Abstract
Background. Initial evidence suggests that the integrity of the ipsilesional corticospinal tract (CST) after stroke is strongly related to motor function in the chronic state but not the treatment gain induced by motor rehabilitation. Objective. We examined the association of motor status and treatment benefit by testing patients with a wide range of severity of hemiparesis of the left and right upper extremity. Method. Diffusion tensor imaging was performed in 22 patients beyond 12 months after onset of stroke with severe to moderate hemiparesis. Motor function was tested before and after 2 weeks of modified constraint-induced movement therapy. Results. CST integrity, but not lesion volume, correlated with the motor ability measures of the Wolf Motor Function Test and the Motor Activity Log. No differences were found between left and right hemiparesis. Motor performance improved significantly with the treatment regime, and did so equally for patients with left and right arm paresis. However, treatment benefit was not associated with either CST integrity or lesion volume. Conclusion. CST integrity correlated best in this small trial with chronic long-term status but not treatment-induced improvements. The CST may play a different role in the mechanisms mediating long-term outcome compared to those underlying practice-induced gains after a chronic plateau in motor function.
Keywords
Introduction
The corticospinal tract (CST) is implicated in the recovery of motor function after stroke.1-4 For example, Fries and colleagues 1 report clinical evidence for a critical role of ipsilesional motor efferences in functional recovery and restitution after stroke. These findings led to the proposition that CST integrity of the CST may predict motor recovery.5-7
In a previous study with 10 patients, 8 this hypothesis was tested using a recently developed neuroimaging technique to map the corticofugal fibers and quantify the integrity of these fiber tracks in vivo. 5 This method combines diffusion-weighted imaging with probabilistic tractography to determine a CST variability map in healthy controls. This probability map is then used to estimate CST integrity in patients through a voxel-based quantification of the overlap between the lesion and the CST variability map derived from controls. Using this method, we found a strong correlation of long-term outcome and CST integrity in 3 patients with circumscribed subcortical white matter damage. 5 We replicated this association in 10 patients with chronic hemiparesis due to lesions in cortical and subcortical regions. 8 For the present study, we examined the association of CST integrity and long-term outcome in a larger sample of patients with moderate to severe left and right hemiparesis rather than by lesion extent or location. Residual functional ability was determined by the Motor Activity Log (MAL 9 ) and the Wolf Motor Function Test (WMFT 10 ). Our hypothesis is that if CST integrity were the critical determinant for motor recovery in the chronic state, CST integrity but not lesion volume would be associated with functional outcome measures.
Stinear et al 11 further proposed that CST integrity may be a predictor for functional recovery induced by practice-based motor rehabilitation in the chronic phase. This view builds on the theory that CST integrity is intrinsically linked to recovery because it is instrumental to functional reorganization.5-7 We therefore speculated that the efficacy of neuroplasticity-facilitating rehabilitation techniques may also be related to CST integrity. However, our previous study 8 showed that, at least in a group of patients presenting with low-functioning hemiparesis, motor-practice benefits were not associated with CST integrity. In the present study, we explored this issue further.
Methods
Participants
Twenty-two patients (12 male, 10 female: aged 42-69 years, mean = 57.0 ± 1.7), 12 with left and 10 with right-sided hemiparesis, participated in the study. All patients had a first stroke 12 to 107 months (mean = 40.5 ± 6.8 months) prior to testing and presented with severe to moderate hemiparesis of the upper limb. At intake the average quality of movement score of the Motor Activity Log (MAL QoM)9,12 was 1.2 ± 0.2, indicating primarily severe to moderate levels of functioning, with 11 patients scoring less than 1 point, 5 patients scoring between 1 and 2 points, and 6 patients with scores between 2 and 3 points. The maximum score for MAL QoM is 5, indicating prestroke levels of functional ability. Poor functional ability was further indicated by an average Frenchay Arm Test (FAT) 13 score of 2.4 ± 0.4, with 9 patients obtaining scores between 0 and 2, 5 patients scoring between 2 and 3, 1 patient scoring between 3 and 4, 2 patients scoring between 4 and 5, and 5 patients achieving the full score of 5. The FAT has a maximum rating of 5, which represents good recovery of basic hand function. Patients were selected on the basis of symptom presentation, not the location of the lesion, which in most cases was unknown to the general practitioner (GP). Analysis of the T1 magnetic resonance imaging (MRI) images conducted by a trained neurologist (AC) later revealed that there was no systematic lesion pattern. The lesion characteristics are summarized in Table 1.
Summary of Patient Details and Lesion Characteristics.
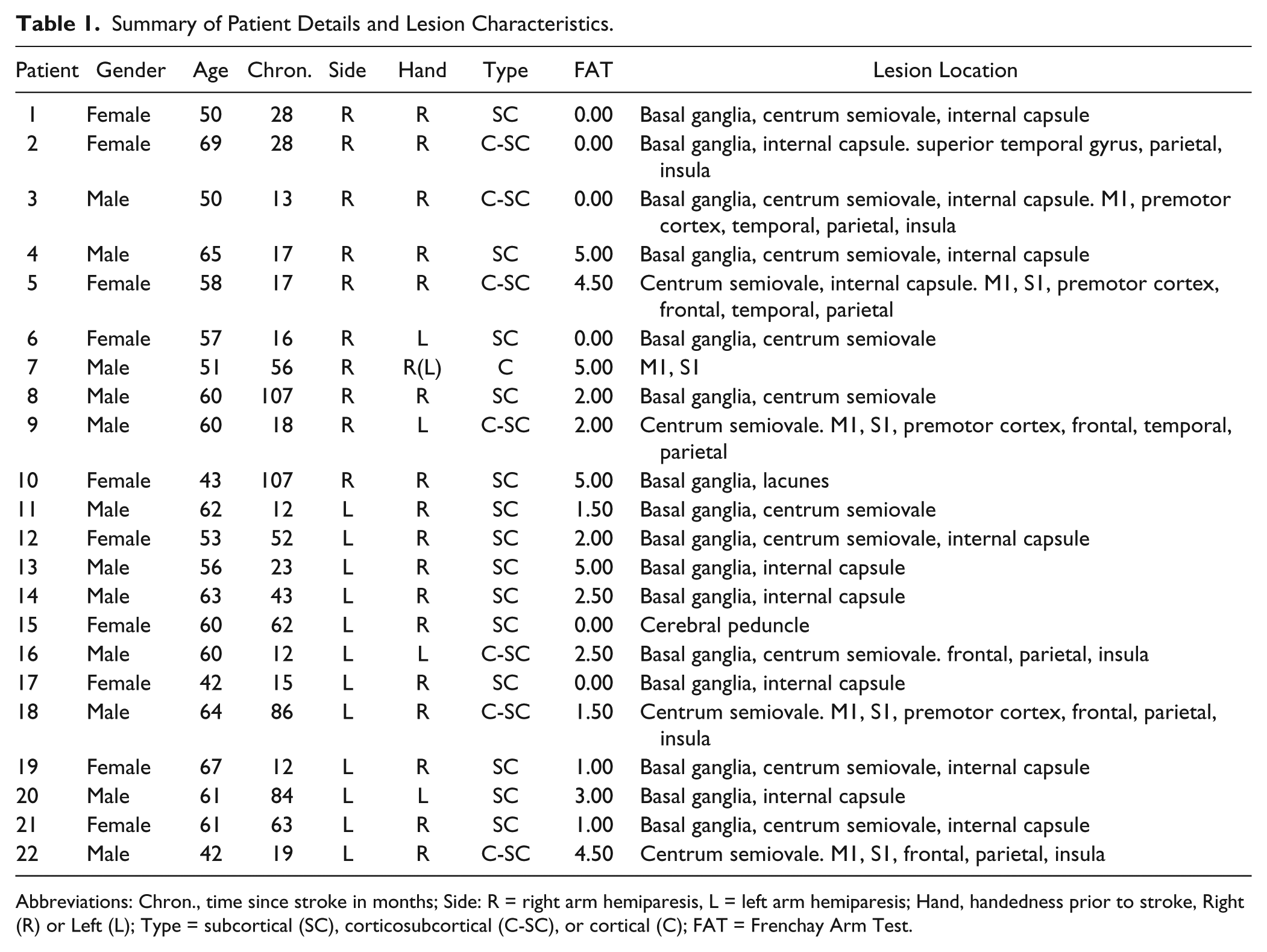
Abbreviations: Chron., time since stroke in months; Side: R = right arm hemiparesis, L = left arm hemiparesis; Hand, handedness prior to stroke, Right (R) or Left (L); Type = subcortical (SC), corticosubcortical (C-SC), or cortical (C); FAT = Frenchay Arm Test.
Participants were recruited through flyers, newspaper, and Web advertisements. Patient respondents were prescreened by phone and attended a subsequent screening session at the University of Surrey in which further information was obtained on physical and mental health history, and the level of hemiparesis using the FAT. 13 The criteria for patient selection were the following: (a) first ever stroke with chronicity > 9 months, (b) Mini-Mental State score 14 ≥ 24, and (c) ability to understand the therapist and to communicate needs.
In addition, structural MRI data were obtained from 22 right-handed healthy individuals (5 male, 17 female: aged 19-35 years with a mean age of 23.7 years) to determine the CST variability map. Control participants were recruited through e-mail lists and flyers.
The study was approved by the NHS National Research Ethics Committee and the Ethics Committee of the University of Surrey. Written informed consent was obtained prior to participation.
Study Schedule and Behavioral Parameter Extraction
Participants took part in a program of affected arm training, which was based on the shaping method of constraint-induced movement therapy.12,15 For 2 weeks patients practiced functional movements repetitively with the hemiparetic arm while task difficulty was gradually increased. Shaping tasks are structured so that the level of difficulty could be adjusted on several dimensions and had directly observable outcomes. This allowed patients to understand and trace their progress.
Training was either delivered by a therapist for 90 (n = 12) or 180 minutes (n = 5) a day, or as a home training program (n = 5) where patients were instructed to practice the shaping tasks at home daily for at least 90 minutes per day. Motor data were collected at a baseline 2 weeks prior to the intervention, as well as before and after the intervention (baseline, pre, post). Structural MRI data were obtained at the pretesting session only.
The WMFT, 16 a comprehensive laboratory-based test of gross and fine motor function, and the MAL, 9 a subjective assessment of everyday activities, were used to measure motor ability and real-world use. For the WMFT, the functional ability score (WMFT-FAS, range = 0-7) and speed (WMFT-TT, “time taken” in seconds) were extracted for statistical analysis. From the MAL, the amount of use (MAL-AoU) and quality of movement (MAL-QoM) were used. Residual motor ability and real-world use of this ability were indexed as the average of the baseline and pretreatment scores. Treatment benefit was calculated as the difference score between the average of the baseline and pretreatment values, and posttreatment scores. T tests calculated prior to averaging baseline and pretreatment scores indicated no significant differences (WMFT TT: t[18] = −2.0, P = .06; WMFT FAS: t[18] = 1.1, P = .29; MAL QOM: t[18] = 1.0, P = .32; MAL AOU: t[18] = 1.0, P = .33; FAT: t[18] = −1.2, P = .23).
Prior to further analysis, the treatment effects obtained in the 3 different treatment groups were tested for group differences (t tests, all P > .1). Neither the MAL nor the WMFT showed significant differences. The data were hence collated, and all subsequent analyses refer to the whole group of 22 patients.
MRI Data Acquisition
Participants were scanned on a 3T Siemens Trio scanner (Erlangen, Germany). T1-weighted anatomical scans and diffusion-weighted images (DWI) were acquired. High-resolution 3D brain MRI images were obtained using a T1-weighted magnetization prepared rapid acquisition gradient echo (MPRAGE) pulse sequence, with the following characteristics: TR = 1830 ms, TE = 4.43 ms, inversion time = 1100 ms, 1 acquisition, flip angle = 11°, FOV = 256 mm, 176 slices, voxel size = 1 × 1 × 1 mm3, in-plane matrix = 256 × 256.
DWI data were acquired using a single-shot diffusion-weighted echo-planar imaging sequence, with diffusion gradients applied along 12 directions (b0 = 0, 1 image, and b1 = 1000 s/mm2, 12 images), and TR = 8900 ms, TE = 100 ms, number of averages = 4, 55 slices, voxel size = 2.5 × 2.5 × 2.5 mm3, in-plane matrix = 88 × 128.
Preprocessing
For both patients and healthy control participants, DWI images were motion corrected and realigned to the mean image using the diffusion II toolbox (http://sourceforge.net/projects/spmtools/) in SPM8. Corrected images were normalized to Montreal Neurological Institute (MNI) space. The gradient information of each image was updated by reorientation, which applied the rotations performed during realignment and normalization to the gradient direction vectors. The diffusion tensor eigenvalues and eigenvectors were calculated to generate the fractional anisotropy (FA) maps for each participant.17,18
Methods to Determine CST Integrity
CST integrity of patients was determined by summing the fraction of overlap in each axial slice between the lesion and the CST variability map extracted from the control group. The respective analysis steps are detailed below.
CST Variability Map
DTI Studio 19 software (https://www.dtistudio.org) was used for fiber tractography in the control participants, based on the fiber assignment by continuous tracking (FACT) method. 20 Fibers were tracked in MNI space starting from all voxels with a threshold of FA > 0.2 and a tract-turning angle <41°.21,22 The CST was then reconstructed for each control participant between 2 regions of interest (ROI), the cerebral peduncles and the precentral gyrus of the left and right hemisphere, respectively. These ROIs were chosen to conform with the original article on CST integrity. 5 The ROIs were determined using anatomical landmarks, as described in other studies,5,11,21,23,24 and marked by hand on the FA maps. The left and the right hemispheric CSTs derived for each control participant were saved as binary masks and then summed across all 22 control participants to produce a trajectory variability map for each hemisphere (see Figure 1).
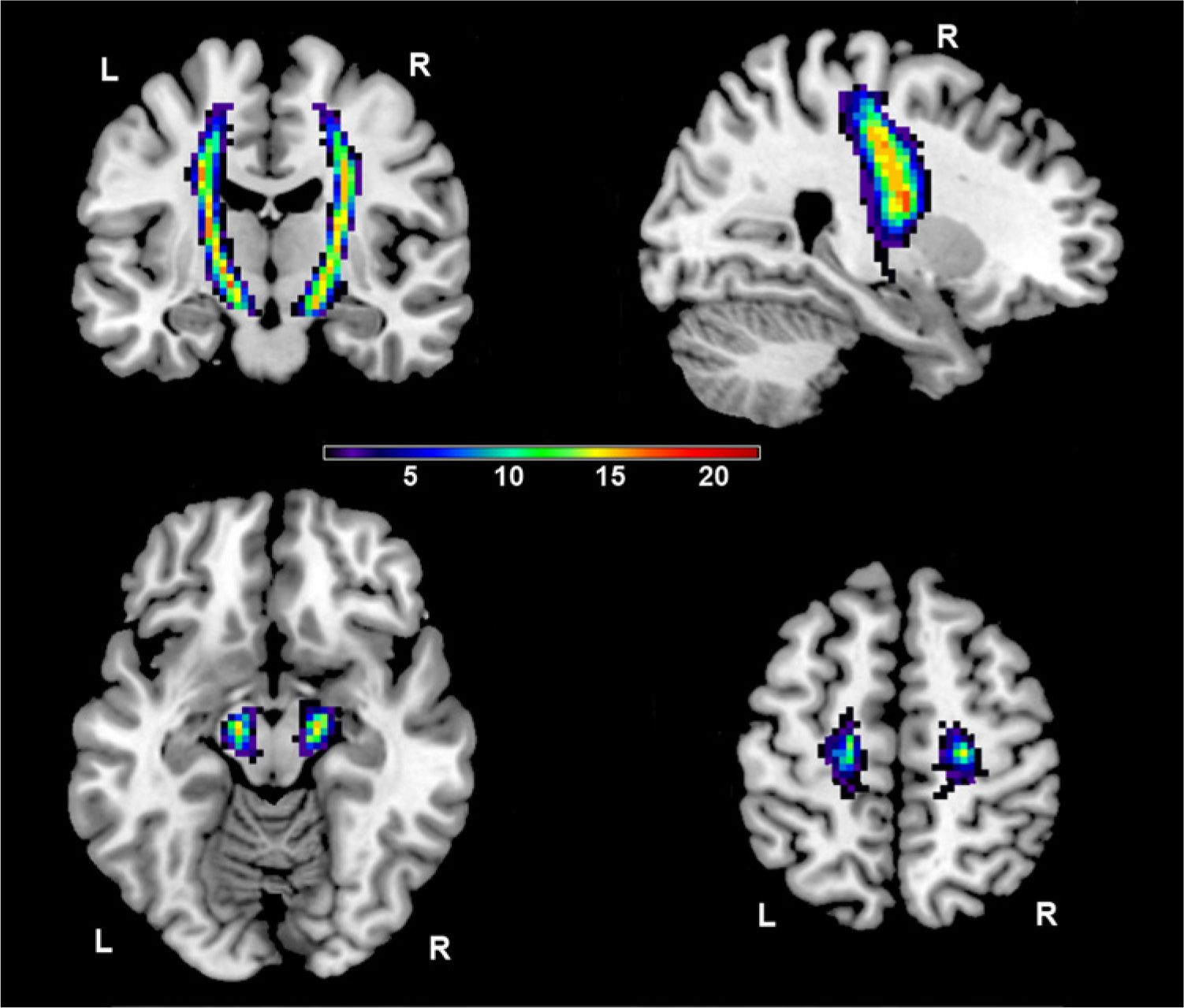
Corticospinal tract (CST) trajectory variability maps displayed on a T1-weighted image.
Lesion Identification
All FA maps were smoothed with a Gaussian kernel of full-width at half-maximum (FWHM) = 8 mm. A one-way ANOVA was then computed to compare the map of each patient with the trajectory variability map derived from controls on a voxel-by-voxel basis. Brain volume, age, and gender were included as covariates. Lesioned tissue was identified as significant anisotropy reduction (P < .01, uncorrected). A binary lesion mask was created for each patient and subsequently used to calculate CST integrity.
Quantification of CST Integrity
Each lesion mask was overlaid onto either the left or right CST variability map. CST integrity was then measured by summing the fraction of overlap in each axial slice between the lesion and the weighted CST variability map, calculated as follows:

where N is the number of axial slices in each image. Higher values indicate lower CST integrity.
Absolute Lesion Volume
In addition to CST integrity, absolute lesion volume was calculated as the sum of the values of the voxels with significantly reduced anisotropy identified in the lesion identification analysis.
Statistical Analysis
Spearman correlations were used to examine (1) the association of residual motor ability and (a) the degree of CST integrity and (b) lesion volume, and (2) the association of motor improvement induced by motor practice and (a) CST integrity and (b) lesion volume. These analyses were first calculated across the whole group and then separately for left hemiparetic (N=12) and right hemiparetic (N=10) patients. T tests were then performed for group comparisons.
Results
CST Integrity and Residual Motor Ability
The mean residual MAL scores (QoM: 1.18 ± 0.21 units; AoU: 1.27 ± 0.25 units) suggested low levels of affected arm use in everyday situations. The residual WMFT confirmed that abilities with the affected arm were generally poor (FAS: 4.43 ± 0.29 units; time taken [TT]: 29.25 ± 5.71 seconds). Neither MAL nor WMFT scores showed significant differences between left and right affected patients (all P > .2).
The magnitude of lesion overlap was similar in left and right hemispheric patients as indicated by a nonsignificant independent samples t test (t[20] = −1.19, P = .25). The correlations between lesion overlap and either MAL or WMFT were not significant (all P > .09) when left (n = 12) and right (n = 10) affected patients were tested separately. However, the scatterplots, presented in Figure 2, are suggestive of a positive association between poorer residual motor ability and greater lesion overlap in the 2 subgroups.
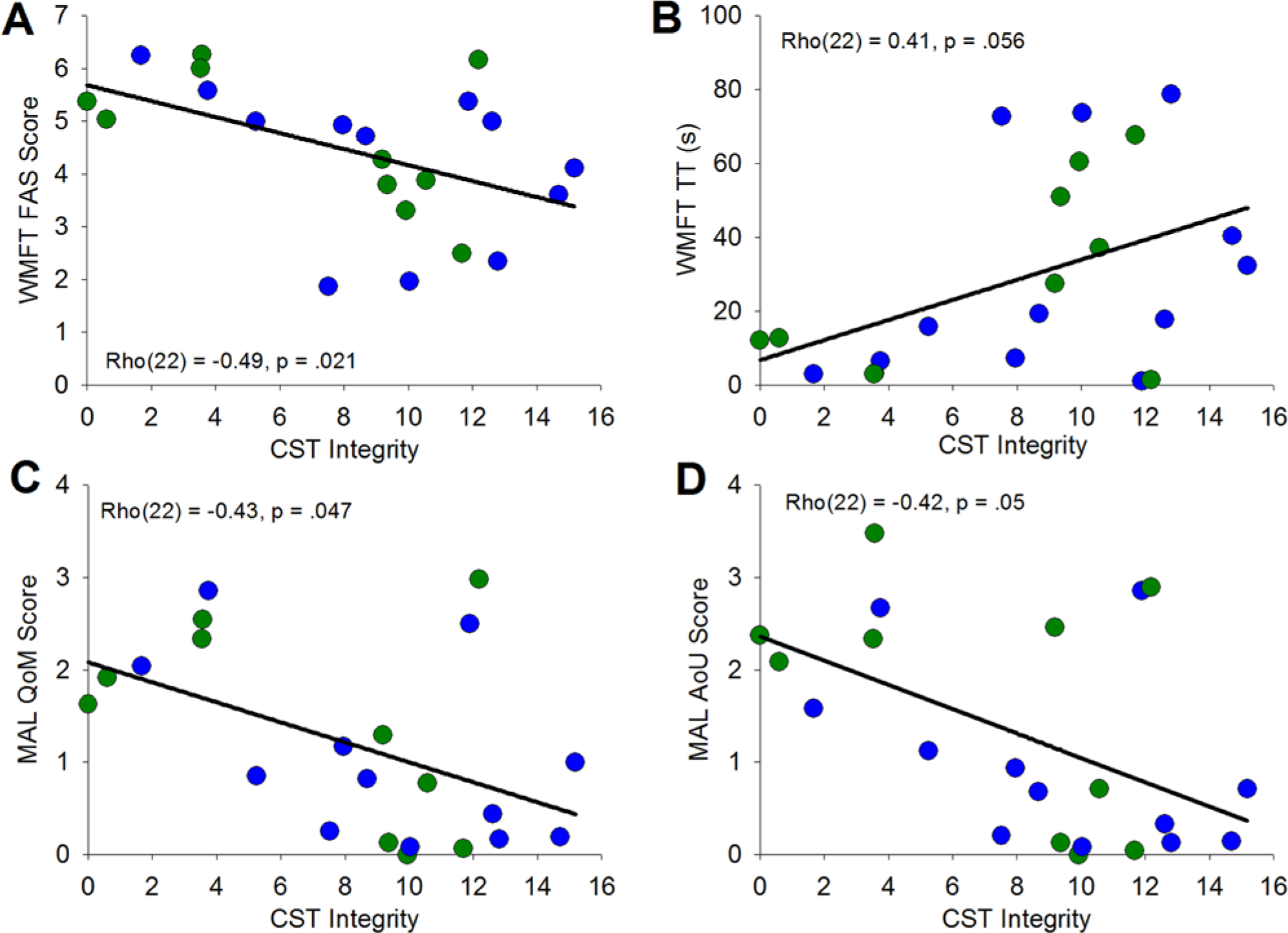
Scattergraphs showing the association between corticospinal tract (CST) integrity and residual ability for the Wolf Motor Function Test (WMFT; A, B) and Motor Activity Log (MAL; C, D), across all participants.
The correlation analysis across the whole group revealed significant associations between CST integrity and the WMFT FAS scale (ρ[22] = −0.49, P = .021), as well as the MAL scales (QoM: ρ[22] = −0.43, P = .047; AoU: ρ[22] = −0.42, P = .05), and approached significance for WMFT TT scale (ρ[22] = 0.41, P = .056).
CST Integrity and Treatment-Induced Recovery
Motor ability improved significantly with motor practice with a mean increase of 0.4 ± 0.1 for WMFT-FAS (t[21] = 4.15, P < .001), 0.7 ± 0.1 for MAL-QoM (t[21] = 8.02, P < .001), and 0.6 ± 0.1 for MAL-AoU (t[21] = 4.72, P < .001), but no significant decrease for WMFT-TT (2.7 ± 1.8; t[21] = −1.56, P = .13). These effects were also present when left and right hemipheric patients were tested separately (left hemispheric: WMFT-FAS: t[11] = 3.21, P = .008; MAL-QoM: t[11] = 5.72, P < .002; MAL-AoU: t[11] = 3.99, P = .002; WMFT-TT: t[11] = −0.79, P = .45; right hemispheric: WMFT-FAS: t[9] = 2.53, P = .032; MAL-QoM: t[9] = 5.58, P < .001; MAL-AoU: t[9] = 2.91, P = .017; WMFT-TT: t[9] = −1.34, P = .22). The magnitude of this improvement did not differ between these groups, as indicated by nonsignificant independent sample t tests for the respective measures (WMFT-FAS: t[20] = −0.08, P = .94; WMFT-TT: t[20] = −0.80, P = .44; MAL-QoM: t[20] = 0.91, P = .37; MAL-AoU: t[20] = 0.63, P = .54).
The scatterplots relating to the correlation analysis are illustrated in Figure 3. Visual inspection, as well as statistical analysis, suggested that treatment benefit was not systematically associated with CST overlap as indicated by nonsignificant correlations for all motor parameters (WMFT-FAS: ρ[22] = 0.12, P = .59; WMFT-TT: ρ[22] = −0.13, P = .57; MAL-QoM: ρ[22] = 0.04, P = .86; MAL-AoU: ρ[22] = 0.15, P = .51).
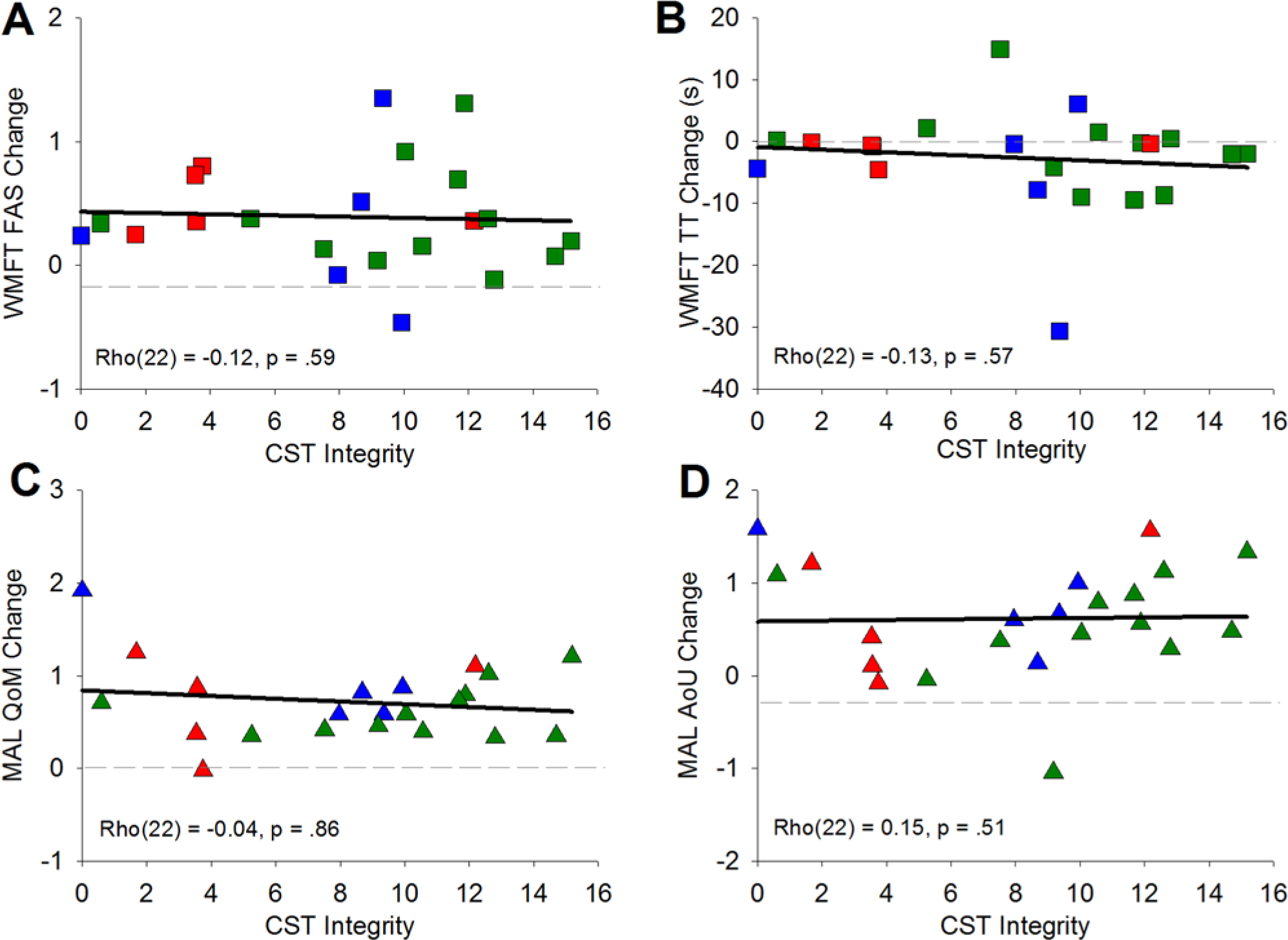
Scattergraphs showing the association between corticospinal tract (CST) integrity and treatment benefit for the Wolf Motor Function Test (WMFT; A, B) and Motor Activity Log (MAL; C, D), across all participants.
Lesion Volume
The absolute lesion volume showed no significant association with residual motor ability (WMFT-FAS: ρ[22] = 0.09, P = .7; WMFT-TT: ρ[22] = 0.13, P = .56; MAL-QoM: ρ[22] = −0.22, P = .33; MAL-AoU: ρ[22] = −0.16, P = .47) or treatment benefit (WMFT-FAS: ρ(22) = −0.27, P = .22; WMFT-TT: ρ[22] = 0.20, P = .38; MAL-QoM: ρ[22] = 0.10, P = .66; MAL-AoU: ρ[22] = 0.03, P = .9). The lesion volume was similar in left and right hemispheric patients as indicated by a nonsignificant independent samples t test (t[20] = 0.01; P = .99).
Discussion
This study assessed the association of CST integrity and long-term motor outcome in patients with left- and right-side hemiparesis. For this purpose, we used a method that allows a quantitative in vivo estimation of the overlap between infarct lesion and the CST on the basis of diffusion-weighted MR images. 5 In line with a previous study, 8 we found that patients with poorer CST integrity, defined as greater overlap of the infarct lesion with the CST variability map derived from healthy controls, demonstrated lower levels of upper limb function in the chronic state. This association between CST integrity and long-term outcome was similar for patients with left- and right-sided hemiparesis. In addition, there were no group differences for the magnitude of CST overlap or the functional ability measured with the MAL and the WMFT. This suggests that, at least in our sample, long-term motor outcome in left- and right-sided chronic hemiparesis is comparable and that this outcome is not differentially mediated by CST integrity. The data further confirmed the observation that lesion volume was not associated with the level of motor deficit sustained in the chronic state.8,25 Again, this was equally true for left and right affected patients.
Taken together, our results support the hypothesis that the structural integrity of the CST, rather than absolute white matter lesion volume, is a critical parameter for the long-term outcome of upper-limb function.1,2,4,7,26,27 Our findings further suggest that this fundamental lesion–outcome relationship applies to both left- and right-sided hemiparesis.
CST and Long-Term Outcome
It is tempting to conclude from our data that CST integrity is predictive of long-term outcome. However, our findings only represent a structural and behavioral “snapshot” long after the stroke has occurred. Recent research suggests that altered sensorimotor input also leads to white matter changes in the adult human brain. 28 Therefore, plasticity processes and the effects of early rehabilitation may not only have shaped motor outcome but also influenced brain structure. 29 It follows that structural characteristics are likely to evolve throughout acute and postacute phases of recovery. The association between CST integrity and residual ability found in the present study therefore only holds implications for the chronic state.
The present study measured long-term motor outcome through 2 instruments, the laboratory-based WMFT and the self-rated MAL. The WMFT assesses the speed and quality of a range of upper-limb movements through a 16-item standardized laboratory test and represents a very sensitive measure for the actual motor ability. The MAL, on the other hand, is a subjective instrument in which patients estimate affected arm use and ability in everyday activities. The measure has high ecological validity for real-world behavior and correlates highly with the WMFT. Both measures showed a significant association with CST integrity. This is an important point because a patient’s ability and the usage of this ability in everyday life can be dissociated, a phenomenon most poignantly evident in learned nonuse behavior.30-32 The fact that CST integrity is correlated with both WMFT and MAL makes it a strong and comprehensive structural marker for the restitution of movement abilities that are relevant for the real-world setting.
CST and Treatment Benefit
Patients tested in the present study also completed a 2-week program of motor rehabilitation. The treatment data confirmed earlier findings that practice-based interventions improve affected arm function in the chronic state.15,33,34 Even though our study was not designed to specifically investigate how CST integrity might interact with the efficacy of motor practice protocols, it provided a good opportunity to explore this relationship. Confirming previous observations,8,35 we found that neither CST integrity nor lesion volume was significantly related to treatment outcome. These findings suggest that the benefit of motor training in the chronic state might not entirely depend on CST integrity or lesion volume.
However, this interpretation can only be suggested since the treatment outcomes were achieved through 3 slightly different versions of motor training. Despite the absence of statistical differences between the treatment protocols in our sample, the possibility cannot be excluded that, for example, an individual receiving 1.5 hours of daily training would have obtained better outcome with 3 hours of training, perhaps changing the association between CST integrity and treatment outcome. The data nevertheless provide valuable insights for future directions of research. The notion that the association between CST integrity and motor training success in the chronic phase might be relatively weak is certainly encouraging news for patients as it suggests that motor training in the chronic phase might be beneficial to patients with a range of lesions and varied levels of CST preservation.
The findings raise interesting questions regarding the mechanism mediating long-term outcome (residual ability) versus those subserving treatment efficacy (treatment-induced recovery) in the chronic state. Thus, the apparently differential role of CST integrity in long-term outcome and training-induced recovery in the chronic state might indicate slightly different mechanisms. These mechanisms could either be a function of time, a function of the factors involved in the rehabilitation process, or a combination of both. In other words, it is possible that the mechanisms supporting long-term outcome and those supporting an improvement of function specifically obtained through motor practice in the chronic state may differ in their reliance on CST integrity. Long-term outcome is the compound product of spontaneous and treatment-induced recovery processes that include physiological, neurological, and psychological aspects, which collectively shape a person’s residual abilities and their use in everyday life. In contrast, motor practice in the chronic phase is targeted to further improve motor function of the affected hand. Albeit measurable and significant, these improvements are on a much smaller scale than found in long-term outcome following usual care and may therefore lack the power to show differential effects in relation to CST integrity, in particular with smaller sample sizes. In addition, the quantification of CST integrity as well as lesion volume is an approximation in itself and is hence subject to a margin of error. It may therefore be the case that the lack of association in the present study is due to a combination of the magnitude of treatment effects and the approximate nature of CST integrity estimation. Only through larger scale studies will it be possible to fully investigate the nature of the relationship between the recovery potential released through motor practice in the chronic state and structural lesion parameters such as CST integrity.
Limitations
The CST integrity measured here is only an approximation of the true CST damage the lesion has caused. This lies in the nature of the approach, which relies on a CST probability map derived from control participants. In our case, these control participants were not fully matched for age or gender. Moreover, we mapped the CST between the precentral gyrus and the cerebral perduncles, to keep with the protocol of published work. This circumscribed ROI may not have captured the CST in its entirety. Finally, it has been shown that substantial ventricular dilation can either visibly distort the course of the CST 36 or obliterate its detection. 37 These findings highlight the challenges involved in the noninvasive investigation of CST integrity. The CST probability map is a good proxy; however, this proxy does not take potential structural deformations into account.
Conclusion
CST integrity, described by the overlap between the infarct lesion and the CST variability maps derived from healthy volunteers, is strongly associated with the level of residual motor ability in patients with chronic left- and right-sided hemiparesis. However, CST integrity is not associated with the benefits achieved through motor practice in the chronic phase, despite all participants showing positive change in motor ability. This suggests that these treatment effects can be obtained despite substantive CST damage.
Footnotes
Acknowledgements
Dr Katherine Herron, Jenny Sanders, and Amy Saunders are gratefully acknowledged for their help with treatment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Medical Research Council UK.
