Abstract
Background: Task-specificity, repetition and progression are key variables in the acquisition of motor skill however they have not been consistently implemented in post-stroke rehabilitation. Objective: To evaluate the effectiveness of a stroke rehabilitation plan of care that incorporated task-specific practice, repetition and progression to facilitate functional gain compared to standard physical therapy for individuals admitted to an inpatient stroke unit. Methods: Individuals participated in either a circuit training (CTPT) model (n = 72) or a standard (SPT) model (n = 108) of physical therapy, 5 days/week. Each 60 minute circuit training session, delivered according to severity level, consisted of four functional mobility tasks. Daily exercise logs documented both task repetition and progression. Results: The CTPT model was successfully implemented in an acute rehabilitation setting. The CTPT group showed a significantly greater improved change in gait speed from hospital admission to discharge than the SPT group (0.21± 0.25 m/sec vs. 0.13± 0.22 m/sec; p = 0.03). The difference between groups occurred primarily among those who were ambulatory upon admission. There were no significant differences between the two cohorts at 90 days post-stroke as measured by the FONE-FIM, SF-36 and living location. Conclusions: Therapy focused on systematically progressed functional tasks can be successfully implemented in an inpatient rehabilitation stroke program. This circuit-training model resulted in greater gains in gait velocity over the course of inpatient rehabilitation compared to the standard model of care. Community-based services following hospital discharge to maintain these gains should be included in the continuum of post-stroke care.
Introduction
Stroke is a leading cause of long-term disability, with most survivors requiring assistance in completing mobility tasks for daily function. Although the focus of rehabilitation is to reduce disability to attain independence, 90% of individuals at discharge have some functional disability and lack independence in basic mobility skills. 1 Poststroke sensorimotor deficits require relearning the most basic skills: rolling and sitting up over the bedside, sit to stand, sitting and standing balance, transfers, gait, and negotiating stairs. The charge to rehabilitation professionals is to structure therapy sessions to provide the most efficacious means of attaining these skills for return home and attainment of independence.
Task-specific training, first described in the strengthening 2 and postural control literature 3,4 is now advocated in stroke rehabilitation, with the rationale that we learn what we practice. It is well established that task-specific practice is a prerequisite for motor learning. 5 “Task-oriented” 6 or “task-related” 7 are also used to designate the emphasis on practice of functional tasks rather than the practice of isolated movement patterns. Tasks must be at the appropriate level for the patient’s ability, and as they improve, task difficulty progresses. Task-oriented training studies, conducted primarily in chronic stroke, demonstrate enhanced recovery of locomotion, 8-10 improved paretic limb weight-bearing in sitting 11 and sit to stand, 12 and improved cardiorespiratory fitness. 13 Upper-extremity kinematics, 7 strength, 14,15 and function 14-16 also improve following functional task training.
Repetition is also a requisite for motor learning. The most fundamental principle in motor skill learning is that the degree of performance improvement is dependent on the amount of practice. 5 Behavioral experience promotes neuroplastic changes in the brain, but simply engaging a neural circuit in task performance is not sufficient to drive plasticity. 17 Repetition of a newly learned or relearned behavior is required to induce lasting neuronal changes. Behaviorally induced changes in cortical organization are at once encouraging yet sobering as rehabilitation professionals recognize the amount of practice necessary for central reorganization 18-21 and the disparity between this requirement and the amount occurring in clinical practice.
Although most inpatient physical therapy sessions are 1 hour in length, and services across disciplines are provided 3 to 4 h/d, behavioral mapping studies reveal that the time patients are engaged in activity is much less—between 36 and 62 min/d. 22-25 Observational studies designed to count repetitions of therapeutic activities during both inpatient 26 and outpatient therapy 26,27 sessions further revealed the paucity of practice or task repetition in rehabilitation. Repetitions of purposeful movements were far below those reported in motor learning studies.
Valid reasons exist for the disparity between the numbers of repetitions that occur in motor learning studies versus rehabilitation sessions. Patients are not simply completing repetitions, but interaction and feedback between the therapist and patient is critical. Patients do not simply practice 1 task within a session but are faced with the necessity to practice numerous motor tasks. Given these needs, it is imperative that the limited therapy time is maximized by repetitive, progressive practice of functional motor tasks necessary for daily mobility. Planning the contents of a session in advance with a predetermined progression of tasks may allow more time for in-session practice.
Circuit training, a mode of exercise using a series of systematically progressed workstations, is an intervention approach that may provide the necessary platform to encourage more practice (task repetition). This model has been examined primarily in chronic, ambulatory cohorts ranging from 3 months 9,28 to 6 months, 10,12 to greater than 1 year 13,29 from stroke onset. Participants were community dwelling and did not require physical assistance to exercise in the stations. Circuit training has been studied in more acute stroke (5-7 weeks postonset) 30-32 however, this time frame is still longer than the poststroke onset status of individuals admitted for rehabilitation in the United States. 33 Even in these more acute studies, participants needed to be able to ambulate 30,32 or stand 31 to participate in the circuit training—skills not often present in those admitted to inpatient rehabilitation following acute stroke.
In this program evaluation study, we first determined the feasibility of implementing a circuit training model of care on admission to an acute rehabilitation setting. Could patients fully participate without undue fatigue in a program of care that emphasized task-specific practice, repetition, and progression? Second, we evaluated the effectiveness of this program of care with regard to balance, gait, sensorimotor recovery, discharge destination, and mobility status at 90 days poststroke compared with a current model of inpatient rehabilitation.
Method
Participants
Participants were recruited from the Stroke Service at Brooks Rehabilitation Hospital, Jacksonville, Florida. Medical charts of consecutive new admissions were reviewed for eligibility. Criteria included (1) stroke diagnosis, (2) >18 years old, (3) living in community prior to admission, (4) ability to follow 1 step command, (5) cleared for full rehabilitation program, and (6) length of stay >5 days. As our intent with this feasibility study was to capture as broad a spectrum of stroke presentation as possible, we had no exclusion criteria related to sensorimotor, aphasia, or visuospatial neglect sequelae. Patients were only excluded if they had (1) previous stroke with residual motor deficits, (2) preexisting neurological disorder, (3) history of major head trauma, (4) lower-extremity (LE) amputation, (5) renal dialysis or end-stage liver disease, (6) legal blindness or severe visual impairment, and (7) history of significant psychiatric illness.
Study Design
A nonblinded, nonrandomized controlled trial compared the current model of physical therapy to a delivery model that emphasized high repetition task-specific practice. In this program evaluation, each model, delivered for 1.5 h/d, 5 d/wk, served as the standard of care for all stroke admissions for a 3-month period. All eligible patients admitted from October to December 2007 entered the standard physical therapy (SPT) group, and those admitted from March to May 2008 entered the circuit training physical therapy (CTPT) group. Neither patients nor therapists were blinded to group allocation. Data were only collected on those who signed an Informed Consent Form approved by the University of Florida Institutional Review Board.
Interventions
Participants received the determined mode of intervention for the duration of their inpatient stay. Occupational therapy, speech therapy, and psychology services were provided to both groups per usual multidisciplinary stroke rehabilitation practice. We did not track the number of hours of these therapy services each patient received, but there were no systemwide changes in the Stroke Program, personnel, or delivery of these services between the two 3-month periods studied. The broad aim of both types of physical therapy interventions was to improve participants’mobility to permit safe discharge home or to a destination with the appropriate level of support.
Standard physical therapy
Patients participated in a total of 1.5 h/d of physical therapy (one 60- and one 30-minute session), 5 d/wk, with a 1:1 therapist to patient ratio. Activities performed, time on task, number of repetitions, and progression of those activities were determined individually by the treating therapist. Therapeutic activities included gait training, transfers, bed mobility, sitting and standing balance, lower- and upper-extremity passive and active range of motion, and strengthening. Intervention was not based on any particular treatment philosophy.
Circuit training physical therapy
Patients participated in a total of 1.5 h/d of physical therapy (one 60-minute and one 30-minute session), 5 d/wk, with a 1:1 therapist to patient ratio. The 60-minute session was the designated circuit training session and was divided into 4 task-specific stations, 13 to 14 minutes each, tailored to patients’ mobility levels. The 30-minute session was dedicated to other critical aspects of inpatient rehabilitation: family education, orthotic and wheelchair prescription, and home program education.
For CTPT, patients’ level of stroke severity was determined on admission by the 5-m Walk Test (5MWT) gait speed: 34 1 = nonambulatory; 2 = severe (≤0.2 m/s); 3 = moderate (>0.2 ≤ 0.4 m/s) and 4 = mild (>0.4 m/s). Four levels of circuit training were established, each level consisting of a bank of 6 to 10 functional tasks. Initial severity determined at which level of circuit training participants would begin. Patients progressed within and across the 4 levels of functional tasks throughout their course of rehabilitation. For example, activities for nonambulatory patients consisted of rolling, supine-to-sit-to-supine, sitting balance, sit-to-stand, transfers, and standing balance. The activity bank for those with a mild stroke consisted of dynamic standing balance, continuous gait for endurance, stairs, ramps, dual-task gait, and an obstacle course. Appendix 1 includes a comprehensive list of exercises for each level of severity. The therapist and patient would mutually decide which 4 activities to practice at each session. This provided clinical decision-making opportunities for the therapist and empowered patients as active decision makers in their rehabilitation. Each activity was practiced for 13 to 14 minutes before rotation to the next. The therapist recorded the level of the task, assistance required, and number of repetitions/distance or time on task for each activity in a daily treatment log.
Progression
Each functional task was progressed such that the level of difficulty, complexity, and number of repetitions matched each individual’s ability. A systematic task progression was provided for each exercise to guide therapists with the progression and to ensure that patients were attempting tasks with sufficient challenge. The daily treatment logs served as a cue for the therapist to progress the patient from the previous session’s activity level. For example, sit-to-stand tasks progressed from coming to stand from a 23-inch surface with feet in parallel to coming to stand from a 13-inch surface with paretic LE behind nonparetic LE to enforce weight bearing through the paretic limb. Dynamic sitting balance was progressed from alternating weight shift between ischial tuberosities to using feedforward control to bat objects. If a patient was able to repeat a given task 10 times safely and without deterioration of form, he or she was progressed in that task.
The intent was for the participant to be continuously active for the 1-hour circuit training session. Blood pressure guidelines, 85% target heart rate, and Rate of Perceived Exertion scales were positioned around the exercise gym. Therapists used these American Heart Association and American College of Sports Medicine guidelines 35-37 to determine if an activity should be stopped.
Evaluations
Individuals were assessed on all primary outcome measures on admission and discharge. Follow-up assessments occurred at 90 days poststroke via phone. As the aim of this feasibility study was to determine if a circuit training model of therapy delivery could be implemented within the current structure of an inpatient stroke program, the patients’ assigned therapist delivered the intervention and conducted the assessment. All assessments were outlined in a manual of procedures, and all assessors received identical instruction in the primary outcome measures and were standardized in their delivery.
Stroke severity, depression, cognition, and premorbid functional status—factors known to influence recovery—were assessed on admission. 38-41 To better characterize our population, severity was assessed by the NIH Stroke Scale 42 and the Orpington Prognostic Scale, 43 depression by the Patient Health Questionnaire (PHQ-9) Depression screen, 44 cognition by the Mini-Mental State Exam, 45 and premorbid functional status by the Short Form (SF-36), physical functioning domain. 46,47
Outcome Measures
Primary outcome measures were gait speed as measured by the 5MWT, 34 functional balance as measured by the Berg Balance Scale (BBS), 48 paretic lower-extremity motor control and sensation as measured by the Lower Extremity Fugl-Meyer Motor (LEFM-M) and Sensory (LEFM-S) Assessment, 49 Functional Independent Measure—Mobility portion (FIM-M; bed transfers, toilet transfers, shower/tub transfers, locomotion, and stairs), 50 and discharge destination. For the 5MWT, participants walked at their comfortable pace along a corridor marked at 2, 7, and 9 m from the start position. A stopwatch was used to record the time to walk the middle 5 m of the walkway. If participants were unable to advance their paretic LE independently the 5MWT was not conducted; they were considered nonambulatory. Assistance during paretic limb stance and balance control was allowed.
The BBS and LEFM-M and LEFM-S were assessed according to protocols established by a current clinical trial of poststroke recovery. 51 Secondary outcome measures—living arrangement, phone version of the Functional Independence Measure (FONE-FIM) 52 and Stroke Impact Scale (SIS)-16 53 —were conducted via phone at 90 days poststroke. Sociodemographic characteristics and presence of stroke risk factors were obtained from medical records.
Intensity of Intervention Delivery
We defined intensity of intervention as the number of repetitions of a functional activity completed during the 60-minute session. We randomly observed both SPT and CTPT sessions and counted the number of repetitions of 8 predetermined functional mobility activities (number of gait steps, transfers, static standing, dynamic standing, static sitting, dynamic sitting, sit to stand, and number of stair steps).
Statistical Analyses
Descriptive statistics were used to compare the baseline characteristics between the SPT and CTPT cohorts. A Wilcoxon rank sum test was used to compare the 2 cohorts in the number of repetitions per session for each of the 8 observed functional tasks. Change scores for the primary outcome measures were computed by subtracting admission from discharge scores. Change scores were then compared between the SPT and CTPT groups using t tests.
In addition, regression analyses were conducted to investigate the interaction effect of treatment group (SPT vs CTPT) by baseline ambulation status (ambulator vs nonambulator) adjusting for baseline PHQ-9 and SF-36. Paired t tests were used to investigate whether there were significant changes between admission and discharge across all participants when the SPT and CTPT groups were combined. For the 90-day outcomes measures, group means were also compared using t tests. All statistical tests were performed at a .05 significance level using SAS version 9.1 (SAS Institute, Cary, North Carolina).
Results
In all, 182 individuals consented to participate and received physical therapy in either the SPT or CTPT mode of delivery according to month of admission (Table 1). Overall, both treatment groups showed significant improvement between inpatient rehabilitation admission and discharge in 5MWT, LEFM-M, LEFM-S, BBS, and FIM.
Patient Enrollment a
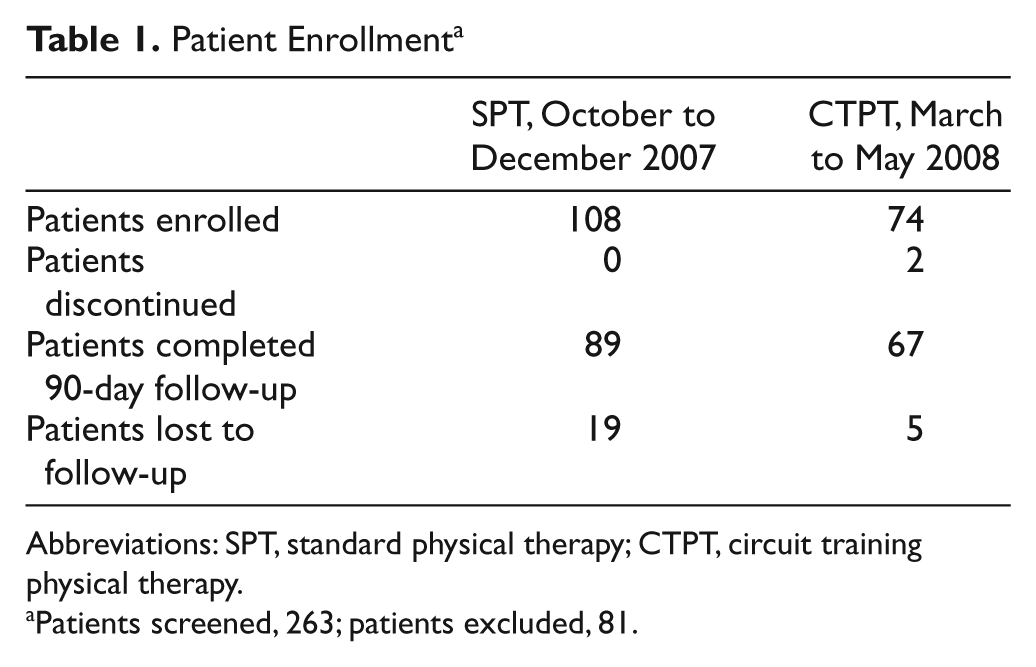
Abbreviations: SPT, standard physical therapy; CTPT, circuit training physical therapy.
Patients screened, 263; patients excluded, 81.
Feasibility of CTPT Intervention in Acute Stroke
Of the patients assigned to the CTPT group, 97% (72/74) were able to fully participate in this mode of therapy throughout their rehabilitation stay. Just 2 patients (3%) were discontinued from this intervention mode as they were unable to participate in a full 60-minute treatment session secondary to excessive fatigue. All participants assigned to the SPT group continued participation for the duration of their rehabilitation. There were no adverse events for either cohort.
Intensity of the Intervention
To quantify the intensity of the intervention sessions, the number of functional task repetitions was counted during 46 SPT and 32 CTPT randomly observed sessions. Wilcoxon rank sum test revealed that the CTPT cohort had a significantly higher intensity of task repetition for dynamic sitting, dynamic standing, sit to stand, and stair stepping tasks compared with the SPT group (Table 2). This demonstrated that it was feasible to increase the number of repetitions practiced during a 60-minute session.
Repetitions per Session of Functional Tasks
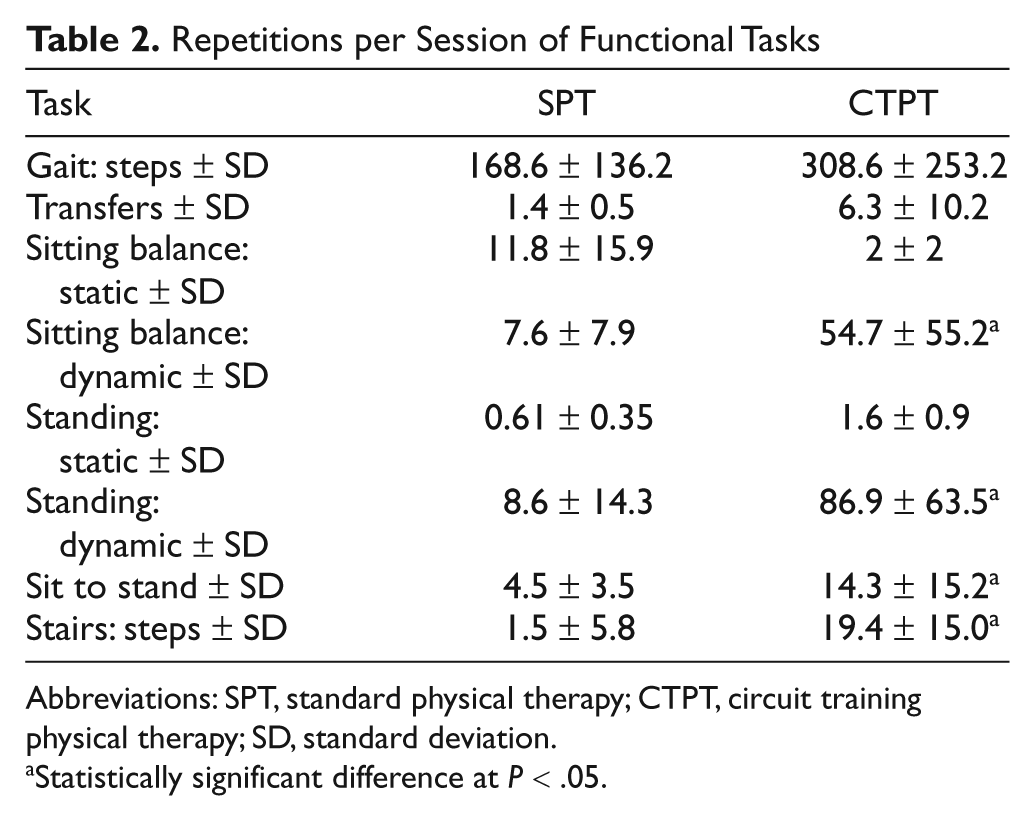
Abbreviations: SPT, standard physical therapy; CTPT, circuit training physical therapy; SD, standard deviation.
Statistically significant difference at P < .05.
Effectiveness of CTPT Versus SPT
Demographics, baseline characteristics, and predictors of stroke recovery
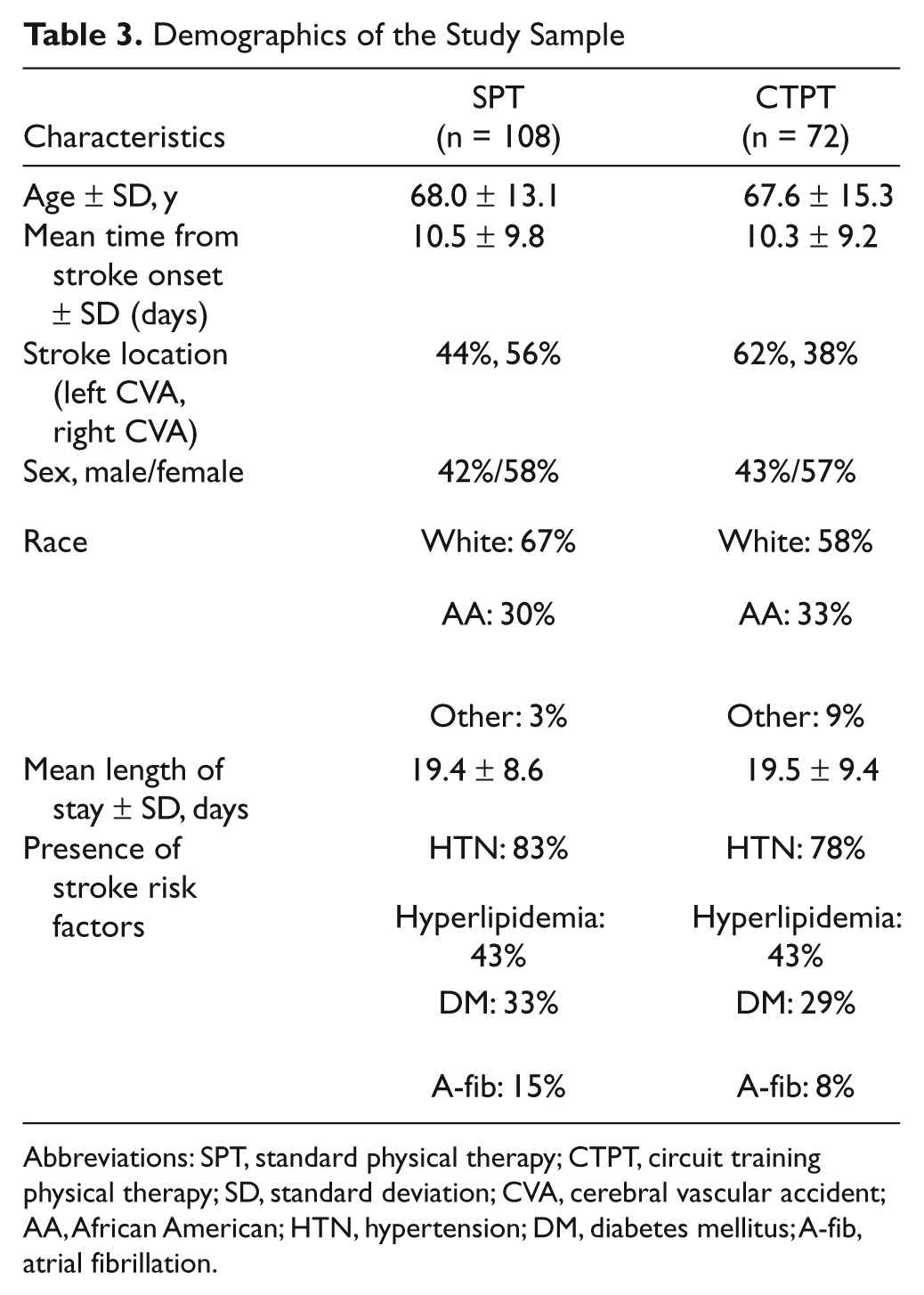
There was no significant difference in demographic characteristics between the 2 groups (Table 3). Times from stroke onset to rehabilitation admission (SPT, 10.5 ± 9.8 days; CTPT, 10.3 ± 9.2 days) and length of stay (SPT, 19.4 ± 8.6 days; CTPT, 19.5 ± 9.4 days) were the same for both groups. Table 4 presents baseline characteristics of 5 predictors of stroke recovery. The SPT group was significantly more depressed as assessed by the PHQ-9 than the CTPT group (8.8 ± 5.7 vs 5.7 ± 5.8; P < .05). There was a trend for those in the SPT group to have a lower premorbid SF-36 score than those in the CTPT group (23.9 ± 5.8 vs 25.6 ± 5.7; P = .06). The 2 groups were not significantly different from one another on any of the outcome measures at baseline (Table 5).
Demographics of the Study Sample
Abbreviations: SPT, standard physical therapy; CTPT, circuit training physical therapy; SD, standard deviation; CVA, cerebral vascular accident; AA, African American; HTN, hypertension; DM, diabetes mellitus; A-fib, atrial fibrillation.
Baseline Assessments for Predictors of Stroke Recovery
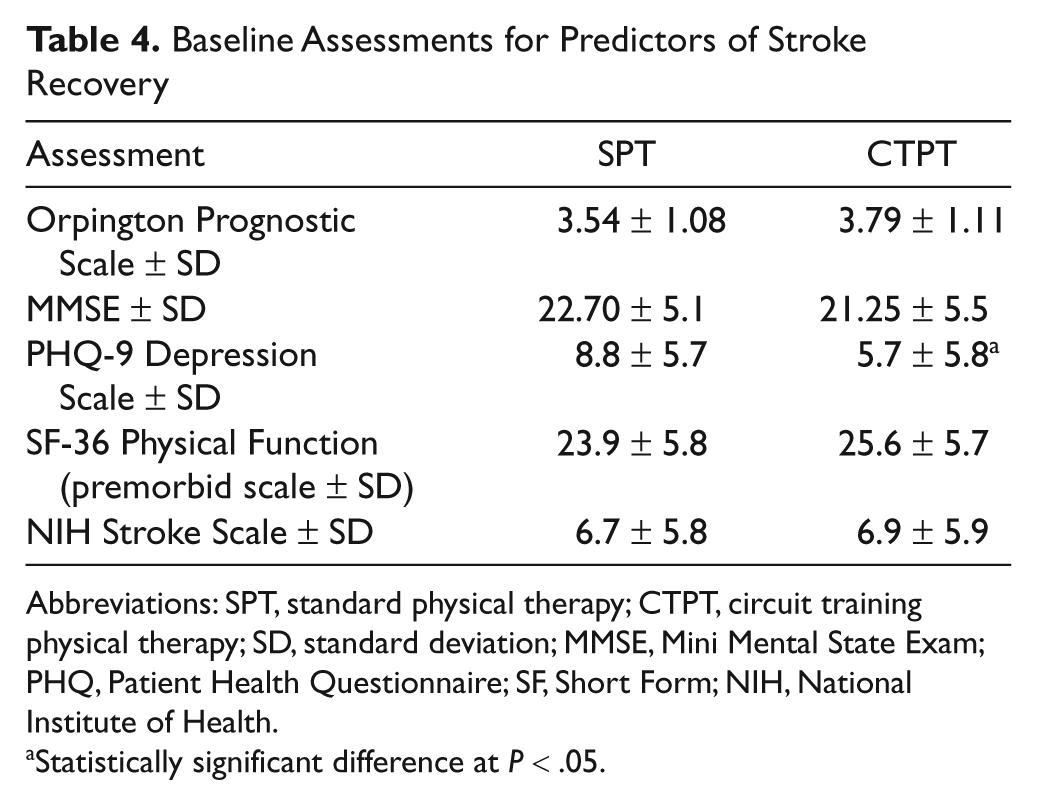
Abbreviations: SPT, standard physical therapy; CTPT, circuit training physical therapy; SD, standard deviation; MMSE, Mini Mental State Exam; PHQ, Patient Health Questionnaire; SF, Short Form; NIH, National Institute of Health.
Statistically significant difference at P < .05.
Outcome Measures at Baseline
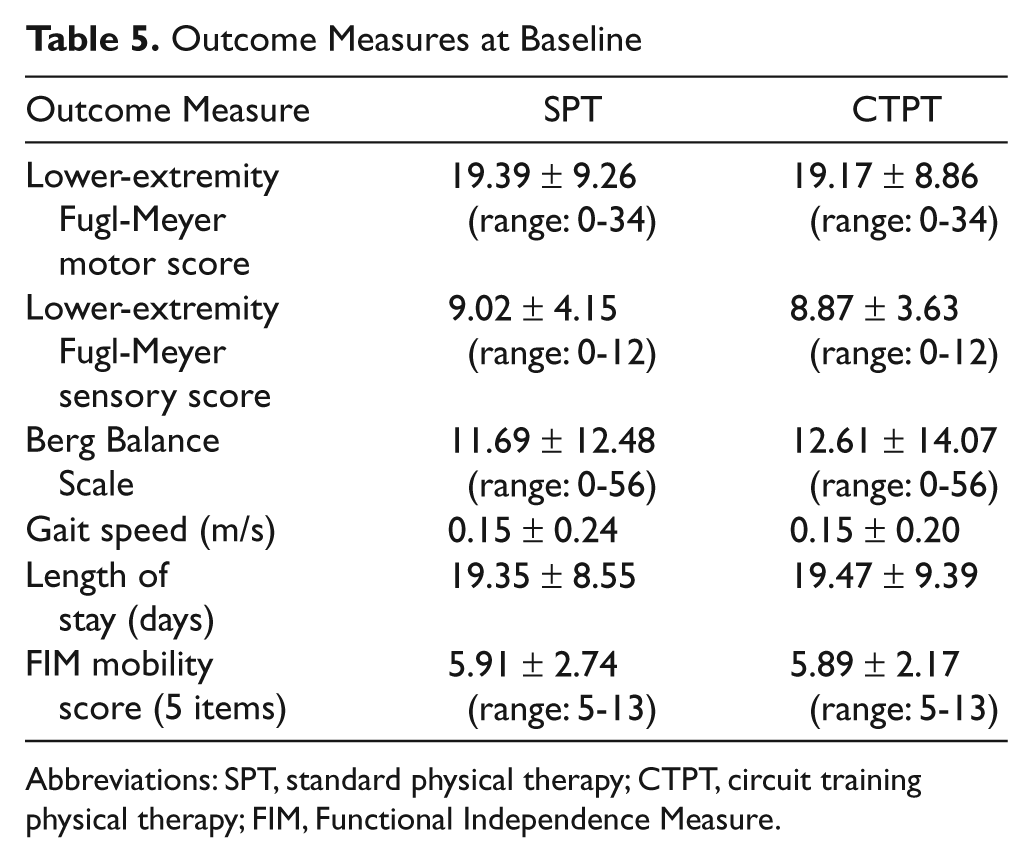
Abbreviations: SPT, standard physical therapy; CTPT, circuit training physical therapy; FIM, Functional Independence Measure.
Change scores from admission to discharge
A 2-sample t test showed that the CTPT group had a significantly greater change in gait speed (5MWT) from admission to discharge than the SPT group (0.21 ± 0.25 vs 0.13 ± 0.22; P = .03). Furthermore, regression analysis revealed that there was a significant interaction effect (F = 9.23; P = .003) between group and baseline severity as measured by the 5MWT. The difference between groups occurred among those who were able to complete the 5MWT at baseline (mean gait speed changes of 0.28 vs 0.11 m/s for CTPT and SPT, respectively), with negligible difference among those unable to ambulate on admission (mean changes of 0.16 vs 0.17 m/s for CTPT and SPT, respectively). There were no significant differences between the 2 groups in the other outcome measures, although there was a trend in the data toward a greater change in BBS and LEFM-S for the CTPT than the SPT group (Table 6).
Change Scores Between Admission and Discharge
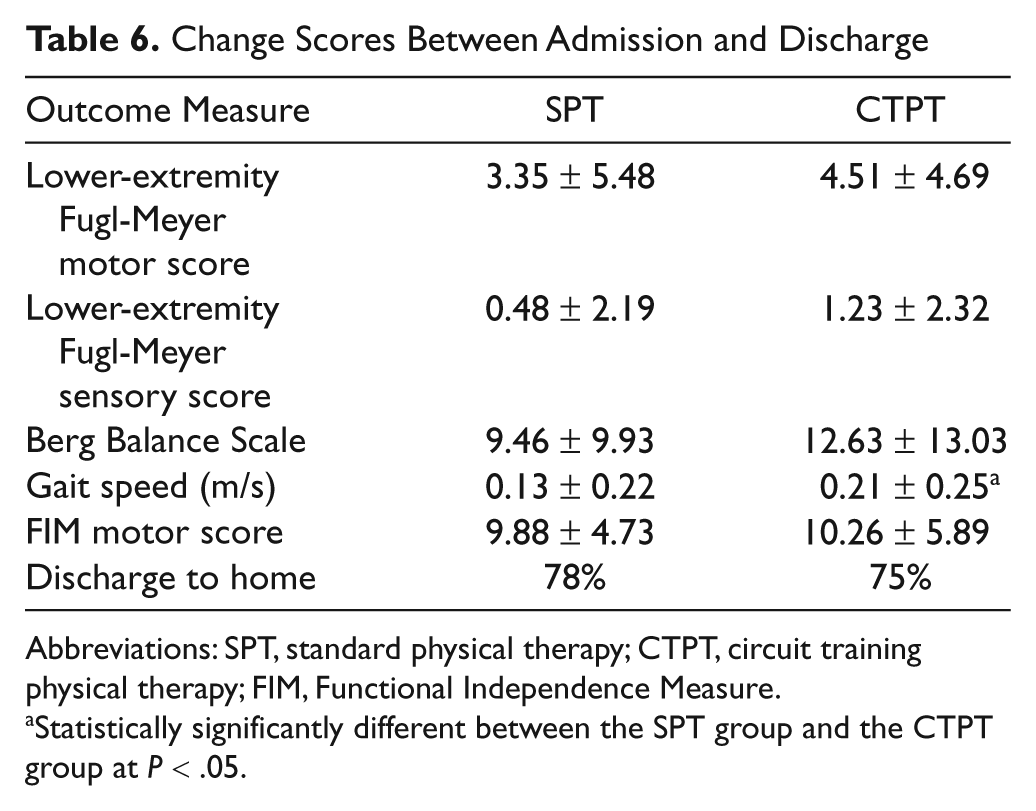
Abbreviations: SPT, standard physical therapy; CTPT, circuit training physical therapy; FIM, Functional Independence Measure.
Statistically significantly different between the SPT group and the CTPT group at P < .05.
90-Day follow-up
Across all participants, 156 (87%) were able to be contacted at 90 days poststroke. There were no significant differences between the 2 treatment cohorts on the 90-day follow-up outcomes measured by FONE-FIM, SIS-16, and living location.
Lost to follow-up
Those who were lost to follow-up had a slower baseline gait speed (0.17 ± 0.21 m/s vs 0.33 ± 0.31 m/s; P < .05) and a smaller change in gait speed (0.05 ± 0.16 m/s vs 0.17 ± 0.24 m/s; P < .05) between admission and discharge compared with those not lost to follow-up. There were no other significant differences in baseline or change scores between these 2 groups.
Discussion
This is the first study to examine circuit training of functional task practice stations in an inpatient acute rehabilitation setting. The primary finding of this feasibility study is that individuals only 10 days poststroke tolerated an intervention mode that provided intense task practice focused on repetition and progression. This study is novel in that we determined that individuals at this acuity level were able to perform systematically progressed functional task practice exercises for 60 minutes. Although the number of rest periods during a session was not counted, they were sufficiently minimal for therapists to document full treatment sessions throughout the study. There were no adverse events associated with CTPT intervention.
Just 2 participants assigned to CTPT (3%) were unable to participate in 4 functional tasks during a 60-minute session. They were not able to sit upright unsupported, and it could be argued that they may not have been candidates for inpatient rehabilitation; however, they were admitted to the hospital and CTPT was attempted. The majority of the cohort (97%), however, was able to fully participate in CTPT, providing evidence that a structured therapy session, with attention to ensuring task repetition and task progression can be successfully used in an acute inpatient rehabilitation setting.
Patients assigned to CTPT had a statistically significant greater change in gait velocity between admission and discharge compared with those in SPT. Further analysis revealed that this greater change in the CTPT compared with the SPT group was specifically for those who were able to complete the 5MWT on admission, that is, had some degree of ambulation capability, suggesting that circuit training may be most beneficial for those with some motor ability in the lower extremity and sufficient trunk control to remain upright. Although gait speed change was significantly greater for the CTPT group, their gait speed at the time of inpatient discharge remained significantly below that needed for community ambulation. There was a trend in the data for those in the CTPT group to have a greater improvement in balance ability, as measured by the BBS, compared with the SPT group. The circuit training focus on whole-body functional tasks translated into greater improvements in gait and the functionally based BBS.
There were no differences in discharge destination between the 2 interventions. Numerous factors determine discharge destination other than patients’ physical ability, such as social and caregiver support, finances, and accessibility, which may have accounted for this lack of difference. Additionally, there were no differences at 90 days poststroke in the FONE-FIM or SIS-16, suggesting that the advantage of a circuit-training intervention mode observed at discharge was not maintained at home. Similar results are reported by Mudge et al. 10 A greater initial improvement in walking endurance immediately following 12 sessions of circuit training compared with a control intervention did not persist at a 3-month follow-up assessment. This suggests the need for a continuance of activity and exercise to sustain and advance the initial gains realized in the inpatient rehabilitation setting. We advocate the implementation of community-based wellness programs to address this need. 54-56 A second explanation for our results at 90 days poststroke is also provided by Mudge and colleagues. 10 Although a postintervention assessment revealed greater walking endurance for the circuit-trained compared with the control group, there were no differences between the groups on 2 self-report measures of mobility. This may be because of lower perceived ability compared with actual ability and may be the case in this present study as well.
One of the contributing factors to decreased time in active exercise and mobility is the precautionary approach by therapists to not overstress patients’ cardiovascular systems or overfatigue their musculoskeletal systems, providing rest periods when they may not be warranted. Doubt or concern is often expressed that patients will not tolerate therapy that is continuous and constantly progressed.
More than 75% of individuals poststroke have comorbid cardiovascular disease, 57 so indeed, established exercise guidelines must be followed. However, we may be exercising caution to a fault and thereby not providing appropriate cardiovascular and musculoskeletal stress and sufficient challenge that will provide our patients the opportunity to improve. Although with these compromised systems following stroke it may not be feasible to exercise continuously for 60 minutes, this study demonstrated that individuals, even in the acute phase of rehabilitation, can tolerate intense practice. Health professionals, by frequently inquiring if patients “need a rest” or suggesting “you should rest now,” risk perpetuating a “sick patient” in contrast to a “return to health” mentality. A rest is often provided out of habit rather than physiological need. Participants in the CTPT group anecdotally remarked, “It feels good to work hard” and “This feels like real exercise.” We do not deny that rest may be needed during a 60-minute session but encourage therapists to determine if the rest is physiologically warranted rather than assume that the patient is tired and provide rest out of habit. Using published guidelines on heart rate, blood pressure, and rate of perceived exertion (RPE) for those with compromised cardiovascular systems can guide the therapist regarding providing appropriate rest. 35-37
Including the patient in task selection and documentation of repetitions and progression, as done in wellness and fitness programs, helps patients perceive themselves as “well persons” who need to return to activity rather than as “sick persons” who need to be treated. The therapist–patient relationship becomes one akin to coach–athlete or teacher–student instead of the provider–receiver relationship of health care. 58,59
Study Limitations
This was not a randomized controlled trial but rather a feasibility study on the implementation of a circuit training model of care in an inpatient acute stroke rehabilitation unit. As such, the patient’s assigned therapist served as both the interventionist and the evaluator, precluding blinded evaluations. The focus of this feasibility study was on implementation of CTPT; the study was not designed to incorporate blinded evaluators. Participants were nonrandomized because the model of physical therapy delivery in operation at time of hospital admission determined group allocation. The number of intervention sessions was not fixed across patients as this was dependent on length of stay. Because of a decline in referrals during the CTPT delivery, there were an unequal number of patients in the 2 cohorts. Therapy services were not tracked between discharge and the 90-day phone calls, so their role in the 90-day results is unknown. Follow-up assessments were limited to self-report phone questionnaires in which functional abilities may have been overestimated or underestimated. We did not directly assess gait and balance once patients were discharged from the rehabilitation hospital. Patients in the SPT group were significantly more depressed than those in the CTPT group. Depression cannot be excluded as a factor that could contribute to the results.
Conclusion
This study demonstrated that rehabilitation focused on systematically progressed functional tasks can be successfully implemented with individuals 10 days poststroke. This circuit-training model resulted in greater gains in gait velocity during inpatient rehabilitation compared with standard therapy. These gains were not maintained at 90 days poststroke. Providing community-based services as part of the continuum of poststroke care may be a mechanism to maintain these gains.
Footnotes
Appendix
Examples of Exercises Included in Circuit Training for Each Severity Level
| Four activities were practiced within each 60-minute session, 13 to 14 minutes per activity: |
| Nonambulatory group Rolling Scooting in supine Sidelying to sit Static sitting balance/Equal weightbearing Dynamic sitting balance Scooting in sitting Transfers Standing Sit to stand |
| Severe group Rolling Sidelying to sit Dynamic sitting balance Transfers Sit to stand Standing Pregait Gait |
| Moderate group Transfers Sit to stand Dynamic standing balance Gait Stepping onto a block Stairs |
| Mild group Dynamic standing balance Sit to stand Gait Dual task gait Gait with adaptability Stairs |

Acknowledgements
Brooks Community Health Foundation; Janet Collins, Analytics IT Manager; Brooks Rehabilitation Stroke Team Therapists; and Brooks Rehabilitation Hospital Stroke Program are gratefully acknowledged. The contents of this manuscript do not represent the views of the Department of Veterans Affairs or the United States Government.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Dorian Rose, Erin Crews, and Pamela Duncan received financial support for this research from the Brooks Community Health Foundation.
