Abstract
Background
Patients with poststroke pusher syndrome (PS) require longer duration of rehabilitation and more supplemental care after discharge. Effective treatment of PS remains a challenge. The role of repetitive transcranial magnetic stimulation (rTMS) for PS has not been examined.
Objective
Assess the efficacy of rTMS for patients with poststroke PS in reducing pushing behavior, enhancing motor recovery and improving mobility.
Methods
A randomized, patient- and assessor-blinded sham-controlled trial with intention-to-treat analysis was conducted. Thirty-four eligible patients with poststroke PS were randomly allocated to receive either rTMS or sham rTMS for 2 weeks. Pushing behavior on the Burke lateropulsion scale and scale for contraversive pushing, motor function on Fugl-Meyer assessment scale-motor domain (FMA-m) and mobility on modified Rivermead mobility index were measured at baseline, 1 and 2 weeks after intervention. Repeated-measures analysis of covariance was used for data analysis.
Results
There was no significant interaction between intervention and time on Burke lateropulsion scale (F = 2.747, P = .076), scale for contraversive pushing (F = 1.583, P = .214), or change of modified Rivermead mobility index (F = 1.183, P = .297). However, a significant interaction between intervention and time was observed for FMA-m (F = 5.464, P = .019). Post hoc comparisons of FMA-m show better improvement in rTMS group with mean differences of 12.7 (95% CI −7.3 to 32.7) and 15.7 (95% CI −4.6 to 36.0) at post-treatment week 1 and week 2 respectively.
Conclusions
rTMS did not demonstrate significant efficacy in improving pushing behavior and mobility in patients with PS. However, rTMS might have potential effect in enhancing motor function for patients with PS.
Registration:
The study was registered in the Chinese Clinical Trial Registry (registration No. ChiCTR2200058015 at http://www.chictr.org.cn/searchprojen.aspx) on March 26, 2022.
Introduction
Pusher syndrome (PS), or lateropulsion, is a common postural impairment in patients after stroke. 1 It is characterized by patients actively pushing toward their hemiparetic side and exhibiting resistance to passive correction of the body to the vertical upright position. 2 PS was recently reported in 41% of stroke patients. 3 The motor recovery and functional abilities were found to be significantly poorer among pushers compared to nonpushers at 3 months post-onset. 4 This highlights the necessity for a longer duration of rehabilitation and more supplemental care after discharge from an inpatient rehabilitation setting.1,4,5 This also implies an increase in the burden of care on the health system.
Various interventions for poststroke PS have been reported in the literature, but their efficacy remains uncertain. 6 Previous studies have predominantly demonstrated the beneficial effects of interventions focusing on conscious visual feedback for patients with PS, as indicated by observational reports.7-11 Observational reports or non-randomized controlled trials have also shown that transcranial direct current stimulation (tDCS) has a positive effect on improved posture in patients with poststroke PS.12-15 However, the certainty of evidence is low due to the limitations in study design. For controlled trials, robotic, or machine-assisted somatosensory cue training shows better outcomes than visual feedback training16-18 or general postural training. 19 However, the somatosensory input only plays a relatively minor role in PS. 20
Repetitive transcranial magnetic stimulation (rTMS) is a method of noninvasive neuromodulation (NINM) using a high-intensity magnetic field generated by an electric current passing through an inductive coil to modulate the excitability of cerebral cortex. 21 The existing literature consistently supports the positive effect of rTMS in improving motor, leg mobility, activity level, walking, and balance after stroke with minimal or no occurrence of adverse events.22-24 Clinical practice guideline indicates that rTMS can effectively ameliorate neglect symptoms similar to PS with an evidence of class IIb, level B. 25 Considering its ability to directly modulate cortical excitability in the brain regions responsible for the egocentric postural reference system, the application of rTMS could be considered as a potential intervention for PS, aiming to restore bilateral hemispheric balance. However, no clinical trial investigating the efficacy of rTMS in treating PS has been conducted so far.
The inferior parietal lobe (IPL) has been implicated as a potential contributor to the development of PS in previous studies.26,27 This region evaluates and integrates various sensations to generate a perception of postural vertical and establish an egocentric reference system for maintaining upright posture. 26 Moreover, it may also play a role in processing gravity perceptive information that affects the perception of postural upright. 27 Lesion in this region disrupts the subjective orientation of postural vertical. 28 A recent investigation, employing a case-control design with a large sample size and matched comparison group, as well as utilizing sophisticated multivariate statistical approach for lesion symptom mapping, further identified the IPL, extending from Brodmann area 2 to Brodmann area 40, as a crucial neuroanatomical determinant for PS. 29 One may speculate that modulating IPL activity via rTMS may be a rational therapeutic strategy for PS. Therefore, the research questions for this randomized, patient- and assessor-blinded, controlled trial were as follows:
In patients with poststroke PS, does combined rTMS applied over the IPL and usual rehabilitation (UR) ameliorate pushing behavior more than sham rTMS (S-rTMS) combined with UR?
How do the rTMS and S-rTMS compare regarding their effects on motor recovery and mobility, as well as adverse events?
Methods
Study Design
This parallel, two-group, randomized controlled trial was conducted with concealed allocation, blinding of patients and assessors, and intention-to-treat analysis using a repeated-measures design. Eligible participants were randomly allocated into either the rTMS group (experimental group) or the S-TMS group (control group) for 2 weeks. Considering that the type of stroke could be a possible confounding factor, stratified blocked randomization 30 was performed to balance the number of patients after ischemic stroke or hemorrhagic stroke in both groups with two separate allocation sequences generated for patients after each type of stroke. After the allocation sequences were generated by an independent researcher not involving in the delivery of interventions, the sequences were placed in sequentially numbered sealed opaque envelopes and were stored in a locked cabinet. When an eligible participant was recruited into the trial, the therapist would contact the independent researcher to open the sealed envelope to determine the allocation of that particular participant. The assessors and patients were blinded to the group assignment. Outcomes were measured at baseline (T0) and after 1 week (T1) and 2 weeks (T2) of intervention. The trial was publicly registered with the protocol published in a peer review journal. 31
Participants
Patients with poststroke PS were recruited by a researcher from the inpatient and outpatient Department of Rehabilitation Medicine in West China Hospital, Sichuan University, China. To be eligible for inclusion, participants had to be aged between 18 and 80 years; ≤6 months following an ischemic or hemorrhagic stroke; with poststroke PS defined as a Burke lateropulsion scale (BLS) score of 3 or greater during the initial examination; ability to follow two-step commands, “take the paper in your unaffected hand and fold it in half”; and absence of other neurologic disorders. Exclusion criteria were visual field deficits or eye muscle paralysis; orthopedic conditions limiting participation, for example, fracture, severe osteoporosis, contractures of the lower extremities; having an unstable medical condition, or being unable to safely perform exercise; and presence of metal implants, cardiac pacemakers, brain tumors, meningitis, or epilepsy. Written informed consent was obtained from all participants or caregivers.
Interventions
All patients received the UR program according to the clinical practice guideline for adult stroke rehabilitation. 25 The UR lasted approximately 4 hours daily for 5 days per week for 2 weeks, with exercise graduated according to their impairments and recovery. The detailed UR program had been shown in the published protocol. 31
Experimental Group
In addition to UR, participants in the experimental group received rTMS sessions over the intact IPL at the junction of Brodmann area 2 and Brodmann area 40, using a rapid magnetic stimulator (YIREUIDE®, Wuhan, China) after UR. A figure-eight coil was oriented at a tangent to the target scalp. Given the absence of prior research on rTMS for poststroke PS, we employed a continuous theta burst stimulation (cTBS) protocol, 32 which was an inhibitory rTMS technique used for neglect treatment and had been endorsed in update evidence-based guidelines on the therapeutic use of rTMS. 22 Ten sessions of cTBS were administered over two weeks (10 work days), and the detailed parameters used in each session were set as follows:
a. Nonlesional IPL as the target area, corresponding to the CP3 or CP4 site of the international EEG 10-10 system based on Lefaucheur et al.’s study. 22 Localization method based on recommendations from American Clinical Neurophysiology Society and the International Federation of Clinical Neurophysiology 33 was shown in Supplemental file 1.
b. The intensity was set at 80% of the resting motor threshold. The resting motor threshold, defined as the minimum stimulation intensity required to elicit motor evoked potentials exceeding 50 μV in at least 5 out of 10 trials at rest, was determined prior to treatment. This measurement was performed over the primary motor cortex of the unaffected hemisphere using Rossini’s procedure 34 under electroneuromyography control.
c. The cTBS protocol comprised 801 pulses delivered in a continuous train of 267 bursts. Each burst consisted of three pulses at 30 Hz, repeated at 6 Hz. The duration of one single cTBS train was therefore 44 seconds.
d. Eight cTBS protocols with an interval of 30 seconds were applied per session per day.
Control Group
In addition to UR, participants in the control group were given pseudostimulation after UR for 2 weeks. The treatment parameters of the control group were identical to those of the experimental group, with the exception that the coil was positioned perpendicular to patients’ heads and patients were exposed to the stimulation sound without receiving actual stimulation. The positioning of the coil for rTMS group and S-rTMS group was shown in Supplemental file 2.
Outcome Measures
Baseline characteristics of patients were collected in both groups, such as age, gender, stroke characteristics (duration, type, lesion side, and lesion location), handedness, BLS scores and presence of neglect or aphasia. Baseline assessment was performed before randomization.
The primary outcome measures were the BLS and scale for contraversive pushing (SCP) to assess poststroke pushing behavior. The secondary outcome measures were the Fugl-Meyer assessment scale-motor domain (FMA-m) and the modified Rivermead mobility index (MRMI) to assess motor functions and mobility. Safety was reported as the start date, stop date, description, severity, and amounts of adverse events or serious adverse events.
BLS
The BLS had recently been recommended as the preferred tool to evaluate PS. 35 The BLS is both a reliable and a valid assessment of lateropulsion following stroke and has sound clinimetric properties.36-38 BLS might be more sensitive for detecting mild pusher behavior in standing and walking. 37 A 17-point ordinal scale is used in the BLS to evaluate postural alignment according to how much resistance is assessed by the examiner while the patient performs the following functional activities: rolling, sitting, standing, transferring, and walking. 38 Scores range from 0 for those without lateropulsion to a maximum score of 17. The cutoff for the diagnosis of pusher behavior is ≥3 points. 39 Change in BLS had been considered significant when the change value was more than 1 point. 17 However, without the availability of the minimal clinically important difference (MCID) of BLS from the literature, the most common and well-described distribution-based formula for MCID calculation using the half standard deviation (SD) (MCID = 0.5* SD) of baseline scores would be adopted in this study. 40
SCP
The clinimetric properties of the SCP had been tested extensively. 36 The validity of the SCP had been established, with the interrater reliability of the SCP reported to be good to excellent with regard to both each subscore and the total score. The internal consistency was very high, along with correlations between the subscore and total score of the scale. 41 The construct validity of the SCP was demonstrated by significant moderate to high correlations with mobility, functional, and balance scores. 42 There was almost perfect agreement with clinical diagnosis with a cutoff >0 in each category. 42 The SCP includes three components with a total score ranging from 0 to 6: (1) the symmetry of spontaneous body posture, (2) the use of nonparetic extremities to push away from the unaffected side of the body, and (3) the resistance to passive correction of the tilted posture. 43 Each component is tested in sitting and standing positions, yielding a maximum score of 2 per component. The half SD of SCP scores at baseline was set as the MCID.
FMA-m
The FMA is a well-designed, feasible, and efficient clinical examination method that has been tested widely in the stroke population. 44 The FMA scale is divided into five domains: motor function, sensory function, balance, joint range of motion, and joint pain. The motor domain is highly recommended as a clinical and research tool for evaluating changes in motor impairment following stroke. 44 The FMA-m includes items measuring movement, coordination, and reflex action about the shoulder, elbow, forearm, wrist, hand, hip, knee, and ankle. Each item is scored on a 3-point ordinal scale. The motor score ranges from 0 to a maximum of 100 points, divided into 66 points for the upper extremities and 34 points for the lower extremities. A minimum change of 15 points, with at least 9 points in the upper extremities45,46 and 6 points the in lower extremities, 47 was set as the MCID of FMA-m.
MRMI
MRMI is a short and simple test of mobility in routine clinical practice. The MRMI has good to excellent measurement properties with good content validity, high responsiveness, adequate predictive validity, excellent test-retest reliability, high internal consistency, and unidimensionality. 48 The MRMI consists of eight items, including turning over, changing from lying to sitting, maintaining sitting balance, going from sitting to standing, standing, transferring, walking indoors, and climbing stairs. The MRMI score ranges from 0 to 40. Scores are assigned based on direct observations of the patient’s performances in given items. The half SD of MRMI scores at baseline was set as the MCID.
Data Collection
All outcome data were collected by two independent researchers who were blinded to the group assignment and not involved in the delivery of interventions to the participants. The assessors had more than 10 years of working experience in stroke rehabilitation and in using the outcome measures, especially BLS and SCP. To improve the reliability of scoring, all the assessment scales were administered jointly by the two independent researchers.
All baseline data and raw data were recorded on case report forms in a complete, accurate, and clear manner immediately upon data acquisition. The two assessors crossed check to ensure the accuracy and completeness of the data. A database using the Microsoft Excel was used to manage the data. All research data were entered into this electronic database on the last day of the month. Data input and proofreading were performed by the two independent researchers, with double checking of correct data entry and storage.
Data Analyses
Sample size for the trial was calculated using G*Power software version 3.1.9.2. An effect size of 1.0 was used to compare the change scores of BLS using an independent t test between the two groups, with a two-tailed level of significance of .05 and a statistical power of 80%. A total sample size of 34 was needed.
Categorical variables were expressed as frequencies and percentages. Continuous variables were expressed as the means and SDs. Demographics and baseline clinical characteristics of participants were compared between groups using independent t tests or Mann-Whitney U tests (for continuous variables), or Chi-squared tests or Fisher’s exact tests (for categorical variables). The normality of continuous data was examined using the Shapiro-Wilk test. Continuous variables were added as covariates in the repeated-measures analysis of covariance (rmANCOVA) when obvious or important baseline differences existed. Categorical variable with baseline difference was added as the independent variable in the rmANCOVA. Mauchly’s test of sphericity was used to examine the assumption of sphericity of the rmANCOVA. Post hoc multiple comparisons with Bonferroni correction were performed to find out the significant differences once the interaction effect was significant. All analyses were performed using IBM SPSS Statistics for Windows version 25.0 (Armonk, NY: IBM Corp). Following the Consolidated Standards of Reporting Trials (CONSORT) statement, all our analyses followed the intention-to-treat principle. The missing data of any variable at T2 were analyzed and filled using with multiple imputation method in IBM SPSS. 49 The level of significance was set at .05 for all analyses.
Results
Compliance with the Trial Protocol
The interventions were applied as described in the registered protocol. Due to the management policy of hospitals during the COVID-19 epidemic, the length of hospital stay for patients was limited. The planned 3-week intervention could not be implemented, and participants only received the intervention for 2 weeks. All of the registered primary and secondary outcomes were reported after 1 and 2 weeks of intervention.
Flow of Participants Through the Study
Trial recruitment was started on 26 March 2022 and closed on 25 July 2023 due to the slower than anticipated recruitment rate during the COVID-19 period. After 118 participants had been screened, 34 fulfilled the eligibility criteria. Randomization resulted with 18 participants allocated to the experimental group and 16 participants to the control group. Figure 1 shows the flow of participants through the study.
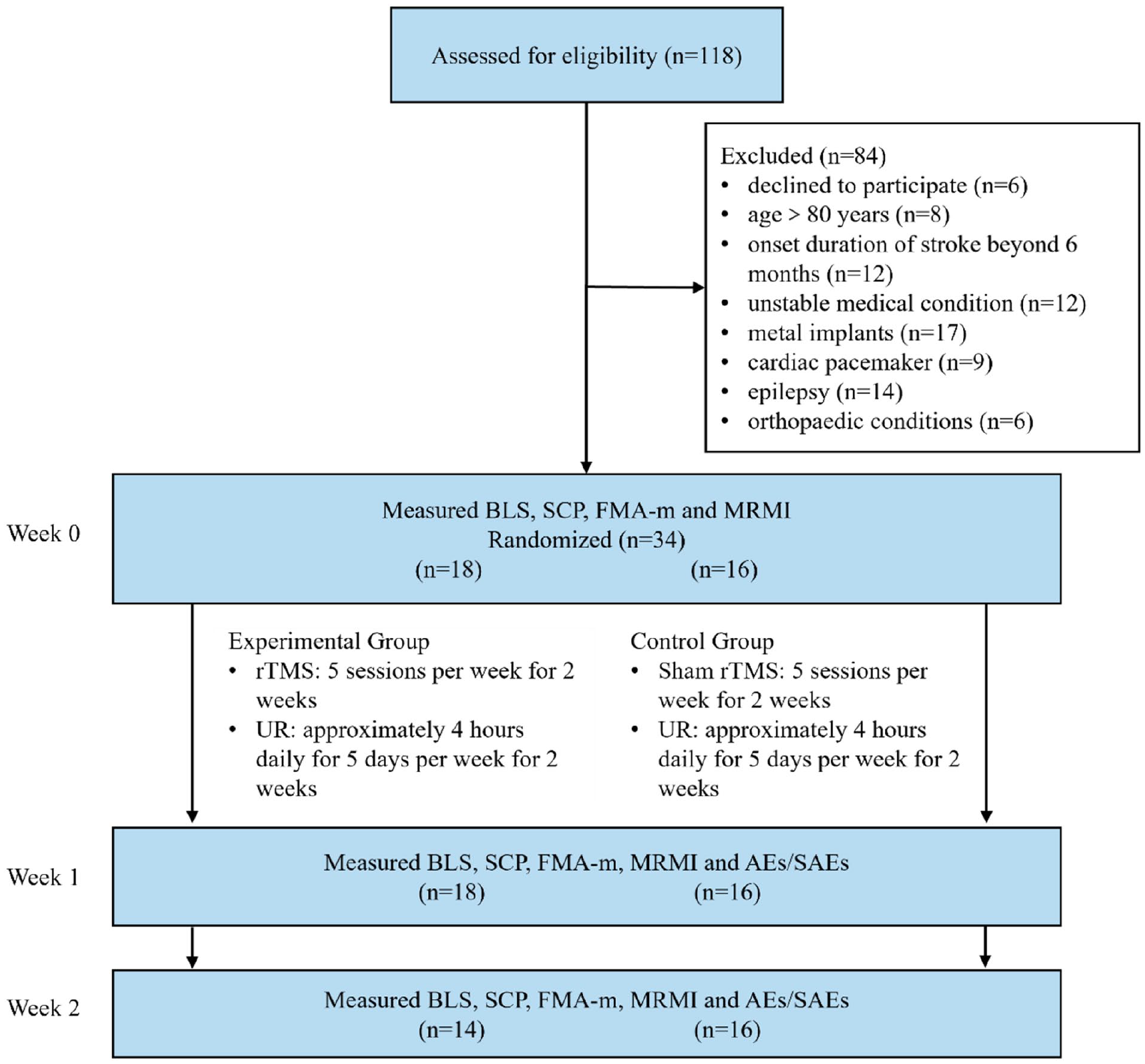
Flow of participants through the trial.
Characteristics of Participants
The demographic characteristics are presented in Table 1, and baseline scores of outcome measures are presented in Table 2. Participants in the rTMS group exhibited a higher mean age (P = .041), a greater proportion of female individuals (P = .046), and a lower MRMI score (P = .018) compared to those in the S-rTMS group. Therefore, rmANCOVA was conducted for BLS, SCP and FMA-m with age and baseline MRMI as covariates, group and gender as between-subjects factors, and time as within-subjects factor. For MRMI, rmANCOVA was performed to analyze three change scores from baseline (T1−T0, T2−T0, and T2−T1) with age and baseline MRMI as covariates, group and gender as between-subjects factors, and change in MRMI as within-subjects factor.
Baseline Characteristics of the Participants.
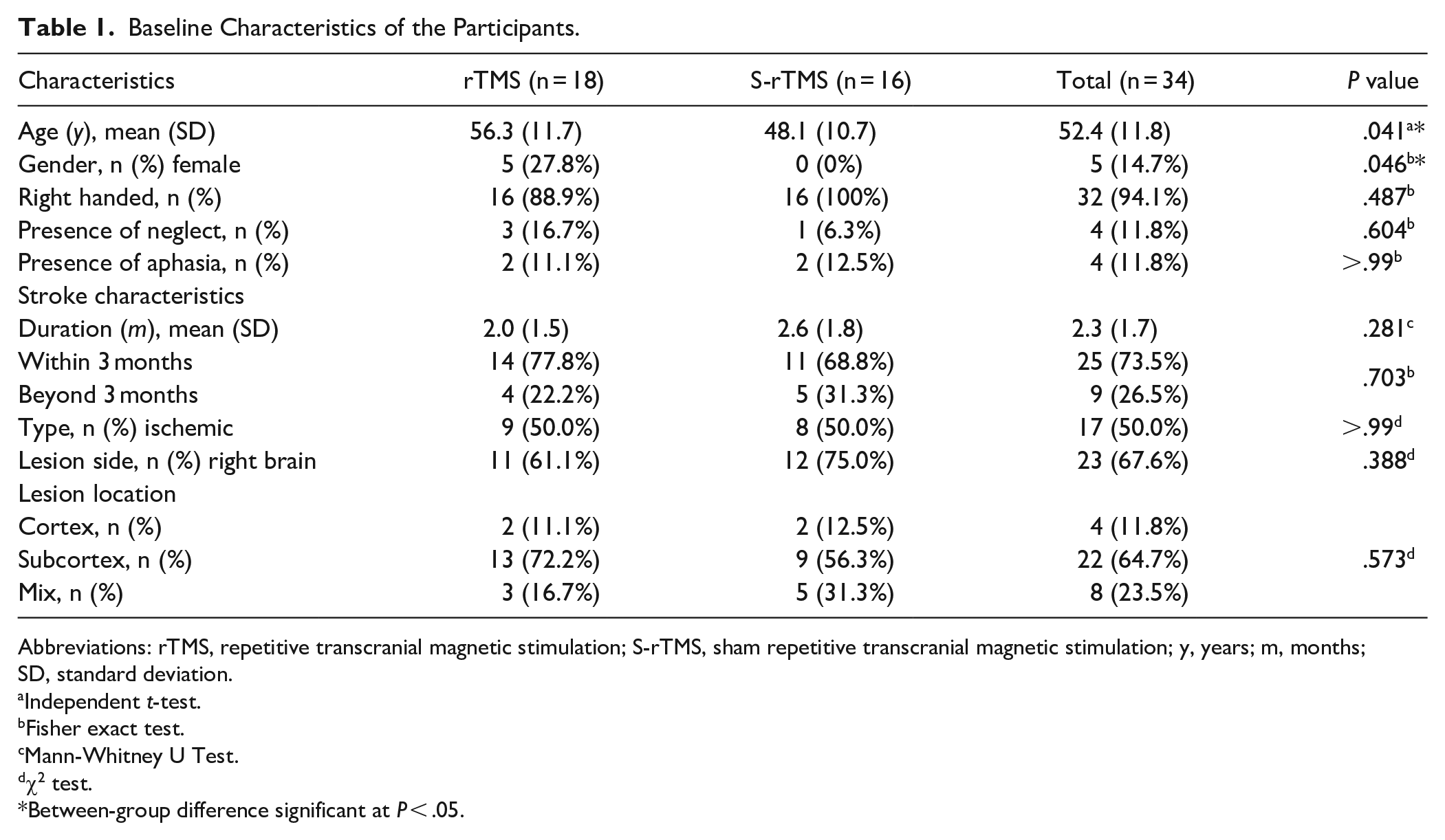
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; S-rTMS, sham repetitive transcranial magnetic stimulation; y, years; m, months; SD, standard deviation.
Independent t-test.
Fisher exact test.
Mann-Whitney U Test.
χ2 test.
Between-group difference significant at P < .05.
Analysis of the Effect of Time and Intervention on the Primary and Secondary Outcomes Measures.
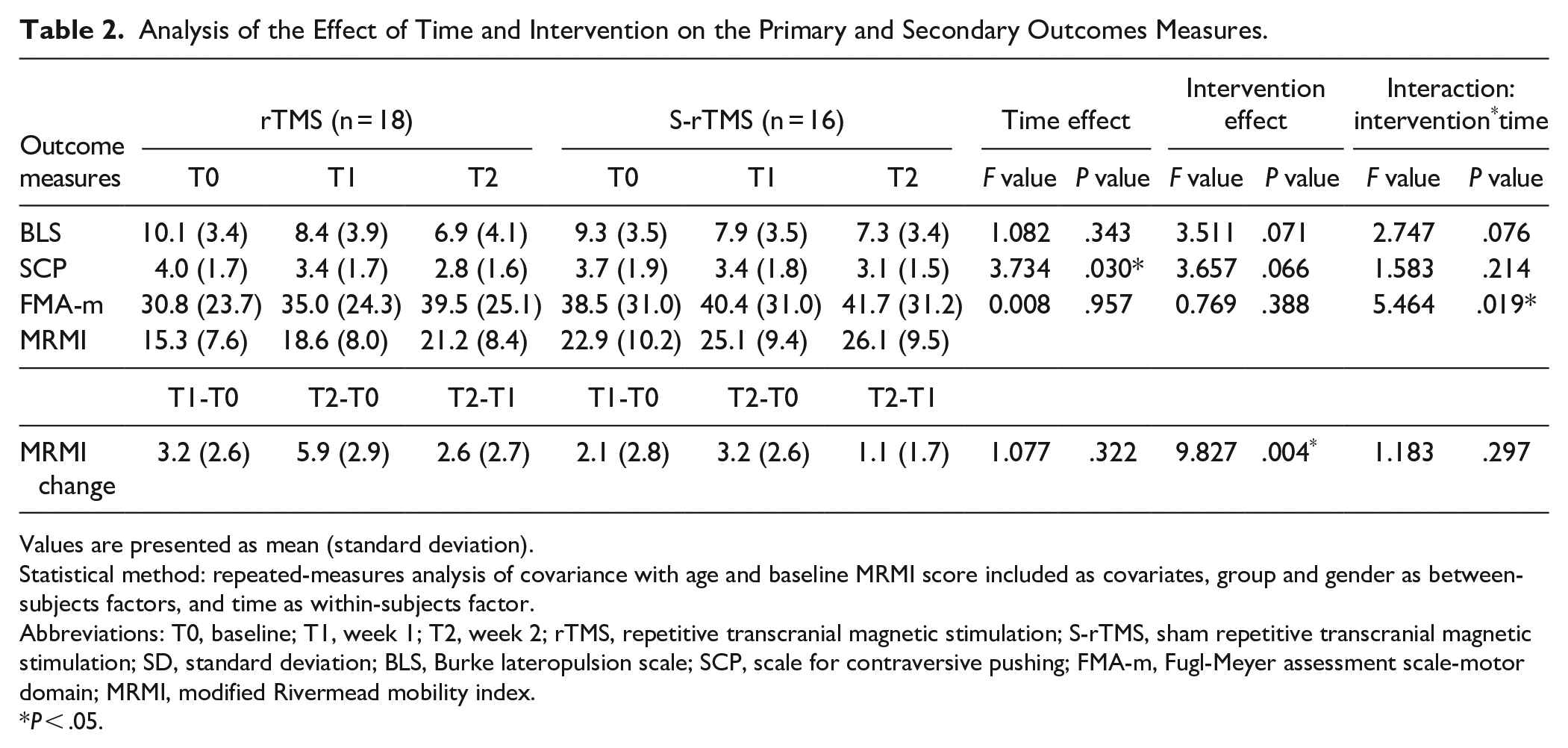
Values are presented as mean (standard deviation).
Statistical method: repeated-measures analysis of covariance with age and baseline MRMI score included as covariates, group and gender as between-subjects factors, and time as within-subjects factor.
Abbreviations: T0, baseline; T1, week 1; T2, week 2; rTMS, repetitive transcranial magnetic stimulation; S-rTMS, sham repetitive transcranial magnetic stimulation; SD, standard deviation; BLS, Burke lateropulsion scale; SCP, scale for contraversive pushing; FMA-m, Fugl-Meyer assessment scale-motor domain; MRMI, modified Rivermead mobility index.
P < .05.
Effects of Intervention
Primary Outcome
The rmANCOVA did not reveal any significant interaction effect of intervention, time and gender or interaction effect of time and gender (F = 0.310, P = .722) on BLS scores. Additionally, there was no significant interaction effect observed between intervention and time (F = 2.747, P = .076) on BLS scores (Table 2). There was no significant difference in BLS scores across different time points (F = 1.082, P = .343), as well as no significant effect of interventions (F = 3.511, P = .071) (Table 2).
The rmANCOVA revealed a non-significant interaction effect of intervention, time and gender on SCP scores, as well as a non-significant interaction effect between time and gender (F = 0.251, P = .779). Furthermore, no significant interaction effect was observed between intervention and time (F = 1.583, P = .214) on SCP scores (Table 2). The SCP scores varied with time and demonstrated statistical significance in the main effect of time (F = 3.734, P = .030) (Table 2). However, the main effect of the intervention on SCP did not reach statistical significance (F = 3.657, P = .066) (Table 2).
Secondary Outcomes
The analysis did not reveal any significant interaction effect of intervention, time and gender, or interaction effect of time and gender (F = 0.184, P = .721) on FMA-m scores. However, a significant interaction between intervention and time was observed for FMA-m scores (F = 5.464, P = .019) (Table 2). Post hoc comparisons with the inclusion of the gender as one of the between-subjects factor revealed significant improvements in FMA-m scores only in the rTMS group at T1 compared to T0 (4.2 [95% CI 0.9 to 7.5], P = .009), at T2 compared to T0 (8.4 [95% CI 4.0 to 12.9], P < .001), and at T2 compared to T1 [4.2 (95% CI 2.3 to 6.2), P < .001] (Table 3). However, there was no significant differences in FMA-m scores between the two groups at either T1 (12.7 [95% CI −7.3 to 32.7], P = .205) or T2 (15.7 [95% CI −4.6 to 36.0], P = .124) (Table 3).
Post Hoc Multiple Comparisons with Bonferroni Correction for FMA-m.

Values are presented as mean change (95% CI), P value.
Abbreviations: T0, baseline; T1, week 1; T2, week 2; rTMS, repetitive transcranial magnetic stimulation; S-rTMS, sham repetitive transcranial magnetic stimulation; FMA-m, Fugl-Meyer assessment scale-motor domain.
P < .05.
For MRMI change, the main effect of intervention was found to be significant (F = 9.827, P = .004). However, no significant interaction effect between intervention, time, and gender was observed. Additionally, the analysis revealed no interaction effect between time and gender (F = 2.663, P = .104) or between intervention and time (F = 1.183, P = .297) (Table 2).
There was no adverse event reported in either the rTMS group or the S-rTMS group. Four patients in the rTMS group did not complete the study between T1 and T2. One patient experienced a rib fracture, while three others exhibited exacerbated medical conditions. None of these events were attributed to rTMS. There was a significant difference in the attrition rate between the groups (P = .045).
Discussion
This study represents the first randomized controlled clinical trial investigating the efficacy of rTMS in patients with poststroke PS. The findings from this investigation suggest that rTMS does not confer superior benefits compared to sham stimulation in improving pushing behavior and mobility for patients with poststroke PS. However, the significant interaction effect between intervention and time on FMA-m scores, along with the significant improvements within the rTMS group and the exceeding MCID for between-groups comparison at T2 during post hoc analysis, suggests that rTMS might be effective to improve the motor function for patients with poststroke PS. Despite employing the multiple imputation method to address missing data, it remains uncertain whether the higher attrition rate observed in the rTMS group compared to the S-rTMS group may have introduce some bias in the results.
Few existing studies have shown that tDCS, another NINM technique, has a positive effect on improved posture and pushing behavior in patients with PS.12-15 Yamaguchi T et al. applied tDCS to the parietal area at an intensity of 2 mA for a duration of 20 minutes per day, over a period of 8 days. This intervention resulted in remarkable improvement of SCP and BLS, as well as an increase in static sitting time for a patient with PS. 15 Nakamura J et al. found that tDCS applied on the skin over bilateral mastoid resulted in significant improvements in SCP and BLS for two patients with PS, when applied 20 mintues per day, 5 days per week for 2 weeks. 12 In a study conducted by Babyar S et al. 13 , a 20-minutes session of tDCS at an intensity of 2 mA was applied to nine patients who were able to sit at least 25 minutes. 13 The results indicated that tDCS over bilateral parietal-insular vestibular cortex (PIVC) let to a small yet significant increase in the mean speed of the center of pressure, whereas anode placement over the ipsilesional PIVC or ipsilesional mastoid did not produce similar effects. 13 Another study by Babyar S et al. 14 demonstrated that a 15-minutes session of anodal tDCS at an intensity of 0 to 2 mA, targeting either the ipsilesional PIVC or ipsilesional mastoid, resulted in significant improvements in seated haptic center of pressure for ten patients with PS. 14
In this study, rTMS was not more beneficial than the sham intervention in reducing pushing behavior measured by BLS and SCP. This finding is not consistent with those of previous NINM studies due to variations in stimulation target selection, treatment duration and dosage between this study and these prior investigations. Furthermore, differences in study design and participant inclusion criteria may account for the divergent results observed here compared to those reported in these studies. Among these four existing studies, two employed a self-controlled design13,14, and two were case(s) studies12,15. Three studies included participants with PS diagnosed by BLS scores of two or higher12-14, and one study did not report the specific BLS scores. 15 In our study, we used a BLS cutoff score of ≥3 for diagnosing PS based on recent findings. This threshold indicates more severe balance disturbance and verticality perception impairment, and includes subjects more consistent with PS as assessed by the SCP. 39
The findings from a case series study suggest that tDCS applied over bilateral mastoid do not result in significant improvements in FMA-m of lower limbs or functional ambulation category for two patients with PS. 12 However, the significant interaction effect between intervention and time on FMA-m scores, along with the significant improvements within the rTMS group and the exceeding MCID for between-groups comparison at T2 during post hoc analysis in this study, suggests that rTMS might have positive effect to improve the motor function. It can be inferred that application of rTMS over nonlesional IPL, which is responsible for evaluating and integrating various sensory inputs, enhances sensory-motor function and consequently improves motor performance. The lack of a significant difference between groups in terms of FMA-m during post hoc multiple comparisons is likely due to the wide distribution of FMA-m scores and the small sample size. The contributing factor to the lack of significant difference in mobility between groups was the non-significant improvement in pushing behavior. Patients with PS face challenges in maintaining an upright posture, which is likely to impede their ability to achieve greater improvements in mobility.
The rTMS protocol used in this study has been shown to be effective for poststroke neglect.22,32,50,51 PS is very similar to poststroke neglect, which is a perceptual defect resulting from interhemispheric imbalance.52,53 Pérennou et al. suggested that a major component of PS is an implicit active body postural alignment with the perceived titled vertical. 54 Patients with PS experience a tilted perception of postural vertical which leads to tilted body posture and loss of lateral balance. 54 This tilted perception is probably attributed to interhemispheric imbalance, following the concept of stroke-induced dysbalanced interhemispheric interactions. 55 The parietal cortices are integral components of both interhemispheric and intrahemispheric frontal-parietal pathways. Consequently, damage to one parietal cortex results in disinhibition of the other parietal cortex leading to pathological overactivation of the latter. Based on this understanding, we hypothesized that the application of an inhibitory rTMS protocol over nonlesional IPL could potentially restore interhemispheric balance and normalize patients’ perception of postural vertical in cases of PS. However, our findings did not support this hypothesis. Several possible reasons can be considered. Firstly, there was a higher attrition rate observed in the rTMS group compared to the S-rTMS group. Secondly, some patients with PS might have spontaneous recovery during the three-month study period as suggested by Danells et al. 4 . Thirdly, it was possible that the intervention duration was too short to produce significant effect. Lastly, the sample size was insufficient to detect statistically significant differences between the groups.
This trial has some strengths. First, this is the first randomized controlled clinical trial to explore the efficacy of rTMS in patients with poststroke PS. Second, blinding of the patients and assessors could reduce bias and assess the role of rTMS in the treatment of PS with more certainty. However, the study has several limitations. First, the higher attrition rate observed in the rTMS group compared to the S-rTMS group might introduce bias to the results. Second, there was no objective measurement of the pushing behavior of the patients. Third, this trial has the limitation of not assessing the effect of 3 weeks of rTMS for PS as planned, as more than half of the participants were lost to follow-up due to the pandemic.
In conclusion, rTMS did not demonstrate significant efficacy in improving pushing behavior and mobility in patients with PS. However, rTMS might have potential effect in improving motor function for patients with PS. To further determine the clinical benefit of rTMS in patients with poststroke PS, future work should consider a larger sample size with more precise patient inclusion criteria (such as onset duration of stroke, diagnostic criteria for PS and severity of PS) and longer intervention duration.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241268537 – Supplemental material for rTMS for Poststroke Pusher Syndrome: A Randomized, Patient-Blinded Controlled Clinical Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683241268537 for rTMS for Poststroke Pusher Syndrome: A Randomized, Patient-Blinded Controlled Clinical Trial by Lijiao Meng, Yanlei Ge, Raymond C.C. Tsang, Wenyue Zhang, Xingyu Liu, Siyi Li, Jingyu Zhao, Xiaoyue Zhang and Qingchuan Wei in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683241268537 – Supplemental material for rTMS for Poststroke Pusher Syndrome: A Randomized, Patient-Blinded Controlled Clinical Trial
Supplemental material, sj-docx-2-nnr-10.1177_15459683241268537 for rTMS for Poststroke Pusher Syndrome: A Randomized, Patient-Blinded Controlled Clinical Trial by Lijiao Meng, Yanlei Ge, Raymond C.C. Tsang, Wenyue Zhang, Xingyu Liu, Siyi Li, Jingyu Zhao, Xiaoyue Zhang and Qingchuan Wei in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank all participants as well as the clinicians involved in this trial.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program (2020YFC2008500).
Ethics
Ethics approval was obtained from the Biomedical Ethics Committee of West China Hospital, Sichuan University (approval No. 2022-133) on March 23, 2022.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
