Abstract
Background. Treatment of genetic degenerative ataxia is currently based on symptom management and maintenance of function. However, utilization of rehabilitation is limited due to a lack of evidence supporting its efficacy. Objective. This systematic review evaluated rehabilitation interventions for individuals with genetic degenerative ataxia. In addition, long-term outcomes from rehabilitation and optimal duration and intensity of rehabilitation were examined. Methods. A comprehensive search of 4 databases (MEDLINE, CINAHL, PEDro, and Cochrane) identified randomized, nonrandomized controlled, and cohort studies published from inception through to January 2016. The studies included at least one measure examining function, ataxia, balance, or gait. Methodological quality was assessed with the Australian National Health and Medical Research Council (NHMRC) Hierarchy of Evidence and the randomized controlled trials were rated according to the PEDro scale. Results. Seventeen studies met eligibility criteria. Five randomized controlled trials were included; however, the majority were classified as level III-3 and IV studies. Of 292 participants included, 148 had autosomal dominant ataxia, and 85 had autosomal recessive ataxia. Rehabilitation interventions included coordination and balance training, multifaceted inpatient rehabilitation, a cycling regime, balance exercises with technology assisted biofeedback, respiratory muscle training, and treadmill training. Two studies examined adjuncts to rehabilitation. Fifteen of the 17 studies demonstrated a statistically significant improvement in at least 1 outcome measuring ataxia, function, gait, or balance. Less than half of the studies included assessment of long-term outcomes and follow-up time frames varied considerably. Conclusion. There is consistent evidence that rehabilitation improves function, mobility, ataxia, and balance in genetic degenerative ataxia.
Genetic degenerative ataxia encompasses a heterogeneous group of autosomal recessive, autosomal dominant, X-linked, and mitochondrial ataxias. 1 The dominant ataxias are classified into more than 40 subtypes of spinocerebellar ataxia (SCA), 1 while Friedreich ataxia (FRDA), ataxia-telangiectasia (AT), and ataxia with oculomotor apraxia types 1 and 2 are the most common of the recessive ataxias.1,2 In some ataxias, the neuropathology is purely cerebellar; however, extrapyramidal, spinocerebellar tract, dorsal column, or peripheral nerve pathology is present in many. Disease onset, clinical presentation, and progression are variable between, and even within disease categories. Nevertheless, a universal feature is significant loss of function and mobility secondary to ataxia. 2
Although a number of trials are underway, proven pharmacological therapies for these disorders is rare. 3 Primary treatment is generally based on symptom management and maintenance of function, 4 of which rehabilitation and physical therapy is a crucial component. However, despite the known benefits in other neurological degenerative diseases,5,6 inaccessibility to quality care, therapists’ lack of understanding of rehabilitation potential, and minimal scientific evidence limit the utilization of physical therapy for individuals with degenerative ataxia. 7 In 2013, a review of prospective trials examining high-intensity coordination training in individuals with degenerative cerebellar ataxia demonstrated beneficial effects on mobility. 8 These improvements were almost equivalent to regaining 1 year of natural disease progression 8 ; however, only 4 studies met the stated inclusion criteria. Therefore, the focus of the review was limited to measuring the impact on gait and stance. In 2014, Fonteyn et al 9 examined all available allied health interventions for individuals with ataxia. The authors found inconclusive evidence to inform guidelines for physical therapy programs and suggested rehabilitation was most effective earlier in the disease course. However, a potential limitation of this review was the inclusion of nondegenerative and relapsing-remitting conditions. Therefore, outcomes may not reflect the specific challenges associated with degenerative ataxia. 10
A comprehensive understanding of the effects of physical rehabilitation will facilitate development of effective and targeted treatment strategies for improving function and quality of life, and enhance the cost-effectiveness of health care. 11 Thus the main overarching objective of this systematic review was to evaluate the effectiveness of rehabilitation interventions and potential adjuncts to rehabilitation, for individuals with genetic degenerative ataxia. We aimed to expand findings from the previous two systematic reviews by including both prospective and retrospective studies and both ambulant and non-ambulant participants. Furthermore, we also examined long-term outcomes and ideal duration and intensity of rehabilitation.
Methods
A comprehensive search of 4 electronic databases (Cochrane Library, Physiotherapy Evidence Database (PEDro), CINAHL and MEDLINE) was performed to identify publications from inception to, and including, January 2016. In addition, reference lists of included articles and the authors’ personal libraries were manually searched for further publications. The search was restricted to English-language articles. A combination of the following index terms and keywords or their synonyms were used for the search: “physical therapy” OR “exercises” OR “hydrotherapy” OR “physical training, balance” OR “rehabilitation” OR “occupational therapy” AND “Friedreich ataxia” OR “cerebellar ataxia” OR “spinocerebellar ataxia” OR “spinocerebellar degenerations” OR “ataxia” OR “cerebellar disease.”
Publications met the inclusion criteria if they examined the effectiveness of rehabilitation or physical therapy, or an adjunct therapy to physical therapy, in individuals with genetic degenerative ataxia and, included at least one measure examining function, ataxia, balance, mobility, or quality of life in the analyses. Randomized, nonrandomized controlled, and cohort study designs were included. Studies examining the effects of interventions on individuals with cerebellar ataxia due to non-genetic causes, such as stroke, tumor or spinal cord injury were excluded. However, publications were included if the study population comprised multiple etiologies; and the data from the genetic degenerative ataxias was identifiable in the analyses or greater than 65% of participants were diagnosed with a genetic degenerative ataxia. For this review, idiopathic cerebellar ataxia (IDCA) and sporadic adult-onset ataxia (SAOA) were included, as studies have shown up to 29% of cases have an undiagnosed genetic cause.11,12 Studies of pharmacological interventions were excluded from this review.
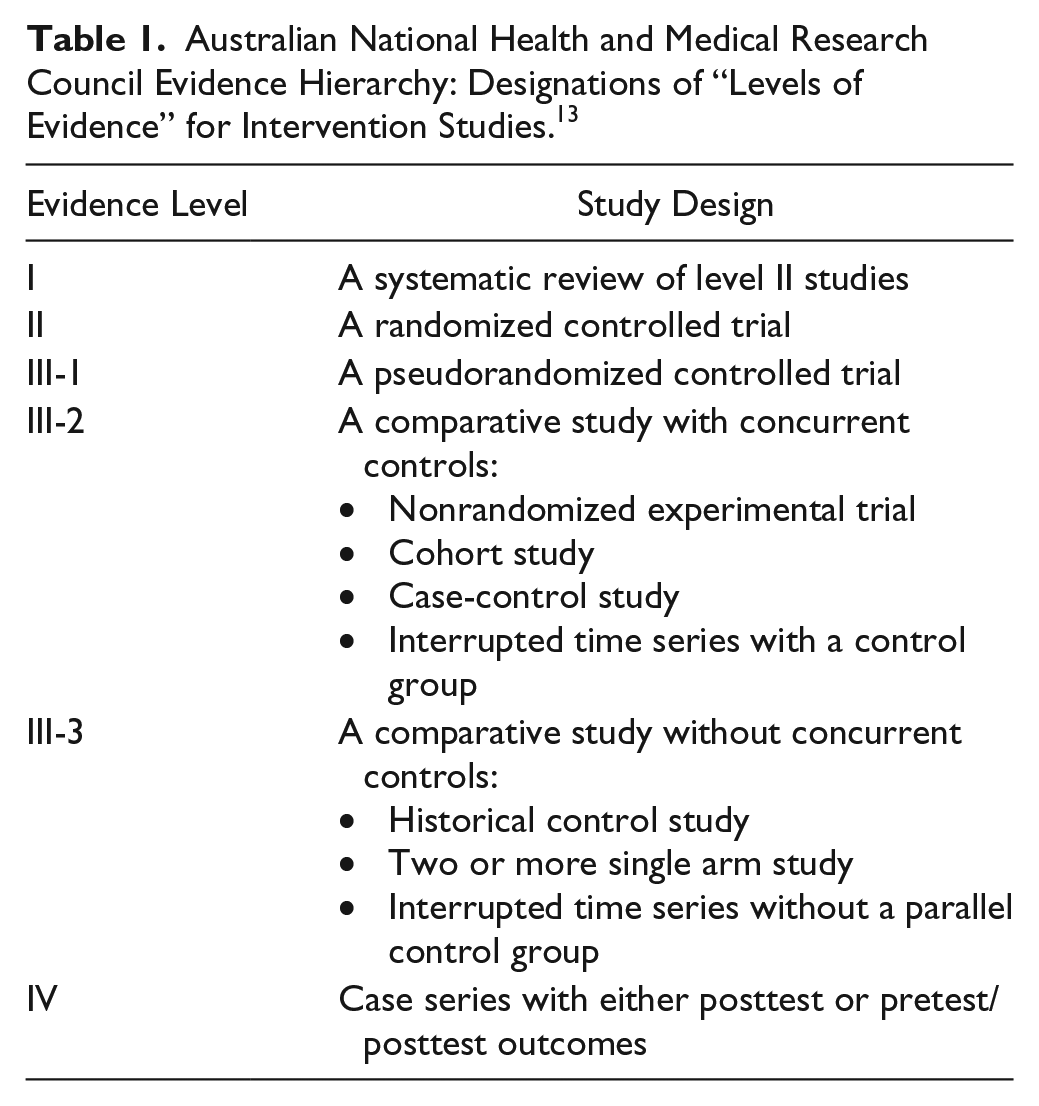
Articles were graded according to the Australian National Health and Medical Research Council (NHMRC) Evidence Hierarchy 13 (Table 1). This hierarchy assigns levels of evidence based on the ability of a study’s research design to answer a specific research question. 13 Intervention studies range from level I to level IV with the highest level of evidence, level I, a systematic review. Methodological quality of included randomized controlled trials (RCTs) was further rated according to the PEDro scale. 14 The PEDro scale was developed by Delphi consensus, 14 with 2 additional items reporting follow-up adequacy and between-group statistical comparison. The maximum score for the PEDro scale is 10, with studies rated as excellent (score 9-10); good (6-8); fair (4-5), and poor (<4). 15 Because of the large variability in inclusion criteria, and the heterogeneity of outcome measures, a meta-analysis was not performed. However, where data were available, within-group data were expressed as a percentage of improvement. This was calculated as described by Aman et al. 16
Australian National Health and Medical Research Council Evidence Hierarchy: Designations of “Levels of Evidence” for Intervention Studies. 13
Results
A total of 1010 publications were identified, 17 of which met the eligibility criteria. One of these publications was a long-term follow-up 17 from an original study 18 so these results were combined. Characteristics of the included studies are summarized in Table 2.
Summary of Included Studies.
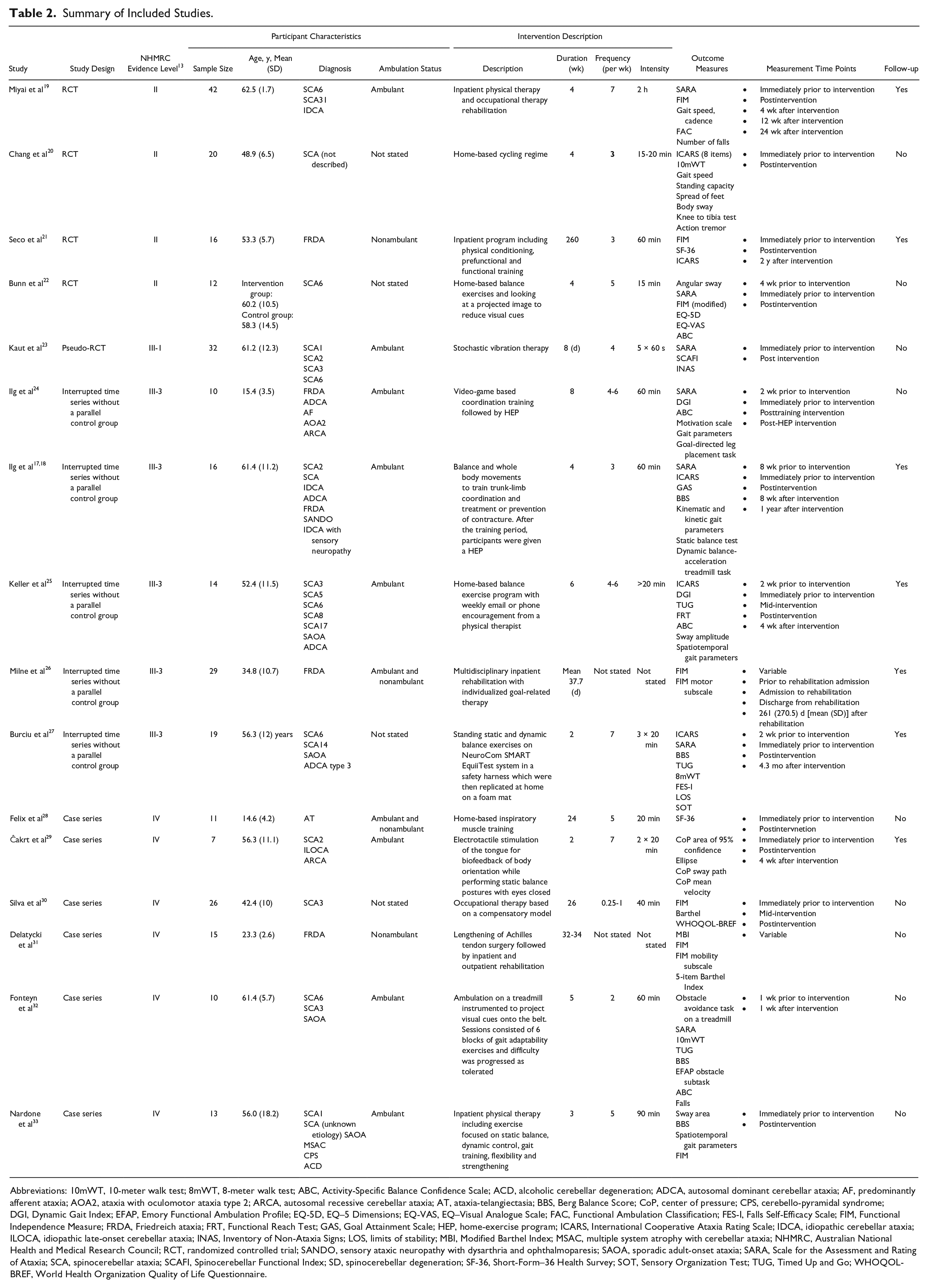
Abbreviations: 10mWT, 10-meter walk test; 8mWT, 8-meter walk test; ABC, Activity-Specific Balance Confidence Scale; ACD, alcoholic cerebellar degeneration; ADCA, autosomal dominant cerebellar ataxia; AF, predominantly afferent ataxia; AOA2, ataxia with oculomotor ataxia type 2; ARCA, autosomal recessive cerebellar ataxia; AT, ataxia-telangiectasia; BBS, Berg Balance Score; CoP, center of pressure; CPS, cerebello-pyramidal syndrome; DGI, Dynamic Gait Index; EFAP, Emory Functional Ambulation Profile; EQ-5D, EQ–5 Dimensions; EQ-VAS, EQ–Visual Analogue Scale; FAC, Functional Ambulation Classification; FES-I, Falls Self-Efficacy Scale; FIM, Functional Independence Measure; FRDA, Friedreich ataxia; FRT, Functional Reach Test; GAS, Goal Attainment Scale; HEP, home-exercise program; ICARS, International Cooperative Ataxia Rating Scale; IDCA, idiopathic cerebellar ataxia; ILOCA, idiopathic late-onset cerebellar ataxia; INAS, Inventory of Non-Ataxia Signs; LOS, limits of stability; MBI, Modified Barthel Index; MSAC, multiple system atrophy with cerebellar ataxia; NHMRC, Australian National Health and Medical Research Council; RCT, randomized controlled trial; SANDO, sensory ataxic neuropathy with dysarthria and ophthalmoparesis; SAOA, sporadic adult-onset ataxia; SARA, Scale for the Assessment and Rating of Ataxia; SCA, spinocerebellar ataxia; SCAFI, Spinocerebellar Functional Index; SD, spinocerebellar degeneration; SF-36, Short-Form–36 Health Survey; SOT, Sensory Organization Test; TUG, Timed Up and Go; WHOQOL-BREF, World Health Organization Quality of Life Questionnaire.
Methodological Quality
Five studies were RCTs19-23; however, only 4 were graded as NHMRC level II evidence 13 as Kaut et al 23 used a block (quasi) randomization method to allocate participants to the intervention or sham group. The mean (standard deviation) PEDro score for these trials was 6.4 (1.8), and ranged between fair and excellent quality (see Table 3 for individual scores). All studies met criterion 11 by including measures of variability and point measures for at least one key outcome 14 ; however, no other criterion was unanimously met. The majority of RCTs had blinded-assessors. Similar to most studies examining physical rehabilitation, the total PEDro score in 3 studies was reduced by the inability to blind therapists and participants.19,20,22 However, Seco et al 21 reported that participants and therapists were blinded to the group allocation by conducting the interventions at different locations, while Kaut et al 23 were able to include a sham stochastic vibration therapy to blind participants to their allocation. Of the remaining 11 studies, the evidence level ranged from level III-317,18,24-27 to IV28-33 (Table 2). The level III-3 studies all used an intraindividual controlled design measuring outcomes at least twice prior to the study intervention. Six studies in this review were classified as level IV studies and did not have a control.
PEDro Score for Randomized Controlled Trials. a

Individual PEDro scale item descriptions: 1, eligibility criteria were specified; 2, subjects were randomly allocated to groups; 3, allocation was concealed; 4, groups were similar at baseline regarding the most important prognostic indicators; 5, blinding of all subjects; 6, blinding of all therapists who administered the therapy; 7, blinding of all assessors who measured at least one key outcome; 8, measures of at least one key outcome were obtained from more than 85% of the subjects initially allocated to groups; 9, all subjects for whom outcome measures were available received the treatment or control condition as allocated or, where this was not the case, data for at least one key outcome were analyzed by “intention to treat”; 10, results of between-group statistical comparisons are reported for at least one key outcome; 11, study provides both point measures and measures of variability for at least one key outcome.
Item 1 not included in total score.
Study Population
A total of 292 participants, including 148 with autosomal dominant ataxia, 85 with autosomal recessive ataxia, 27 with IDCA and 28 with SAOA, were included in the studies that met inclusion criteria for this review. From the studies with mixed etiologies, 4 participants with nongenetic ataxias were included in the analysis. These diagnoses included multiple system atrophy, cerebello-pyramidal syndrome and alcoholic cerebellar degeneration. Of the autosomal dominant ataxias, 140 participants had a diagnosis of SCA, with SCA6 (n = 57) and SCA3 (n = 39) the most frequent. Rehabilitation outcomes were investigated in 67 participants with FRDA. Four participants had an autosomal recessive ataxia of an unknown or undescribed genetic cause24,29 while 1 study examined respiratory muscle training in 11 participants with ataxia-telangiectasia (AT). 28 The mean age of all participants was 49.4 years with an age range between 11 and 82 years. A total of 228 participants were ambulant, and 72 were nonambulant. In 2 studies, ambulation status was not reported.
Cerebellar Versus Afferent Ataxia
Nine studies distinguished between participants with a more pure cerebellar ataxia and those with an afferent ataxia component. Five studies included participants with degenerative ataxias with pure cerebellar ataxia19,22,27,28 or excluded participants with sensory impairment. 33 One of these studies was the largest NHMRC level II study (n = 42) 19 and demonstrated significant changes in Scale for the Assessment and Rating of Ataxia (SARA) 34 and the Functional Independence Measure (FIM) 35 after rehabilitation. Three studies included participants likely to have a significant afferent component to their ataxia, including 2 studies with individuals with FRDA26,31 and 1 study with SCA3. 30 Both FRDA studies demonstrated significant changes in function; however, neither utilized ataxia measures or other neurological outcomes. In the study with SCA3 participants, there were no significant within-group changes after occupational therapy. Seven studies17,18,23-25,29,32 included multiple degenerative ataxias, neither specifically including nor excluding participants with afferent ataxia or sensory impairment. One study performed a subgroup analysis comparing the effects between a “cerebellar” group and an “afferent” group. 18 This study indicated that four weeks of training was less effective for individuals with afferent ataxia, with less sustain in positive results. However, baseline ataxia scores were significantly worse in the afferent group, which may have influenced results.
Study Intervention
There were 7 main categories of training identified in this review: coordination and balance training, multifaceted inpatient rehabilitation, a cycling regime, balance training, treadmill training, occupational therapy, and inspiratory muscle training. Corrective foot and ankle surgery and stochastic vibration sat outside these categories as adjuncts to rehabilitation. Within each of these categories the type of training was varied. Therapy was carried out in an inpatient setting in 4 studies19,21,26,33; 4 were conducted in an outpatient setting18,23,29,32 and 4 were home based.20,22,25,28 Three utilized a mixed program, with training carried out at home after 10 to 14 days of training in a supervised outpatient environment in 2 studies,24,27 and a combination of inpatient and outpatient in the other. 31 One study did not describe the setting. Figure 1 provides an overview of the within-group percentage change (pre- and postintervention) on the most frequently used outcomes, the International Cooperative Ataxia Rating Scale (ICARS), 36 the SARA, the Berg Balance Scale (BBS), 37 and the FIM. Coordination and balance training and multifaceted inpatient rehabilitation demonstrated the greatest percentage change in the SARA, while multifaceted inpatient rehabilitation appeared to have the greatest impact on function. Three studies did not report data for any of these measures.25,28,29
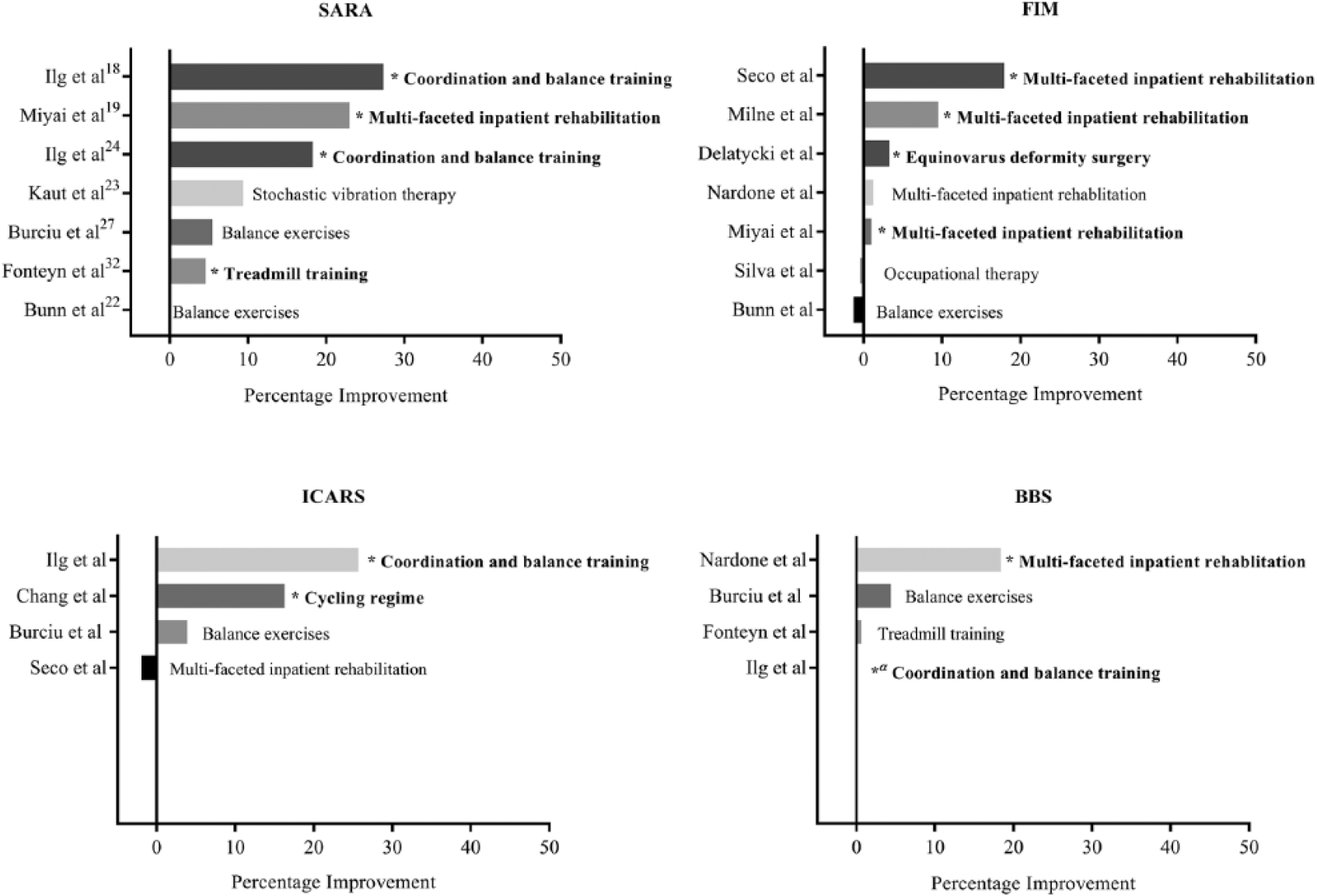
Effectiveness of rehabilitation intervention—percentage improvement. BBS, Berg Balance Score; FIM, Functional Independence Measure; ICARS, International Cooperative Ataxia Rating Scale; SARA, Scale for the Assessment and Rating of Ataxia. *Significant within-group difference; α, insufficient data to calculate percentage improvement.
Coordination and Balance Training
Ilg et al 18 investigated the effects of coordination training on motor performance in ambulant adults with either degenerative cerebellar or afferent ataxia. The training program in this prospective cohort study comprised physical therapy aimed at improving balance and trunk-limb coordination, and preventing falls and lower limb contracture. Compared with baseline scores, there was a significant decrease in ICARS and SARA scores, and an increase in the BBS score following intervention, all indicative of clinical improvement. Additionally, gait speed, step length, and temporal variability in hip-knee coordination improved in the cerebellar group; however, there was no significant change in the afferent ataxia group.
Ilg et al 24 examined video game–based coordination training targeting goal-directed limb movements, dynamic balance, and whole-body coordination in children and adolescents (aged 12-20 years) with autosomal recessive or dominant ataxia. SARA scores, step variability and lateral sway improved after the initial training period of 2 weeks. Further improvement in ataxia, step variability, and lateral sway occurred after 6 weeks of home training.
Balance Training
Keller et al 25 described a prospective intraindividual control cohort study examining a 6-week home exercise program (HEP). Participants were included if they had a genetic disease that primarily involved the cerebellum. Significant improvements were seen in the Dynamic Gait Index, Timed Up and Go (TUG), and gait velocity following the HEP. However, there were no significant improvements in participants’ ICARS scores.
Burcui et al 27 undertook a prospective cohort study to evaluate a 2-week postural training program on a moving force platform with visual biofeedback. 27 Participants were diagnosed with SCA or SAOA, and served as their own controls. Compared with pre-training scores, there were no significant improvements in ICARS or SARA scores, or gait velocity. However, there were significant improvements in limits of stability (LOS) testing, the BBS, and the Sensory Organization Test scores.
Bunn et al 22 investigated balance exercises while looking at a projected image to reduce visual cues for individuals with SCA6. This randomized controlled feasibility trial compared 4 weeks of home-based exercises to a control group who completed no intervention. Angular sway, SARA score, FIM, and Activity-Specific Balance Confidence Scale were the outcome measures used. No statistical significance was found in any of the outcome measures.
Treadmill Training
Fonteyn et al 32 evaluated a 5-week treadmill training program on individuals with SCA or SAOA. The program focused on gait adaptability and utilized projected visual cues to simulate obstacles. In this study, there was no control group or intraindividual control. After training, there was a significant improvement in the SARA score; however, there were no changes in the BBS, TUG, or 10-meter walk test (10mWT) scores.
Multifaceted Inpatient Rehabilitation
Miyai et al 19 investigated the effects of a multidisciplinary inpatient rehabilitation program in ambulant participants with SCA or IDCA. Participants were randomized to an immediate start or a 4-week delayed start. Interventions included strengthening and balance exercises, spinal mobilization, relaxation, coordinative and dual motor task practice, and gait, stair climbing, and activities of daily living practice. Facilitatory tactile and verbal techniques were applied to enhance motor learning. The FIM, SARA, gait velocity, and number of falls significantly improved in the intervention group compared to the delayed-start group.
Seco et al 21 conducted an RCT comparing a 5-year inpatient rehabilitation program and usual pharmacological management to pharmacological management alone, in individuals with FRDA. The inpatient rehabilitation provided by physical therapists, included physical conditioning, prefunctional training, and functional training. The intervention group showed a significant improvement in FIM and SF-36 scores, while FIM and SF-36 scores were significantly worse in the control group. There was no significant change in the ICARS score in the intervention group; however, the ICARS score increased in the control group.
Milne et al 26 reported the results of a study of individual goal-related inpatient rehabilitation provided by a multidisciplinary team for individuals with FRDA. Duration of intervention was variable, ranging between 2 and 166 days, and was dependent on the participant’s goals. The rehabilitation focused on risk management; hip, pelvic, and spinal control; spasticity management; hand function; communication and swallowing; cardiovascular exercise; and specialized equipment prescription. Development of a home or community program was incorporated in the rehabilitation program. The primary outcome measure in this study was function, as measured by the FIM. Compared with functional decline seen prior to inpatient rehabilitation, function significantly improved following the period of rehabilitation. The greatest improvement was seen in independence with transfers, dressing the lower body, and toileting.
Another retrospective study evaluated a 3-week inpatient rehabilitation program. 33 The study conducted by Nardone et al 33 was exclusive to ambulant participants with degenerative ataxia due to varied etiologies. Interventions, designed and conducted by a physical therapist, targeted static balance, dynamic balance control, gait training, and flexibility and strengthening exercises. There was a significant improvement in BBS score and cadence during gait; however, there was no significant change in gait velocity or FIM score following the intervention.
Cycling Regime
Chang et al 20 conducted an RCT comparing a 4-week home-based cycling regime (n = 10) with a control group (n = 10) completing verbal health education and upper limb exercises. All participants had a diagnosis of “pure” SCA. There was a significant improvement in the ICARS score following the cycling intervention and no change in the control group.
Inspiratory Muscle Training
Felix et al 28 described the outcomes from inspiratory muscle training at 40% to 60% of maximal inspiratory pressure in 11 individuals with AT. Participants completed a 24-week home-based inspiratory muscle training program. Although the primary outcome was lung volume and respiratory muscle strength, a Brazilian-translated SF-36 was used to measure quality of life. After training, an improvement in lung volumes and respiratory muscle strength corresponded to a significant improvement in the general health and vitality domains of the SF-36.
Occupational Therapy
Silva et al 30 described the outcomes for individuals with SCA3 following a 6-month occupational therapy program based on a compensatory model. The Barthel Index (BI) 38 and Portuguese version of the FIM were utilized to measure function and the World Health Organization Quality of Life (WHOQOL) questionnaire measured quality of life. After the intervention, there was no significant change to FIM, BI, or WHOQOL.
Stochastic Vibration
One quasi-randomized trial investigated stochastic vibration as a possible adjunct to physical therapy. 23 In this trial, participants with SCA were randomized into an intervention group (n = 17) receiving 4 sessions of stochastic vibration therapy or a control group receiving sham vibration over the same duration. 23 There was no significant change in the total SARA or Inventory of Non-Ataxia Symptoms score in the vibration therapy group compared with the sham group. However, there was a significant improvement in the SARA gait and posture subscores and 10mWT time in the intervention group.
Corrective Foot and Ankle Surgery
Delatycki et al 31 reported the results of surgery to correct equinus deformity in 7 nonambulant participants with FRDA, followed by intensive inpatient and outpatient rehabilitation. After the intervention there was a significant increase in the FIM mobility subscale and 5-item BI scores. This study reported a clinically significant change in the ability to transfer independently.
Intensity and Dosage
With the exception of the 5-year and 6-month interventions provided by Seco et al 21 and Silva et al, 30 respectively, the mean (standard deviation) duration of rehabilitation was 6.3 (6.5) weeks. Sessions ranged from 15 to 60 minutes in duration with an average session time of 46.5 (23.8) minutes. Rehabilitation was conducted on average 4.4 times per week, ranging from once a week to twice daily. Figures 2 and 3 provide an overview of the impact of intensity and duration of therapy on the percentage change to SARA, FIM, ICARS, and BBS scores. However, because of the heterogeneity of outcome measures, the impact of training intensity or duration could not be determined. Nevertheless, in 2 studies, greater intensity of training was related to better outcomes.17,18,24 Conversely, Keller et al 25 found no impact on outcome from the frequency of training; however, increased balance challenge during training was related to improved gait velocity.
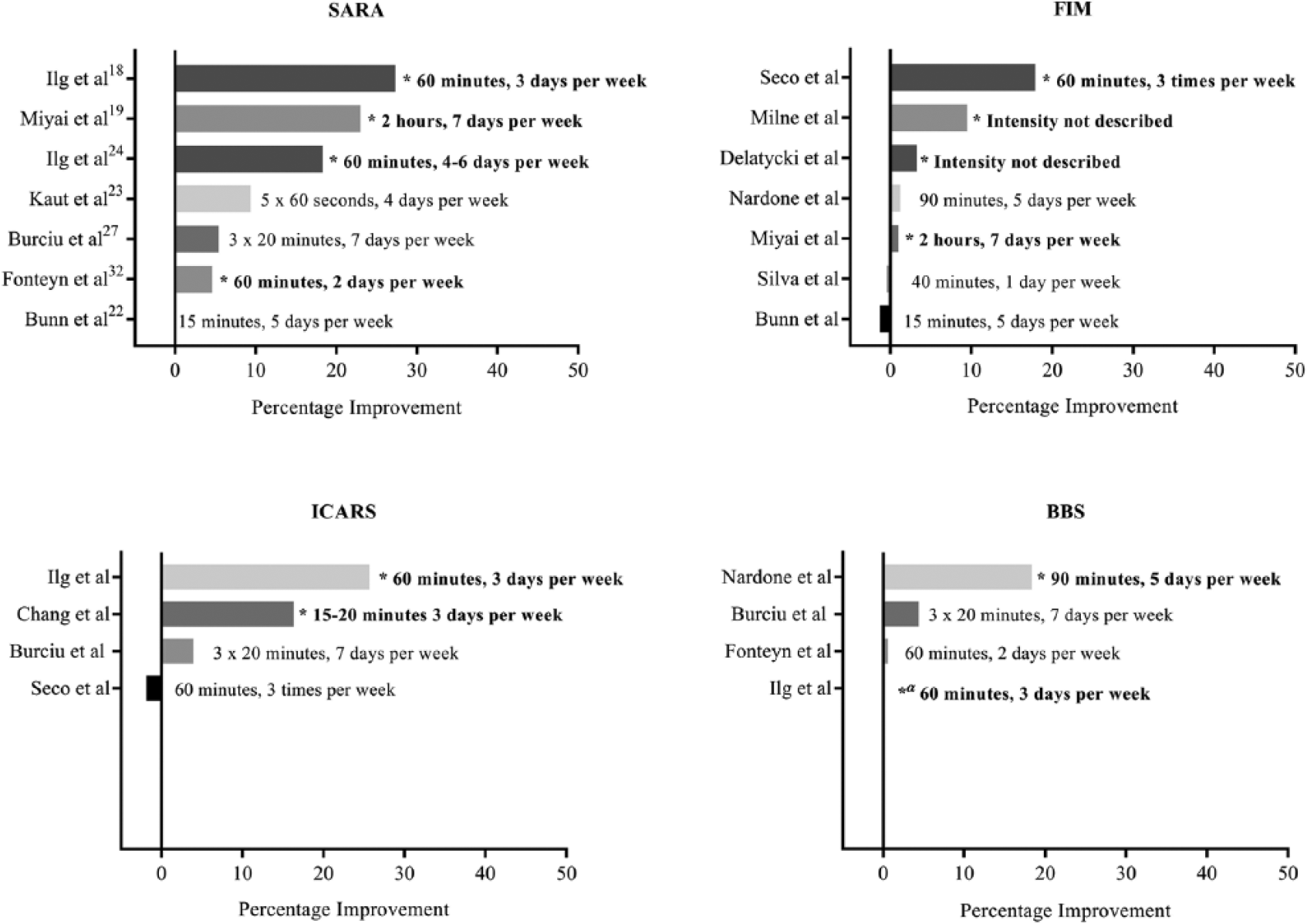
Effectiveness of rehabilitation intensity—percentage improvement. BBS, Berg Balance Score; FIM, Functional Independence Measure; ICARS, International Cooperative Ataxia Rating Scale; SARA, Scale for the Assessment and Rating of Ataxia. *Significant within-group difference; α, insufficient data to calculate percentage improvement.
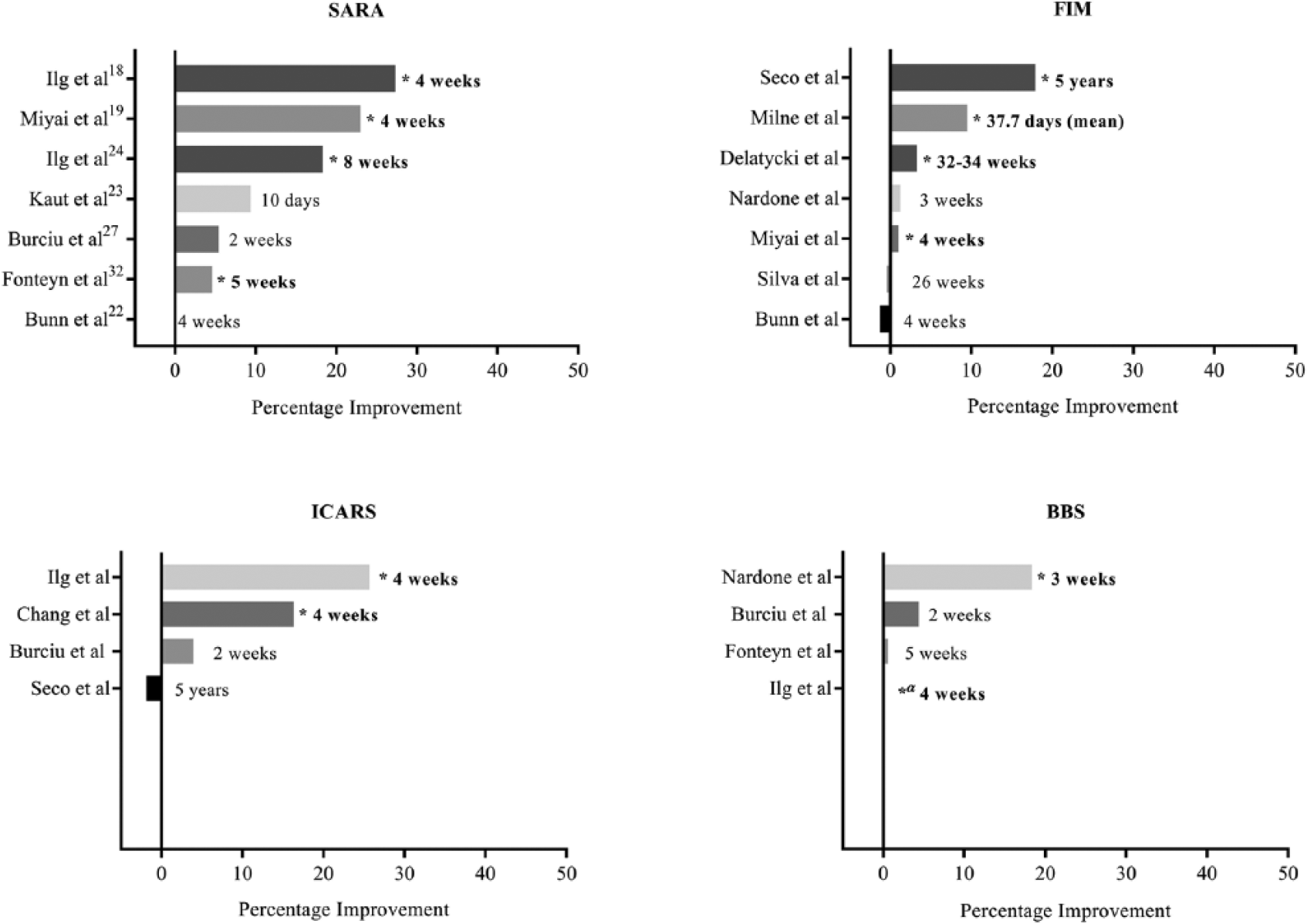
Effectiveness of rehabilitation duration—percentage improvement. BBS, Berg Balance Score; FIM, Functional Independence Measure; ICARS, International Cooperative Ataxia Rating Scale; SARA, Scale for the Assessment and Rating of Ataxia. *Significant within-group difference; α, insufficient data to calculate percentage improvement.
Sustainability of Results Postrehabilitation
Seven studies conducted a follow-up assessment. Follow-up duration ranged between 1 month19,25,29 and 2 years. 21 Four studies compared follow-up outcomes to baseline19,21,25,29 and 3 evaluated change with postintervention scores.21,27,29 One study measured follow-up outcomes compared with prerehabilitation change scores. 26 Only 4 studies included an assigned HEP or measured continued exercise following the rehabilitation intervention.17,18,25,26
All studies with a 4-week follow-up found significant improvements in balance, ataxia, gait speed, or function when compared with baseline, in most, but not all outcomes.18,19,25,29 Two studies with a 3-month follow-up found a significant improvement in SARA score and gait speed, but unchanged FIM score compared with baseline scores 19 ; and no significant decline in LOS, BBS score, and sway length compared with postintervention scores 27 .
Three studies evaluated follow-up at 6 months or greater after inpatient rehabilitation.19,21,26 In 1 study at 6 months, 3-month improvements in the SARA score were eradicated, while normalized gait speed was the only measure still significantly improved compared with baseline. 19 However, in 2 studies, FIM scores remained significantly higher compared with the progressive decline prior to rehabilitation 26 or to baseline measures. 21 Follow-up periods were an average of 261.7 (270.5) days 26 and at 2 years, 21 respectively in these studies. However, Seco et al 21 found a significant worsening in FIM scores between the end of the intervention and the 2-year follow-up. At this time point, the rehabilitation group had stable ICARS scores, while participants in the control group had a significant worsening in the ICARS score.
Ilg et al 17 evaluated 1-year follow-up outcomes after intensive coordination training and an ongoing HEP. SARA scores worsened from postintervention values and changes in the BBS score and gait velocity were not sustained. 17 However, SARA scores were still significantly improved compared with baseline for participants with pure cerebellar ataxia.
Discussion
The purpose of this review was to examine the evidence for rehabilitation and potential rehabilitation adjuncts in improving function, balance, mobility, ataxia, and quality of life in individuals with genetic degenerative ataxia. Seventeen publications, 5 of them RCTs, were identified. All of the studies, with the exception of the feasibility study 22 and the compensatory occupational therapy intervention, 30 demonstrated a statistically significant within-group improvement in at least one outcome of gait, function, balance and/or ataxia. Miyai et al 19 provided the most conclusive evidence (NHMRC level II study), reporting significant between-group improvements in ataxia, function, number of falls, and balance after 4 weeks of inpatient rehabilitation for individuals with more pure cerebellar ataxia. In this review, rehabilitation interventions were varied. Interventions included intensive coordination and balance training, 18 video- or technology-facilitated training22,24,27,29,32 and inpatient rehabilitation.19,21,26,33 Two of the inpatient rehabilitation studies were RCTs.19,21 Seven studies included a HEP as either the primary intervention or as a component of their intervention.17,18,20,22,24,25,27,28 Two studies investigated possible adjuncts to rehabilitation: equinovarus deformity surgery 31 and stochastic vibration therapy. 23 Less than half of the publications examined the persistence of gains from rehabilitation by a follow-up assessment. When assessed, follow-up time frames varied considerably, being between one month and two years postrehabilitation.17-19,21,25-27,29 Given the inconsistency in results, no conclusion about the long-term effect of rehabilitation could be drawn from this review.
This review highlights the significant heterogeneity in intervention type, intensity, and duration of rehabilitation interventions. This inconsistency renders drawing conclusions difficult. However, preliminary findings suggest that multifaceted programs incorporating more than one focus, such as coordination and balance training or multidisciplinary inpatient programs, may have greater effect than singularly focused therapies such as balance training or occupational therapy alone. Furthermore, significant within-group outcomes with an examination of the percentage of improvement in the SARA, ICARS, FIM, and BBS suggest that improvements in balance can occur at 3 weeks, and improvements in ataxia require a minimum of 4 weeks. The duration of therapy required for functional gains is less clear. Intensity appears to similarly impact the effectiveness of interventions. Consistent with subgroup analyses indicating improved effectiveness with greater rehabilitation intensity,17,18,24 60 minutes or greater for 2 days or more per week appears more effective than less intensive therapy. However, it is difficult to ascertain if there is a difference in benefit of beyond 3 days. Unfortunately, 2 of the inpatient studies, likely to have a greater intensity of training, did not report their training intensity so this could not be compared.26,31 However, Miyai et al, 19 with the greatest intensity of rehabilitation, demonstrated that individuals with degenerative ataxia benefit and tolerate a two hour, daily program. These preliminary summations must be considered carefully as, all but one study demonstrated within-group statistical significant effects only. Furthermore, utilizing percentage of change to examine the magnitude of effectiveness does not take into account variability of effects or baseline scores.
The inconsistency of training type and intensity is almost certainly related to an evolving understanding of the cerebellum’s role in learning-dependent neuroplasticity. Rehabilitation is based on the assumption that motor learning and motor adaptation contributes to motor recovery in individuals with neuropathology.39,40 Motor adaptation is crucial for the adjustment of movement to account for variability in demands or situation of a task, 40 essential for everyday activities. However, it is severely affected after cerebellar lesions.40,41 The unique and varied multisystem pathology within genetic degenerative ataxia similarly presents further challenges to rehabilitation effectiveness. 42 Cerebellar dysfunction typically results in increased reliance on the extracerebellar regions8,27,43 and a greater dependency on afferent information to perceive movement. 8 However, these compensatory mechanisms for individuals with extracerebellar and afferent pathology are often significantly compromised, 27 resulting in even fewer avenues for successful motor learning 27 and a further reduction in cortical excitation for facilitation of motor learning.8,44 Moreover, the variable neuropathology of genetic degenerative ataxia, 45 along with the somatotopic and functional organization of the cerebellum, suggests that disease specific interventions may be required. A greater understanding of the cerebellum’s role in learning-dependent neuroplasticity is critical for future design of rehabilitation interventions. Nevertheless, this review demonstrates, despite the differences in clinical presentation and precipitating neuropathology, there is evidence for positive outcomes from physical intervention in many of the ataxias.
A further understanding of neuroplasticity and mechanisms that underlie motor leaning are also vital in developing a scientific framework to guide rehabilitation interventions. 46 Principles of neurorehabilitation such as variable practice, practice intensity, reward during practice, and off-line skill learning (ie, during sleep) 46 enhance motor learning and thus should influence the design of interventions. Concurrent neuroplasticity augmentation therapies such as I-wave periodicity transcranial magnetic stimulation (iTMS) offer exciting new avenues to establish the best mechanisms for functional change.47,48 Aerobic exercise and other adjunct pharmacotherapies, including the utilization of neurotransmitters, dopamine and serotonin, may further enhance neuroplasticity.49-51
This review expands the previous review by Synofzik et al, 8 including all rehabilitation interventions aimed at both ambulant and nonambulant cohorts. Surprisingly, interventions aimed at nonambulant individuals provided similar improvements, although effect sizes were unable to be compared due to the heterogeneity of outcome measures and interventions. Interventions targeted at improving ambulation are more likely to be beneficial earlier in the disease course, where there is less likely to be diffuse and permanent neuronal and axonal loss. Consequently fewer structural neuroplastic changes may be required for functional change 52 and avenues for neuroplasticity are potentially more accessible 53 during rehabilitation. However, this review suggests that even individuals with advanced disease course, including those who were no longer ambulant, benefit from rehabilitation with improvements at both the body structure and function level.
The utilization of many and varied outcome measures when evaluating the effects of rehabilitation (Table 4) is evident in this review. In the genetic ataxias, there are a number of validated measures of ataxia, including the SARA and ICARS.54,55 However, there are no measures of balance validated for use specifically in degenerative ataxia. Consequently, 14 different measures of balance and a range of measures of gait were used in the included studies. Furthermore, the 25-foot walk test 56 and spatiotemporal gait parameters, validated to measure gait in some genetic ataxias,56,57 are yet to be examined for clinical meaningfulness or responsiveness after interventions. Although this review highlights the benefits from rehabilitation, the effects are inconsistent. Given the multitude of outcome measures used in these studies, it is difficult to ascertain if this is due to the effect of rehabilitation, the type and specificity of the intervention, the choice of measure or the difference in neuropathology underlying the differing diseases. Conversely, given the anticipated trajectory of decline associated with degenerative ataxia, lack of change may actually indicate either a halt or reversal in functional deterioration. There are potential funding and recruitment challenges associated with rare diseases. 58 Thus, it is critical for future meta-analyses and thus the overall accurate evaluation of interventions, that the same responsive, reliable, valid, and clinically meaningful outcomes are used in clinical trials. Furthermore, effect sizes should be reported.
Outcome Measures and Results From Included Studies.
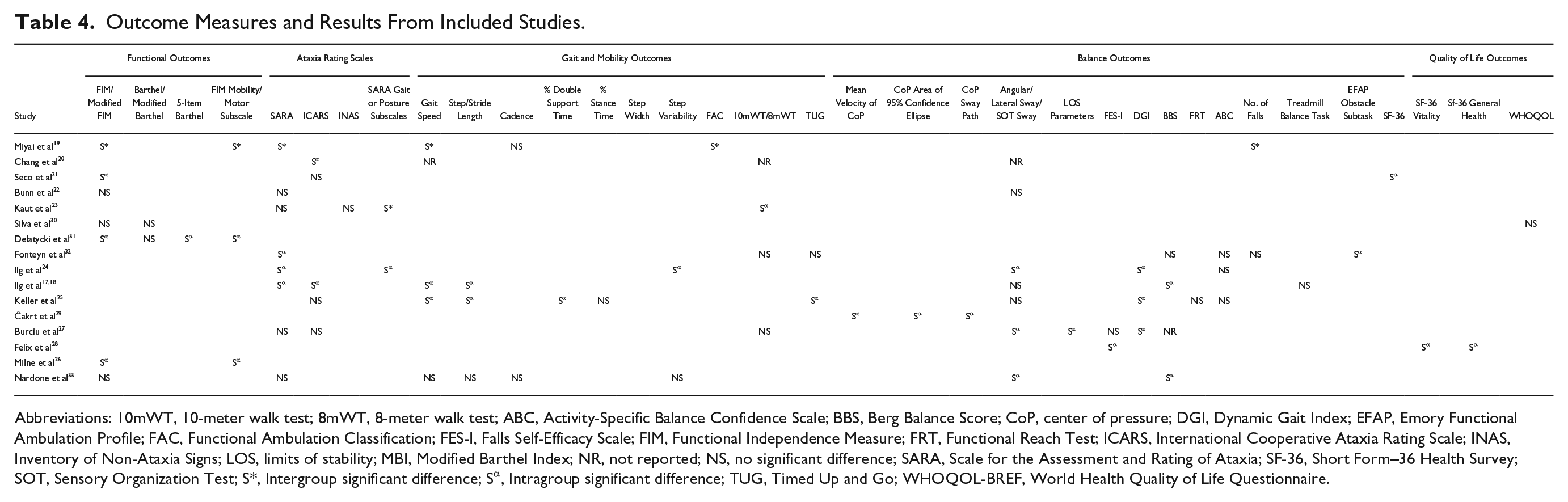
Abbreviations: 10mWT, 10-meter walk test; 8mWT, 8-meter walk test; ABC, Activity-Specific Balance Confidence Scale; BBS, Berg Balance Score; CoP, center of pressure; DGI, Dynamic Gait Index; EFAP, Emory Functional Ambulation Profile; FAC, Functional Ambulation Classification; FES-I, Falls Self-Efficacy Scale; FIM, Functional Independence Measure; FRT, Functional Reach Test; ICARS, International Cooperative Ataxia Rating Scale; INAS, Inventory of Non-Ataxia Signs; LOS, limits of stability; MBI, Modified Barthel Index; NR, not reported; NS, no significant difference; SARA, Scale for the Assessment and Rating of Ataxia; SF-36, Short Form–36 Health Survey; SOT, Sensory Organization Test; S*, Intergroup significant difference; Sα, Intragroup significant difference; TUG, Timed Up and Go; WHOQOL-BREF, World Health Quality of Life Questionnaire.
The main limitation of this review was the use of a descriptive synthesis approach. Because of the varied outcome measures used and the varying characteristics of participants it was not possible to conduct a meta-analysis or measure overall effect sizes. However, this overview provides a preliminary analysis and highlights the need for rigorous research and further RCTs in rehabilitation for individuals with genetic degenerative ataxia.
Conclusion
Despite scarce high-quality evidence evaluating rehabilitation interventions for individuals with degenerative ataxia, this review highlights emerging evidence that rehabilitation may improve function, mobility, ataxia and balance in genetic degenerative ataxia. Although these conclusions are based primarily on moderate to low-quality studies, the consistency of positive effects verifies that rehabilitation is beneficial. Thus, this review provides valuable encouragement and preliminary guidance for rehabilitation clinicians in the treatment of individuals with degenerative ataxia. One RCT has provided evidence to support inpatient rehabilitation for ambulant individuals with pure cerebellar ataxia. However, further high-quality multisite RCTs are urgently required to provide conclusive evidence as to the efficacy of rehabilitation interventions, particularly for individuals with afferent ataxia and for those who are nonambulant. Therapies should include multifaceted interventions targeting more than one area of impairment. The next challenge is to extend these results further, to determine the optimal intensity, duration, and type of intervention. This will ensure individuals with genetic degenerative ataxia are provided with the best possible evidence-based care.
Footnotes
Acknowledgements
The authors would like to thank the Monash Health Physiotherapy Department for its support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Friedreich Ataxia Research Association (Australasia), the Friedreich Ataxia Research Alliance (United States of America), and Murdoch Childrens Research Institute for their ongoing financial support. SCM is a recipient of a National Health and Medical Research Council of Australia Postgraduate Scholarship (APP1093259), EMY is an Early Career Fellow of the National Health and Medical Research Council of Australia (APP1073323).
