement accuracy. This precaution reduced the risk of classifying patients with marginal trunk orientation due to measurement uncertainty as tilted. Therefore, a trunk tilt was defined as a trunk orientation outside the defined range of normality.
The distribution of VV orientation for the 3 settings was normal for each group (all Kolmogorov–Smirnov tests: P > .2), and the distribution of the VV within-subject variability was normalized after log transform (all Kolmogorov–Smirnov tests: P > .2). A 2-way ANOVA was performed on both VV orientation and variability with groups (controls, ISP, non-ISP) as between factors and setting (nonmaintained, partially maintained, maintained) as a within factor. Tukey HSD tests were used for post hoc analyses. The α risk was fixed at P < .05.The partial eta squared (
Results
VV Orientation (Figure 2A)
The 2-factor ANOVA (group, setting) showed group, F(2, 53) = 10.2, P < .001,
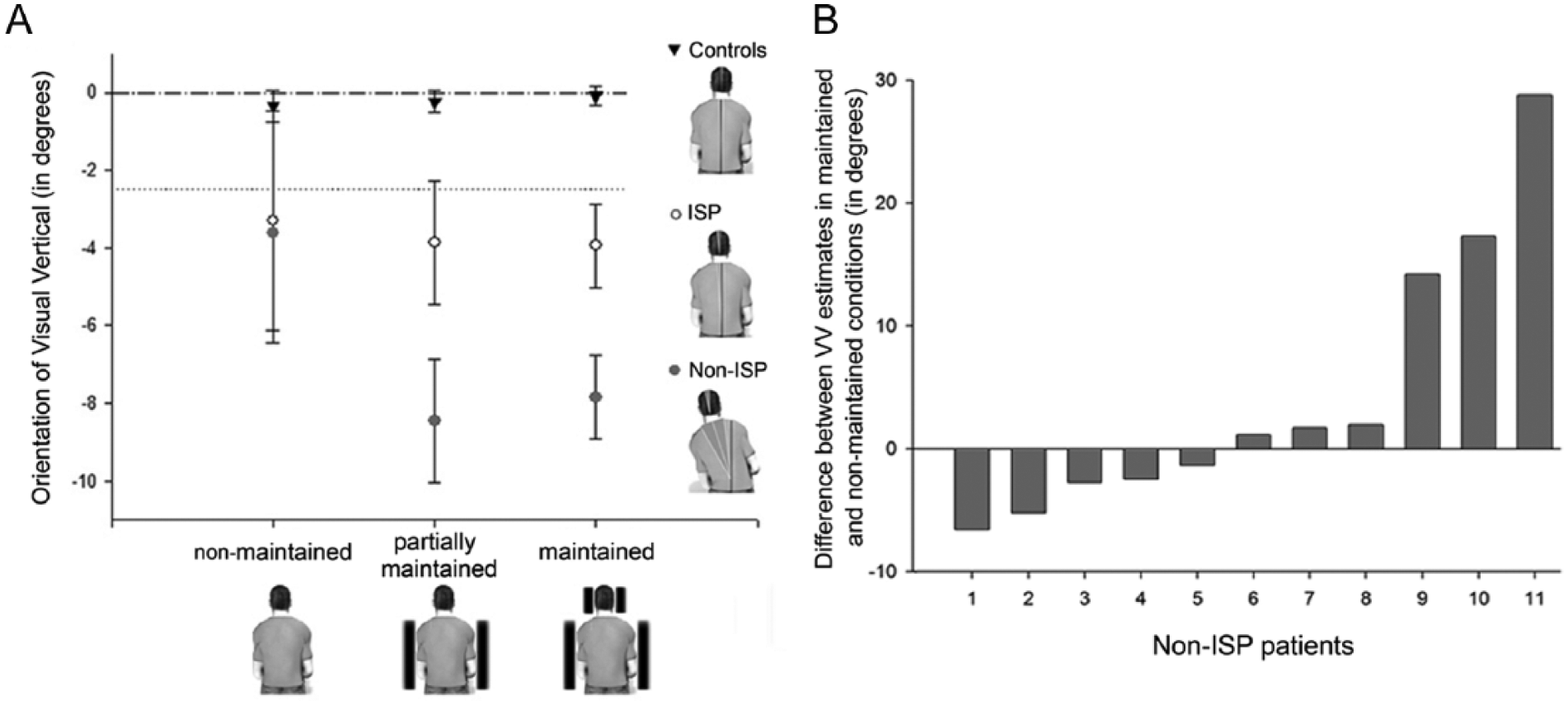
(A) Visual vertical (VV) orientation as a function of group (controls, patients with Independent Sitting Posture [ISP] and without [non-ISP]) and setting (nonmaintained body, partially maintained body, maintained body). The error bars represent the standard error of the mean. (B) Individual differences between VV estimates in the maintained and nonmaintained settings for the 11 non-ISP patients. Positive values indicate a more contralesionally biased VV and negative values an ipsilesional VV bias in the nonmaintained setting.
VV Variability (Figure 3)
The 2-factor ANOVA (group, setting) showed a difference between groups, F(2, 53) = 17, P < .001,
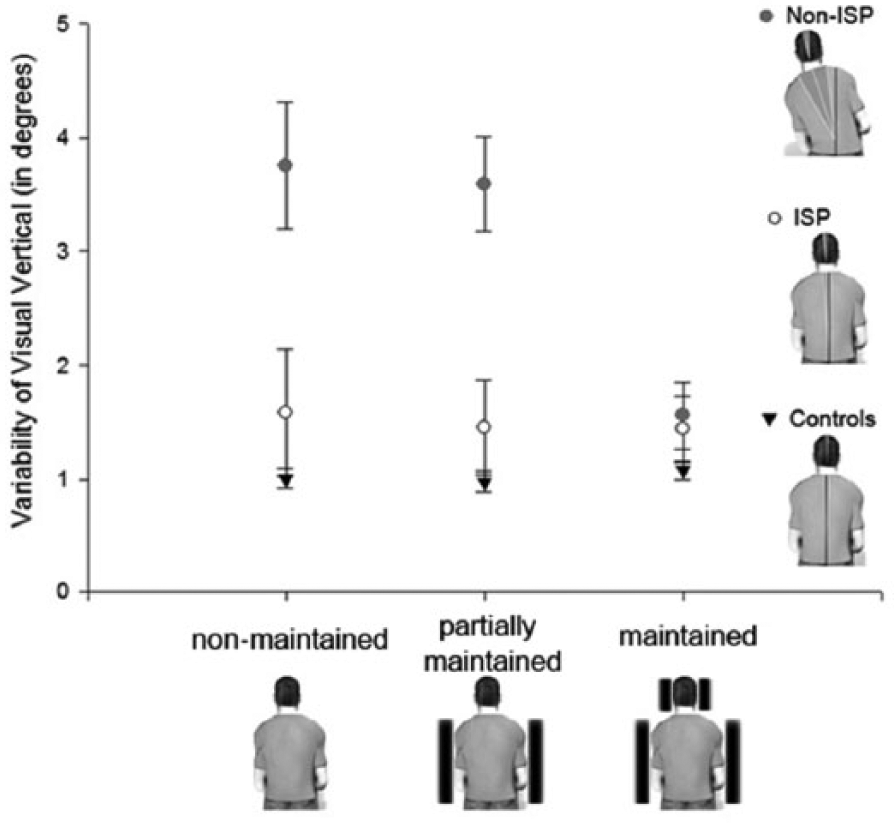
VV variability as a function of group (controls, patients with Independent Sitting Posture [ISP] and without [non-ISP]) and setting (nonmaintained body, partially maintained body, maintained body). The error bars represent the standard error of the mean.
Spontaneous Segmental Body Orientation in Roll Trunk Orientation (Figure 4A)
In the nonmaintained setting, t tests against zero showed that the trunk was spontaneously upright for controls (0.01 ± 1.5°), t(19) = 0.03, P = .9, but tilted to the contralesional side for patients. Trunk contralesional tilts were slight for ISP patients (−1.8 ± 2.6°), t(24) = −3.38, P = .002, and pronounced for non-ISP patients (−18.4 ± 11.7°), t(10) = −4.99, P < .001, with an effect of group, F(2, 53) = 41.8, P < .001,
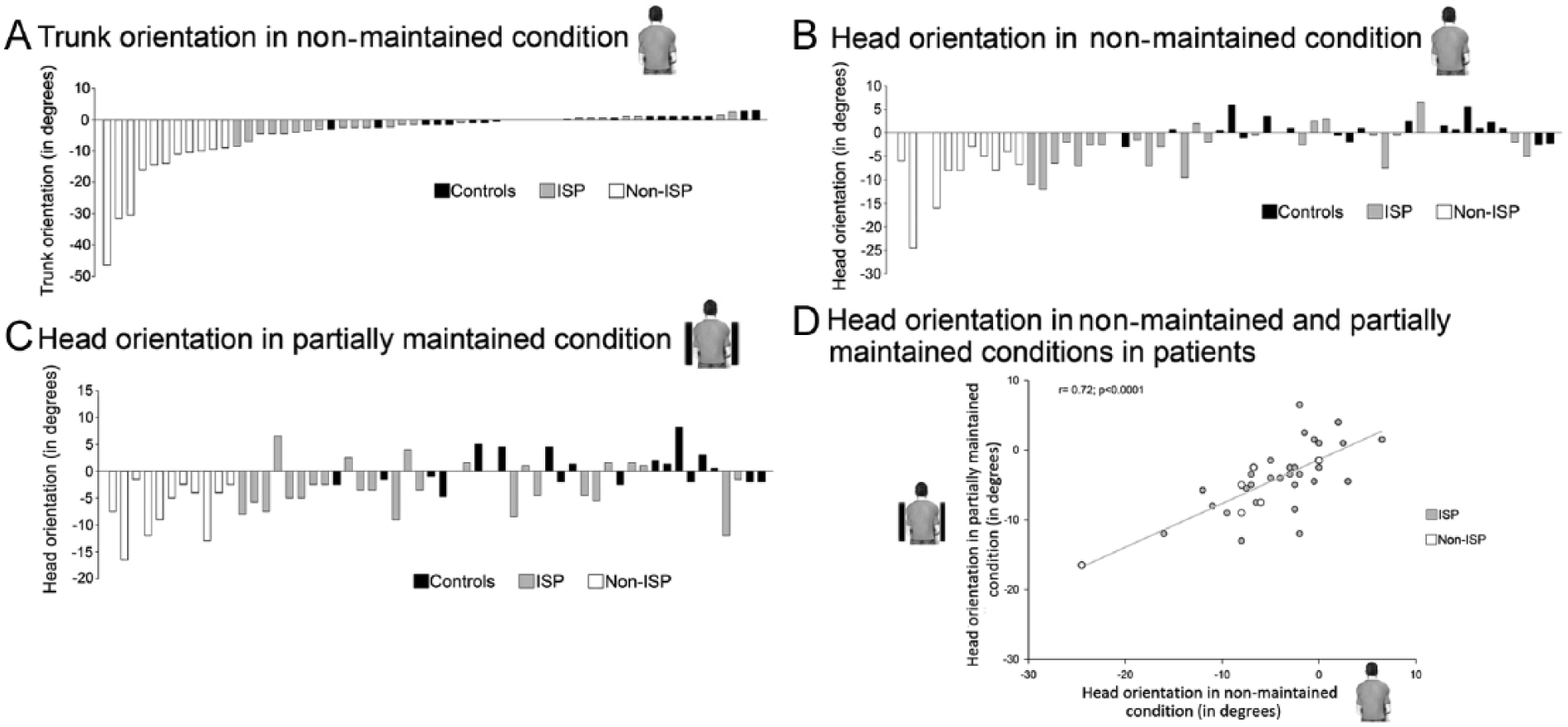
Individual mean orientations of (A) trunk axis and (B) head axis in nonmaintained setting, (C) head axis in partially maintained setting (trunk maintained). The data were classified from the most pronounced contralesional tilt (negative values) to the most pronounced ipsilesional tilt (positive values), for trunk orientation (A), and this order was maintained for the classification of head orientation data (B and C). Controls are in black, patients with Independent Sitting Posture (ISP) are in gray, patients with Non-Independent Sitting Posture (non-ISP) are in dark gray. (D) Correlation of head orientation of patients (ISP and non-ISP) between nonmaintained and partially maintained settings.
Head Orientation (Figure 4B and C)
In the nonmaintained setting, t tests against zero showed that the head was spontaneously upright for controls (0.8 ± 2.3°), t(19) = 1.5, P = .15, but tilted to the contralesional side for patients, with a slight magnitude for ISP patients (−2.8 ± 4.3°), t(24) = −3.2, P < .01, and a pronounced magnitude for non-ISP patients (−8.1 ± 6.4°), t(10) = −4, P < .01, and a group effect, F(2, 53) = 14.4, P < .001,
Similarly, in the partially maintained setting, the head was upright for controls (0.6 ± 3.5°), t(19) = 0.8, P = .44, but tilted to the contralesional side for patients, with a slight magnitude for ISP patients, (−2.9 ± 4.4°), t(24) = −3.2, P < .01, and a pronounced magnitude for non-ISP patients (−7 ± 4.7°), t(10) = −4.7,
Head orientation did not differ between the nonmaintained and partially maintained settings for both non-ISP (P = .34) and ISP patients (P = .93). Furthermore, head orientation in these 2 settings was strongly correlated (r = .72, P < .001; Figure 4D). Therefore, head orientation was independent of trunk orientation.
Influence of Spontaneous Body Orientation on Postural Disorders
Partial correlation analysis indicated a link between trunk orientation and PASS scores (r = .49, P < .05) when controlling for head orientation. Non-ISP patients had the greatest trunk tilt (Figure 4A). The positive predictive value of trunk tilt on the capacity to maintain an independent sitting posture was 69% (11/16) and the negative predictive value was 100% (20/20) (Fisher exact probability test: P < .001; Table 2).
Positive (69%) and Negative (100%) Predictive Values of Trunk Tilt on the Capacity to Maintain an Independent Sitting Posture a .
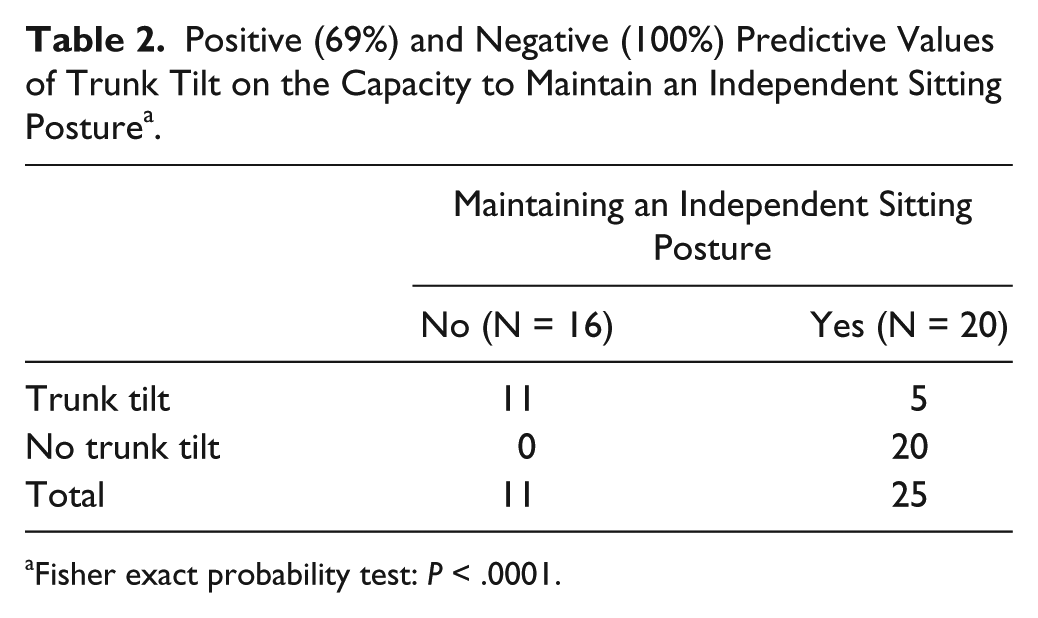
Fisher exact probability test: P < .0001.
Partial correlation analysis of head orientation and PASS scores were not significant (r = .31, P = .07) when controlling for trunk orientation. Both ISP and non-ISP patients showed head tilt (Figure 4B and C).
Discussion
Measurement of verticality perception is now part of the assessment of balance disorders in current clinical practice. Despite abundant literature, guidelines for clinical measurements, especially regarding participant settings for measuring VV, are lacking. We aimed to analyze the interest of controlling body orientation while stroke patients estimate VV. We quantified both trunk and head orientations and analyzed their association with patients’ ability to sit on their own as well as the impact on VV.
Our study revealed that (a) trunk and head setting had a major impact on VV measurements, especially in patients with postural disorders, and (b) the recovery of a spontaneous upright trunk (and not head) orientation affected the recovery of an independent sitting posture in patients with subacute stroke.
Influence of Setting on VV Orientation
In the maintained setting, most patients presented “severe” contralesional VV biases as expected and as reported in several studies.2-6,11,23
Postural setting had no effect on VV orientation in controls and ISP patients, all able to sit upright. One may assume that the pressure exerted on the body (trunk and head) by the lateral wedges of the device was weak, rendering negligible the modulation of verticality representation by this additional element of somatosensory information.
In contrast, postural setting greatly affected VV orientation in non-ISP patients who were unable to sit. These patients, with severe postural disorders, all presented a right-sided lesion, in agreement with the literature.37-40 Given this lateralization of the lesion and its large size in most cases, we would have predicted contralesional VV biases of great magnitude.6,10,11,23 These expected results (−8°) were found only when the trunk was maintained. When neither the trunk nor the head was restrained (nonmaintained setting), the magnitude of contralesional VV biases was halved (−3.6°) and similar to the magnitude found in ISP patients, regardless of testing conditions. The ANOVA showed that the nonmaintained setting did not discriminate the profiles of patients in terms of verticality perception on one hand and on the other hand induced unexpected behavior in terms of VV estimates in non-ISP patients. This result is robust, given that assumptions for ANOVA were met. This counterintuitive result was due to large-scale dispersion of individual behavior. When non-ISP patients had to actively maintain a seated posture, some showed an amplified or a stable contralesional VV bias and the others surprisingly showed a marked ipsilesional VV bias, whereas most of these patients presented the expected6,10,11,23 severe contralesional VV tilts in trunk maintained settings. Therefore, VV perception was not improved when patients had to actively maintain their seated posture. These very different individual behaviors indicated that VV orientation assessed with the trunk not maintained upright was multidetermined and did not reflect just the internal model of verticality. Our study reveals that, in this setting, interpreting and determining what is really measured with VV estimates is difficult, so testing VV in a free sitting posture is not valid for stroke patients showing postural disorders and especially lateral trunk tilt, although these patients are the most interesting to investigate.6,20 Assessing VV orientation in these patients requires maintaining the trunk.
Influence of Setting on VV Variability
VV variability was low in all settings for both controls and ISP patients and twice as high for non-ISP patients in nonmaintained or partially maintained settings. Therefore, VV estimates were consistent for controls and ISP patients but not non-ISP patients. In the latter, VV variability was as low as in controls and ISP patients only when both the trunk and head were maintained upright and thereby aligned. We assume that this congruency between the axes (ie, between somatosensory and vestibular graviception) optimizes the construction of the internal model of verticality, which becomes more robust (lower within-subject variability). This interpretation agrees with a previous study, 23 showing enhanced robustness of verticality representation when both somesthetic and vestibular graviceptive signals were available and congruent. Moreover, awareness of the body orientation itself modulates the perception of VV, thus leading to a reduced within-subject variability. 41 In our experiment, we may assume that because patients were aware of being upright in the maintained setting, their VV variability was lower. Limited variability is an essential criterion for the validity of a measurement.
Which Type of Setting Is Needed for Measuring VV in Clinical Practice?
To perform this experiment, we used a device called the “wheel paradigm,” designed for measuring the postural perception of the vertical, 6 which allows for precise control of trunk and head orientations. In patients with postural disorders, we showed that maintaining the trunk upright was a condition for the validity of results on VV orientation, the main criterion given by this test, if not the only criterion for clinical practice.2-6,9,10,16-18,20,23,33,34,41 To maintain the body upright during VV assessment, most clinical teams or research laboratories use a chinrest with an adjustable height.2,7,16,20,33,34 This setting is assumed to be sufficient to control the orientation of the trunk, ensuring a valid VV in stroke patients with lateral postural disorders. Beyond postural disorders, our experiment shows that measuring VV orientation without any body maintaining3,5,8-10 is valid in subjects with satisfactory balance abilities and able to sit upright. The use of a chinrest for VV assessment in most previous studies2,7,16,20,33,34 was probably sufficient to ensure the validity of the results in terms of VV orientation, because supporting the head maintains the trunk upright.
A complementary criterion of VV assessment is the within-subject variability.3,5,20,23,41 We showed that to be valid, this criterion must be obtained with the head maintained upright. If a chinrest maintains the trunk upright and controls the head position on the vertical axis, it does not preclude any head rotation in roll. Our study suggests that valid within-subject VV variability requires maintaining the head upright with a suitable device.4,6,11 This is not the usual situation. However, this criterion is poorly assessed3,20,23 with a clinical usefulness that remains to be further investigated.
Trunk Orientation Affects a Patient’s Ability to Sit
Postural balance is ensured when the projection on the ground of the center of mass is inside the base of support. To reduce the destabilizing effect of gravity, the body must be oriented vertically (postural orientation) and the control of the oscillations around this orientation optimized (postural stabilization).42,43 This conception, which involves dissociated postural control of the orientation and stabilization, allows for better understanding postural disorders in brain-damaged patients.
In our study, no patients with severe contralesional trunk tilt (>10°) were able to sit independently (Figure 4A). All patients with an upright trunk were able to sit independently. This finding confirmed for the first time the clinical intuition that spontaneous upright trunk orientation is necessary for recovering an independent sitting posture after stroke. Interestingly, our results revealed that a moderately tilted trunk (under 10°) is not sufficient to compromise the ability to sit.
Regarding the organization of the postural control,42,43 postural stabilization in sitting is not a challenging task, given the low height of the center of mass in terms of the base of support and its size. In contrast, orienting the body with a pronounced lateral trunk tilt in relation to a biased verticality perception 6 shifts the center of mass toward the limit of stability. The lateral head tilt did not affect the ability to sit for stroke patients.
Our study suggests that postural orientation deficits, with a critical role of trunk orientation, are a major cause of lateral postural disorders after a stroke.
Dissociated Control for Trunk and Head Orientations
When not maintained, the trunk and head were contralesionally tilted for both ISP and non-ISP patients. These tilts were more pronounced for non-ISP than ISP patients. Maintaining the trunk upright did not modulate the head orientation in space. The independence of trunk and head orientations in roll has been noted in stroke patients performing a dynamic sitting task on a laterally unstable seesaw. 44 Patients with “pusher behavior” presented a pronounced contralesional tilt of the trunk and a correct head orientation in space. Such dissociated control for trunk and head orientation is probably due to the different types of sensory information involved: mainly somatosensory information for the trunk and vestibular and visual information for the head.45-47
Limitations of the Study
The perception of VV may be affected by ocular torsion. Although ocular torsion may occur spontaneously after vestibular lesion or in a small number of patients with hemisphere lesions, 2 our results should not have been affected by ocular torsion. Indeed, we did not include patients with signs of vestibular disorders, and our results mainly rely on the individual modulation of VV estimation according to postural settings.
Owing to the relatively small sample size of our study, we cannot draw any conclusions about the prevalence of trunk and head orientation biases after stroke. Studies with a larger sample based on quantitative analysis of lesion location are needed to further investigate the prevalence of head and trunk tilts after stroke as well as its anatomical correlates.
Trunk and head orientations were manually measured. However, these measurements were performed by one physiotherapist (BS) expert in stroke rehabilitation, who was trained to measure trunk and head orientations, was blinded to VV estimates and to the study hypotheses. Measurements accuracy was under 1°. The differences between groups and settings were >1°. In a clinical perspective, our study shows the clinical interest of such simple measurements of trunk and head orientations in stroke rehabilitation.
Head orientation in roll was estimated on the basis of the orientation of the bipupillary line with respect to the horizontal plane. The bipupillary line allows for good quantification of the orientation of the head in roll except in patients with skew deviation due to vestibular disorders. 2 Patients with signs of vestibular disorders and especially skew deviation were not included in the study, which limited this possible methodological bias.
We used a blinding procedure for trunk and head orientation measurements (physiotherapist blinded to the study hypotheses and protocol) but not VV assessment (operator informed of experimental conditions and hypotheses). Nevertheless, the possible influence of the operator on VV estimations was limited because the operator ignored the patient’s PASS scores determining the patient’s group (ISP and non-ISP patients).
Conclusion
This study reveals that participant setting strongly affects the estimation of VV in stroke patients who have difficulties maintaining a seated posture. Trunk and head maintained upright was the optimal setting for posture. Only this setting had good discriminative ability (able to distinguish profiles of patients based on VV orientation) and low variability. In addition, this setting is also interesting to quantify the changes of verticality perception regardless of patients’ postural behavior, due to the mastering of both trunk and head orientations.
Footnotes
Acknowledgements
The authors thank the team of physiotherapists of the neurorehabilitation unit of the University Hospital of Grenoble for help with data collection regarding motor weakness, sensory loss, balance, and lateropulsion scales.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was part of a project funded by La Fondation de l’Avenir, programs ETO-300 and ETA-382. CP was funded by a grant from “La Société Francophone Posture Equilibre Locomotion” (SOFPEL).
