Abstract
Background. Disrupted sleep is common after traumatic brain injury (TBI) particularly in the inpatient rehabilitation setting where it may affect participation in therapy and outcomes. Treatment of sleep disruption in this setting is varied and largely unexamined. Objective. To study the feasibility of instituting a sleep hygiene intervention on a rehabilitation unit. Methods. Twenty-two individuals admitted to a brain injury unit were enrolled and allocated, using minimization, to either a sleep hygiene protocol (SHP) or standard of care (SOC). All participants wore actigraphs, underwent serial cognitive testing, and had light monitors placed in their hospital rooms for 4 weeks. Additionally, participants in the SHP received 30 minutes of blue-light therapy each morning, had restricted caffeine intake after noon, and were limited to 30-minute naps during the day. SHP participants had their lights out time set according to preinjury sleep time preference. Both groups were treated with the same restricted formulary of centrally acting medications. Results. Of 258 patients screened, 27 met all study inclusion criteria of whom 22 were enrolled. Nine participants in each group who had at least 21 days of treatment were retained for analysis. The protocol was rated favorably by participants, families, and staff. Actigraph sleep metrics improved in both groups during the 4-week intervention; however, only in the SHP was the change significant. Conclusions. Sleep hygiene is a feasible, nonpharmacologic intervention to treat disrupted sleep in a TBI inpatient rehabilitation setting. A larger study is warranted to examine treatment efficacy. ClinicalTrials.gov Identifier: NCT02838082.
Keywords
Introduction
There is a growing awareness in society of the importance of sleep in health and well-being. Popular media outlets often highlight sleep health–related stories and, in the United States, interventions to improve sleep are a multibillion-dollar industry. 1 Research has shown that sleep is vital for memory, cognition, creativity, mood, immune system integrity, cardiovascular, and cerebrovascular health, and even life expectancy.2-6 Good sleep has been associated with positive health outcomes in the general population and within groups of individuals with various disabilities including traumatic brain injury (TBI).7-9
Moderate to severe TBI is extremely costly to society and often holds lifelong impairment for the individuals who sustain such an injury.10,11 Moreover, sleep disorders in individuals with moderate to severe TBI are associated with more severe injury and higher costs in both acute and rehabilitation centers. 12 In the rehabilitation hospital setting, the prevalence of disrupted sleep in individuals with TBI has been estimated to be between 67% and 86%.12-15 Disrupted sleep has long been known to the clinician taking care of patients with moderate to severe TBI; however, investigation into the mechanisms and treatment of sleep disruption in individuals with TBI is relatively new.13,15-25
A small clinical trial among individuals with moderate to severe TBI showed a relationship between improved sleep efficiency and the resolution of confusion. 7 A safe discharge back into the community for an individual with TBI often cannot occur until this confusion clears. It has also been shown that disrupted sleep continuity in normal individuals can lead to impaired memory encoding, reaction time, and executive function. 26 These same deficits are hallmarks of an individual in a rehabilitation center emerging from a moderate to severe brain injury. As such, disrupted sleep in this setting is an obvious target for treatment.
To adequately address sleep disturbance in patients with TBI during inpatient rehabilitation, additional research is needed to investigate possible factors, aside from injury, that might cause sleep disruption. Specifically, it is necessary to consider the rehabilitation hospital setting itself and investigate whether aspects of an institutional environment such as nighttime vitals, incontinence care, scheduled turning of immobile patients, and hallway light and noise may have a negative impact on patients’ sleep. To date, the impact of the hospital environment on sleep in individuals with TBI has been largely unstudied.
Sleep hygiene, a nonpharmacologic intervention, is an effective and recommended first line treatment for disrupted sleep in the general population 27 and is likely to be similarly effective for treating sleep disturbances after TBI. 28 Furthermore, before trying other interventions to address the problem of disrupted sleep in TBI, such as pharmacology, the sleep environment must first be fully optimized to remove the potential confound of a poor sleep context contributing to poor sleep in patients.
The purpose of the present study was to assess the feasibility of implementing a sleep hygiene intervention on an acute, inpatient, brain injury rehabilitation unit. A thorough review of the literature shows that this is the first study to prospectively compare a sleep hygiene intervention to the standard of care among individuals undergoing inpatient rehabilitation for TBI using actigraphy and a matched control group design. 29 The 2 primary aims of the study were to (1) characterize and estimate the incidence of disrupted sleep in the early rehabilitation phase of recovery after TBI and (2) determine the feasibility of implementing a sleep hygiene intervention for patients within an inpatient TBI rehabilitation setting. Additionally, we sought to explore the relationship between sleep quantity and quality and recovery of cognitive function and rehabilitation outcomes among individuals with TBI.
Methods
This was a prospective, unblinded, 2 arm, pilot study designed to assess the feasibility of implementing a 4-week sleep hygiene protocol (SHP) for improving the sleep of individuals with TBI who were undergoing inpatient rehabilitation. The study was approved by the hospital’s institutional review board and was conducted in a 26-bed specialized TBI unit in a rehabilitation hospital in the western United States. All rooms were equipped with light blocking shades and ambient light sensors. Rooms of participants in the SHP were equipped with blue-light therapy boxes (Philips HF3332 goLite). Consecutive admissions of individuals with TBI were screened for sleep disturbance with actigraphy during the first 72 hours of admission; the minimal length of time recommended by the manufacturer for obtaining sleep metrics. A set rest period from 21:00 to 07:00 hours was used for this study because it is the facility’s standard of practice. Sleep efficiency (SE), a core actigraphy metric defined by the duration of sleep divided by the amount of time in bed, was used to identify individuals with disrupted sleep. Individuals with a SE of less than 80% during the 10-hour set rest period underwent secondary screening. All eligible individuals or their designated decision maker were approached for consent. Consenting participants were subsequently allocated into either the SHP or standard of care (SOC) using computer-generated minimization 30 —a method of randomization that balanced the groups on age, time postinjury, and the presence/absence of posttraumatic amnesia (PTA). All participants wore actigraphs while undergoing their respective protocols for 4 weeks.
Participants
To provide a homogeneous group of subjects in terms of mechanism of injury, only individuals with closed head injuries were included in the study. Causes of injury in this cohort included motor vehicle accident, fall, blunt assault, and sports-related accidents. Patients were excluded if they had a previous moderate to severe brain injury, a history of, or a high clinical likelihood of having a sleep related breathing disorder, 31 or a history of any other sleep disorder or neurodegenerative condition. (Please see the online supplementary material for full inclusion and exclusion criteria.)
Sleep Hygiene Protocol
The sleep hygiene intervention was developed and based on the work of Morin and colleagues32,33 employing concepts of both sleep restriction and stimulus control. The SHP consisted of 6 principal components: (1) improved nighttime sleep environment, (2) increased daytime activation, (3) enhanced circadian stimuli, (4) consistent morning wake time and activity of daily living (ADL) routines each day, (5) 30 minutes of blue-light therapy in the morning, and (6) no caffeine intake after 12:00 pm. Participants in the SHP had sleep periods set according to preinjury chronotype as reported by the family. The full SHP employed in this study has been described in a previous publication by our group and is available in the online content. 34
Standard of Care Protocol
Participants allocated to the SOC group underwent standard TBI rehabilitation care and wore an actigraph for the duration of their participation in the study. No systematic attempt was made to modify or influence sleep or wake times or to reduce or enhance light exposure.
Participants in both groups participated in physical therapy, occupational therapy, and speech-language therapy for a minimum of 3 hours per day Monday through Friday. Attending physicians of patients in SOC and the SHP adhered to a single restricted pharmacy list per protocol.
Measures
Demographics
Demographic data collected included ethnicity, gender, injury severity, time since injury, duration of PTA, and presence or absence of PTA on admission to rehabilitation.
Actigraphy
An actigraph is a watch-sized accelerometer typically worn on the wrist that is used to monitor rest and activity cycles in patients with a variety of sleep disorders. Actigraphs have been used in sleep research for over 20 years. Sleep scoring algorithms determine sleep parameters for the designated rest interval according to threshold levels of movement, which signify wakefulness. Below threshold values or the absence of movement has been correlated with sleep. Actigraphy was obtained using the Actiwatch Spectrum (Philips Respironic, Inc), a device with an integrated light monitor. The following default settings were used for data analysis: sleep was scored as immobile time, sleep initiation and termination required 10 minutes of either immobile or active states, wake threshold was set at medium sensitivity (40 activity counts per each 30-second epoch). The white-light threshold was set to 100 lux. Primary outcome measures were defined by metrics obtained from continuous actigraphy monitoring, including total sleep time (TST), sleep efficiency (SE), and wakefulness after sleep onset (WASO) over the duration of the patients’ participation in the study.
The nightly sleep rest interval was set from either 21:00 to 07:00 or from 22:00 to 08:00 hours depending on premorbid sleep history obtained from family as to whether, prior to injury, the participant tended to be phase-advanced or -delayed in their biologic chronotype. A report was then generated to provide summary sleep statistics for bedtime, get up time, time in bed, TST, SE, WASO, and number of awakenings.
Light Monitoring
Light was continuously monitored in participants’ rooms with a centrally placed light meter (Extech SDL400). In addition, participants in both groups wore an actigraph for the duration of the study with a built-in light meter. For participants in the SHP, the continuous readout on the room light meter cued nursing staff to keep ambient room light above 100 lux during the active wake period and below 100 lux during the nighttime rest/sleep interval. To quantify participants’ light exposure, daytime light dose was taken from the wrist actigraph, under the assumption that it would provide light exposure during the active period when the participant was in or out of their room for therapies or other activities. The wrist actigraph total time above illuminance threshold (TALT) was set to 100 lux. The daytime TALT minutes were calculated by dividing the total number of minutes of the active interval (840 minutes) to obtain a percentage above 100 lux. The room light monitor was used to measure nighttime light exposure because wrist actigraphs are typically covered by bed sheets or blankets while participants are in bed.
Cognitive Assessments
Cognitive and behavioral testing was obtained prospectively throughout the study and included the Orientation Log (O-Log),35,36 the Confusion Assessment Protocol (CAP), 37 and the Agitated Behavior Score.38,39 Participants were enrolled into the study both in and out of PTA. For participants with ongoing PTA, multiple weekly assessments of PTA were measured using the O-Log, which was administered by unit-based speech-language therapists. Participants were considered out of PTA after their O-Log scores were ≥25 on 2 consecutive days. For participants who had emerged from PTA, measures of reaction time, working memory and recognition memory were tested weekly using the CNS Vital Signs (https://www.cnsvs.com/) computerized assessment platform. These cognitive tests have been used in previous studies of both TBI and sleep. 40
Disability Rating Scale
The Disability Rating Scale (DRS)41,42 was originally developed in an inpatient rehabilitation setting to track individuals with moderate to severe brain injury from “coma to community.” A person without disability would score zero. The maximum score of 29 is associated with severe disability or extreme vegetative state. In this study, DRS scores were calculated on admission and discharge. Scores were used to compare treatment and control groups and to look at change in DRS scores over time.
Fidelity
Adherence to the SHP was assessed with a daily checklist completed by nursing staff with a goal of an 80% adherence rate. Both groups’ compliance with the restricted formulary was monitored by the hospital pharmacy. Both groups’ light exposure was monitored via in room light monitors and wrist actigraphs. In order to reduce potential crossover of the SHP elements to the SOC group, no nursing fidelity checklists were used with the SOC group. The SOC group did not have access to blue-light therapy.
Satisfaction Survey
Participants, their significant others, and nursing staff were given surveys to rate how satisfied they were with the study. From the nursing perspective, we sought to determine how arduous it was to implement the SHP. From the participants and family members, we sought to determine whether they thought the intervention was helpful or made an impact on recovery. Each survey consisted of 10 questions using a 1-to-5 Likert-type scale to determine a response. (Full surveys are attached in the online supplementary section of this article.)
Statistical Analysis
Demographic data were aggregated into means with standard deviations or percentages. Fidelity was aggregated into percentages meeting criteria per 24-hour periods and satisfaction data were aggregated into percentages per response category. Actigraphy and outcome data were aggregated into weekly medians and ranges and median change (delta scores).
Given the small sample size, data were conservatively analyzed nonparametrically. Between-group (SOC and SHP) analyses were conducted for week 1, week 3, and change scores with the Mann-Whittney U test. Within-group change from week 1 to week 3 was analyzed with the Wilcoxon sign rank test. In order to examine relationships between sleep, cognitive functioning, and rehabilitation outcomes, a combined cohort analysis was performed using Spearman correlations for actigraphy measures (TST, SE, and WASO) and their relation to cognitive test score (O-Log total and CNS Vital Signs age-corrected standard scores for Verbal Memory, Visual Memory and 4-Part CPT), and the DRS (DRS total).
Results
The CONSORT (Consolidated Standards of Reporting Trials) diagram (Figure 1) shows the flow of participants through screening, enrollment, intervention, and follow-up. A total of 258 individuals who were admitted consecutively to an inpatient rehabilitation unit with a diagnosis of TBI and who had actigraphy data in the first 72 hours were screened for the study between April 2016 and September 2017. The screened sample (n = 258) was composed primarily of male (79%), young adults (average age = 35.7 ± 15.6 years), who were on average 35.2 (SD = 36.9) days postinjury (Table 1). The mean actigraphy initial 3-day SE was impaired at 74.6% ± 14.5% for the 10-hour sleep period (Table 1). Twenty-seven (7.5%) met all inclusion/exclusion criteria. Five patients or their medical proxies declined to participate and 22 patients or their medical proxy were consented and enrolled in the study. One participant was administratively withdrawn after consenting, but before assignment to a study arm, because of medical issues.
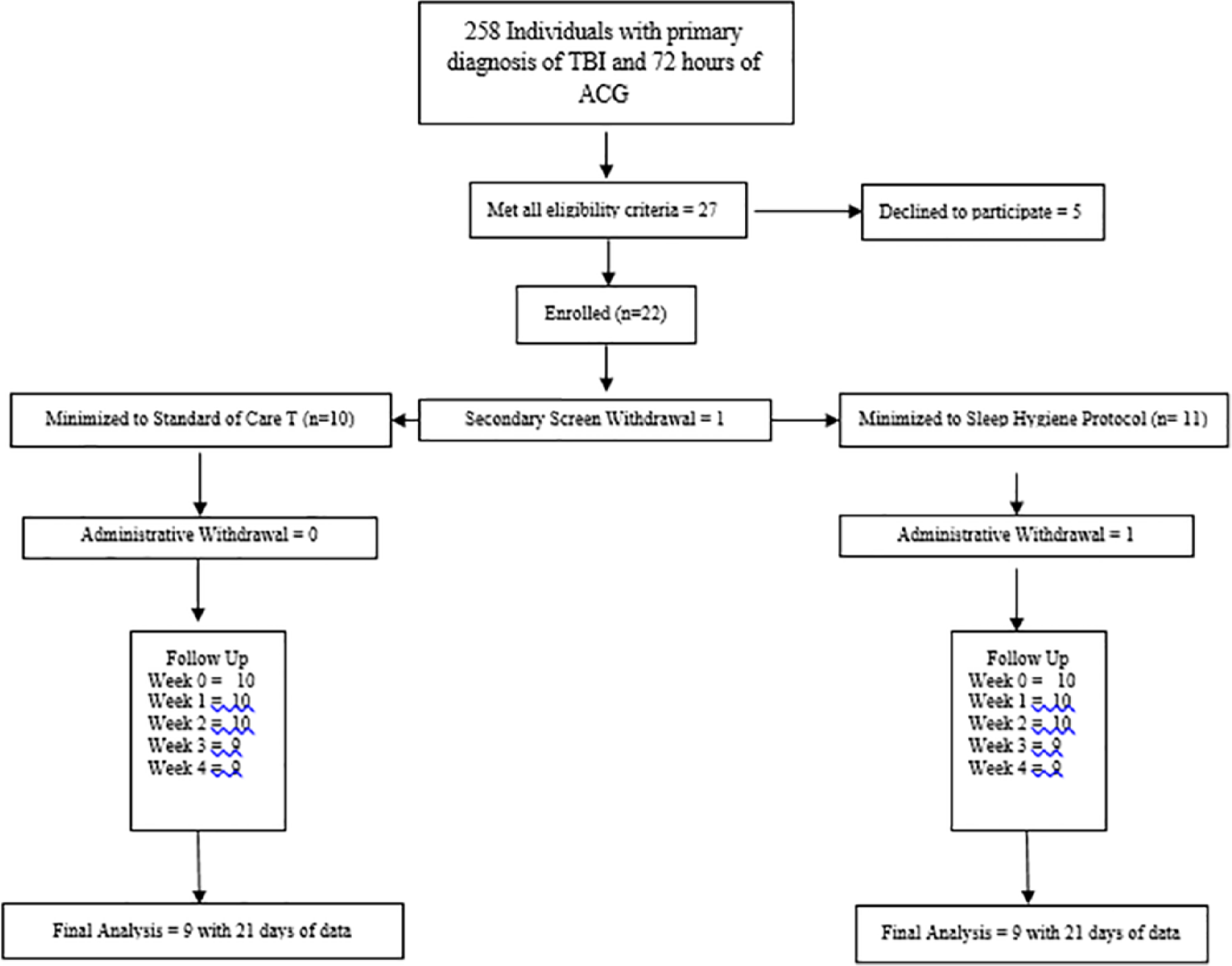
CONSORT (Consolidated Standards of Reporting Trials) diagram.
Demographics for All Patients Screened (Screening) and for OSABI Participants.
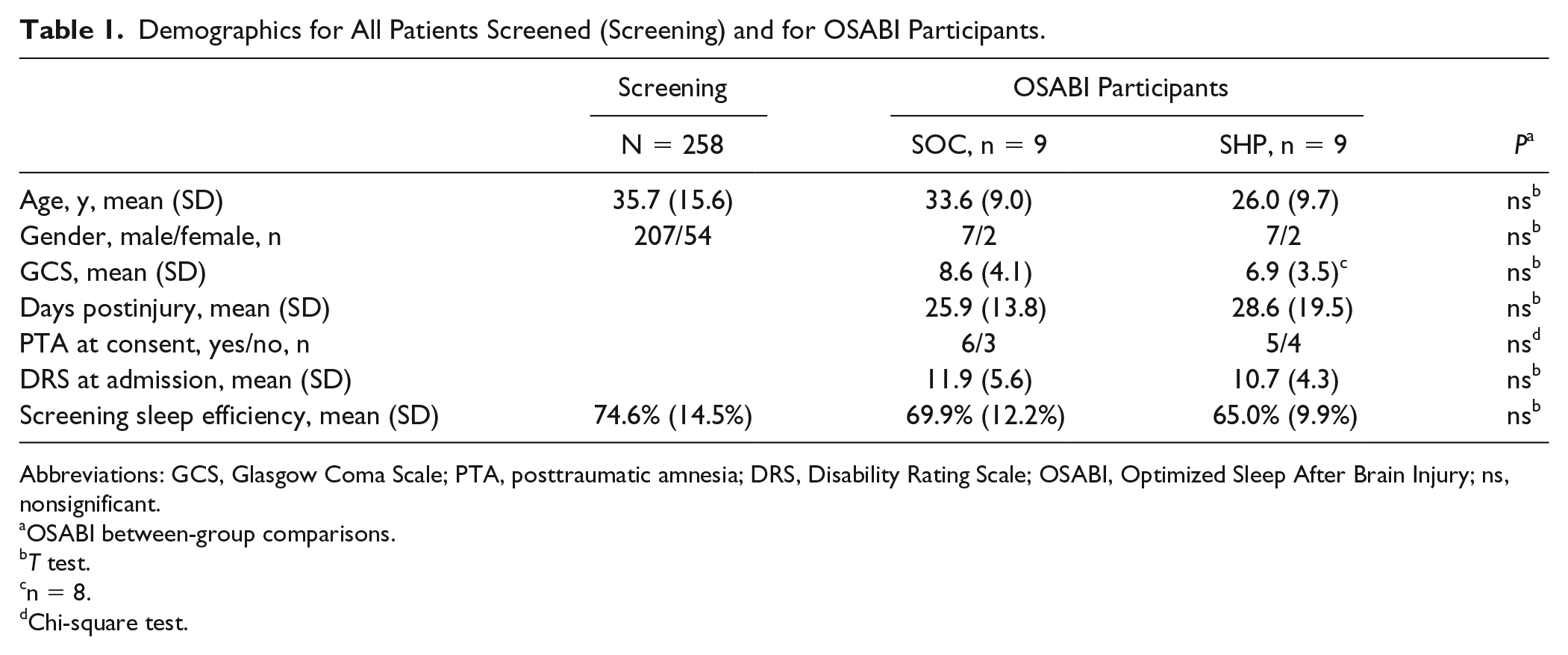
Abbreviations: GCS, Glasgow Coma Scale; PTA, posttraumatic amnesia; DRS, Disability Rating Scale; OSABI, Optimized Sleep After Brain Injury; ns, nonsignificant.
OSABI between-group comparisons.
T test.
n = 8.
Chi-square test.
A total of 11 subjects participated in the SHP and 10 in the SOC. One participant was withdrawn from the SHP due to emerging agitation, which the attending physician felt would interfere with the participant’s ability to comply with the study protocol. One additional participant from each group was dropped from analysis because they had less than 21 days of data collection secondary to early discharge. For analysis, nine participants in each group completed a minimum of 21 days of data collection. Table 1 shows the baseline characteristics of the 9 participants analyzed in each group. The groups were similar in terms of age, gender, initial injury severity as measured by Glasgow Coma Scale (GCS), admission DRS score, time postinjury, and presence of PTA. In terms of rehabilitation outcomes, there were no significant differences between the groups in their DRS scores at discharge, DRS change over the course of the study, or in their length of stay in rehabilitation (Table 2).
TST, SE, WASO for Standard of Care (SOC) and Sleep Hygiene Protocol (SHP) Measured Across the First Week of Treatment (Week 1), the Third Week of Treatment (Week 3), and the Change Over Time (Week 3 − Week 1).
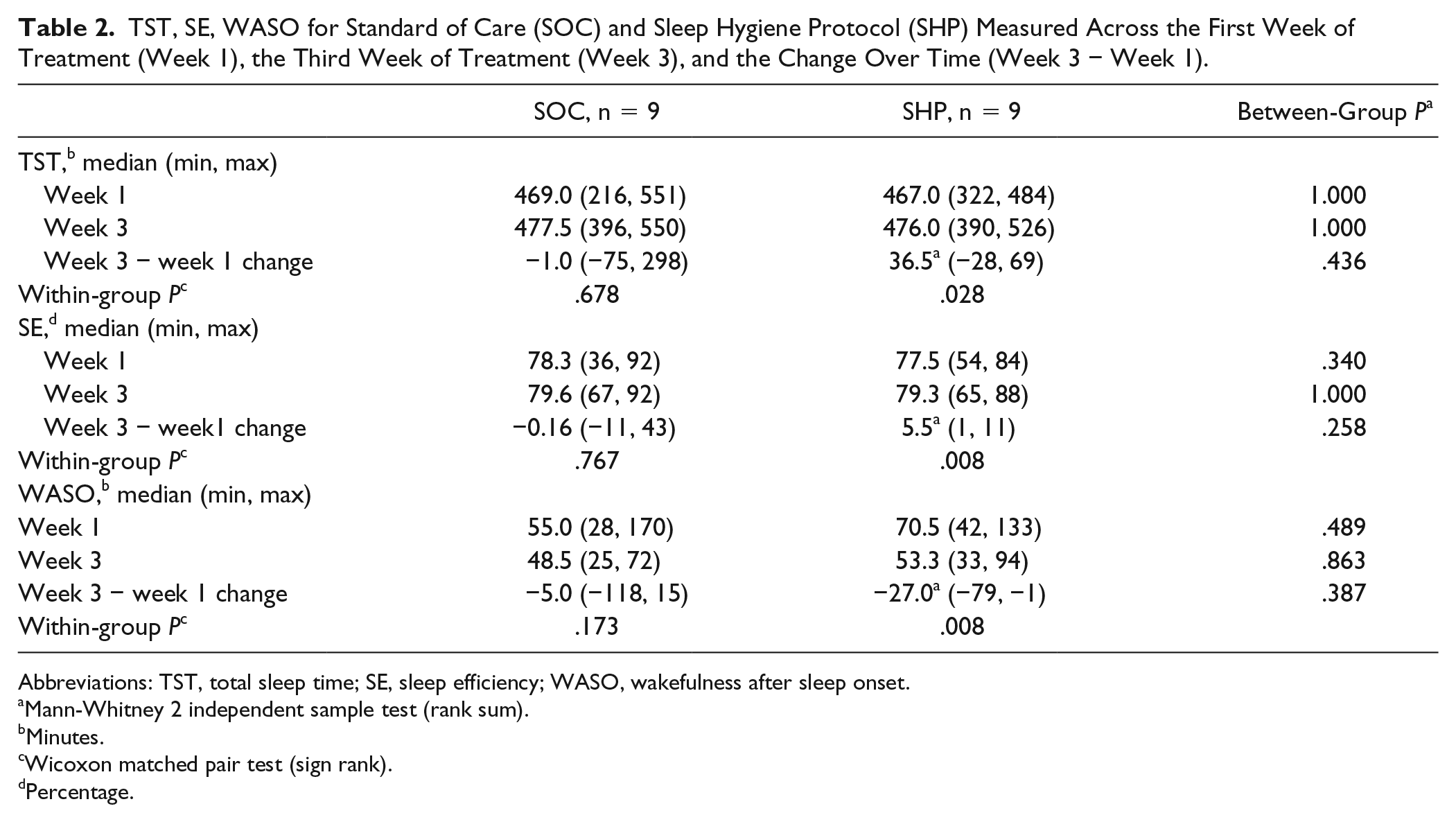
Abbreviations: TST, total sleep time; SE, sleep efficiency; WASO, wakefulness after sleep onset.
Mann-Whitney 2 independent sample test (rank sum).
Minutes.
Wicoxon matched pair test (sign rank).
Percentage.
Fidelity
An overall fidelity rate of 77% was achieved. (See Supplemental Material for adherence rates of individual SHP components.) There was only 1 incident of a medication administration outside of the restricted formulary when one participant received a single 3-mg dose of melatonin on the first night of their participation.
Light Exposure
The wrist actigraph total TALT of 100 lux during the active daytime period was 35% for the SHP group and 25% for the SOC group. Both groups met the fidelity criteria for nighttime ambient lighting being less than 100 lux measured by the room light meters at least 80% of the time: SOC 96% and SHP 89%. The average nighttime lighting lux was slightly lower for the SHP group 88.7 (±21.3), than for the SOC group 96.1 (±12.1).
Satisfaction With Protocol and Treatment of Sleep
Fourteen participants (70%), 15 family members (75%), and 36 nursing staff completed and returned the satisfaction questionnaires. Satisfaction was rated as positive with a score of 3, 4, or, 5 on the Likert-type scale. Eighty-eight percent of SHP participants reported satisfaction with sleep treatment while 100% of SOC participants reported satisfaction with sleep treatment. One hundred percent of family members of participants in the SHP reported satisfaction with their family members sleep treatment and 86% of family members of participants in the SOC group reported being satisfied.
Specifically, in response to the SHP, 88% of participants indicated that the intervention helped their sleep. Of those 88%, 50% indicated the blue-light therapy was helpful. Thirty-eight percent of the SHP participants indicated they planned to continue specific sleep hygiene components, such as regular sleep/wake times and limiting daytime naps and caffeine, after discharge. All family members surveyed indicated they felt the SHP helped their family members’ sleep. Family members rated limiting daytime naps to 30 minutes at a time as the most difficult part of the protocol. Sixty-three percent of family members planned to continue specific sleep hygiene components, such as regular sleep/wake times and limiting daytime napping and caffeine, after discharge.
The majority of nursing staff (60%) rated the SHP as helping their patients’ sleep and 80% reported being likely to integrate aspects of the SHP into their clinical practice. Twenty percent of the nursing staff reported that the SHP was hard to follow and that it had a negative impact on workflow. It should be noted that 10% of nursing staff also found the SOC protocol hard to follow and that it had a negative impact on workflow.
Actigraph Data
Between- and within-group actigraphy data are presented in Table 2. The median actigraphy TST, SE, and WASO scores were not significantly different between groups during week 1 or week 3 (presented in rows). Both groups’ overall sleep quantity and quality improved over the course of the study; however, the SOC group showed more extreme variability and did not significantly change, while the SHP improved in a more consistent and statistically significant manner (presented in columns). The SHP group exhibited significant improvements for TST, SE, and WASO, while the SOC group exhibited minimal change with considerably greater variability on the same sleep metrics (Figure 2).
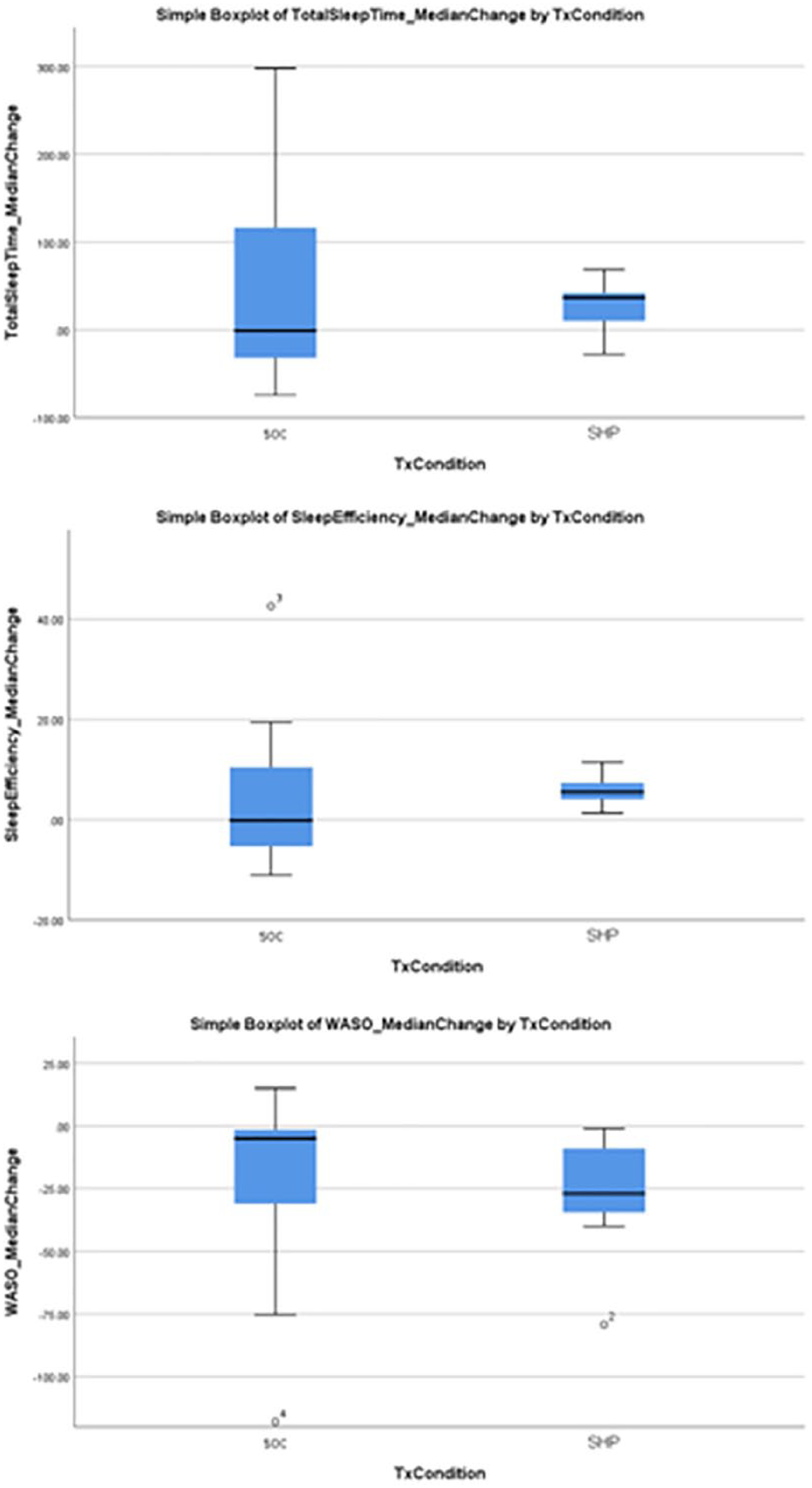
Box Plots of Actigraphy Measures per Group.
Outcome Measures
There were no significant differences between the SOC and SHP groups in discharge DRS, amount of DRS change, or rehabilitation length of stay (Table 3). In the combined cohort analysis of the relationship between sleep and outcome, improvement of TST, SE, and WASO were associated with DRS improvement, which reached significance for WASO (Spearman’s ρ = −0.472, P = .048).
Rehabilitation Outcomes: Discharge Disability Rating Scale (DRS) and Length of Stay (LOS).
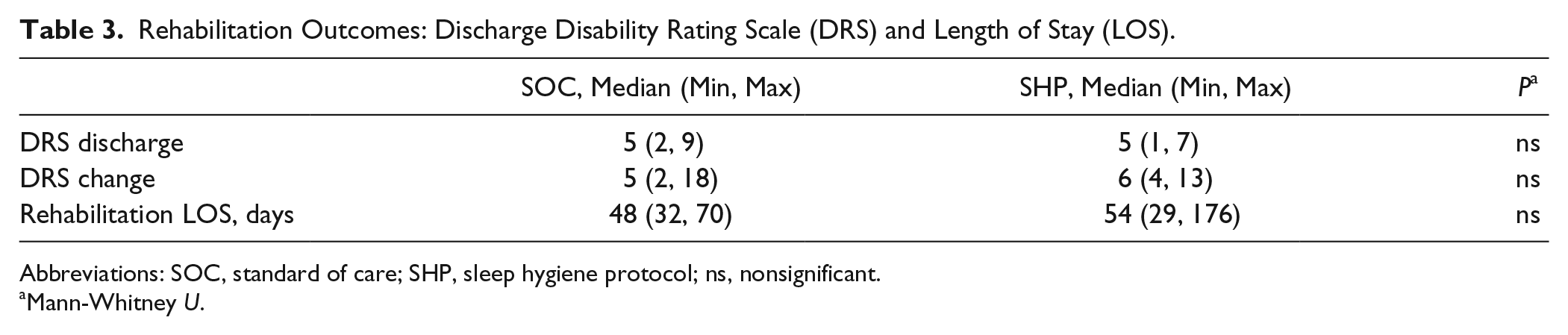
Abbreviations: SOC, standard of care; SHP, sleep hygiene protocol; ns, nonsignificant.
Mann-Whitney U.
Cognition Measures
In terms of sleep and cognition, median actigraphy sleep scores for the first week were compared between the 6 participants who were in PTA and the 12 who were not. Although participants in PTA had slightly worse TST, SE, and WASO, none of the comparisons were significant. Further analysis of cognitive testing for a combined cohort of participants who had emerged from PTA found no significant correlations between TST, SE, or WASO or the CNS Vital Signs standard scores for reaction time, working memory, and recognition memory.
Discussion
To our knowledge, this is the first study using continuous actigraphy to assess a sleep hygiene intervention compared to standard of care in patients with moderate to severe TBI. This study is unique in its approach to adjusting nighttime sleep-rest intervals in the sleep hygiene intervention according to patients’ preinjury biologic preference or chronotype. This was a pilot study to determine the feasibility of conducting a larger, clinical trial and not powered to detect treatment efficacy. Overall, we found a high prevalence of disrupted sleep in patients with moderate to severe brain injury admitted to an inpatient rehabilitation unit and that it is feasible to implement a sleep hygiene intervention for these patients. We also found patients and their families were enthusiastic about joining the protocol.
This pilot feasibility study revealed provocative results in the analysis of continuous actigraphy measures of sleep obtained prospectively over a 3-week period in this small cohort. A combined analysis of patients in both groups showed that participants with better sleep metrics (TST, SE, WASO) tended to have better rehabilitation outcomes (DRS); for WASO, this improvement was significant. In other words, participants in both arms moved in the direction one would expect from previous studies looking at the positive effect of sleep on health in healthy individuals and individuals with brain injury.2,7,26,43-48 Moreover, our analysis within treatment arms suggests that, as a group, the SHP appeared to have a similar experience over the time of the study, while the data in the SOC showed no such cohesion (Figure 2). Although both groups’ sleep improved with time, it is compelling that the change in sleep metrics over time in the SHP group was significant, while it was not in the SOC group. It is possible that this group coherence in the SHP was related to the entraining of a similar circadian harmonic generated by the sleep hygiene intervention.
Actigraphs have been increasingly utilized to study the sleep patterns of patients with all levels of brain injury from mild to severe. We found these devices were well tolerated by our patients with moderate to severe injury. Although polysomnography is considered the gold standard for objective study of sleep stage architecture, actigraphs have the advantage of being small and unobtrusive, yet able to capture periods of sleep and activity over days to weeks.
Additionally, a confused, agitated patient emerging from a moderate to severe brain injury will often not be able to tolerate the elaborate monitoring apparatus involved in a single night of polysomnography. Actigraphy has been demonstrated to show a concordance with polysomnography in patients with moderate to severe brain injury.49,50 There are, however, significant issues with the use of these devices in this patient population. Because a confused patient emerging from moderate to severe brain injury cannot be expected to indicate beginning and end of their sleep rest period with a sleep diary, this study used an institutionally set rest period to score sleep metrics. The rationale for using a set interval to score sleep in the confused brain injury patient over nursing logs and device autoscoring has been described in our previous work, 51 which showed large variability in sleep-rest intervals between the device autoscore function and hand scoring (considered the standard for scoring sleep intervals). Specifically, device autoscoring was found to result in either under or over estimation of the sleep interval, while set interval scoring had the highest correlation with hand scoring. 52 For this reason, a 10-hour institutional set sleep-rest interval was used in this study. Institutional sleep-rest periods are common in rehabilitation hospitals and a patient’s performance in therapies is likely predicated on the quality of sleep they get during this interval. Thus, this interval seems a reasonable and clinically relevant one to study.
With regard to the acceptability of the intervention, participants in the SHP were less satisfied (88%) than participants in the SOC (100%). Slightly decreased satisfaction with the SHP is understandable given the demands of the treatment, such as limiting naps and caffeine consumption. Furthermore, the difference between 88% and 100% satisfaction in this small sample likely has limited clinical relevance. In addition, we found that family members of participants in the SHP were more satisfied than family members of participants who were in the SOC. This result is not surprising given the context of the study. During enrollment, our observation was that families’ enthusiasm for participating in OSABI was often based on observations that their family member had poor sleep. Thus, allocation of their family member to the non-treatment condition may have led to lower satisfaction. Overall, we believe the surveys show a general acceptance of the intervention paradigm by a majority of patients and family members.
Limitations
The primary limitations of this pilot study were a small cohort at a single site and a treatment intervention that could not practically be blinded. Also, this study included participants who were, on average, 30 days from acute injury. Thus, these results may not be applicable to people earlier in their course of recovery and may not generalize to a larger population of individuals with TBI. Additionally, the study’s inclusion and exclusion criteria were rigorous in the hopes of keeping balance between the 2 treatment groups, but this limited the number of patients that could be approached for enrollment and led to a recruitment rate of 7.5%. A larger study would likely need to broaden the inclusion and exclusion criteria to allow for a timely recruitment of needed subjects.
Another limitation of the study was being unable to identify the most relevant aspect of the SHP. Although this was beyond the scope of the present study, attempts were made in the design to objectively quantify light and sound exposure of patients. Early in the study, we found downloaded sound data from study monitors to be so inconsistent that these data were abandoned. Light data from the wrist actigraphs during the day were also inconsistent. Other investigators have found a similar discrepancy between wrist-worn and wall-mounted light meters in the study of light exposure among hospitalized patients. 53 Because of these challenges and discrepancies, future studies designed to track light exposure in subjects should use methods other than wrist-worn actigraphs for more accurate measurements.
Using a 10-hour set interval for sleep rest time could also be seen as a limitation of this study because it may limit generalization to a larger population of patients or comparison to other sleep studies using a different interval to generate actigraph data. Using a 10-hour time interval could potentially lower sleep scoring metrics, in particular, sleep efficiency, which is a measure of time the actigraph scores as sleep during the time the subject is in bed. Again, the rationale for our choice of an institutional set interval seems to make sense in the clinical context. A patient in a rehabilitation hospital is expected to be asleep during this time frame and the patient’s daytime function will likely be related to the quality and quantity of sleep obtained in this period. Another aspect that could be considered a limitation in using a set interval is that measures of latency to sleep onset become invalid in those instances when the patient’s actual onset of sleep is ahead of the start time of the set interval. Such an occurrence would be scored by the actigraph’s scoring algorithm as a sleep latency of zero. Other actigraph sleep metrics, however, were found to be unaffected using this method.
Although not statistically significant, participants who were enrolled with ongoing PTA tended to have worse SE than participants who had emerged from PTA. This is consistent with previously published research by our group. 7 Prospective cognitive testing failed to show a relationship between improved sleep and improved cognition as has been shown in previous studies.2,7,45 There are multiple factors that can influence an individual’s performance on cognitive testing while undergoing acute rehabilitation that could have obscured associations in this small sample.
Implications/Conclusions
Disrupted sleep is highly prevalent in individuals with moderate severe brain injury, as shown in this study where 75% of patients screened showed impaired SE on admission to the rehabilitation center. This finding is consistent with previous studies from our group and others and underlines the magnitude of the problem for patients in this setting.12-14 Our findings support the feasibility of implementing a sleep hygiene intervention on an acute inpatient rehabilitation unit. Families were enthusiastic about participating in the study and having their family members’ disrupted sleep identified and treated. While the surveys were generally favorable to the intervention from a nursing perspective, the investigators found that actual implementation of a SHP required a significant change in culture among nursing staff. Finally, although the study was not powered to show efficacy of treatment, it was of interest that patients in the SHP exhibited significant improvement in actigraphy metrics over the study period while the SOC group did not. We believe that these findings support the need for a larger study powered to show efficacy of this treatment.
Supplemental Material
CONSORT_2010_Checklist_OSABI_2019 – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, CONSORT_2010_Checklist_OSABI_2019 for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Supplemental Material
CRAIG_HOSPITAL_SLEEP_HYGIENE_PROTOCOL_ABF – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, CRAIG_HOSPITAL_SLEEP_HYGIENE_PROTOCOL_ABF for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Supplemental Material
Fidelity_table – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, Fidelity_table for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Supplemental Material
Modified_STOP_BANG_QUESTIONNAIRE – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, Modified_STOP_BANG_QUESTIONNAIRE for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Supplemental Material
Nursing_Staff_Feasibility_Questionnaire – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, Nursing_Staff_Feasibility_Questionnaire for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Supplemental Material
OSABI_INCLUSION_AND_EXCLUSION_CRITERIA – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, OSABI_INCLUSION_AND_EXCLUSION_CRITERIA for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Supplemental Material
Sleep_Hygiene_Protocol_Questionaire-Family_Member – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, Sleep_Hygiene_Protocol_Questionaire-Family_Member for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Supplemental Material
Sleep_Hygiene_Protocol_Questionnaire-Participant – Supplemental material for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury
Supplemental material, Sleep_Hygiene_Protocol_Questionnaire-Participant for Optimized Sleep After Brain Injury (OSABI): A Pilot Study of a Sleep Hygiene Intervention for Individuals With Moderate to Severe Traumatic Brain Injury by Michael J. Makley, Don Gerber, Jody K. Newman, Angie Philippus, Kimberley R. Monden, Jennifer Biggs, Eric Spier, Patrick Tarwater and Alan Weintraub in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of Dr Richard Allen from Johns Hopkins School of Medicine, Dr Jeanine Duffy of Harvard School of Medicine, and Dr Simon Beaulieu-Bonneau from Laval University for their valuable input on the development of the protocol. The authors would also like to acknowledge the involvement and support of the caring and dedicated rehabilitation nursing and therapy staff of Craig Hospital’s Brain Injury Unit as well as the patients and families who participated in this study.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Craig Hospital Foundation and MINDSOURCE—Brain Injury Network, formerly the Colorado Brain Injury Trust Program, under grant number NCT02838082
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
