Abstract
Background. Physical therapy intervention is the primary treatment for gait ataxia and imbalance in individuals with cerebellar damage. Our aim was to determine if a home balance exercise program is feasible for improving locomotor and balance abilities in these individuals. Methods. A total of 14 patients with cerebellar ataxia participated in a 6-week individualized home-based balance exercise program and attended 5 testing sessions (2 pretraining, 1 midtraining, 1 posttraining, and 1 one-month follow-up visit). Pretraining, posttraining, and follow-up testing included a neurological assessment, clinical gait and balance tests, and laboratory assessments of balance and walking. Participants kept logs of the frequency and level of balance challenge during their training. Results. Walking speed improved across visits, as did stride length, percentage double-limb support time, Timed Up and Go (TUG), and Dynamic Gait Index. Post hoc comparisons in these measures revealed that significant rehabilitative improvements occurred over the 6-week training period, and all but TUG gains were retained 1 month later. There were no changes across the other measures for the group. Regression analysis indicated that improvements in walking speed were affected by the level of balance challenge but not by age, ataxia severity, proprioception, or duration of exercise. Conclusions. Improvement in locomotor performance in people with cerebellar ataxia was observable after a 6-week home balance exercise program. The exercise program must be designed to provide a significant challenge to the person’s balance.
Introduction
Individuals with cerebellar degenerative disease exhibit progressive worsening of motor coordination or ataxia. In walking, ataxia is evident by a shortened stride length, high step pattern, and decreased push-off and veering. 1 These gait deviations have been closely linked to the severity of the individual’s balance deficits. 2 These deficits lead to taxing and unsafe mobility. With no available disease-modifying pharmacological treatments for ataxia, 3 physical therapy intervention is the primary treatment option. 4 However, efforts to restore normal movement are complicated because individuals with cerebellar disease also have deficits in motor learning. 5 It has, therefore, been reasoned that these patients may get limited benefit from rehabilitation that relies on motor learning. 6
Despite these considerable challenges, recent evidence suggests that individuals with cerebellar disease may benefit from long-term motor training. Following an intensive, high-level 4-week training program, cerebellar patients showed the ability to improve motor skills and functional performance.4,7-9 These studies, however, involved supervised exercise in a clinical setting. Beyond these studies and some case studies, research concerning exercise and physical therapy for ataxia is severely lacking.10-15
Here we asked if a home-based balance exercise program could improve locomotor and balance abilities in individuals with degenerative cerebellar disease. Given that voluntary leg-coordination deficits play less of a role than balance deficits in gait ataxia, 2 the exercise program focused on static and dynamic balance activities rather than direct gait training. The exercises included balancing in both sitting and standing positions, which allows both highly functional individuals (able to stand on 1 leg) and more severely affected individuals (requiring hand support to stand) to use the program. Clinical and laboratory measures were used to assess changes in balance and gait over the course of training and 1 month later. We also examined the influence of age, ataxia severity, duration of exercise, and level of balance challenge on training-induced changes in walking speed.
Methods
Participants
Individuals with degenerative cerebellar disease participated in the study. All participants were able to stand and take steps, although 4 participants used walkers and 1 used a cane for walking. Cerebellar damage for all participants was confirmed by magnetic resonance imaging or a computer tomography scan. Results of genetic testing were obtained when available. Individuals who had a genetic disease that primarily involved the cerebellum (eg, spinocerebellar ataxia 6 [SCA6]) or had extracerebellar signs (eg, SCA2 and SCA3) were enrolled as long as their primary problem was ataxia. Clinically, all participants had evidence of at least mild ataxia (International Cooperative Ataxia Rating Scale [ICARS] score >5) 16 for more than 3 months. Participants were excluded if they had other neurological diseases (eg, Parkinson’s disease), cognitive impairment (eg, Mini-Mental State exam score < 22) or if they were medically unstable (eg, uncontrolled hypertension and unstable angina). All participants gave informed consent according to the human studies guidelines of the Johns Hopkins Institutional Review Board.
Of the 25 participants who were screened and who consented, 14 participated in the complete study, and only their data are included in the results. It was found that 4 participants were unable to ambulate sufficiently for laboratory measures to be determined; 3 participants discontinued the study because of burden of travel; 2 discontinued because of unrelated medical complications; and 2 participants did not meet the testing schedule and exercise compliance requirements.
Paradigm
All participants performed a home-based balance exercise program for 6 weeks and attended 5 testing sessions (2 pretraining, 1 midtraining, and 2 posttraining visits; Figure 1). Participants served as their own controls because we included 2 pretraining visits spaced 2 weeks apart. The exercise intervention was initiated at the second pretraining visit. A physical therapist contacted the participants weekly by e-mail or phone to answer any questions about the exercise protocol. One midtraining visit 3 weeks into the program was used for evaluating the program’s progression. Participants logged their exercise effort both in terms of frequency and level of balance challenge. Participants were retested when the training ended and again 4 weeks later to assess the longevity of the rehabilitation effects. Each testing session included a neurology assessment, clinical gait and balance tests, and laboratory assessment of balance and walking kinematics.
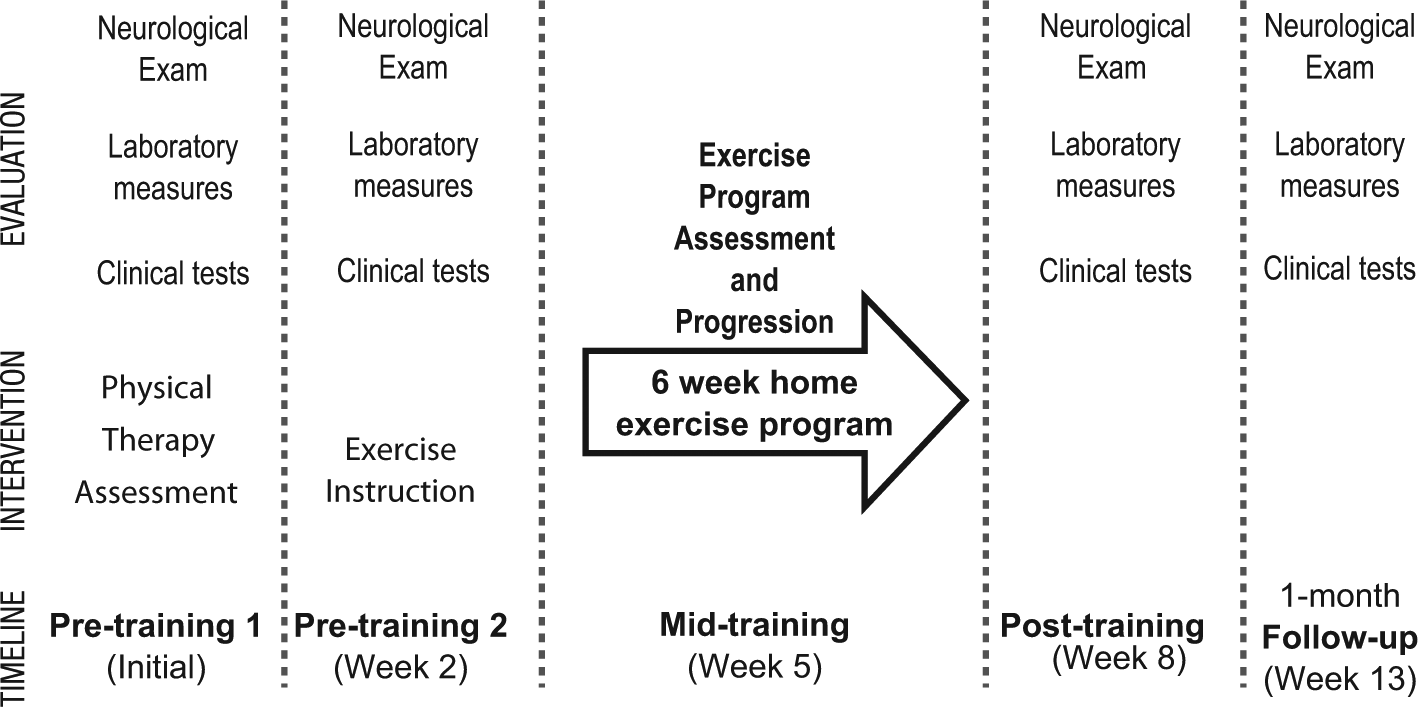
Study paradigm: All participants performed a home-based exercise program for 6 weeks. Two sets of pretraining measurements were taken 2 weeks apart to assess repeatability of our measures. One midtraining visit was used to reevaluate the program progression. Participants were retested after 6 weeks of training and, again, 4 weeks after the completion of training to assess retention.
Assessment Tools
Ataxia severity was measured using the ICARS. 16 The ICARS evaluates the degree of ataxia in 4 domains: posture and gait, limb kinetics, speech, and oculomotor. We also assessed fine touch in the great toe using Semmes Weinstein monofilaments and proprioception via clinical assessment of great toe motion. Spasticity and hyperreflexia at the ankle and knee were assessed and, if present, noted.
The following clinical gait and balance tests were performed according to established protocols: (1) Dynamic Gait Index (DGI), 17 (2) Timed Up and Go (TUG), 18 (3) Functional Reach Test (FR), 19 and (4) Activities Specific Balance Confidence Scale (ABC). 20
Static standing balance was assessed by measuring postural sway during two 20-s trials. Participants stood with arms crossed over their chest and feet shoulder-width apart under 2 conditions: eyes open and eyes closed. Data were collected at 1000 Hz using a Kistler 9281 force plate (Kistler Instrument AG, Winterthur, Switzerland). Balance deficits were calculated as the magnitude of postural sway (sway amplitude) using custom Matlab software (MathWorks Inc, Natick, MA).
For the walking assessment, participants walked as fast as possible on a 6-m runway 6 times, and we averaged the results.of 3-6 trials. Walking speed was our main outcome measure. We also collected marker position data from infrared emitting diodes placed bilaterally at the fifth metatarsal heads, lateral malleoli, lateral knee joint spaces, greater trochanters, pelvic crests, and acromiums at 100 Hz using a 3-dimensional Optotrak motion capture system (Northern Digital, Inc, Waterloo, ON, Canada). Custom Matlab software was used to calculate joint position and determine walking parameters. Stride length was calculated as the distance between initial foot contact of 1 foot and the next contact of the same foot. The percentage time in double-limb support was the time that both feet were in contact with the floor, expressed as a percentage of the stride time.
Intervention
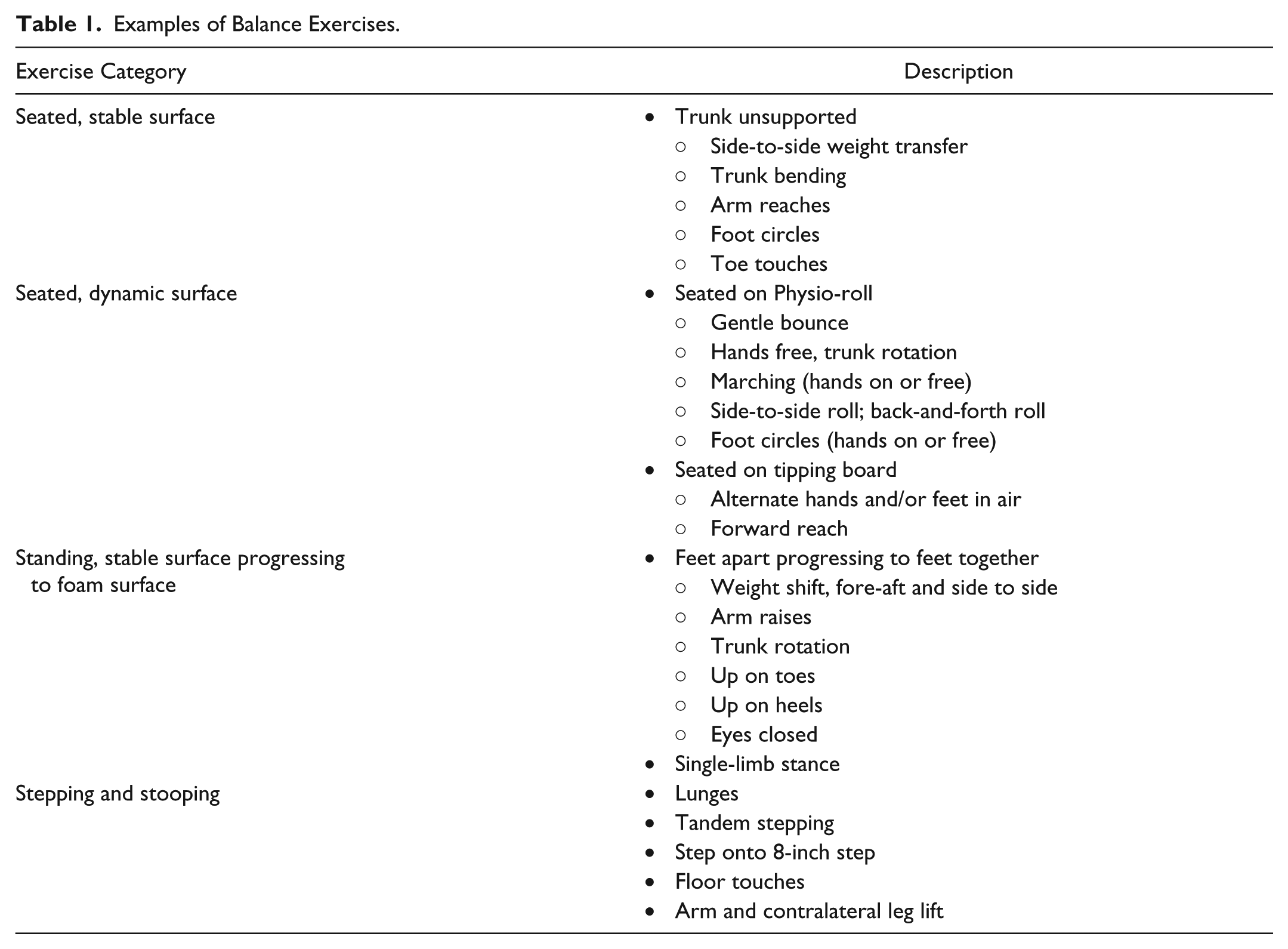
The 6-week home-based exercise program focused on balance training. Both static and dynamic exercises were done in sitting and standing positions. For each participant, a tailored home balance exercise program was developed by a physical therapist based on the testing results of the first pretraining visit. Because of the range of ataxia severity in the participants, sitting exercises were performed using 1 of 4 surfaces: standard chair, 6-inch dense foam, exercise ball (standard or peanut shaped), or balance disc. Similarly, the amount of upper-extremity support used for safety during the sitting or standing exercises varied from holding on to a stable object (ie, walker or chair) with 2 hands, to no support. Exercises started with stabilizing in a challenging static position and progressed to dynamic arm and leg movements in the same or modified position. Participants were given 6 study exercises designed to challenge their balance. Depending on the participant, an additional warm-up exercise was given to allow time to adjust to the equipment. Table 1 describes examples of the different types of exercises that were given. The categories and type of exercise were tailored to each participant’s abilities. Participants were instructed to perform the exercises for approximately 3 to 5 minutes each for a total minimum of 20 min/d, 4 to 6 d/wk. At the end of the 6-week program, participants were told that they were no longer required to do the exercises as part of the study.
Examples of Balance Exercises.
Participants were provided with instructions on how to perform the exercises using PhysioTools General Exercises 1st edition software (PhysioTools, Tampere, Finland), in-person demonstrations, and a DVD for home review. Participants acknowledged understanding of the exercise program through return demonstration initially and at the midtraining visit. General exercise guidelines, with an emphasis on safety, were reviewed and provided in a booklet. An exercise log was provided for entering dates and duration (minutes) of exercise. Additionally, participants were asked to rate their confidence with each exercise on a 100% scale, where 0% was no confidence at maintaining their balance during the task and 100% was complete confidence. The level of challenge of each exercise was defined as 100% minus the confidence rating. The average challenge rating of the 6 exercises on day 1 was used as an overall measure of the level of balance challenge for each participant.
Data Analysis
Intraclass correlation coefficients (ICCs) were calculated to determine repeatability of the measures taken during the 2 pretraining visits. ICC (1, 2) was used for the laboratory measures, which were averages of more than 1 trial, and ICC (1, 1) was used for the clinical 1-trial measures. 21 The averages of the measures from the 2 pretraining visits were used in the rest of the analyses. We used ANOVA to determine change across visits. Repeated-measures ANOVAs were used for laboratory data. Friedman’s ANOVA (nonparametric measures) was used for clinical rating scale data, which did not meet assumptions for parametric tests. Post hoc tests of Tukey HSD (honestly significant difference) or the Wilcoxon matched-pairs test were used to compare the change over the training period and retention of any benefits at 1 month. The coefficient of variation was determined for stride length data, and repeated-measures ANOVA was used to assess for change across visits. A forward stepwise regression was used to assess the extent to which walking speed change in the group was affected by age, ataxia severity, duration of exercise (in number of days performed), and level of balance challenge.
Results
Demographic data and exercise log results of the 14 participants who completed the study are shown in Table 2. Pretest assessment of the group showed that they had considerable ataxia, with an average overall ICARS score of 39 ± 15. The gait and posture ICARS subscore for the group was 15 ± 7. The clinical measures showed impairments in gait (DGI median = 14; range = 6-22) and balance (TUG = 15.2 ± 9.5 s; FR = 12.25 ± 3.25 inches) and decreased confidence with mobility (ABC median = 61%; range = 36%-85%). Proprioception was intact in the lower extremity as tested in the great toe for all but 4 participants (numbers 4, 11, 13, and 14). Light touch sensation was present for all but 3 participants (numbers 4, 7, and 11) who showed loss of protective sensation (light touch sensation impaired >10g force via 5.07 Semmes Weinstein monofilament). Reflexes were increased at the patella in 2 participants (numbers 4 and 9).
Participant Characteristics Pretraining and Exercise Log Summary.
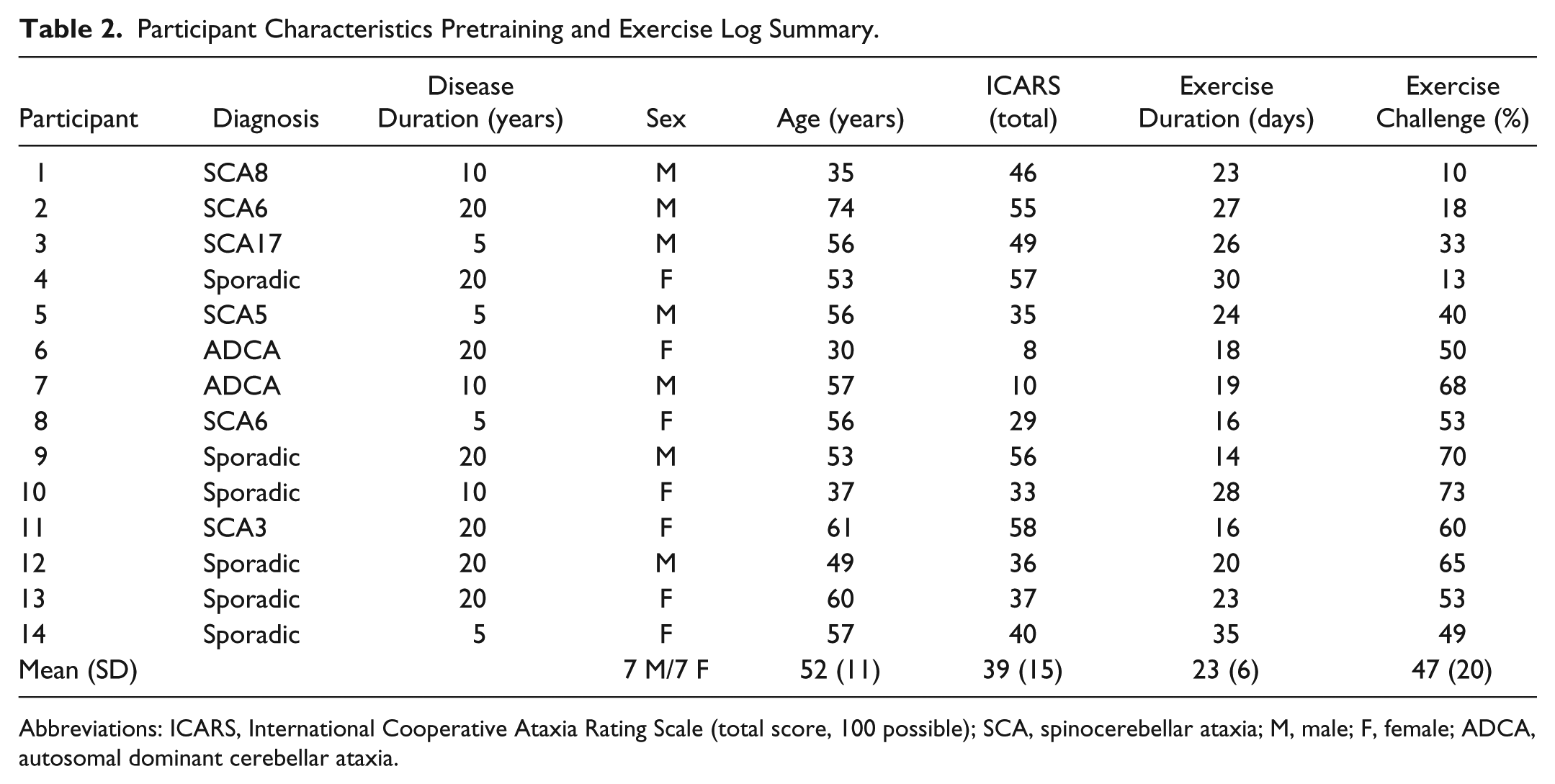
Abbreviations: ICARS, International Cooperative Ataxia Rating Scale (total score, 100 possible); SCA, spinocerebellar ataxia; M, male; F, female; ADCA, autosomal dominant cerebellar ataxia.
The pretest laboratory measures were as follows: walking speed at fastest possible pace was 0.9 ± 0.3 m/s; stride length was 1.08 ± 0.22 m; percentage stance time was 64 ± 4 %; and percentage double-support time was 14 ± 4 %. Postural sway amplitude in static standing (eyes open feet apart) was 10.4 ± 4.6 mm. Despite the inherent variability seen in the movement patterns of the participants, the ICCs for most of our measures was high: walking speed, 0.90; stride length, 0.84; percentage double-support, 0.72; static standing, 0.99; ICARS (total), 0.91; DGI, 0.72; TUG, 0.94; FR, 0.99; ABC, 1.00. Figure 2 depicts this strong relationship for walking speed.
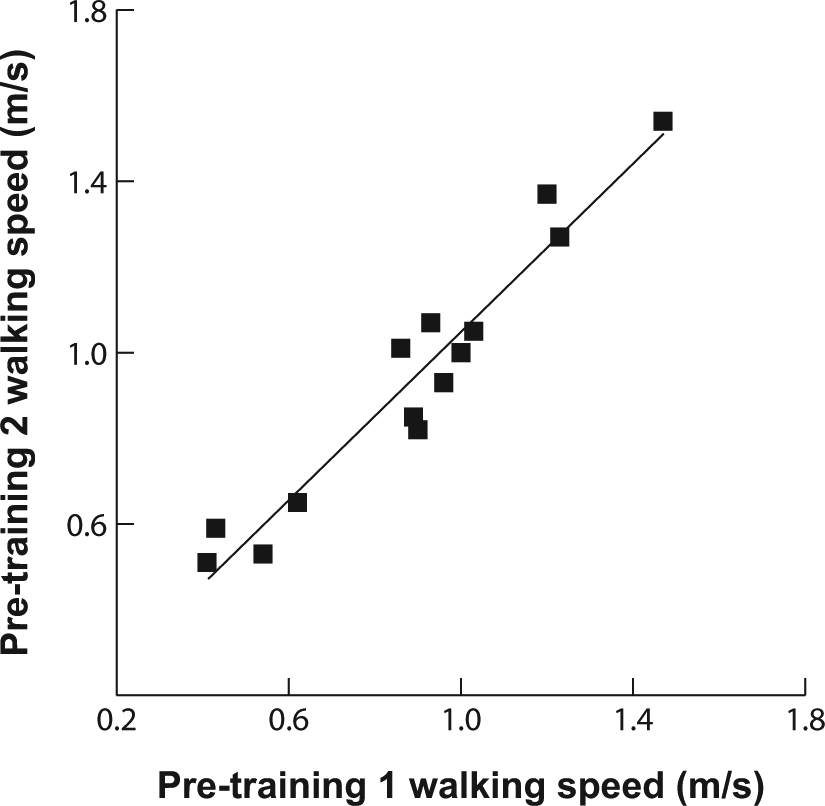
Walking speed reliability across the 2 pretraining sessions was high (intraclass correlation coefficient = 0.9). The scatter plot shows walking speeds for both pretraining visits for all participants.
Overall, participants were compliant with the exercise program, although a few reported difficulty in challenging their balance at home (Table 2). Of the 14 participants, 11 performed their individualized home program 3 or more days per week; 10 of the participants reported at least 40% balance challenge (ie, 60% confidence) with the exercises. Comparing ratings of the first and second halves of training, each participant’s challenge rating of their exercises was within 5% of the value listed in Table 2 except for 2 participants (number 12 increased 10%; number 13 decreased 25%). At 4 weeks after the training period (ie, the follow-up visit), 4 participants indicated that they had discontinued all balance exercises. Across the remaining 10 participants, a reduction in the frequency and duration of performance was reported. No participant reported falls while performing the exercises.
The group showed a significant improvement across pretraining, posttraining, and follow-up visits in both clinical measures and laboratory tests (Figure 3). Note that participant number 11 was unable to complete all clinical measures on the final visit because of illness and, therefore, was excluded from the overall clinical analysis. Of our clinical measures, ANOVA of TUG and DGI revealed improvements in gait and balance across visits (P = .02 and P < .001, respectively). Post hoc tests of these measures between pretraining and posttraining visits revealed that significant rehabilitation effects occurred in the 6-week training period (both P < .05, Figures 3A and 3B). Pretraining to follow-up visit comparisons showed that the effects were retained 1 month later in the DGI (P = .002) but not the TUG (P > .05). The FR, ABC, and ICARS scores did not change across visits for the group (all P > .05), although there were individual participants who improved in each of these measures.
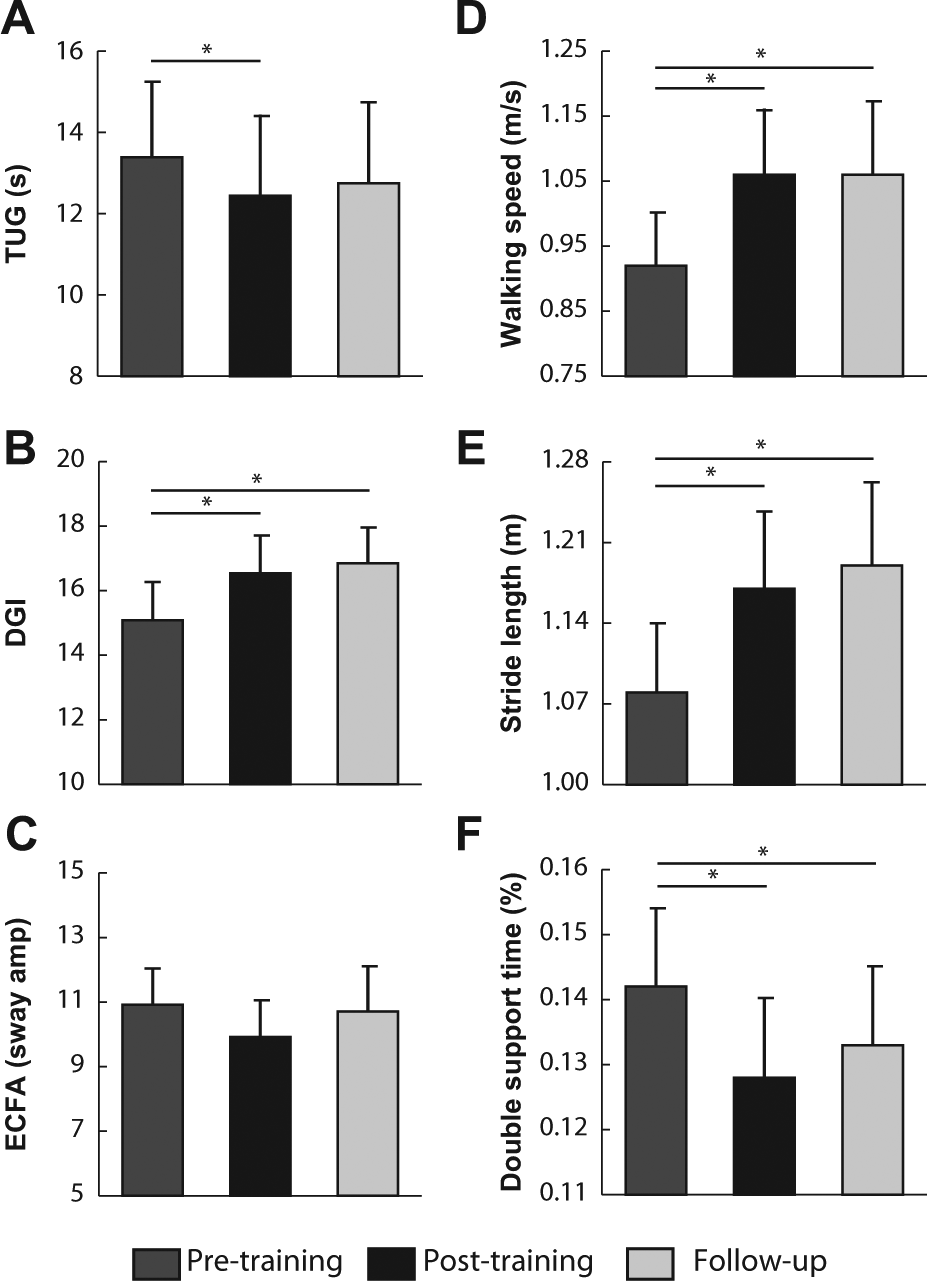
Results of training: selected results from the comparison across pretraining, posttraining, and follow-up visits. Clinical measures: Timed Up and Go (TUG; A) and Dynamic Gait Index (DGI; B). Static balance measure (C): sway amplitude for eyes closed feet apart (ECFA) condition (average of all participants except numbers 9, 11, 12, and 13, who were unable to perform the task). Walking parameters: walking speed (D), stride length (E), double-support time percentage (F). Average values ± 1 standard error indicated for all measures; asterisks indicate post hoc comparison for measures with significant differences (P < .05).
An ANOVA revealed an effect of visit for our primary outcome measure, average walking speed (P < .001), which increased 15% from pretraining to posttraining. This is nearly 3 times the minimally important difference reported for people with other neurological disorders.22,23 Post hoc comparison showed that the difference was significant between pretraining and posttraining, indicating that change occurred over the 6-week training period (Figure 3D, P < .001). The change was also significant from the pretraining to the follow-up visit, indicating that the improvement was retained (P < .001). Of the other walking parameters, ANOVA revealed that stride length and percentage double-support time also showed improvement across visits (P < .001 and P < .01, Figure 3E and 3F, respectively), whereas percentage stance time did not show significant change (P > .05). Note that participants 2 and 4 were not included in the percentage stance and percentage double-support analyses because of marker occlusion from their walkers. Postural sway amplitude did not change significantly across visits (P > .05). Results for postural sway amplitude for condition 2 (eyes closed) are shown in Figure 3C.
We also measured the coefficient of variation of stride length to assess stride-to-stride variability within each participant. Higher values of this measure indicate more variability. The pretraining group average was 9.2 ± 1.6; posttraining and follow-up averages were 6.1 ± 1.3 and 6.0 ± 1.1, respectively. Repeated-measures ANOVA showed a trend toward less variability after training (P = .10).
Regression analysis indicated that the change in group walking speed was affected only by the level of balance challenge (Figure 4) and not by age, ICARS gait and posture subscore, proprioception, or duration of exercise. We used the ICARS gait and posture subscore in this regression because it correlated with pretraining walking speed and was thus a good indicator of walking impairment.
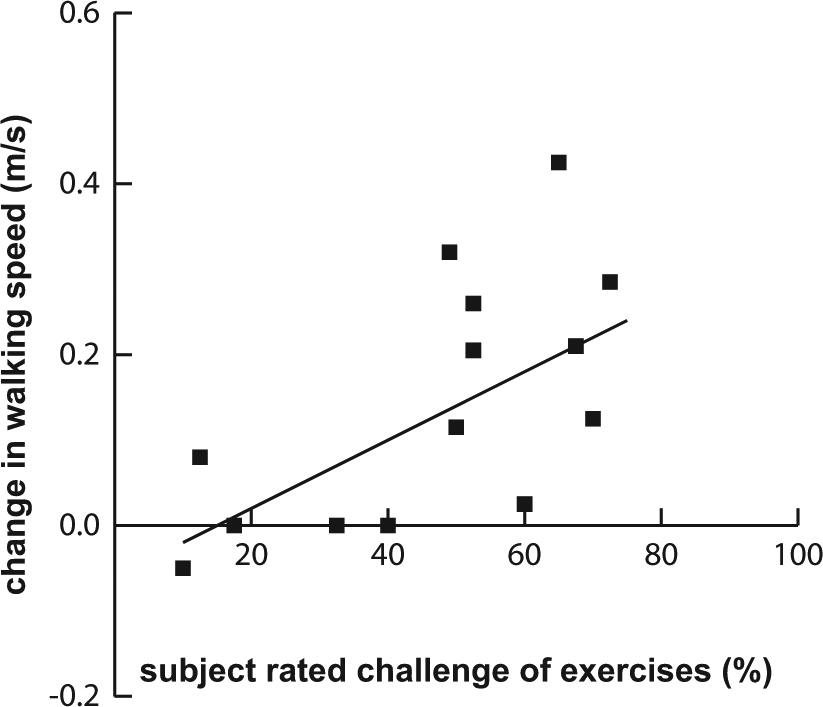
Effect of degree of balance challenge: scatter plot showing the correlation between change in walking speed (WS) and self-rated challenge of exercises. Regression equation: ΔWS = −0.06 + 0.004 × Challenge; R2 = 0.43; P = .01.
Discussion
We found that cerebellar patients with ataxia can benefit from a home exercise program focused on balance training. Despite only including 14 patients, we found statistically and clinically significant improvements after just 6 weeks. An important finding is that the level of challenge to balance was more important than the duration of exercise in producing positive effects. Therefore, individualizing the program and offering continued training and progression might be necessary to effect retention. This study provides valuable new insight because very few previous studies have looked at exercise programs for cerebellar ataxia and in particular home programs with limited one-on-one practice with a therapist.3,7,24 In addition to the well-known benefits that come from improving walking parameters, the advantages of the observed effects in this study may include increased sense of control and independence and improved quality of life. The home program may also provide a means to optimize the use of financial and health care resources in the face of the often functionally limiting progressive disease of degenerative cerebellar ataxia. 25
The pretraining tests indicated that although our group of patients was ambulatory, they were severely affected relative to individuals with ataxia who have been previously studied: the average fast walking speed of 0.9 m/s is comparable to self-selected walking speeds reported in other studies of individuals with degenerative cerebellar disease. 4 Although more severe ataxia results in greater variability of movement, we were able to get stable pretraining measurements, which suggests that our participants could validly serve as their own controls.
The exercise intervention was individualized to optimize challenge and ensure safety. Sitting balance exercises were an important component of the program because the level of challenge could be varied by varying the support surface. Standing exercises were often limited to dynamic arm and trunk movements performed from a stable starting point. For most participants, balance tasks during walking were too challenging to be done safely without assistance. Direct instruction in the program was limited to the second pretraining visit, with reassessment and progression at midtraining (week 3 of 6). Even with the limited training, the group demonstrated significant improvement in both clinical and laboratory measures after 6 weeks.
Walking improved significantly for the group across the pretraining, posttraining, and follow-up visits. A significant increase in fast walking speed was observed after training and was retained 1 month later. This is an important result because walking speed has been shown to reflect overall functional independence and good quality of life. Two other locomotor features were improved: stride length increased and percentage double-support time decreased. Thus, participants could take longer steps and spent less time transitioning from one foot to the other. Both our clinical measures of walking, TUG and DGI, also improved. The DGI improvement may reflect changes in functional skills (eg, stepping around cones and over an obstacle) that may be more affected by leg placement deficits where visual guidance is needed, in addition to deficits in balance or general walking. Improvements in walking are striking considering that balance training was primarily done in sitting and standing positions with little focus on gait activities.
With the observed gains in walking, it is surprising that we did not find changes in our clinical standing balance measure (Functional Reach) or in our laboratory balance measure (postural sway during static standing). We can only speculate that training, which included both dynamic and static balance exercises, affected dynamic balance more than static balance, and we did not test dynamic balance. Improving dynamic balance during gait may be reflected in the trend toward reduced variability in stride length across visits. In the future, a dynamic balance test, such as shift of center of gravity, should be assessed.26,27
ICARS and ABC scores did not change over the 13 weeks. Previously, we showed that the ICARS is a sensitive measure for assessing the decline in degenerative ataxia over a 1-year period. 27 Because other measures were sensitive to improvement in this study, it appears that the ICARS is not sensitive to more subtle improvements that occur over shorter time frames.
The ABC scale measures participants’ balance confidence, but many participants were unable to recall the last time they had attempted many of the skills rated in the scale (eg, standing on a chair and walking on ice). In future studies another measure for perceived change should be used, such as the individualized measure of the goal attainment score.
It is interesting to note that there were self-reports of improvements in everyday life after training: 3 participants, for example, noted that they were able to close their eyes in the shower when washing their hair, which they were previously unable to do; 2 noted greater confidence starting and stopping while walking in the city; and 1 participant no longer needed a hiking stick to walk and work in the family farm.
We wanted to determine what factors influenced the changes observed following participation in the home exercise program. Previously, we showed that the posture and gait subscore of the ICARS is a predictor of severity of ataxia and function at 1 year after testing for degenerative cerebellar disease. 27 We suspected that in addition to severity of ataxia, age, balance challenge, and duration of practice could be factors that would predict improvement. However, we found that higher level of balance challenge was the only factor that predicted greater improvement in walking speed. Without a control group, this finding is limited but does emphasize the importance of designing an appropriately challenging program as well as determining an individual’s ability to follow a home program. This finding is in contrast to that of Ilg et al, 7 who found that frequency was an important factor in retaining changes.
The improvements in walking speed after the 6-week exercise program are similar in magnitude to those seen after an intensive program with direct intervention. 4 It is valuable to demonstrate that a home program can be as effective as direct care. Home programs are less costly, require less travel, and may be more convenient for patients who do not have access to a clinic. With the necessity of challenging an individual’s balance, direct intervention, however, may be necessary for individuals who are unable to safely challenge their balance alone at home.
Determining the appropriate dose of intervention is an important next step. Although rehabilitation will not prevent progression of cerebellar degeneration, future longitudinal studies can examine what doses of training optimize function throughout the course of the disease. Additionally, because our program primarily included sitting and standing balance exercises, individuals who are no longer able to walk can potentially use the program. Safer transfers and greater mobility in later stages of cerebellar degeneration would greatly improve patients’ quality of life.
As with most home programs, participant compliance both in terms of frequency and quality of performance was limited to monitoring by self-report and demonstration at follow-up. Challenging balance in the home was emphasized but for safety reasons was difficult for some participants. Because our results suggest that greater levels of challenge of the exercises is crucial for improvement, for participants who could not safely challenge themselves at home, additional training sessions or direct supervision of the exercise program may have been more beneficial.
Conclusions
This study demonstrates improvement in both clinical and laboratory measures of mobility in cerebellar patients with ataxia after a 6-week home training program. After limited one-on-one practice with a therapist, the home exercise program, which focused on balance training, improved locomotion. Individualizing the training program may be critical because our results suggest that challenging balance is more important for improving walking characteristics and speed than increasing exercise frequency. Continued training and progression of the program may be necessary to see optimal retention of results.
Footnotes
Acknowledgements
The authors would like to thank Kristin Musselman for helpful review of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by NIH R01 HD040289.
Authors’ Note
The roles of the authors were as follows. JLK, acquisition, analysis, and interpretation of data and writing of manuscript. AB: study concept and design; acquisition, analysis, and interpretation of data; writing of manuscript; and study supervision.
