Abstract
Background. After stroke, deregulated interhemispheric interactions influence residual paretic hand function. Anodal or cathodal transcranial direct current stimulation (tDCS) can rebalance these abnormal interhemispheric interactions and improve motor function. Objective. We explored whether dual-hemisphere tDCS (dual-tDCS) in participants with chronic stroke can improve fine hand motor function in 2 important aspects: precision grip and dexterity. Methods. In all, 19 chronic hemiparetic individuals with mild to moderate impairment participated in a double-blind, randomized trial. During 2 separate cross-over sessions (real/sham), they performed 10 precision grip movements with a manipulandum and the Purdue Pegboard Test (PPT) before, during, immediately after, and 20 minutes after dual-tDCS applied simultaneously over the ipsilesional (anodal) and contralateral (cathodal) primary motor cortices. Results. The precision grip performed with the paretic hand improved significantly 20 minutes after dual-tDCS, with reduction of the grip force/load force ratio by 7% and in the preloading phase duration by 18% when compared with sham. The dexterity of the paretic hand started improving during dual-tDCS and culminated 20 minutes after the end of dual-tDCS (PPT score +38% vs +5% after sham). The maximal improvements in precision grip and dexterity were observed 20 minutes after dual-tDCS. These improvements correlated negatively with residual hand function quantified with ABILHAND. Conclusions. One bout of dual-tDCS improved the motor control of precision grip and digital dexterity beyond the time of stimulation. These results suggest that dual-tDCS should be tested in longer protocols for neurorehabilitation and with moderate to severely impaired patients. The precise timing of stimulation after stroke onset and associated training should be defined.
Keywords
Introduction
Stroke is one of the leading causes of long-term disability. According to the World Stroke Organization, only approximately 12% of stroke survivors achieve complete motor recovery after 6 months. 1 The majority of stroke patients present with hemiparesis, characterized by abnormal muscle activation and coordination in the paretic arm 2 and difficulties in strength control, 3 digital dexterity, 4 interjoint coordination, 5 and precision grip. 6 After a stroke, poor upper-limb motor recovery is coupled with a relative imbalance in interhemispheric excitability—namely, a downregulation of excitability in the ipsilesional primary motor cortex (M1) and/or an enhanced excitability in the contralesional M1. 7 Therefore, rebalancing the deregulated interhemispheric interactions is an appealing therapeutic option to improve motor recovery after a stroke. 8 Indeed, noninvasive brain stimulations, such as repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS), have been shown to rebalance interhemispheric excitability and improve motor function in stroke patients.9-12 Anodal tDCS applied to the ipsilesional M1 or cathodal tDCS applied to the contralesional M1 improve motor performance of the paretic hand. 13 When compared with rTMS applied to both hemispheres 14 or complex combinations of rTMS and tDCS on opposite hemispheres,11,15 dual-hemisphere tDCS (dual-tDCS) is particularly attractive for treating stroke patients because tDCS is a simple, safe, and inexpensive method to rebalance disturbed interhemispheric interactions and improve paretic hand function. The pioneer studies that demonstrated that tDCS improves hand motor performance in stroke patients focused on relatively crude or basic measurements of speed and errors in task execution or maximal force during voluntary contraction.11,13,16,17 On the other hand, rTMS or theta burst stimulation (TBS) have been shown to improve the precision grip dynamics and dexterity of the paretic hand.18-20 Currently, the potential of tDCS to improve precision grip or dexterity has not yet been explored.
The goal of the present study was to test the hypothesis that, compared with baseline, 2 important aspects of fine hand motor function relevant from an ecological point of view—the motor control of precision grip and digital dexterity—improved more after real than sham dual-tDCS in chronic stroke patients. We also explored the early time course of functional changes induced by dual-tDCS.
Methods
The study was approved by the local ethical committee and conducted according to the recommendations of the Helsinki Declaration. Written informed consent was obtained at enrollment.
Patients
A total of 19 chronic stroke patients were included in the study (Figure 1). The inclusion criteria were (1) being a chronic (>6 months) stroke patient aged 18 to 80 years, (2) with an initial motor deficit in the upper limb clinically evident during at least 1 week, and (3) having a hemispheric vascular brain lesion demonstrated by cerebral imaging. The exclusion criteria were the presence of (1) intracranial metal, (2) epilepsy, (3) alcoholism, (4) pregnancy, (5) cognitive impairment or psychiatric disorder, and (6) being unable to perform the task or understand the instructions. In all, 17 patients had an ischemic stroke. Of these, patients 12 and 16 had a secondary hemorrhagic transformation, and patients 4 and 8 had an intracerebral hemorrhage. Also, 11 patients presented with subcortical stroke (Figure 2). The degree of overall disability was quantified with the modified Rankin Scale (mRS), 21 and manual ability was quantified with the ABILHAND Scale22,23 (Table 1). ABILHAND is a measure of manual ability that has been defined as the capacity to manage daily activities requiring the use of upper limbs, whatever the strategies involved. 22 During a structured interview, the patients scored the 23 ABILHAND items as “impossible,” “difficult,” or “easy.” The ABILHAND items refer to bimanual tasks such as fastening the zipper of a jacket, tearing open a pack of chips, cutting meat, or hammering a nail. Patients’ responses were classified according to the published calibration, ranging approximately from −3.5 to 6 logits, where smaller logits (−3.5) are associated with a self-perceived greatest difficulty to perform the task. 22 The digital dexterity impairment was also quantified by the mean score of 3 trials with each hand on the Purdue Pegboard Test (PPT)24,25 (Table 1). Unfortunately, the National Institutes of Health Stroke Scale (NIHSS) 26 score was not available for a majority of patients. Nevertheless, all presented upper-limb motor deficits, such as hand weakness and/or disorders in fine alternating movements or dysmetria, as explicitly reported in all the medical records. At the time of inclusion, all the stroke patients were able to perform a reaching movement and a precision grip between the thumb and index finger.
CONSORT flow diagram. Abbreviations: tDCS, transcranial direct current stimulation; dual tDCS, dual-hemisphere tDCS. Allocation/randomization method: a first experimenter established an inclusion list with the codes for real and sham dual-tDCS in a pseudorandomized and balanced order. These codes were used by a second experimenter to apply dual-tDCS in a double-blind fashion.
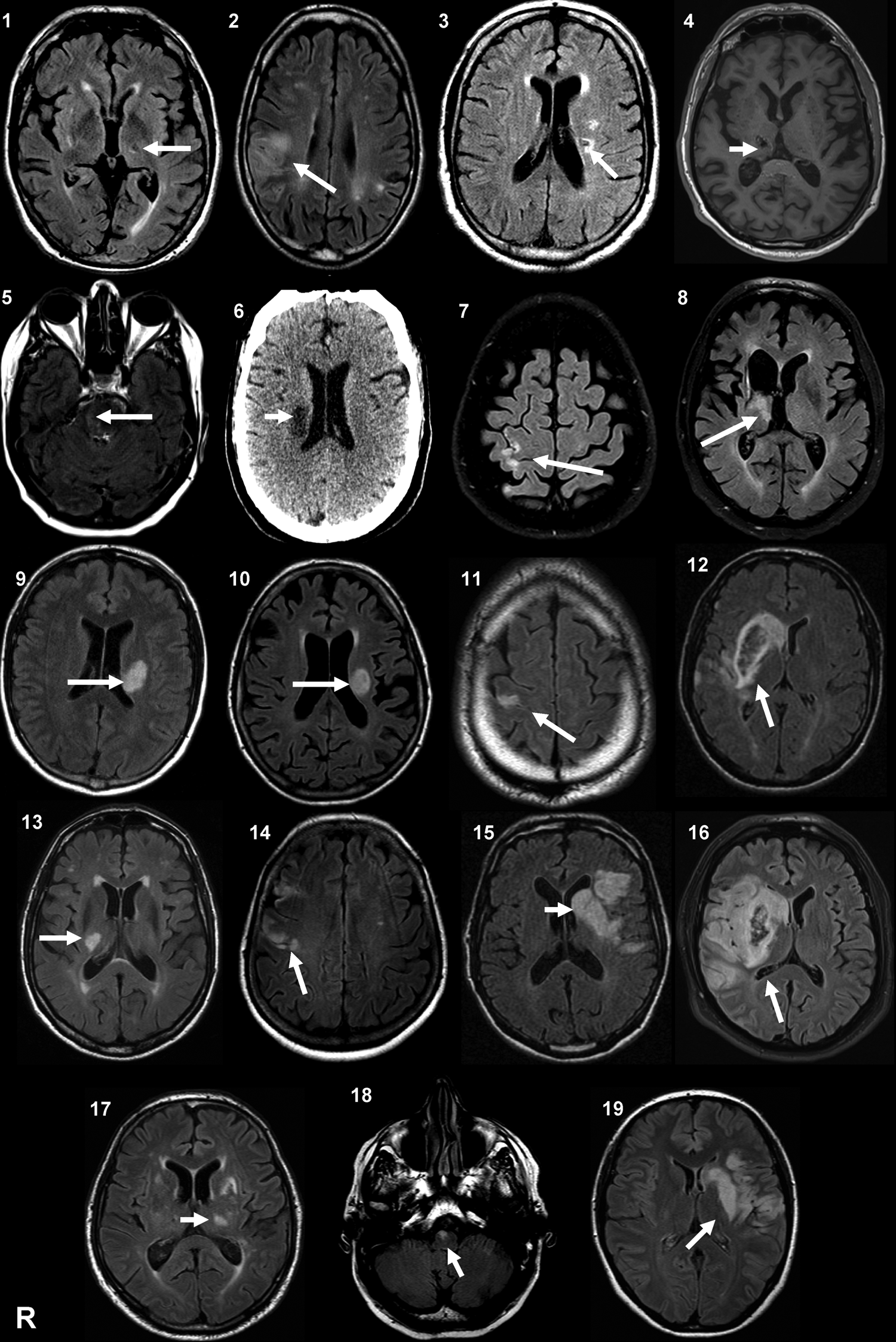
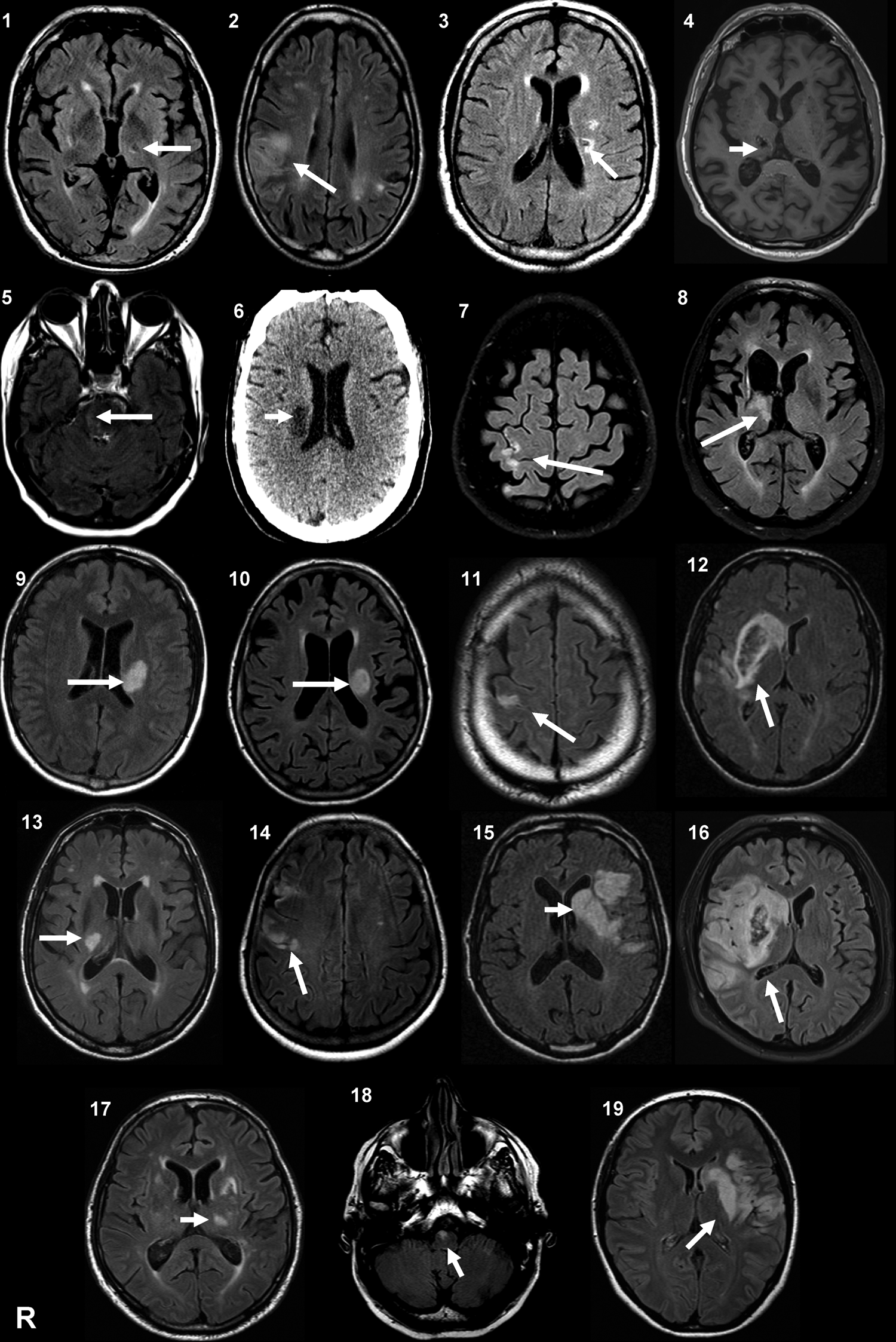
Brain imaging of the stroke patients. MRI or CT (patient 6) at the level of the main stroke injury; T2-weighted FLAIR except for patient 4 (T1). Patients 4 and 8 had an intracerebral hemorrhage. There was a slight secondary hemorrhagic transformation in patients 12 and 16. Patients 2, 14, and 19 had at least 1 other lesion compatible with a previous, minor stroke. Patients 8, 11, 13, and 17 had associated leukoaraiosis. Patients 1, 2, 11, 13, and 18 had some small chronic subcortical infarcts. Patients 3, 4, and 18 had some small chronic subcortical lacunar infarcts.
Baseline Characteristics of the Chronic Stroke Patients. a
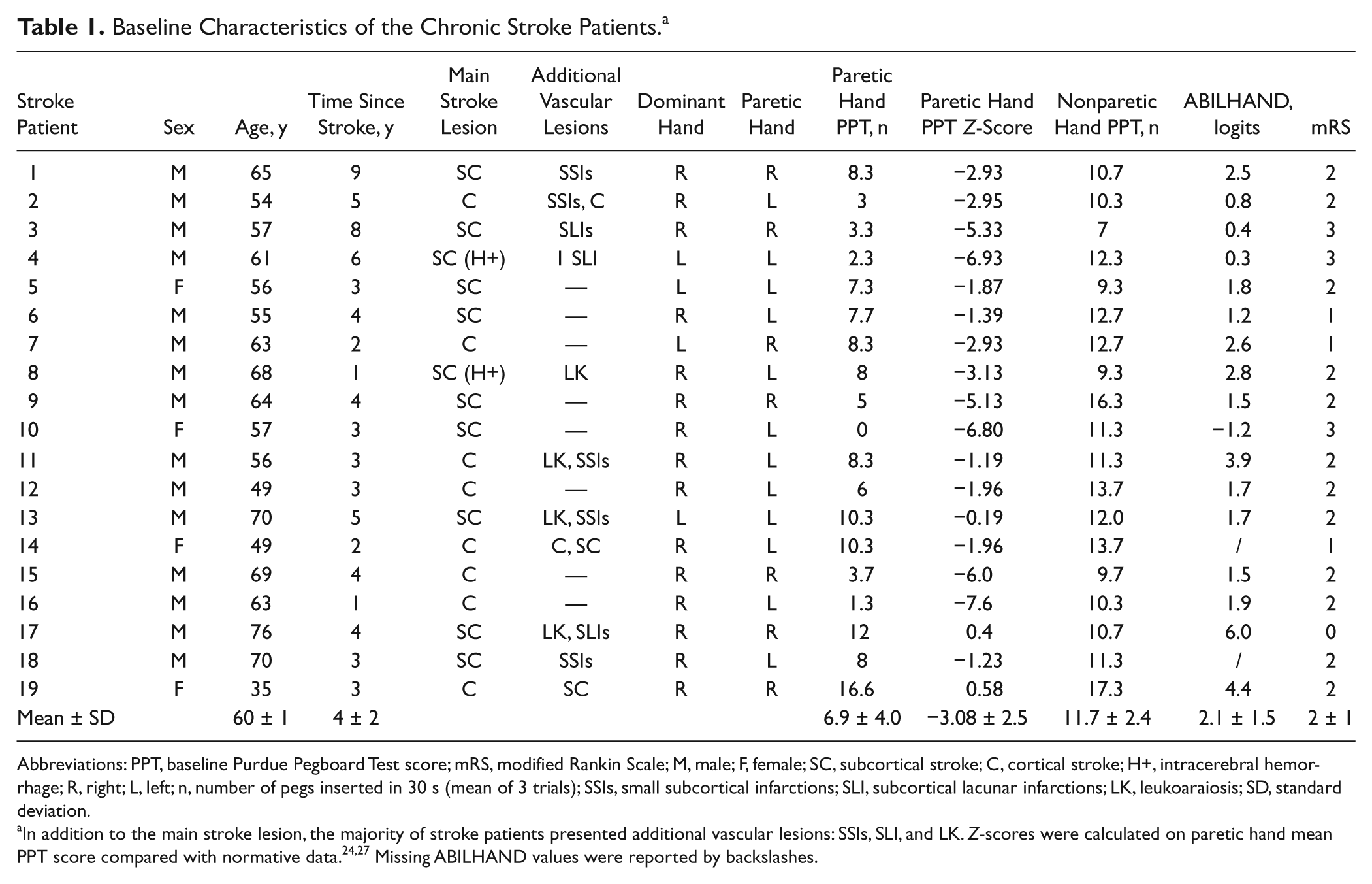
Abbreviations: PPT, baseline Purdue Pegboard Test score; mRS, modified Rankin Scale; M, male; F, female; SC, subcortical stroke; C, cortical stroke; H+, intracerebral hemorrhage; R, right; L, left; n, number of pegs inserted in 30 s (mean of 3 trials); SSIs, small subcortical infarctions; SLI, subcortical lacunar infarctions; LK, leukoaraiosis; SD, standard deviation.
Design
The patients participated in a randomized controlled trial consisting of 2 sessions (real/sham dual-tDCS) performed at least 1 week apart in a balanced order (inclusion list), with a double-blind, placebo-controlled experimental design.
During familiarization, each patient performed 10 grip-lift movements with the paretic hand to allow for fast adaptation/learning effects. 28 Each session was divided into 4 evaluation periods—prior to tDCS (“baseline”), during tDCS (“during”), immediately after tDCS (“after”), and 20 minutes after tDCS (“after 20 minutes”)—during which the patients performed 10 grip-lifts with the paretic hand and the PPT, 3 times with each hand.
Intervention
Dual-tDCS was delivered by an Eldith DC-Stimulator (NeuroConn, Ilmenau, Germany). The electrodes (35 cm2) were soaked in 0.9% NaCl. A Magstim 2002 (Magstim Company, UK) with a figure-of-eight coil was used to determine the hot spot eliciting consistent movements in the contralateral hand. The anode was positioned over the ipsilesional M1, and the cathode was placed over the contralesional M1. During real dual-tDCS, the stimulator delivered 20 minutes of stimulation at 1 mA (fade-in/-out 8 s). During sham dual-tDCS, a short up-ramp (8 s fade-in) was followed by 40 s of direct current and 8 s of fade-out, after which ineffective current pulses (110 µA over 15 ms, peak current 3 ms) were delivered every 550 ms. The first experimenter established an inclusion list with the Eldith codes (real/sham) for each session. These codes were used in a double-blind fashion by the second experimenter.
Hand Function Assessment
The primary outcome measures were the preloading phase duration (PLD) and grip force/load force ratio (GFL/LFL) for the precision grip—2 variables that are typically impaired in chronic stroke patients6,29,30—and the PPT score for quantifying digital dexterity.31,32
To measure the forces (perpendicular: left and right GFs, averaged as the global GF; and tangential: the LF) during the grip-lift task, a manufactured manipulandum fitted with 3 strain gauges (force transducers) and weighing 275 g was used (GLM Arsalis, Louvain-la-Neuve, Belgium). Analog signals were amplified, filtered with a Bessel 150-Hz cutoff low-pass fourth-order filter, and sampled at 2000 Hz. Data were analyzed offline.
During the grip-lift task, the patients were seated with their hand resting on a desk. The manipulandum was placed in front of them. The patients were asked to apply the minimal forces necessary to grasp the manipulandum between the thumb and index finger, lift it 20 cm above the desk, hold it stationary for 3 s, and then replace it.
Temporal parameters of grip-lift movements were assessed by measuring the durations of 3 periods 28 : (1) the PLD, the delay between the onset of GF and the onset of LF; (2) the loading phase duration (LD), the delay during which both GF and LF increased until LF equaled the weight of the manipulandum (2.75 N); and (3) the unloading phase duration (ULD), when LF dropped below the manipulandum’s weight until the end of the movement. Dynamical parameters were assessed by measuring the maximum downward force applied during the preloading phase and the efficiency of GF scaling relative to the load induced by raising the manipulandum. The latter was computed as the ratio between GF and LF (GFL/LFL) at the end of the lift. The coordination between LF and GF was quantified by a cross-correlation function between the first derivative of LF and GF: dLF/dt (LF rate) and dGF/dt (GF rate). 33 This cross-correlation function was computed for the loading period. The cross-correlation was characterized by a time shift (TS1) quantifying the delay to obtain the best possible overlap between the dGF/dt and dLF/dt curves with 0.5-ms steps and a correlation coefficient (R) that reflected the strength of this correlation.
To assess digital dexterity with the PPT, the patients had to pick up as many pegs as possible one by one and insert them into the holes of a board in 30 s.24,31 The PPT score was the mean number of pegs placed in the holes during 3 trials for each hand 24 (Table 1).
Statistical Analysis
The analysis of grip-lift parameters was performed using regression of repeated measures with generalized estimating equations to consider the multiplicity of intercorrelated values in each patient. 34 This analysis was used to evaluate the impact of “stimulation” (real/sham) and “time” (“baseline,” “during,” “after,” and “after 20 minutes”) for each grip-lift parameter. Repeated-measures analysis of variance (RM-ANOVA) was used to explore the effects of stimulation and time on the mean PPT scores. For pairwise post hoc comparisons, t tests corrected for multiple comparisons (Bonferroni) were computed between each period and baseline and separately between sham and real dual-tDCS for the “after 20 minutes” period.
PPT score improvement at the period “after 20 minutes” with real dual-tDCS was correlated with age, mRS, and ABILHAND by Pearson coefficient. The PPT score improvement was compared according to the localization of the lesion and whether the paretic hand was dominant or not, using the Student t test. A P value of .05 was considered statistically significant. All the statistical tests were two-tailed. Statistical analyses were performed using SPSS 15.0 (SPSS Inc, Chicago, Illinois).
Results
Precision Grip
A significant interaction between Time and Stimulation was observed for 3 parameters with the generalized estimating equations analysis (Figure 3): the PLD (P < .001), the GFL/LFL (P = .009), and the ULD (P < .001), suggesting that real dual-tDCS led to greater improvements than sham over time. At baseline, there were no statistically significant differences between real and sham dual-tDCS for any of the precision grip parameters. For the “after 20 minutes” period, only real dual-tDCS significantly improved the PLD from “baseline” (real: −86 ms, P = .023, −18%; sham: +27 ms, P = .4, +6%) as well as the GFL/LFL (real: −0.29, P = .014, −7%; sham: =0.04, P = .9, +1%). The ULD was significantly improved from “baseline” only with real dual-tDCS both during the “after” period (real: −37 ms, P < .001, −4%; sham: +72 ms, P = .5, +7%) and the “after 20 minutes” period (real: −22 ms, P = .004, −2%; sham: +10 ms, P = .8, +1%). In addition, there was no Time × Stimulation interaction for TS1, but the TS1 showed significant improvement from “baseline” to “after 20 minutes” with both real (+19 ms, P = .007, +63%) and sham (+20 ms, P = .039, +70%) dual-tDCS. Finally, for the “after 20 minute” period, comparisons between sham and real dual-tDCS showed a significant improvement for PLD (P = .036; effect size = 0.4) and only a nonstatistically significant trend for ULD (P = .1) and GFL/LFL (P = .1).
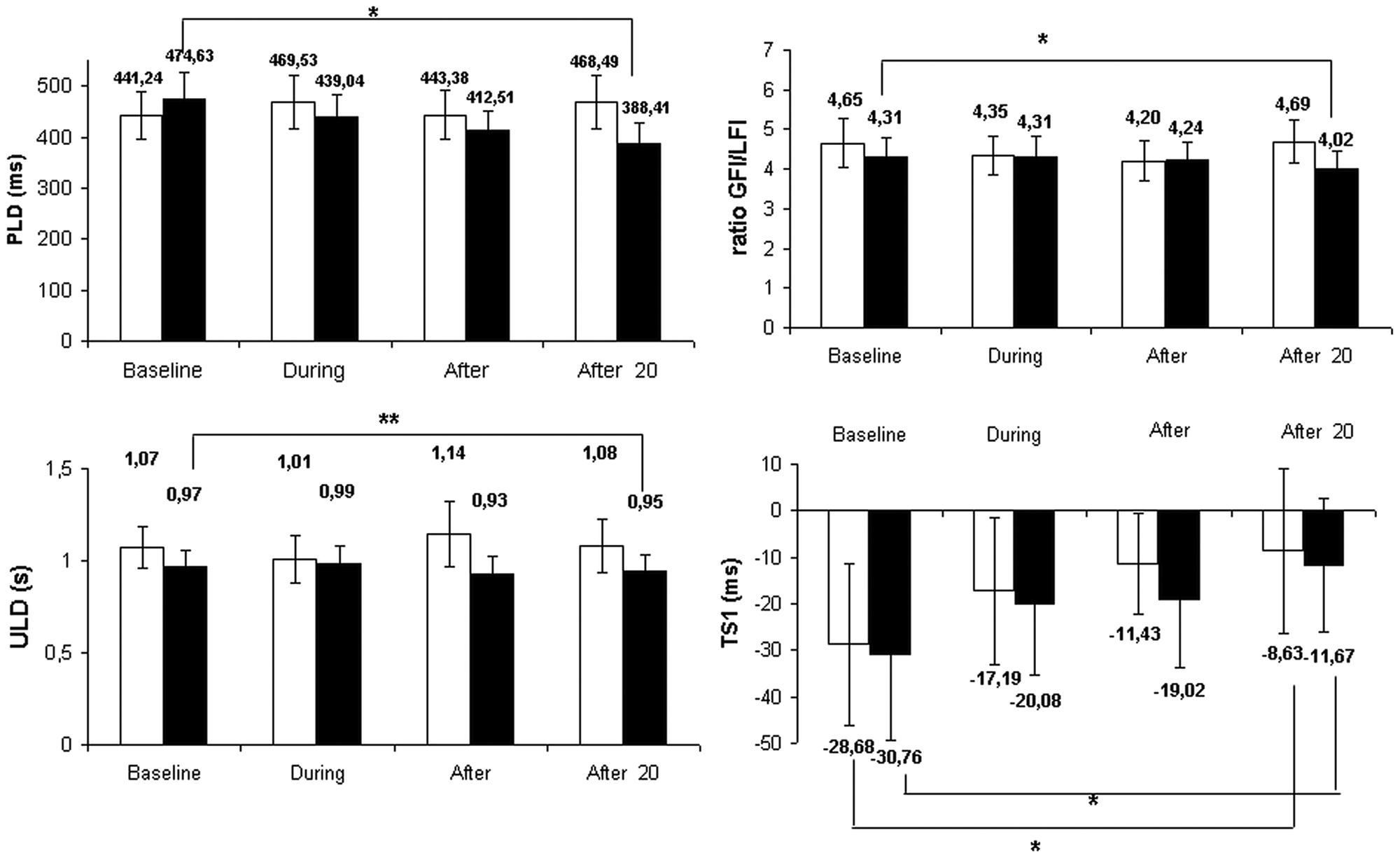
Precision grip parameters under sham and real dual-tDCS: changes at the group level (mean ± SEM) under real (black bars) and sham (white bars) dual-tDCS over the 4 evaluation periods (baseline, during, after, and after 20 minutes).
Digital Dexterity
For the paretic hand, there was continuous improvement over time only with real dual-tDCS, and the maximal improvement was observed after 20 minutes (+2.1 pegs in 30 s, +38% after real dual-tDCS vs +0.4 pegs in 30 s, 5% after sham; Figure 4). The RM-ANOVA demonstrated a significant interaction between Time and Stimulation (P < .001), suggesting that real dual-tDCS led to greater improvements than sham over time. Post hoc analyses confirmed that there was no significant performance improvement with sham dual-tDCS over time, whereas there were statistically significant performance improvements with real dual-tDCS between “baseline” and “during” (P = .003), “baseline” and “after” (P < .001), and “baseline” and “after 20 minutes” (maximal improvement: +2.1 pegs in 30 s, +38%, P < .001). There was a statistically significant difference between real and sham dual-tDCS exclusively for the “after 20 minutes” period (P < .001; effect size = 0.3).
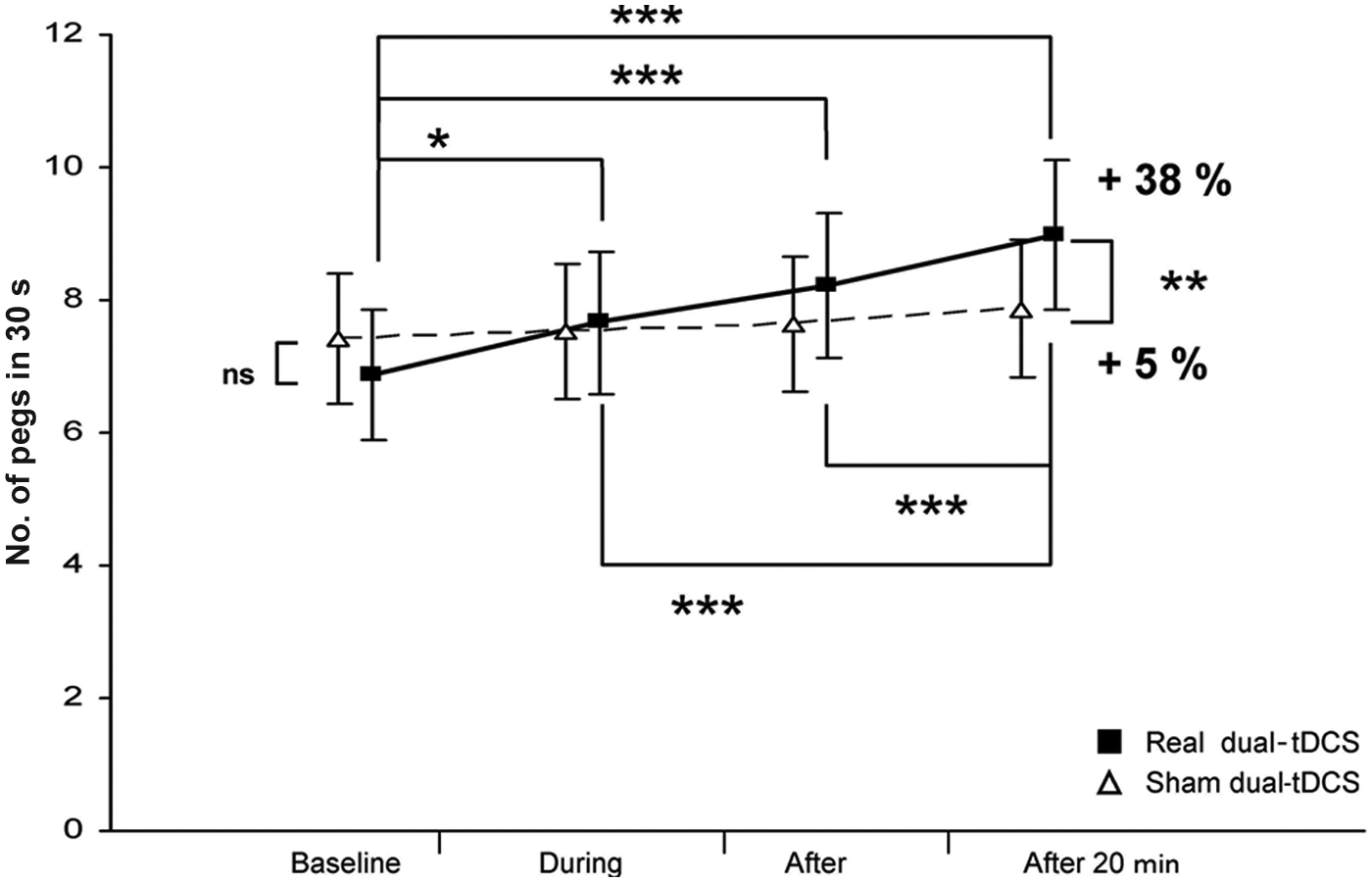
Purdue Pegboard Test (PPT) scores of the paretic hand under sham and real dual-tDCS: changes in the PPT score (mean ± SEM) for the paretic hand under real (black squares) and sham (white triangles) dual-tDCS over the 4 evaluation periods (baseline, during, after, and after 20 minutes). Abbreviations: dual-tDCS, dual-hemisphere transcranial direct current stimulation; SEM, standard error of the mean; NS, not statistically significant. *P < .05; **P < .005; ***P < .0005.
For the nonparetic hand (Figure 5), RM-ANOVA showed that there was only a significant effect of the factor Time (P < .001) and no effect of Stimulation (P = .9) and interaction between Time and Stimulation (P = .3). These results suggest a progressive performance improvement regardless of the stimulation type (real/sham), with a maximal improvement after 20 minutes (real: +0.8 pegs in 30 s,+6%; sham: +1.3 pegs in 30 s, +11%).
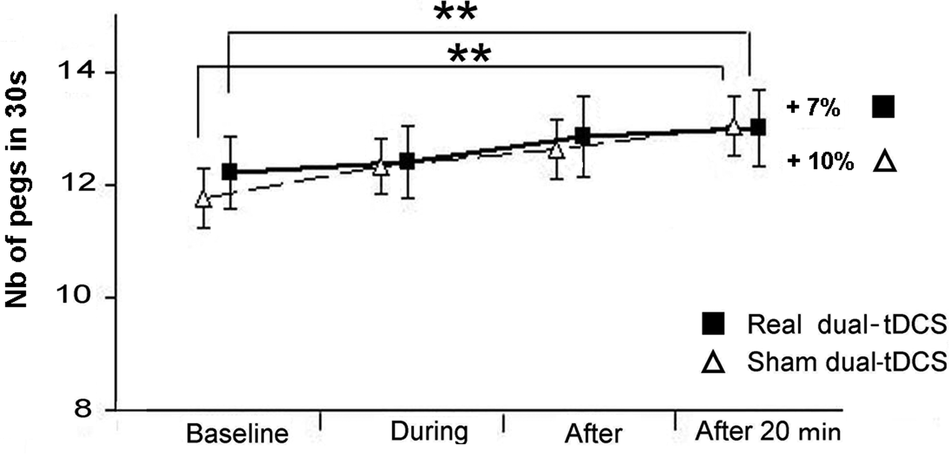
Purdue Pegboard Test (PPT) scores of the nonparetic hand under sham and real dual-tDCS: changes in the PPT score (mean ± SEM) for the nonparetic hand under real (black squares) and sham (white triangles) dual-tDCS over the 4 evaluation periods (baseline, during, after, and after 20 minutes). Abbreviations: dual-tDCS, dual-hemisphere transcranial direct current stimulation; SEM, standard error of the mean. *P < .05; **P < .005.
Correlation Analyses
Correlations were performed to determine whether baseline clinical characteristics could predict the individual percentage of improvement at the period “after 20 minutes” with dual-tDCS. The patient’s age, localization of the lesion (cortical/subcortical), whether the paretic hand was dominant or not, and the mRS did not significantly correlate with the PPT score improvement (P = .87, P = .60, P = .4, and P = .2, respectively). In contrast, the ABILHAND score significantly correlated with the PPT improvement under dual-tDCS (r = −0.54; P = .025).
Discussion
The main finding of this study is that in chronically and mild to moderately impaired persons after stroke, 20 minutes of dual-tDCS induced a large, rapid, and protracted improvement in performance with the paretic hand relative to baseline on a complex digital dexterity task (PPT) and a smaller, delayed improvement on the dynamics of precision grip after real dual-tDCS, compared with sham.
Dual-tDCS Improves Precision Grip and Dexterity With the Paretic Hand
Stroke has devastating effects on the precision grip ranging from eradication to typical impairments such as a prolonged PLD, an excessive GF leading to an abnormal GF/LF ratio, an abnormal time shift between the GF and LF increases, or an excessive preload force.4,6,35-37 The precision grip impairments that we observed in stroke patients were in line with those reported in previous articles.18,28,36
Real but not sham dual-tDCS improved the GF/LF ratio of the paretic hand compared with baseline (−7%, ie, a decrement of excessive GF, versus sham dual-tDCS +1%; effect size = 0.3), which may reflect a finer control of the GFs, a better processing of the somatosensory feedback, or a more accurate planning. 38 By comparison, inhibitory rTMS over the contralesional M1 led to a larger improvement in the GF/LF ratio (−30%, vs sham rTMS −4%; effect size = 0.8, n = 12) in patients with acute subcortical stroke. 18 Compared with sham, real dual-tDCS also improved the rapidity of execution of the precision grip task by diminishing the PLD (−18%, vs sham dual-tDCS +6%; effect size = 0.4) compared with baseline; inhibitory TBS over the contralesional M1 also led to a similar significant diminution of the PLD (−20%, vs sham TBS +12%; effect size = 0.5, n = 16). 19 Whereas the unload period 28 has received little attention in previous studies, dual-tDCS induced a small but statistically significant shortening of the unload period duration (−2%). Finally, the improvement of the time shift relative to baseline during both real and sham dual-tDCS may suggest a nonspecific training effect. The surprising finding was that in contrast to rTMS studies18,19,39 and other tDCS studies,13,17 the improvements were not observed during or just after dual-DCS but 20 minutes after the end of the stimulation period. This delayed improvement will be discussed below.
Dexterity is another aspect of fine sensorimotor function that may be severely impaired by stroke.4,6,35,36,40 In the current experiment, unlike sham, real dual-tDCS induced a strong and rapid “online” improvement in digital dexterity of the paretic hand relative to baseline.24,31,41 Strikingly, the maximal improvement was protracted and observed 20 minutes after the termination of dual-tDCS. The magnitude of this improvement was +38% (vs +5% after sham; effect size = 0.3). By comparison, classical tDCS in stroke patients led to a 10% improvement on the Jebsen Taylor Test (vs 2% deterioration after sham; effect size = 1.6, n = 6) 41 and to 6% improvement on simple reaction time (vs 5% deterioration after sham; effect size = 1.7; n = 11). 17 In another study with stroke patients, rTMS improved the PPT score by 33% (vs 5% after sham; effect size = 0.8, n = 10). 32
In the current study, dual-tDCS induced various improvements in fine motor functions of the paretic hand, some improvements were equivalent (PLD) and others weaker (GF/LF ratio and PPT) compared with what was found in previous studies using classical tDCS or rTMS. It is worth noting that the differences may arise from the heterogeneity in stroke populations, stimulation paradigms, and outcome measures. To demonstrate a superiority of tDCS over rTMS for improving hand motor function in stroke patients or vice versa, a formal comparison has to be carried out with a randomized trial.
Temporal Dynamic of the Improvements Driven by Dual-tDCS
In previous studies with rTMS18,39 and tDCS,13,41 the improvements of the paretic upper limb functions were measured during or just after noninvasive brain stimulation; the temporal dynamic of these improvements after the end of stimulation has received little attention. Strikingly, in the current experiment, the dexterity of the paretic hand improved continuously over time and culminated 20 minutes after the end of dual-tDCS, coincidentally with the delayed improvement in precision grip. This may reflect a protracted/delayed effect of dual-tDCS on fine functions of the paretic hand that were previously overlooked by less-sensitive measures and tests.
Alternatively, dual-tDCS may have strongly improved a training-dependent effect, that is, motor skill learning, at least for the PPT. If this interpretation is correct, dual-tDCS could become an extremely efficient add-on therapy to boost neurorehabilitation because the amount of practice with the paretic hand, if any, was very small (50 precision grip trials, 12 PPT trials). According to the current results, the application of dual-tDCS during motor practice of complex tasks (precision grip and PPT) might place the motor system of chronic stroke patients in an optimal state for improving training-dependent performances, after a short break to avoid fatigue.
Differential Impact of Dual-tDCS on Precision Grip and Dexterity
Why did dual-tDCS induce a greater performance improvement on PPT (+38%) than on precision grip parameters (−18% at best)? The precision grip was relatively well recovered in the majority of our chronic stroke patients, and their residual performance level may have been too high to be sensitive to a single 20-minute session of 1-mA dual-tDCS; the delayed improvement may represent a warm-up effect driven by the combination of practice and dual-tDCS. Alternatively, the precisions grip’s dynamics may be less sensitive to dual-tDCS-driven performance improvement, given the fact that the grasping movements may have been performed thousands of times by the stroke patients in everyday life, whereas the PPT was more novel and challenging, leaving more room for improvement. Thus, performing the PPT may lead to a broader and stronger recruitment of the cortical areas devoted to attention, motor planning and control, and feedback processing than precision grip. In turn, this would both increase the natural afferent inputs toward M1 and lead to functional improvement under dual-tDCS. Indeed, one of the hypothesized mechanisms of action of tDCS is a modulation of the neuronal resting membrane potential, which tunes the receptiveness of the target cortical area to ongoing afferent inputs. 42 In other words, if dual-tDCS modifies the receptivity of the target areas (M1) to ongoing afferent inputs, then the large improvements in the PPT may be caused by an additive modulation through a potential increase of ongoing afferent inputs driven by the more challenging, novel PPT.
Relevance to Neurorehabilitation
The improvements in paretic hand performance were not at the expense of nonparetic hand function because the PPT scores with the nonparetic hand did not deteriorate but rather improved slightly over time. Even though the precision grip of the nonparetic hand has not been assessed, it would be surprising to observe a deterioration because inhibitory rTMS applied over the contralesional hemisphere did not induce a negative effect. 18 Thus, dual-tDCS does not seem to carry a risk of impairing the fine functions of the nonparetic hand, at least in stroke patients with characteristics similar to those involved in the current study.
As suggested by the correlation between the ABILHAND scores and the improvement on the PPT, dual-tDCS had a stronger impact in the more impaired stroke patients. Despite the fact that none of them had a very severe impairment of hand function, the present cohort closely matches the characteristics of stroke patients seen in real life (ie, multiple vascular lesions, different types of vascular injuries, and older age). These results are thus encouraging in the perspective of implementing dual-tDCS as a new tool in the neurorehabilitation of a broad range of stroke patients, with different lesion locations, natures, and extents.
Limitations of the Study
The current experiment has several limitations. First, the full temporal dynamic of dual-tDCS remains to be explored because the last measurements were performed 20 minutes after the end of stimulation. Second, the current experiment has been undertaken with the idea to modulate abnormal interhemispheric interactions in stroke patients on the basis of previous studies using unilateral noninvasive brain stimulations.7,8,10,11,13 Because no measure of cortical excitability with TMS has been performed in the current study, such experiments should be performed to explore the mechanisms of the improvements driven by dual-tDCS. Third, the sample of chronic stroke patients was heterogeneous. We think that this is both a weakness and strength because dual-tDCS seems to have a beneficial effect on fine motor function in patients with different forms of stroke and with an extensive lesion burden.
Conclusions
The current study is the first to demonstrate that dual-tDCS applied in chronic stroke patients improves the dynamic of precision grip and the digital dexterity of the paretic hand—2 important aspects of fine hand motor function. This improvement is independent of stroke type and does not cause deterioration of motor performance with the nonparetic hand.
Given the fact that tDCS may be easily implemented in clinical settings, is able to enhance fine motor function of the paretic hand in stroke patients, is painless, easy, and safe to use, tDCS is in the pole position for a successful bench-to-bedside translation. The full temporal dynamic of the improvements induced by dual-tDCS need to be established to ensure an optimal implementation of dual-tDCS in the neurorehabilitation of the paretic hand in stroke patients.
Footnotes
Acknowledgements
We thank the stroke patients who took part in this study, and we thank A. Findik for his help in screening and recruiting patients at the Cliniques Universitaires Saint-Luc (UCL, Brussels). We also thank Dr F. Evrard and J-M. Raymackers for referring patients. We are grateful to Professor E. Olivier for the use of his TMS laboratory at UCL Brussels (Institute of NeuroScience, IoNS). We also thank D. Dispa and Ph. Vincent for their help with the precision grip data analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work of YV was supported by the following grants: Fonds de la Recherche Scientifique Médicale (FRSM) 3.4.525.08.F in 2008, 2010, and 2012 and Université Catholique de Louvain (UCL) Fonds Spécial de Recherche (FSR) in 2008 and 2010. The work of SL was supported by UCL FSR grants in 2008 and 2010. The Eldith DC-Stimulator (NeuroConn, Ilmenau, Germany) and the Magstim 2002 (The Magstim Company Ltd, UK) were purchased thanks to the FRSM grants (3.4.525.08.F 2008 and 2010), the UCL FSR grants (2008 and 2010), and a grant from the Fondation Van Goethem-Brichant.
Authors’ Note
Trial Registration: NCT01503073 Noninvasive Brain Stimulation for Stroke (NIBSstroke).
