Abstract
Background. Sleep-wake disturbances are among the most persistent sequelae after traumatic brain injury (TBI) and probably arise during the hospital stay following TBI. These disturbances are characterized by difficulties sleeping at night and staying awake during the day. Objective. The aim of the present study was to document rest-activity cycle consolidation in acute moderate/severe TBI using actigraphy and to assess its association with injury severity and outcome. Methods. In all, 16 hospitalized patients (27.1 ± 11.3 years) with moderate/severe TBI wore actigraphs for 10 days, starting in the intensive care unit (ICU) when continuous sedation was discontinued and patients had reached medical stability. Activity counts were summed for daytime (7:00-21:59 hours) and nighttime periods (22:00-6:59 hours). The ratio of daytime period activity to total 24-hour activity was used to quantify rest-activity cycle consolidation. An analysis of variance was carried out to characterize the evolution of the daytime activity ratio over the recording period. Results. Rest-activity cycle was consolidated only 46.6% of all days; however, a significant linear trend of improvement was observed over time. Greater TBI severity and longer ICU and hospital lengths of stay were associated with poorer rest-activity cycle consolidation and evolution. Patients with more rapid return to consolidated rest-activity cycle were more likely to have cleared posttraumatic amnesia and have lower disability at hospital discharge. Conclusions. Patients with acute moderate/severe TBI had an altered rest-activity cycle, probably reflecting severe fragmentation of sleep and wake episodes, which globally improved over time. A faster return to rest-activity cycle consolidation may predict enhanced brain recovery.
Introduction
Sleep disturbances are among the most persistent and disabling sequelae after traumatic brain injury (TBI), reported by more than 50% of patients1-6 and are known to compromise recovery in chronic TBI. 7 These disturbances are characterized by difficulties sleeping at night and staying awake during the day.2,8-11 Although sleep-wake disturbances are common in TBI patients, little is known about their origin and evolution. They probably appear in the first days after injury, when patients are hospitalized in the intensive care unit (ICU). A recent study carried out in the postacute phase of TBI reported that mild to severe sleep-wake disturbances were present among 84% of patients on rehabilitation admission and persisted for 66% of patients 1 month postinjury. 12 Although no sleep studies have been performed among TBI patients in the ICU, previous ICU studies in non-TBI patients have shown that both sleep-wake cycle and sleep architecture are highly disturbed,13,14 with a large proportion of sleep occurring during the day and up to 96% of total sleep spent in the lighter, less restorative sleep stages (stages 1 and 2).15,16
Sleep disturbances in the ICU are likely to have the same deleterious effects as chronic sleep restriction in healthy individuals on cognition, blood pressure, 17 glucose metabolism, 18 activation of the hypothalamic-pituitary-adrenal axis, and inflammatory response.19-21 The lack of deep sleep can have severe consequences during early rehabilitation of patients with TBI by slowing processes of physical recovery and exacerbating cognitive and neurobehavioral impairments, especially hippocampal-dependent memory processes. 22 Thus, there is a critical need to understand the nature and evolution of sleep disturbances in acute moderate to severe TBI, from the ICU to hospitalization in regular wards.
Polysomnography is the gold standard for measuring sleep architecture; however, it does not enable long-term measurement and is poorly tolerated by highly monitored or confused patients. An interesting alternative is wrist actigraphy, a valuable instrument to measure long-term rest-activity cycle in a clinical setting. In healthy individuals, the rest-activity cycle has been strongly correlated with the sleep-wake cycle and is often used as an indirect measure of the sleep-wake cycle. 23 Indeed, because of its low invasiveness and cost, actigraphy has been successfully used among different clinical populations.24-27
The aim of the present study was to document rest-activity cycle consolidation during the acute phase of moderate and severe TBI using actigraphy. Moreover, this study aimed to explore the clinical characteristics and outcome measures correlated with prolonged absence of rest-activity cycle consolidation. We hypothesized that patients would exhibit a severely disturbed rest-activity cycle that would improve over time. Our second hypothesis was that poorer rest-activity cycle consolidation would be associated with more severe TBI and poorer outcome at hospital discharge.
Methods
Participants
The study comprised 16 hospitalized patients (mean age = 27.1 ± 11.3 years) with moderate or severe TBI recruited from Hôpital du Sacré-Coeur de Montréal, a level-1 trauma center (see Table 1 for patient characteristics). The study was approved by the hospital ethics committee. Consent for participation was obtained from patients’ families. Patients who eventually became cognitively able to provide informed consent for themselves were asked to sign a consent form for study protocol to continue.
Demographic and Clinical Characteristics of Patients.
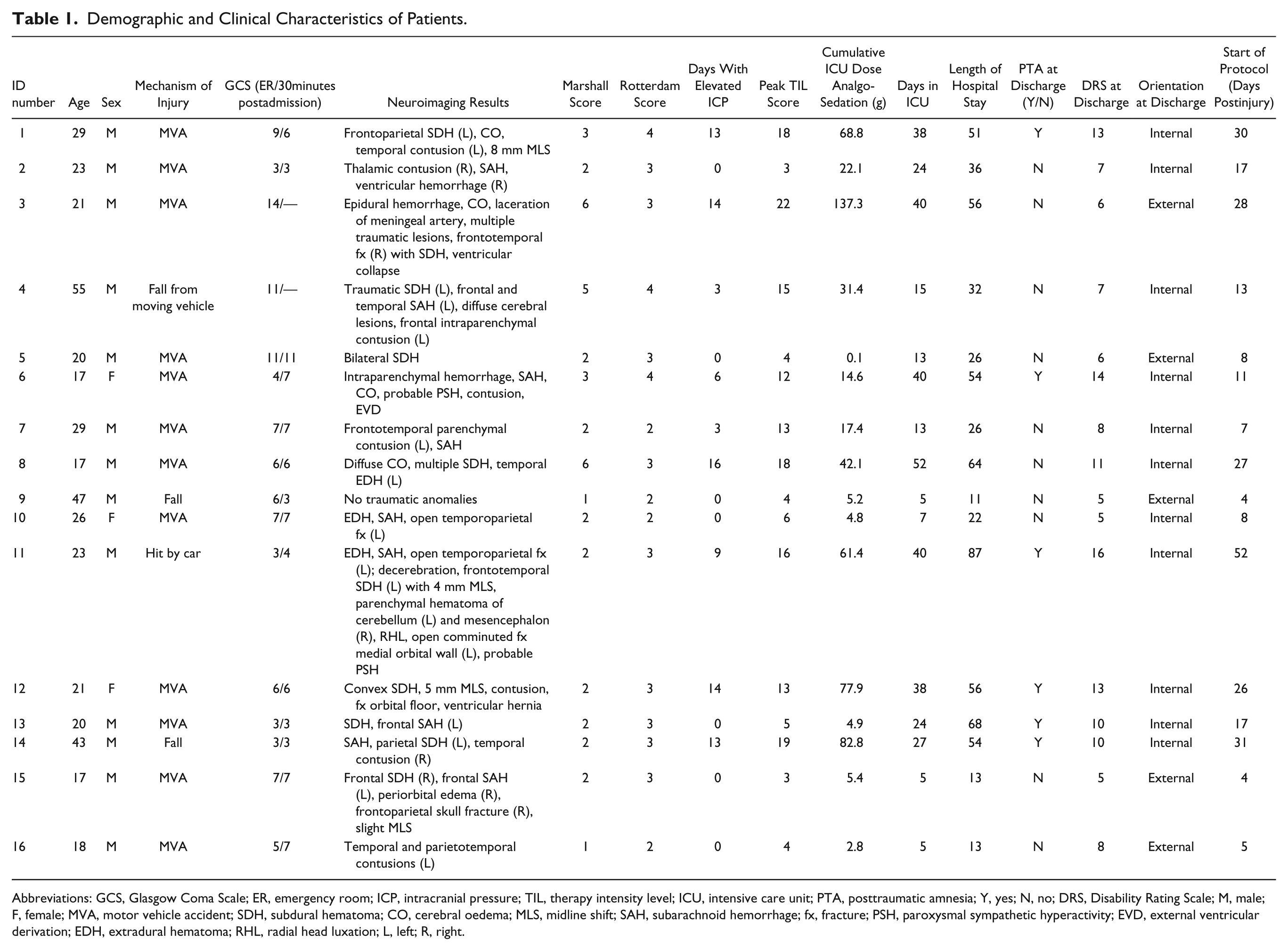
Abbreviations: GCS, Glasgow Coma Scale; ER, emergency room; ICP, intracranial pressure; TIL, therapy intensity level; ICU, intensive care unit; PTA, posttraumatic amnesia; Y, yes; N, no; DRS, Disability Rating Scale; M, male; F, female; MVA, motor vehicle accident; SDH, subdural hematoma; CO, cerebral oedema; MLS, midline shift; SAH, subarachnoid hemorrhage; fx, fracture; PSH, paroxysmal sympathetic hyperactivity; EVD, external ventricular derivation; EDH, extradural hematoma; RHL, radial head luxation; L, left; R, right.
Inclusion/Excluson Criteria
TBI was defined as an alteration in brain function or other evidence of brain pathology caused by an external force. 28 TBI severity was assessed on emergency room admission using the Glasgow Coma Scale (GCS) 29 and was reassessed 30 minutes later to provide a postreanimation index of TBI severity. Patients were included if they had a GCS score of 3 to 12 on emergency room admission and had to be hospitalized in the ICU. One person with a GCS score of 14 at emergency room admission was included because he subsequently suffered elevated intracranial pressure (ICP), rapidly dropped to a GCS of 3, and received a diagnosis of severe TBI.
Patients were excluded if they were nonfluent in French or English before injury; were quadriplegic; had a history of substance abuse, psychiatric, or neurological disorders; or had diagnosed sleep disorders. Such information was obtained from patients’ families through psychosocial assessments carried out by the ICU social worker and were confirmed by research assistants. Patients were also excluded for pregnancy, incarceration, and homelessness or if they suffered any damage to both eyes or the optic nerve (modifying light perception).
Protocol
Clinical Variables
Injury severity
Length of ICU stay, hospital length of stay, duration of continuous sedation/analgesia, number of days with elevated ICP (>20 mm Hg), Marshall 30 and Rotterdam 31 scores, and the summary therapy intensity level (TIL) 32 were documented for each day in the ICU. Cumulative administered dose of sedative and analgesic medication (lorazepam, midazolam, propofol, morphine, hydromorphones, and fentanyl) in the ICU was calculated according to the previously described method. 33 Sedative, analgesic, antiepileptic, and neuroleptic medications were also noted for all days of actigraphy recording that took place in the regular units (see supplementary materials for details).
Cognitive and functional outcome
Cognitive and behavioural functions were assessed daily by occupational therapists using the Rancho Los Amigos Scale 34 and the Galveston Orientation and Amnesia Test (GOAT). 35 Duration of posttraumatic amnesia (PTA) was calculated from the day the patient reached a GCS score of 9, as suggested in the standard GOAT protocol. 35 Emergence from PTA was designated as the first of 2 consecutive days with a GOAT score ≥76. Because 6 patients were still in PTA at hospital discharge and no PTA duration could be documented for them, no analyses were performed on PTA duration. Rather, patients were divided according to the presence or resolution of PTA at discharge. The Disability Rating Scale (DRS) 36 score at discharge (last 72 hours of hospitalization) was calculated by the occupational therapist. The DRS is a 29-point scale, for which absence of disability is scored 0 and extreme vegetative state is scored 29. It is commonly used among the population with moderate-severe TBI.
Actigraphy
Patients wore a wrist actigraph (Actiwatch-2, MiniMitter Philips Healthcare, Andover, MA) on a nonparalyzed arm during hospitalization. The actigraph is a small, watch-like device that contains an accelerometer, which records physical motion in all directions with a sensitivity of 0.05 g. Motion is then converted to an electric signal, which is digitally integrated to derive an activity count per 1-minute epochs.
Data were acquired during hospitalization, beginning in ICU for most patients and continuing throughout hospitalization in regular wards. Actigraphy recording began when continuous sedation and analgesia had ceased for at least 24 hours. By that stage, patients were no longer intubated and had reached a level of medical stability defined by the absence of elevated ICP, hemodynamic instability, and fever or active infections. Moreover, the actigraph was installed only once patients reached a Rancho Los Amigos score ≥III, indicative of a more apparent physical reactivity to internal and external stimuli.
Approximately every 3 days, data were uploaded into dedicated software (Actiware 5.0). Removal of the actigraph for data uploads took on average 7.9 ± 6.2 minutes, during which time wrist activity was not recorded.
Data Analyses
Because of the absence of a consolidated 24-hour rest-activity cycle on most recording days, the cosinor analysis, which is typically used to quantify the rest-activity cycle, 37 would have led to nonvalid results. In the present study, the rest-activity cycle consolidation was thus estimated with the ratio of daytime activity to total 24-hour activity. For each 24-hour day and for each participant, the activity counts were summed separately for daytime (07:00-21:59 hours) and nighttime (22:00-6:59 hours) periods. Nighttime was defined according to the schedule of the hospital unit, and was characterized by lower levels of activity and light. Total 24-hour activity (07:00-06:59 hours) was the sum of the daytime and nighttime periods. Each 1-minute epoch during which no recording took place (because of actigraph removal for data uploads) was attributed the average activity count of the period (daytime or night-time) during which the actigraph was removed. The percentage of total 24-hour activity occurring in the daytime was calculated to obtain the daytime activity ratio: Daytime activity ratio = (Daytime activity/24-Hour activity) × 100. A ratio of 50% indicated a level of activity evenly distributed across daytime and nighttime periods, whereas a ratio of 100% indicated that all activity occurred during the daytime. According to our preliminary analyses, a daytime activity ratio of 80% was chosen to designate an adequate consolidation of the rest-activity cycle, synchronised to the day-night cycle. A total of 5 variables were used to evaluate the state and evolution of rest-activity cycle consolidation: mean daytime activity ratio for the total recording period, daytime activity ratio averaged over the first and last 48 hours of recording, percentage of improvement between the first and last 48 hours of recording, and number of days with daytime activity ratio ≥80%.
Actiware 5.0 automatically scored a minute of recording as “moving” according to a threshold value of ≥10 activity counts per epoch. The quantity of minutes moving was then calculated for each day of recording, for each patient. Minutes during which the actigraph was removed for data uploads were scored as “moving,” because the actigraph was removed only when participants were not resting.
Statistical Analyses
A within-subject ANOVA with Time as the repeated factor was carried out to compare the daytime activity ratio over the 10 days of recording. This analysis was only carried out on patients who had 10 days of recording. All other analyses included all 16 patients. Paired t tests on daytime activity ratio were also performed to compare the first and last 48 hours of actigraphy recording. To test our second hypothesis, a series of Pearson correlations were conducted between actigraphic variables and each variable of injury severity and outcome. Subgroups of patients with and without PTA at hospital discharge were compared using independent-samples t tests. Exploratory testing of each subgroup’s improvement of rest-activity cycle consolidation was performed using an ANOVA with 1 repeated measure (time). If not otherwise mentioned, the data are presented as mean ± standard deviation. Significance was set at P < .05.
Results
Figure 1 and the Supplementary Figures A1, A2, and A3 show the actigraphy recordings of all patients. Patients began wearing the actigraph 18 ± 13.3 days postinjury and a delay of 13.9 ± 13 days was observed between the last day of actigraphy recording and hospital discharge. Details on patients’ clinical characteristics and actigraphy recordings are presented in the Results section of supplementary materials.
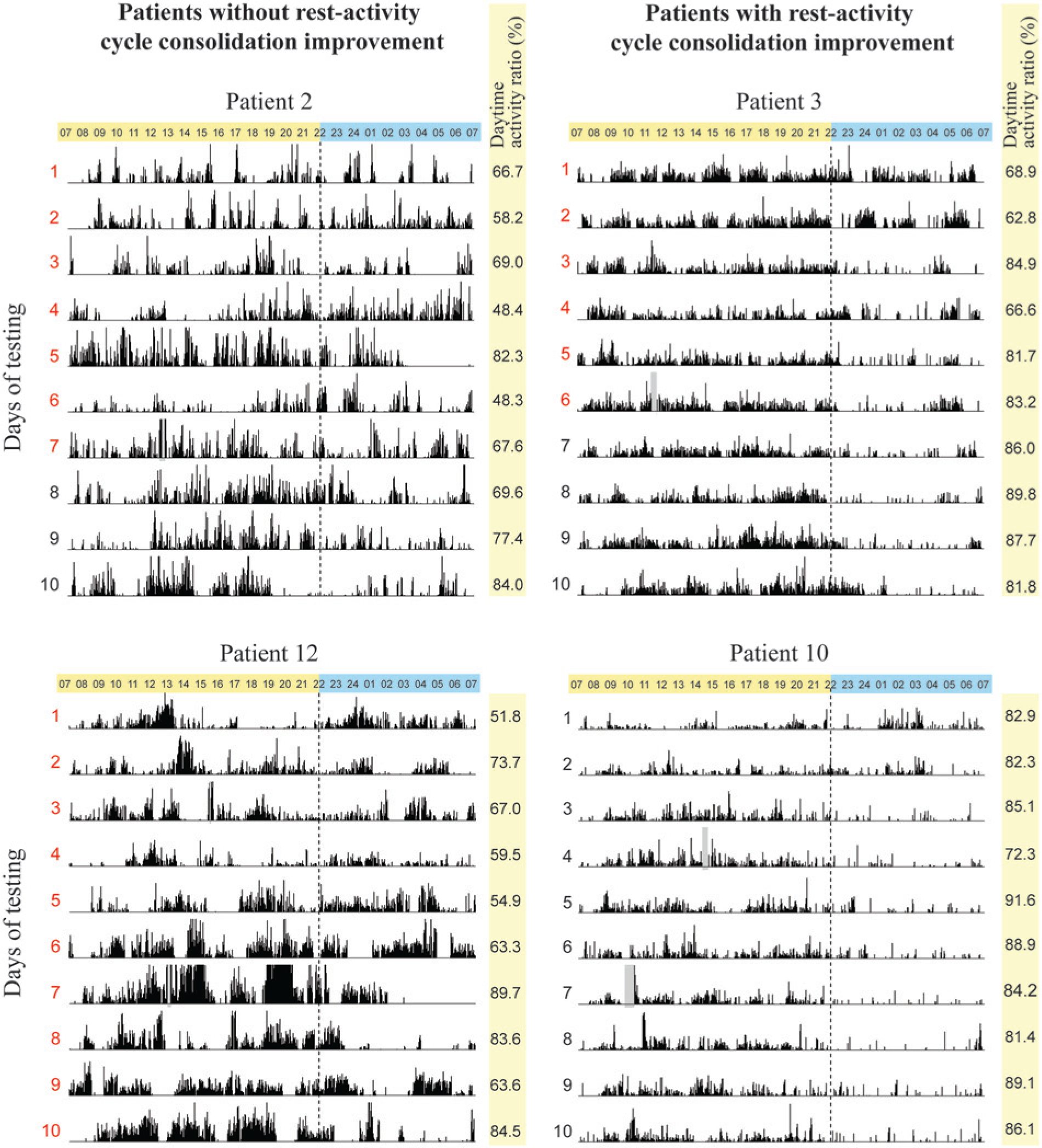
Examples of actigraphy recordings: examples of 10-day actigraphy results from 4 representative patients. Each of the 10 days is represented on a separate line, from 07:00 to 07:00 hours. Total activity counts for each minute of recording is illustrated by vertical dark lines. The same scale of 0 to 1000 activity counts was used for all participants and all days of recording. Hours included in the day period (07:00 to 22:00 hours) are shown in yellow and those included in the night period (22:00 to 07:00 hours) are in blue at the top of each graph. Daily percentages of daytime activity on total 24-hour activity are indicated on the right side of each actigram. Periods with no recording are represented by gray rectangles. Days when the recording took place in the intensive care unit are in red.
Consolidation of the Rest-Activity Cycle in Acute TBI
When all patients (n = 16) and all days of recording (n = 148) were considered, daytime activity represented 75.1% ± 13.7% of total activity. Overall, the criteria of ≥80% daytime activity, representing a consolidation of the rest-activity cycle, was met in 69 (46.6%) of all days of recording. Cycle consolidation was present during only 18 of 63 days (28.6%) in the ICU but increased to 52 of 85 days (61.1%) in regular wards. In the 3 patients who recovered rapidly and had less than 10 days of actigraphic recording, most days (93.3%) showed ≥80% daytime activity, suggesting that the ≥80% criterion was valid, although arbitrary.
Figure 1 shows the actigraphy recordings of 4 patients, which are an adequate representation of all patients tested. Two general patterns of rest-activity cycle were observed. The first pattern, represented by patients 2 and 12, was observed in 8 patients and illustrates a continuous absence of rest-activity cycle consolidation over all days of recording. Several of these patients also had only brief periods of consolidated rest over the 24 hours, as shown by the brief periods during which the actigraph was not active. The second pattern, illustrated by the actigrams of patients 3 and 10, was observed in 8 patients and depicts an improvement in the consolidation of the rest-activity cycle over the 10 days of recording.
During the first 48 hours of actigraphy, only 4 patients (25%) showed a consolidated rest-activity cycle; however, this evolved to being present in 10 patients (62.5%) during the last 48 hours of recording. Furthermore, only 3 patients (18.8%) reached a daytime activity ratio ≥80% while in the ICU. Average daytime activity ratio of the first 48 hours of recording was 70.8% ± 10.1%, increasing to 79.1% ± 13.7% for the last 48 hours, thus showing a significant improvement of 9.3% ± 11.3% between the beginning and end of recording: t(30) = −2.1; P < .05. Of the 12 patients with 10 days of actigraphy, a significant linear trend of improvement was observed from day 1 to day 10 (see Figure 2): F(1, 11) = 11.92; P < .01. When patients were considered individually, improvement from the first to last 48 hours of actigraphy was quite diverse, ranging from −22.39% to 26.97%.
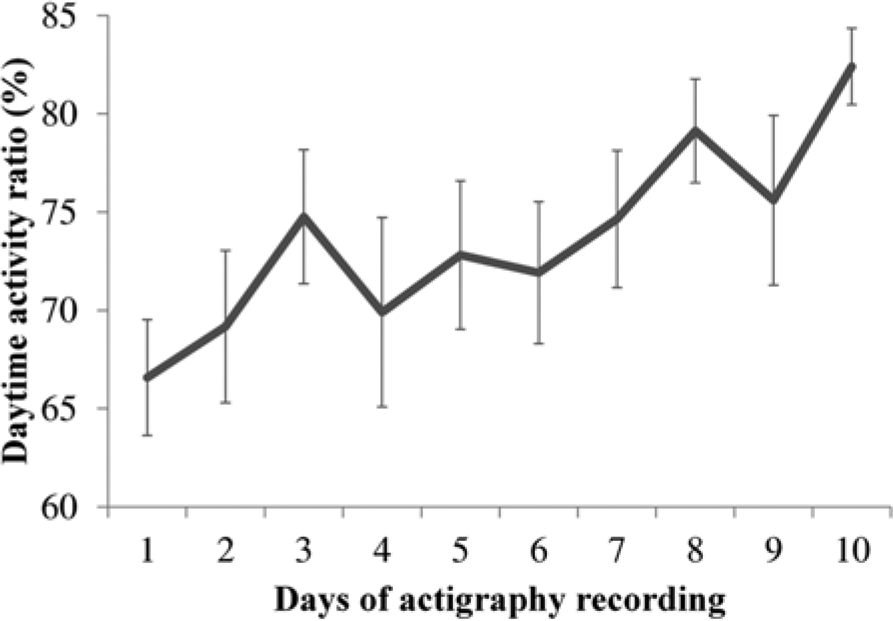
Evolution of the consolidation of rest-activity cycle over 10 days: evolution of the mean (±SEM) daytime activity ratio in the 12 patients having 10 days of recording. The linear trend of improvement was statistically significant (P < .01).
On average, patients had 827.2 ± 233.2 minutes scored “moving” per 24 hours (57.4% per day) during the first 48 hours of recording and 845.8 ± 166.0 minutes per 24 hours (58.7% per day) during the last 48 hours of recording. The percentage of time moving per 24 hours is provided for each patient and for each day of recording in Supplementary Table 1.
Association Between Rest-Activity Cycle and Clinical Variables
Table 2 presents the Pearson correlation coefficients showing the associations between rest-activity variables and clinical variables. Better or improving rest-activity cycle consolidation was associated with higher GCS at admission, a shorter ICU stay, and a shorter hospital stay. The daytime activity ratio of the first 48 hours of actigraphy was not related to any variable of injury severity or outcome. The Marshall and Rotterdam scores, peak TIL score, number of days of elevated ICP, and cumulative ICU dose of analgosedation were not significantly associated with any rest-activity variable.
Pearson Correlation Coefficients Between Rest-Activity Cycle and Clinical Variables. a
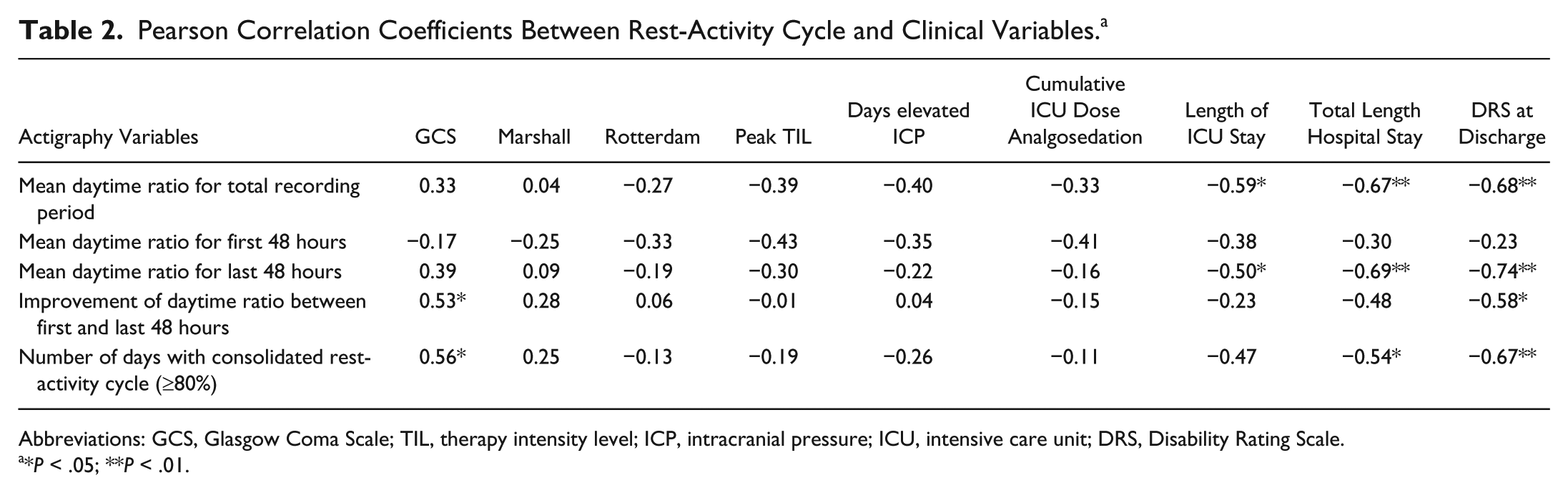
Abbreviations: GCS, Glasgow Coma Scale; TIL, therapy intensity level; ICP, intracranial pressure; ICU, intensive care unit; DRS, Disability Rating Scale.
*P < .05; **P < .01.
Better rest-activity cycle consolidation was associated with lower disability at discharge (Figure 3). More specifically, a higher daytime activity ratio for the total recording period, a higher daytime activity ratio in the last 48 hours of recording, a higher percentage of improvement from the first to last 48 hours, and a larger number of days with rest-activity cycle consolidation were associated with lower DRS scores (r values ranging from −0.53 to −0.69, P < .05).
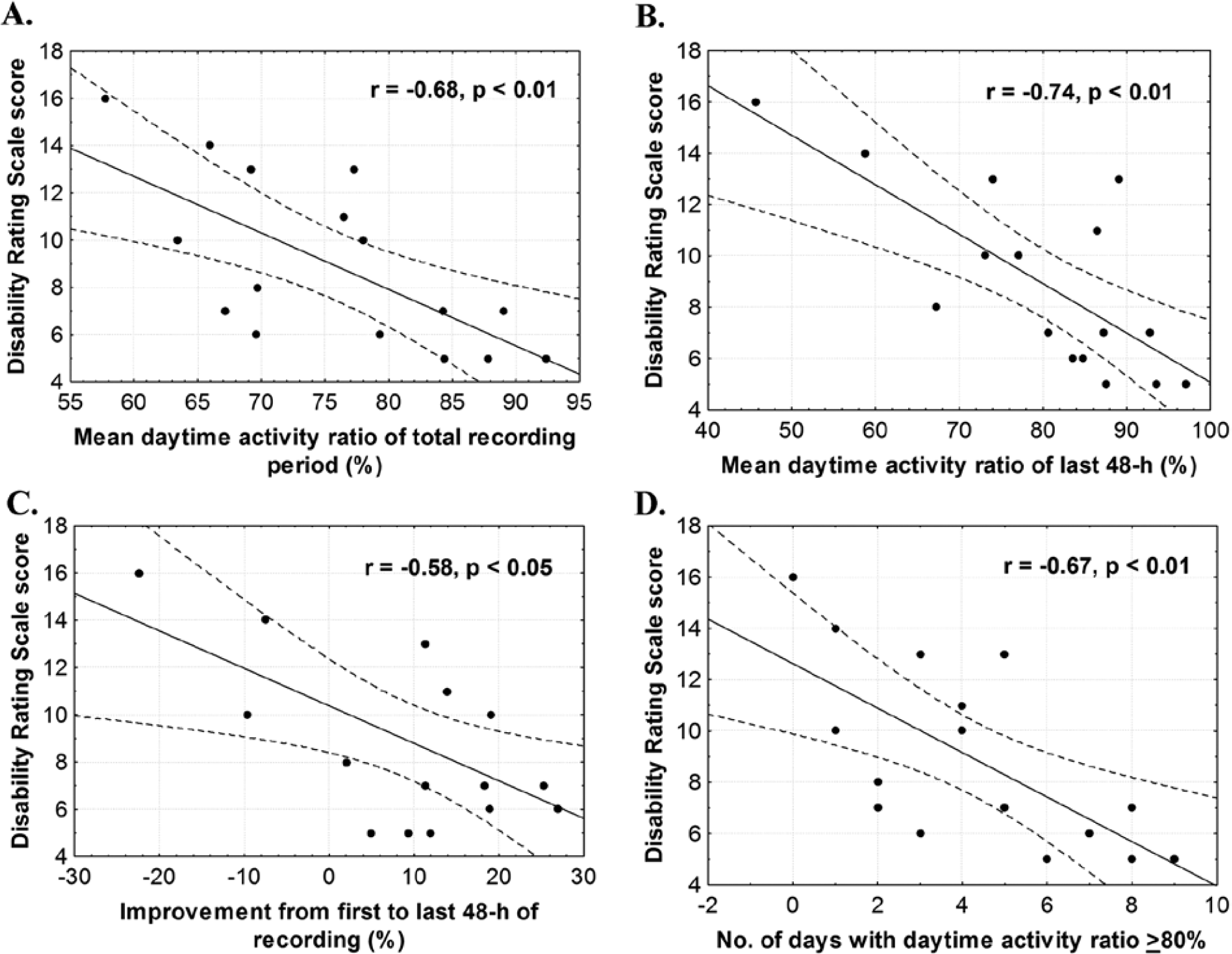
Illustration of the significant Pearson correlations between the daytime activity ratio and the score on the Disability Rating Scale (DRS) at discharge: A. Negative correlation between the daytime activity ratio averaged over the total recording period and the DRS at discharge. B. Negative correlation between the daytime activity ratio averaged over the last 48 hours of actigraphy recording and the DRS at discharge. C. Negative correlation between the percentage of improvement between the first and last 48 hours of recording and the DRS at discharge. D. Negative correlation between the number of days with a daytime activity ratio >80% and the DRS at discharge. Dashed lines represent the 95% confidence interval.
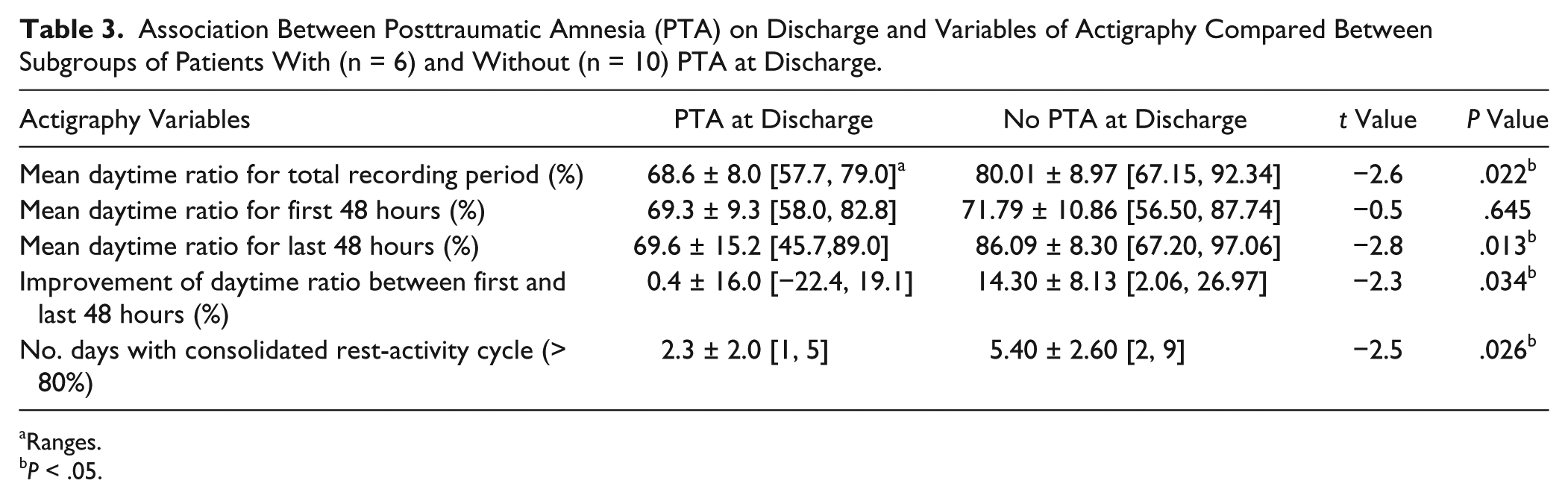
Table 3 shows the actigraphy results of patient subgroups divided according to presence or absence of PTA at hospital discharge. Patients no longer in PTA at discharge had significantly higher daytime activity ratios over the total recording period, higher daytime activity ratio in the last 48 hours of recording, greater improvement between the first and last 48 hours of recording, and more days of consolidated rest-activity cycle (t and P values detailed in Table 3). Moreover, the only actigraphy measure that did not differ between groups was the daytime activity ratio of the first 48 hours of recording. Therefore, the 2 subgroups were very similar during the first 48 hours of recording; however, the patients no longer in PTA at discharge showed significantly greater improvement over time. The ANOVA performed over the 10 days of recording confirmed a significant evolution of consolidation of the rest-activity cycle in patients no longer in PTA at discharge: F(1, 6) = 8.85; P < .05. This evolution was not significant in patients still in PTA at discharge: F(1, 4) = 2.61; P = .18. When compared on variables of injury severity and outcome, patients still in PTA at discharge had a longer ICU stay (33.5 ± 6.9 vs 17.9 ± 16.2 days, t(14) = 2.2, P < .05), a longer hospital stay (61.7 ± 13.8 vs 34.1 ± 17.7 days, t(14) = 3.7, P < .01), and a higher DRS score at discharge (12.4 ± 2.5 vs 6.7 ± 1.8, t(13) = 5, P < .001).
Association Between Posttraumatic Amnesia (PTA) on Discharge and Variables of Actigraphy Compared Between Subgroups of Patients With (n = 6) and Without (n = 10) PTA at Discharge.
Ranges.
P < .05.
Discussion
Poor Circadian Rest-Activity Cycle Consolidation in Acute TBI
Our aim was to characterize the quality and evolution of the rest-activity cycle in acute moderate to severe TBI. We found that rest-activity cycle consolidation was predominantly absent during the first days of actigraphy recording but globally improved over time during their hospital stay. In fact, when a daytime activity ratio ≥80% was used as a threshold for adequate rest-activity cycle consolidation, consolidation was observed for 28.1% of days in the first 48 hours, whereas this percentage increased to 68.8% in the last 48 hours of recording. Two distinct patterns of evolution of the rest-activity cycle were observed: 50% of patients showed an absence of rest-activity cycle consolidation throughout the recording period, whereas the other 50% had reached a daytime activity ratio ≥80% by the last 48 hours of recording.
This absence of rest-activity cycle consolidation is likely associated with fragmentation of both sleep and wake episodes. The number of minutes moving was similar during the first and last 48 hours of recording and covered nearly 60% of the day. This observation suggests that the absence of consolidation of the rest-activity cycle was not caused by constant rest but rather reflects the dispersion of activity bouts all over the 24 hours. Because actigraphy has not been validated with polysomnography in this bed-ridden population, it cannot be concluded that the absence of activity represents sleep. However, the presence of activity over the 24 hours shows that the rest episodes were highly fragmented by intervening activity bouts, thereby possibly preventing the occurrence of any deep sleep episodes of significant duration.
Association Between Consolidation of the Rest-Activity Cycle, Injury Severity, and Recovery
This study revealed significant associations between TBI severity and the rest-activity cycle. More precisely, less-severe TBI was associated with greater improvement between the first and last 48 hours of activity recording and with more days of consolidated rest-activity cycle. Shorter lengths of ICU and hospital stay were associated with a higher average daytime activity ratio over all days of recording and with a higher average daytime activity ratio in the last 48 hours of recording. A shorter duration of total hospital stay was also associated with more days of consolidation of the rest-activity cycle. Although these results will need to be confirmed in a larger sample, they do point to the role of TBI in the persistence of rest-activity cycle disturbances in acute care.
Our study also revealed significant associations between the rest-activity cycle and outcome at hospital discharge. First, we found that patients no longer in PTA at discharge had better rest-activity consolidation over the total recording period and during the last 48 hours of recording, had greater improvement of rest-activity consolidation between the first and last 48 hours of recording, and had more days of consolidated rest-activity cycle. Moreover, patients with a more rapid return to a consolidated rest-activity cycle over the 10 days of actigraphy were more likely to clear PTA before hospital discharge. Similar results were also found for disability severity, where patients with better rest-activity cycle consolidation and with greater improvement of cycle consolidation showed lower DRS scores at discharge. These associations between rest-activity cycle consolidation and outcome at hospital discharge were observed even if there was an average delay of 13.9 ± 13 days between the last day of recording and hospital discharge, suggesting that the degree of rest-activity cycle consolidation is associated with cerebral recovery and may predict short-term outcome in acute TBI.
An association between sleep and PTA has already been reported among 14 TBI patients in a rehabilitation setting, where patients who had cleared PTA before admission to the rehabilitation center showed better sleep efficiency, as measured with wrist actigraphy, compared with patients with ongoing PTA. 11 These results point to a close link between sleep consolidation and cognitive functioning, but it is currently difficult to establish a causal relationship in the context of TBI. In fact, considering that sleep has a crucial role in memory consolidation, learning, cerebral plasticity, and neurogenesis,38,39 sleep fragmentation may, probably by preventing the apparition of deep sleep, 22 impede PTA resolution in patients with TBI. However, it is also possible that structural brain damage simultaneously affects memory and sleep, either through dissociated or common pathways. In the latter case, PTA and rest-activity cycle consolidation or increased sleep efficiency may occur simultaneously when the brain recovers sufficiently to allow both sleep consolidation and PTA resolution.
Possible Causes of the Lack of Consolidation of the Rest-Activity Cycle
The rest-activity cycle is the most easily observed manifestation of endogenous circadian rhythms. 40 The main circadian biological clock, located in the hypothalamus, generates an oscillation of about 24 hours that is normally synchronized to the environmental 24-hour day, mainly by exposure to the light-dark cycle but also by regular timing of social contacts, food intake, and so on. In humans, the endogenous circadian clock synchronizes physiological, cognitive, and behavioral functions, such that those associated with activity happen in the daytime, whereas those associated with rest and sleep occur during the night. Circadian disruption occurs when the main biological clock is not synchronized to the 24-hour day and/or when it produces a circadian signal too weak to entrain properly the peripheral clocks located in other regions of the brain and body. The first manifestation of circadian disruption is a decreased consolidation and abnormal timing of the rest-activity cycle and sleep-wake disorders. 41 The ICU is devoid of regular day-night environmental cues, and the ICU stay is associated with severe medical conditions, analgesia, and sedation, all known to affect the temporal structure of circadian rhythms and induce circadian desynchrony.42,43 The fact that there was no correlation between the number of minutes moving per 24 hours and the daytime activity ratio in our patients suggests that the daytime activity ratio was not affected by the quantity of activity but rather measures the circadian organization of activity and rest periods. Although a low daytime activity ratio could also signal a poor timing of consolidated sleep, such as sleep that occurs with a phase advance (eg, 19:00 to 3:00 hours) or phase delay (eg, 3:00 to 11:00 hours), visual inspection of actigraphy data suggests that it was not the case in our patients. When patients had a low daytime activity ratio, they rather had fragmented rest and activity dispersed over 24 hours. The concurrent measure of robust circadian markers would be necessary to assess the contribution of circadian disruption to the decreased consolidation of the rest-activity cycle in TBI patients.
TBI itself could be responsible, at least in part, for altered circadian rest-activity rhythm. In a previous study, ICU patients with neurological injury showed more severe circadian rhythm disturbances than patients without neurological injury. 44 Because of our small sample size, it is currently not possible to understand the role of specific TBI characteristics, such as location of brain lesions; however, we can hypothesize that patients with brain injury in hypothalamic regions, for example, will be at greater risk of circadian rhythm disturbances.
Actigraphy in a Hospital Setting
It has been shown that actigraphy underestimates wakefulness,45-47 particularly when individuals lie immobile in bed in a nonsleeping state, 48 and especially among a critically ill population. 49 Therefore, the already short rest episodes observed on actigraphy recordings probably overestimate the quantity of sleep our patients actually experienced.
A daytime activity ratio ≥80% was used to designate the presence of rest-activity cycle consolidation. Because participants frequently reached a ratio above 85% to 90% in the last days of recording, 80% was a conservative threshold to denote an acceptable rest-activity rhythm. The significant association of this threshold of consolidation with GCS and outcome supports its usefulness as a measure of rhythmicity within this patient population and setting.
Study Limitations
Although the rest-activity cycle has been strongly correlated with the sleep-wake cycle among healthy individuals, 23 such an association has not been formally validated among those hospitalized in critical care. Because of the small number of patients, our study serves as an exploratory initiative aiming to understand the nature of sleep and rest-activity cycle disturbances in the acute phase of moderate-severe TBI. Therefore, the use of actigraphy and the parameters chosen to analyze the data collected during the acute phase of the moderate-severe TBI population will need to be formally validated in a larger cohort and compared with concurrent polysomnography recordings.
Conclusions
This study is the first initiative aiming to understand the nature and evolution of sleep and circadian rhythm disturbances in acute moderate to severe TBI. Despite the difficulties encountered in a hospital setting, our study demonstrates that it is possible to effectively use actigraphy to assess the rest-activity cycle, even in acute care. Although a larger sample will be needed to perform a formal validation, the associations found between the rest-activity cycle and variables of outcome at hospital discharge suggest that actigraphy could become an important clinical tool for the monitoring and prognosis of TBI patients.
Our data revealed severe disturbances of the rest-activity cycle in the acute phase of moderate-severe TBI. Such disturbances had not been systematically documented and quantified previously for an extended period of time among this population. The lack of consolidation of the rest-activity cycle was associated with rest episodes of very short duration not compatible with the occurrence of the deepest stages of sleep. Considering the role of sleep in cerebral plasticity and neurogenesis, sleep disturbances in acute TBI may impede short- and long-term cognitive and neuronal recovery in this population. Efforts to restore circadian synchrony while patients are hospitalized may help prevent the development of chronic sleep and wake disturbances and optimize recovery.
Footnotes
Acknowledgements
The authors would like to acknowledge Pierre-Olivier Gaudreault, Katia Gagnon, and Andrée-Ann Baril for their contribution to the monitoring of patients during actigraphy recordings.
Authors’ Note
Ethics approval was provided by the Ethics Committee of Hôpital du Sacré-Coeur de Montréal. This article was not commissioned and was externally peer reviewed.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by the Canadian Institutes of Health Research (CIHR), by the Fonds pour la recherche du Québec, Santé (FRQS), by the University of Montréal (studentship to CD), by the Fondation Neurotrauma Marie-Robert (studentship to CD), by the Réseau provincial de recherche en adaptation-réadaptation (REPAR) (studentship to CWH), and by the J. A. De Sève foundation (studentship to CD).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
