Abstract
Functional recovery following central nervous system injuries is strongly influenced by rehabilitative training. In the clinical setting, the intensity of training and the level of motivation for a particular task are known to play important roles. With increasing neuroscience studies investigating the effects of training and rehabilitation, it is important to understand how the amount and type of training of individuals influences outcome. However, little is known about the influence of spontaneous “self-training” during daily life as it is often uncontrolled, not recorded, and mostly disregarded. Here, we investigated the effects of the intensity of self-training on motor skill acquisition in normal, intact rats and on the recovery of functional motor behavior following spinal cord injury in adult rats. We used a custom-designed small animal tracking system, “RatTrack,” to continuously record the activity of multiple rats, simultaneously in a complex Natural Habitat–enriched environment. Naïve, adult rats performed high-intensity, self-motivated motor training, which resulted in them out-performing rats that were conventionally housed and trained on skilled movement tasks, for example, skilled prehension (grasping) and ladder walking. Following spinal cord injury the amount of self-training was correlated with improved functional recovery. These data suggest that high-impact, self-motivated training leads to superior skill acquisition and functional recovery than conventional training paradigms. These findings have important implications for the design of animal studies investigating rehabilitation and for the planning of human rehabilitation programs.
Introduction
Behavioral experience is one of the most potent modulators of central nervous system (CNS) structure and function.1,2 For example, learning a motor skill is associated with dendritic changes and cortical map reorganization2-5 and skilled prehension training after a cortical lesion6-9 or locomotor training after a spinal lesion10-13 leads to greater functional recovery, meaning the regaining of preinjury movement patterns post-injury, in both animals and humans. In addition, following injury, enhanced activity, both behavioral and neuronal (electrical), following a unilateral pyramidotomy lesion in the brainstem positively influences plastic sprouting in the spinal cord.14-16
Possible explanations for the differences in functional outcome following CNS injury in both preclinical and clinical data relate to the amount of time spent actively training, or the dose of rehabilitation, which, in comparison to the nontraining time period, is often very little.17,18 In this case, “motor training” refers to the extended practice of a goal-directed task that leads to motoric improvements in performing that particular task, that is task-specific improvements. This should be distinguished from “motor learning,” which instead refers to the better selection of actions and the improved execution of these actions for performing a particular task that follows successful motor training. Hence, successful motor training would result in motor learning. The dose of training is increasingly being seen as an important feature of rehabilitation programs19,20 but no clear guidelines for the actual amount required to see significant changes currently exist. 21 “Self-training,” that is, activity within the home cage (animals) or outside of the clinical rehabilitation program (humans), is difficult to control and assess experimentally but could interfere with the effects of the specific training. For example, the effects of self-training were investigated in adult rats following spinal cord injury. To eliminate the effects of self-training in the home cage the group immobilized the hindlimbs with specially designed wheelchairs. They found that immobilization (15-18 hours per day, 5 days per week) impaired spontaneous recovery of hindlimb locomotor function. 22 This study nicely illustrates the importance of self-training for the normal course of recovery from spinal cord injury. Environmental enrichment, that is, experimental animal housing conditions that provide increased opportunities for motor and sensory stimulation and self-training, is one possible way to influence self-training and has been used with positive effects on motor outcome in a number of preclinical spinal cord injury experiments23,24 and stroke experiments 25 ; for reviews, see Cramer 26 and Nithianantharajah and Hannan. 27 However, the high variability observed in these studies points to large interindividual differences in the amount (dose) and type of training, an aspect that was not investigated in these earlier studies.
In preclinical studies, one method to assess activity levels experimentally is to track individuals. Some behavioral radiofrequency identification (RFID)–based tracking systems for small animals do exist28,29; however, they work in 2-dimensional (2D) environments and focus on investigating social interactions. As our aim was to investigate the effects of enhanced self-training we needed a different system that could function robustly within a complex, 3D enriched environment (EE). We developed a custom-designed tracking system (RatTrack) and were able to accurately measure the activity of multiple individual rats simultaneously over long time periods. We report that naïve animals housed in the Natural Habitat Cage (NH Cage), which is an example of a complex EE, for 4 weeks outperform standard housed and conventionally trained naïve rats on a number of motor function tasks. We observed that following spinal cord injury, activity within the NH Cage correlated with lesion size and functional recovery.
Material and Methods
Animals
Adult female Long-Evans rats (200-250g, n = 61) were obtained from a specific pathogen-free breeding colony (Charles River, Erkrath, Germany). Animals were kept on a standard regimen of 12-hour light/dark and food and water ad libitum. All experiments were performed in accordance with the guidelines of the Veterinary Office, Zurich, Switzerland.
Transponder Implantation
Animals were anesthetized with isoflurane (5% in O2) and implanted subcutaneously with 2 RFID transponders (ISO Standard 11784; T-IS 8010 FDX-B, Datamars, Switzerland) one in the neck and one above the tail. Each transponder had a unique RFID code. All rats were implanted with 2 RFID transponders, so that only when consecutive signals from both were recorded at the same time was it counted as an antenna passage.
Antennas
For details about the specifications of the antennas, see Supplementary Information.
Nighttime Infrared Video Recordings
The grasping boxes, grasping grid, and wheel were filmed at night with an infrared camera (TVCCD 118 B/W camera, Monacor Schweiz AG, Risch, Switzerland) and infrared illumination (IR-12/65LED, Monacor Schweiz AG). Videos were analyzed using VirtualDub (VirtualDub 1.7.8, Avery Lee, http://virtualdub.org). For each training device, the percentage of entries that resulted in active training was calculated.
Behavioral Training
Naïve animals were divided into 3 groups: nontrained (n = 10), conventionally trained (n = 19), and NH Cage trained (n = 10). All naïve–conventionally trained rats were pretrained on the single pellet skilled prehension and horizontal ladder tasks (see below) for 4 weeks. Spinal cord injured and naïve–NH Cage training rats were housed in the NH Cage in separate experiments, that is, the naïve animals were part of the first experiment and were housed in the NH Cage while their control group were housed in conventional cages. Following the completion of this experiment, the SCI animals were housed in the NH Cage while their controls were housed conventionally. For full details of the training paradigms, see Supplementary Information.
Naïve rats (nontrained) were housed under standard housing conditions (4-5 rats/cage, 1 plastic tube, food and water ad libitum). Sugar pellets were given 2× per week and animals were acclimatized to the behavioral tasks. Additionally, we exposed these rats to the same novel food items that were given in the “Penthouse” area of the NH Cage at the same frequency and amount in both.
Naïve rats (conventionally trained) were also housed under standard housing conditions. Additionally, as above, we exposed these rats to the same novel food items that were given in the Penthouse area of the NH Cage at the same frequency and amount in both.
Naïve rats (NH Cage trained) were housed in the NH Cage after 4 to 5 days of standard housing. The rats remained in the NH Cage for 4 weeks. Naïve rats were handled and acclimatized to the behavioral tests and sugar pellets as described for Naïve rats (non-trained).
Behavioral Testing
Testing of the naïve animals on the behavioral tests was performed on days 1 to 3 after the completion of training (detailed above). The single pellet skilled prehension task was carried out as described previously. 30 Prior to testing all NH Cage animals were removed from the NH Cage and kept in conventional cages and food deprived for the three consecutive tests of grasping. These rats remained out of the NH Cage during the entire testing period for these tests. Skilled prehension success was scored as described previously 16 for the preferred forelimb only. The horizontal ladder test was carried out as previously published.14,31 Forepaw placement was scored as was done previously. 14 The staircase test was carried out and scored as described previously.16,32
Spinal Cord Injury Group
All spinal cord injured rats (n = 22) were pretrained for 6 sessions on each task of the MotoRater 33 prior to lesion. Following baseline recordings, bilateral dorsal hemisections at T9 (n = 22) were performed as reported previously. 33 Animals were checked twice daily for the entire experiment and bladders were manually expressed until normal function returned. Half of the spinal cord injured rats (n = 11) were housed in the NH Cage from day 3 post-lesion onward for 4 weeks in total, whereas the remaining spinal cord injured rats (n = 11) were conventionally housed for 4 weeks. Spinal cord injured rats had baseline recordings taken on the MotoRater 33 prior to lesion and were then tested on days 3, 8, and 34 post-lesion. The MotoRater was carried out and analyzed as described previously. 33 At the completion of behavioral testing, spinal cord injured rats were perfused as described previously. 16 Transverse sections (50 µm) of the spinal cord containing the lesion site (T1-L2) were cut on a cryostat. In order to measure the percentage of remaining tissue at the lesion sites, cresyl violet–stained sections were reconstructed in 3D using Neurolucida (MBF Bioscience, Magdeburg, Germany).
Statistical Analysis
All behavioral data from naïve animals were analyzed using one-way analysis of variance (ANOVA) with Bonferroni post hoc analysis. Behavioral data from the spinal cord injured animals were analyzed using two-way ANOVA with Bonferroni post hoc analysis. Data where only one time point was used were analyzed using an unpaired t test. Spearman correlations were carried out between the success rate on the horizontal ladder and the number of ladder passages. All statistical analyses were conducted using GraphPad Prism software. Data are presented as means ± standard error of the mean, single data points represent single animals, and asterisks indicate significances: *P < .05, **P < .01, and ***P < .001 and P < .0001.
Results
The Natural Habitat Cage Allows Intense, Nonforced Self-Training of Various Behaviors
In order to investigate self-training, we first investigated how often a passage into a training device resulted in training. To do this, we analyzed nighttime infrared video recordings of the wheel, grasping boxes, and grasping grid in conjunction with the RatTrack tracking data. On average 96.5% of the entries through the wheel antenna resulted in training on the wheel (black bar, Figure 1G). In the grasping boxes, 79.2% of the entries resulted in grasping, whereas for the grasping grid, 51.1% of entries resulted in grasping training (black bars, Figure 1G). The lower value for the grasping grid is because of the fact that the animals liked to sleep in this secluded area.

RatTrack is a novel small animal tracking system contained within the Natural Habitat (NH) Cage, an example of an enriched environment, able to accurately track the activity of multiple animals. (A) The NH Cage comprises three floors, each containing specific tasks to train the fore- and/or hindlimbs. The ground floor includes a standard home cage, a tube, and a ladder to the first floor. The first floor contains a number of devices for forelimb training: grasping boxes (B and F), grasping grid (C and G), and a rope. In addition, it also contains a house and two ladders (appear as blue diagonal lines). The second floor contains locomotor training devices: a running wheel (D), a small ladder, and two beams, as well as a penthouse containing food treats (E). (F) A higher power image of one grasping box to show the design. Five of these were attached together to comprise the grasping boxes in the NH Cage. (G) A higher power image of the grasping grid to show the wells of the grid filled with sugar pellets and seeds. Eight custom-designed antennas comprise the tracking system and are each placed at the entry to the training devices. (H) The design of the NH Cage and tracking system does not allow certain sequences of passages. Red circles represent antennas and solid black lines represent possible movements between them. (I) Nighttime infrared video recordings of the wheel, grasping boxes, and grasping grid were used to assess the amount of training. On average, 96.5% (black bar) of the entries through the wheel antenna resulted in training. Values were lower for the grasping boxes (79.2%) and grasping grid (51.1%).
These data allowed us to compare the amount of training animals in the NH Cage performed in comparison with conventional, task-specific training. In our lab conventional, experimenter-assisted ladder walk training of rats consists of 3 to 10 passages of a 1-m long horizontal ladder 5 days a week, which is comparable to published data. 34 In the NH Cage, rats ran on average 117.3 ± 6.4 m of ladder per day, 7 days a week, which was significantly different from conventionally trained rats (P < .0001, unpaired t test, least active animal, 77.3 m per day; most active animal, 139.6 m per day). Animals within the NH Cage spent 187.9 ± 33.6 min/wk grasping, which is considerably more than conventional grasping training, which, in our lab, is 50 min/wk again, comparable to previously published data.30,35,36 The RatTrack readings showed that there were large variations between individual animals with the least active animal grasping for 58.5 min/wk and the most active animal grasping for 286.2 min/wk. In the running wheel, RatTrack recordings showed that naïve animals ran on average 685.9 ± 114.3 m per day (least active animal, 261.8 m per day; most active animal, 1346.9 m per day).
Adult rats, housed in the NH Cage were most active during the dark (Figure 2A). The total activity of each animal (all RatTrack recordings from all 8 antennas over the 4-week period) varied across the animals from 10 130 to 18 406 passages over the 4 weeks, and revealed that the most active animals were 70% to 80% more active that the least active rats (Figure 2B). In general, the locomotor tasks (ladders and wheel) were used more frequently than the grasping tasks (grasping grid, grasping boxes, and penthouse). For the wheel, the grasping grid and the house, the intensity of use varied 2 to 5 times between individual animals. Interestingly, the tracking data also showed that individual animals had preferences for certain tasks (Figure 2C and D). By day 8, the animals were familiar with the NH Cage and this was reflected in the activities they performed, which varied very little in the remaining 3 weeks in the NH Cage (Figure 2E) or throughout any given week (Figure 2C-E).
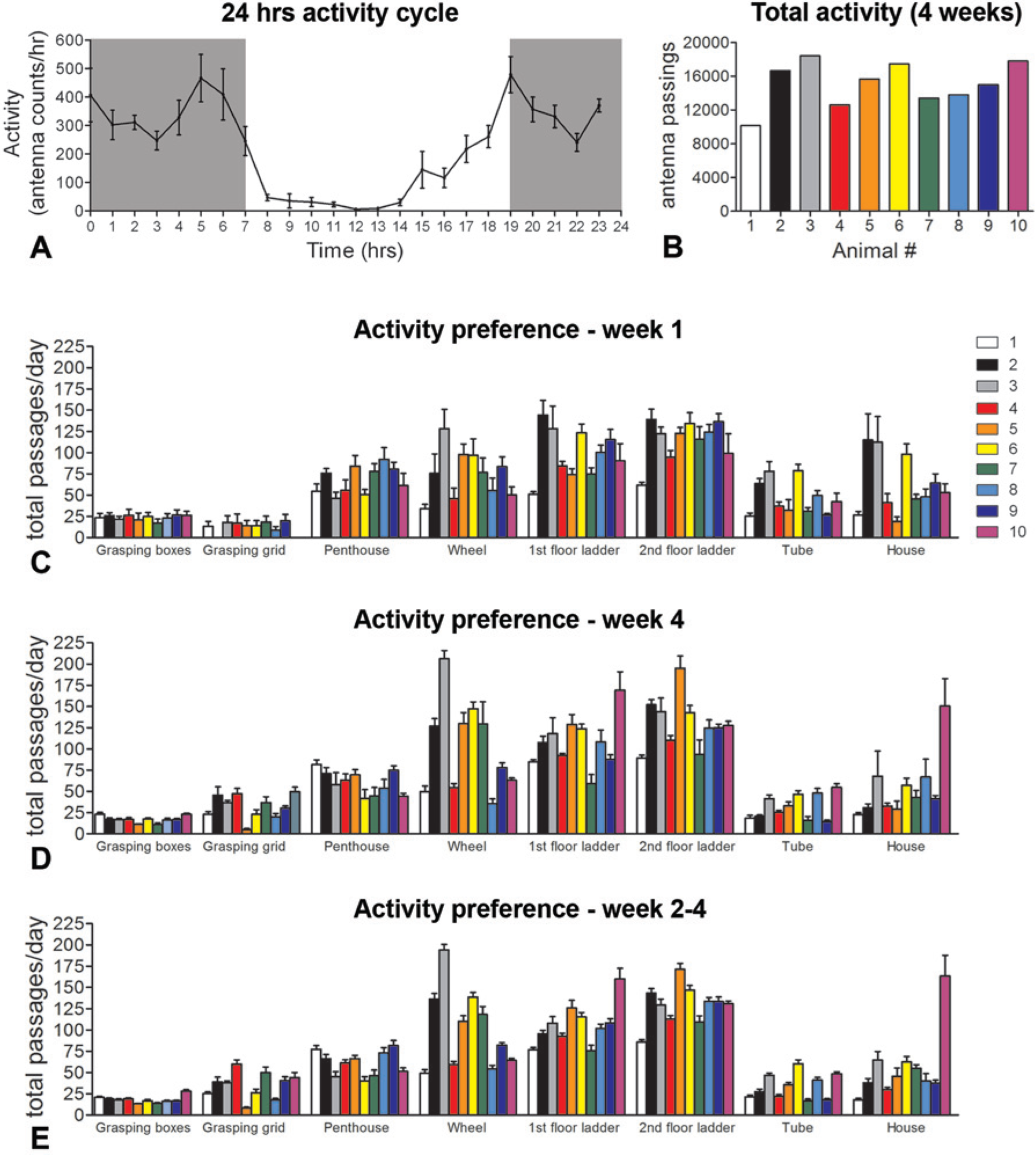
RatTrack provides information about the self-training behavior of individual animals within the Natural Habitat (NH) Cage. (A) A 24-hour activity profile of adult rats in the NH Cage. Rats are more active during the dark phase (gray boxes) than during the day time (white section). (C and D) Activity profiles and preferences of individual rats (1-10) during the first (C) and fourth week (D). (E) Activity profiles and preferences of individual rats during the final three weeks in the NH Cage (weeks 2-4) showing low intra-animal variation. Data are presented as means ± standard error of the mean.
Self-Training Leads to Enhanced Functional Outcomes Compared With Conventional Training
We compared the motor function ability of rats housed in the NH Cage for 4 weeks with standard housed rats, which were either nontrained or conventionally trained. The single pellet skilled prehension task is a demanding task requiring highly skilled forelimb movements. Skilled prehension is one of the most difficult motor tasks for a rat to learn. Therefore, the aim was to test whether it was possible for rats to gain a high level of prehension skill from the NH Cage alone, that is, whether EE, including several opportunities for skilled prehension, would provide an effective training for prehension. This had not been seen previously when the grasping training was included within the EE 37 but positive effects were seen when it was given separately but in addition to EE.25,38 Over 3 consecutive trial days despite making a similar number of grasping attempts as the other groups nontrained rats performed poorly at this grasping task achieving a success rate of only 5.5% ± 4.9% (first trial), 7.0% ± 5.0% (second trial), and 5.8% ± 4.1% (third trial; Figure 3A). Rats trained conventionally on the single pellet grasping task (25 pellets grasped per day, for 20 days, 10-min sessions) were significantly better than nontrained rats and had success rates of 56.9% ± 3.9% (first trial), 58.9% ± 3.5% (second trial), and 58.2% ± 3.2% (third trial; Figure 3A, P < .0001, one-way ANOVA). Whereas, NH Cage housed rats that had grasped in 2 separate paradigms (grasping boxes and grasping grid) the NH Cage but were completely unfamiliar with the single pellet task showed a steep learning curve 10.8% ± 7.9% (first trial), 53.8% ± 11.2% (second trial), and 77.5% ± 9.5% (third trial), thus outperforming the other groups by the final test session (Figure 3A, P < .0001, one-way ANOVA).
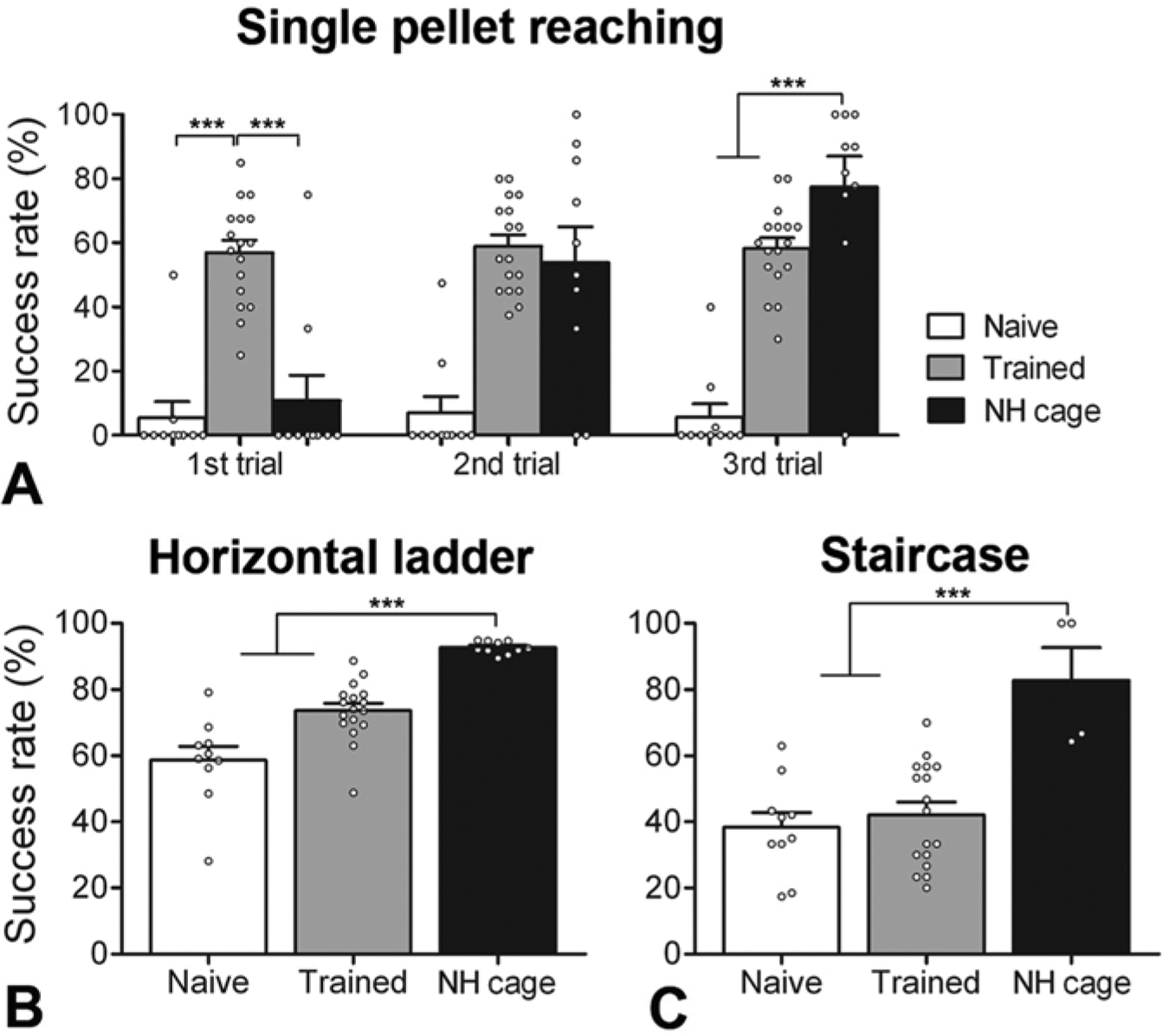
Intense, nonforced, self-training of multiple behavioral tasks leads to enhanced functional ability in naïve, adult rats. (A) Rats housed in the Natural Habitat (NH) Cage for four weeks were compared with nontrained/standard housed rats and standard housed rats that had been trained conventionally on the selected tasks. Nontrained rats performed poorly on the single pellet grasping task on each of the three trial sessions (one trial per successive day). Rats trained on this task performed well and consistently during testing. NH Cage housed rats rapidly improved their performance on this task during testing such that they outperformed the trained rats by the third trial session. (B) Rats housed in the NH Cage outperformed nontrained rats and conventionally trained rats on the horizontal ladder task. (C) All rats were tested on the staircase as a novel task. NH Cage housed rats again performed significantly better on this task than the other groups (P < .001, one-way analysis of variance). Data are presented as means ± standard error of the mean; open circles indicate individual animals. ***P < .001.
Housing rats in the NH Cage also affected their ability to cross a horizontal ladder. Nontrained rats had a success rate for correct, precise foot placement on the rungs of 58.6% ± 4.2% (Figure 3B) when crossing the ladder whereas rats conventionally trained to cross the horizontal ladder (3-4 crossings per day of a 1-m long ladder over 12 days) had a success rate of 73.6% ± 2.2% (Figure 3B). Rats that had been housed in the NH Cage, which contained several ladders that were somewhat different from the test (diagonally positioned and regular rung placement), had a success rate of 92.7% ± 0.6%, which was significantly better than the 2 other groups (Figure 3B, P < .0001, one-way ANOVA).
Most interestingly, we tested all rats on a novel task, the Staircase task (rats had to grasp up to 3 food pellets from different height steps on a bilateral staircase), which none of the groups had experienced previously. NH Cage housed rats, which had self-trained grasping within the NH Cage, performed best on this task achieving a success rate of 82.7% ± 10.0% (Figure 3C, rats that made no attempts to grasp during their Cage time were removed from the analysis), which was significantly better than the standard housed/nontrained (38.3% ± 4.5%) and standard housed/conventionally trained rats groups (42.2% ± 3.8%, Figure 3C, P < .0001, one-way ANOVA). Thus, rats housed in the NH Cage for 4 weeks with enhanced opportunities for self-training, outperformed nontrained and conventionally trained rats on all tasks.
Recovery Following Spinal Cord Injury From Self-Initiated Training
In order to test the ability of the NH Cage to enhance motor recovery post-lesion, we chose a spinal cord injury model. The reason for choosing a spinal cord injury instead of another CNS injury model such as a stroke was first, because this lesion model is extensively used in the field to test a large variety of lesion interventions,39,40 and second, it is a harsh injury affecting gross locomotor function, and thus a good test of whether such animals could use the NH Cage. Finally, the majority of the NH Cage contains tasks that do not require highly skilled training, for example, ladders, beams, rope, running wheel, and so on and thus using a model that affects the forelimb, such as a stroke or pyramidotomy, would not test the NH Cage to its full potential as most of the tasks would be easily accessible for animals with such a lesion model. In order to test the effects of enhanced self-training on recovery of function following spinal cord injury rats received thoracic dorsal hemisection lesions, which were made intentionally either large (about 60% of the spinal cord cross-section, including dorsal funiculi, dorsal gray matter, and part of the lateral funiculi, Figure 4A and C) or small (dorsal columns with corticospinal tract only, Figure 4B and D). Rats were housed in the NH Cage for 4 weeks after the injury. They were tested using standardized locomotor assessments from the MotoRater: horizontal ladder (correct foot placement, Figure 4J), overground locomotion (BBB [Basso–Beattie–Bresnahan] score, Figure 4K), and swimming (compensatory forelimb use, Figure 4L) at 3, 8, and 34 days post-lesion. At the end of the experiment, the lesion sites were reconstructed (Figure 4C and D) from histological sections (Figure 4A and B), and the rats were assigned to either the small (Figure 4A and C) or large (Figure 4B and D) lesion group.
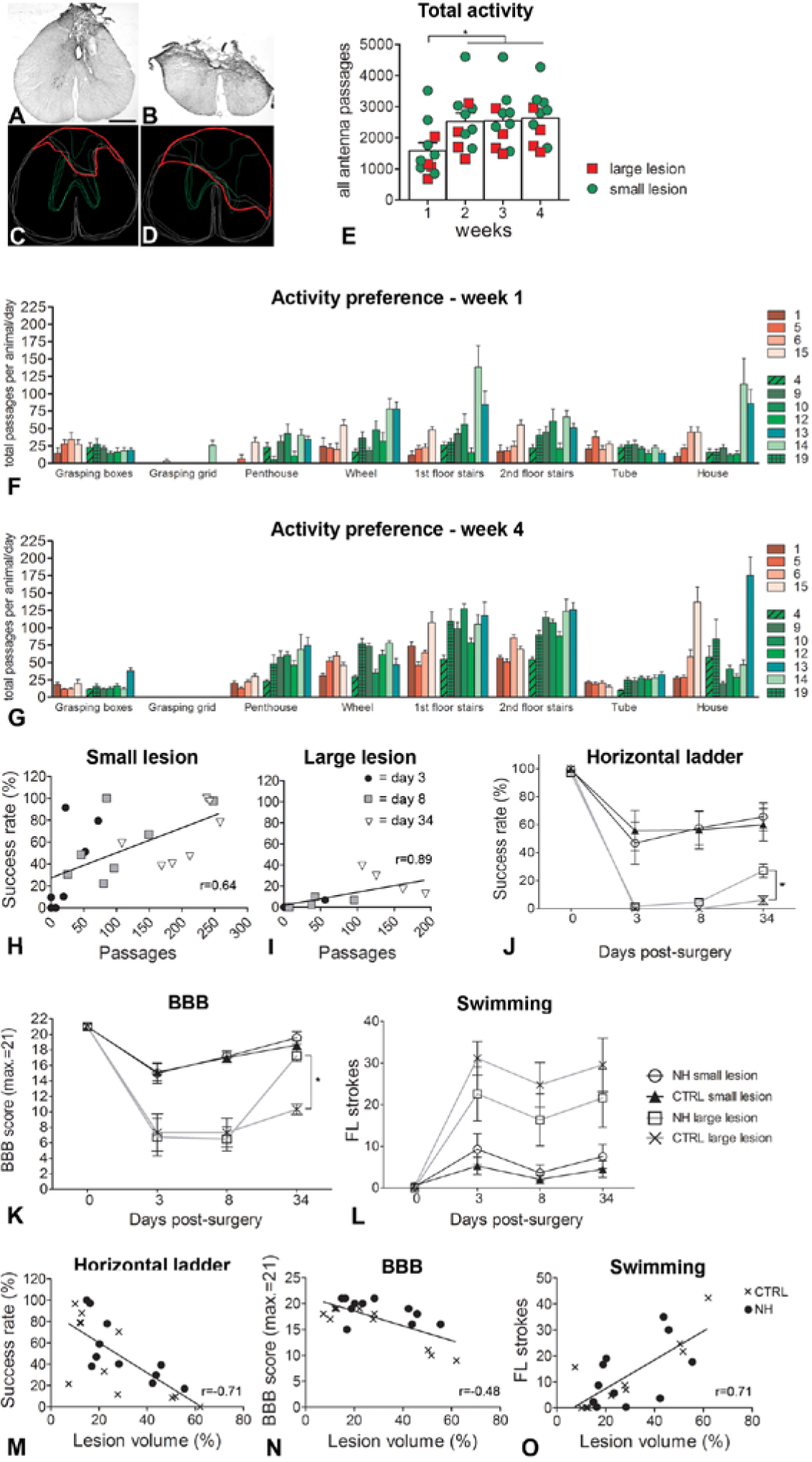
Self-training is correlated with functional recovery following spinal cord injury (SCI). Rats received dorsal hemisection lesions of the thoracic spinal cord producing small (A and C) and large (B and D) lesion groups. (E) Injured rats were significantly less active in week 1 than in weeks 2 to 4 (P < .001, Mann–Whitney). Rats with large lesions (red squares) were less active than those with small lesions (green dots). Individual activity profiles for week 1 (F) and week 4 (G) showed partial and lesion-dependent recovery of locomotor activities. Both the small (green series of bars) and large (red series of bars) lesion groups contained active and inactive rats. (H) The number of self-motivated passages individuals with small lesions made across the ladders was directly correlated with their success rate on the horizontal ladder test (P < .01, r = 0.64, Spearman correlation). (I) In animals with large injuries, there was also a correlation between the number of self-motivated passages across the ladders in the Natural Habitat (NH) Cage and their success rate on the horizontal ladder test (P < .001, r = 0.89, Spearman correlation). (J) By day 34 post-lesion, NH Cage housed animals with large lesions recovered fine control of hindlimb function significantly better than those that were conventionally housed (P < .05, two-way analysis of variance). (K) NH Cage housed animals with large lesions recovered locomotor function by day 34 post-lesion (P < .05, two-way analysis of variance). (L) During swimming, NH Cage housed animals with large lesions performed better than those that were conventionally housed. (M) Lesion volume was highly correlated with success rate on the horizontal ladder (P < .001, r = −0.71). (N) Lesion volume was also correlated with BBB (Basso–Beattie–Bresnahan) score (P < .05, r = −0.48). (O) The lesion volume showed a high correlation with forelimb strokes during swimming (P < .001, r = .71). Data are presented as means ± standard error of the mean; open circles indicate individual animals. *P < .05.
As expected, the spinal cord injured rats were overall significantly less active in week 1 (1506 ± 2201 total antenna passages/wk/rat) than in weeks 2 to 4 (week 2 = 2393 ± 250, week 3 = 2409 ± 246, week 4 = 2468 ± 233, Figure 4E, P < .05, one-way ANOVA) and remained significantly less active than intact animals throughout the experiment (8776 vs 15 076 passages over 4 weeks, P < .001, Mann–Whitney 2-tailed test). Animals with large lesions (Figure 4E, red squares) were somewhat less active than animals with small lesions (Figure 4E, green dots). To investigate differences between the groups, we analyzed all activity through all antennas over the 4 weeks and found that the overlap between the two groups was remarkably high; in fact, there was no significant difference between the two lesion groups (Mann–Whitney t test, P = .16). There was also no significant difference between the two lesion groups on the separate antennae (Mann–Whitney t test, P > .05), except for the Penthouse antenna, which the small lesion animals visited significantly more often (Mann–Whitney t test, P < .05). This suggests that it was not the amount but the type of activity within the NH Cage that allowed NH Cage housed rats to perform better on the different motor tasks than those housed in conventional cages.
The RatTrack system allowed us to investigate the individual activity of each spinal cord injured rat (Figure 4F and G). We found that the grasping grid was not visited regularly. The penthouse was also visited infrequently in the first week post-injury but visits increased, in particular by the small lesion group (green series of bars, Figure 4G), by week 4 post-lesion. Interestingly, locomotor tasks were more popular than grasping ones (Figure 4F and G). As with intact rats, there were large individual differences, for example, in the use of the wheel, the first and second floor ladders, and the house (red and green series of bars in Figure 4F & G). However, the intra-animal variation was low within weeks 2 to 4 (and hence only data for week 4 are shown), suggesting that individual animals commonly visited the same tasks to the same extent from one day to the next.
To assess the importance of self-training on recovery of function after spinal cord injury we compared large and small lesioned rats housed in the NH Cage with those that were conventionally housed (control). On day 3 post-lesion, all rats showed deficits in crossing the horizontal ladder, with large lesioned animals performing considerably worse than those with small lesions, regardless of the housing conditions. However, by day 34 postlesion, NH Cage housed animals with large lesions performed significantly better than conventionally housed animals with large lesions (P < .05, two-way ANOVA, Figure 4J). We found that the number of self-motivated passages across the ladders in the NH Cage was highly correlated with success rate on the horizontal ladder test for both small (P < .01, r = 0.64, Spearman correlation, Figure 4H) and large (P < .001, r = 0.89, Spearman correlation, Figure 4I) lesion NH Cage housed groups.
To assess overall locomotor function we tested the open field locomotor behavior of all groups (BBB test 41 ). As with the horizontal ladder, all rats showed deficits on day 3 post-lesion with the large lesion group performing much worse than the small lesion group. Both groups with small lesions (control and NH Cage) recovered to baseline scores by day 34 post-lesion, regardless of housing condition (Figure 4K). However, most interestingly, by day 34 post-lesion, the large lesion NH Cage group recovered significantly better than those that were conventionally housed (P < .05, two-way ANOVA, Figure 4K). In fact, there was no significant difference between the performances of this group with that of the small lesioned animals by day 34 post-lesion.
We also assessed hindlimb function during swimming, an activity that was not trained by either group and was thus a novel task for all animals. All animals with large lesions were heavily impaired in this task at day 3 post-lesion meaning that they had higher numbers of forelimb strokes (Figure 4L). By day 34 post-lesion, NH Cage housed animals with large lesions performed better than those that were conventionally housed, but because of large individual variations, the difference in their performance was not significant.
Finally, we found that lesion volume was strongly correlated with success rate on the horizontal ladder (P < .001 r = −0.71, Figure 4M), the score on the BBB (p < .05 r = −0.48, Figure 4N) and with the number of forelimb strokes during swimming (p < .001 r = 0.71, Figure 4O) regardless of housing condition.
Discussion
After large CNS injuries, rehabilitative training of remaining and compensatory functions is the only option that currently exists. Different kinds of training programs are used in clinics, but there are no generally accepted schemes. Personal motivation is assumed to be key in human rehabilitation but this is based on anecdotal evidence as there are limited data on this and it is difficult to model in animals. Likewise, the daily use of recovered functions, or the dose of rehabilitation provided is thought to be crucial, 42 but there are very few systematic studies of this in humans and none to our knowledge in animals.19-21 Thus, we developed RatTrack, which allowed automated tracking of multiple animals freely moving about a complex 3D environment. When groups of 10 to 12 young adult, female, outbred Long-Evans rats were kept in the NH Cage for 4 weeks, individuals had a high propensity to visit similar tasks to the same degree, suggesting they had individual preferences for certain tasks. Also the overall activity levels differed between individuals. Motivation and personal preference probably make an important contribution to the variability often observed in behavioral readouts.
With increasing (preclinical and clinical) neuroscience studies investigating the effects of motor training, motor learning, and rehabilitation, it is important to understand how the amount and type of training influences outcome. In particular, there have recently been a number of studies investigating task-specific training from our own 16 and other labs.43,44 Some data suggest that specific training, and thus motor learning, of one task comes at the expense of other tasks 43 although we did not find this 16 . Thus, it is interesting to investigate the differences between task-specific training and the training provided by EEs as these two paradigms, both of which modify motor performance, may have very different impacts on motor learning and recovery, as reviewed by Zeiler and Krakauer. 42 In the present study, we directly investigated task-specific training versus EEs (Figure 3). Intact, adult rats were task-specifically trained on the single pellet skilled prehension task (Figure 3A) and the horizontal ladder task (Figure 3B) and were compared with intact, adult rats housed in the NH Cage, an example of EE. We found that those rats housed in the NH Cage performed better than the task-specifically trained rats on all tasks tested. We specifically designed the NH Cage so that it contained many opportunities for skilled prehension and ladder crossing and we showed that animals housed in the NH Cage performed a higher amount, and thus received a higher dose, of training than the task-specifically trained rats. However, in addition to this enhanced cognitive and somatosensory function, reduced fear that has been associated with EE 27 could have influenced the final performance of the NH Cage housed rats on the behavioral tasks tested. These findings are particularly notable as the comparison of training techniques such as task-specific versus EE training has only recently started to be investigated in the literature. 42 The findings that NH Cage housed animals outperform task-specifically trained rats highlights the importance of the NH Cage/RatTrack system as a research tool and training device because of its potential utility in motor learning, motor training, and ultimately, rehabilitation.
Tracking individual animals in all of their daily activities is an effective way to investigate the influence of self-training on functional outcome. Self-training can enhance or interfere with the desired functional outcome, especially if it comprises more of the day than the specific training.18,22,45,46 We found that the amount of individual self-training in the NH Cage following spinal cord injury was correlated with an improved functional outcome, although we cannot say whether this was true recovery of function, that is, the regaining of the same preinjury movements post-injury, or compensation, that is, the use of alternative movements to accomplish the same motor task. Furthermore, NH Cage housed animals with large lesions performed significantly better than animals with comparable lesions that were conventionally housed. In addition, nonforced, self-motivated training of multiple tasks in the NH Cage led to superior performance by naïve animals in tests requiring skilled fore- and hindlimb movements compared with standard housed animals conventionally trained on these specific tasks. NH Cage housed rats were able to translate grasping skills learnt in the NH Cage (grasping grid, grasping boxes, and penthouse) into skill on a novel skilled prehension task (single pellet grasping task) tested outside of the NH Cage. Despite the use of genetically similar (albeit outbred) adult rats of the same age, weight, sex, and supplier, we observed a surprisingly high variability in the activities that individual rats preferred to visit, that is, the self-training behavior of individual rats housed together for four weeks in a complex EE.
We report that housing rats in the NH Cage fostered learning of the single pellet skilled prehension task. This is an interesting finding because NH Cage housed rats performed better than non–NH Cage housed rats and those conventionally, task-specifically trained on the tested tasks. NH Cage housing also altered performance on the BBB test following spinal cord injury. This is of interest because there is no reason to expect that NH Cage housing alone would affect BBB performance in uninjured rats (because of ceiling effects). However, the fact that NH Cage housing per se improved performance on the ladder task in spinal cord injured rats is less astonishing because this occurred independent of injury. This implies that there are features of the NH Cage that enhance test performance generally, for example, similarity in motor tasks trained in the NH Cage and tested outside the NH Cage, or a more general feature, such as habituation to fear-eliciting aspects of the test paradigm. We believe that fear did not play a significant role in our studies as all animals were housed in the same room, all testing was done in the same room as the training took place, at the same time of the day as the training and by the same experimenter who performed the training. Additionally, all animals were habituated to all behavioral tasks prior to testing. What likely played a greater role was the enhanced self-training that the NH Cage rats likely received. This has been shown to be an important determinant of functional recovery following spinal cord injury in the past. 22 Also, as the NH Cage represents an example of EE, there are other features general to the EE situation that could enhance test performance such as enhanced learning, improved memory, superior cognitive and exploratory performance, and improved motor function. 27 We did not test any of these features directly; however, this would be an interesting future step for such experiments.
We used the ladder and grasping devices in the NH Cage for training because these tasks are well established for testing functional recovery of the fore- and hindlimbs after brain and spinal cord injuries31,47 and are frequently used for rehabilitative training in these paradigms.6,43 However, skilled grasping training has been shown to negatively affect recovery of other, related tasks such as crossing the horizontal ladder, 43 and environmental enrichment housing has been shown to interfere with recovery in some but not in other experiments.37,38,48,49 Despite this, environmental enrichment is widely accepted as a positive intervention and it has been shown following spinal cord injury that it can improve gross locomotor function,24,50 and following stroke it was shown to improve fine motor control.38,49 Here, we demonstrated a direct correlation between the intensity of training and the degree of skill (naïve animals) at locomotor and grasping tasks or functional recovery of locomotor tasks after thoracic spinal cord injury in individual rats. This shows the importance of high training volumes for successful recovery of function after large CNS lesions.
Automated tracking set-ups have the advantage that animals can be observed independent of an experimenter, whose presence might affect behavior. Until recently, small animal tracking set-ups had major limitations; they often could not track animals without disrupting the natural behavior and did not work within an elaborate, quasi-natural, 3D environment.51-53 However, the use of the RFID has altered this as it is minimally invasive and allows the investigation of social interactions within complex environments without disrupting the animals’ natural behavior.28,29 These previously published systems used 2D environments and focused on the analysis of social interactions between animals instead of training. Therefore, basing our RFID tracking system within a 3D environment and investigating rehabilitative training is, to our knowledge, novel. RatTrack combined with the NH Cage constitutes a reliable, versatile, and sensitive system for continuously measuring the self-training and nonforced behaviors of multiple animals simultaneously. RatTrack has several advantages compared to older animal tracking systems: multiple animals can be labeled for tracking without affecting their behavior; tracking of multiple animals can occur simultaneously and can take place in the home cage and also in the dark.
In conclusion, the present study explores the differences between task-specific training and enhanced self-training within an EE. We show that self-motivated training of multiple tasks within the NH Cage leads to superior performance in tests requiring skilled fore- and hindlimb movements than restricted task-specific training. This suggests that there are key features of the NH Cage that allow for more superior training, in particular the high intensity of self-motivated training. This is supported by the finding that the amount of self-training within the NH Cage was correlated with functional recovery such that NH Cage housed animals with large spinal cord injuries recovered significantly better than animals with comparable lesions but conventional housing. In particular, NH Cage housing altered performance on the BBB test, which is of interest as this is not expected to occur in uninjured rats. Additionally significant was the finding that naïve animals that self-trained in the NH Cage outperformed conventionally trained animals on tests of fine motor control of the forelimb. We also showed that adult rats developed what appeared to be relatively stable individual preferences for certain activities. Such “personality profiles” probably reflect motivation and are often disregarded in behavioral analyses but likely play an important role in learning and functional outcome in rehabilitation. The NH Cage/RatTrack system allows the modeling of human rehabilitation and addresses key questions such as self-initiated and task-specific training and the role of dose.
Footnotes
Acknowledgements
Special thanks to the late Toni Lipp for his belief in this project and his enthusiastic backing to get it underway. We are especially saddened that he never got to see our collaboration realized. The authors would also like to thank Dr Guillermo Garcia-Alias and Dr Dale Corbett for providing us with the building instructions for the grasping grid and grasping boxes, respectively, and also for helpful discussions, Lukas Bachmann for help with surgery, and Lorenzo Tanadini for help with statistics.
Authors’ Note
Michelle L. Starkey is presently at the University of Zurich, Balgrist University Hospital, Zurich, Switzerland and Björn Zörner is presently at the Neurology Clinic, University Hospital Zurich, Zurich, Switzerland.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors’ lab is supported by grants of the Swiss National Science Foundation (Grant No. 3100AO-122527/1), the National Centre for Competence in Research “Neural Plasticity & Repair” of the Swiss National Science Foundation, The European Union’s Seventh Framework Programme (FP7/2007-2013) under grant agreements No. 201024 and No. 202213 (European Stroke Network, ![]() ), the European Commission under the Seventh Framework Programme–HEALTH–Collaborative Project Plasticise (Contract No. 223524), and the Spinal Cord Consortium of the Christopher and Dana Reeve Foundation.
), the European Commission under the Seventh Framework Programme–HEALTH–Collaborative Project Plasticise (Contract No. 223524), and the Spinal Cord Consortium of the Christopher and Dana Reeve Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
